Abstract
Routine peptide structure-activity relationship screening requires the serial dilution of peptides to produce full concentration-response curves. Established tip-based protocols involve multiple tip changes and high exposure to plasticware. In the case of peptides, this becomes a challenge, since peptides can adsorb to plastic, resulting in an observed loss of potency. Various methods can be employed to prevent peptide loss during compound handling, such as the inclusion of bovine serum albumin or solvents in assay buffer and the siliconization of plasticware, yet protein binding remains unpredictable. The degree of variation by which peptides will adhere to plasticware can confuse results and cause inaccuracies in potency predictions. We evaluated acoustic noncontact methods for peptide serial dilution and compared it with traditional tip-based methods, on the effect on potency curves for glucagon-like peptide-1 and glucagon peptide analogues. The current study demonstrates the benefits of noncontact dispensing for high-density microplate assay preparation of peptides using nanoliter droplets across our entire drug discovery workflow, from in vitro high-throughput screening to drug exposure determinations from in vivo samples.
Introduction
Incretin receptors are a key therapeutic target in the treatment of metabolic disease. 1 In particular, the glucagon-like peptide-1 (GLP-1) receptor represents an ideal target to model an effective antidiabetic drug due to the well-established antihyperglycemic effects of its peptide hormone ligand, GLP-1. 2 In contrast to GLP-1, glucagon plays a hyperglycemic role as the main insulin-counteracting hormone but has raised interest as a potential antidiabetic and antiobesity agent due to its anorexigenic properties.3,4 On binding to their specific G protein–coupled receptors, both GLP-1 and glucagon elicit their effects through the downstream activation of adenylyl cyclase via Gαs and the production of the second messenger cAMP. 5 Thus, the measurement of accumulated cAMP within cells is a functional readout of receptor activation and can be used to screen for active GLP-1 or glucagon-based peptide analogues.
Throughout the lead identification and lead optimization phases of drug discovery, routine half-maximal effective concentration (EC50) determinations performed for peptide structure-activity relationship (SAR) screening involve the serial dilution of samples. This presents a particular problem for peptides, which can stick to plasticware to varying degrees, leading to inconsistency in potency calculations. While various methods can be employed to prevent peptide loss, such as the inclusion of bovine serum albumin (BSA) in buffers and the use of siliconized plasticware, protein binding is unpredictable, and peptides such as GLP-1 vary greatly to the degree by which they adsorb to the different plastics they come into contact with. 6 Historically, we have used tip-based liquid handling robots for the serial dilution of peptides in a Hank’s buffered saline solution (HBSS) buffer supplemented with BSA. This process involves multiple tip changes to transfer peptide samples within polypropylene microtiter plates. Using this method, significant rightward shifts in the EC50 reported for a given peptide under different dilution conditions became apparent, suggesting peptide loss. We observed potency loss over orders of magnitude, which could not be completely prevented by the inclusion of increased BSA concentrations. As a further complication, potential therapeutic peptides have variable plasma protein binding characteristics; thus, elevated assay buffer albumin concentrations are undesirable. Activity could be preserved following serial dilution in DMSO, suggesting DMSO prevents peptides sticking to plasticware; nevertheless, the use of DMSO is undesirable for large molecule biologics, because it interferes with protein integrity and stability.
Acoustic liquid handling has been described to improve SAR screening for small molecules that share the same problem of adhering to plastic. 7 Here, we evaluated the efficiency of liquid handling with Acoustic Droplet Ejection (ADE) with the Labcyte (Sunnyvale, CA) ECHO 550 for peptide serial dilution. Noncontact acoustic dispensing was used for the preparation of peptide samples in functional cell-based assays in both transfected recombinant receptor expressing cell lines and endogenous receptor primary cell lines. Using ADE with Labcyte ECHO demonstrated precise peptide transfer, accurate dilution, and elimination of activity loss resulting from peptides sticking to plasticware.
Materials and Methods
Cell Culture
A stable Chinese hamster ovary (CHO) cell line expressing the rat GLP-1 receptor was a kind gift from AstraZeneca (Mölndal, Sweden). Clonal INS1E cells were isolated from the parental INS1 based on both their insulin content and their secretory responses to glucose 8 and were a kind gift from Prof. Claes B. Wollheim (University of Geneva Medical Center, Geneva, Switzerland).
CHO-rat GLP-1 and INS1E cells were cultured to 80% confluence, cryopreserved in cell freezing medium (Sigma Aldrich, St. Louis, MO), and stored in liquid nitrogen at 1e 7 cells/mL. Cryopreserved rat (Sprague-Dawley) hepatocytes were purchased from Life Technologies (Carlsbad, CA).
Peptide Solubilization
GLP-1 and glucagon were purchased from Bachem (Bubendorf, Switzerland). All other peptides were produced in house at MedImmune. Peptides were reconstituted to 1 mg/mL in DMSO and stored in siliconized Eppendorfs.
cAMP Homogeneous Time-Resolved Fluorescence Accumulation Assay
Cell-based cAMP accumulation assays were used to screen peptides for agonist activity against rat GLP-1 receptors expressed in CHO cells and endogenously expressed in the INS1E rat β-cell line. In addition, peptides were screened for activity against the glucagon receptor in primary rat hepatocytes. Peptide serial dilutions were prepared in DMSO or assay buffer (HEPES-buffered saline solution; 25 mM HEPES, 0.5 mM IBMX, 0.1% BSA, pH 7.4) using a tip-based (Agilent Bravo, Santa Clara, CA, USA) or an acoustic noncontact dispenser (Labcyte ECHO 550) to obtain an 11-point dose-response curve. When DMSO was used as a diluent, the final DMSO concentration that cells were exposed to across both systems was equivalent at 1%.
