Abstract
Automated cell culture incubators generally are considered primary components of fully automated cell culture systems, which are able to monitor cell growth without human interaction. This tutorial is focused on automated cell culture incubators. It emphasizes the impact of automation on throughput and environmental controls (temperature, humidity, and CO2) and proposes some basic protocols to check these functions. In addition, it details practical aspects for switching from manual to automated cell culture incubators.
Introduction
Cell culture has been a basic concept in most laboratories for many years. It is used for many purposes, including toxicity studies, monoclonal antibody production, human viral vaccine production, artificial tissue engineering, and cell and gene therapy. Traditionally, cell culture has been a manual process that is subdivided into many repetitive steps, each requiring highly skilled laboratory personnel to perform meticulous and often tedious manipulations on a daily basis, including weekends. These tasks require stringent environmental constraints and pose a high risk of contamination. It is imperative that all batches are produced under the same conditions to achieve reproducible experimental results.
The main equipment used for cell culture is the cell culture incubator. An incubator enables reproduction of ideal conditions for cell development. Many classic incubators have been on the market for years. They were developed for the manual handling of cell cultures and include sensors to check culture conditions. The evolution of drug-discovery methods toward high-throughput assays, the increasing cost of skilled laboratory personnel, and the need for standardization have driven the cell culture process toward automation. Automated cell culture incubators were developed, allowing automatic handling and management of cell cultures while decreasing interference by human users. Automated incubators are often integrated into a completely automated production line that may include transportation robots and other modules. 1
An automated approach to cell culture requires an understanding of new technologies, methods, and criteria in order for laboratories to successfully evaluate, qualify, purchase, and install automated incubators. This tutorial offers background and insight for users and manufacturers who need to describe specifications and controls for their equipment.
Cell Culture Automation
It is well known that mammalian cell cultures rely not only on media, but also on environmental factors, which must be considered when selecting an incubator. The first key factor to consider is the cell line to be produced. Different cell lines require different culture conditions. For most human cells, protocols have been published using a temperature of 37 °C, relative humidity (
Another major factor for consideration is vessel type. Plates, flasks, bottles, and dishes are all available for cell culture applications. It is important that a robotic incubator be compatible with the vessel type with which a laboratory is working, or an adaptation of the protocol will need to be agreed upon and validated. When vessels are closed or capped, fluctuations inside the culture medium are less than those inside the incubator, thus reducing possible negative effects during loading and unloading.
Before an automated incubator can be properly evaluated, it is necessary to understand its essential purpose, the cell culture vessels being used, and the incubator parameters that are microprocessor-controlled. A clear understanding of the equipment allows users to perform a thorough critical evaluation. Following is a list of features that can be possessed by an automated incubator:
Traditional manually operated glass external doors.
Traditional systems for environmental control, including an input for CO2, a water tank for humidity, a thermal regulation device for temperature control, and an audio and/or visual warning system, among others.
A transport device such as a rotating carousel or Cartesian robot in the storage module.
The inclusion of several berths for microplate storage stackers.
Whether or not stackers are designed to accommodate different types of vessels.
A plate “shuttle” system, such as a robotic arm and tray, that transfers vessels from outside, through an automatic door, and into a stacker (loading); or from a stacker, through an automatic door to the outside (unloading).
A microprocessor-controlled automatic door that opens and closes quickly during loading and unloading.
From this list of features, it is possible to define evaluation parameters.
Evaluation of an Automated Cell Culture Incubator
Loading and Unloading Throughput
Loading and unloading throughputs can be defined as the mean times to load or unload a vessel. When investigating the latter, two different time measurements can be per-formed—the delay between the command and the closing of the gate (minimum time to deliver a vessel) and the delay between the command and the return of the robotic arm to its standby position (minimum time between two deliveries).
Through this last value, it is possible to calculate the unloading throughput per hour. Random loading or unloading of an incubator should be measured with the incubator filled to a capacity of not less than 50%. Results are issued as the mean time ± standard deviation (
Environmental Control
The accuracy, spatial homogeneity, and stability of environmental conditions are crucial for high-quality cell cultures. The main parameters involved are temperature, CO2 level, and humidity.
There are two steps in studying this parameter: (1) temperature stability without opening a manual door or automated gate and (2) the eventual impact on temperature of the loading and unloading process.
