Abstract
The need for predictive, in vitro cardiac safety screening drives further development of automated, high-throughput–compatible drug evaluation based on cardiac cell preparations. Recently, pluripotent stem cells are evaluated as a new, more predictive model for cardiovascular risk assessment pertaining to in vitro assays. We present a new screening platform, the CardioExcyte 96, a hybrid instrument that combines impedance (cell contractility) with extracellular field potential (EFP) recordings. The electrophysiological measurements are noninvasive, label free and have a temporal resolution of 1 ms. This hybrid technology addresses the lack of easy-to-use high-throughput screening for in vitro assays and permits the reliable investigation of short- and long-term pharmacological effects. Several models of cardiomyocyte preparations were successfully validated for use with the CardioExcyte96. Furthermore, the pharmacological effects of a number of reference compounds were evaluated. Compound effects on cell monolayers of human-induced pluripotent stem cell–derived cardiomyocytes are evaluated using a quasi-simultaneous hybrid recording mode that combines impedance and EFP readouts. A specialized software package for rapid data handling and real-time analysis was developed, which allows for comprehensive investigation of the cellular beat signal. Combining impedance readouts of cell contractility and EFP (microelectrode array–like) recordings, the system opens up new possibilities in the field of in vitro cardiac safety assessment.
Keywords
Introduction
A New Approach to Cardiac Safety Screening
Cardiac safety assessment is a vital part of drug development because late withdrawals of compound candidates due to heart liability issues are very costly. Therefore, predictive and cost-efficient cardiotoxicity assays are strongly demanded by the pharmaceutical industry.1,2
Until lately, label-free large-scale screening platforms for cost-effective cardiovascular risk assessment were not available on the market. The CardioExcyte 96 is a new screening platform allowing reliable high-throughput label-free measurements of short- and long-term compound effects for drug safety screenings, contractility assays, and functional investigations of electrical activity of cell monolayers and their developments. This hybrid instrument combines impedance readout (a correlate of cell contractility) with extracellular field potential (EFP) recordings generated by cellular action potentials (their duration was previously correlated to a QT interval in an electrocardiogram). The EFP recording mode provides electrical signals equivalent to microelectrode array (MEA)–like arrangements.
The industry is currently starting to use induced pluripotent stem cells for functional assays to identify cardiac pathologies and their genetic underpinnings (such as long QT syndrome), in addition to cardiotoxicity profiling of pharmaceutical compounds. 3 As stem cell–derived cardiomyocytes are readily accessible compared with freshly isolated cardiomyocytes and at the same time the assay is scalable, this opens the way for the pharmaceutical industry to perform high-throughput screening of new drug candidates.4,5
Several models of in vitro cardiomyocyte preparations were successfully validated for use with the CardioExcyte 96, such as stem cell–derived cardiomyocytes from Axiogenesis (human: Cor4U, murine: CorAt), CDI (iCell), GE Healthcare (Cytiva), Cellectis (human, three-dimensional [3D] clusters: hES-CMC), and ReproCELL (ReproCardio 2). Human-induced pluripotent stem (iPS) cell–derived cardiomyocytes (Cor.4U, Axiogenesis AG, Cologne, Germany; iCell Cardio-myocytes, Cellular Dynamics International [CDI], Madison, WI) were used to compare the effect of well-known compounds on the contractility and extracellular potential of cardiomyocytes.
Basics and Comparison of Impedance and Cardiac Field Potential Assays
Two of the most promising approaches to high-throughput in vitro cardiac safety screening are EFP- and impedance-based cell-monitoring systems.
Recordings of the EFP are most often performed by means of planar MEAs. A typical MEA is composed of several dot electrodes in the dimension of tens of micrometers arranged in a two-dimensional grid, which are overgrown by a functional cell layer. With respect to a reference electrode displaced from the grid, microelectrodes capture changes in the extracellular potential, generated by the electrophysiological processes inside the attached cells. Within the scope of cardiac preparations, MEAs can be used to monitor the electrical activity of spontaneously beating cardiomyocytes. Therefore, EFP recordings present an indirect image of the action potentials generated by cells, allowing noninvasive monitoring of intact networks of cardiomyocytes.6–8
In contrast to this technology, the approach of using impedance as a readout is focused on the mechanical movement of the spontaneously contracting cells. Spectral impedance analysis of biological samples is well established. Impedance spectra recorded from cell-covered electrode pairs have been routinely used to monitor for cell adhesion, growth, proliferation, or cell death, for example. 9
In the field of cardiovascular risk assessment, here, a new application of this technology is introduced. The hybrid instrument CardioExcyte 96 combines impedance readout, which is a correlate of cell contractility, with EFP recordings generated by cellular action potentials. Fixed-frequency impedance readouts with high temporal resolution can be used not only to depict adhesion of cardiomyocytes but also to display the change in morphology of contracting networks of such cells. In this manner, an impedance-based cell assay can be used to investigate effects on the mechanical beating activity of in vitro experiments.10–12
Both methods, MEA and impedance, have in common the label-free and noninvasive principle of measurement, which allows experiments over an extended period of time and the use of various kinds of cardiac cell samples (cell lines, stem cell–derived, primary cells/tissues) without laborious preparation. Besides, both methods are working with intact networks of interconnected cardiac cells and can therefore yield results based on samples that, in contrast to single-cell measurements such as patch-clamp readouts, come close to a physiological cardiac tissue. One difference between both readout methods is the level at which the acquisition of the cells’ beating activity takes place. Although the impedance readout captures the most essential function of the cardiomyocytes, the mechanical contraction, the readout of extracellular voltage changes displays the underlying electrophysiological process. The advantage of EFP measurements is that the results are directly linked to the electrophysiological origins of the contractile activity and allow for a more detailed investigation of effects resulting from ion channel–modulating compounds. Measurements feature a temporal resolution of 1 ms (impedance), respectively 0.1 ms (EFP).
Materials and Methods
The CardioExcyte 96 Instrument
A complete CardioExcyte 96 setup consists of three parts: the main recording unit containing all electronics situated inside an incubator, one or multiple cell-populated CardioExcyte 96 sensor plates, and a personal computer running the CardioExcyteControl software (refer to Fig. 1 ). The main unit encapsulates the digital interface as well as the analog signal processing electronics so that it is well protected from the environmental conditions inside an incubator and disinfection treatment. A USB data link connects the main unit to the computer, allowing the recorded data to be transferred in an all-digital mode, which provides stable, distortion-free measurements. The connection between the CardioExcyte 96 and the sensor plate is ensured by a lever-operated mounting mechanism, which is easily locked and released. The user can operate plate mounting and electrical connections single handed; furthermore, all required cables fit easily through sealed doors or in dedicated orifices of the incubator. The compact housing and the comfortable plate mounting facilitate regular tasks in sterile environments as compound additions or culture medium exchanges. This, in conjunction with an intelligent power management, makes the CardioExcyte 96 well adapted to the needs of cell culture laboratories.
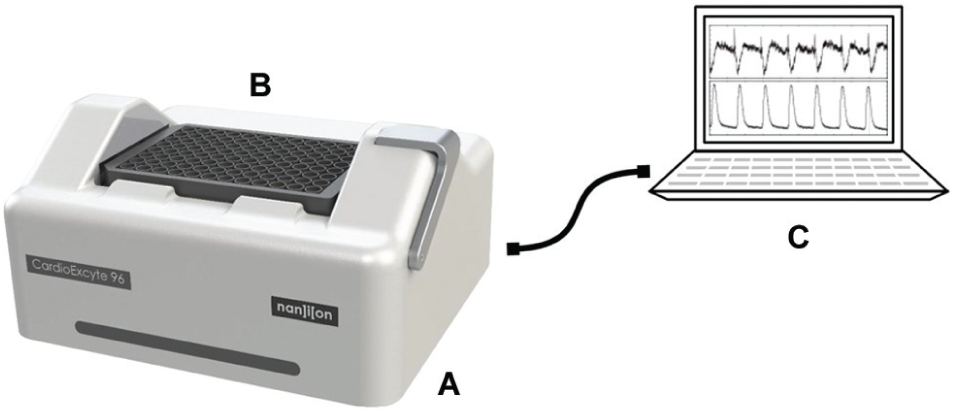
The CardioExcyte 96 setup. The CardioExcyte 96 (
The CardioExcyte 96 Sensor Plates
The CardioExcyte 96 consumables (sensor plates) are industrial standard 96-well microtiter plates with a circular gold sensor electrode on the bottom of each well. The culture chambers of the CardioExcyte 96 sensor plate feature biocompatibility and sustained sensor stability, thereby offering ideal conditions for long-term experiments. The bottom of the well plate is semitransparent and allows observation and verification of the cultured cells by use of standard transmission light microscopy. The embedded planar gold electrodes are designed as a combination of ring and dot electrodes. The circular sensing electrode has a diameter of 2 mm and is surrounded by a reference electrode near the outer wall of the cell cavity ( Fig. 2A ). With this electrode design, the CardioExcyte 96 is capable of recording high-quality impedance readouts from cardiomyocyte monolayers but also covers nonstandard experiments as, for instance, recordings from 3D stem cell clusters. The same CardioExcyte 96 sensor plate can be used for EFP-based and impedance-EFP combined experiments. In the impedance-EFP combined mode, the same biological process is mapped using two complementary electrical parameters, revealing the correlation between cause and effect of cardiac cell activity ( Fig. 2B , exemplary impedance and EFP trace of a single beat).