For the tip-based system, peptides were serially diluted in DMSO or assay buffer in low protein binding 384-well plates (Greiner, Gloucestershire, UK). All sample dilutions were made in duplicate. Then, 5-µL serially diluted peptides were transferred in duplicate to black shallow-well u-bottom 384-well assay plates (Corning, New York, NY). For the acoustic system, peptides were serially diluted in DMSO or assay buffer by the transfer of nanoliter volumes from 384-well source microplates (Labcyte) into assay plates (Corning) prefilled with 5 µL assay buffer. Backfill of DMSO or assay buffer was applied to keep the total transfer volume of 100 nL constant across the 11-point range.
Frozen cryo-vials of cells were thawed rapidly in a water bath, transferred to prewarmed assay buffer, and centrifuged at 240 ×
Data Analysis
Data were transformed to % Delta F as described in the manufacturer’s guidelines and expressed as % activation, where 80 nM GLP-1 or 100 nM glucagon defines maximum effect. The transformed data were analyzed by four-parameter logistic fit to determine EC50 values using GraphPad Prism 6 (GraphPad Software, La Jolla, CA). Correlation plots were generated by fitting mean pEC50 data ± SD using TIBCO Spotfire (TIBCO, Boston, MA), with straight-fit analysis of least squares.
Pharmacokinetic Analysis
Pharmacokinetic (PK) peptide exposure analysis was performed by testing serum samples in a bioassay using the GLP-1R cAMP accumulation assay described above. Estimated concentration of active peptide remaining in rat serum was calculated by comparison of test serum samples with a reference peptide at a known concentration. Typical rat PK study design included three animals per test sample dosed 1 mg/kg intravenously (IV), and 30 µL serum was sampled across five time points from 0 to 2 days.
Results and Discussion
SAR Workflow
Routine SAR can be imagined as a workflow cascade progressing from initial in vitro primary screens in recombinant cell lines, through physiologically relevant cells lines that endogenously express the receptor of choice, to in vivo PK studies that establish peptide in vivo stability ( Fig. 1 ). Typically, the workflow is used as a selection funnel, and as assays become more complex and more expensive, the throughput decreases. Accurate peptide potency determinations are required at each stage of this workflow to understand peptide SAR and to demonstrate translation from high-throughput assays to more physiological readouts.
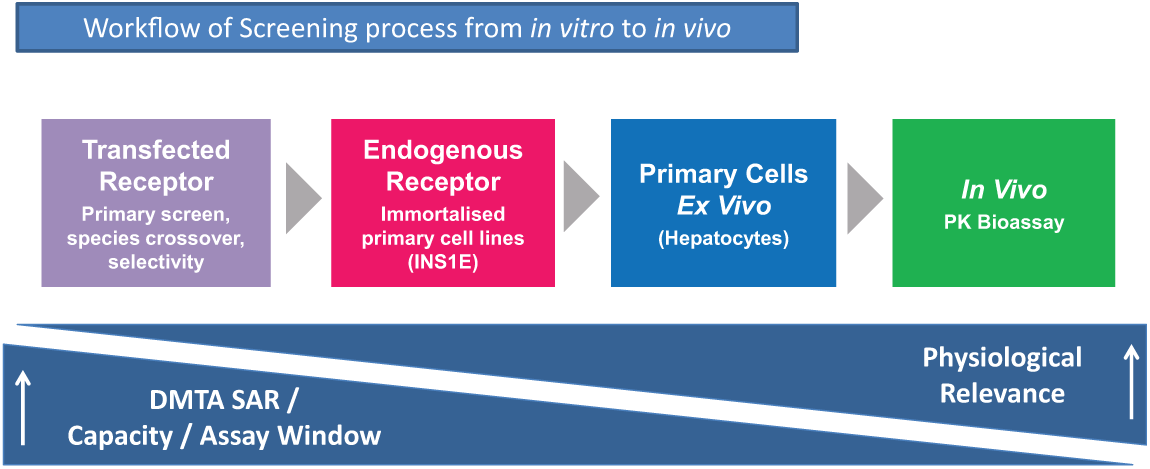
Workflow of screening process from in vitro to in vivo. DMTA SAR, Design Make Test Analyse structure-activity relationship; PK, pharmacokinetic
Peptide Potency Loss against the rGLP-1R
A cAMP accumulation homogeneous time-resolved fluorescence (HTRF) assay was used to determine the potency of peptide agonists against recombinantly expressed rat GLP-1R. Native GLP-1, the reference peptide for this assay, serially diluted in assay buffer with the tip-based system was 8.8-fold less potent compared with equivalent serial dilution using DMSO ( Fig. 2A ). This loss of activity indicates a loss of peptide, most likely due to adsorption to plasticware. Peptide activity was preserved in DMSO, suggesting DMSO prevents peptide binding to plastic. This difference in potency was not observed when serial dilutions were performed using noncontact dispensing ( Fig. 2B ). We tested a range of peptides with activity at the rat GLP-1R and saw a large variation in potencies when diluting in DMSO or assay buffer with the tip-based system, indicating varying degrees of adsorption to plasticware between peptides ( Fig. 2C ). Furthermore, EC50 data generated using noncontact dispensing for serial dilution in assay buffer overlaid DMSO data for the tip-based system, implying no peptide loss for all peptides tested ( Fig. 2D ).
