Abstract
Drug combination testing in the pharmaceutical industry has typically been driven by late-stage opportunistic strategies rather than by early testing to identify drug combinations for clinical investigation that may deliver improved efficacy. A rationale for combinations exists across a number of diseases in which pathway redundancy or resistance to therapeutics are evident. However, early assays are complicated by the absence of both assay formats representative of disease biology and robust infrastructure to screen drug combinations in a medium-throughput capacity. When applying drug combination testing studies, it may be difficult to translate a study design into the required well contents for assay plates because of the number of compounds and concentrations involved. Dispensing these plates increases in difficulty as the number of compounds and concentration points increase and compounds are subsequently rolled onto additional labware. We describe the development of a software tool, in conjunction with the use of acoustic droplet technology, as part of a compound management platform, which allows the design of an assay incorporating combinations of compounds. These enhancements to infrastructure facilitate the design and ordering of assay-ready compound combination plates and the processing of combinations data from high-content organotypic assays.
Keywords
Introduction
Late-stage attrition of candidate drugs in clinical trials as a result of poor efficacy has been a significant factor in the decline of R&D productivity in recent years. 1 In the years 2005 to 2010, there were 50% fewer new molecular entities approved by the major drug administration regulatory bodies compared with the previous 5-year period despite higher levels of financial investment and increasing knowledge of disease. 2 Drug discovery in the postgenome era typically focuses on identifying compounds with selectivity against a given target; however, a number of these therapies have suffered from lack of efficacy in the clinical setting. 3 The genetic complexity of many diseases suggests that ablation of a single target is unlikely to produce a sustained effect because of pathway redundancy and/or resistance mechanisms.4,5 Furthermore, genetic studies have shown that in mammalian models, inactivation of a single gene is often not sufficient to lead to a discernible phenotype. 6 Thus, one strategy to bring improved success to drug discovery is the rational design of drug combinations and multitargeted therapeutics within early drug discovery.7,8
In creating a drug-combination capability, the aim is to develop a scientific competency that integrates informatics, in silico, analytical, and experimental methods to enhance and formalize the prioritization of drug combinations for preclinical and clinical development. Methods to identify effective drug combinations include experiments to identify synergy, independence, or antagonism on a given disease phenotype. 9 The combinatorial space in such studies increases exponentially as the number of agents of interest is increased. As such, identifying optimum drug combinations and concentrations experimentally represents a significant challenge. For example, a study of 50 different drugs, each tested pairwise with three possible doses, requires a minimum of 11,175 experiments. Researchers have aimed to address this challenge using in silico approaches,10–12 closed-loop optimization studies,13–15 and medium-throughput screening.16–21 Challenges exist in the efficient implementation of such a capability within early drug discovery, where there is a need to translate complex drug-combination study designs into the required contents for assay plates and in some cases for medium-throughput screens. Automated processes that are standardized across multiple users could potentially facilitate best-practice design, improved data quality, and speed of data delivery for medium-throughput combination studies.
Within AstraZeneca, the Compound Management (CM) group has the task of supporting numerous projects in various stages of drug discovery, demanding a range of technology, capabilities, and delivery capacities. The combinations intermediate assay-ready plate and screening workflow project was initiated in response to increasing demand for combinations screening in early drug discovery and the consequent need to optimize the process. Prior to the project, combination experiments typically involved bench-top processing of intermediate dilution plates using conventional dispensers. These processes and workflows used substantial amounts of compound and high levels of manual processing and carried a significant risk of error. This project was seen as an opportunity to develop an integrated system that allowed project scientists to request intermediate assay-ready drug combination plates, screen in complex organotypic cell models, and process and analyze combinations data in an efficient manner. Here we describe the agile development of a new software tool, Apothecary, that enables experiment design by the project scientists and the creation of system files to allow automated production of combination intermediate assay-ready plates by the CM group using acoustic droplet technology. 22
The benefit of this software tool as part of a combinations assay approach is the ability to systematically screen potential drug combinations early in drug discovery at acceptable throughput, quality, and cost. In particular, the development of the software tool we describe enabled utilization of acoustic droplet technology for dispensing compound combinations. We further describe a proof-of-concept study as part of this article that demonstrates significant cost and time saving over previously used manual and nonacoustic approaches.
System Development
The end-to-end combinations process is illustrated in Figure 1 . The workflow begins with the design of the experiment followed by experimental test, data formatting, quality control and validation, storage, and results visualization. The software we describe here, Apothecary, enables scientists to design combinations experiments, receive centrally prepared intermediate assay-ready combination plates, and process combinations data automatically using existing data analysis tools.