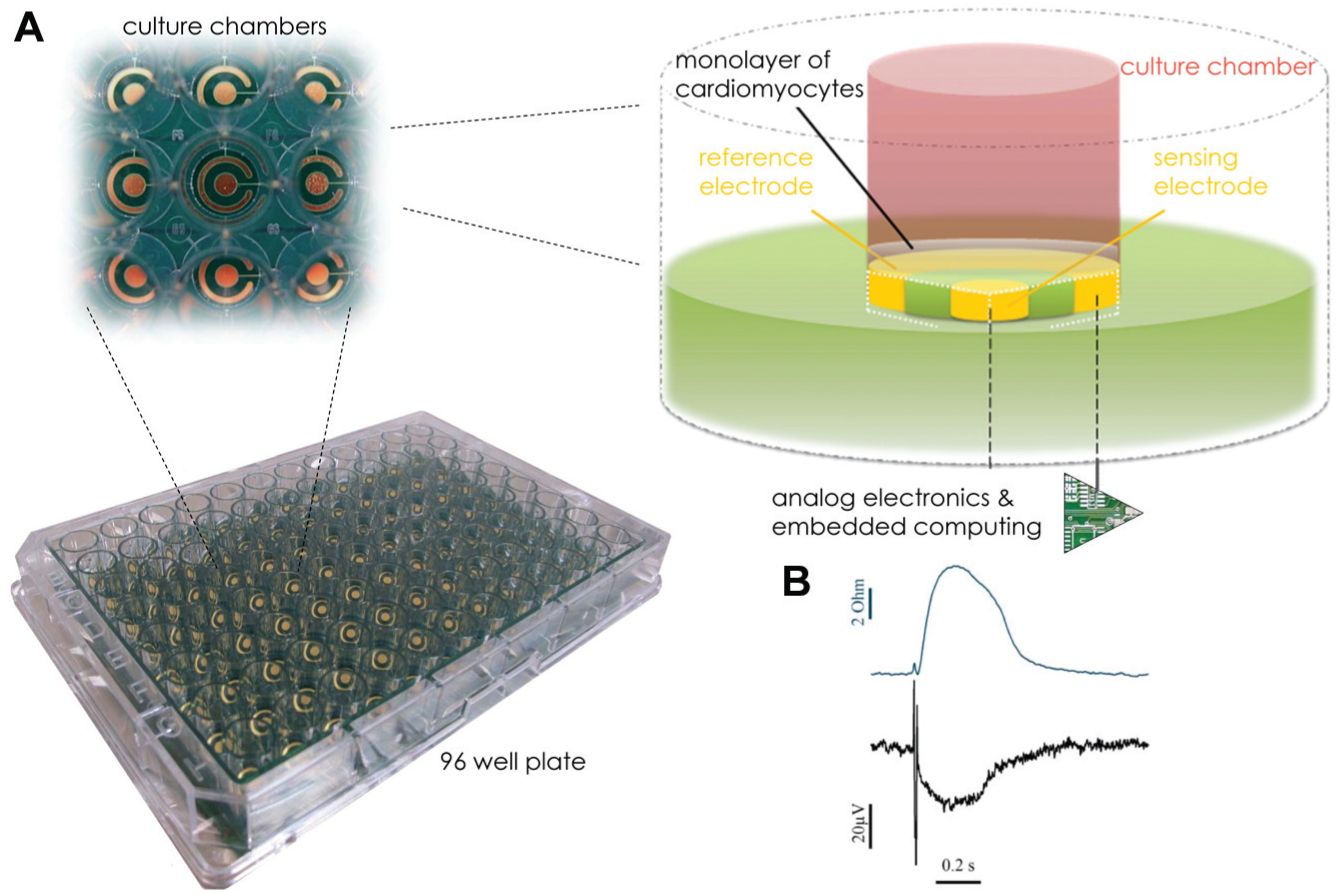
The CardioExcyte 96 sensor plate. A single electrode design is illustrated (
Principle of Impedance Detection
The ability of the CardioExcyte 96 to record and analyze cardiomyocyte activity in real time is based on a fixed-frequency impedance calculation with an outstanding fast sampling rate of 1 ms. With each step of data acquisition, 96 high-resolution impedance values are recorded and transferred to the connected computer. The measured impedance of the employed planar gold electrodes is mainly modulated by the changes in adhesion and morphology of the attached cardiac cells. The quality of adhesion, the number of intact cells, and the density of the cell layer determine the steady component of the impedance signal. On the other hand, the periodic contractile activity of the cells modulates the recorded signal and provides information about the functional condition of the cardiomyocytes. Therefore, the impedance mode on the CardioExcyte 96 is a powerful tool for the evaluation of cardiomyocyte preparations (cell lines, stem cell derived, or primary) and pharmacological cardiac safety assessment. On the CardioExcyte 96, impedance values are determined by imposing a small excitation voltage between the sensing and reference electrode and picking up the resulting current. More details regarding the origin of the impedance signal are depicted in Figure 3B , in which a schematic drawing of a cell monolayer growing on a CardioExcyte 96 sensor plate and the supposable paths of current are illustrated. The first model exemplifies a horizontal current pathway along the attached cell monolayer, passing the cell-to-cell junctions. The second model discloses a vertical current path either across the cell layer passing the membrane or through the intermediate spaces. In this case, most of the current conducts through the electrolytic medium. Depending on the recording frequency and the cell network morphology, both current paths are contributing to the impedance readout. Both current paths between the sensing and reference electrode are to a certain extent determined by an electrical capacitance and resistance caused by the adhering monolayer, which in turn are modified by the changes in cell morphology as part of the cardiac contractile activity.
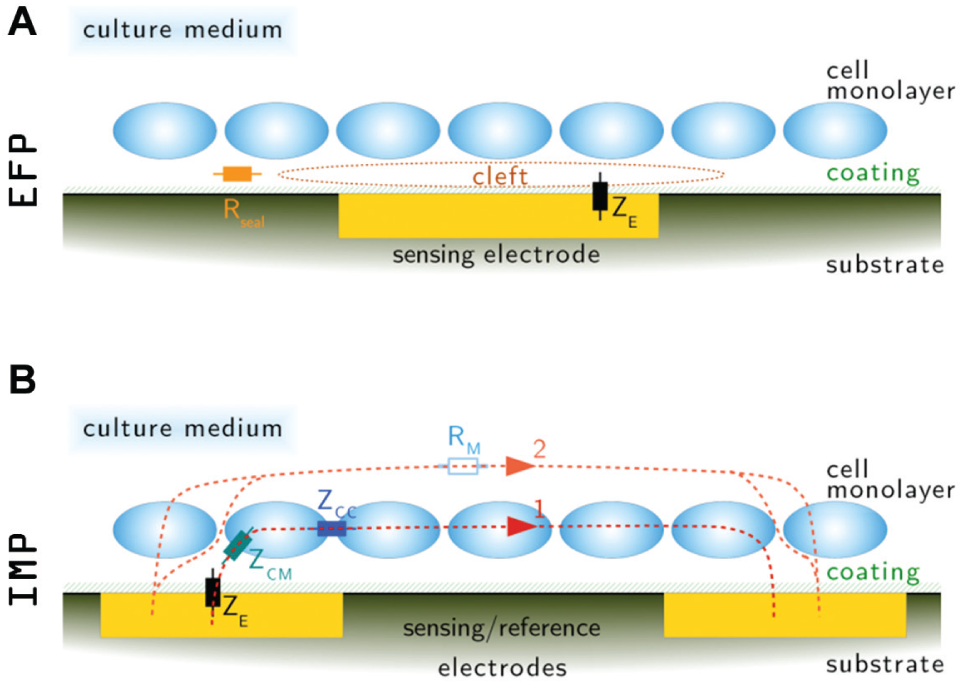
Principle of the impedance and extracellular field potential (EFP) recording mode. The schematic illustration depicts the situation on the sensor surface after the formation of a well-connected cell monolayer (
In accordance with this model concept, the contraction and relaxation of the cardiomyocyte layer modulates the impedance base line and allows for the detection of beating parameters.
Principle of EFP Detection
Measuring the level of variations in the EFP generated by the spontaneously active cell monolayer serves as a perfect complement to the impedance assay. In the EFP mode, the CardioExcyte 96 samples the extracellular signal from each of the 96 wells/electrodes, similar to a system using microelectrode arrays. In contrast to the MEA technique with several micrometer-scale electrodes in each measurement well, the CardioExcyte 96 operates with only one sensing electrode. The principle of measurement is illustrated in Figure 3A . For monitoring high-resolution and low-noise EFP signals, a theoretically noninterrupted cell monolayer has to be plated in the wells of the CardioExcyte 96 sensor plate, so that the cleft between the electrode and the monolayer is separated from the remaining medium by a high sealing resistance. During synchronized electrical activity of the monolayer, ions released by the cells are accumulating in the cleft, thereby changing its potential referred to the surrounding medium, which is set to a zero potential by the reference electrode. The charge buildup in the cleft is released through the seal resistance, but because of its relatively high value, a voltage can be detected between the sensing electrode below the excited cell layer and the reference electrode directly conducting the medium. Because of the relatively large surface area of the sensing electrode, this single-electrode design represents a new approach to extracellular recordings especially adapted to well-connected and synchronized cardiomyocyte preparations. The large electrode with its integrating characteristic sums up the extracellular signals from a greater number of cells and provides the user with an EFP signal representing the average electrical activity of the cells in the respective well. Lacking the spatial resolution and the clearly defined recording position, this approach has the advantage of a more uniform signal shape among the wells and an easier evaluation routine, because a selection of an optimal electrode for each well is not necessary. The single-electrode approach drastically facilitates multiwell assays because the evaluation is based on exactly one conclusive signal per well, so that the elaborate procedure of electrode selection is rendered unnecessary.
The CardioExcyte 96 enables the user to decide at any time during the experiment which readout delivers more significant information and to choose which data set is to be used for succeeding evaluations. The combined mode of measurements reduces the amount of cells needed for a hybrid experiment because the same preparation can be used for both types of recording. Besides that, the system greatly simplifies such experiments because no user interaction is required while the device captures the hybrid data.
