Abstract
Cervical specimens collected in liquid-based cytology (LBC) media are the most common sample type used for high-risk human papillomavirus (HPV) testing. Since preanalytic steps such as vortexing and decapping vials, liquid transfer to a sample input tube with matching unique identifier, and recapping the original vials are required for processing LBC samples prior to running the Abbott RealTime High Risk HPV assay (Abbott, Wiesbaden, Germany), a full manual execution can be complicated, especially in high-throughput diagnostic contexts. Here, a custom-configured worktable setup for the Tecan Freedom EVO (Tecan, Männedorf, Switzerland) designed to automate and control preanalytic steps for ThinPrep (Hologic, Marlborough, MA) samples was used to evaluate the impact of automated versus manual preanalytics. Archival results for manual processing of 226 samples were compared with those obtained with the Tecan protocol, observing a very good overall concordance for final assay interpretation (95.6%). High overall agreement (100%) resulted also from retesting 99 samples by both the preanalytical protocols. High reproducibility was observed analyzing 23 randomly selected samples by automated preprocessing in triplicate. Hence, the new configuration of the Tecan platform translates the manual steps required to process ThinPrep specimens into automated operations, controls sample identification, and allows for saving hands-on time, while maintaining assay reproducibility and ensuring reliability of results, making it suitable for screening settings.
Introduction
High-risk human papillomavirus (HR-HPV) DNA detection in cervical specimens provides 60% to 70% greater protection against invasive cervical carcinomas compared with cell morphology-based cervical cytology, suggesting introduction of HR-HPV DNA testing as primary screening test. 1 The results of several large clinical trials and many pilot studies conducted to evaluate HR-HPV DNA testing as a primary screening mode in organized population-based screening programs led to the revision of European guidelines on cervical screening (e.g., Health Technology Assessment report in Italy 2 ).
Since 2012, many Italian regions started addressing the transition to the new screening algorithm with an organizational and professional appropriate network. Future cervical cancer screening programs based on HR-HPV DNA testing as a primary screening mode will use—in most cases—cervical specimens collected in liquid-based cytology (LBC) compatible with both cytological and molecular testing. Cervical specimens collected in ThinPrep (Hologic, Marlborough, MA) LBC medium represent one of the major LBC sample types currently used in cervical cancer screening, and high-level automation is available for processing cervical cytology with this sample type. Most of the currently marketed HR-HPV DNA tests are validated for use with ThinPrep LBC samples, which are collected in a 20-mL volume of liquid fixative, of which a test-specific fraction is required for HR-HPV DNA testing.
Preanalytic processing of ThinPrep LBC samples for HR-HPV DNA testing involves (1) mixing of the LBC vial according to instructions of the supplier of the test, (2) opening the vial, (3) aspirating a test-specific sample volume (4) followed by liquid transfer to a secondary tube, (5) closing the LBC vial, and (6) labeling the secondary vial with an ID that can be related to that of the original LBC vial. These process steps require significant hands-on time and leave room for errors when carried out manually. Consequently, automation of the preanalytic preprocessing of LBC samples is desirable, particularly in high-throughput screening settings in accredited laboratories carrying out HR-HPV DNA testing for primary cervical cancer screening.
According to International Consensus Guidelines,3,4 only HR-HPV DNA-based molecular tests demonstrating appropriate clinical performance and reproducibility should be applied in European HPV-based screening programs. 5 Hence, we evaluated reliability and ease of use of a custom-configured setup of the Tecan Freedom EVO System (Tecan, Männedorf, Switzerland), a liquid handling robotic platform custom-configured and designed to automate the preanalytic steps required for preprocessing of ThinPrep LBC specimens prior to HR-HPV DNA testing with the automated Abbott RealTime High Risk HPV assay (“RealTime HPV”; Abbott, Wiesbaden, Germany) by comparison of test results observed after manual and automated (“Tecan”) preprocessing.
