Abstract
A low-cost and fast real-time PCR system in a pseudo-isothermal manner with disposable capillary tubes based on thermal convection for point-of-care diagnostics is developed and tested. Once stable temperature gradient along the capillary tube has been established, a continuous circulatory flow or thermal convection inside the capillary tube will repeatedly transport PCR reagents through temperature zones associated with the DNA denaturing, annealing, and extension stages of the reaction. To establish stable temperature gradient along the capillary tube, a dual-temperature heating strategy with top and bottom heaters is adopted here. A thermal waveguide is adopted for precise maintenance of the temperature of the top heater. An optimized optical network is developed for monitoring up to eight amplification units for real-time fluorescence detection. The system performance was demonstrated with repeatable detection of influenza A (H1N1) virus nucleic acid targets with a limit of detection of 1.0 TCID50/mL within 30 min.
Keywords
Introduction
Typical PCR is performed on complicated thermal cyclers that provide repeated temperatures respectively for different reaction stages, including denaturing, annealing, and extension with 30 to 40 cycles. 1 One PCR cycle consists of transition time between different reaction stages as well as dwelling time for each of them. For conventional thermal cyclers, it is difficult to shorten the transition time or the dwelling time because both are decided by the naturally existing large thermal inertia of the heating block.
To bring sensitive molecule diagnostics into a doctor’s office, on-site environment, and rural clinics with poor medical resources (e.g., in point-of-care settings), temperature control for nucleic acid amplification should be simplified to reduce the amplification time as well as the device complexity. 2 More recently, isothermal nucleic acid amplification methods, such as loop-mediated isothermal amplification (LAMP), nucleic acid sequence-based amplification (NASBA), and rolling circle amplification (RCA), have been developed with simplified temperature settings.3–5 The inconveniences with isothermal amplification include the large number of primers and their quantification method of the template.
Another avenue for simplifying temperature control is so-called convection PCR, where an effective thermal cycling of the reaction mixture is realized in a pseudo-isothermal manner by imposing a steady-state vertical temperature gradient on the reaction volume.6,7 This temperature gradient induces a continuous, sustainable circulation of reactants between hot and cool zones of the reaction, thus stratifying the reaction into spatially separate and stable melting, annealing, and extension zones.8,9 Convective PCR eliminates the need for controlled time-domain temperature cycling, which significantly shortens the amplification time.
Here we report for the first time a readily commercialized convective PCR instrument for point-of-care diagnostics with up to eight amplification units. Within the convective PCR instrument, real-time in situ fluorescence monitoring of the reaction eliminates the separate postamplification detection step and shortens the total test time. We developed a novel and low-cost thermal-optical coupling design to implement the real-time capillary convective PCR (CCPCR), and its utility is validated with detection of influenza virus (H1N1) in samples.
Real-Time CCPCR System
System Overview
As shown in Figure 1 , the real-time CCPCR system mainly consists of a controller module, an interfacing module, a heating module, an optical module, and an assisted mechanical module.
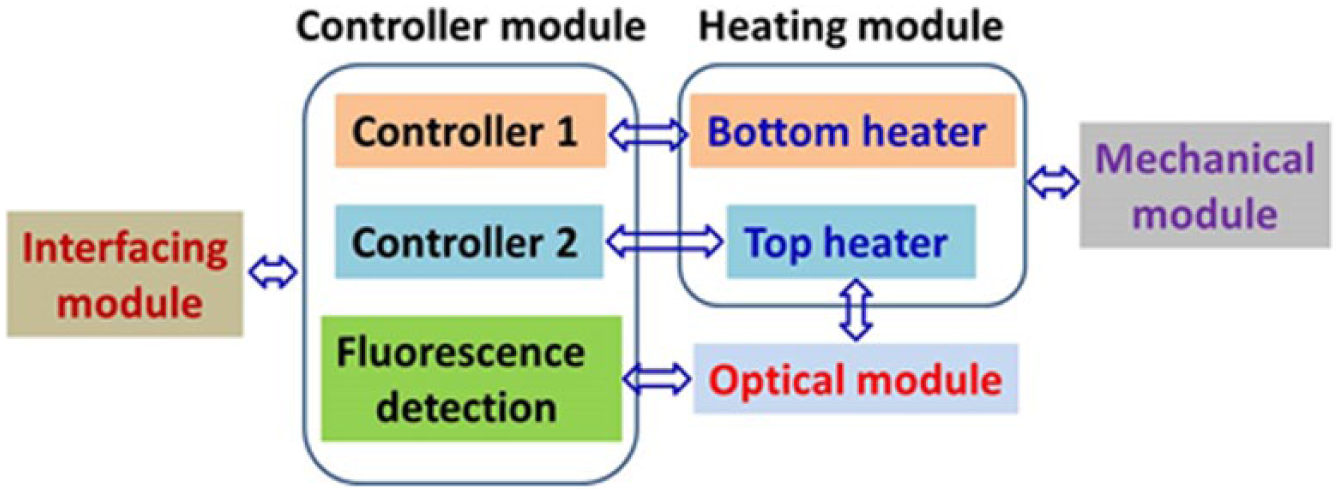
Schematic depiction of the real-time capillary convective PCR system.
