Abstract
A method is described for using custom snap-on lids to protect chemicals in microtiter plates from evaporation and contamination. The lids contain apertures (diameter 1.5, 1.0, or 0.5 mm) through which the chemical building blocks can be transferred. The lid with 0.5 mm apertures was tested using a noncontact acoustic liquid handler; the 1.0 and 1.5 mm lids were tested using two tip-based liquid handlers. All of the lids reduced the rate at which solvents evaporated to room air, and greatly reduced the rate of contamination by water and oxygen from room air. In steady-state measurements, the lids reduced the rate of evaporation of methanol, 1-hexene, and water by 33% to 248%. In cycled experiments, the contamination of aqueous solvent with oxygen was reduced below detectability and the rate at which DMSO engorged atmospheric water was reduced by 81%. Our results demonstrate that the lids preserve the integrity of air-sensitive reagents during the time needed for different types of liquid handlers to perform dispensations. Controlling degradation and evaporation of chemical building blocks exposed to the atmosphere is increasingly useful as the reagent volume is reduced by advances in liquid handling technology, such as acoustic droplet ejection.
Keywords
Introduction
Improvements in liquid handling technology
1
underlie transformative new capabilities for applications such as bioassays
2
and chemical syntheses.
3
In many cases, the frontier for the miniaturization of fluid phase assemblies has been pushed to volume regimes of just a few nanoliters.
4
One challenge is that miniaturization increases the ratio of the surface area to volume and accelerates both the evaporation of nanoscale solvents into room air and their contamination by chemicals in the atmosphere, such as water vapor, oxygen, nitrogen-containing compounds, and carbon dioxide. Techniques for managing air-sensitive and moisture-sensitive chemicals on a

Frustrations of library curation. Collaborators supplied our screening facility with a library of chemical fragments in aqueous buffers containing dithiothreitol (DTT) and DMSO. (
We previously described a simple technique to isolate specific types of experiments from room air. 15 Here, we describe a similar but more general approach for using snap-on climate control lids with small apertures to protect chemical building blocks that are located within source plates from the surrounding atmosphere during dispensation (the liquids are transferred through the small apertures in the lid). By greatly reducing both the evaporation and contamination of solvents, the useful life of valuable and fragile chemicals can be greatly extended. The lids are fastened under an inert atmosphere before liquid transfer begins. When all transfers are complete, the lids are replaced with adhesive plate sealant ( Fig. 2 ).

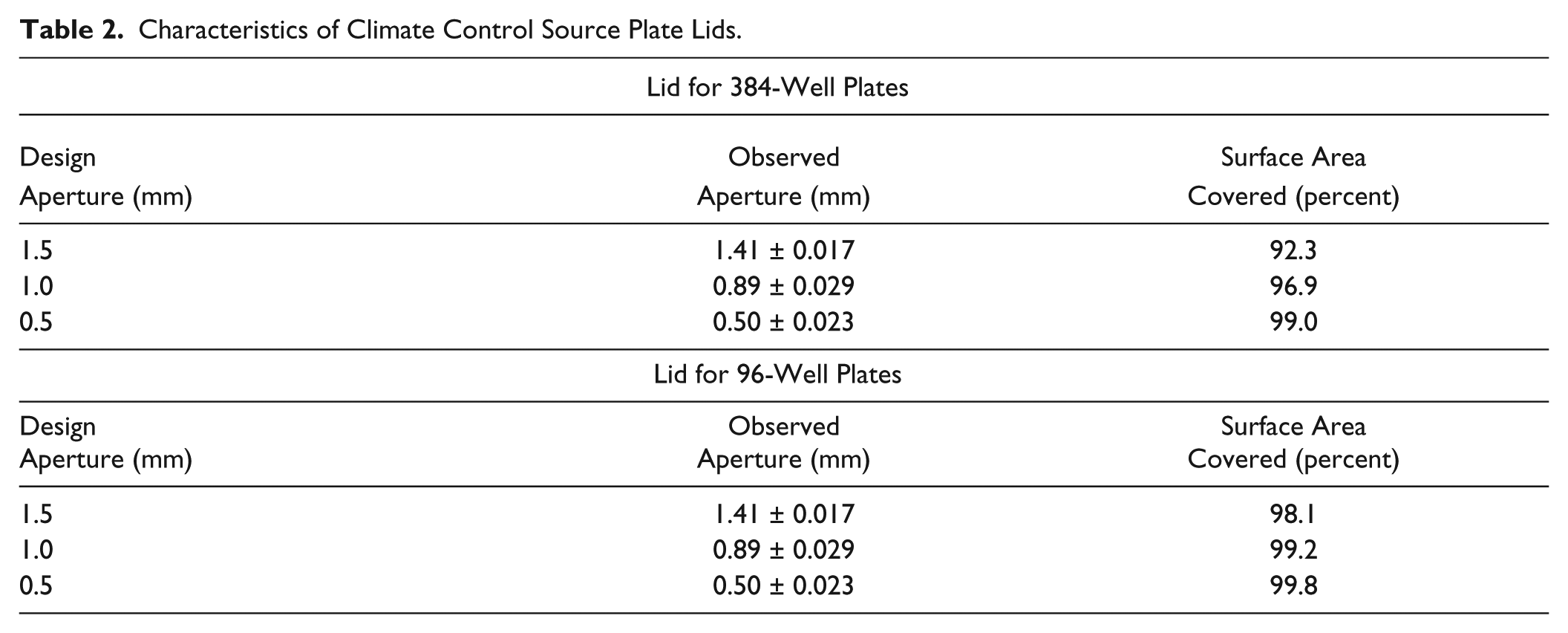
Climate control source plate lids. Three tested source plate lids with target aperture diameters of 1.5, 1.0, and 0.5 mm (A1, B1, and C1, respectively) that were designed to fit a 384-well polypropylene microplate (Labcyte, Inc., Sunnyvale, CA; “PolyPro” source plate hereafter) and three untested source plates that were designed to fit a 96-well Nunc source plate (A2, B2, and C2). We measured the diameter of 20 apertures in each plate type to confirm that the target diameters were accurate (the measured diameters were 1.41 ± 0.017, 0.89 ± 0.029, and 0.50 ± 0.023 mm, respectively). We designed a series of climate control lids to maintain a local microclimate inside of the PolyPro source plate (to preserve volatile solvents or air-sensitive chemicals). The lid designs for the 96-well source plates that were not tested are also available. The plate lids were constructed out of 1 mm thick acrylonitrile butadiene styrene (Amtek P430, Eden Prairie, MN) using a 3D printer (Stratasys Dimension Elite, Arnold, MD).