Materials and Methods
Clinical Specimens
Residual material from 226 cervical specimens collected in LBC medium (PreservCyt solution; ThinPrep; Hologic) were obtained after routine analysis from the diagnostic service at the Unit of Microbiology, S. Orsola-Malpighi University Hospital, Bologna, Italy. Samples included in the study were from a referral population and from a population of patients surgically treated for cervical intraepithelial neoplasia grade 2+ (CIN2+). All specimens were stored at 4 °C for less than 6 months.
Detection of HR-HPV DNA
RealTime HPV (Abbott) is a qualitative multiplex real-time PCR test for the detection of 14 HR-HPV genotypes. The assay is performed on the highly automated Abbott m2000 System: DNA extraction and setup of PCR reactions are carried out on the m2000sp instrument, while amplification, detection of fluorescent signal, interpretation, and reporting of test results are carried out on the m2000rt instrument.
A primer mix consisting of three forward primers and two reverse primers targeting a conserved L1 region is used to amplify HPV targets. Signal for 14 HR-HPV genotypes (HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68) is generated with the use of fluorescent-labeled probes. An endogenous human β-globin sequence serves as an internal control (IC), which is amplified with a primer set and detected with an IC-specific probe. Single-stranded nucleic acid probes specific for HPV 16, HPV 18, non–HPV 16/18 genotypes (other HR-HPV), and IC are labeled with different fluorophores, allowing their signals to be distinguishable in a single reaction. Four signals corresponding to HPV 16, HPV 18, other (non–HPV 16/18) HR-HPV, and IC are evaluated for each sample. Each HPV signal is determined as “detected” if the cycle number (CN) observed is found below a fixed threshold (32.00 cycles) or “not detected” in the absence of signal or in the presence of signal beyond the assay cutoff. HPV signals (HPV 16, HPV 18, or other HR-HPV) are reported with the respective CN values (in parentheses after the target result). Samples with any of the three HR-HPV signals detected will have an interpretation of “HR-HPV detected.” Samples with all three HR-HPV signals not detected will have an interpretation of “not detected.” IC signals below a predefined CN threshold (35.00 cycles) are invalidated by the system in the absence of an HPV target.
Preanalytic Processing of ThinPrep LBC specimens
As previously stated, ThinPrep LBC vials contain 20 mL of fixative; this volume is reduced when preparing microscopic slides, leaving 5 to 15 mL volume in the original LBC vials for the majority of samples. According to the standard protocol indicated by the manufacturer, cervical specimens collected in ThinPrep LBC medium have to be vortexed for 15 to 20 s prior to manual transfer of an aliquot of less than 1 mL to a secondary sample input tube, which is then placed into a dedicated sample input rack of the m2000sp instrument.
A Tecan Freedom EVO System was equipped with the following functionalities to automate the preanalytic process steps required to prepare ThinPrep LBC samples for testing with the RealTime HPV assay: (1) two custom-configured carriers that fit the dimensions of ThinPrep LBC vials (48 samples each), (2) a barcode reader for ThinPrep vials, (3) a vortexer, (4) a de/recapper unit, (5) six m2000 sample racks for carrying secondary sample input tubes with a dedicated barcode reader, and (6) a run script comprising barcode matching of LBC vials and m2000sp sample input tubes and carrying out the preanalytic process steps described ( Fig. 1 ). The system allows for preanalytic processing of 96 ThinPrep LBC vials in one run.

Worktable setup of the new configuration of Tecan Freedom EVO, designed to automate the preanalytical processing of cervical samples collected in vials for liquid-based cytology (LBC): global vision of the instrument (
Data Analysis
GraphPad Prism v.5 software (GraphPad Software, La Jolla, CA) was used for statistical analysis. Proportions are presented with 95% confidence intervals (CIs), estimated by standard methods. Cohen’s κ was calculated to determine the level of chance-adjusted agreement between the two different preanalytic processing procedures.
The difference between signals (CN) observed with RealTime HPV after manual and automated preanalytic processing (ΔCN) was calculated for HPV 16, HPV 18, other HR-HPV, and IC to compare both the manual and the automatic preanalytic processing procedure: ΔCN16/18/other HR/IC = Manual RealTime HPV CN16/18/other HR/IC – Tecan RealTime HPV CN16/18/other HR/IC.
