Abstract
Industrial enzymes are instrumental in many applications, including carbohydrate processing, fabric and household care, biofuels, food, and animal nutrition, among others. Enzymes have to be active and stable not only in harsh application conditions, but also during shipment and storage. In protein stability studies, formulated concentrated enzyme solutions are frequently diluted gravimetrically prior to enzyme activity measurements, making it challenging to move toward more high-throughput techniques using conventional robotic equipment. Current assay methods pose difficulties when measuring highly concentrated proteins. For example, plastic pipette tips can introduce error because proteins adsorb to the tip surface, despite the presence of detergents, decreasing precision and overall efficiency of protein activity assays. Acoustic liquid handling technology, frequently used for various dilute small-molecule assays, may overcome such problems. Originally shown to effectively deliver dilute solutions of small molecules, this technology is used here as an effective alternative to the aforementioned challenge with viscous concentrated protein solutions. Because the acoustic liquid handler transfers nanoliter quantities of liquids without using pipette tips and without sample loss, it rapidly and uniformly prepares assay plates for enzyme activity measurements within minutes. This increased efficiency transforms the nature of enzyme stability studies toward high precision and throughput.
Keywords
Introduction
Industrial enzymes play an enormous role in many applications, ranging from carbohydrate processing and household care to food, animal feed, and biofuel production.3,4 Frequently, industrial enzymes are used in conjunction with many household products, such as laundry detergents, not only increasing the efficiency of the product, but also posing enormous environmental benefits, such as reducing the amount of industrial and chemical waste going to the landfill and waterways as a result of being biodegradable. 5
Enzymes must be active and stable in harsh applications and during shipment and storage. Therefore, accurately measuring enzyme specific activities over various conditions during protein stability studies in formulation development is crucial. In laboratory conditions, these industrial enzymes are commonly tested and utilized in the ultrafiltrate concentrate (UFC) form. Ultrafiltrate concentrates are concentrated enzyme solutions obtained after the bacterial or fungal fermentation broth has been passed through several recovery steps, including cell matter removal and ultrafiltration.6,7 In most cases, the final UFC solution is very viscous, turbid, and contains a high concentration of enzyme (20–200 mg). To add an additional degree of complexity, UFC material is commonly formulated with viscous excipients (e.g., glycerol and sorbitol) to increase the shelf life of the liquid industrial enzyme product during storage.
Measurement of enzymatic activity of the formulated liquid industrial enzyme products using common volumetric pipetting techniques is inaccurate as sample is lost during transfer, making enzyme quantitation difficult. In an effort to accurately quantify enzyme concentration and activity of formulated and concentrated industrial enzymes, scientists rely on gravimetric dilution. Gravimetric dilution is commonly used in analytical chemistry for the quantitative determination of solids as well as nonaqueous liquid samples, as the technique takes into consideration the mass or density properties of the analyte. 1 Although accurate, gravimetric dilution is time-consuming, tedious, and low-throughput. Because formulated concentrated enzyme solutions used in biotechnology are frequently diluted gravimetrically prior to enzyme activity measurements, it is challenging to move toward more high-throughput techniques with conventional robotic equipment. For example, pipette tips made out of plastic materials can trap viscous solutions, despite the presence of surface antibinding agents, decreasing the precision and overall efficiency of protein activity assays. In addition, large amounts of protein and solvents are used, leading to high amounts of environmental waste and increased cost. These problems are compounded with many subsequent dilutions performed to bring the enzyme concentration into a range acceptable for detection in a standard enzymatic assay.
Acoustic liquid handling or acoustic droplet ejection (ADE) technology, as represented by the Echo 550 liquid handler (Labcyte, Sunnyvale, CA), overcomes many of these problems by using focused acoustic energy to eject solutions in nanoliter increments.2,8 Originally shown to effectively deliver dilute solutions of small molecules using acoustic energy 2 and reproducibly determine IC50 values for small molecules,9,10 we show here that it is also effective in handling viscous concentrated protein solutions. Because acoustic droplet ejection technology transfers nanoliter quantities of liquids without using pipette tips and with no sample loss, it significantly decreases chances of contamination and dilution errors. Acoustic liquid handlers can rapidly and uniformly dilute the enzyme of interest within minutes into multiwell plates, already set up for the assays to be performed. This significantly increases efficiency and accuracy as well as transforms assays performed during protein stability studies toward high precision and throughput, while decreasing costs of these long-run studies.
Acoustic droplet ejection technology is preferred for enzyme formulation stability studies over other precise dilution techniques, such as gravimetric and positive displacement methods, due to (1) the possibility for increasing throughput, (2) rapid sample dilution and setup of assay plates, (3) elimination of ergonomic injuries associated with the gravimetric method of analysis and positive displacement pipetting techniques, (4) reduction of operator-caused dilution errors, and (5) reduction of waste. Although applying acoustic liquid handlers for dispensing formulated concentrated enzyme solutions provides an opportunity to the industrial enzyme field, to increase efficiency and speed of enzyme formulation development in the future, the ADE technology has some limitations that need to be overcome in an effort to increase accuracy based on the results shown in this study. Typical samples used in formulation stability studies for industrial enzymes vary in excipient and protein concentration. To enable a rapid analysis of a multitude of samples, it is necessary to produce dilution and assay microtiter plates that contain both analyte and standard curve samples in one plate. In our applications, the samples used for analysis and the standards used for generating a standard curve come from sources of very different protein concentrations and may have very different viscosities. Delivering standard and analyte onto a single assay plate in one run results in delivering volumes of a few nanoliters for highly concentrated standards and several hundred nanoliters for enzymatic analytes. Therefore, the ADE instrument needs to maintain superior precision and speed (to prevent evaporation from other wells) over delivering hundreds of microdroplets of solution into a single well, and this precision has to be maintained over different sample viscosities without having to set up the instrument again.
However, acoustic liquid handlers do ensure a contaminant-free tipless transfer of concentrated and viscous enzyme solutions and remove problems involved with compounding errors with serial dilutions. Replacing the tedious and time-consuming gravimetric enzyme dilutions with acoustic droplet technology will promote increased data collection and data quality. In the future, many concentrations and types of enzymes in the presence and absence of viscous excipients can be delivered with acoustic instruments to prepare enzymes for activity measurement. Acoustic liquid handlers can eventually be adapted to a wide range of concentrated and formulated industrial enzymes, and other challenging sample types with similar properties, as they expand the throughput of enzymatic assays and transform their precision and efficiency.
The feasibility of incorporating the Echo liquid handler in formulation stability studies of industrial enzymes, as well as understanding the current limitations and future opportunities of the ADE technology, was tested by measuring phytase activity of several enzyme preparations using two substrates, the small synthetic substrate
Materials and Methods
The following series of experiments were performed with formulated and unformulated enzyme UFC samples with known (formulated-control, formulated-standard) and unknown activities as a proof of concept that the acoustic liquid handling system can deliver samples precisely for measuring enzymatic activity. The formulated and unformulated phytase enzyme samples were prepared in-house. The liquid density of the enzyme samples ranged between 1.0 and 1.3 g/mL due to the absence and presence of excipients. Density values were determined by using a scale to measure the weights, in grams, of various enzyme volumes. Enzyme volumes (50–250 µL) were added in increments of 50 µL with a calibrated MR-100 Pos-D positive displacement pipette and respective presterilized piston syringe tip (Rainin Instrument LLC, Oakland, CA) to a weighing boat. The weights of the enzymes (g) were graphed as a function of the dispensed enzyme volume (mL), and the slopes were then taken to be the densities.
Phytase Activity Using pNPP Endpoint Assay
pNPP endpoint assay was performed on various phytase enzyme solutions, including unformulated regular-UFC material recovered using conventional industrial methodology, 11 unformulated modified-UFC that was recovered with a proprietary modified industrial process, and purified phytase, to examine the precision and accuracy of the reported activities, using formulated-standards of the same enzyme as a reference. Phytase purification was accomplished by following the procedure described in reference 11.
The activity value of formulated-standard (24,717 FTU/mL) was assigned by the central QA/QC laboratory of Genencor Division, Danisco (now DuPont Industrial Biosciences) in Rochester, NY, following the phytase reference activity assay described in the
In preparation for the pNPP assay, the enzyme solutions were diluted using a combination of manual dilution and the Echo liquid handler. First, each enzyme sample (60 µL) was dispensed manually into individual wells of a Labcyte 384-well source plate (P-05525). Only a 384-well source plate (and not a 96-well plate) could be used due to the Echo’s mechanical configuration. Assay buffer (0.1 M sodium acetate [pH 5.5] in the presence of 0.01% Tween 80) was added manually to a different well in the 384-well source plate, which would be subsequently used for in-Echo backfilling of all the enzymes in the destination plate (the Echo liquid handler would backfill with nanoliter quantities of buffer). Formulated-control of known activity (41,007 FTU/mL) determined using the same phytase activity assay described in reference 12 was used for an additional degree of confidence of the activity measurement for the UFC, and it was added to another well on the source plate.