Thermal stability without door opening: Inside a traditional cell incubator, cartography studies are performed using temperature sensors. In an automated incubator, the mechanical and electrical parts of the robot occupy the internal space. Because these parts may disturb thermal regulation, temperature sensors must be located strategically in response to the heating system's design (hot spots) and to the door or automated gate (cold spots). Knowledge of these hot and cold spots helps in understanding condensation phenomena. Figure 1 shows a schematic recording of temperature obtained from three different sensors located in different places within a cell culture incubator. From this record, the mean temperature and its standard deviation can be calculated for each sensor location along with the offset between the mean and setpoint temperatures, to determine if the incubator performance is acceptable for planned cell culture experiments.
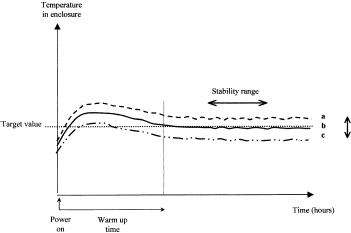
Thermal stability without door opening: a, b, c: different locations.
Thermal recovery after door opening: Door opening may cause fluctuation of the temperature inside an incubator. In theory, the automation of cell culture handling should decrease the exchanges between the external environment and the incubator. A sensor placed near the automatic gate measures the magnitude of fluctuation. Figure 2 presents a recording of such a temperature measurement and illustrates that during door opening, temperature decreases to a minimum value and then increases to reach equilibrium after a short temperature overshoot. Usually, the common recovery threshold is 95% of the previous temperature. The elapsed time between the point when the door opens and when the incubator reaches the recovery threshold temperature is called “recovery time.” With automated incubators, recovery time is often minimal because automated loading and unloading require approximately three-fold less time than manual loading and unloading and results in less air exchange. Coupled with the minimized effects attributed to closed vessels, as mentioned previously, the impact of external environmental fluctuation becomes negligible.
Thermal recovery after door opening: a, initial temperature; b, minimal temperature; c, return to the temperature setpoint; d, overheat.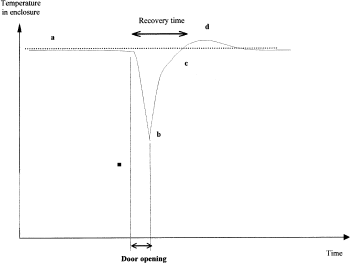
Method: When evaluating an incubator, two assessments must be performed: one at the incubator's highest throughput to determine the impact at the highest frequency of automated gate opening, and one that simulates the typical throughput anticipated once the incubator is integrated in a complete cell culture system.
In cell culture incubators, the level of relative humidity (RH) is often very high, around 95%. This humidity is created by evaporation from the water tank when the heating system is activated. Relative humidity is homogeneous inside the incubator.
Just as with temperature, two steps are required for studying this parameter: (1) basic performance without opening a manual door or automated gate and (2) the eventual impact of loading and unloading on the stability of humidity.
Humidity stability without door opening: Because relative humidity is homogeneous inside an incubator, only one sensor needs to be placed in the center of the storage area. To achieve equilibrium, it is necessary to wait a minimum of 24 h after the heating system is started.
Humidity recovery after door opening: Door opening may cause fluctuation of the humidity inside the incubator. The amplitude of the fluctuation can be measured by placing a sensor inside one stacker and then testing different opening frequencies. Figure 3 shows a schematic recording of such RH measurements. After each gate opening, the temperature decreases slightly, depending on the frequency, and then increases to return to equilibrium.
Recovery time of relative humidity (RH %) according to frequency opening: a, low; b, medium; c, high frequency of door opening.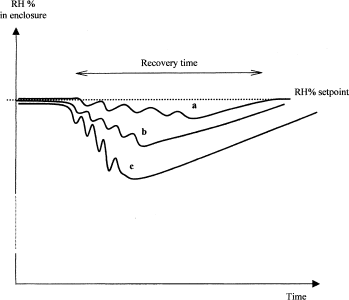
Method: When evaluating an incubator, three assays may be performed: one at the incubator's highest throughput level to determine the highest frequency of automated gate opening, one that simulates the typical throughput anticipated once the incubator is integrated into a complete cell culture system, and one at a low throughput level.