We designed climate control lids that are compatible with a variety of liquid handling instruments. Some liquid handlers require larger apertures to accommodate the liquid transfer mechanism, so we designed lids with apertures having three different diameters (1.5, 1.0, and 0.5 mm). We then measured the reduction in the rate of evaporation of various common solvents when the plates were continuously exposed to room air, as well as the reduction in the rate of contamination by water 16 and oxygen when the plates were cyclically exposed to room air. Our results suggest that using climate control lids can protect the integrity of vulnerable chemicals, greatly extending the useful life of chemical libraries such as fragment, reagent, and ligand libraries. Lids with smaller apertures yielded the greatest improvement, and the acoustic noncontact liquid handler that used the smallest apertures had the additional benefit that there was no chance of a collision due to an error in positioning or alignment.
Methods
Although there is a huge diversity of designs for liquid handling robots, the component responsible for separating each liquid aliquot from its storage reservoir must utilize a washable tip, a disposable tip, or no tip. Each of these strategies imposes a different clearance requirement for the physical space that must remain unobstructed immediately above the storage reservoir (through which the liquid aliquot is dispensed). We compared the clearance requirements (obtained from published material and from inquiries to the manufacturers) for a variety of commonly used liquid handlers ( Table 1 ). With this information, we designed lids with apertures of three sizes to accommodate the majority of the specifications ( Table 1 is divided into three groups of liquid handlers that are compatible with the three designs). To test our designs, we obtained access to two liquid handling robots with a washable tip17,18 and one with no tip. 19 These three instruments could be accommodated by apertures having design diameters of 1.5, 1.0, and 0.5 mm, respectively. Before testing the effectiveness of our climate control lid designs, we verified that each of the three liquid handlers was able to dispense liquids from source trays that were covered by the relevant lid ( Fig. 3 ).
Characteristics of Commonly Used Liquid Handling Robots.


Clearance requirement for three liquid handling robots. The physical space that must remain unobstructed immediately above the storage reservoir is demonstrated. (
An undergraduate engineering intern used the
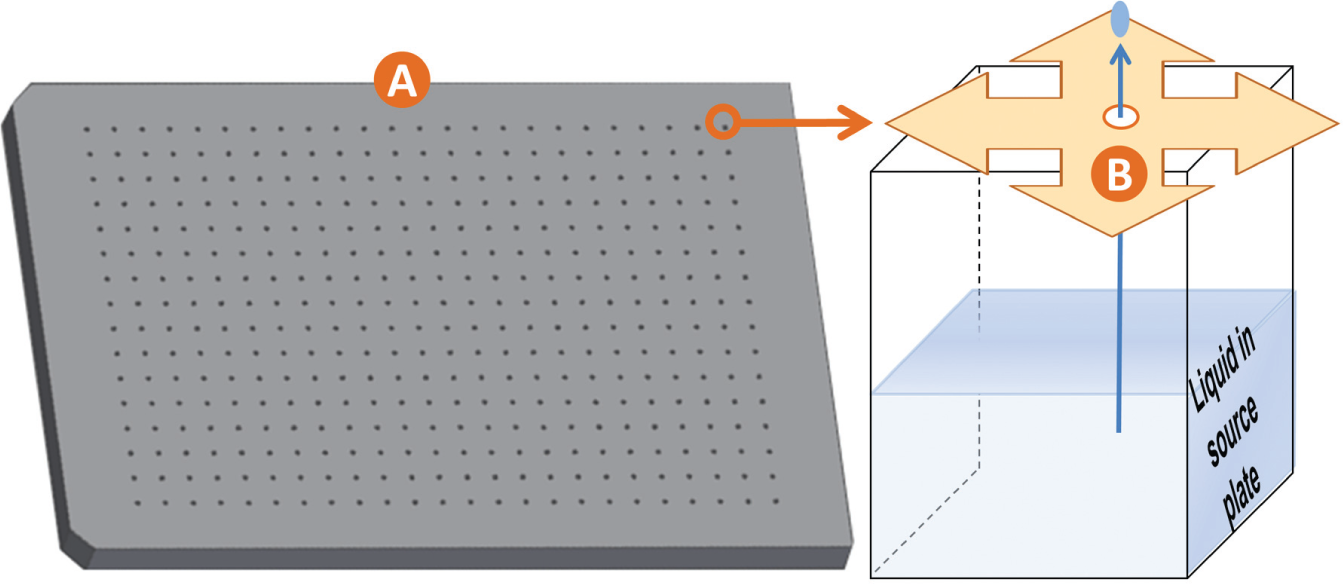
Apertures in climate control lids. Very small volumes of chemical building blocks can be damaged by atmospheric evaporation and contamination. To prevent this, we designed and tested climate control plate lids (
Characteristics of Climate Control Source Plate Lids.