Capillary tubes with excellent smoothness and transparency, which are made from polycarbonate by injection molding, are used for amplification, as shown in Figure 2 . The capillary tube consists of a head for reagent loading and a reaction chamber (~40 µL) in the form of a capillary tube with outside/inside diameters of 3.3 mm/1.7 mm and a length of 19 mm.
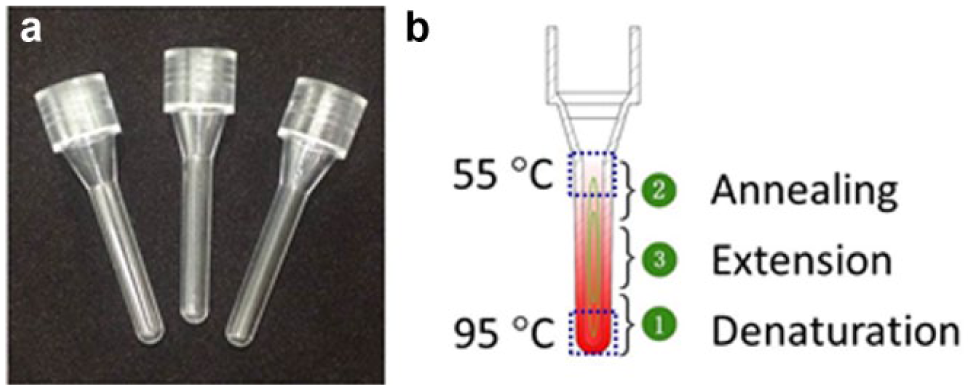
Capillary tubes for real-time amplification: (
Thermal Subsystem
We developed a dual-temperature heating strategy to heat from both the top and bottom ends of the capillary tube simultaneously. A concise heating scheme, as shown in Figure 3 , is adopted to provide precise heating with high efficiency while facilitating easy optical access to the capillary tube for real-time monitoring of reaction progress.
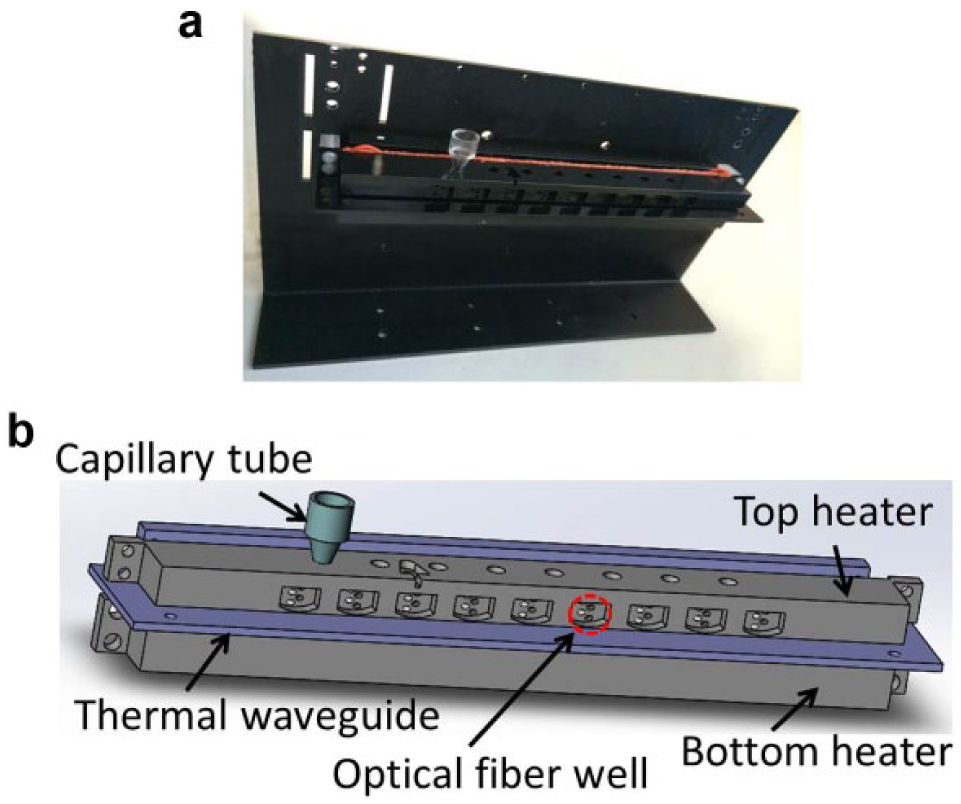
Thermal subsystem of real-time capillary convective PCR: (
Figure 3 shows the thermal subsystem consisting of top and bottom heaters separated by a thermal waveguide. The bottom and top sections of the capillary tube are respectively surrounded by the bottom and top aluminum blocks whose interior sides are respectively attached with two rubberized resistive heater elements.