CardioExcyte 96 Sensor Plate Preparation and Cell Seeding
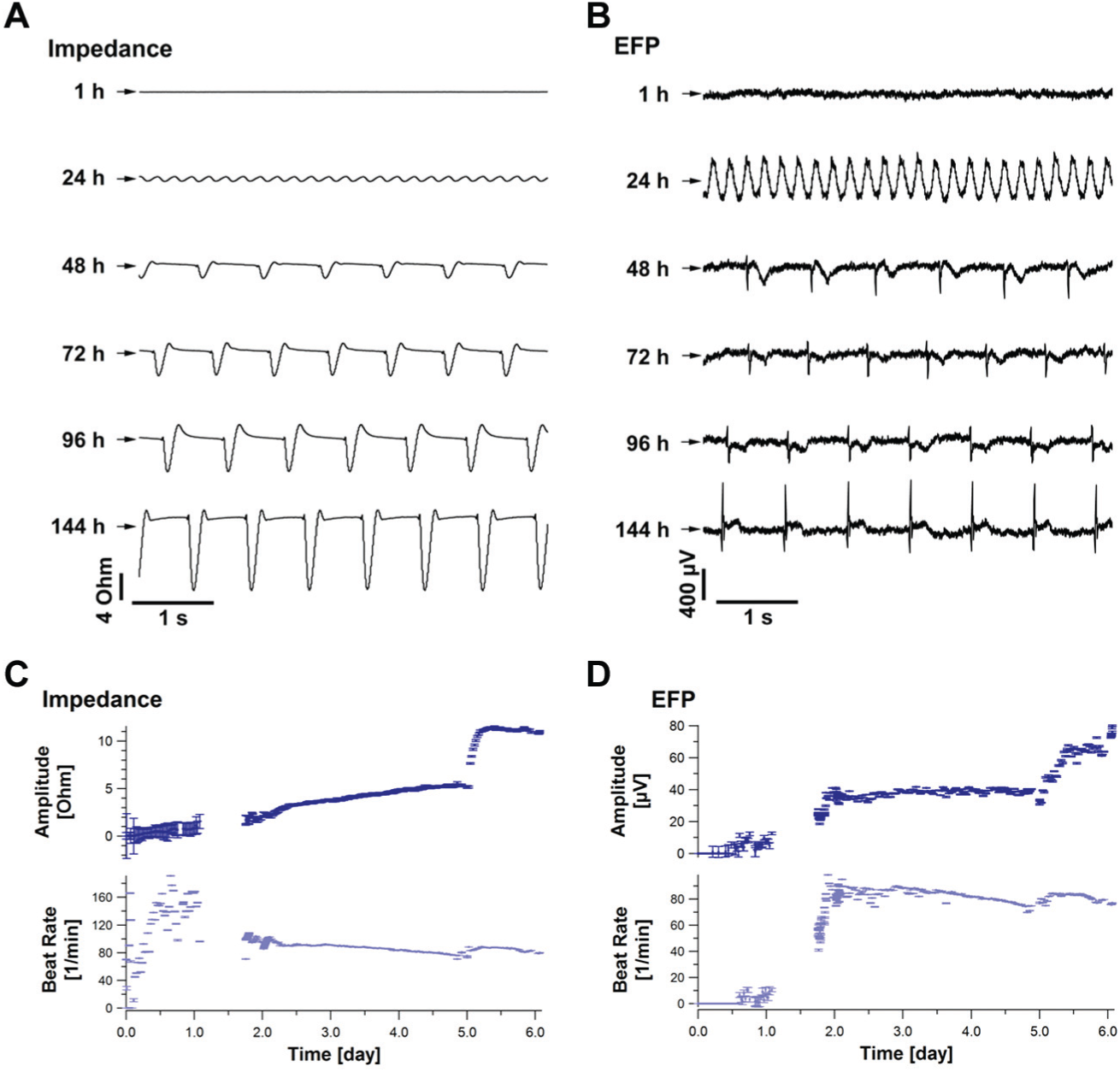
The CardioExcyte 96 enables cost-efficient safety pharmacology and allows investigations of a variety of stem cell–derived cardiomyocytes, 3D clusters, and primary cells. Therefore, it is readdressing the lack of easy-to-use in vitro assays and complements patch-clamp examinations. Several in vitro models were evaluated on the CardioExcyte 96: iPS cells (e.g. iCell Cardiomyocytes, Cor.4U), 13 embryonic stem cells from human (e.g., Cytiva, hESC-CM) 7 and murine (e.g., Cor.AT), cardiac muscle cells (e.g., HL-1), freshly isolated cardiomyocytes (e.g., from rats), 3D cell cluster (e.g., Cellectis, hES-CMC), 14 and ReproCELL (ReproCardio 2). Example data with typical beating patterns of the different cell models are shown in Figure 4 .
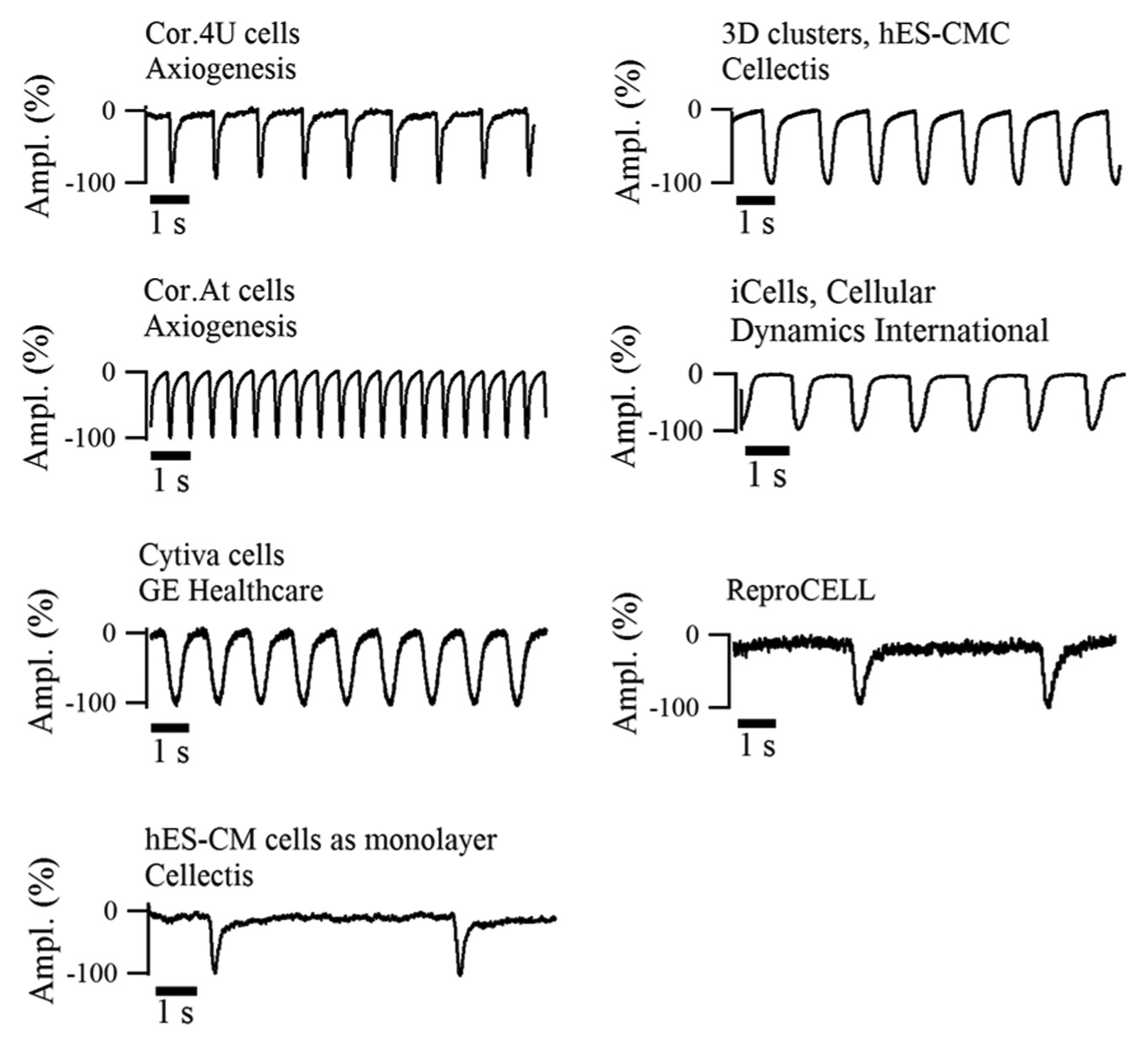
Beating pattern of stem cell–derived cardiomyocytes. Typical beating patterns of well-established stem cell–derived cardiomyocytes are displayed with an impedance sampling rate of 1 ms (sweep length, 10 s).
Frozen cells from nitrogen or cells that were already in culture can be seeded on a CardioExcyte 96 sensor plate. Depending on the number of cells per well, the cells are able to generate a network for a synchronized beating pattern. For most cell models, a cell density of 20,000 cells/well was found to be optimal.
The cell density was tested by seeding at least 10,000 cells to a maximum of 70,000 per well. A cell density of a minimum 20,000 cells proved to be optimal (for Cor.4U and iCells cardiomyocytes), as this concentration produced a stable and large signal, with a good signal-to-noise (SNR) ratio. The beats were uniform without intermitting peaks, which can occur when there are nonsynchronized beating areas on the surface. Mainly, all tested iPS-derived cardiomyocytes also adhere well on noncoated wells, illustrating a good surface condition of the sensor.
The 96-well sensor plates of the CardioExcyte 96 were coated with fibronectin (Sigma Aldrich, St. Louis, MO; 1:100 solution in phosphate-buffered saline without Ca2+/MG2+) for 1.5 h inside the incubator (36.3 °C; 5.0% CO2). A cell suspension of 300 viable cells/µL was prepared mainly out of thawed iPS cells cardiomyocytes (e.g., Cor.4U, iCells). After the fibronectin solution was removed, 100 µL prewarmed medium (37 °C) (e.g., Cor.4U culture medium, iCells plating medium) was filled inside each well, and the cell suspension was added (e.g., 100 µL/well in Cor.4U medium, iCells plating medium). The plate was transferred into the incubator (36.3 °C; 5.0% CO2), and cells were not touched or moved for 48 h. After this time, a medium change was made every 2 to 3 d (e.g., Cor.4U culture medium, iCell maintenance medium).
Operation
Impedance or field potential readouts provide information about the condition of the cells, and contractile activity can be conducted as soon as a prepared CardioExcyte 96 sensor plate is attached to the device. An experiment on the CardioExcyte 96 system consists of consecutive recording periods of configurable length, referred to as sweeps. Sweeps are automatically repeated according to a user-defined repetition interval and directly displayed in the CardioExcyteControl software. During an experiment, the repetition interval can be adjusted to meet the requirements of different phases of a long-term experiment. This method of operation ensures that fast changes in the condition of the cell monolayer can be recorded with sufficient accuracy while the overall amount of data is kept at a reasonable level.
Experimental Scheduling and Drug Application
An experimental schedule is illustrated in Figure 5 that shows the time course for the entire process, starting from seeding cells, over cell adhesion, to the beginning of the cell contraction, reaching synchronized beating, performing the compound addition until the end of an experiment, followed by the data analysis. The circles indicate the repeated workflow during cell culture maintenance and the succesive compound addition. Whereas impedance measurements can be conducted at 3 to 5 d after seeding, EFP recordings might require a longer time period of up to 10 d, because a proper network of cell-cell interactions and a dense monolayer need to be established for a steady propagation of the excitation and synchronized beating (see also Figure 6 ).