Measuring Evaporation Rates for Methanol, 1-Hexene, and Water
We used the EchoSurvey software to deduce the volume in each of the 384 wells in a PolyPro source plate by projecting a sound pulse (or “ping”) through the plastic bottom of the plate and measuring the time needed until the ping was reflected back from the fluid surface. 21 Using this method, we periodically recorded the volume in each of the wells of PolyPro plates (with no lids) that were initially filled with 50 µL of three test solvents (methanol, 1-hexene, and water). Similar measurements were made for the same three test solvents in plates that were covered with climate control lids with aperture diameters of 1.5, 1.0, and 0.5 mm. We recorded the temperature and humidity in the laboratory during our measurements using a sling psychrometer (Sper Scientific, Scottsdale, AZ; 20~120 °F). We periodically checked the consistency of our results (i.e., consistency across all of the wells in each plate) by superposing topographical surface plots of the percent remaining volume in the wells of plates with and without climate control lids (we tracked percent remaining volume to avoid confounding our results with changes during setup) ( Fig. 5 ). We noticed that evaporation from plates without lids was nonuniform (with significantly more evaporation occurring in wells near the edges of the plate compared to wells near the interior of the plate). To prevent transient features such as these “edge effects” from influencing our calculated evaporation rates, we used the slope of a linear best fit to the volume change data as the best estimate of the evaporation rate for each solvent in each plate ( Fig. 6 ).
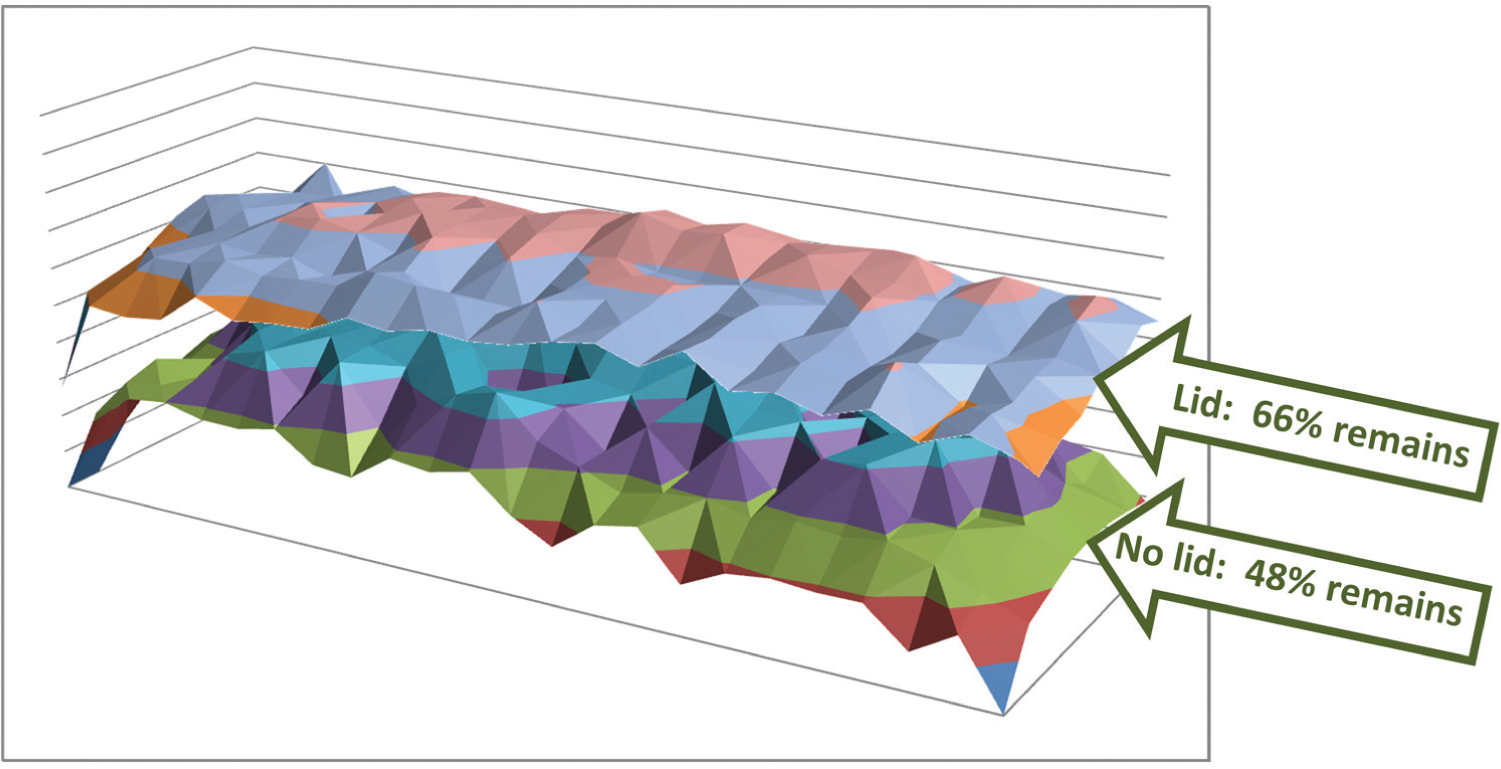
Climate control lids reduce evaporation and reduce edge effects. Climate control lids reduce evaporative changes in the volume of solvents ( Fig. 6 ) and also reduce nonuniform evaporation of solvents located near the edge of the source plate. The final volume at the end of our evaporation trial for methanol is shown for each of the 384 wells in an uncovered source plate (bottom) and also in a source plate covered with a climate control lid containing 1.5 mm apertures (top). The average volume for all of the 384 