To prevent inadvertent overheating of the top block (set point 55 °C) due to its close proximity (3.5 mm) to the bottom heater block (set point 95 °C), a cantilevered aluminum (1.5 mm thick) sheet serves as a planar thermal waveguide to isolate the top block from the bottom block by preventing convective heat transfer in the air gap between blocks and shielding the top block from thermal radiation from the bottom heater. The waveguide is attached to a fan-cooled heat sink at the backside of the frame.
Optical Subsystem
Because the strongest fluorescence signal due to the Taqman probe occurs during the extension step of each cycle, the optical probes are inserted through wells in the top heating block, as shown in Figure 3 . To achieve real-time fluorescence detection of up to eight amplification units with two different wavelengths, an optical network consisting of different optical components was developed, as shown in Figure 4 .
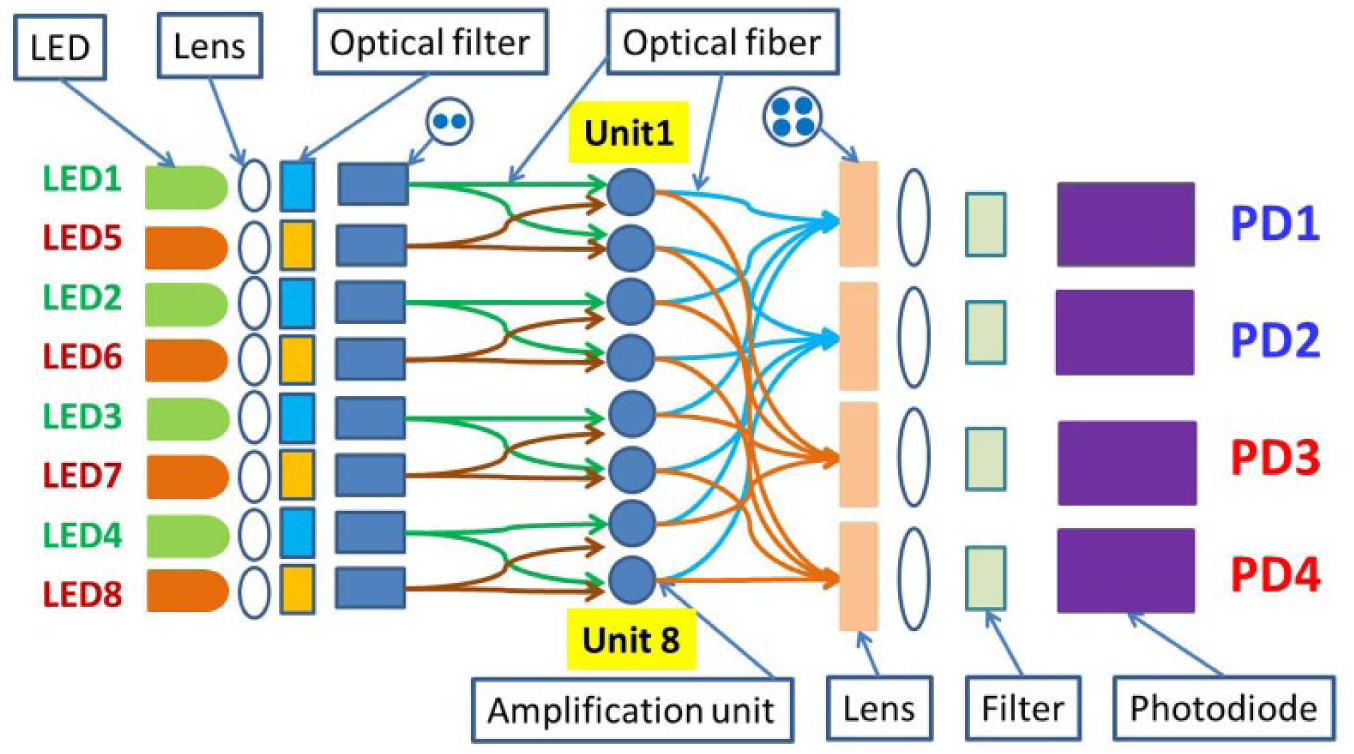
Optical network for real-time in situ fluorescence monitoring.