For the Echo dispensing, the Echo 550 was set to 384PP_AQ_CP mode, which dynamically adjusts for surface tension and viscosity settings for all deliveries at low protein concentrations, as it was the best dispensing mode available based on our various sample types. The Echo 550 was subsequently used to perform dilutions by delivering precise amounts of formulated-standards, formulated-control, unformulated enzyme samples (regular-UFC, modified-UFC, or purified phytase), and buffer ( Table 1 ) into a 96-well assay plate (Corning 3641). Afterwards, the Rainin Liquidator 96 manual pipetting system (Mettler-Toledo LLC, Columbus, OH) was used to uniformly add 64 µL of assay buffer to all wells containing samples (standards, control, and UFC) as a second backfill, so that the final enzyme concentrations for the standards (0–30 FTU/mL), control (22.5 FTU/mL), and UFC (15 FTU/mL) were within the linear range of the enzymatic assay. The target enzyme dilution for the 6-point enzyme standard curve was between 800- and 26,000-fold, while the target dilution for the UFC was near 5000-fold. After the second buffer backfill, the Echo destination plate was then mixed using a tabletop shaker for 5 min to ensure proper mixing prior to performing the enzymatic assay.
Enzyme Dilution for pNPP Endpoint Assay Using ADE Technology.
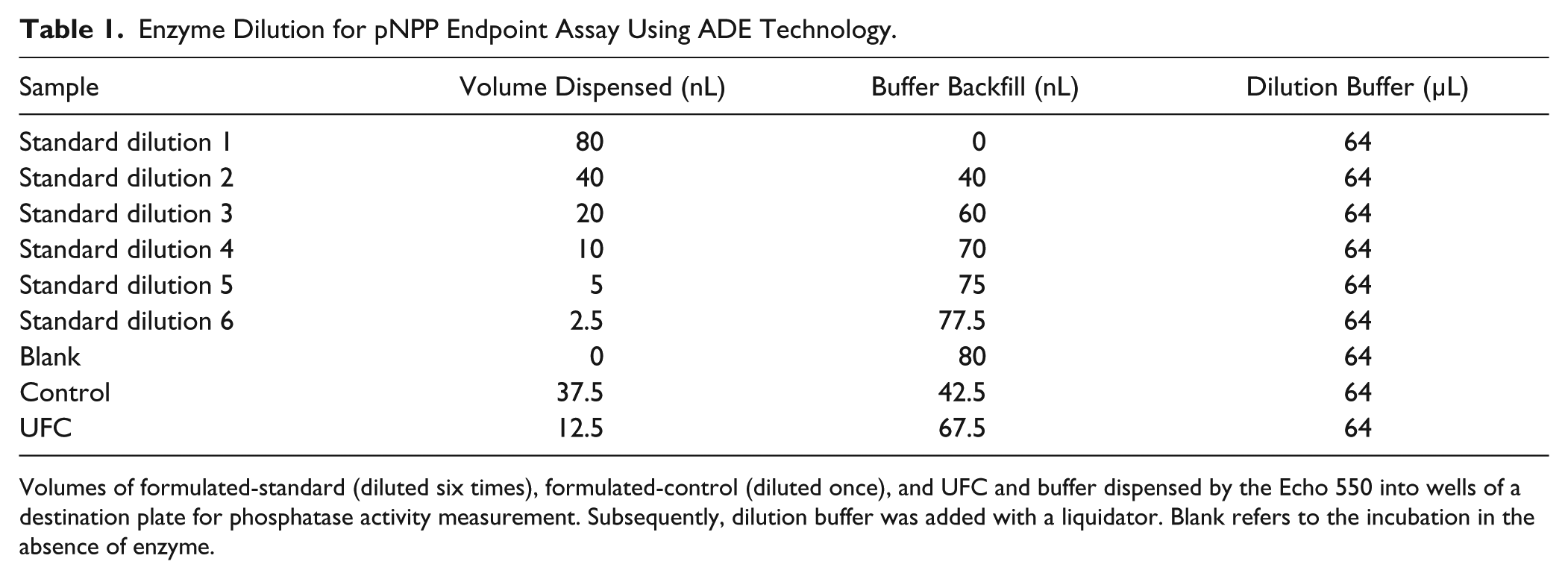
Volumes of formulated-standard (diluted six times), formulated-control (diluted once), and UFC and buffer dispensed by the Echo 550 into wells of a destination plate for phosphatase activity measurement. Subsequently, dilution buffer was added with a liquidator. Blank refers to the incubation in the absence of enzyme.
The pNPP enzymatic assay was then performed on the various phytase samples. The assay described below is a standard pNPP plate assay performed in phytase activity assays at DuPont Industrial Biosciences (Palo Alto, CA). The pNPP endpoint assay was carried out manually by mixing 84 µL of 25 mM pNPP substrate (Sigma N-3254) prepared in assay buffer with 7 µL of Echo-diluted enzyme samples. The reaction was then incubated for 7 min at 25 °C using a Thermo Scientific iEMS incubator/shaker (Thermo Fisher Scientific, Waltham, MA) and quenched with 105 µL of 200 mM Borate (pH 10.2). Absorbance at 405 nm was recorded on a SpectraMax Plus 384 microplate reader (Molecular Devices LLC, Sunnyvale, CA) after 2 min of equilibration. Assay response for the blank reaction was subtracted from all absorbance values. Phytase activities of the UFC and formulated-control were determined using the blank-corrected assay response of the standard curve.
For comparison, a manual pNPP endpoint assay was performed by using enzyme sample (standard, control, UFC) diluted gravimetrically using a Mettler-Toledo XP Analytical Balance by mixing 0.10 g of enzyme with 10 g of buffer in the first dilution. Standard was diluted further by weighing 0.10, 0.51, and 1.0 g of the first diluted standard and mixed with 7.0 g of buffer to form the 3-point standard curve. Formulated-control and UFC sample were diluted once again by mixing 0.2 g with 8.5 g of buffer or 0.10 g with 5.0 g of buffer, respectively. Manual pNPP plate assay was carried out as described above by mixing and incubating 84 µL of pNPP substrate with 7 µL of gravimetrically diluted enzyme samples and subsequently quenching with borate after 7 min at 25 °C. Absorbance at 405 nm was recorded on a SpectraMax Plus 384 microplate reader after 2 min of equilibration.
Precision and consistency of the manual and ADE technology were also studied by measuring phytase activity of modified-UFC over a period of three consecutive days. To clarify the source of the lower-than-expected activity obtained by diluting the modified-UFC sample using ADE technology, sorbitol (VWR, 97062-204) was added to one modified-UFC sample to a final concentration similar to that of the formulated-standard (15%) to make the modified-UFC sample the same viscosity as that of the formulated-standard, while the same volume of assay buffer was added to a second modified-UFC sample. Assay response for the blank reaction was subtracted from all absorbance values. Phytase activities of the UFC and formulated-control were determined using the blank-corrected assay response of the standard curve.
In addition, the phytase activities of purified enzyme at medium (63 mg/mL) and high (306 mg/mL) protein concentrations that were prepared by utilizing either the ADE technology or the manual positive displacement method using calibrated Pos-D positive displacement pipettes (MR-10, MR-100, and MR-1000) and respective presterilized piston syringe tips were assessed. Volumes used for enzyme dilutions using the positive displacement method are listed in Table 2 . Enzyme activity values for standard-UFC, control-UFC, and unknown samples were determined using the same pNPP assay protocol described above.
Enzyme Dilution for pNPP Endpoint Assay Using Manual Positive Displacement Technique.