wells in the uncovered plate was 48% of the starting volume, compared to 66% of the starting volume for the covered plate (this corresponds to the data point that is boxed in green in Fig. 6 ). In addition to the reduction in overall evaporation, the climate control lids also improved the uniformity of the solvent. The increased evaporation in wells located near the edges of the uncovered plate is clearly visible compared to similar wells located in the interior of the plate. Similar results were observed for all of our measurements (data not shown).
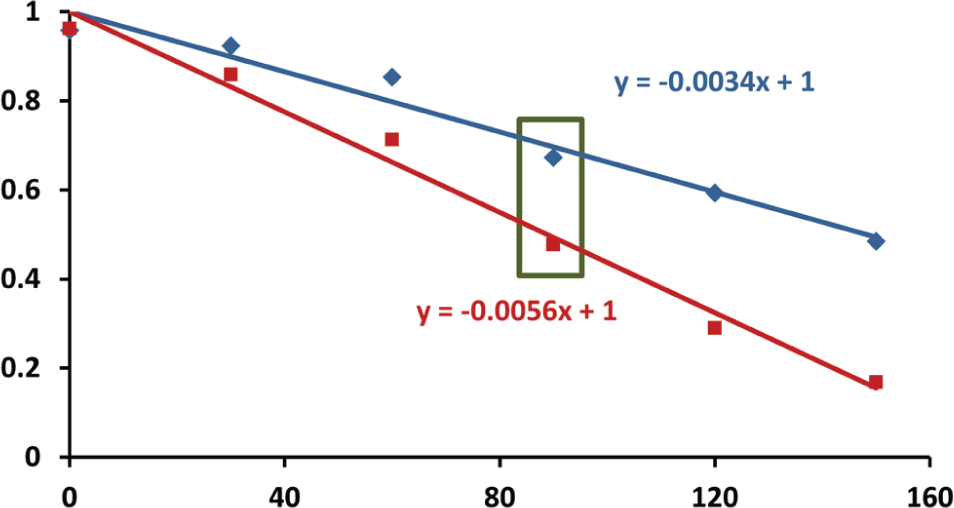
Deducing the evaporation rate. We periodically compared the remaining volume of solvent (methanol, 1-hexene, and water) in each of the 384 wells of an uncovered PolyPro plate with the remaining volume in a PolyPro plate that was covered with a climate control lid (the two data points obtained from Fig. 5 are boxed in green here). Each solvent was tested using no lid, using a lid with 1.5 mm apertures, using a lid with 1.0 mm apertures, and using a lid with 0.5 mm apertures. For each of the three tested solvents and for each of the three aperture sizes, the reduction in the rate of evaporation was taken as the slope of the linear best fit to the data obtained from plates fitted with a climate control lid divided by the slope of the data obtained from a plate with no lid. As an example, the remaining volume (in percent) is plotted as a function of the time (in minutes) for source plates containing methanol that were covered with a plate lid containing 1.5 mm apertures (blue data points) and that were not covered (red data points). In this case, the slope obtained from the source plate that was covered by a climate control lid is −0.34%/s, and the slope obtained from the uncovered lid was −0.56%/s. Consequently, we calculate that the evaporation rate was reduced by {(0.56 − 0.34)/(0.56)} = 39%. More importantly, we deduce that the total time available before reaching an acceptable level of evaporation is increased by {(−0.56/−0.34) − 1 } = 64.7%. Figure 7 shows the observed reduction in evaporation of each of three tested solvents using lids with each of three aperture sizes (the data from Fig. 6 is boxed in green in Fig. 7 ).