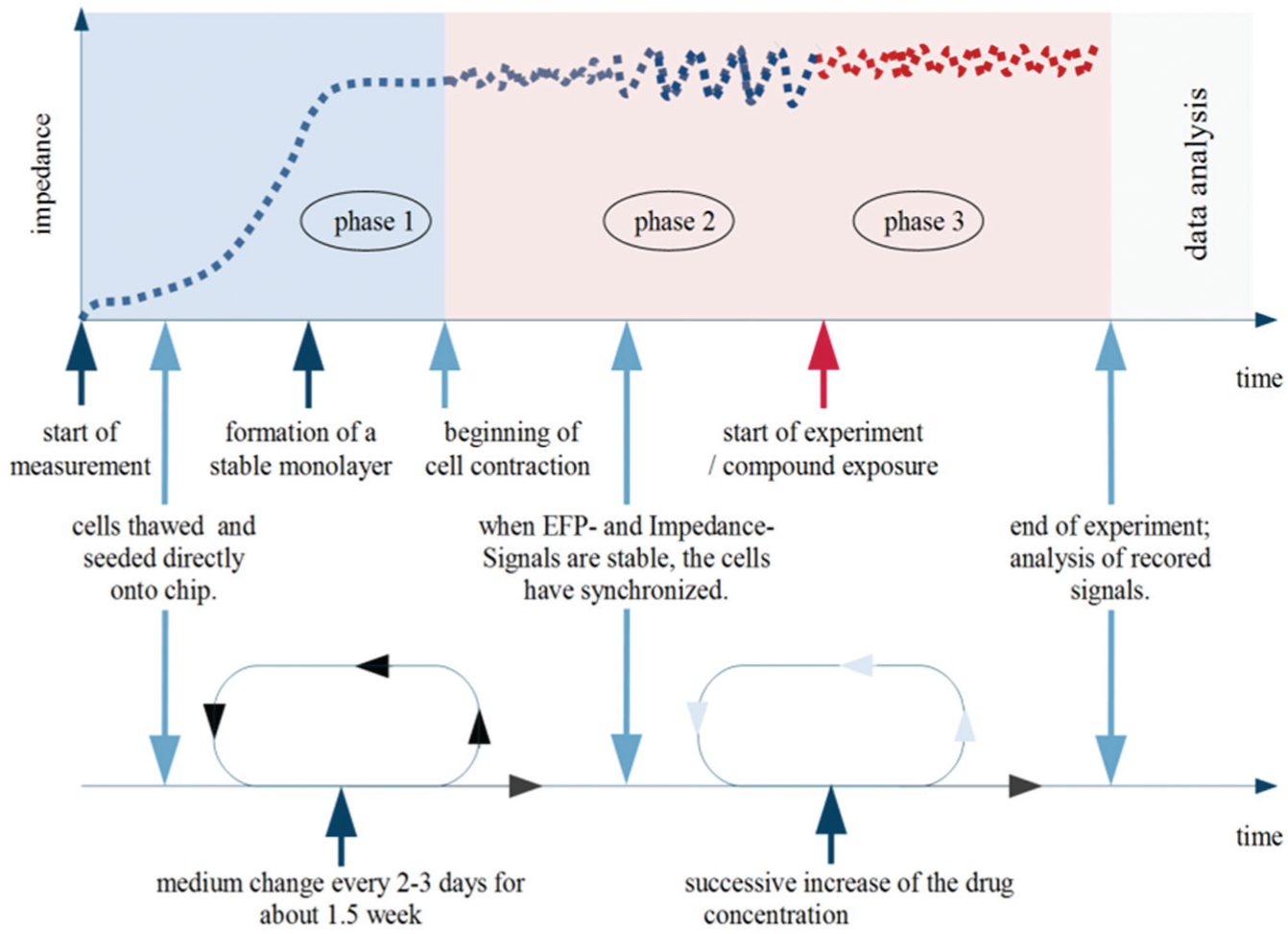
Experimental time course of the impedance signal. The impedance signal rises after seeding a monolayer of cardiomyocytes according to the increasing cell adherence (phase 1). The impedance reaches a plateau phase at which the complete monolayer adhered. In phase 2, the cell monolayer starts to execute sporadic contractions, leading to a modulation of the electrode’s base impedance. These small differential impedance changes are recorded by the CardioExcyte 96 in high temporal and absolute resolution. When the complete monolayer is beating in a synchronized manner, compounds can be applied and the real experiment can be started (phase 3).
Time-dependent development of the impedance and extracellular field potential (EFP) during cell adhesion. Representative traces of the impedance and EFP signal are presented (
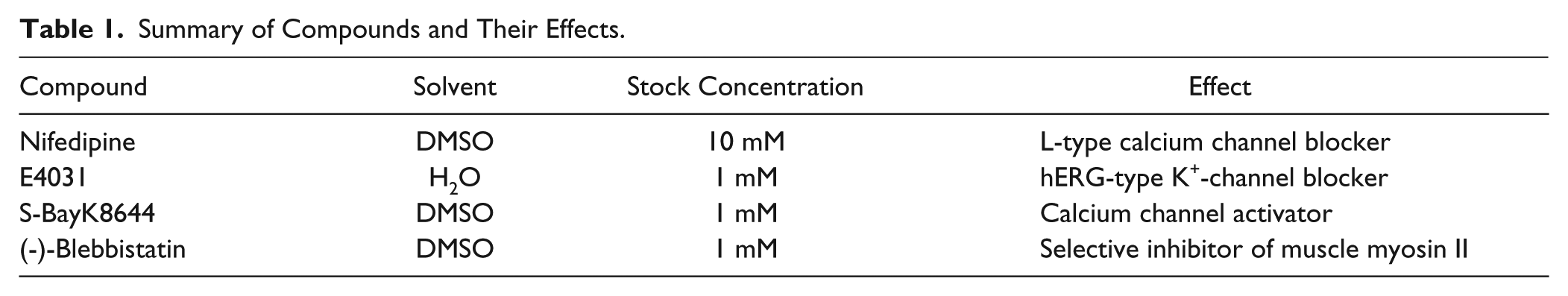
Several drug-induced effects have been investigated on the CardioExcyte 96. The effects of four compounds (
Table 1
) on the impedance and EFP signal are presented and compared. Drug applications were performed with successively increasing concentrations (0 nM, 3 nM, 10 nM, 30 nM, 100 nM, and 300 nM) for Nifedipine, E4031, and S-BayK8644, whereby a zero concentration (medium only) was added in the beginning to manifest a baseline. After the baseline equilibration, the compounds were added successively every 12 min, as illustrated in
Figure 5
. Data were collected in six wells, whereby two of them served as controls (DMSO 0.001%
Summary of Compounds and Their Effects.
Software
The CardioExcyte 96 system is accompanied by a powerful software package assisting the user with the preparation of experiments, management of acquired data, and the evaluation and export of experimental results. Prior to the start of a new measurement, the user can define the duration and repetition rate of the recordings and set up a library of compounds, which are to be applied throughout the experiment. To use the automated online analysis (OA) feature of the software CardioExcyteControl, the user can choose and configure several analysis parameters in the OA Editor ( Fig. 7 ), which are applied to the raw data right after recording and visualized in online analysis graphs progressing with the ongoing experiment. The software includes common OA parameters such as amplitude, beat rate, pulse width, and so forth. In addition, primary and secondary beats can be distinguished by defining a threshold. An overview and illustration of some of the parameters are presented in Figure 8B . Depending on whether EFP or impedance data are loaded, different parameters and analysis settings are available. A special feature of the CardioExcyte 96 software is the Mean Beat View, which automatically calculates a single beat shape based on the data of one sweep ( Fig. 8A ). This mean beat is, in addition, used for the calculation of the OA parameters. Instead of looking at the sequence of beats in the raw data trace, the mean beat allows a more descriptive and condensed view.
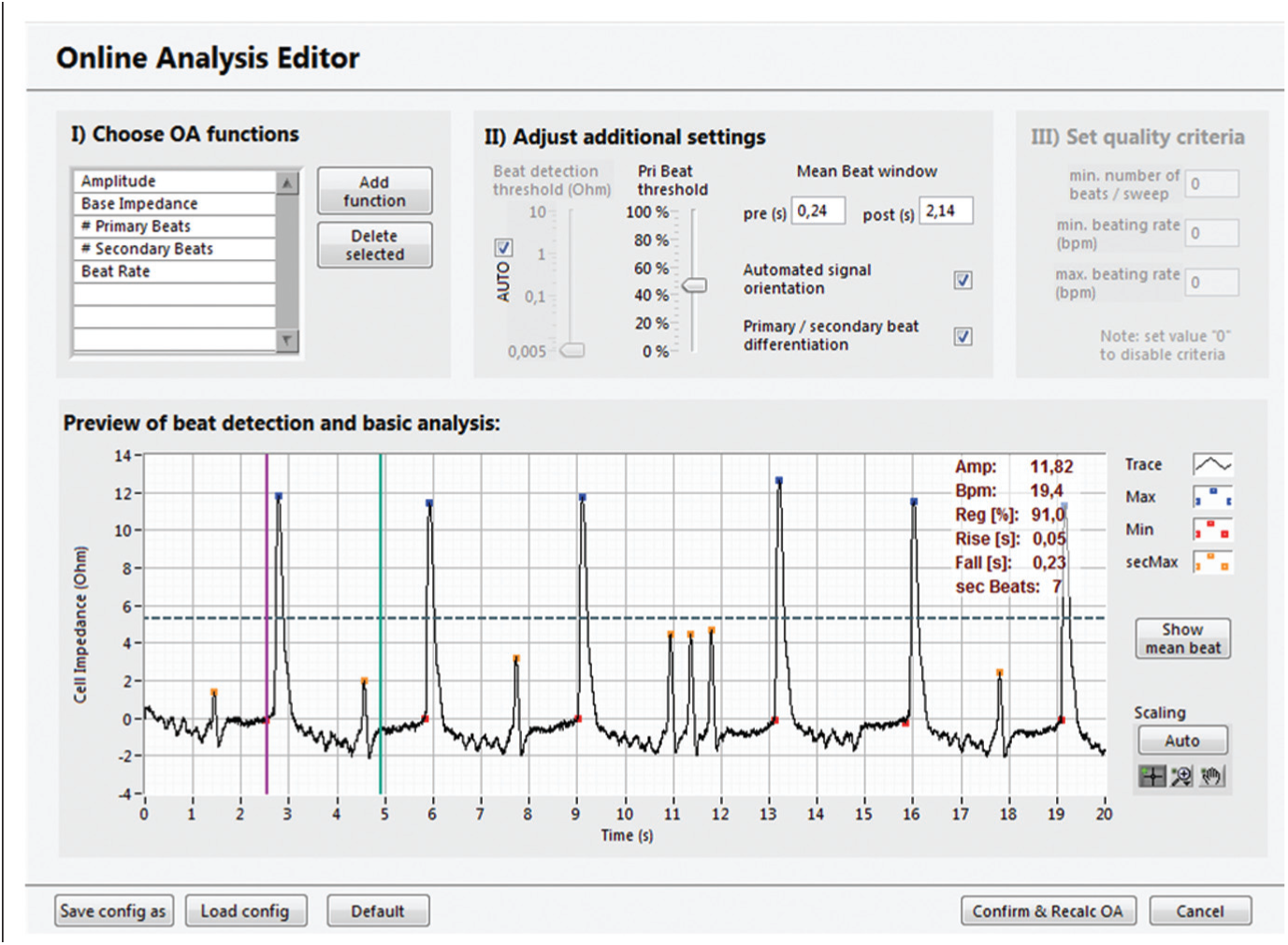
The Online Analysis Editor. The Online Analysis Editor allows the configuration of customizable data analysis. Prior to a new experiment, the user can chose which parameters are going to be available while the experiment is running. A finalized set of experimental data can be reanalyzed with new settings at any time. The software features versatile analysis parameters and an intelligent peak detection engine, with primary-secondary beat distinction.