Intra-assay reproducibility was assessed by calculating the overall concordance of results from triplicates of automatically preprocessed specimens (i.e., concordance if at least two-thirds replicates showed the same final interpretation). The average CN value, the corresponding standard deviation (SD), and the coefficient of variability (CV = SD/average CN) were calculated for both the HPV and the IC signal.
Results
Overall Concordance of Test Results after Manual and Automated Preprocessing of ThinPrep Samples
All samples used in this evaluation had been preprocessed manually prior to testing with the RealTime HPV assay during the laboratory routine, and results will be referred to as “manual 1” in this report. In a first approach, 226 LBC vials with residual volume (5–15 mL) were preprocessed, in three consecutive runs, with the Tecan instrument, followed by testing with the RealTime HPV assay; results will be referred to as “Tecan 1.”
Among the 226 samples tested in the first round of this study, 83 (36.7%) were HR-HPV positive and 133 (58.8%) were HR-HPV negative for both preanalytical methods used, leading to an overall concordance of 95.6% (95% CI, 92.0–97.7; κ = 0.907) ( Table 1 ). Ten samples showed discordant results and were retested in a parallel mode (i.e., the samples were preprocessed manually [“manual 2”] and automatically [“Tecan 2”] by the same operator on the same day prior to retesting with the RealTime HPV assay in the same run); 7 of 10 samples were concordant and 3 of 10 remained discordant for the final test interpretation ( Table 2 ).
Overall Concordance of RealTime HPV Results after Manual (Archival Results, Manual 1) and Tecan Preanalytic Processing of ThinPrep Liquid-Based Cytology Samples (Tecan 1).
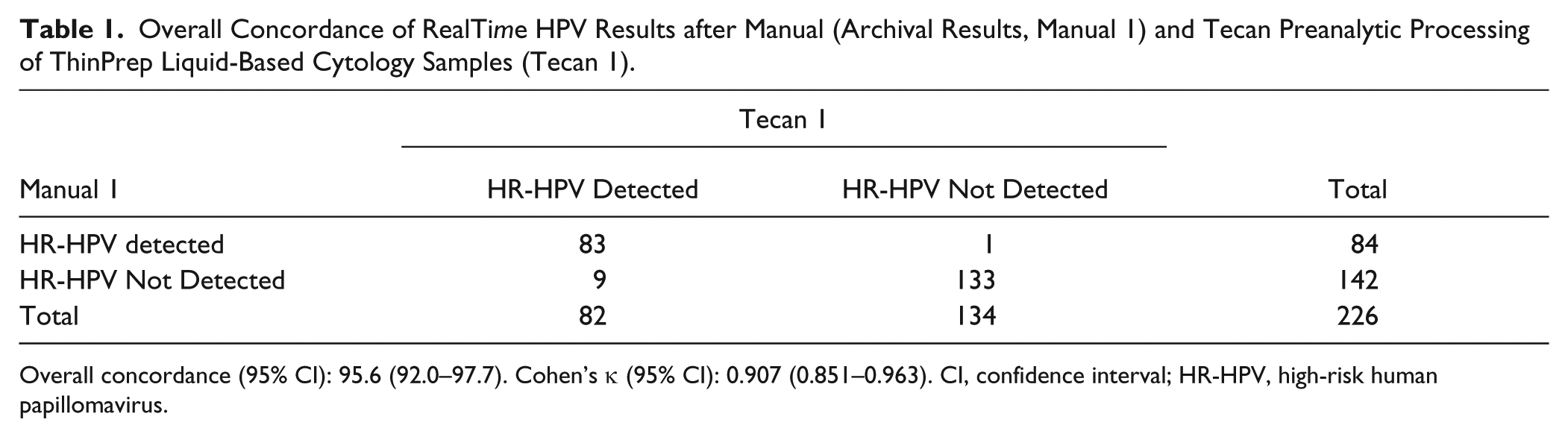
Overall concordance (95% CI): 95.6 (92.0–97.7). Cohen’s κ (95% CI): 0.907 (0.851–0.963). CI, confidence interval; HR-HPV, high-risk human papillomavirus.