Measuring Contamination of an Aqueous System by Oxygen
Although there are many techniques to accurately measure the concentration of oxygen in a test fluid, most of these cannot be used to track the oxygen infiltration into each of the 384 wells of a source plate in real time. We used the oxidation of a reduced form of methylene blue (immobilized by 2% agar) as an indicator of the distance that oxygen penetrated into each of the 384 wells of a source plate. 22
We heated 100 mL of 2% w/v agarose to 100 °C in an Erlenmeyer flask for 5 min, and then cooled and maintained the solution at 60 °C. We then added glucose (140 mM final), sodium hydroxide (80 mM final), and 120 µL of 1% methylene blue. During the preparation, nitrogen gas was bubbled through the solution to remove oxygen. The resulting reduced “indicator gel” was colorless and translucent. We transferred 79 ± 11 µL of indicator gel into all 386 source wells of two PolyPro source plates and then allowed the source plates to be fully cleansed of oxygen in an oxygen-free box (
Both tested plates (covered with a 0.5 mm diameter plate lid and an uncovered control) were cycled into the oxygen-free acrylic box for 0.5 min and into room air for 5.0 min. The plate lid was removed from the covered plate while it was in the acrylic box, and replaced when it was moved to room air (to reproduce the experimental conditions that would be observed if a chemical-containing plate were repeatedly used for liquid dispensation and then returned to storage). Every 4 h during 20 h of observation, one cuboidal indicator gel from the gel-containing wells in each of the source plates (control and covered) was individually transferred to water that had been deoxygenated using nitrogen bubbling (as described earlier) for visualization (Leica MZ16 F).
Measuring Contamination of DMSO by Water
We used the EchoSurvey software to deduce the percentage of DMSO and the percentage of engorged water in each of the 384 wells in four PolyPro source plates (one without a lid and three with lids) that were cycled between a dry nitrogen chamber (for 0.5 min) and a 100% humidity chamber (for 5 min). We used the same apparatus as was used to measure oxygen contamination (
Extending the Usable Life of Grubbs’ Catalyst
We prepared a saturated solution of Grubbs’ catalyst by adding 300 mg to 10 mL of toluene (first generation, Sigma-Aldrich, St. Louis, MO; no. 579726) and dispensed the solution to two 386-well plates (50 µL aliquot per well) in a single-use nitrogen glove bag, retaining leftover solution in two tubes to serve as benchmarks. Both tested plates (covered with a 0.5 mm diameter plate lid and an uncovered control) were cycled into the oxygen-free acrylic box for 0.5 min and into room air for 5.0 min for 4 h (as described earlier). We regularly monitored the volume in each well and used a Hamilton microsyringe to replace evaporated solvent (toluene is volatile and we did not want to confound reduction in evaporation with reduction in contamination by room air). After 4 h, the solution from the wells in each plate was combined and sealed in a centrifuge tube. After 48 h in the centrifuge tubes, we used a NanoDrop spectrophotometer (Thermo Scientific, Waltham, MA; ND-2000c) to obtain a UV-Vis spectrum from each specimen, and also from an uncontaminated benchmark that was never exposed to room air and from an oxidized benchmark that was intentionally damaged by mixing with nondegassed water for 24 h.
Results
There are two classes of problems to avoid during nanoliter dispensations: those that cannot be mitigated by a glove box (such as solvent evaporation) and those that can (such as oxygen poisoning and absorption of room water). Climate control lids protect liquids in two ways during dispensations: (1) they prevent, or slow, the evaporation of solvents, and (2) they protect the chemical properties of the solvated chemicals from contamination by outside air. In the steady-state measurements, climate control lids significantly reduced the rate of evaporation of methanol, 1-hexene, and water. In cycled experiments, the contamination of aqueous solvent with oxygen was reduced below detectability and the rate at which DMSO engorged atmospheric water was reduced by 81%. Our results demonstrate that climate control lids can be instrumental for preserving the integrity of air-sensitive reagents during the time needed for different types of liquid handlers to perform dispensations.
Climate Control Lids Reduce Evaporation Rates for Methanol, 1-Hexene, and Water
Figure 7 shows that using source plate lids effectively reduced the rate of evaporation of methanol, 1-hexene, and water for all of the tested aperture sizes. The reduction in evaporation rate was well correlated with the volatility of the solvent. The ratio of the average rate of evaporation for methanol and 1-hexene was 1:1.46, which is in good agreement with relative volatilities of the two solvents (1:1.37) (the ratio for water is omitted because it is confounded by room humidity). Consequently, we conclude that the reduction in the evaporation rate of solvents by using climate control lids increases as a function of the volatility of the solvent.