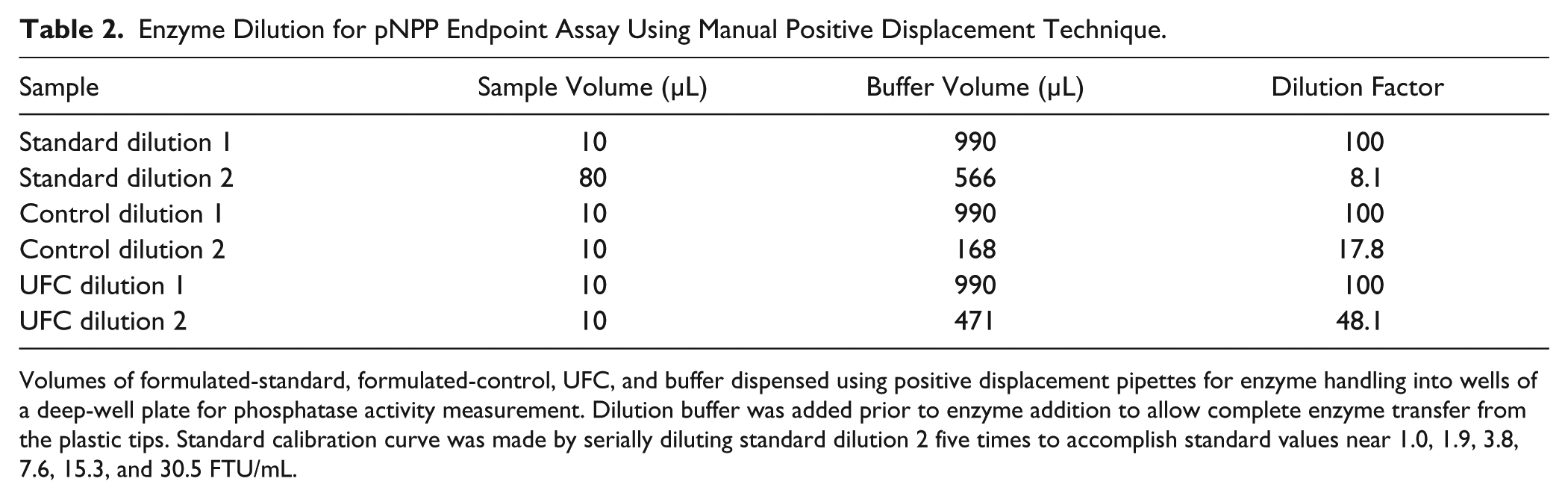
Volumes of formulated-standard, formulated-control, UFC, and buffer dispensed using positive displacement pipettes for enzyme handling into wells of a deep-well plate for phosphatase activity measurement. Dilution buffer was added prior to enzyme addition to allow complete enzyme transfer from the plastic tips. Standard calibration curve was made by serially diluting standard dilution 2 five times to accomplish standard values near 1.0, 1.9, 3.8, 7.6, 15.3, and 30.5 FTU/mL.
Phytase Activity Using Malachite Green Endpoint Assay
The Malachite Green endpoint assay directly quantifies inorganic phosphate that is released from a more complex, natural substrate (phytic acid; Sigma P0109) that lacks colorimetric properties, by complexing the inorganic phosphate with molybdate to form phosphomolybdic acid, detected at 620 nm under acidic conditions. 13 Pi Blue Phosphate Stop Solution (POPB) from BioAssay Systems (Hayward, CA) was used as the source of molybdate. The Malachite Green assay was performed on the industrial UFC solution of a phytase enzyme, by either Echo dilution or manual dilution using Pos-D positive displacement pipettes to examine and compare the precision and accuracy of the reported activity using formulated-standards of the same enzyme in a more complicated assay setup. This assay differs from the pNPP assay because standards, control, and UFC samples must be diluted several millionfold, as opposed to several thousandfold, making enzyme dilution a more challenging process. Therefore, the Echo 550 is needed to precisely perform two consecutive dilutions. The tested phytase UFC is estimated to have an activity ranging from 70,000 to 80,000 FTU/mL based on the same Malachite Green enzyme activity assay performed with gravimetric enzyme dilution and the same formulated-standard as calibrator.
The test plate was first prepared using a combination of the Echo liquid handler and the Liquidator 96 manual liquid handler to set up the diluted enzymes. Again, the viscosity-adjusted delivery mode (384PP_AQ_CP mode) of Echo 550 was used, and the Echo 550 performed two subsequent dilutions. The Echo 550 performed the first dilution of formulated-standard, formulated-control, and UFC with assay buffer according to
Table 3
, into a Labcyte Echo 384-well destination plate, and backfill buffer (39 µL) was added with the Rainin Liquidator 96 manual pipetting system. Formulated-control of known activity was used here for an additional degree of confidence of the activity measurement for the UFC. Samples were diluted so that the final enzyme concentrations for the standards (0–0.025 FTU/mL), control (0.01875 FTU/mL), and UFC (0.0125 FTU/mL) were within the linear range of the enzymatic assay. The resulting plate was rapidly mixed with an Applikon RAMbio Benchtop Acoustic Shaker (Applikon Biotechnology, Delft, Netherlands) for 20 s at 5
First Enzyme Dilution for Malachite Green Endpoint Assay Using the AE Technology.
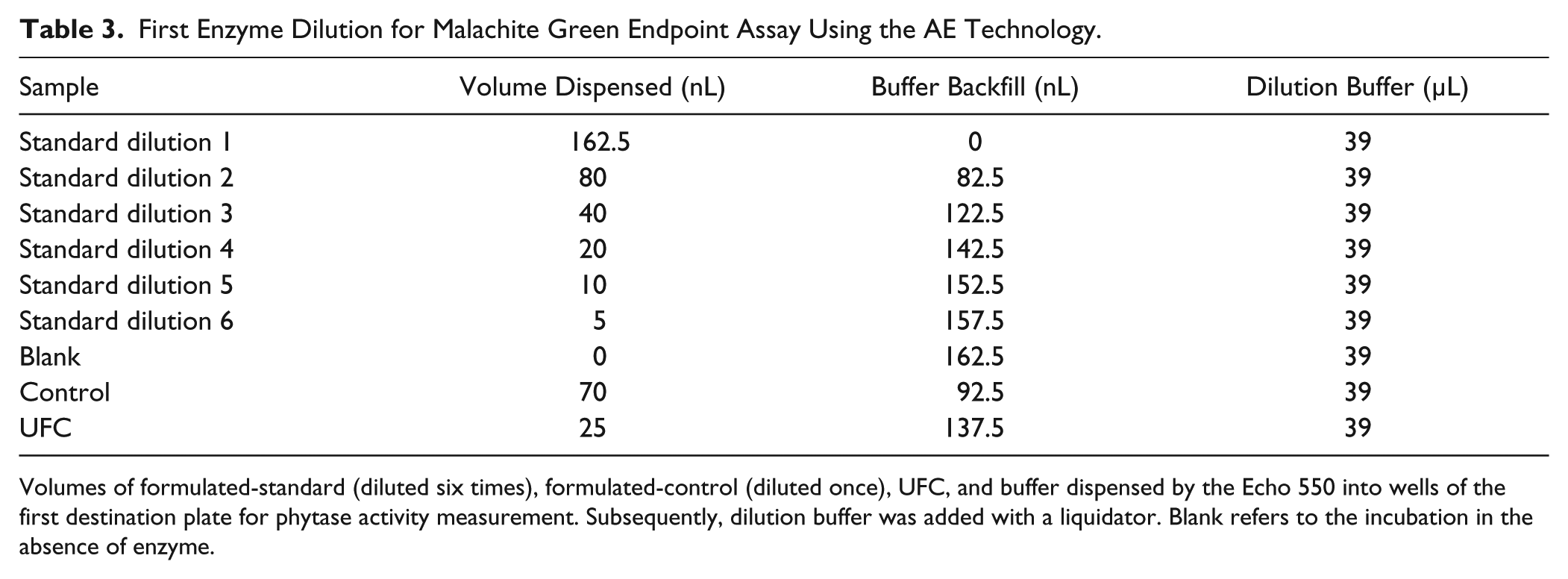
Volumes of formulated-standard (diluted six times), formulated-control (diluted once), UFC, and buffer dispensed by the Echo 550 into wells of the first destination plate for phytase activity measurement. Subsequently, dilution buffer was added with a liquidator. Blank refers to the incubation in the absence of enzyme.
For the second dilution step, 10 nL of all samples in the second Echo source plate were delivered to a new 96-well destination plate (Corning 3641). The new destination plate was then backfilled with 40 µL of assay buffer using the Rainin Liquidator 96 manual pipetting system. The second Echo destination plate was mixed using a tabletop shaker for 5 min to ensure proper mixing prior to enzymatic assay.
Enzyme samples in the 96-well plate were then directly used in the Malachite Green assay. To perform the assay, 20 µL of diluted enzyme solution was incubated with 20 µL of the phytic acid substrate (150 µM), prepared in assay buffer without added detergent, for 10 min using an iEMS incubator/shaker set at 25 °C with 900 rpm mixing. After 10 min of incubation, 85 µL of Pi Blue Phosphate Stop Solution was added to each of the wells. Following 30 min incubation at 25 °C and 300 rpm, absorbance was recorded at 620 nm using a SpectraMax Plus 384 microplate reader. Assay response for the blank reaction was subtracted from all absorbance values. Phytase activities of the UFC and formulated-control were determined using the blank-corrected assay response of the standard curve.
The precision of Echo dilution was then compared side by side with the manual dilution using calibrated Pos-D positive displacement pipettes (MR-10, MR-100, and MR-1000) and respective presterilized piston syringe tips. The procedure for the manual dilution to prepare samples for the Malachite Green assay is described in Table 4 . Phytase activities of control dilution 4 and UFC dilution 4 in Table 4 were measured.
Enzyme Dilution for Malachite Green Endpoint Assay Using Manual Positive Displacement Technique.