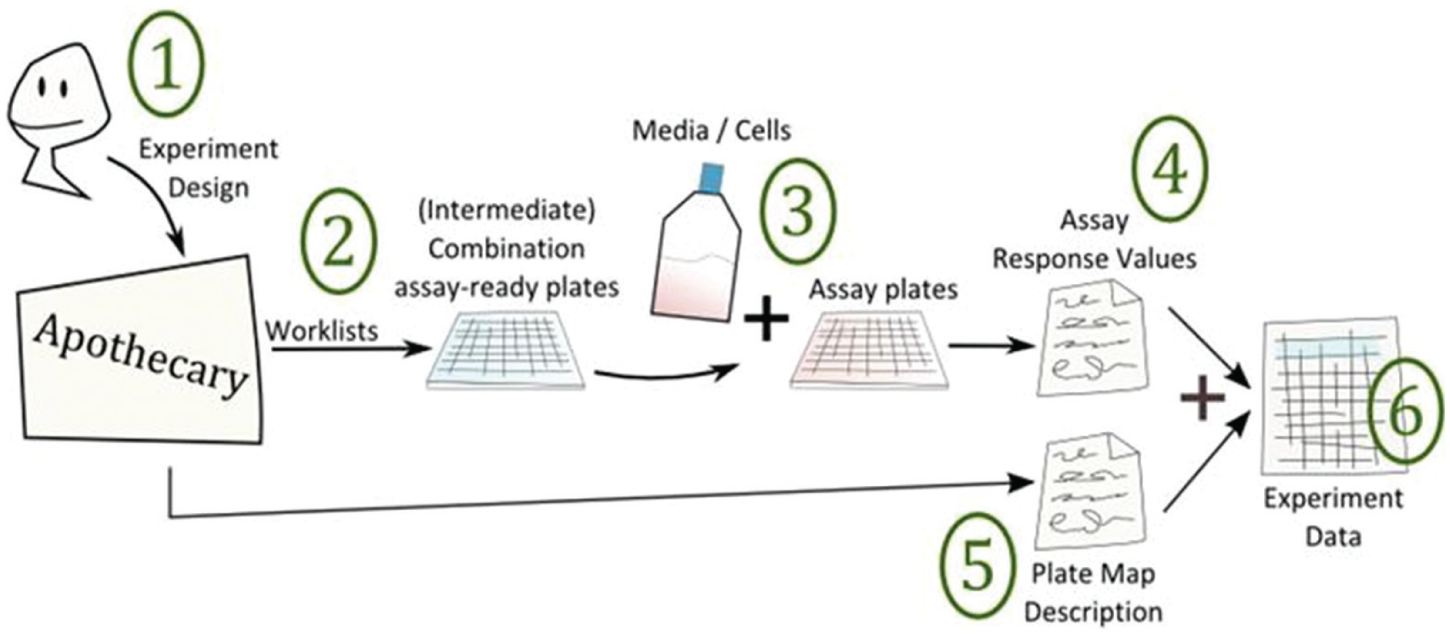
Combinations screening workflow overview from experimental design to data interpretation. The end-to-end workflow is: 1. design of experiment, 2. intermediate assay-ready plate generation, 3. experimental test, 4. results processing, 5. data formatting, quality control, and validation, 6. data storage and results visualization.
To ensure a fit-for-purpose solution, requirements were gathered from the multiple disciplines involved: informaticians, assay scientists, and CM. Multiple test cycles of the developing software were completed to drive usability and further refine specifications using an agile process. Assay scientists required the ability to request and receive intermediate assay-ready combination plates for pairwise combinations in a range of containers and using an array of experimental designs. The ability to predict experiment size/cost and track progress and content of ordered intermediate assay-ready plates was also important, as was the aim to deliver combinations data within the standard cycle time for monotherapy assays. The CM group required a solution that delivered automatic translation of experiment designs into definition of the source compound and concentrations required for the Echo (Labcyte, Sunnyvale, CA) work cell platform, definition of the location of compounds in the intermediate assay-ready plates, as well as integration with the global assay-ready plate requesting and dispensing system. CM also required information on the total volume to dispense in the intermediate assay-ready plates to ensure it was sufficient for replicate assays, the final dilution factor (how much more concentrated the plate that CM provided would be than the given dose-point dilutions), and the position and substance of plate controls. Visualization of combination data presented many challenges, in particular the ability to visualize and interrogate complex data composed of multicompound treatments. As part of the project, significant effort was expended in identifying a flat-file format for data, that when uploaded into visualization and analytical tools, would retain multicompound treatment information for interrogation. Informatics demand also required experimental results and definitions to be associated and for standardized data files compatible with downstream visualization, analytical tools, and data storage platforms to be produced. The user requirements identified here led to the specification of a number of inputs and outputs required at specific stages of the experiment design and intermediate assay-ready plate ordering process ( Fig. 2 ).
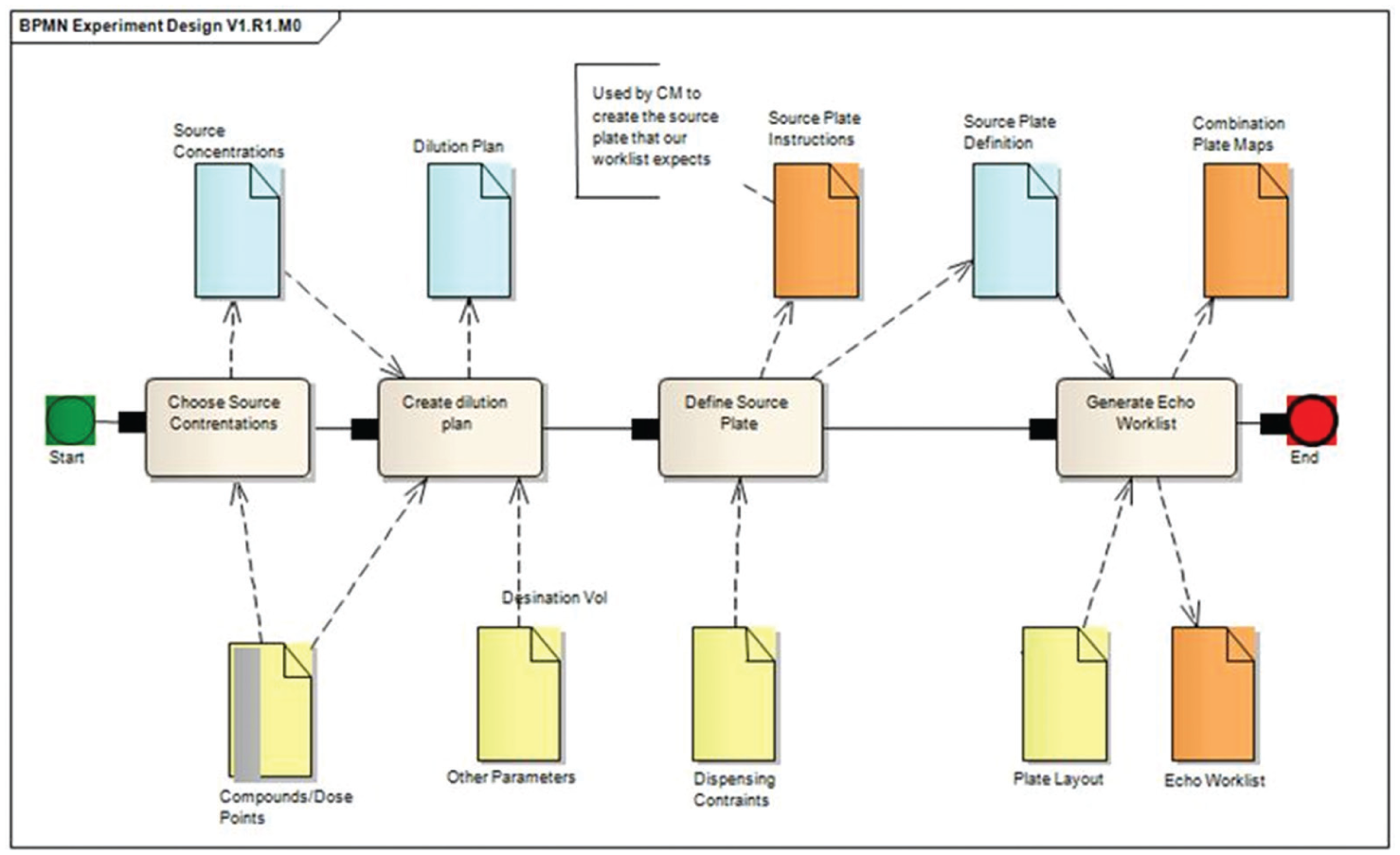
Overview of Apothecary system experimental design showing required inputs and outputs. The process flow involves: 1. choose source concentrations (both source and compound dose range), 2. create dilution plan, 3. define source plate, and 4. generate echo work lists. Required inputs and outputs are illustrated by arrows to and from the process flow, respectively.