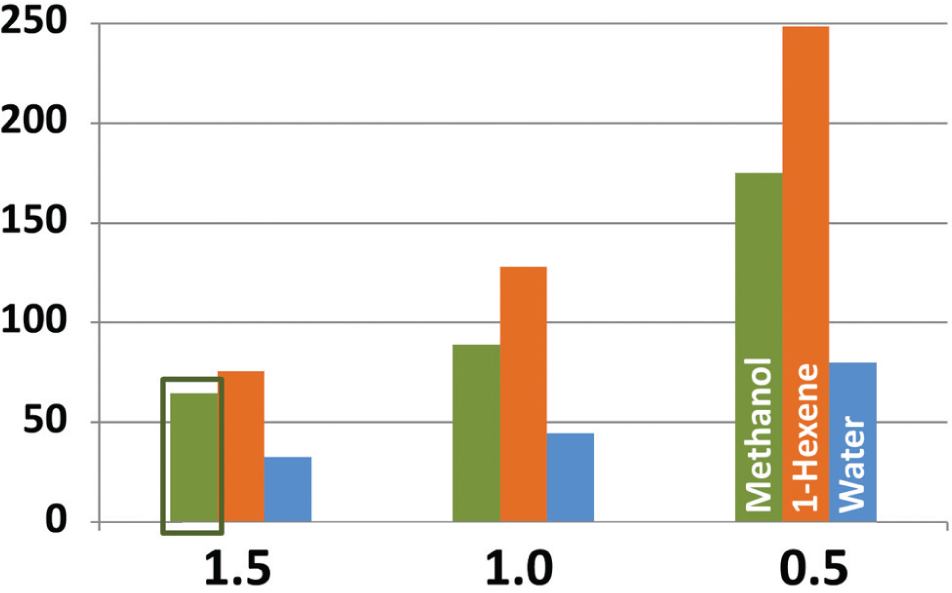
Climate control lids reduce solvent evaporation. The reduction in the evaporation rate for plates with climate control lids compared to uncovered plates was deduced from the ratio of the slope of the percent volume change as a function of time (as shown in Fig. 6 ). The data point obtained from Figure 6 is boxed in green here.
In addition to reducing the average evaporation rate for all wells in a source plate, the climate control lids also improve the uniformity of performance across all wells in the plate. Figure 5 shows that solvents stored in unprotected source plates undergo differential evaporation because chemicals located near the interior of the source plate are less exposed to room air than chemical located at the edges of the source plate. 16 Use of plate lids greatly reduces these edge effects, because the plate lids uniformly protect all of the chemicals in the plate.
Climate Control Lids Reduce Contamination of Aqueous System by Oxygen
Figure 8 shows that the climate control lid with 0.5 mm diameter apertures significantly reduced the penetration of oxygen into the indicator gel. After 20 h of cycled exposure to room air, the entire colorless indicator gel was oxidized to a blue color in the control plate, compared to no observable discoloration in the plate that was protected by the climate control lid. Lids with larger aperture sizes slowed the rate of oxygen penetration somewhat, as evidenced by a reduced rate of oxidation of the indicator column (data not shown). However, only the 0.5 mm apertures allowed the indicator column to fully recover from each 5 min oxygen exposure during the subsequent 0.5 min in the nitrogen box, allowing the indicator column to remain colorless indefinitely.

Climate control lids reduce the contamination of chemicals by oxygen. Agarose was used to immobilize a methylene blue–based oxygen indicator in each well of a source plate to compare to the rate of oxygen penetration into the agar column in a source plate that was fitted with a 0.5 mm aperture climate control lid with the rate of penetration in an uncovered plate. When the source plates were continuously exposed to room air, we observed no difference in the oxygen penetration rate (deduced from the depth of color change) between covered and uncovered plates. However, a striking difference in deduced oxygen penetration was observed when both plates were cyclically rotated between an oxygen-free nitrogen box (for 30 s, both plates uncovered) and room air (for 5 min, one plate covered). Similar measurements made using 1.0 and 1.5 mm aperture lids showed a smaller reduction in the oxygen penetration rate (data not shown).
Climate Control Lids Reduce Contamination of DMSO by Water
We used the EchoSurvey software to demonstrate that climate control lids reduce DMSO engorgement in source plates that were cycled between dry nitrogen and high humidity (the cycling reproduces the environmental changes experienced during repeated liquid dispensations followed by plate storage). We compared the rate of DMSO engorgement of water for source plates with and without lids. Water engorgement was reduced for plates protected by all of our designs, but the reduction was most significant using lids with 0.5 mm apertures (
Fig. 9
). Overall, the climate control lids reduced the rate of water engorgement by DMSO by 34%, 61%, and 81% using lid apertures of 1.5, 1.0, and 0.5 mm, respectively (see
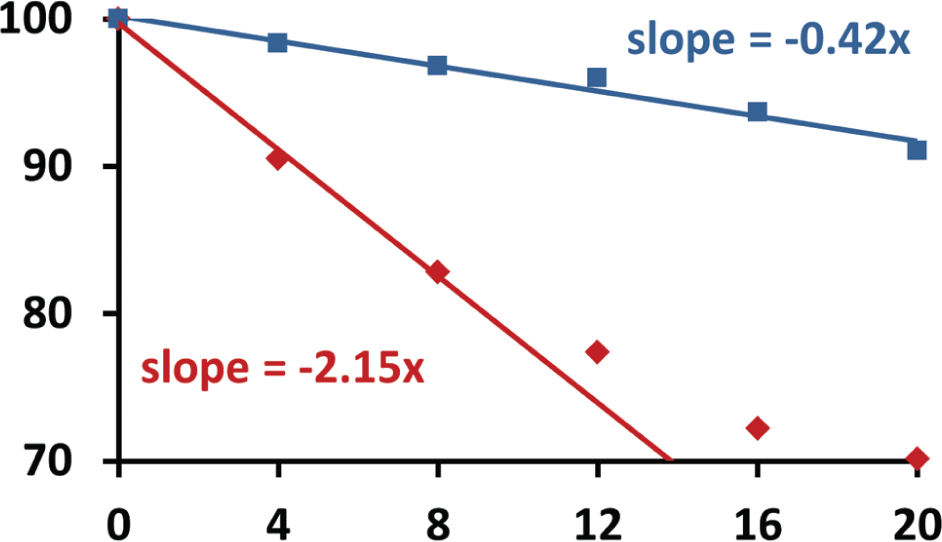