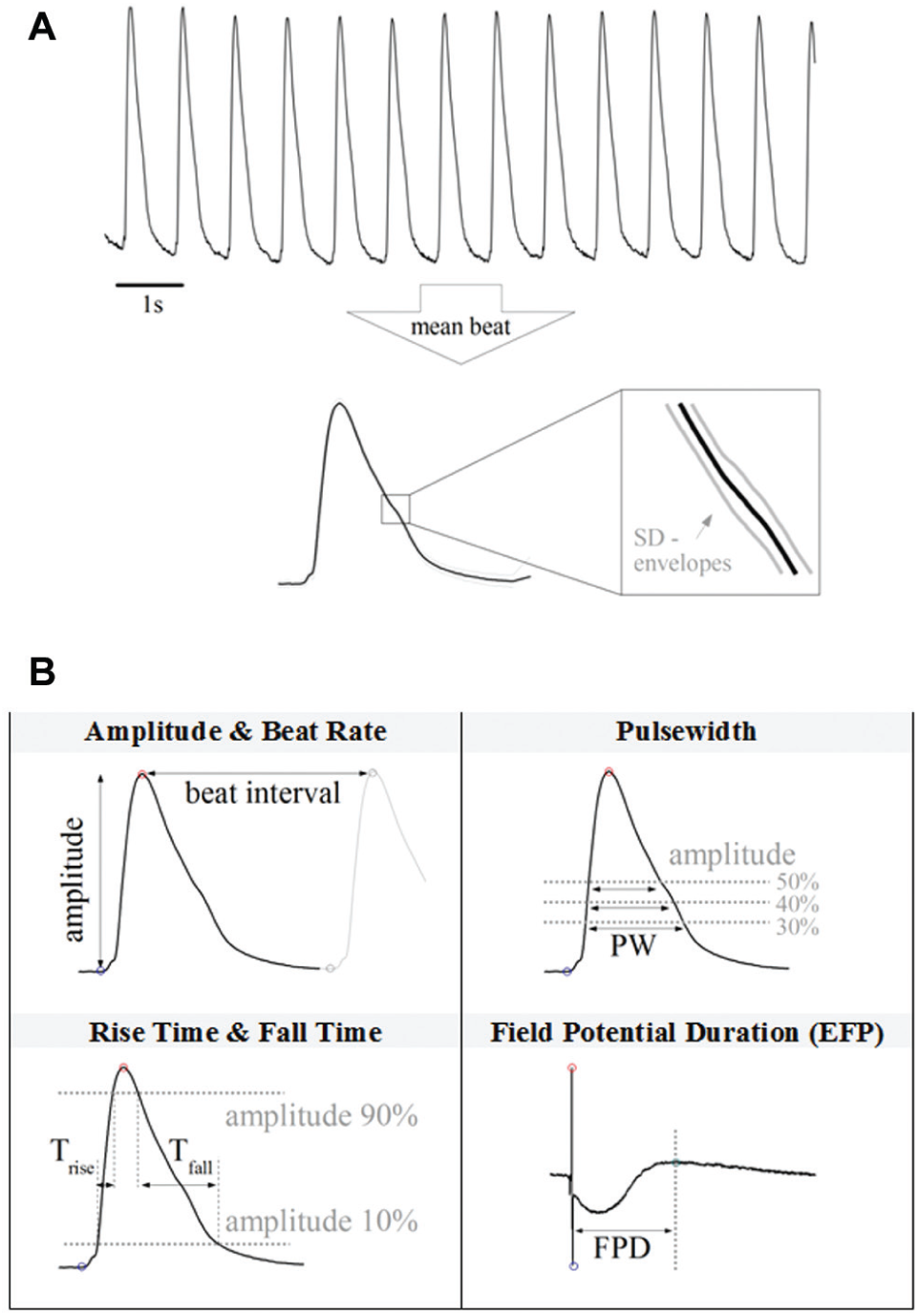
Online analysis (OA) parameter calculation. (
As assistance in experiments including addition of compounds, the software allows virtual additions of predefined substances with specific concentrations. Markers are added to the measurement file, which can then be used for tracking the additions and for compound- and concentration-related data evaluation. This also includes sophisticated IC50 analysis and calculation of dose-response curves.
During an active experiment as well as in replay mode (in which the user can navigate through previous recordings), the software offers great capabilities for keeping track of the cells’ condition on the entire well plate. The user can highlight 16 wells for an extended view and choose between raw data, single-beat, and OA graphs at any time ( Fig. 9 ).
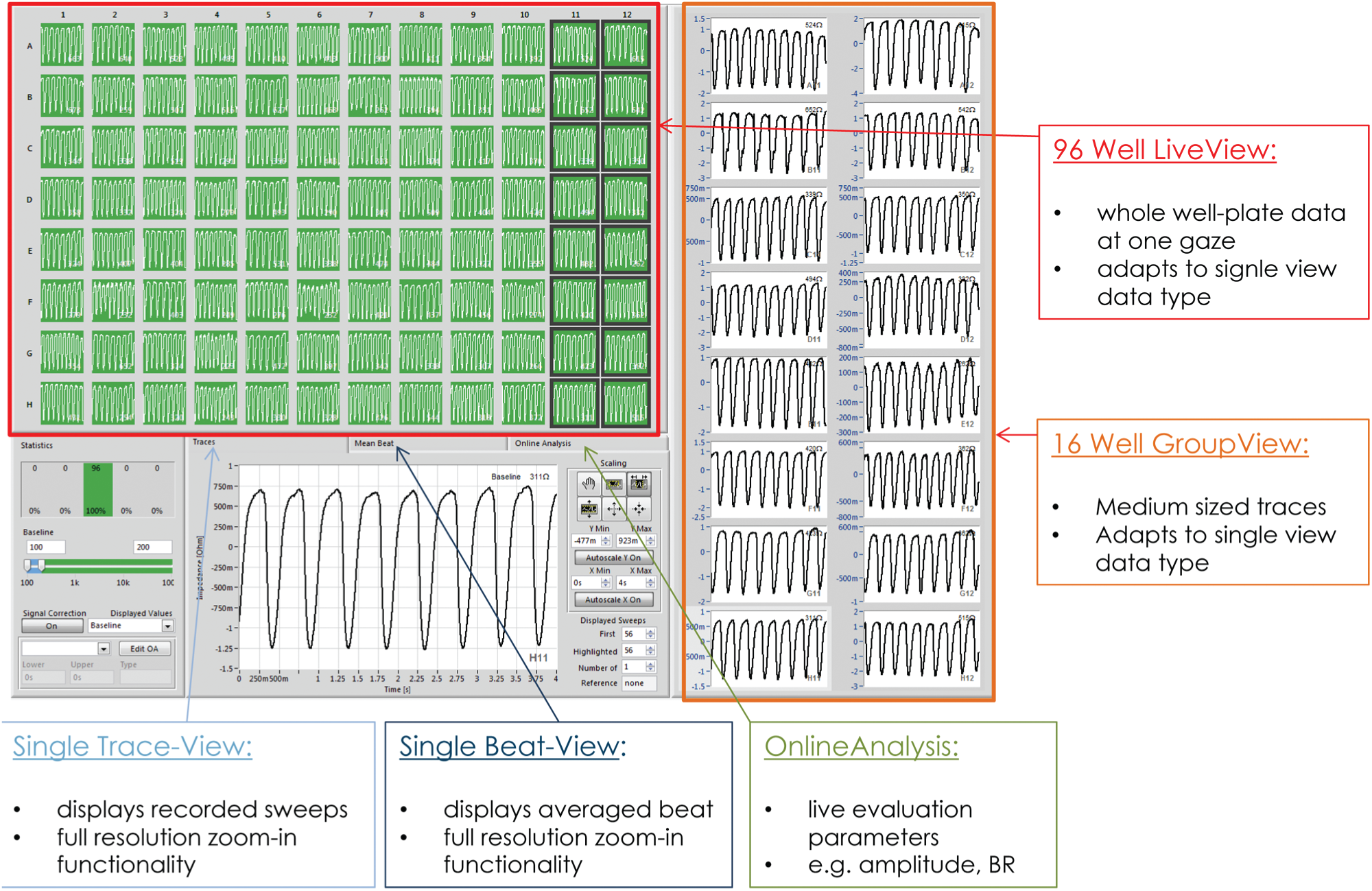
Screenshot of the software interface in replay mode. Various differently scaled graph objects provide the user with an overview of the cell activity on the entire sensor plate as well as a more detailed view of highlighted wells. Furthermore, the user can switch from a raw data view to a more condensed single-beat view.