The Apothecary system was developed with the operating principles and specifications as outlined as a bolt-on to AstraZeneca’s standard compound ordering platform that also integrated with the end-to-end combinations workflow. Apothecary was designed to enable a combinations assay process that used Echo (LabCyte) acoustic droplet ejection technology both for integration with AstraZeneca’s standard compound ordering platform and to realize the benefits of acoustic droplet ejection technology for compound combination dispensing. Use of the Compounds On Demand for Assay (CODA) system (The Automation Partnership [TAP], Royston, UK) 23 and existing BioCel automation platforms (Agilent, Santa Clara, CA) allowed rapid deployment of new capability, centralization, and standardization of combination work into one process. This approach also exploited existing sample management infrastructure without any additional hardware cost. Key to the exploitation of the CODA platform for combinations was the acoustic droplet technology present within CM. This platform provides the means to dispense very low volumes of liquid to high levels of precision, allowing conservation of liquid stocks, without the requirement for tip washing, thus eradicating any possibility of cross-contamination that exists with conventional contact liquid dispensers incorporating tips. 22 Additional benefits over manual combinations intermediate assay-ready plate production include the ability to screen drugs beyond pairwise combinations, reduced labor intensity, and increased quality coupled to a more robust dispensation, alongside plate and reagent cost reduction. Integration of Apothecary with an existing automated CM system also potentially provides opportunity to screen at a medium throughput and add randomized placement of combinations across plates.
Software and Principles of Operation
The Apothecary system takes combination experiment design inputs alongside hardware system constraints to generate work lists that allow CM to produce intermediate assay-ready combination plates on any automation platform using Labcyte Echo (Labcyte). Currently within AstraZeneca, these platforms are CODA (TAP) or a BioCel (Agilent). The system further allows result interpretation by output of target plate data in a standardized format to allow data analysis by the requesting scientist. The software was programmed in Java and is accessed through a web-based graphical user interface.
User Input
Apothecary uses an experimental design wizard to obtain all required user inputs. This comprises three steps: definition of compounds and concentration, test combinations and plate layouts, and controls. Step 1 requires input of the identifiers of compounds or compounds to be used in the study as well as the concentrations to be tested ( Fig. 3 ). The concentrations are the final assay concentrations and are displayed in µM. The values are entered either manually, manually with a dilution wizard, or from a pre-prepared comma- or tab-separated file. At this point, users also assign an experimental reference number to the study, for example, an electronic lab notebook reference number. This unique study identifier is used within file names and emails created by the system to indicate association with a specific study.
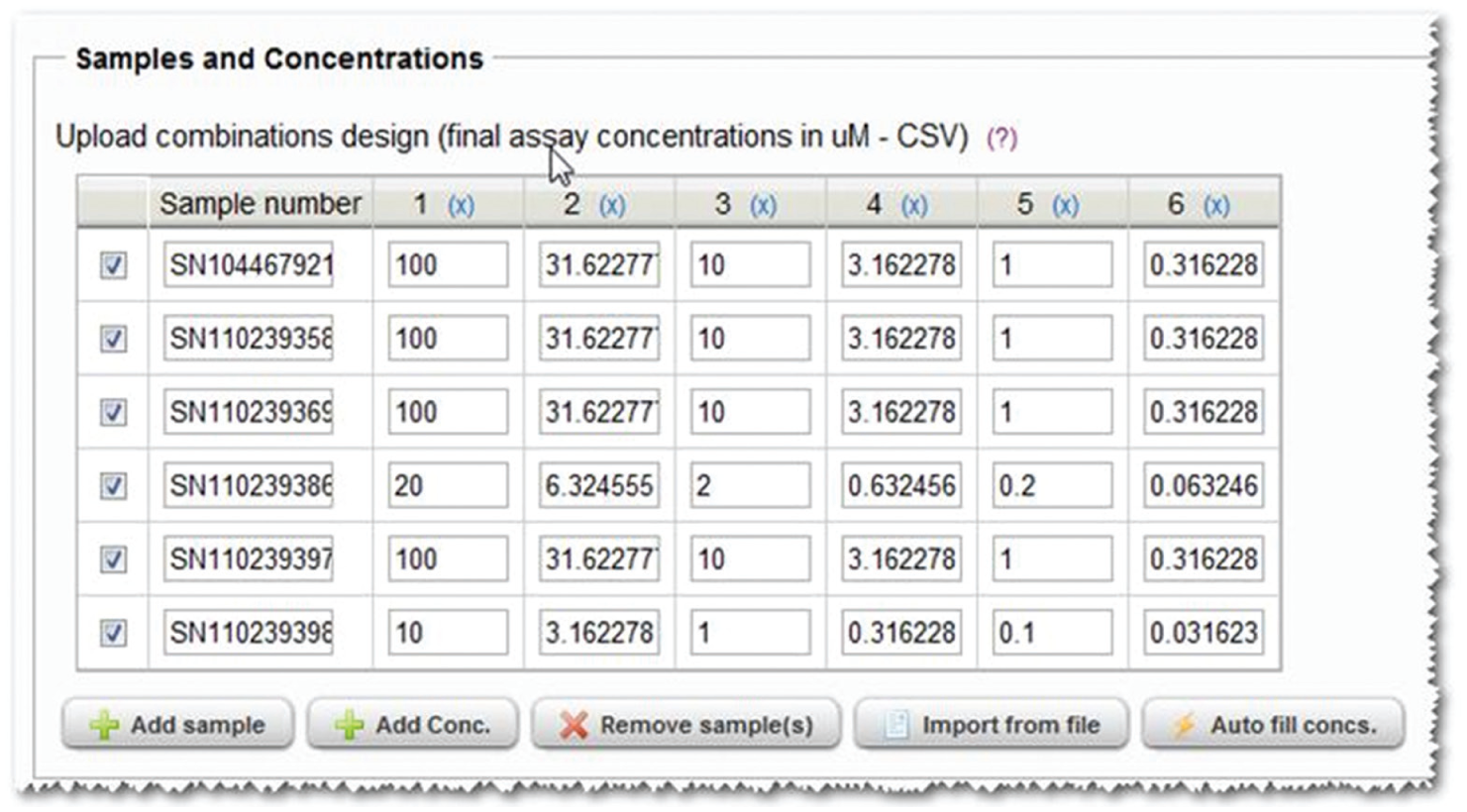
Apothecary experimental design wizard screenshot. The screenshot shows the sample ID and final assay concentration software wizard allowing the user to design the respective combinations experiment.