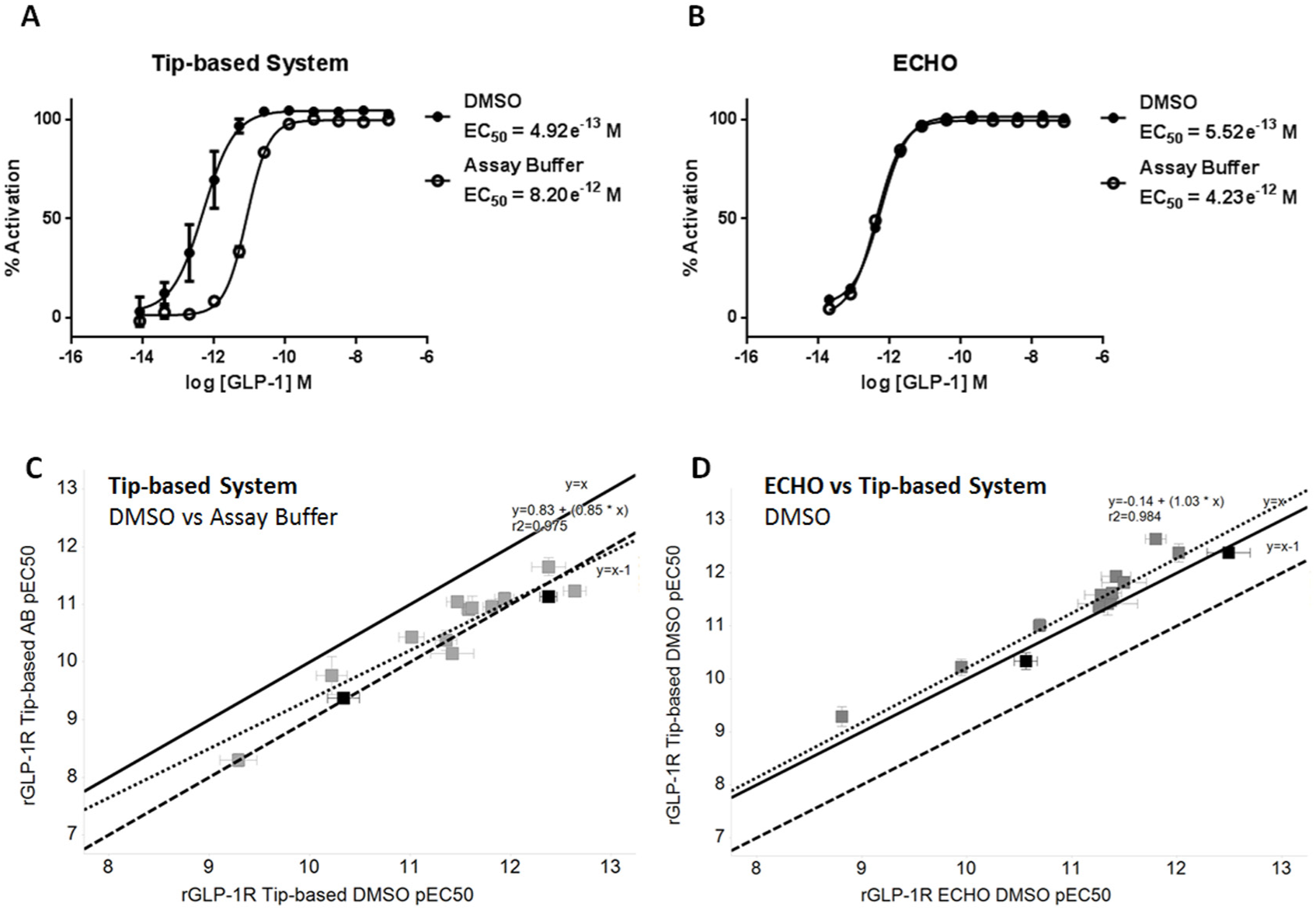
Effect of dilution method on peptide-induced cAMP response in Chinese hamster ovary (CHO) cells expressing rat glucagon-like peptide-1 receptor (GLP-1R). Mean ± SD data (representative of three independent experiments) are analyzed by four-parameter logistic fit to determine EC50. Closed circles = DMSO serial dilution, open circles = assay buffer serial dilution. Serial dilution of glucagon-like peptide-1 (GLP-1) in assay buffer using the tip-based system causes an 8-fold shift in EC50 compared with DMSO (
Cell Line DMSO Tolerance
DMSO prevented peptide binding to plasticware; however, DMSO itself can alter cell functionality9,10 and has been speculated to cause synergistic effects with GLP-1 11 and increase cAMP accumulation. 12 We therefore investigated the DMSO tolerance of our rat GLP-1R and INS1E cell lines. DMSO concentrations up to 1% are tolerated ( Fig. 3 ), and it is therefore important not to exceed this concentration during routine screening. Furthermore, this suggests that the shift in EC50 observed following serial dilution with the tip-based system ( Fig. 2 ) is not due to DMSO since concentrations are below 1% in this assay. The stability of some peptides (e.g., recombinant or PEGylated) can be jeopardized by reconstitution in DMSO; however, serial dilution using a noncontact dispenser negates the use of DMSO completely for such peptides and instead allows for the use of aqueous assay buffer without causing peptide loss. Furthermore, the ECHO will allow synthetic and recombinant peptides to be tested from same dilution plate while keeping final DMSO concentrations <1%.