Results
Cell Adhesion
Starting a recording directly after seeding a monolayer of cardiomyocytes, the cell adhesion process and the establishment of the cell network can be monitored online. A representative time course of the first 140 h is displayed in
Figure 10
. The base impedance rises from about 600 Ohm to 800 Ohm (+33%), reaching a stable level after about 80 h, according to the increasing adherence of the cardiomyocytes. Every medium exchange induced a transient increase in the base impedance signal caused by a small change in temperature and CO2 concentration (dashed lines in
Fig. 10
). These artifacts can be reduced if the plate is left on the CardioExcyte 96 and the complete device is moved under a sterile hood for a medium exchange. The development of the cell network can be monitored by the amplitude of the synchronized beating and the beat rate (
Figs. 6
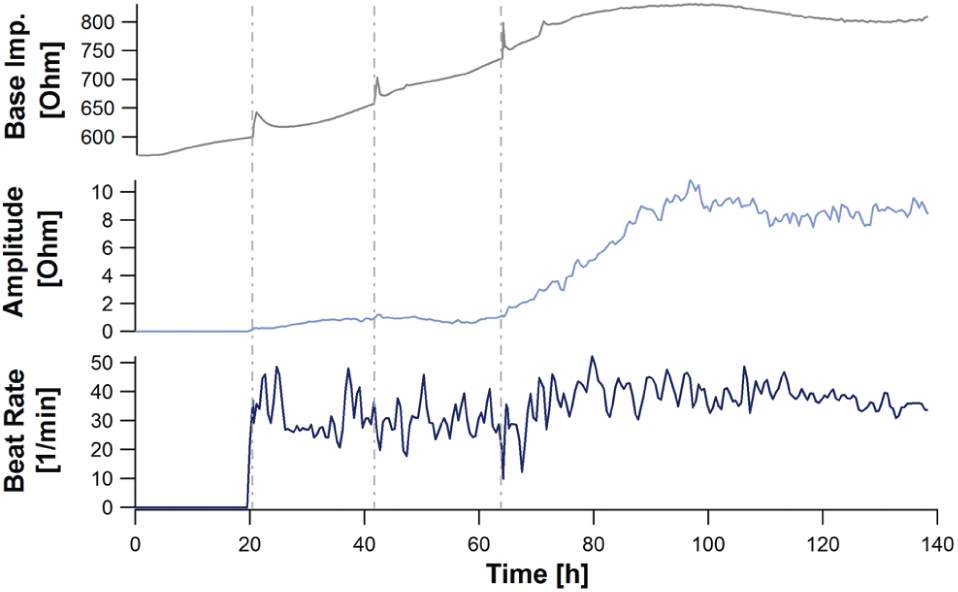
Representative time course of an impedance readout. The three major online analysis parameters are presented, Base impedance, amplitude, and beat rate during the attachment of cardiac cells (iCell cardiomyocytes; T = 0: cell thawing/seeding). Data are derived from 32 wells and are shown as an across-well average. Medium was changed at 22 h, 44 h, and 64 h after seeding (indicated by the dashed lines). The base impedance steadily increases during the formation of a cell monolayer. As soon as a critical quality of cell-cell interconnection is reached, the contractions of the cells can be captured by the beat rate analysis. The impedance amplitude rises while the pulsatile activity of the monolayer is synchronizing and reaches a plateau phase depending on the intensity of the contractions.
By observing more than one well, it is also possible to derive an interwell variation, which can give some indication of the quality of the cell model and the qualification for precise and reliable compound assays. At the end of the adhesion phase, the entire plate is showing uniform beating in every well (about 100 h after plating, ~4 d in culture). At this time, the cells are ready for an experiment.
Taking a closer look at the EFP beat pattern during the cell adhesion and network development, a switch in the orientation of the beat shape can be recognized ( Fig. 6C ). In the first days (while the cells are not synchronized), the beat shape seemed to point downward. The signal flipped upward and developed to its common shape, revealing typical characteristics of extracellular recordings, when the cells were fully adhered to the sensor plate surface. The signal flipping over time is an empirical finding and occurred as well in the impedance mode and was also seen by others (verbal notices) using similar technology. Impedance signals might point upward in the first days and flip downward when the monolayer is completely adhered (data not shown). After the impedance and EFP readouts reach a stable value, a firm signal is measured, and no further flipping occurs. Over time, the impedance and EFP signal sharpened and the SNR increased because the cell signals gained strength. The beat pattern measured in the EFP and impedance mode showed typical characteristics and a significant synchronicity after the cell network had been properly established ( Fig. 6A , C ). Exemplarily, the amplitudes and the beat rates of the 10 s sweeps, detected from the impedance ( Fig. 6A ) and the EFP ( Fig. 6C ) signal, were averaged over 12 wells (n = 12) and plotted versus time. Whereas a stable beat rate was already reached after about 2 d ( Fig. 6B , D ), the amplitude increased further on, reaching a steady-state value after about 5 d ( Fig. 6B , D ). The standard deviation of the 12 wells used for the interwell average decreased over time, indicating a stable condition throughout all cardiac preparations.
Compound Effects on Impedance and EFP
The CardioExcyte 96 software allows a free selection of control and compound measurements. The 96-well–based sensor plate permits several compound applications in one experiment. A good alignment for concentration-dependent compound analysis resulting in reliable data outputs is presented in Figure 11 . Six compounds, each with four different concentrations and a statistical population of three, can be easily analyzed per senor plate with one control well per concentration. The screenshot in Figure 11 illustrates nicely the effect of the added compounds on the beat rate and amplitude in a concentration-dependent manner.
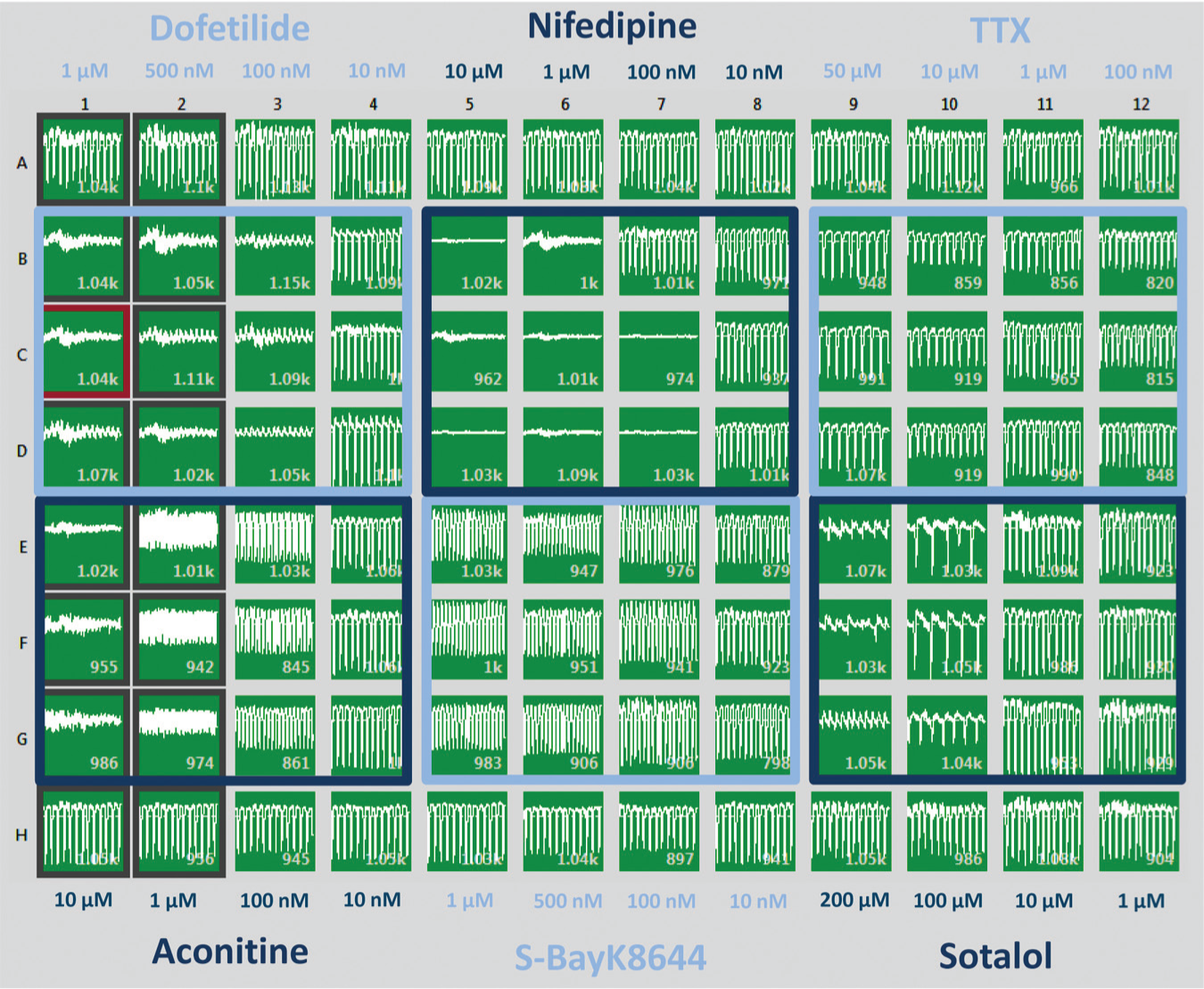
Ninety-six-well view of an experimental layout. Different concentrations of compounds were tested on Cor.4U cardiomyocytes. The compounds induced effects on the beat rate, the amplitude, and the beat irregularity (arrhythmia).
Nifedipine
Nifedipine is an L-type calcium channel blocker that leads to QT shortening. Its effect on the impedance and EFP signal is exemplarily shown in Figure 12 by a representative experiment.