Climate control lids reduce the engorgement of water by DMSO. The percentage of DMSO is shown as a function of time for a source plate that was covered with a climate control lid containing 0.5 mm apertures (blue) and compared with the percentage for an uncovered control plate (red); the extra volume is composed of engorged water. The initial concentration of DMSO was scaled to 100% (the measured initial DMSO concentration was slightly less than 100% because some water was engorged during plate preparation). The software that was used to deduce the DMSO ratio is unreliable when there is more than 30% water, so all compositions below 70% DMSO are reported as 70% (consequently, the DMSO composition asymptotically approaches 70%). Linear best fits are shown for both data sets. Only the first three data points were used to determine the best fit for the control plate composition, because of the measurement limits described above (some of the outeredge wells had engorged 30% water by the fourth measurement). The slopes for the two best-fit lines are 0.42% water engorgement per hour (for the plate covered with a climate control lid) and 2.15% water engorgement per hour (for the control plate). The rate of water engorgement was reduced by 81% for the plates covered with lids compared to the control plates. Similar measurements made using 1.0 and 1.5 mm aperture lids showed a smaller reduction in the rate of water engorgement (see
Usable Life of Grubbs’ Catalyst Extended ~4.5-Fold
Figure 10 shows spectroscopy measurements and microscope pictures of each of the four Grubbs’ catalyst solutions. Note that the fully oxidized benchmark does not account for losses of Grubbs’ catalyst during the oxidation procedure (e.g., due to precipitation of damaged catalyst). We used the region between 475 and 675 nm to estimate the degree of protection conferred by the climate control lid by extrapolating the difference between the uncontaminated control and the lid-covered data by 4.5-fold to best fit the uncovered data. Those derived data points are also shown in Figure 10 , corresponding to a 4.5-fold reduction in the rate of change observed in the absorbance between 475 and 675 nm.
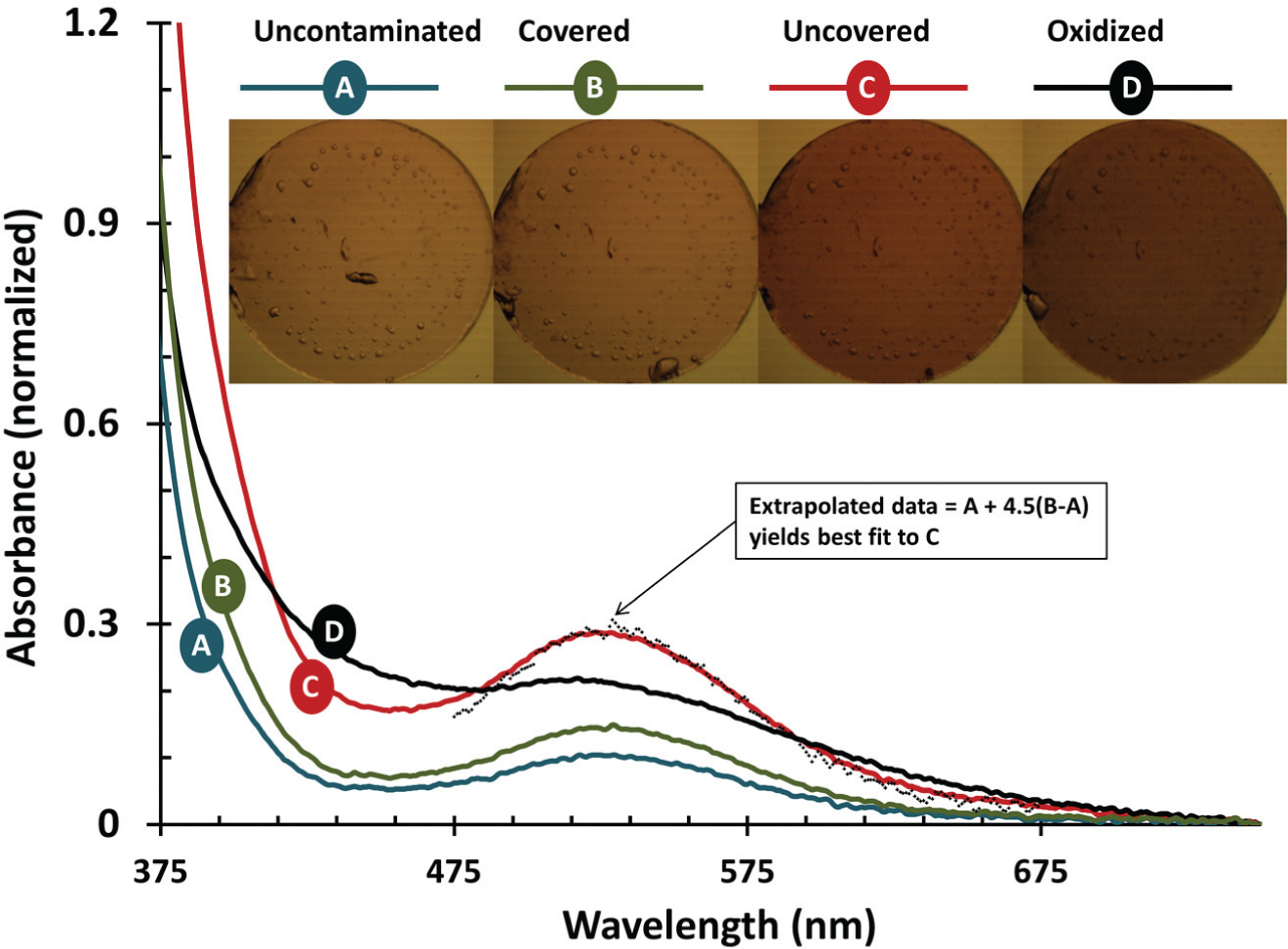
Climate control lids reduce the rate of contamination of Grubbs’ catalyst. Pictures and UV-Vis spectra are shown for solutions of Grubbs’ catalyst in toluene that were never exposed to room air (blue line A), that were protected by climate control lids (green line B), that were not protected (red line C), and that were intentionally oxidized (black line D). Extrapolating the A→B trend by 4.5 best fits C, which we postulate may indicate that the rate of damage caused by room air is decreased by a factor of 4.5 using the climate control lids (+ symbols).
Discussion
Nanoliter liquid handling can facilitate high-throughput screening applications such as fragment-based drug discovery and organic synthesis. 24 These improvements can make prominent research more affordable for well-funded private sector businesses as well as more accessible to academic laboratories with limited resources. Crystallography groups (including ours) have leveraged miniaturization to decrease the cost of specimen preparation25,26 and to increase the speed of crystallographic bioassays.27,28 These advantages have motivated numerous efforts to improve the scale and efficiency of liquid handling robots using technologies such as conventional air displacement and piezoelectric ink jets. 29 Clearly, the advantages of miniaturizing liquid handling can only be realized if the savings are reflected in lowered consumption of chemicals stored in the source plate. One challenge has been that chemicals in source plates can degrade before they are fully utilized. This degradation can occur because of evaporation of the solvent or because of contamination of the chemical by environmental gasses. The importance of the physical and chemical integrity of building blocks is highlighted by the extensive efforts to prevent cross-contamination of specimens and to reduce contamination by the sample container. 