Analysis of the 10 Samples with Discordant Results after the First Preanalytical Processing of Selected Samples with the Manual (Manual 1) and the Automated (Tecan 1) Procedures.
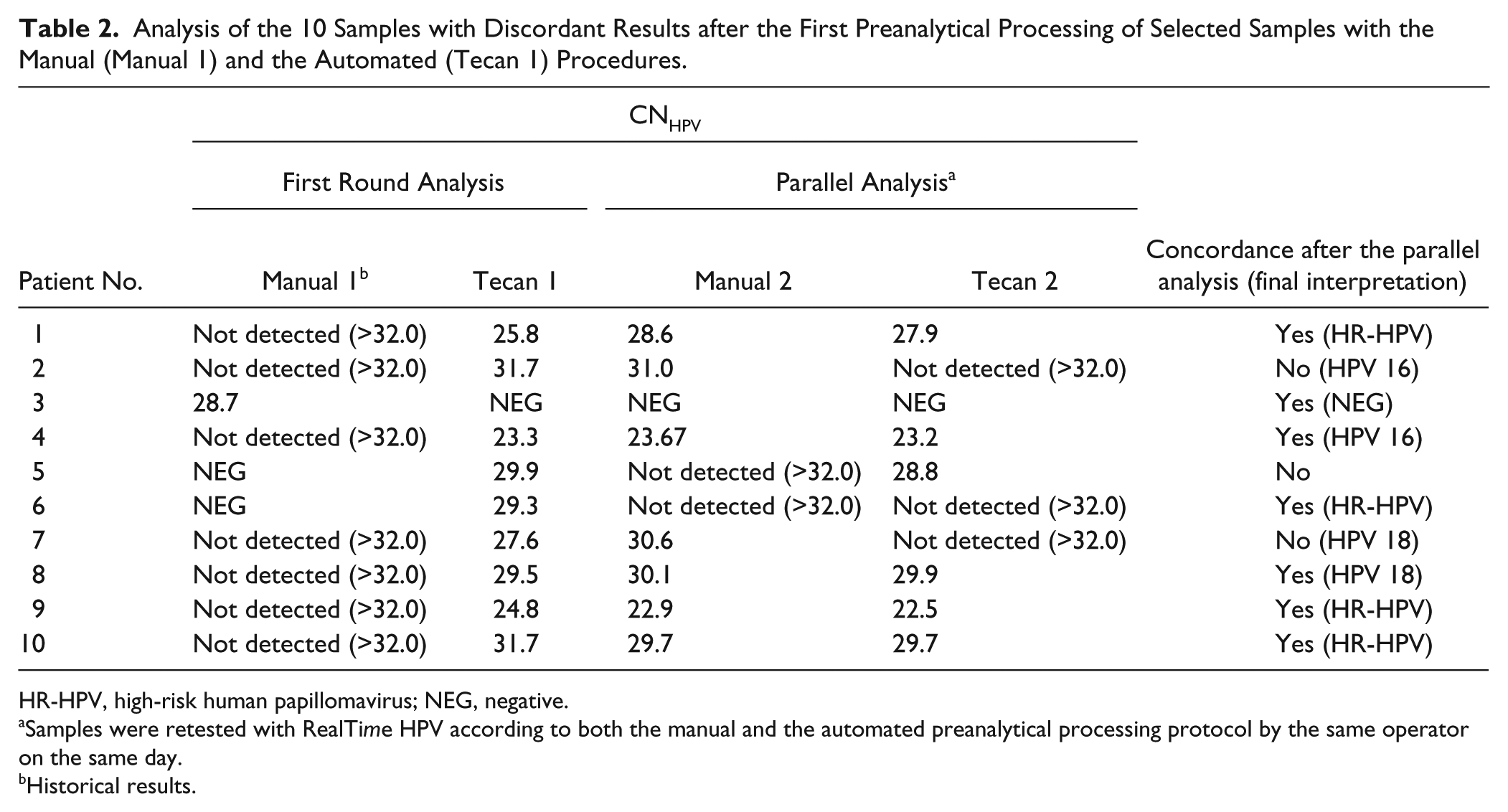
HR-HPV, high-risk human papillomavirus; NEG, negative.