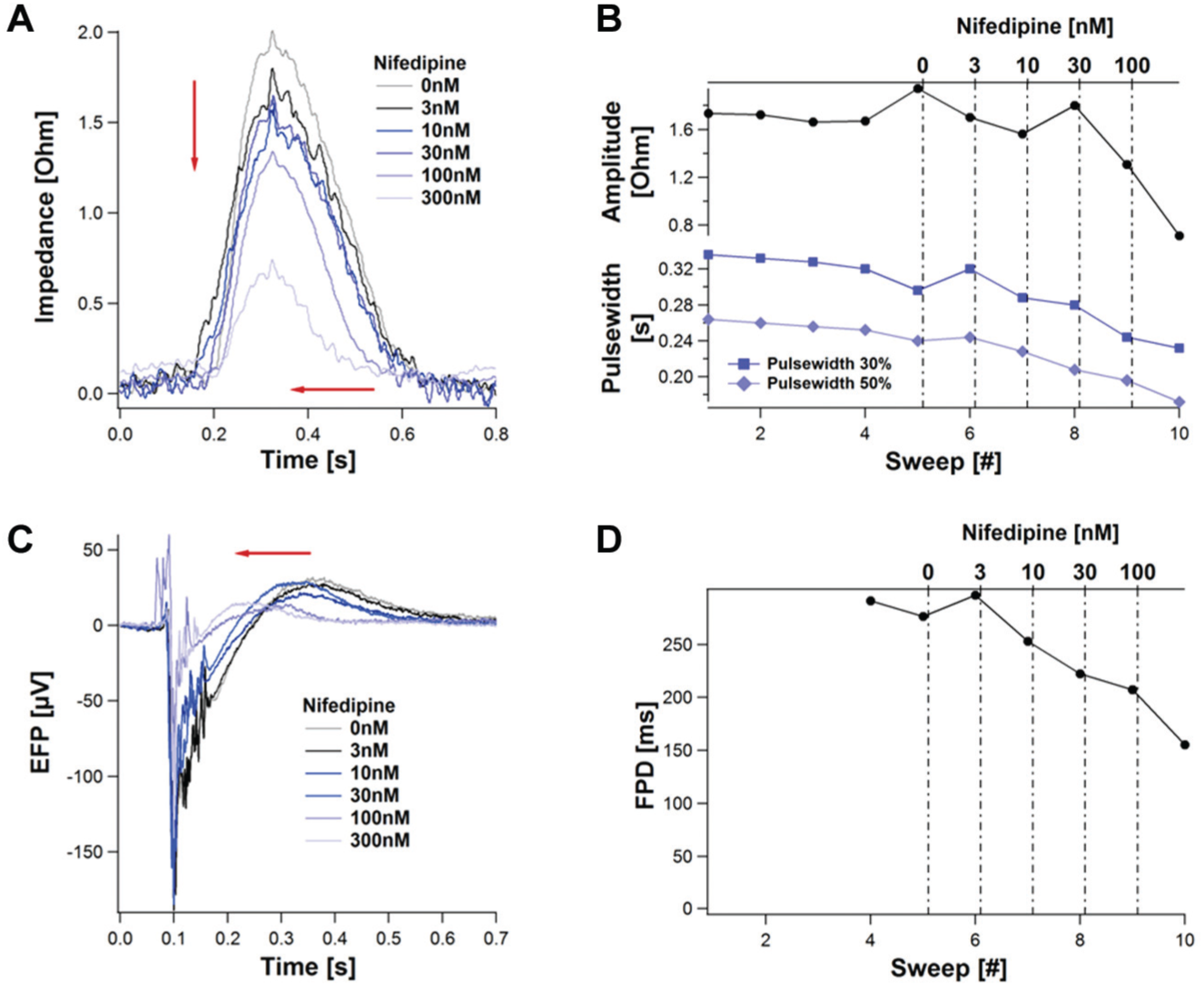
Dose-dependent effect of Nifedipine on the impedance and the extracellular field potential (EFP). Compounds were applied as indicated to a monolayer of 20,000 Cor.4U cells. The mean beats, derived from 10 s sweeps, are shown in (
In the impedance mode, the addition of increasing Nife–dipine concentrations induced a concentration-dependent decrease of the amplitude. In addition, the maximum is shifting in a dose-dependent manner to the left, which corresponds to a decreasing rise time. The decrease in the pulse width 50% is higher than the decrease in the pulse width 40%, leading to the assumption that the area under the curve is decreasing with a higher rate than the amplitude. This is similar to reports from other groups. 12
In the EFP mode, Nifedipine led to a dose-dependent shortening of the field potential duration (FDP), consistent with the effect on an action potential. This result is in good agreement with other reported data.7,15
E4031
The effect of the well-known hERG channel blocker E4031on the impedance and EFP signal is summarized in Figure 13 .
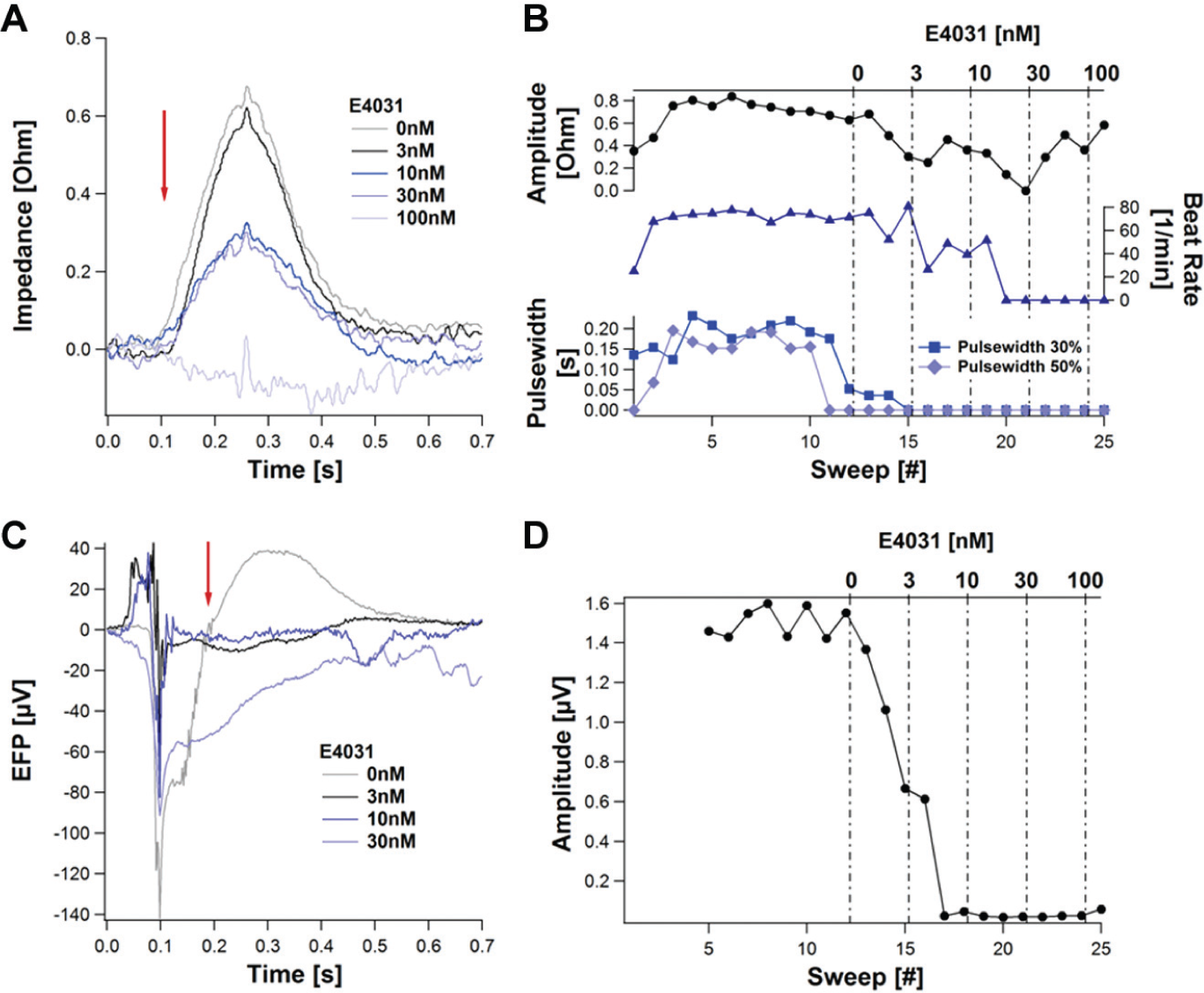
Dose-dependent influence of the hERG blocker E4031 on the impedance and extracellular field potential (EFP). A monolayer of 20,000 Cor.4U cells was seeded per well. Mean beats from 10 s sweeps are presented (
Although the impedance amplitude decreased dose dependently without any change in the rise time, the beat rate remained unaffected up to a concentration of 10 nM. At higher concentration, the beating diminished and terminated at 100 nM. Secondary beats early afterdepolarizations could be observed (data not shown) as well as beat pattern irregularities, which are typical effects of this K+ channel blocker. 11
Because E4031 suppressed the contraction of the cardiomyocytes after a concentration of 100 nM was applied, only concentrations below this level could be used to calculate parameters. A prolongation of the FPD signal can be observed; however, the automated calculation of the FPD was hindered by the appearance of EADs. This was reported in other experiments as well. 7
S-BayK8644
S-BayK8644, which is a calcium channel activator, caused a massive effect on the beat rate in the impedance mode. The effect on the beat rate is illustrated in Figure 14 . The shape of the signal is not influenced at all (beat rate regularity stays at 100%), which shows that the signal remains stable over the 10 s sweep. After the concentration reached a value of 100 nM, the amplitude dropped because of the lack of the recovery time after each beat. 11
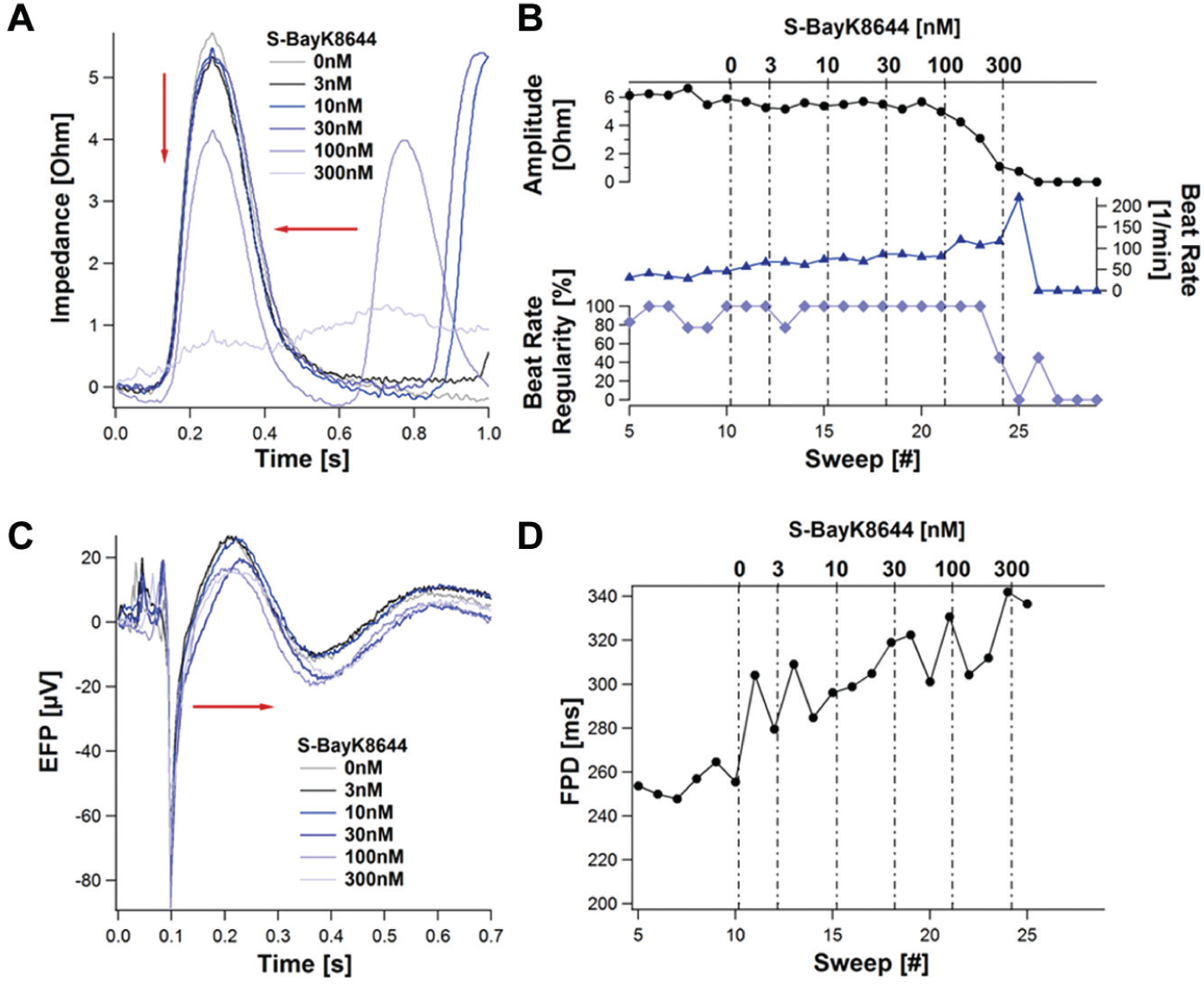
Calcium channel activator S-BayK8644 induced effects on the impedance and extracellular field potential (EFP). Presented are the mean beats of the impedance (
The automated calculation of the FPD derived from EFP measurements was hindered because the hump (which follows after the fast negative deflection; refer to Fig. 8 ) used for the calculation was not apparent in some traces. To achieve an efficient automated calculation of the FPD, a filtering step was required. Using a band pass filter of 2 Hz and 4 kHz revealed a dose-dependent increase of the FDP, which complies with results obtained with a standard MEA setup. 7
Hybrid Impedance and EFP Data Evaluation
Because both measurements are performed on the same sensor plate, using the same well with a time difference of about 5 s, both signals can be compared with each other in a powerful and very efficient way. Furthermore, this leads to the assumption that both signals can be related to each other and provide a more comprehensive picture of the cell monolayer activity. For example, Nifedipine seemed to induce a related effect on the pulse width and the FDP duration as well as on the impedance amplitude and the EFP amplitude ( Fig. 15 ).