30 Unsurprisingly, much of the successful nanoliter biology and chemistry has occurred in apparatuses with no air–liquid interface, such as microfluidics and microbatch.31,32 However, combinatorial chemistry (for bioassays or for chemical synthesis) is difficult to do with microfluidic technologies because the diversity of tested chemicals is too great.
We have demonstrated that source plate lids can greatly reduce the problems commonly encountered when miniaturizing liquid handling that has an air–liquid interface. Evaporation of the solvent is reduced in the steady state, and contamination of the chemicals is reduced when the plate is cycled between dispensation and storage. Lids with the smallest apertures, appropriate for use with acoustic droplet ejection, most effectively maintained a protective microclimate in each source plate. Acoustic droplet ejection is a noncontact method that has the additional advantage that misalignment between the source plate lid and the dispensation robot cannot cause a collision. The rapid advances in advanced liquid handling robots partnered with simple techniques to protect the smaller (and consequently more vulnerable) chemical aliquots may be natural partners to allow the overall quality of assays to be governed by the improvements in liquid handling, as opposed to plateauing because of environmental contamination issues. 33 These new technologies (in combination with rigorous testing of the performance of liquid handling robots34,35) will allow future biological and chemical innovations to probe smaller specimens and exploit more challenging opportunities.
Footnotes
Author Contributions
We thank Labcyte, Inc., and especially Joe Olechno, Richard Ellson, and Richard Stearns, for their technical support and guidance. A.S.S. designed the experiment and wrote the paper with input from all coauthors. B.J.F., A.M.D., M.T.B., D.L., B.A., N.P., N.Z., and A.S.S. tested the labware, prepared specimens, obtained data, and analyzed data. A.S.S., A.S.R., and R.J. designed and built the labware. A.S.S. trained and supervised undergraduate interns with the support of R.M.S. and S.M.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Personnel for this study were recruited largely through the 2015 spring and summer sessions of the Science Undergraduate Laboratory Internships Program (SULI) and High School Research Program (HSRP), supported through the U.S. Department of Energy, Office of Science, Office of Workforce Development for Teachers and Scientists (WDTS). Major ongoing financial support for the LSBR specimen preparation laboratory was through the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, under contract no. DE-AC02-98CH10886. The Life-Science and Biomedical Technology Research Resource for NSLS-II is supported by the DOE Office of Biological and Environmental Research, proposal E-SC0012704, and by the National Institutes of Health, National Institute of General Medical Sciences, including P41GM103473 and P41GM111244.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