Samples were retested with RealTime HPV according to both the manual and the automated preanalytical processing protocol by the same operator on the same day.
Historical results.
Considering the ΔCN analysis, for the IC we observed that most of the samples (159/226, 70.4%) showed a |ΔCNIC|<1.5, indicating an almost identical result after the manual and automated pre-analytic processing of LBC specimens. However, 67/226 samples (29.6%) showed |ΔCNIC|>1.5. The overall correlation between test results observed after application of the two different pre-analytic processing methods was rather low (R2 = 0.578; Fig. 2).
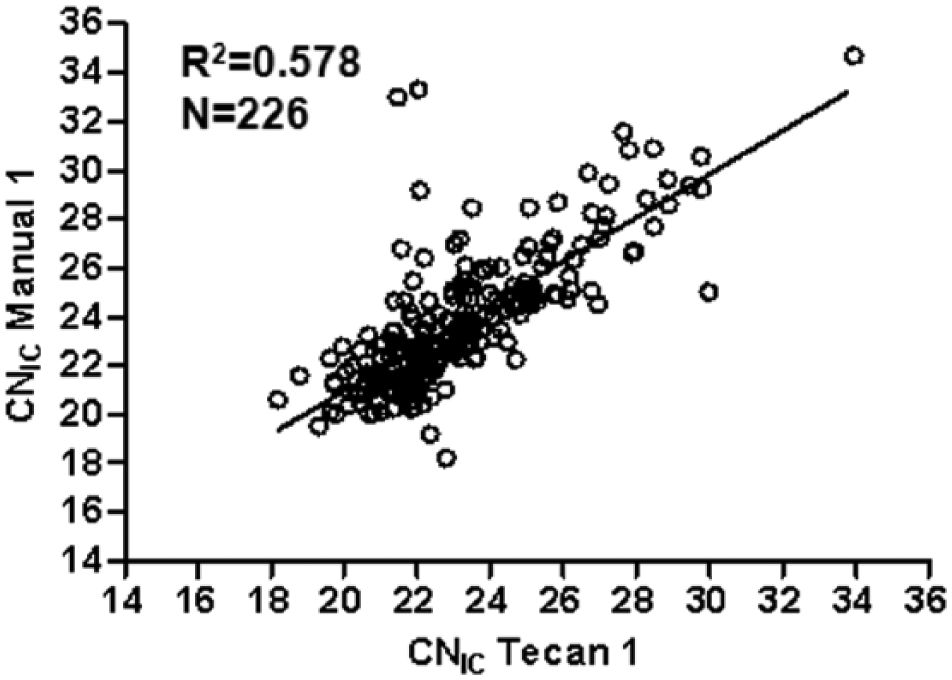
Linear regression analysis relative to 226 selected PreserCyt liquid-based cytology samples comparing historic CNIC values resulting from manual preprocessing (manual 1) with those observed after the preanalytic handling of the Tecan Freedom EVO (Tecan 1).
To eliminate both the possible interoperator variability and the variability due to the storage, we decided to retest 99 samples in a parallel way, as it was described above for discordant samples. After retesting, an overall concordance of 100% was observed for RealTime HPV test results, and the |ΔCNIC| was <1.5 for all the samples except for four samples, leading to an excellent correlation of signal observed with both preanalytical protocols (R2 = 0.958; Fig. 3 ).
