Abstract
The blood circulatory system links all organs from one to another to support and maintain each organ’s functions consistently. Therefore, blood vessels have been considered as a vital unit. Engineering perfusable functional blood vessels in vitro has been challenging due to difficulties in designing the connection between rigid macroscale tubes and fragile microscale ones. Here, we propose a generalizable method to engineer a “long” perfusable blood vessel network. To form millimeter-scale vessels, fibroblasts were co-cultured with human umbilical vein endothelial cells (HUVECs) in close proximity. In contrast to previous works, in which all cells were permanently placed within the device, we developed a novel method to culture paracrine factor secreting fibroblasts on an O-ring-shaped guide that can be transferred in and out. This approach affords flexibility in co-culture, where the effects of secreted factors can be decoupled. Using this, blood vessels with length up to 2 mm were successfully produced in a reproducible manner (>90%). Because the vessels form a perfusable network within the channel, simple links to inlets and outlets of the device allowed connections to the outside world. The robust and reproducible formation of in vitro engineered vessels can be used as a module to link various organ components as parts of future body-on-a-chip applications.
Introduction
In vitro cell culture methods have been widely used drug-screening applications to screen potential drugs rapidly and efficiently. Increasingly, three-dimensional (3D) cell culture methods and organ-on-a-chip devices are being developed that better simulate in vivo conditions. To overcome the limitations of conventional 2D in vitro assays, microfluidics have been used to provide a new approach for drug screening.1,2 By precisely controlling the physical and chemical microenvironment around the cells, microfluidic devices have evolved into sophisticated, single organ-on-a-chip systems such as those for guts, lungs, kidney, and other organs.3–5 Furthermore, the body-on-a-chip platform has been proposed as a toxicological assay. 6
Blood vessels, the most important conduit for organs, were recently engineered on a microfluidic platform. 7 Kim et al. have reported that co-culturing lung fibroblasts (LFs) and endothelial cells (ECs) could generate a perfusable blood vessel network in vitro. The paracrine effect from LFs induced EC assembly and the formation of tube structures that can be perfused. We were able to follow the in-vivo-like response of blood vessels, such as vasculogenesis, angiogenesis, and transendothelial migrations.9–13
A notable function that limited investigations to decouple the effect of paracrine factors was the permanent placement of LFs in close proximity to human umbilical vein endothelial cells (HUVECs) inside the microfluidic device. We wanted to develop a generalizable method to decouple the presence of other cell types needed to achieve differentiation of a particular cell type in microfluidic culture. Thus, a flexible culture system in which the paracrine secreting cells can be transferred was desirable.
In this article, we engineered a millimeter-scale perfusable network of blood vessels that can serve as a module to connect various organ components as a part of body-on-a-chip applications. Successful generation of the vessel network was helped by the adoption of LFs loaded into a removable hydrogel loop due to increase in the number of LFs. With the loop as a physical barrier, we could obtain perfusable microcapillary vessels that were composed only with endothelial cells after the vessel formation, without any interference in the EC region. The module has a single microchannel with an inlet–outlet pair, and it is thus able to support the connections with other organs and can represent a system that mimics blood circulation. Therefore, we expect this microscale blood vessel module (µBVM) to be used as a functional module of future body-on-a chip platforms.
Materials and Methods
Fabrication of the Microchannel
A microfluidic device was fabricated using soft lithography and other published techniques. 14 Briefly, a plasma-treated silicon wafer was spin-coated with 150 µm thick negative photoresist, SU-8 100 (MicroChem, Boston, MA). After prebaking at 65 °C for 10 min and 95 °C for 30 min, the wafer was exposed to 405 nm ultraviolet (UV) light (Shinu MST, Gyeonggi-do, Korea) for 500 mJ. After the exposure, the wafer was baked at 65 °C for 1 min and 95 °C for 10 min. SU-8 developer (Microchem) was used to remove the unexposed part of the photoresist. Using this wafer as the master mold, PDMS precursor Sylgard 184 (Dow Corning, Midland, MI) was poured, baked, and replicated. After punching for the inlets and reservoirs, the PDMS block was attached on the coverslip by plasma treatment (Femto Science, Gyeonggi-do, Korea). The attached device was heated on the 80 °C dry oven overnight to turn the surfaces hydrophobic.