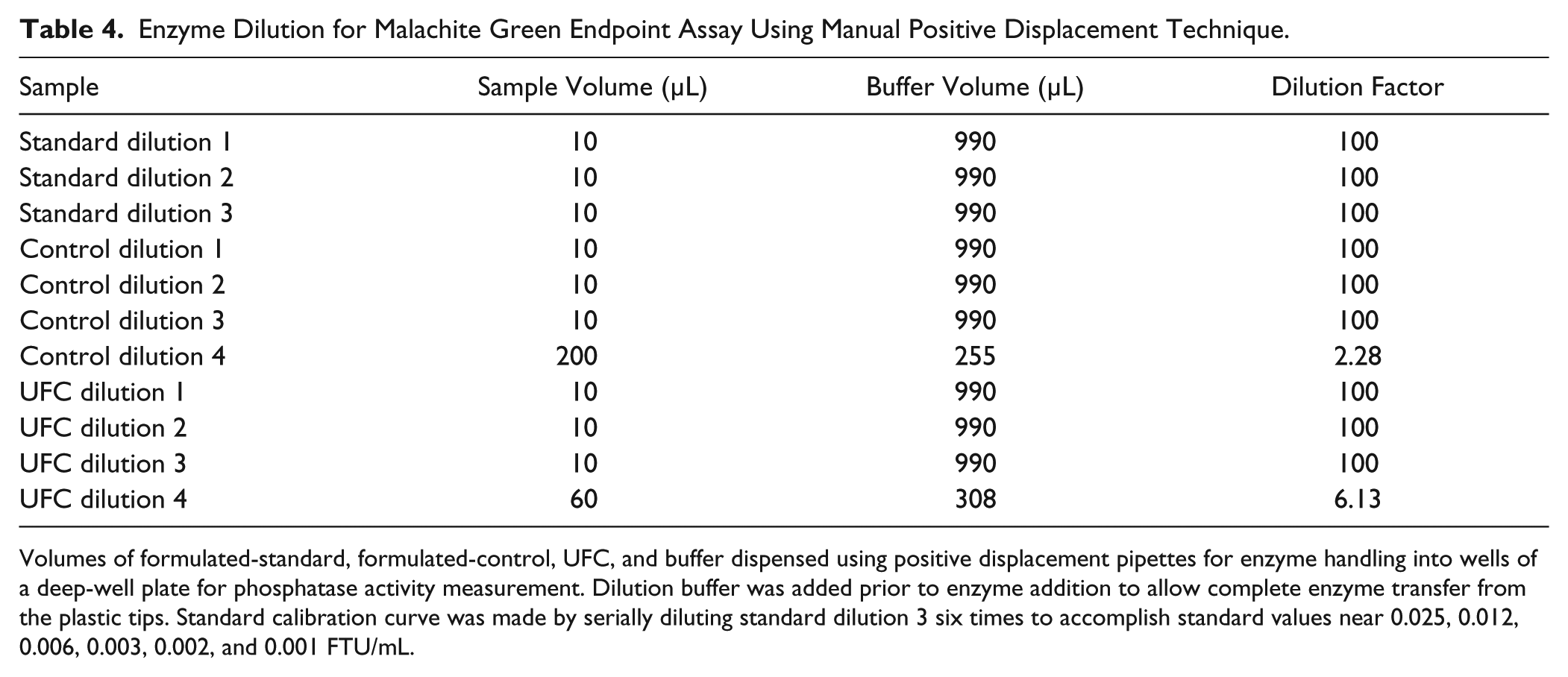
Volumes of formulated-standard, formulated-control, UFC, and buffer dispensed using positive displacement pipettes for enzyme handling into wells of a deep-well plate for phosphatase activity measurement. Dilution buffer was added prior to enzyme addition to allow complete enzyme transfer from the plastic tips. Standard calibration curve was made by serially diluting standard dilution 3 six times to accomplish standard values near 0.025, 0.012, 0.006, 0.003, 0.002, and 0.001 FTU/mL.
Fluorescent Dye Test of Delivery: Manual versus ADE Dilution
In an effort to understand some of the limitations of the ADE technology compared to the manual positive displacement technique, several tests were performed to examine whether the Echo 550 delivered viscous proteins accurately and linearly across dilutions of a formulated-control phytase protein and modified-UFC using a fluorescent dye as reporter. 14 8-Aminopyrene-1,3,6-trisulfonic acid (APTS) (0.12 mM; Sigma 09341), a fluorescent dye typically used for derivatizing sugars, was added to a stock formulated-control protein (1 mL, 93.2 mg/mL) and modified-UFC (1 mL, 202.3 mg/mL), to a resulting 1:25 ratio of APTS–enzyme sample, with final protein concentrations of 89.6 and 194.5 mg/mL, respectively. Subsequently, the fluorescent formulated-control protein was serially diluted by hand using assay buffer 1.5-fold six times using calibrated Pos-D positive displacement pipettes to yield seven resulting samples of the following concentrations: 7.9, 11.8, 17.7, 26.5, 39.8, 59.7, and 89.6 mg/mL. Similarly, the fluorescent modified-UFC was serially diluted by hand to yield seven resulting samples of the following concentrations: 17.0, 25.6, 38.4, 57.6, 86.5, 129.7, and 194.5 mg/mL. Each diluted enzyme sample (60 µL) was dispensed manually in individual wells of a Labcyte 384-well source plate (P-05525). Assay buffer was added manually to a different well in the source plate for in-Echo backfilling of the destination plate.
The Echo 550 was then used to deliver 107.5 nL of each of the seven prediluted samples and 550 nL of buffer into seven different wells on a black 96-well destination plate (Costar 3915), to a resulting volume of 657.5 nL in each of the seven wells on the destination plate. This experiment is referred to as Echo 1. The seven wells containing samples were then backfilled with 104 µL of buffer, using a Rainin Liquidator 96 manual pipetting system, for a final volume of 104.658 µL. The fluorescence intensity of the resulting plate (excitation wavelength at 420 nm and emission at 500 nm) after 1 min centrifugation at 3000 rpm was recorded on a SpectraMax M2 fluorescence plate reader (Molecular Devices). Final formulated-control concentrations were 0.008, 0.013, 0.019, 0.028, 0.043, 0.064, and 0.096 mg/mL, and final modified-UFC concentrations were 0.018, 0.027, 0.041, 0.062, 0.092, 0.139, and 0.208 mg/mL.
The fluorescence intensities of formulated-control and modified-UFC sample that were serial diluted to similar final concentrations using solely positive displacement pipettes were also determined for accuracy comparison between manual dilution and ADE technology. In this case, final formulated-control concentrations were 0.011, 0.017, 0.025, 0.038, 0.057, 0.085, and 0.128 mg/mL, and final modified-UFC concentrations were 0.006, 0.012, 0.025, 0.049, 0.099, and 0.198 mg/mL.
A change in sample dispensing method for modified-UFC was carried out to understand if the observed nonlinear relationship between fluorescence intensities as a function of protein concentration was due to error associated with the large dispensed volume (107 nL) in the previous experimental setup (Echo 1). This new experiment is referred to as Echo 2. In this case, 60 µL of fluorescent modified-UFC at a concentration of 194.5 mg/mL was transferred manually to a Labcyte 384-well source plate well. Assay buffer was added manually to a different well in the source plate for in-Echo backfilling of the destination plate. The Echo dispensed 5, 10, 20, 40, 60, 80, and 100 nL of fluorescent modified-UFC into seven different wells of a black 96-well destination plate (Costar 3915), followed by 652.5, 647.5, 637.5, 617.5, 597.5, 577.5, and 557.5 nL of assay buffer, respectively, which resulted in a volume of 657.5 nL in each of the seven wells on the destination plate. The seven wells containing samples were then backfilled with 104 µL of buffer, using the same Rainin Liquidator 96 manual pipetting system as before, for a final volume of 104.658 µL. The fluorescence intensity of the resulting plate (excitation wavelength at 420 nm and emission at 500 nm) after 1 min centrifugation at 3000 rpm was recorded on a SpectraMax M2 fluorescence plate reader (Molecular Devices). Final modified-UFC–control concentrations were 0.010, 0.019, 0.039, 0.077, 0.116, 0.155, and 0.193 mg/mL.
Results and Discussion
Phytase Activity Using pNPP Endpoint Assay
The synthetic substrate pNPP is commonly used to quantify phosphatase activity 15 and was chosen as test substrate for phytase because it typically correlates with the production of inorganic phosphate from phytic acid. Enzyme activities are reported in FTU/mL. One FTU is defined as the amount of enzyme that releases 1 µmol of inorganic phosphate from sodium phytate per minute at pH 5.5 and 37 °C. The tested UFC was estimated to have an activity ranging from 75,000 to 85,000 FTU/mL based on the pNPP endpoint activity assay performed with gravimetric enzyme dilution and the same formulated-standard as calibrator.