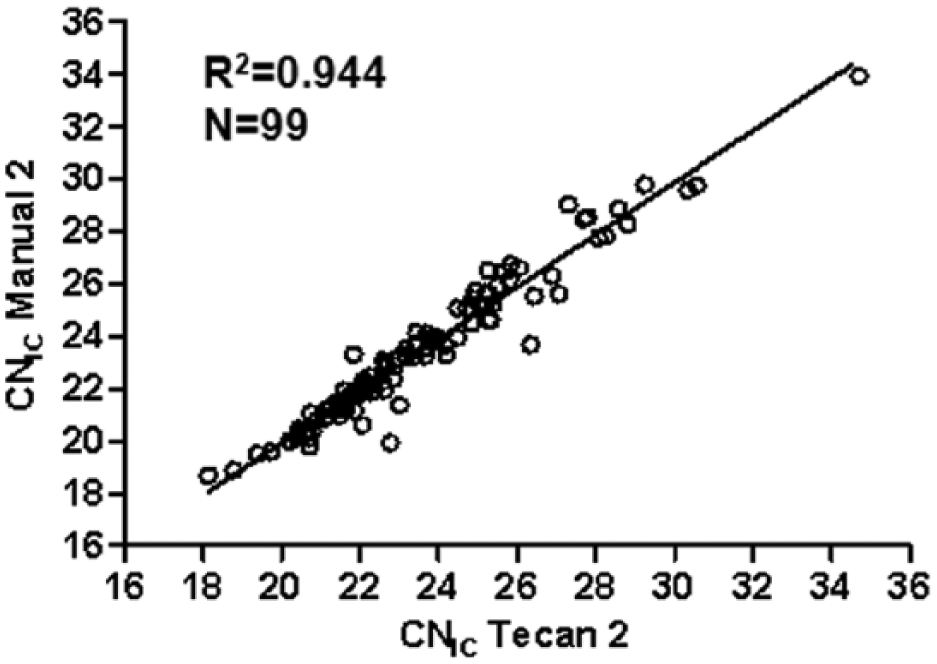
Linear regression analysis relative to the 99 retested samples comparing the CNIC values obtained after the automatic preanalytical processing protocol for ThinPrep liquid-based cytology specimens using the Tecan Freedom EVO (Tecan 2) and manual 1 (new results obtained at the time of this study, manual 2).
Inter- and Intra-Assay Reproducibility
For the 99 retested samples, we also compared the CNIC values and the CNHPV values obtained after each round of manual/Tecan-coupled RealTime HPV testing to assess the effect of the two types of preanalytical processing methods on interassay reproducibility ( Fig. 4 ). Reagents used for this experiment were from the same lots used for the previous experiment.
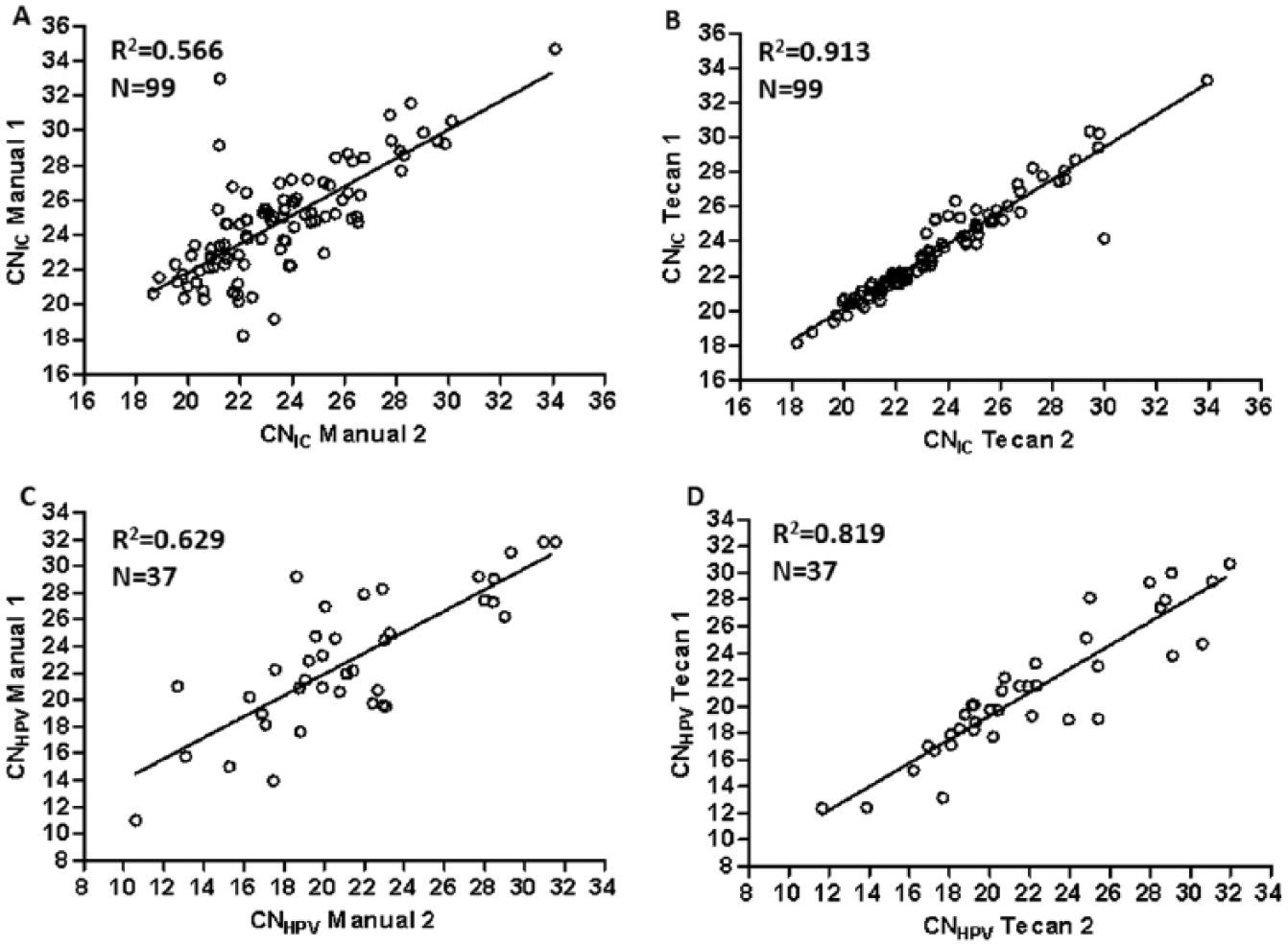
Linear regression analysis relative to the 99 retested samples showing the interassay reproducibility of signal intensity observed with RealTime HPV preceding manual (
As indicated in Figure 4A , C , the manual procedure showed good, even if not completely satisfying, correlations for both the CNIC and CNHPV values (R2 = 0.566 and R2 = 0.629, respectively). The Tecan protocol showed a very good correlation for both the CNIC and the CNHPV (R2 = 0.913 and R2 = 0.819, respectively; Fig. 4B , D ).
In addition, to assess the effect of the automatic preprocessing protocol on the RealTime HPV intra-assay reproducibility, 23 samples were randomly selected from the specimen collection and preprocessed in triplicate with the Tecan instrument on the same day prior to testing with the RealTime HPV assay in a single run. The overall