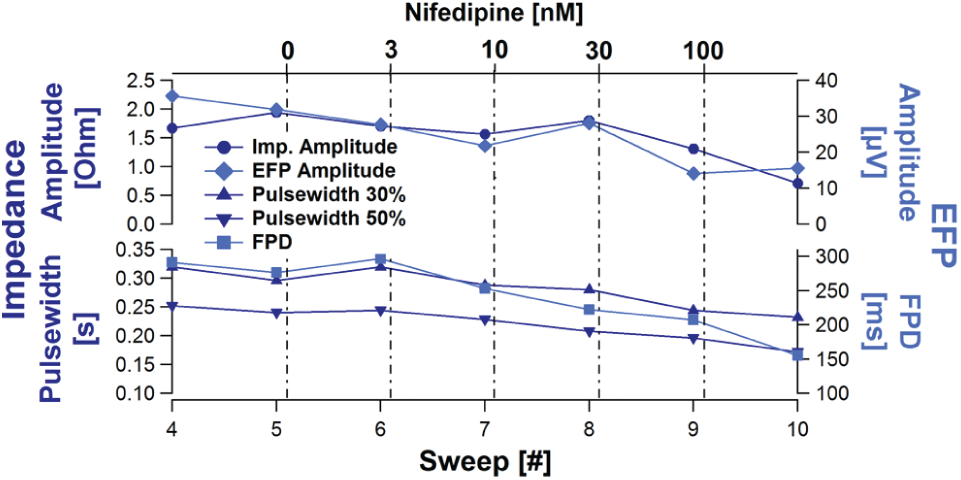
Comparison of the Nifedipine effect on the impedance and extracellular field potential (EFP). The concentration-dependent time course of the impedance and EFP amplitude (top) as well as the impedance pulse width and the extracellular field potential duration (FPD; bottom) are compared. Nifedipine concentrations are indicated.
For deeper investigations of the impact mechanism of a compound, it might be even necessary to evaluate both the impedance and the EFP. A good example is Blebbistatin, which inhibits the contraction of cardiomyocytes but not on the ion channel level. Blebbistatin blocks myosin II in an actin-detached state, leaving the ion channels unaffected. 16 This impact level was also observed when comparing impedance and EFP measurements: the contraction of cardiomyocytes was blocked in impedance measurements, and the EFP signal stayed unaffected ( Fig. 16 ). The impedance amplitude was completely blocked by Blebbistatin (3 µM, 6 µM), whereas the higher concentration acted more rapidly.
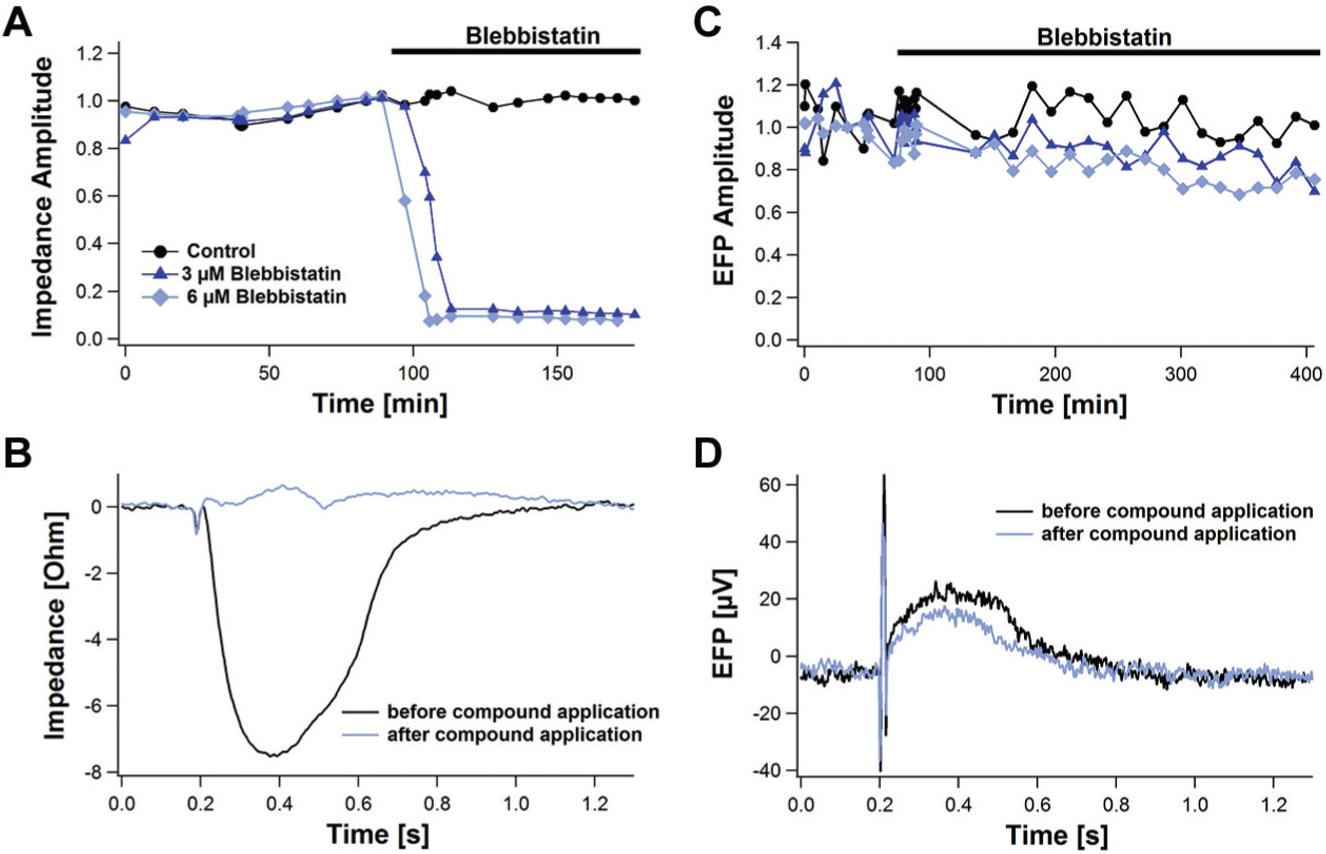
Time course of the Blebbistatin effect on the impedance and extracellular field potential (EFP). The effect of Blebbistatin on the impedance and EFP amplitude is presented in
Conclusion
High-throughput methods combined with human stem cell–derived tissues offer great potential for improved, more predictive assay formats for safety testing in the early drug development phase.17–19
In this article, we have introduced a new approach to recording electrical and mechanical properties of stem cell–derived cardiomyocytes using the CardioExcyte 96 platform. This new instrument allows combined impedance measurements with an ultrafast time resolution of 1 ms and MEA-like EFP measurements.
In impedance mode, the morphological properties of beating cardiac cells are measured with a 1-kHz resolution. This high bandwidth provides a very detailed picture of the time course of the cell contraction during the actual beating, which reflects all properties of the action potential. Obtained signals are very reproducible and stable, as the recordings are performed from a confluent layer of approximately 20,000 cardiomyocytes beating in synchrony. This gives a very robust assay format, which enables monitoring of the subtle modulation of the electrophysiological signal upon relevant compound addition. This has been shown using standard reference compounds active on the different classes of ion channels involved in the cardiomyocytes’ excitability and action potentials.
In EFP mode, the same electrodes are employed to record the EFP of the cardiomyocyte cell monolayer. The EFP recordings provide a view of the electrical properties of the cardiomyocytes at an even higher time resolution of 10 kHz, which is complementary to the contractility data. The analysis of the FPD upon compound addition enables a very information-rich readout for performing a pharmacological test in analogy to classical action potential duration studies using, for example, the patch-clamp method.
The described unique combination of high-frequency impedance and field potential measurements for the first time allows a comparison between FPD and pulse width in the contractility signal obtained from impedance measurements in the same instrument. It could be shown that the width of the impedance signal corresponds very well with the FPD and therefore strengthens the relevance of contractility data.
The CardioExcyte 96 addresses the lack of easy-to-use high-throughput and high-content in vitro assays. It permits the reliable investigation of short- and long-term pharmacological effects of compounds acting on the cardiac cells. The straightforward cell culture processes in combination with the 96-well format with a single electrode structure allows for robust high-throughput and cost-efficient cardiovascular drug screening. This makes the CardioExcyte 96 a valuable cardiotoxicity screening tool, specifically addressing the upcoming requirements as defined in the CiPA initiative. 20
Footnotes
Acknowledgements
We thank Cellular Dynamics International (CDI), Madison, Wisconsin, for the collaboration and for providing us with cardiomyocytes (iCell cardiomyocytes). We also thank Axiogenesis AG, Cologne, Germany, for the collaboration and for providing us with cardiomyocytes (Cor.4U).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work presented here was funded in part by the Bayerische Forschungsstiftung (grant AZ-1036-12).