Dilution of concentrated formulated enzyme samples using the Echo liquid handler is reproducible and similar to the conventional method of gravimetric dilution. This is evident when superimposing the pNPP endpoint assay standard curves made either by gravimetric dilution of the formulated enzyme standards (represented by equation
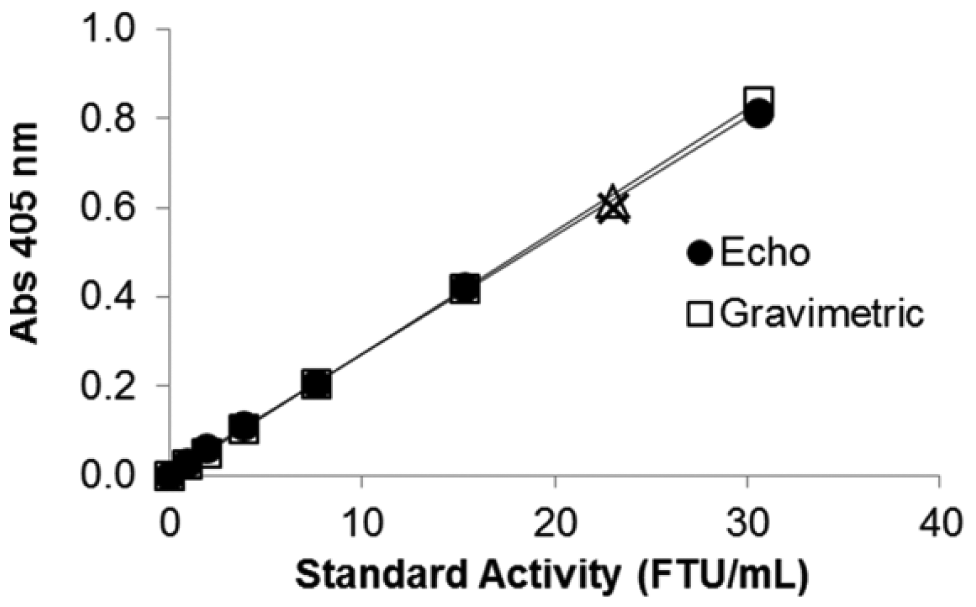
Comparison of pNPP endpoint assay standard curves made either by gravimetric or Echo dilution: absorbance at 405 nm as a function of formulated-standard concentration in terms of enzyme activity that was diluted six times either gravimetrically (□) or using the Echo 550 (•) handler. Each data point is the average of triplicate measurements. Linear standard curve made gravimetrically is represented by equation
Six independent tests performed in an effort to determine phytase activity of the UFC sample by diluting the enzyme stock with Echo 550 gave an average phytase UFC activity of 79,000 ± 1000 FTU/mL. The phytase activities for the UFC samples obtained for each test using a standard curve made by diluting samples with the Echo handler were as follows: 80,691, 79,167, 78,211, 78,699, 77,276, and 78,150 FTU/mL. Overall, the low standard deviation and CV of 1.5%, detected indirectly through the pNPP endpoint assay, suggest that the Echo liquid handler consistently (<5% CV) and accurately delivers the ultraconcentrated enzyme in the initial dilution, with consistent results across different trials.
In an effort to understand the limitations of the ADE technology, other relevant sample types used in biotechnology were studied and subjected to similar analysis. Fresh regular-UFC material recovered using conventional industrial methodology, modified-UFC material that was recovered with a proprietary modified industrial process, and purified enzyme at a concentration that is one-third of both UFC samples were diluted together with the formulated-standard and formulated-control using either the gravimetric, manual positive displacement, or ADE technology represented by the Echo 550 handler ( Fig. 2 ). Phytase activities of regular-UFC, modified-UFC, and purified enzyme were determined using the pNPP endpoint assay with the formulated-standard as calibrator and formulated-control as reference. While all three dilution methods used reported similar phytase activities for regular-UFC and purified enzyme, the reported phytase activitiy for modified-UFC was different depending on the dilution method ( Fig. 2 ).
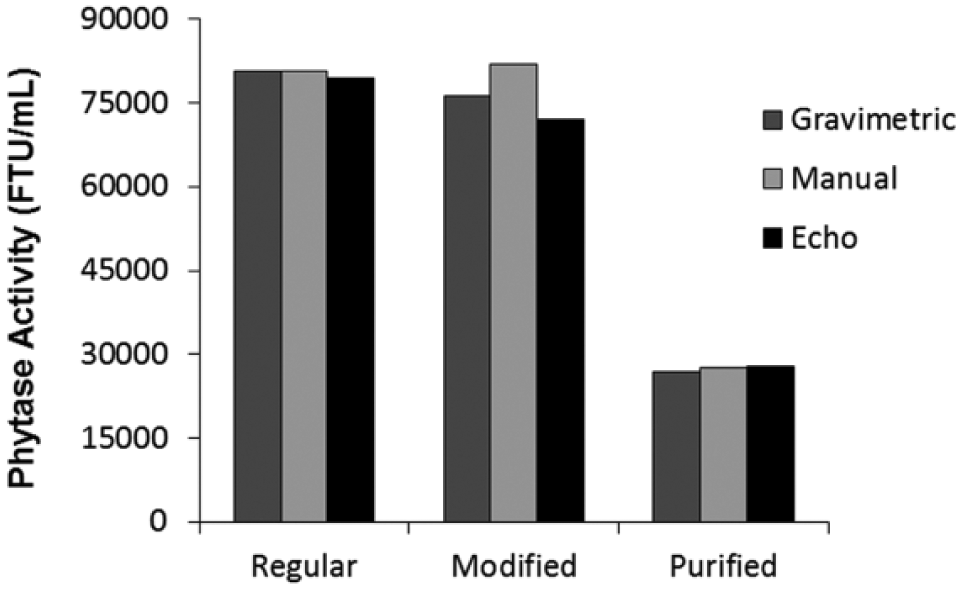
Comparison of phytase activity of different types of industrial concentrated enzyme solutions that were diluted either gravimetrically, manually, or using the Echo: phytase activities of regular-UFC (Regular), modified-UFC (Modified), and purified enzyme (Purified) were determined using the pNPP endpoint assay with formulated-standard as a calibrator and formulated-control as a reference. Samples, standard, and control were diluted either gravimetrically, manually using positive displacement pipettes (Manual), or using the Echo 550 handler (Echo). Each data point is the average of triplicate measurements.
Three independent tests performed to determine phytase activity of regular-UFC and purified enzyme by diluting each sample stock with Echo 550, gravimetrically or manually, gave an average phytase activity for regular-UFC of 80,320 ± 740 FTU/mL (0.93% CV), and purified enzyme activity of 27,400 ± 570 FTU/mL (2.07% CV) ( Fig. 2 ). All three dilution methods were consistent on the reported phytase activity. On the other hand, diluting modified-UFC using the three techniques resulted in a large variation in the enzyme activity (77,000 ± 5000 FTU/mL) with a CV of 6.54% ( Fig. 2 ). It is evident that this sample type reports a wider range of phytase activity values depending on the sample dilution method used. Interestingly, in this case, the ADE technology seems to be more aligned with the gravimetric dilution method, which is considered more accurate and precise.
Precision of the manual and ADE technology was also studied by measuring phytase activity of modified-UFC over a period of three consecutive days, to examine the potential for ADE technology in long-term stability studies taking place over weeks and months. Having a dilution method such as ADE that can provide increased throughput in formulation stability studies is highly desired, but having a method that provides increased precision as well is very important, as formulation stability studies are typically long experiments taking place on a timescale of weeks to months. Phytase activity of a freshly made modified-UFC was determined using the pNPP endpoint assay with the formulated-standard as a calibrator and formulated-control as a reference. Sample, standard, and control were diluted either manually using positive displacement pipettes or using the Echo 550 handler. While reported phytase activity for modified-UFC during three consecutive days was different depending on the dilution method used, activity values were precise for each method based on the calculated %CV. Average phytase activity for modified-UFC was 89,000 ± 2000 FTU/mL (2.45% CV), determined by diluting the sample manually, and 72,700 ± 700 FTU/mL (0.92% CV), determined by diluting the sample using the ADE technology ( Fig. 3 ). Phytase activity of modified-UFC determined by diluting the sample gravimetrically, and using formulated-standard and formulated-control, resulted in 89,000 FTU/mL (dash line in Fig. 3 ). The low %CV suggests that the activity of the enzyme stays fairly constant over the 3 days of our test. In applying this technique to stability studies, major changes can therefore be attributed to decreased stability and activity of the enzyme, rather than experimental error. Interestingly, in this case, phytase activity determined by the gravimetric dilution method is the same as that estimated using the manual dilution and not the ADE dilution method.