concordance for the final test interpretation was 100% (95% CI, 83.1%–100%). Considering the CNIC analysis, we observed that 22 of 23 samples (95.6%) showed a coefficient of variation (CV) <2.0% (range, 0.1%–2.6%), and for the CNHPV analysis, 9 of 12 HPV-positive samples (75.0%) showed a CV <5.0% (range, 0.8%–8.6%).
Discussion
Cervical cancer screening will enter a new era with replacing cervical cytology by HR-HPV DNA frontline testing and using cytology as part of the triage of screen-positive women.
Sampling of cervical specimens in LBC media allows performing cervical cytology and HR-HPV DNA testing in cervical cancer screening on the same specimen. However, scrape samples from the cervical mucosal epithelium fixed in solution are a challenging specimen type due to their inhomogeneous nature and the settling properties of this material in alcoholic fixatives. Preanalytic processing of LBC samples for HPV testing requires appropriate mixing prior to sampling aliquots for nucleic acid extraction from the original LBC vial. Manual preanalytical processing of LBC specimens is laborious, is time-consuming, and leaves room for labeling errors.
We have evaluated a new worktable setup of the Tecan Freedom EVO system custom-configured to automate the preanalytic steps required to prepare cervical specimens collected in ThinPrep LBC vials for HR-HPV DNA testing with the Abbott RealTime High Risk HPV assay, which has been validated in both the triage of the referral population and primary cervical cancer screening6–8 to assess the impact of automated versus manual preanalytics on assay performance and ease of use.