The high relative humidity inside the incubator creates a condensation risk that may contaminate cell cultures. To locate condensation spots, the maximum anticipated humidity can be achieved by filling the incubator to capacity with plates filled with water and then observing the eventual condensation phenomena. Condensation may appear as a mist, stationary droplets, or streaming drops. Precise temperature measurements should be performed during these observations to explain the condensation phenomena.
CO2 levels are often established at 5% and regulated by a CO2 injection device that is activated to counterbalance any loss. As with temperature and RH, there are two steps in studying this parameter: (1) basic performance without opening a manual door or automated gate and (2) the eventual impact of loading and unloading on the stability of CO2.
CO2 stability without door opening: Because CO2 pressure is homogeneous inside an incubator, only one sensor is required in the center of the storage area. To achieve equilibrium, it is necessary to wait a minimum of 12 h after the system starts. CO2 pressure can then be recorded for a 2-h period. After the first 2 h, the mean CO2% and the amplitude of the fluctuation (standard deviation/mean) can be calculated.
CO2 recovery after door opening: Because diffusion of CO2 through the gate to the outside happens quickly, door opening causes significant CO2% fluctuations inside the incubator. The amplitude of these fluctuations can be measured by placing a sensor inside one stacker. Figure 4 shows a schematic recording of such CO2 measurements. After a gate opening, the CO2 decreases rapidly, and then successive injections (small peaks on the graph) compensate for the loss of CO2 to return to equilibrium. The curve illustrates that the lowest levels of CO2 occur after gate openings. The number of injections required to achieve desired levels is a measure of recovery time.
CO2 recovery after door opening.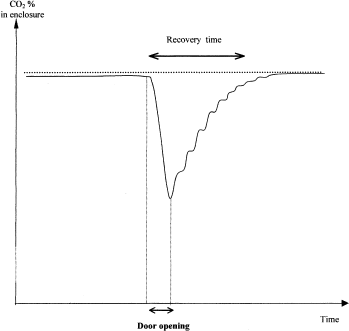
Practicability
User manual: In addition to standard instructions concerning the setup and daily use of an incubator, clear drawings, or better yet, a CD-ROM with animated illustrations showing how to handle the mechanical parts of the incubator, is necessary.
Noise: Noise level should be less than 85 decibels.
Traceability—Culture environment: Parameters should be continuously recorded, and any drifts inside the incubator should trigger an alarm.
Plate position: the location of cell cultures contained in the incubator should be accessible to the user and recorded.
Maintenance: Unlike a manual cell culture incubator, automated cell culture incubators often require the disassembly of many mechanical parts to facilitate proper cleaning. This process sometimes requires two people. Compatible cleaning agents must be determined by the various materials being used—stainless steel, plastics, etc. Special attention must be given to any rough surfaces, condensation spots, the water tank, and any hidden places where contaminants could grow.5, 6 Decontamination must be performed in an autoclave. For parts that are impossible to disassemble or for materials that cannot endure the heating process, decontamination can be performed with volatile products such as glutaraldehyde or hydrogen peroxide. Finally, because of the high level of relative humidity, watertight joints need to be checked frequently and replaced when necessary. In addition, contents drained from the water tank should be considered a biohazard and regarded as dangerous liquid waste.
Alarms: To ensure good working order, alarms must be checked regularly. This can be done by purposely causing a failure such as interrupting an incubator's CO2 input. These purposeful failures also can be used to help train laboratory personnel.
Integration into a completely automated cell culture line: The cell culture process includes cell growth, harvesting, reseeding, and analysis. Fully automated systems provide different modules in globally controlled environments. Controls include atmospheric control, evaluation of pH, nutrient and waste concentration, and an information system that is able to manage operations, especially vessel handling. Each module should be able to function independently to protect cell cultures from spoilage in case of a hardware or software failure.
Conclusion
Increased throughput demands and standardization requirements are driving cell culture toward automation. Because this change requires that mechanical and electrical components function in warm and humid closed areas, the growth of contaminants poses a threat and requires new knowledge for using and maintaining automated cell incubators. Before purchasing equipment, users should carefully evaluate their options and verify specifications to ensure the equipment they purchase is qualified to meet their cell culture needs. When critical details are well defined, the advantages of installing and using automated incubators instead of manual incubators become clear. In turn, quality documents are easier to write, ensuring the level of performance of an automated cell culture incubator in routine use, especially when integrated into a fully automated cell culture system with a single information system.