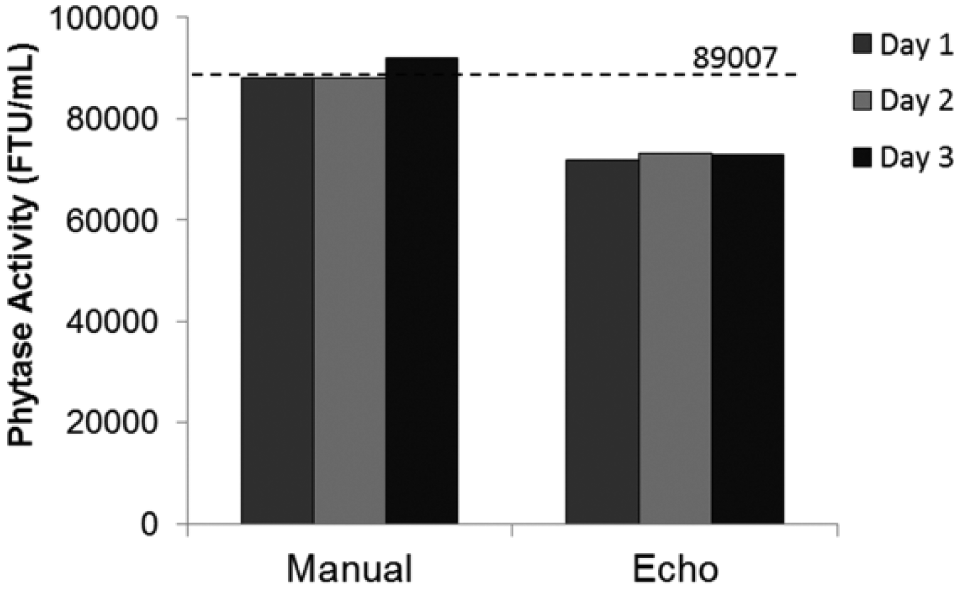
Day-to-Day reproducibility of phytase activity for modified-UFC diluted either gravimetrically, manually, or using the Echo: phytase activity of modified-UFC determined using the pNPP endpoint assay with formulated-standard as a calibrator and formulated-control as a reference. Sample, standard, and control were diluted either gravimetrically (dash line at 89,007 FTU/mL), manually using positive displacement pipettes (Manual), or using the Echo 550 handler (Echo) on three consecutive days. Each data point is the average of triplicate measurements.
Although so far we have shown that the ADE technology provides high precision for industrial enzyme activity measurement, several challenges of incorporating the ADE technology in high-throughput formulation stability studies become evident. Among the challenges of applying the ADE technology in formulation stability studies are (1) the wide variety of excipients tested in a single study and (2) the wide range of enzyme concentrations that need to be dispensed into one assay plate. Using excipients with different viscosities as well as various concentrated enzyme solutions in one assay plate might promote differences in the impedance of the acoustic wave, delivering droplets of various sizes when at extreme conditions.
To probe the lower-than-expected activity of the modified-UFC observed upon diluting the sample with ADE technology compared to the manual gravimetric dilution, sorbitol was added to the freshly made modified-UFC to a final concentration (15%) similar to that present in the formulated-standard that was used to assign phytase activity to match the excipient composition between modified-UFC and formulated-standard. Enzyme activity after addition of sorbitol or buffer was determined by the pNPP endpoint assay using formulated-standard as a calibrator and formulated-control as a reference ( Fig. 4 ). Sample, formulated-standard, and formulated-control were diluted either manually using positive displacement pipettes or using the Echo 550 handler. Average phytase activity for modified-UFC determined by the manual dilution method was similar in the presence and absence of sorbitol (91,661 FTU/mL), while phytase activities determined using the Echo dilution method were 72,876 (20% lower than that determined by the manual dilution method) and 84,869 FTU/mL (7% lower than that determined by the manual dilution method) in the absence and presence of sorbitol, respectively ( Fig. 4 ). Although reported phytase activities for modified-UFC in the absence and presence of sorbitol were different depending on the dilution method used, the activity value for the sample diluted using the Echo in the presence of sorbitol (15% final concentration) was higher and closer to the expected phytase activity value reported based on the positive displacement dilution.
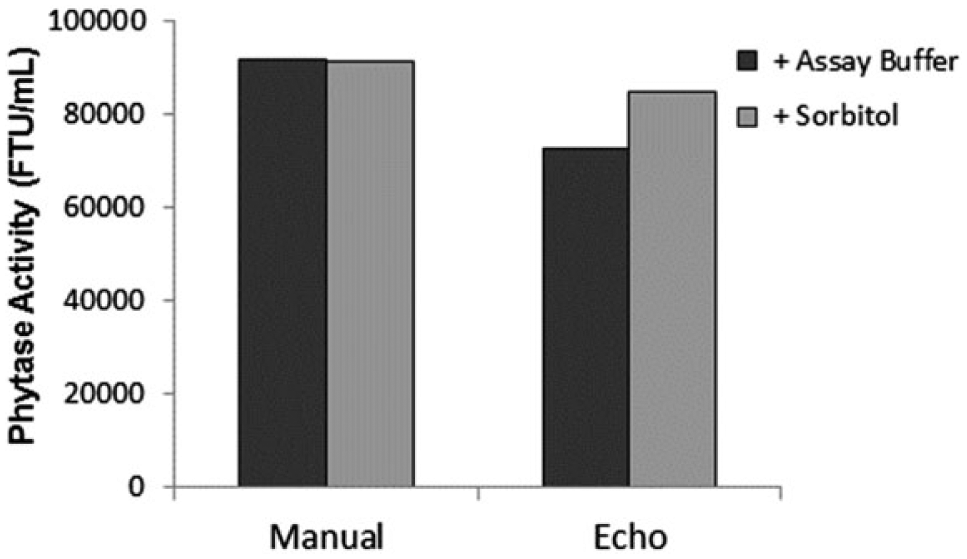
Effect of adding sorbitol on the reported phytase activity of modified-UFC diluted either manually or using the Echo: phytase activity of modified-UFC was determined in the presence of 15% sorbitol (final concentration) or assay buffer and using the pNPP endpoint assay with the formulated-standard as a calibrator and formulated-control as a reference. Sample, standard, and control were diluted either manually using positive displacement pipettes (Manual) or using the Echo 550 handler (Echo). Each data point is the average of triplicate measurements.
Addition of sorbitol to modified-UFC at a concentration similar to that present in the formulated-standard increased the estimated phytase activity, making it closer to the activity value determined by performing the same enzyme assay using the manual positive displacement method. This result suggests that matching the viscosity properties of modified-UFC to those of formulated-standard and formulated-control improves, to some extent, the accuracy of the activity measurement for modified-UFC. Even though the viscosity properties of modified-UFC were similar to those of the formulated-standard after addition of sorbitol, reported enzyme activity values were still lower than those determined using the manual positive displacement dilution method. Thus, we could speculate that there is an additional factor in modified-UFC that prevents the ADE technology from dispensing consistent droplets. Although the Echo liquid handler is calibrated to dispense up to 50% glycerol using the 384PP_AQ_CP mode, the intrinsic viscosity of protein solutions might not behave the same way as glycerol, imposing a protein-dependent dispense challenge. It can be speculated that different proteins will dispense differently based on their individual intrinsic viscosity properties when at high concentrations.
To further understand the lower-than-expected phytase activity of modified-UFC when diluting the sample with the ADE technology compared to the manual gravimetric dilution, the effect of protein concentration on the reported enzyme activity was assessed. Protein concentrations in modified-UFC, formulated-standard, and formulated-control are 202.3, 56.2, and 93.2 mg/mL, respectively. These protein concentration values are somewhat high and typical in samples from industrial enzymes and protein crystallography studies. In an effort to identify the impact of dispensing extreme protein concentrations with the ADE technology, phytase activities of purified enzyme at medium (63 mg/mL) and high (306 mg/mL) protein concentrations were determined using the pNPP assay with the formulated-standard as calibrator and formulated-control as reference. Sample, standard, and control were diluted either manually using positive displacement pipettes or using the Echo 550 handler ( Fig. 5 ). The reported phytase activity of 63 mg/mL of purified enzyme was similar between dilution methods (27,756 FTU/mL) and in accordance to the specific activity of phytase (440 FTU/mg). At high enzyme concentration (306 mg/mL), the activity value for the sample diluted using the Echo (120,941 FTU/mL) was lower than expected based on the manual dilution (134,508 FTU/mL) and the specific activity of the phytase. These results suggest that the viscosity of superconcentrated phytase (306 mg/mL), compared to that of phytase at 63 mg/mL, is much higher, and therefore it prevents delivery of equivalent drop sizes by the ADE technology across a wide range of protein concentrations.