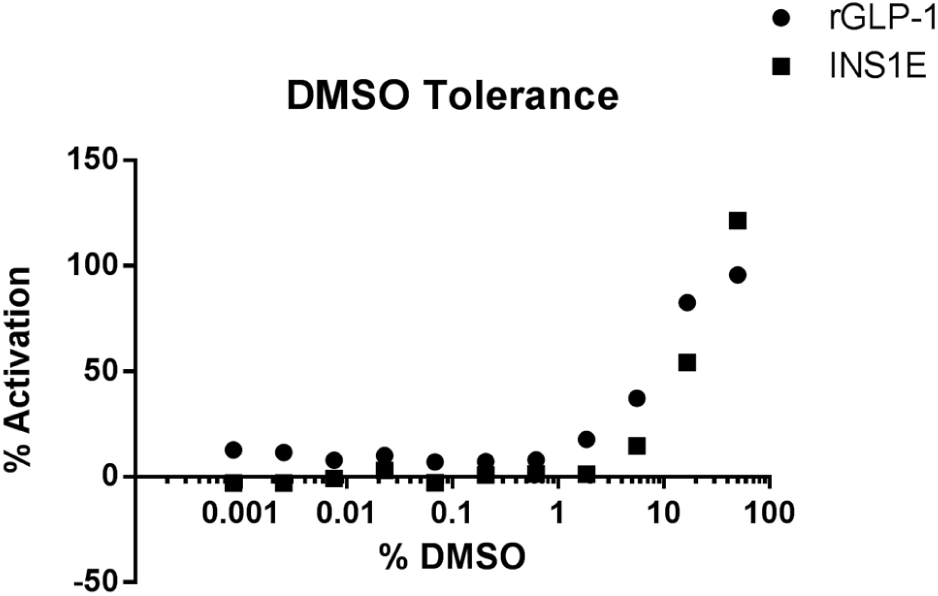
DMSO Tolerance of cAMP assay in rat glucagon-like peptide-1 receptor (GLP-1R) expressing cell lines. Effect of increasing concentrations of DMSO on Chinese hamster ovary (CHO) cells expressing recombinant rGLP-1R (closed circles) or INS1E (closed squares). Representative figure of
High Concentrations of BSA Do Not Protect from Peptide Loss
The inclusion of increasing concentrations of BSA in the assay buffer caused a leftward shift in the GLP-1 EC50 curve toward that of DMSO dilution; however, it could not fully rescue peptides from sticking to plasticware during tip-based serial dilution ( Fig. 4A ). EC50 values generated following noncontact serial dilution did not shift, regardless of the presence of BSA, confirming a reduction in peptide loss through adsorption in tip-based methodology ( Fig. 4B ). The inclusion of 10% BSA did not alter GLP-1 EC50, which is as expected since the peptide is only 20% plasma protein bound (in-house data).
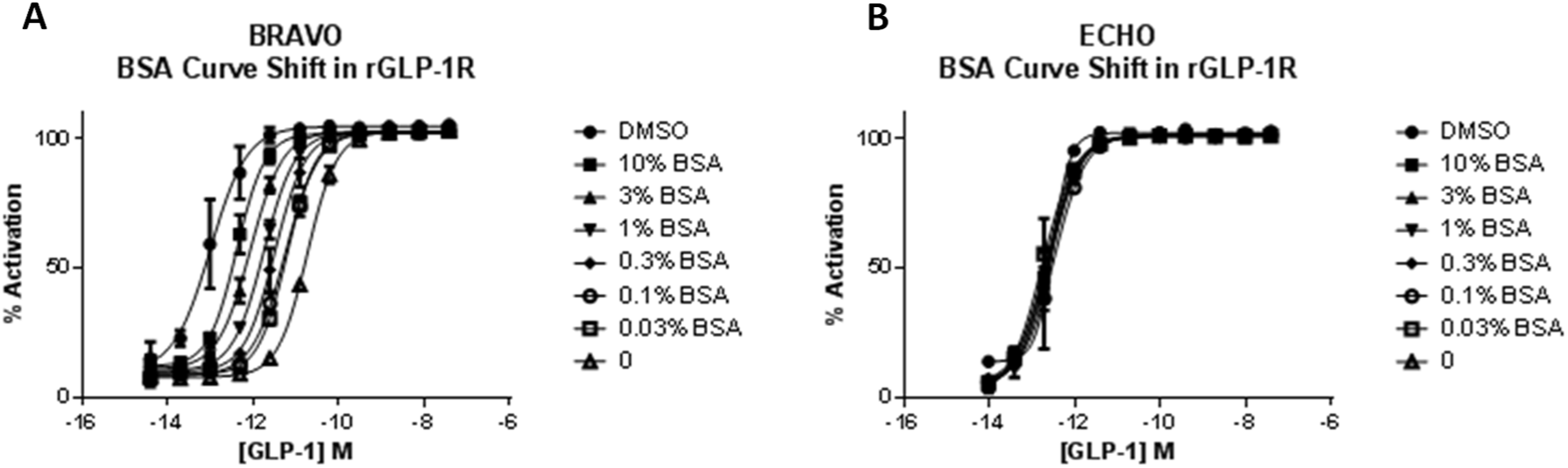
Effect of increasing bovine serum albumin (BSA) concentration on glucagon-like peptide-1 (GLP-1)–stimulated cAMP in Chinese hamster ovary (CHO)–rGLP1 cells. Mean ± SEM data (representative of two independent experiments) are fitted with four-parameter logistic fit to determine EC50. Serial dilution in assay buffer with increasing concentrations of BSA using the tip-based robotic system causes a leftward shift in EC50 (
Impact on Peptide Potency at Endogenous Receptors Is Also Affected