Excellent agreement (95.6%; 95% CI, 92.0%–97.7%; κ = 0.907) of the RealTime HPV assay resulted from automatically preprocessed ThinPrep LBC samples (Tecan 1) and archived data obtained following the manual preanalytical procedure (manual 1). Our data compared well with intralaboratory reproducibility results previously published by Poljak et al. 9 (κ = 1.00) and Hesselink et al. 6 (κ = 0.96) and observed after manual preprocessing of ThinPrep samples.
Retesting of the 99 selected samples with both the automatic and the manual procedure, with the latter carried out after careful mixing of the specimens in compliance with the manufacturer’s instructions, improved the correlation between the two preanalytical protocols from both a qualitative and a quantitative point of view (i.e., no discordant samples for the final RealTime HPV assay interpretation and R2 = 0.958 for the CNIC, respectively), leading to almost identical performance of the preprocessing methods. In addition, the CNIC/HPV analysis revealed that the Tecan preanalytical processing of LBC samples improved interassay reproducibility.
Moreover, the 100% overall concordance of interpretation and the acceptable CVs for the IC (<3%) and of HPVs (<9%) obtained in the intra-assay reproducibility analysis allows us to state that the use of the Tecan system does not affect the final clinical interpretation and does not alter in any way the robust analytical performances of the RealTime HPV assay. 10
The main advantages of automated preprocessing of ThinPrep LBC samples with the custom-configured worktable setup of the Tecan Freedom EVO evaluated in this study are (1) reduction of hands-on time spent on preanalytic processing, (2) minimizing operator exposure to infectious pathogens, (3) significant reduction of the risk of sample misplacement (complete sample tracking), and (4) process documentation.
The Tecan instrument can preprocess 96 ThinPrep LBC samples within 1.45 h, if the vortexing time is set at 15 s, and its turnaround time is unlikely to be exceeded by manual operation. However, the significant advantage provided by the use of the automated preanalytic processing of LBC specimens is the saving of hands-on time of about 1 h and 30 min per run. Moreover, monitoring the initial 15- to 20-s vortexing step of the RealTime HPV assay protocol prior to sample dispensation may pose a challenge to the laboratory personnel, particularly when processing large batches of samples in a high-throughput setting, which will occur with implementation of primary HR-HPV DNA screening in the future. In addition, the Tecan system’s ability to identify and match barcodes of LBC vials and secondary tubes helps to avoid possible human error in tracking the specimen identification along the diagnostic workflow, and a robust LIS (laboratory information system) integration for sample identification to downstream systems allows for rapid reporting and turnaround time of test results.
The reproducibility of HR-HPV DNA test results associated with the complete automation of the preanalytic processing is particularly desirable both in the context of primary screening, since they will trigger the entire downstream cascade, and in laboratories, such as ours, performing HR-HPV DNA testing in the management of abnormal cytological lesions and in the follow-up after treatment for CIN2/3. As reported by Poljak et al., 11 the common background rationale of all recommendations is for all HPV tests used in clinical practice (screening and diagnosis) to be held to very similar clinical performance standards (clinically validated), with little, then, to distinguish them in terms of ease of use, automation, and high-throughput capacity. Moreover, the availability of many commercial HPV-DNA tests clinically validated and their adaptation to automated platforms (both preanalytic and analytic) will hopefully lead to high reproducibility of the results, shortening of hands-on time, and a significant price reduction in the next few years. The Tecan Freedom EVO in combination with the Abbott RealTime High Risk HPV assay seems to respond efficiently to all these critical points.
In conclusion, the new custom-configured version of the Tecan Freedom EVO robotic platform translates the manual steps required to preprocess ThinPrep LBC specimens prior to running the Abbott RealTime High Risk HPV assay into automated operations, controls sample identification matching between primary and secondary vials, and improves the efficiency of the laboratory, while ensuring reproducibility and reliability of test results, making it suitable for both the screening and the second-level laboratories.
Footnotes
Acknowledgements
The authors thank Abbott Molecular Italy for donating materials and consumables required to perform this study and Dr. Roberto Restivo from Abbott Molecular Italy for supplying technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