As shown in Figure 4 , eight LEDs with two wavelengths and four photodiodes are used. Each LED is coupled through an optical lens and filter to two optical fibers, which respectively reach one side of two capillary tubes. The fluorescence emission of each of the eight amplification units is collected with their own optical fibers on the side of the capillary tubes opposite the excitation fibers and then focused by a lens before being coupled through an optical filter to a photodiode. A control strategy to turn on the LED and read the photodiode according to a predefined sequence was developed for fluorescence detection.
Instrumentation
A dual-loop control system was developed for both top and bottom heating. As shown in Figure 5 , on top of the instrument (290 × 190 × 130 mm), a 7-inch touch screen, which works as the interfacing module (as shown in Fig. 1 ), is used to provide graphical interface for easy operation.

A photograph of the capillary convective PCR instrument with eight independent caps.
As shown in Figure 5 , a separate cap is assigned to each amplification unit to allow each one to run independently. Different from normal PCR, capillary tubes can be put into amplification as needed, even when parts of them have started running, which is beneficial for immediate diagnostics in the emergency room or other point-of-care diagnostics.
Results and Discussion
The average ramp rates for the bottom and top heating blocks are 0.28 °C/s and 0.23 °C/s, respectively. With proper tuning, overshoot is negligible (<0.1 °C), and the steady-state temperatures are maintained within ±0.1 °C. The temperature uniformity between eight amplification units for both the top and bottom heaters is reasonably under 0.3 °C.
Without the thermal waveguide, the temperature of the unpowered top heater reaches 43 °C because of heat from the bottom heater at a room temperature of 25 °C. It can be anticipated that when the room temperature is close to 40 °C, the temperature of the top heater will most probably exceed 55 °C because it has no cooling capacity. Therefore, the waveguide is essential for precise maintenance of the 55 °C to 95 °C temperature difference over the length of the capillary tube since neither block can efficiently dissipate heat.
Detection of Influenza Virus
Tenfold serial dilutions of influenza A (H1N1) virus culture stock samples (100–1.0 TCID50/mL) with negative control (NC) were respectively tested. The primer sequence used with the influenza H1N1 template is expected to yield a 105-bp amplicon. In other experiments, it has been proved that up to an 800-pb amplicon with an RNA template of enterovirus 71 (EV71) can be successfully obtained with the developed CCPCR within 30 min.
The CCPCR reaction mix comprised a 3-µL mixture of primers (Beijing Wantai Biological Pharmacy Enterprise, Ltd., Beijing, China) and a Taqman probe labeled with FAM at the 5′ end and Eclipse at the 3′ end, 4 mM dNTP and 4 µL Fast Buffer I (Mg ion buffer) (both Takara Bio, Shiga, Japan), 0.4 U AMV reverse transcriptase (Promega, Madison, WI), 1 U SpeedSTAR HS DNA polymerase (Takara Bio), 10 µL purified influenza A (H1N1) RNA as per above protocol, and molecular-biology grade water to a total reaction volume of 40 µL. After loading the PCR reaction mixture, the reaction was sealed with a layer of 10 µL sterile mineral oil (Sigma-Aldrich, St. Louis, MO).