GLP-1 and GIP receptors have been reported to be expressed on pancreatic β-cells. 13 Rat insulinoma-derived INS1E cells constitute a stable β-cell surrogate useful for screening lead peptides against receptors at physiologically relevant levels of expression at the next step in our workflow. GLP-1 serially diluted in assay buffer with the tip-based system was 5-fold less potent compared with equivalent serial dilution using DMSO ( Fig. 5A ). This difference in potency was not observed following peptide serial dilution using the noncontact dispenser ( Fig. 5B ). As demonstrated for the rat GLP-1 receptor cell line, large variation in peptide potencies was observed when diluting in DMSO or assay buffer with the tip-based system ( Fig. 5C ). EC50 data generated using noncontact dispensing for serial dilution in assay buffer overlaid DMSO data for the tip-based system, implying no peptide loss ( Fig. 5D ).
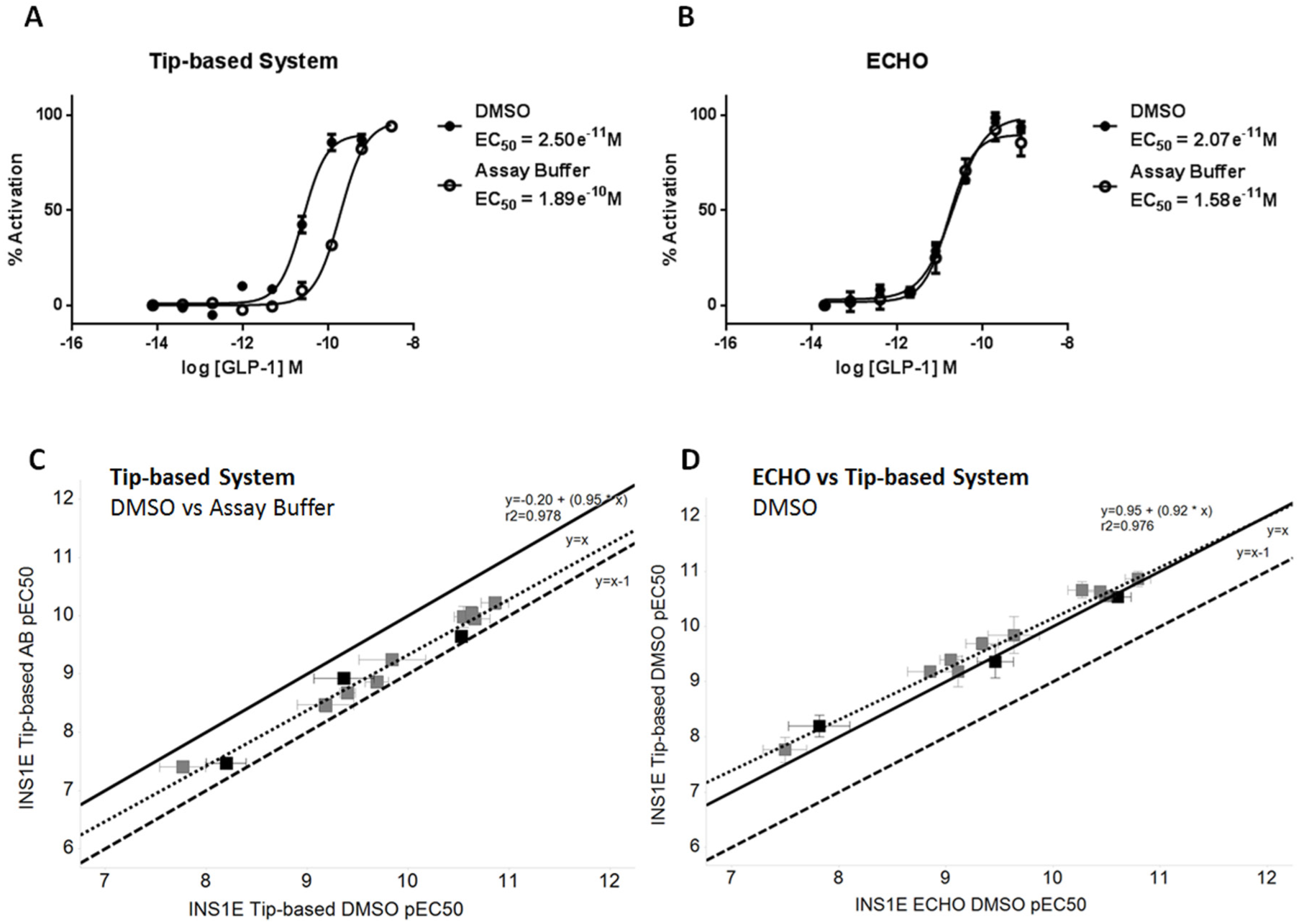
Effect of peptide dilution method on the glucagon-like peptide-1 (GLP-1)–induced cAMP response at endogenous receptors expressed in INS1E cells. Mean ± SD data (representative of three independent experiments) are fitted with four-parameter logistic fit to determine EC50. Closed circles = DMSO, open circles = assay buffer. Serial dilution of GLP-1 in assay buffer using the tip-based system causes a 5-fold shift in EC50 compared with DMSO (
Hepatocyte Primary Cell Line