Configuring Dose and Compound Combinations
In many cases, a combination study will be designed such that all compounds are tested against each other at all dose points for one compound against all dose points for the other compounds. In some cases, experimental designs may represent a subset of all the possible combinations of drugs in a given study, for example, for synthetic lethal screening in which the effect of inhibiting a number of different targets (compounds B–Z) is tested in the presence of compound A or where availability of test compound is limiting. Step 2 of the experimental design wizard allows users to specify a combinatorial matrix (
Fig. 4a
), including the desired drug and dose matrix (
Fig. 4a

Apothecary combinations design wizard screenshot. The screenshot shows the software wizard allowing bespoke design of a combinatorial experiment. (
Setting Plate Layouts, Controls, and Standards
Apothecary will take user-defined parameters of compound, dose, and combinatorial matrix to identify all unique combinations to be tested and distribute across wells of as many intermediate assay-ready plates as are required to complete the experiment. User input is required so that the system takes account of the desired plate layout including the identity and position of controls and standards. Step 3 of the experimental design wizard enables users to select one of several prepopulated plate layout templates. These are plate layouts consistent with common standards for plate-based assays. The system also has sufficient flexibility to allow novel plate layouts to be used by enabling manual design within the software tool ( Fig. 5 ). Plate layouts contain a set of controls and/or standards within it. For each control or standard, the system allows specification of whether the compound should be dispensed by CM into the assay plates or whether the well should be left empty for the end user to add manually later. The final values that must be explicitly provided and defined are the number of copies of the intermediate assay-ready plate to dispense (e.g., for experimental replicates), the final required volume in the intermediate assay-ready plate (nL), and the cell plate dilution factor. The cell plate dilution factor is the dilution that will take place by transferring contents of intermediate assay-ready plates to the cell assay plates combined with media. There is additionally an option to randomize the position of different combinations.
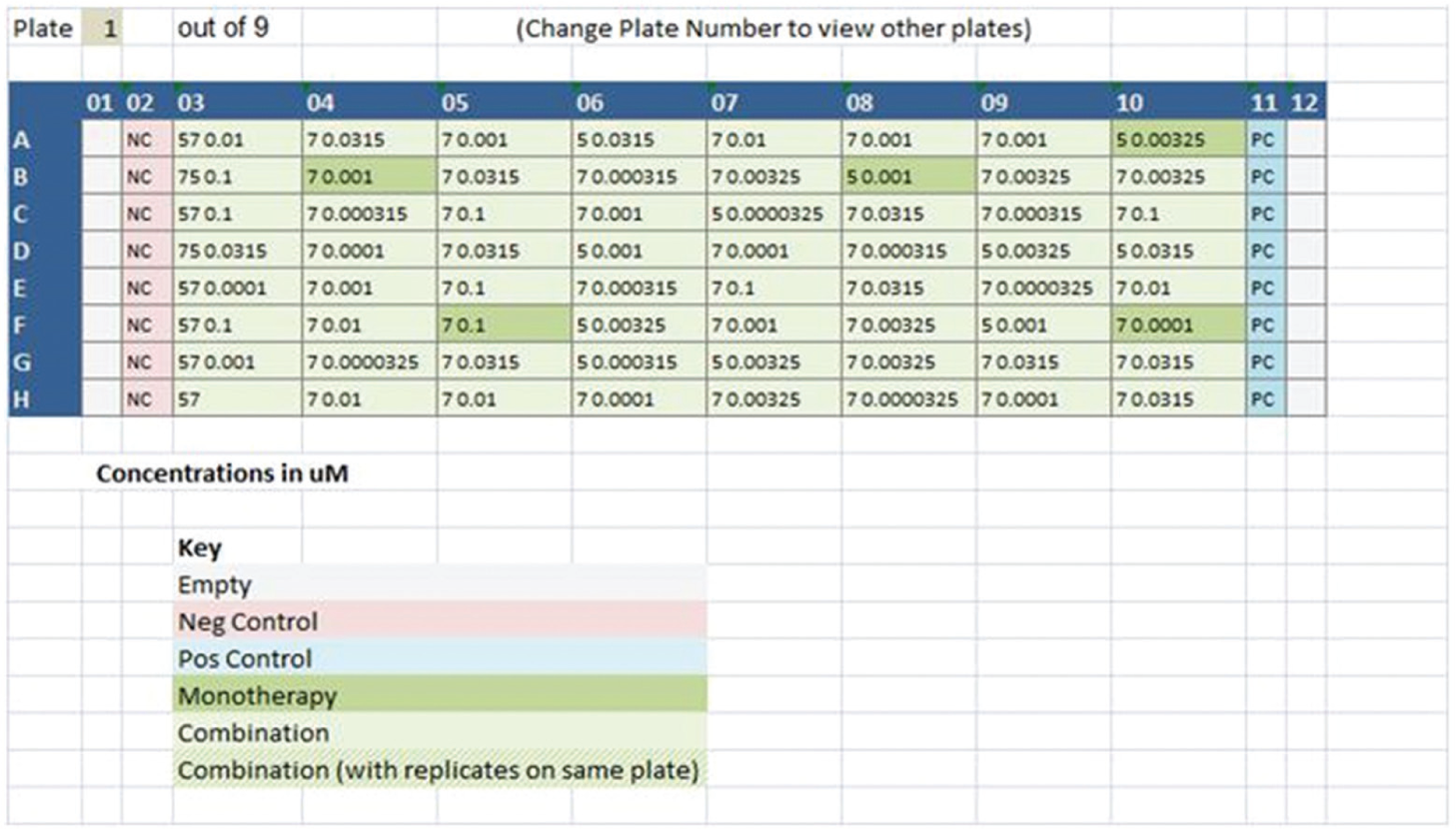
Manual microplate layout design tool featured in Apothecary software. The screenshot shows the section of the software wizard where upload of the plate map, final assay concentration positions, and controls or monotherapies can be easily entered by the user to aid in design of the respective combinations experiment.