The production of amplicons with Taqman probes labeled with FAM was monitored in real time by measuring fluorescence via optical fiber probes as described above. As shown in Figure 6 , the real-time fluorescence intensity curves saturated after approximately 25 min, and thus the amplification time can be reduced accordingly without a loss in sensitivity.
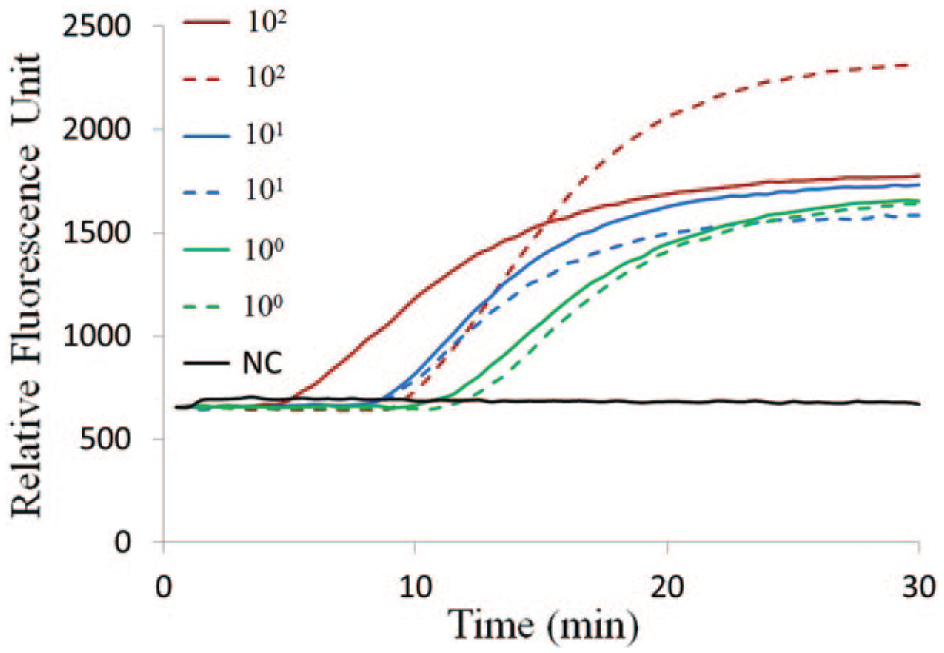
Real-time capillary convective PCR with H1N1 influenza virus RNA template. The detected fluorescent intensity for each sample is indicated as a function of the amplification time. The two red lines (solid and dotted), two blue lines (solid and dotted), two green lines (solid and dotted), and one black line represent, respectively, samples with concentrations of 100, 10, 1.0 TCID50/mL, and negative control (NC).
For the H1N1 clinical test, a limit of detection of 1.0 TCID50/mL is feasible with commercial kits. Different from the conventional PCR, in CCPCR, there is no periodic cycling, and thus analogous quantification of a template based on the number of cycles is not feasible. However, as with isothermal amplification techniques (where a cycle number is not relevant), the amplification time to reach a threshold fluorescence intensity is expected to be inversely related to the log of the template concentration. Because currently, the reproducibility of convection characteristics from run to run in the same instrument and between different instruments is challenging, as shown in Figure 6 , the two curves for 100 TCID50/mL are significantly different in both intensity and the time that they start to increase. For quantitative detection, the accurate correlation of the template with amplification time merits further study.
In conclusion, we successfully developed and tested a real-time CCPCR instrument that is able to shorten PCR amplification time from typically 1 to 2 h to less than 30 min with significant convenience. We have developed a simple, efficient, and concise heating strategy that facilitates optical access for real-time monitoring of the reaction progress. A thermal waveguide was proofed to be essential to maintain the accurate temperature for the top heater. A custom optical network consisting of different optical components was developed for fluorescence detection. The real-time CCPCR instrument is able to detect influenza A (H1N1) virus sample with a concentration of 1.0 TCID50/mL repeatedly. The cost of the real-time CCPCR instrument is extraordinarily lower than that of typical real-time PCR instruments. The real-time CCPCR instrument with its flexible operation mode provides a promising solution for simple, fast, low-cost, and sensitive nucleic acid–based analysis in point-of-care settings.
Footnotes
Acknowledgements
Mr. Qi Guo, Shuo Yang, and Liang Feng provided help on software development of the instrument system.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the National Natural Science Foundation of China (No. 81371711) and the Fundamental Research Funds for the Central Universities (ZZ1329,YS1404).