Several peptides agonists of the rat GLP-1R in both transfected and INS1E cell lines were also active against the glucagon receptor. Glucagon receptors are natively expressed by hepatocytes 14 ; therefore, quantification of cAMP can be used to selectively evaluate the potency of lead peptides in primary hepatocytes. Again, peptides serially diluted in assay buffer with the tip-based system were consistently found to be less potent against primary rat hepatocytes than when serially diluted in DMSO. Glucagon serially diluted in assay buffer using the tip-based system was 26-fold less potent compared with equivalent serial dilution in DMSO ( Fig. 6 ). Glucagon serially diluted in assay buffer using the noncontact dispenser showed the same potency as tip-based serial dilution in DMSO, indicating there is no peptide loss ( Fig. 6 ). The EC50 values of synthetic peptides serially diluted in DMSO correlated closely to the EC50 values derived from peptides serially diluted by noncontact dispensing ( Table 1 ).
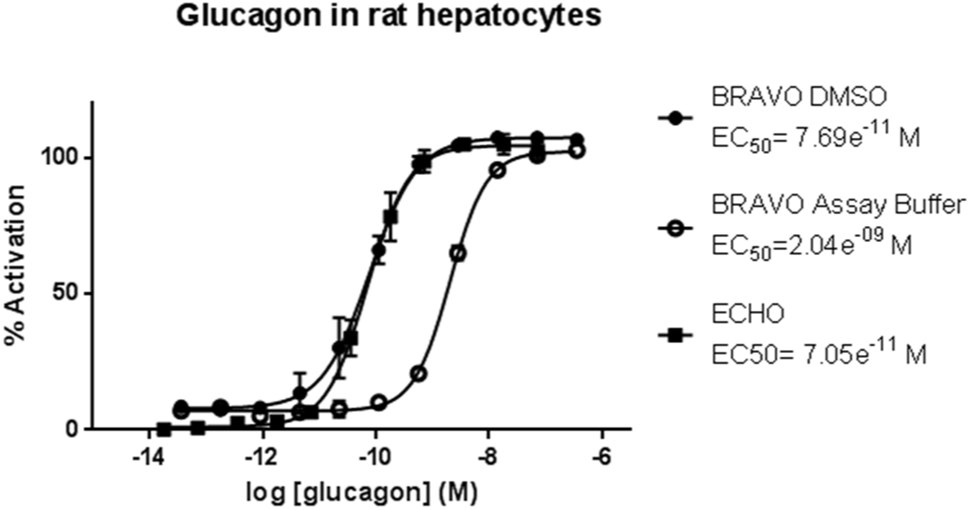
Effect of peptide dilution method on glucagon-stimulated cAMP response at endogenous receptors expressed in rat hepatocytes. Mean ± SD data (representative of four independent experiments) are fitted with four-parameter logistic fit to determine EC50. Closed circles = DMSO serial dilution, open circles = assay buffer serial dilution, closed square = assay buffer serial dilution using ECHO.
Comparison of EC50 Obtained in Rat Hepatocytes for Peptides Serially Diluted in Assay Buffer with ECHO or in DMSO with the Tip-Based Robotic System.