Computation and Output
The entered values are applied to an algorithm written to translate the entered high-level combinations experiment design to detailed operational instructions. Upon completion of computation, Apothecary produces an experimental summary that details the input parameters provided to describe the experiment, the scale of the experiment (i.e., number of plates and wells), and the volume of compounds required to create the experiment. Hard coded into the algorithm are hardware constraints such as the need for the Echo (Labcyte) to dispense in multiples of 2.5 nL. Where constraints are such that modifications are required, for example, concentrations of compounds, the system identifies the deviation and requests user approval. To complete the dispensing request, Apothecary creates a zip of all required output files ( Fig. 6 ). These can either be downloaded to a network-connected machine or emailed to CM. Emailing the files provides CM with the necessary information to start processing the order. The CSV data files contained in the email are used by CM to drive Echo (Labcyte) dispense systems incorporated into the respective work cell. These work cells are controlled using OverLord 2 software (Peak Analysis and Automation [PAA], Farnborough, UK) or by Agilent VWorks (Agilent) allowing a fully automated solution to combination intermediate assay-ready plate production. An additional plate file is generated by Apothecary that specifies the contents of each well in the intermediate assay-ready plate. In this file, errors in dispensing can be identified (e.g., where nothing was dispensed when there should have been), and this can be used in a number of utilities for downstream data analysis. A plate map viewer tool, written using a macro in Microsoft Excel, can be used to visualize these data in a user-friendly format.
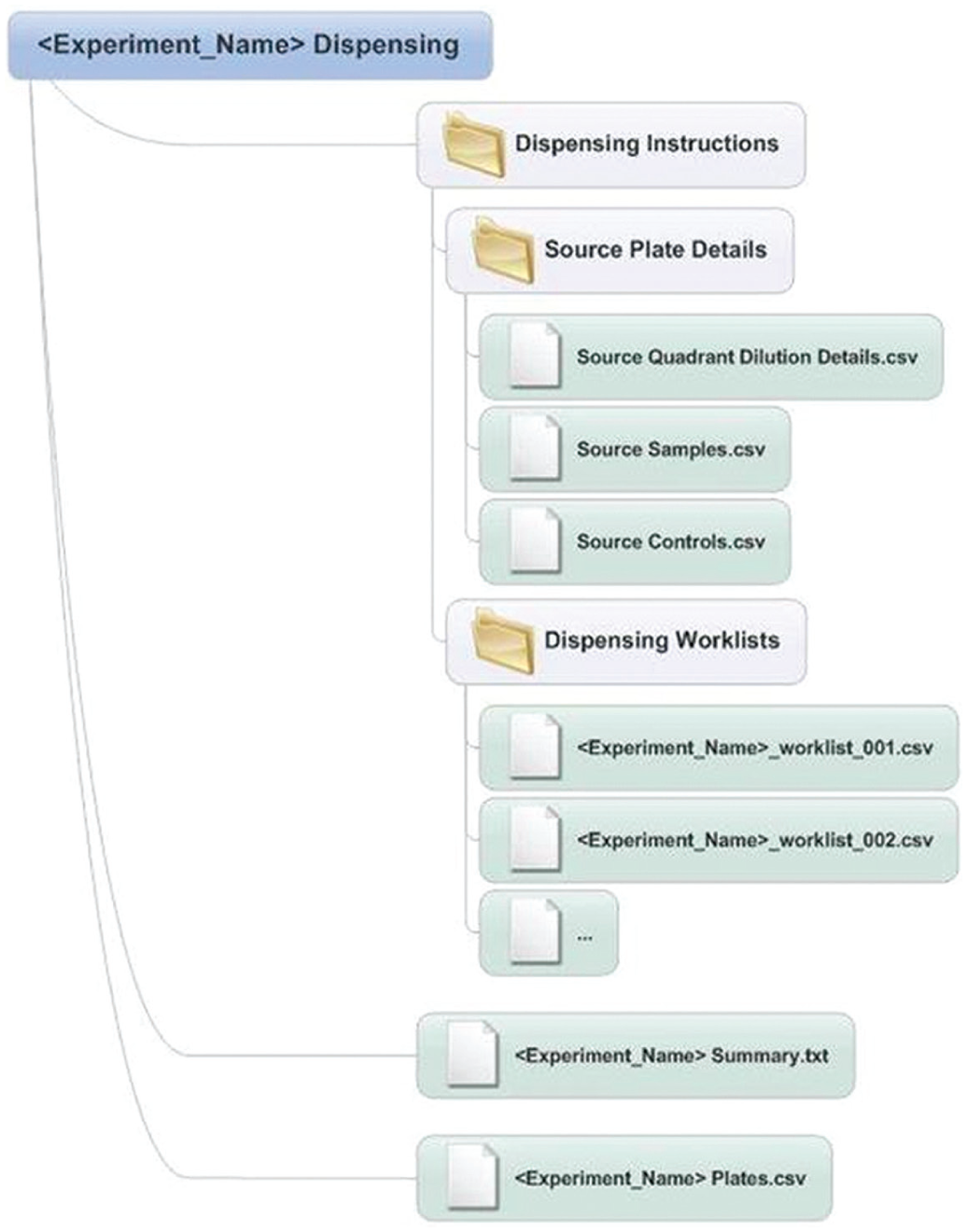
Overview of Apothecary output files for creation of combination intermediate assay-ready plates. Diagram showing the output files emailed to Compound Management staff in order for the designed experiment to be dispensed. These files contain all the details for production of the intermediate assay-ready combinations plates. Also contained in this folder are the details of the respective plate maps and concentrations for the end user, which are used as part of data analysis.