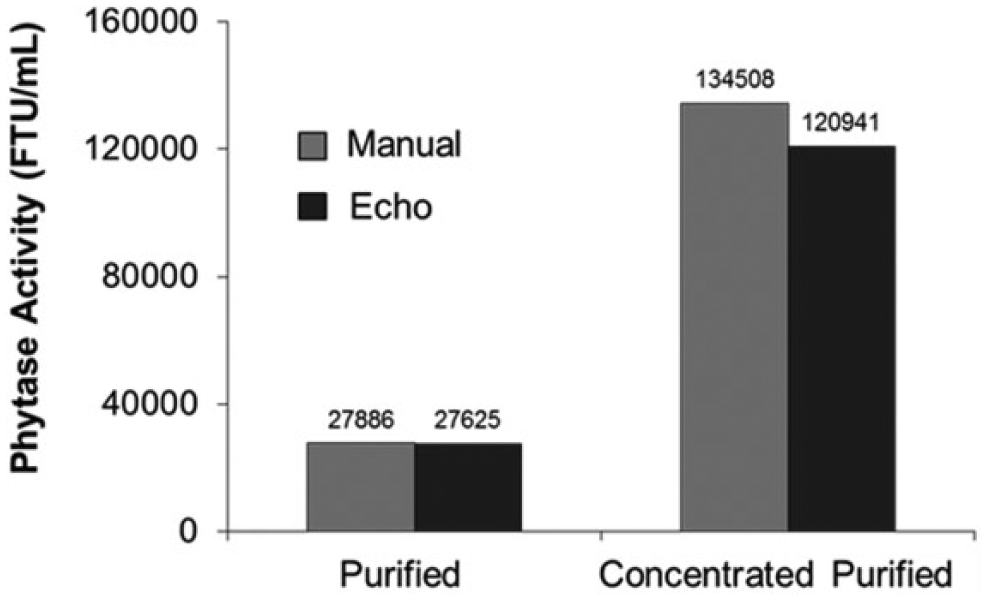
High protein concentration impedes proper sample ejection by the Echo as reported by the phytase activity of purified enzyme: phytase activity of purified enzyme (Purified, 63 mg/mL) or concentrated purified enzyme (Concentrated Purified, 306 mg/mL) determined using the pNPP endpoint assay with formulated-standard as a calibrator and formulated-control as a reference. Sample, standard, and control were diluted either manually using positive displacement pipettes (Manual) or using the Echo 550 handler (Echo). Each data point is the average of triplicate measurements.
Phytase Activity Using Malachite Green Endpoint Assay
The dilution of concentrated formulated enzyme samples using the Echo liquid handler is reproducible. This is evident when superimposing the Malachite Green endpoint assay standard curves made by diluting the formulated enzyme standards using the Echo liquid handler on two different days ( Fig. 6B ) compared to standard curves made by diluting the formulated enzyme standards manually using the positive displacement method ( Fig. 6A ). It is apparent that the signal for the standard curve and control sample is higher when samples are diluted with the manual positive displacement method than when they are diluted with the same dilution made by the Echo. At this point, it is unknown why the Abs 620 signal for the samples diluted manually is higher than that for the same samples dispensed by the Echo, but it could be speculated that (1) viscous sample in the exterior of the plastic tips is carried out throughout the different dilution steps, or (2) the Echo, although precise, might be consistently underdelivering these sample types. An advantage of the ADE system over manual dilutions is that liquid transfer is done without tips, ensuring a contaminant-free and carryover-free transfer of concentrated and viscous solutions. Moreover, utilizing the ADE technology allows the user to save reagents and reduce waste by evading intermediate dilution steps necessary for conventional dilution methods. In the case of the Malachite Green endpoint assay for measuring phytase activity, the ADE technology requires 5–163 nL of enzyme sample and up to 79.16 µL buffer for every unique sample dilution, while conventional dilution requires 10 µL enzyme sample and up to 3300 µL of buffer per unique sample dilution.
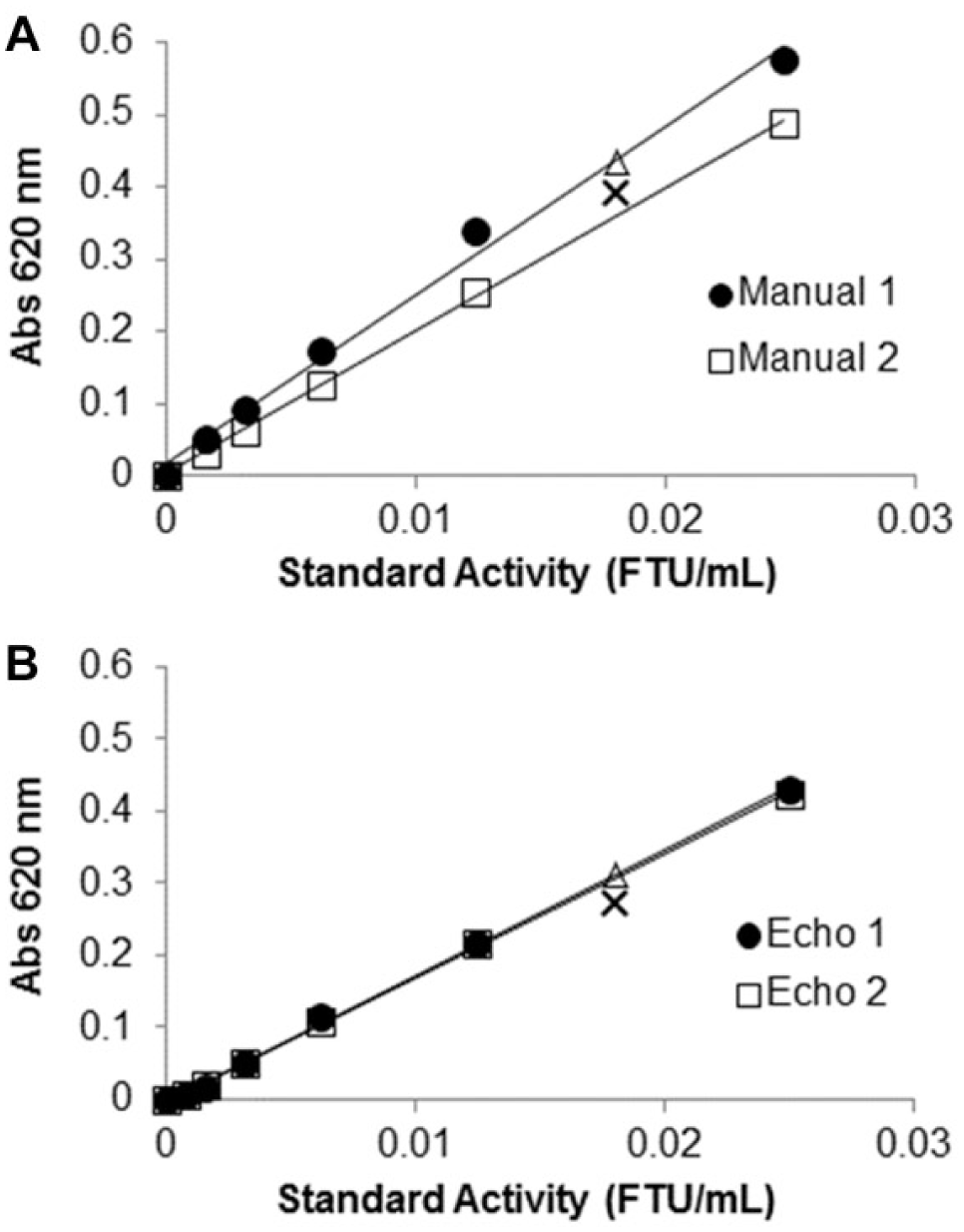
Reproducibility of Malachite Green endpoint assay standard curves made with Echo dilution: absorbance at 620 nm as a function of formulated-standard concentration in terms of enzyme activity diluted (
Three independent tests, all including triplicate measurements, were performed in an effort to determine the phytase activity of the UFC sample by diluting the enzyme stock with Echo 550. The following UFC activities were obtained using a standard curve made by diluting samples with the Echo handler: 72,275, 71,165, and 72,347 FTU/mL. This produced an average activity of 72,000 ± 700. Overall, the low standard deviation and CV of 0.92%, detected indirectly through the Malachite Green endpoint assay, again suggest that the Echo liquid handler consistently delivers the ultraconcentrated enzyme in the initial dilution. The lower value for activity (72,000 FTU/mL) of UFC in the Malachite Green assay compared to that reported by the pNPP endpoint assay (79,000 FTU/mL) is likely due to lower phosphatase activity (which may exist as an impurity in the phytase UFC) with the natural substrate compared to a synthetic substrate. Because the Malachite Green assay is frequently used to assess phytase activity using the natural substrate and requires a millionfold dilution of the enzyme prior to performing the activity assay, the successful delivery and dilution of phytase UFC by the Echo 550 suggests many possibilities for its use in the future, in particular for assessing enzyme activity with high precision during long-term protein stability studies.
Fluorescent Dye Test of Delivery: Manual versus ADE Dilution
Several tests were performed to examine whether the Echo 550 delivered proteins accurately and linearly across dilutions of a formulated-control phytase protein and modified-UFC with a wide range of protein concentrations, using the fluorescent dye APTS as reporter for quantifying volume transfer. Samples used in this study are typical in industrial applications and similar in protein concentration to those used in protein crystallography. In an effort to understand the effect of protein concentration in sample dilution by the ADE technology, similar fold dilutions were performed manually with positive displacement pipettes for the same samples for comparison.