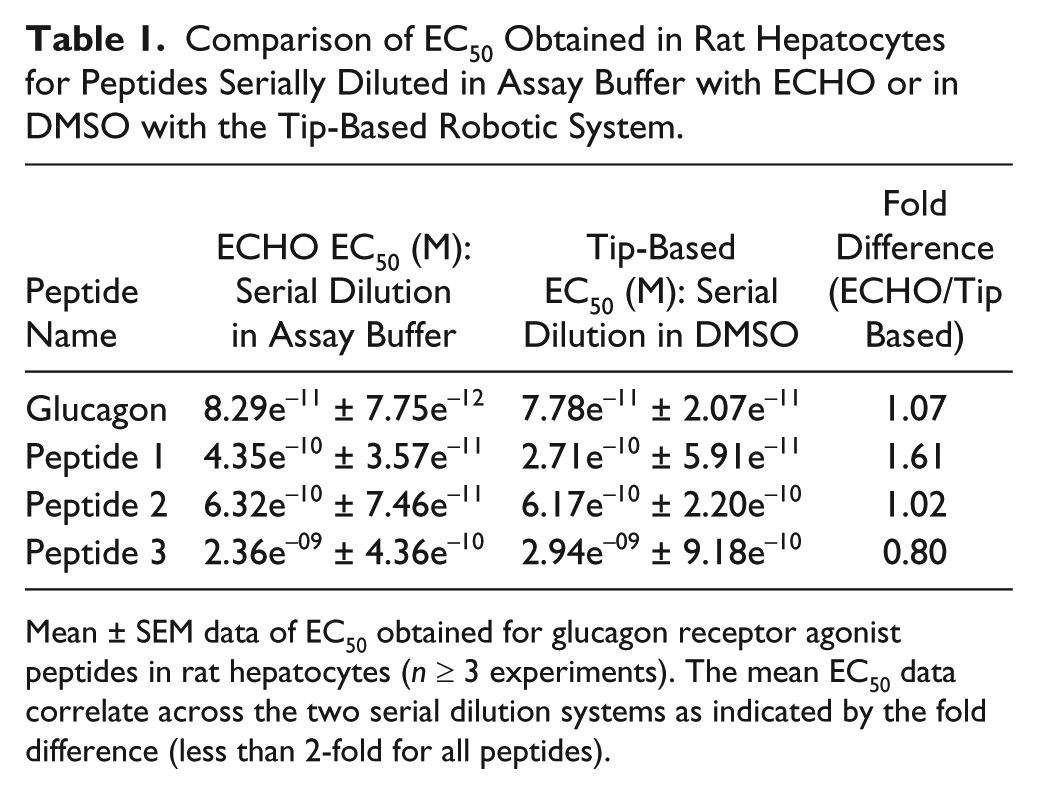
Mean ± SEM data of EC50 obtained for glucagon receptor agonist peptides in rat hepatocytes (
PK Bioassay
For the final stage of the workflow, lead peptides progress to in vivo rat PK and pharmacodynamic (PD) studies. The activity of peptides remaining in rat serum over time can be indirectly measured using the cAMP accumulation assay as a bioassay from which unknown concentrations of peptide in serum can be back-calculated from a reference curve. As an example, GLP-Fc activity can be detected in serum up to 24 h after IV administration in rats at 1 mg/kg ( Fig. 7A ). Estimated concentration data can be plotted against time to estimate apparent half-life for key peptides ( Fig. 7B ). As well as supporting the entire workflow cascade, ECHO provides high-quality data by eliminating the opportunity for peptides to adsorb to plasticware and facilitates reduced sample volumes, equating to a reduction in animal usage through reduction in required blood sampling volumes ( Table 2 ).
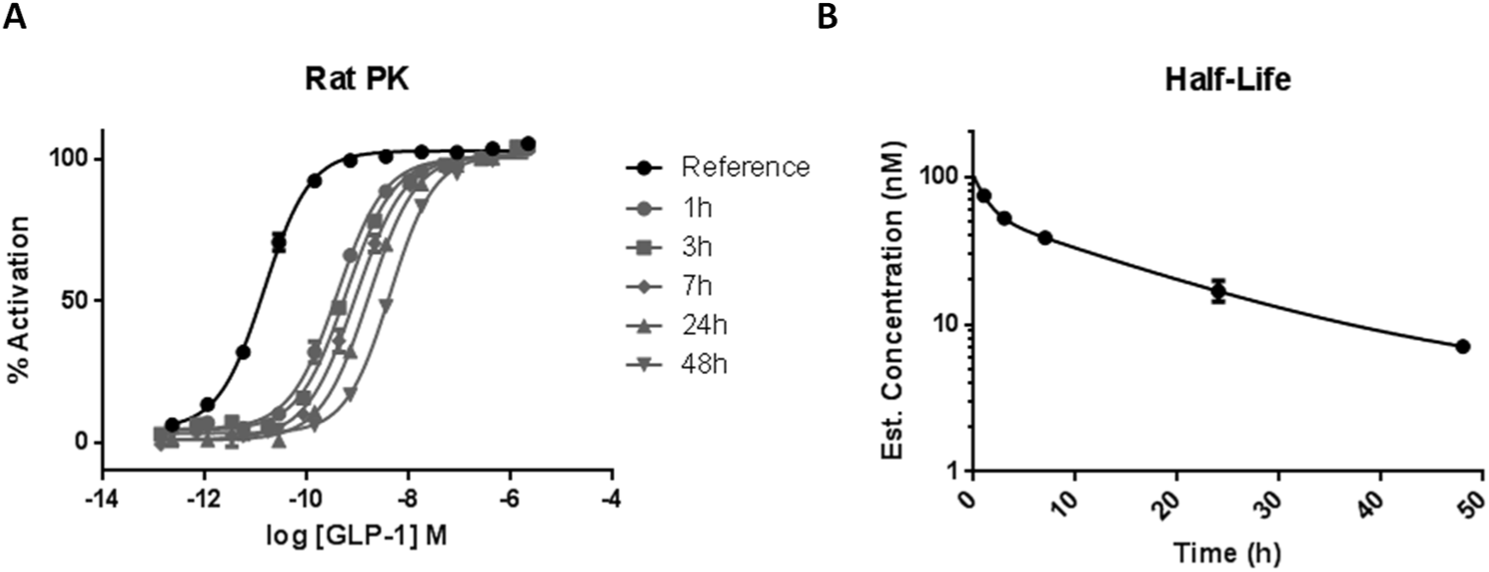
Pharmacokinetic (PK) bioassay for peptide concentration determination in rodent serum. Mean SEM data are fitted with four-parameter logistic fit to determine EC50; closed circles = glucagon-like peptide-1 (GLP-1) reference. Over time, concentration of active peptide remaining in serum decreases, seen as a rightward shift in EC50 (
Comparison of Serum Volume Requirements for Pharmacokinetic Bioassay.