Simulation
As part of the Apothecary software tool development, we also created an Echo simulator that allows users to simulate the Echo (Labcyte) operations that would occur in CM production of the assay-ready combination plates. Initially, the tool was developed to allow validation of the developed algorithm by testing the work list that was to be given to the Echo dispenser. This test demonstrated that the described work list would not dispense more from a well than was possible (accounting for dead volume). This allowed faster development rather than manually working through the Echo work list files. Subsequently, the tool was used to predict experimental cost. The simulator takes the files generated by Apothecary for a CM operator and runs a simulation to predict what would happen to the initial compound source when dispensing a combinations study. The simulator produces a graphical plate viewer display showing where and how much the system has dispensed ( Fig. 7a–d ).
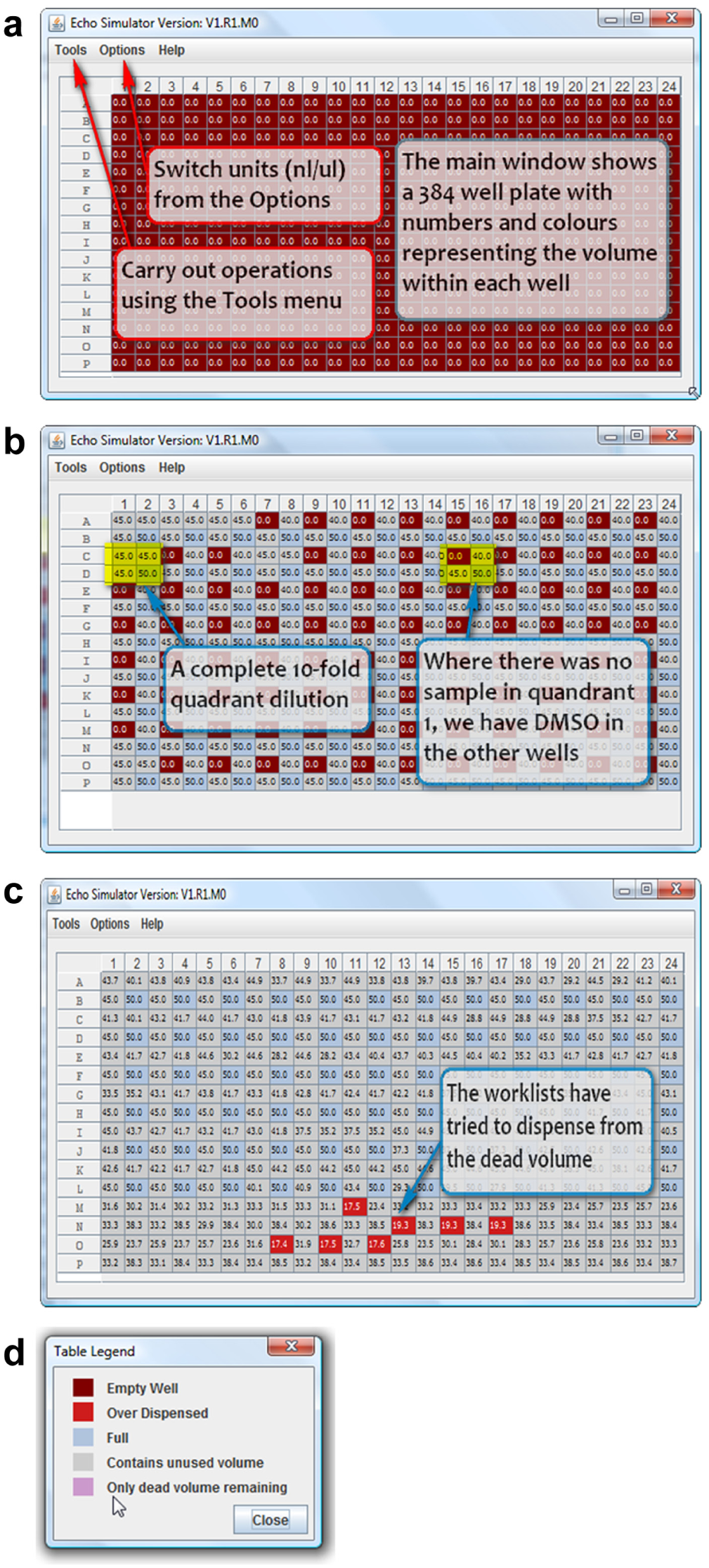
(
Hardware
Apothecary has the ability to work across multiple hardware platforms. This is delivered by design as the system produces files to drive the Echo acoustic droplet unit (Labcyte) and does not attempt to create protocol files for a given operating system or in fact be an operating system itself. This leaves the operator free to create protocols to run whichever automation system is currently in use. The complex Echo driver files are created for the operator to be called upon when required to produce the combination intermediate assay-ready plates. We define intermediate assay-ready plates as compound-containing microplates that can simply be removed from a freezer, have buffer/reagent dispensed into them, and undergo only one further step for the plate to be ready to use in assay. An example of this further step could be a n-fold dilution onto a cell-seeded assay plate.