The resulting fluorescence intensities (λex = 420 nm, λem = 500 nm) of the formulated-control sample diluted using the ADE technology represented by Echo 550 (
Fig. 7A
) were as follows (from lowest to highest protein concentration): 5.13, 7.27, 10.23, 15.39, 23.63, 36.10, and 54.52 FU. The graph of fluorescence intensity as a function of Echo-diluted formulated-control concentration is represented by equation
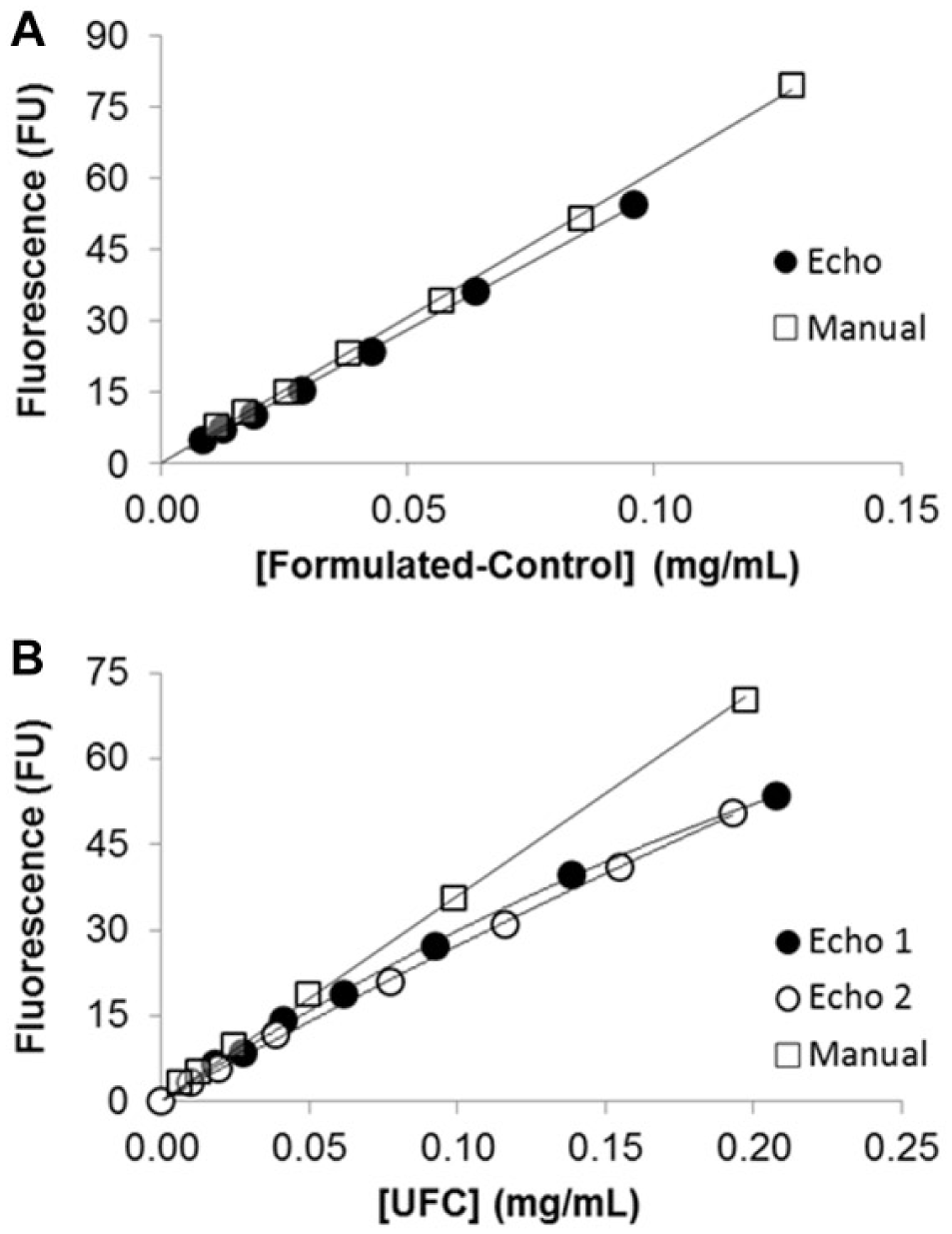
Protein delivery test by Echo 550 using APTS as fluorescence reporter: relationship between APTS fluorescences (λex = 420 nm, λem = 500 nm) as a function of final concentration of formulated-control (
When testing the delivery of a more challenging sample (modified-UFC, 107.5 nL) of the same phytase enzyme containing 2.2-fold higher protein concentration than that present in the formulated-control sample (194.5 mg/mL vs 89.6 mg/mL), it was evident that protein concentrations of more than ~20 mg/mL seem to limit the ejection of droplets with the desired volume (
Fig. 7B
, Echo 1). The resulting fluorescence intensities (λex = 420 nm, λem = 500 nm) of the modified-UFC sample diluted by mixing 107.5 nL with 550 nL of assay buffer using the Echo handler were as follows (from lowest to highest protein concentration): 6.69, 8.60, 14.23, 18.79, 27.25, 39.63, and 53.44 FU. The graph of fluorescence intensity as a function of Echo-diluted modified-UFC concentration is represented by the equation
The Echo dispensing method for modified-UFC was altered to be able to understand if the nonlinear relationship between fluorescence intensities as a function of protein concentration of prediluted modified-UFC is due to error associated with the large volume dispensed (107 nL) in the previous experimental setup (Echo 1). In the new experimental setup (Echo 2), the Echo dispensed 5, 10, 20, 40, 60, 80, and 100 nL of fluorescent modified-UFC (194.5 mg/mL) into seven different wells on a black 96-well destination plate, followed by addition of assay buffer for a total volume of 657.5 nL, and then sample was diluted further to the same extent as in the previous experiment, Echo 1, to be able to compare the fluorescence intensities of manual and Echo dilution. Use of the Echo dilution method, where different volumes of modified-UFC were dispensed into the read plate, resulted in a response in fluorescence intensity ( Fig. 7B , Echo 2) that was very similar to that observed in Figure 7 , Echo 1. This result suggests that the nonlinear response of fluorescence intensity when diluting modified-UFC with the ADE technology is most likely due to the increased impedance of the acoustic energy when transferring extremely high protein concentrations. Thus, the Echo liquid handler appears to transfer smaller-than-expected volumes of protein at high protein concentrations, compared to when at lower concentrations. Although droplets dispensed by the Echo in experiment 2 (Echo 2) originated from the same concentrated fluorescent modified-UFC, the error for transferring multiple droplets (2 drops for dispensing 5 nL and 40 drops for dispensing 100 nL) magnified the inaccuracy, as the error is compounded after each droplet delivery.
Understanding the limitations of the Echo in terms of reduced accuracy is essential when delivering such high protein concentrations relevant in enzyme development for industrial enzymes or protein crystallographic studies. Although accuracy is reduced when delivering samples with extreme protein concentrations by the ADE technology (Echo reports lower fluorescence values than the manual dilution), the ADE technology offers high precision (<5% CV) while dispensing these extremely challenging samples.
Conclusion
Acoustic liquid handling technology represented by the Labcyte Echo 550 liquid handler is extremely efficient and precise (<5% CV) in dispensing ultraconcentrated, viscous, and formulated protein solutions. It successfully overcomes many problems previously encountered in delivering highly concentrated and formulated industrial enzymes with conventional liquid handlers. In addition, because it delivers only nanoliter quantities of enzymes, cost and enzyme waste are decreased in the long term. Acoustic droplet ejection allows for more reproducible enzyme activity measurements in formulation stability studies, compared to the manual positive displacement method, necessary for enzyme formulation development. ADE also allows miniaturization of enzymatic assays for formulated enzyme solutions in an effort to explore a wider formulation space for a particular industrial enzyme in a high-throughput manner. For any laboratory that requires high-quality and high-throughput enzyme activity assessments, incorporating ADE technology (as was used in the formulation studies reported here) in place of gravimetric/volumetric pipetting techniques is no longer an option, but an essential evolution. Without the ADE technology, high-throughput formulation stability studies for formulated concentrated enzyme solutions or similar sample types will be more challenging.
Footnotes
Acknowledgements
The authors would like to thank Roopa Ghirnikar and Douglas Dale for reviewing this manuscript, Allison Gauthier and Grant Ganshaw for providing UFC and purified phytase, respectively, and DuPont Industrial Biosciences for the undergraduate summer internship opportunity (JDH) and support throughout this project. Special gratitude goes to Mike Travis and Howard Lee from Labcyte for answering many questions about the ADE technology.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