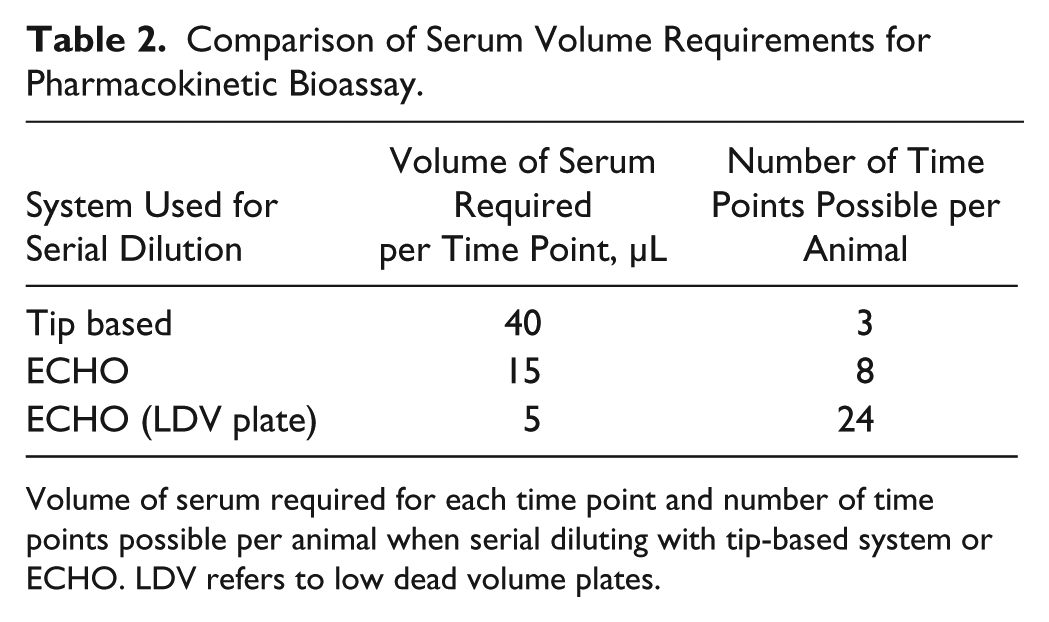
Volume of serum required for each time point and number of time points possible per animal when serial diluting with tip-based system or ECHO. LDV refers to low dead volume plates.
In conclusion, the use of ADE with Labcyte ECHO is a unique tool for serial peptide dilution and allows the prediction of accurate peptide potencies at all stages of our drug discovery Design Make Test Analyse (DMTA) SAR workflow. Tip-based methods for serial dilution change tips at every step to remove the risk of carryover; however, this exacerbates the issue of peptide adsorption to plasticware. We have demonstrated that noncontact acoustic dispensing reduces this problematic peptide loss.
DMSO is the preferred diluent for most synthetic peptides in tip-based systems and can prevent the reduced potency associated with loss of peptide. Not all peptide formats can be handled in DMSO; thus, there is a necessity to use alternative methods such as ADE for serial dilutions. DMSO itself was reported to enhance GLP-1–induced insulin secretion in INS1 cell lines. 11 Our data suggest that these results may have been confounded by a decreased amount of peptide loss via adsorption and could be avoided through the use of ADE.
The potency determinations from our cAMP assays that drive our SAR translate to physiologically relevant functional assays in vitro and in vivo. The relative potency ratios determined from the INS1E β-cell cAMP assay reported here correlate closely with in vivo efficacy readouts such as interperitoneal glucose tolerance tests (in-house data). Likewise, the relative potency ratios derived from the hepatocyte functional cAMP assays described directly correlate with data from fresh primary hepatocyte functional assays such as hepatic glucose output (in-house data). This demonstrates the physiological relevance of the data determined in the early SAR in vitro assays. Using ADE has positively affected the data quality at each stage of our early preclinical drug discovery workflow.
In this study, we have shown that ADE with Labcyte ECHO enables accurate, automated peptide dilution across our entire workflow and minimizes the risks associated with standard techniques, including peptide adsorption to plasticware. The improvement in peptide potency we observed with the ECHO was dramatic, in some cases over orders of magnitude for potent molecules. Furthermore, we demonstrate that ADE can be applied to a cell-based assay used for the analysis of plasma samples. This has potential to reduce volume of serum required per time point, increasing number of time points and data per animal and reducing the number of animals per study. ADE is successfully used across our lead identification and lead optimization workflow, from in vitro high-throughput screening to a bioassay for exposure determination from in vivo samples.
Footnotes
Acknowledgements
We thank the MedImmune ADPE peptide platform group for generating the test peptides used in this study and MedImmune BSU staff for performing the PK studies. Furthermore, we thank David Baker and Carol Moreno-Quinn for helpful comments on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