Within AstraZeneca CM, there are currently two platforms for the production of assay-ready plates that Apothecary can provide such files to respectively. The first of these platforms is a BioCel system (Agilent) comprising a humidity- and temperature-controlled enclosure, a Direct Drive Robot (Agilent), a combi-nano dispenser (Thermo Fisher Scientific, Waltham, MA), Plateloc heat sealer (Agilent), V-spin centrifuge (Agilent), Nexus de-sealer (Brooks, Chelmsford, MA), and an Echo acoustic dispense unit (Labcyte). This system is operated using V-Works (Agilent) and is a standalone system dedicated to small, involved pieces of work.
The second platform is known as the CODA system (TAP). 23 The CODA (TAP) system consists of two work cells allowing for peaks in demand and cell redundancy for maintenance. The work cells were designed and built by TAP, who partnered with PAA and Labcyte to deliver a fully automated acoustic dispensing platform. These cells are integrated into the AstraZeneca database and are used for production of nearly all assay-ready plates for lead identification and lead optimization work. Each of the two cells comprises three modules: a plate-handling module, an Echo (Labcyte) feeding module, and a carousel unit. CODA has been designed to be able to deliver both large-scale primary screening work and smaller-scale secondary screening follow-up in one automated platform. The system can deliver up to four assays running concurrently at a rate of 100,000 compounds per day for primary screening. The system can also support production of plates for secondary screening of up to four assays running concurrently at a rate of up to 5000 compounds per week per assay. These typically produce curves of between 7 and 12 different concentration points. The hardware is driven using Overlord 2 software (PAA) linked to a commercially available operating system, Concerto (TAP).
The two-work-cell infrastructure available on CODA (TAP) enabled business-as-usual delivery while evaluation and development of Apothecary took place simultaneously (BioCel not in operation at the time of proof of concept). Due to the complexity of combinatorial compounds existing within one container (e.g., two compounds per well of a microplate), the process, although being automated, is handled in an offline fashion while in CM. With this in mind, the CODA operating system, Concerto (TAP), is bypassed for combination orders, and the underlying third-party controller OverLord 2 (PAA) is used to drive the work cell. A standardized script, coupled with the automatically produced Echo (Labcyte) driver files from the Apothecary software, allows automated production of the target labware through the work cell.
Once the combination intermediate assay-ready plates arrive with the end user, the associated data files produced by Apothecary are also available to the scientist to facilitate easy data interpretation downstream.
Exploitation
We validated the developed combinations approach through the testing of pairwise combinations of six compounds in a human primary co-culture model of angiogenesis. Angiogenesis is the growth of new blood vessels from preexisting vasculature. It is a fundamental biological process essential to the survival of the tumor. As a result of drug resistance and pathway redundancy, currently available vascular endothelial growth factor–targeted therapies display suboptimal clinical efficacy.
24
Hence, there exists a rationale to exploit a combinations approach to antiangiogenic drug development. In this study, we tested six compounds pairwise with each compound being tested in combination with each other compound. To calculate the size of such an experiment, the formula
The angiogenesis assay was run according to previously published protocols. 25 In brief, human umbilical vein endothelial cells (pooled) and normal human dermal fibroblasts were cultured in 96-well microplates for 10 d, with compound treatment and/or media replenishment at days 0, 4, and 7 in co-culture. After 11 days, co-cultures were fixed, immunolabelled (anti-CD31 and Hoechst), and imaged using a high-throughput fluorescent imaging microscope. Automated image analysis was used to quantify the total tubule area per well, as well as to capture additional features of endothelial tubule and fibroblast cell morphology. 25 Biological activity was measured as percentage inhibition of the total tubule area. Combinations data were analyzed by integration of Apothecary and Image Analysis output files using Pipeline Pilot (Accelrys, San Diego, CA) to generate a single flat file that preserved compound combination data. This file was imported to the CombinatoRx Chalice software tool (Horizon, Cambridge, MA) for statistical analysis and visualization. In brief, biological activity of compound combinations was visualized as a dose matrix ( Fig. 8a , percentage inhibition data) and combination effects characterized by comparing each data point with that of a combination interaction Lowe additivity reference model derived from the single agent curves ( Fig. 8a , Lowe model). The predicted additive response was subtracted from the actual result to give the excess inhibition response ( Fig 8a , excess inhibition). All values were then summed to provide a single specific synergy score for any of the combination of drugs, allowing comparisons across different drug combinations, in which positive values indicate that the effect seen in combination is better than additive ( Fig. 8b ). On this occasion, the data indicated that compound combinations tested were all additive. This application of Apothecary and acoustic droplet technology realized a number of benefits over the previously employed manual methods. For example, the volume of each compound stock required for the experiment was reduced more than 13-fold, with 2 mL and 150 µL stock required for manual and Apothecary methods, respectively. Furthermore, the time taken from design to dispense of compound combination plates was significantly reduced, as was the likelihood of dispense error. This practical application of the developed system demonstrates the potential to adopt this solution to the testing of other combination hypotheses in different multicellular disease models in a robust and efficient manner.
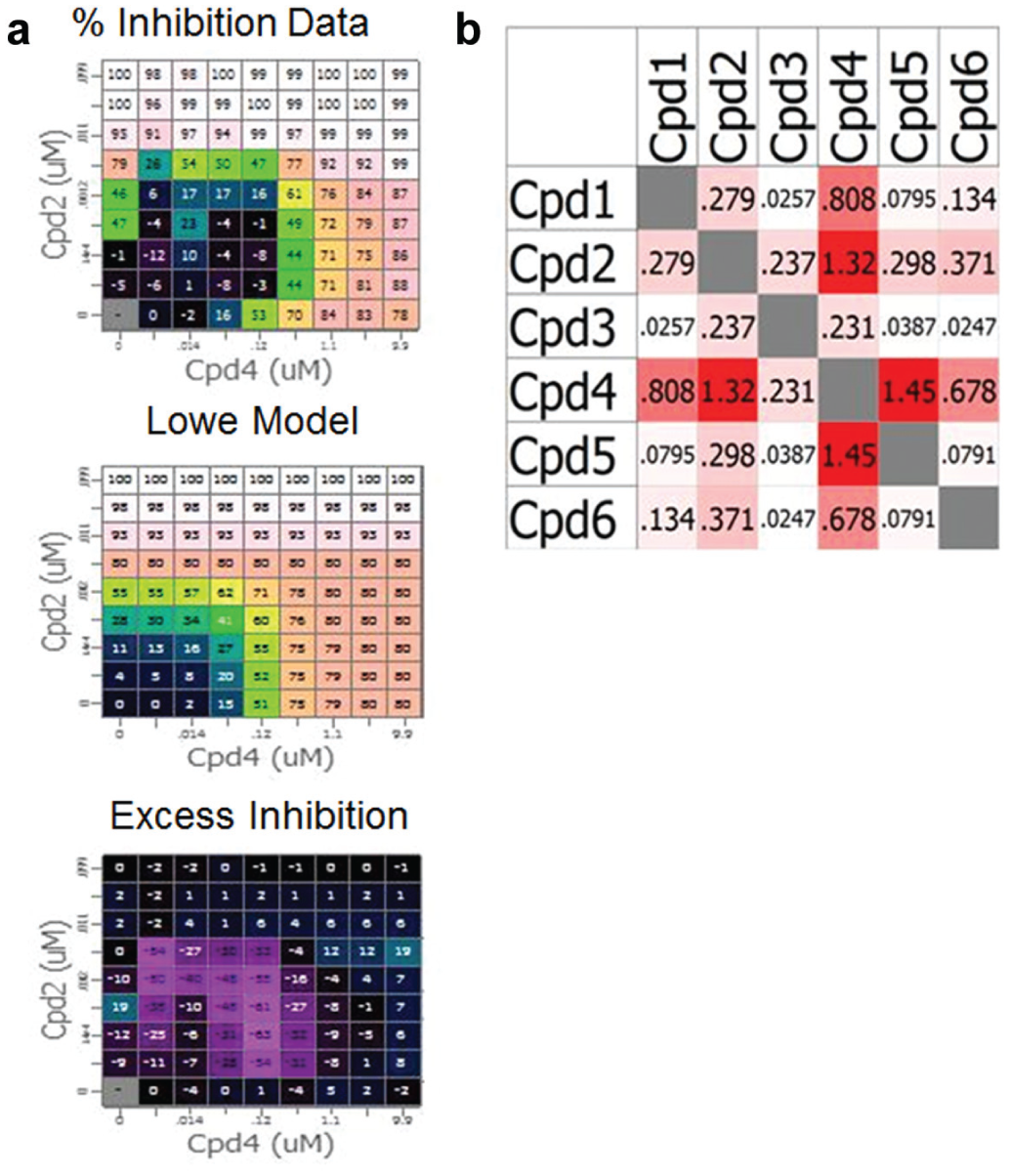
(
Discussion
We have developed an integrated screening approach for the preclinical study of drug combinations in phenotypic physiologically relevant models of disease. The major success of this work was the development and integration of a software platform, Apothecary, that enabled the design and dispense of intermediate assay-ready compound combination plates in a cost-effective manner. The utility of the developed approach was validated in a study of the effect of drug combinations on the process of angiogenesis in a physiologically relevant high-content assay.
The concept of assaying drugs in combination to identify those with potential for improved efficacy or to overcome resistance is not new; however, the process does encompass several separate and potentially inefficient operations. The system we developed aimed to implement a fully integrated process for combinations experiments by addressing the need to deliver intermediate assay-ready combination plates to user-specified designs in an efficient and cost-effective way. Where possible, the solution used and integrated with existing workflows and in particular was based on the use of acoustic droplet technology. The advantage of this over previously employed manual approaches was the reduced volume of stock liquid compound used in the process. This equated to a reduction from 2 mL to 150 µL per compound in the case study used to validate the newly developed system. In addition, our new approach reduced the time required to generate a combination plate from 45 min to 5 min and reduced the possibility of dispensing error. Apothecary was further integrated with the automated processing of combinations data through the use of the third-party tool, CombinatoRx Chalice. Chalice allowed rapid analysis and visualization of combinations data. The success of the platform depended on an agile process for development in which the end-user scientists, informaticians, and sample management staff worked closely and iteratively with the software and algorithm developers. Critical to this process was the use of simulation tools to refine the dispensing algorithms in rapid iterative test cycles without the expense of wet experiments.
Enhancements
We applied and validated our combinations approach by testing pairwise combinations of compounds in a phenotypic human primary co-culture model of angiogenesis. The case study was chosen to illustrate functionality of the platform for a routinely used combinatorial experimental design at a medium throughput. With wider exploitation and adoption of the developed tool and workflow, we recognize the potential for diverse requests that may result in the need for future enhancements to the platform. For example, a planned future enhancement of the developed tool is an ability to assess compounds beyond pairwise combination to greater-order combinations such as triples. The system could also be developed for the automated dispensing of combination biologics or indeed small-molecule-biologics combinations. The main challenge this would introduce would be the need to take account of whether an aqueous dispense (biologics) or DMSO dispense (small molecules) is required. If Apothecary was to be extended to support larger experiments, multiple source plates would be required and the dispensing algorithm enhanced such that source plates are not required to be constantly swapped on to the Echo instrument. One solution may be to define a target plate set that could be produced from a single-source plate and change the randomization to spread wells across the set rather than all plates in all sets. Deployment of the currently developed system should in time help identify more areas for future development that may have the greatest benefit and be targeted for future enhancement.
In conclusion, we present a method to establish an integrated combinations workflow within a drug discovery setting. The developed software, Apothecary, comprised a guided user interface and dispensing algorithm. This enabled sophisticated combination experiment designs to be dispensed in an automated, cost-effective fashion through the use of acoustic droplet technology. We are now extending this work to the testing of other combination hypotheses in other multicellular disease models to ascertain whether the approach delivers improved early predictive power for combinations opportunities.
Footnotes
Acknowledgements
The authors wish to thank Clive Green, Ian Sinclair, and Sunil Sarda for critical review of the manuscript; Zalicus for access to CombinatoRx Chalice bioinformatics software under a collaboration agreement with AstraZeneca; and Grant Zimmerman (Zalicus) for helpful discussions on the analysis of combinations data using Chalice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