Cell Culture
HUVECs (Lonza, Basel, Switzerland) were cultured in the endothelial growth medium (EGM-2; Lonza), and cells between passages 4 to 5 were used. Primary human LFs (Lonza) were cultured in fibroblast growth medium (FGM-2; Lonza), and passages 6 to 10 were used for the experiment. To harvest the cells, they were rinsed with phosphate buffered saline (PBS) and treated with 0.25% trypsin–EDTA (Gibco, Carlsbad, CA). After 2 min, M199 (Lonza) containing 10% fetal bovine serum was added to neutralize the effect of the enzyme. Detached cells were collected and centrifuged for 2 min in 1100 rpm, and diluted in EGM-2 to reach a certain cell number in the suspension.
LF–Hydrogel Loop Preparation
A plastic loop (Loop and Needle, SPL Life Science, Gyeonggi-do, Korea) was trimmed and used for capturing the LF–fibrin mixture. LFs were cultured and harvested from a tissue culture dish, and they were suspended in EGM-2 to 2.5~4×106 cells/ml. Bovine fibrinogen solution (2.5 mg/ml fibrinogen with 0.15 U/ml aprotinin) was added to the cell suspension, and the LF-containing hydrogels were mixed with thrombin (0.5 U/ml) immediately before loading on the inside loop portion. A total of 20 µl LF-containing hydrogels was then gently suspended at the inner surface of the plastic loop. After placing the gel-loaded loop into a 96-well plate, it was allowed to gel for 5 min. When the gels solidified, 200 µl of medium was filled and incubated for a day before use.
Live/Dead Assay
A live/dead assay kit (Molecular Probes L3224; Invitrogen, Carlsbad, CA) was used quantify LF viability when cultured in a hydrogel loop. The LF loop was gently rinsed with PBS and dipped into antibodies for 30 min. Hoechst 33342 was used to stain the nucleus. The LF loop was then rinsed with PBS and prepared for fluorescent imaging. Fluorescent images were taken with the confocal microscope FV1000 (Olympus, Tokyo, Japan) with a 3 µm interval. Stacked images were then deconvoluted into a 3D image using IMARIS (Bitplane, Zurich, Switzerland).
Preparation of µBVM
Fibrinogen was prepared as described above. HUVEC was mixed with the fibrinogen and injected into the microchannel. Mixture fluid was placed only in the microchannel because of the surface tension at the edge of the hydrophobic microchannel, which prohibits the further progress of the fluid. After 5 min incubation at room temperature to solidify the hydrogel, the fresh medium was added. The LF loop was then dipped into each reservoir to let HUVECs differentiate. The fresh medium was changed every 2 days.
Immunostaining and Imaging of the Capillary
To configure the physiological relevance of the blood vessel, samples were fixed with 4% paraformaldehyde. They were permeabilized with 0.15% Triton X-100 for 15 min and treated with PBS solution containing 3% bovine serum albumin (BSA; Sigma-Aldrich, St. Louis, MO). To visualize the lumens of the blood vessel, green fluorescent protein–conjugated Phalloidin (Molecular Probes) was used with a ratio of 1:200, and Hoechst 33342 for the nucleus. Fluorescent images of the capillary were taken with the confocal microscope with every 3 µm height. The stacked images were deconvoluted into 3D volume using IMARIS.
Results and Discussion
Loop–Hydrogel Culture of the Lung Fibroblast in µBVM
For the sustained secretion of growth factor from LFs, verification of the loop culture was conducted. We have tested the loop sample with the live/dead assay and compared their viability while they were at 1 and 10 days in vitro ( Fig. 1B ). Live and dead cells were distinguished by counting the cell nuclei. Over 95% of the fibroblast appeared alive after 10 days in hydrogel. referring no significant difference to DIV1 ( Fig. 1C ). The total number of cells can be controlled because the maximum amount of the hydrogel cell cocktail depends on the size of the diameter of the loop. We have counted 2.5~4×106 cells/ml in the hydrogel mixture, and the loop (3.8 mm inside diameter) holds up to 20 µl. Referring to previous publications, 13 LFs in the channel counted 30,000, which is half of the amount of ours. However, we have experienced that a higher concentration of LFs induced detachment of hydrogel from the loop surface, resulting in a non-uniform distribution of cells.
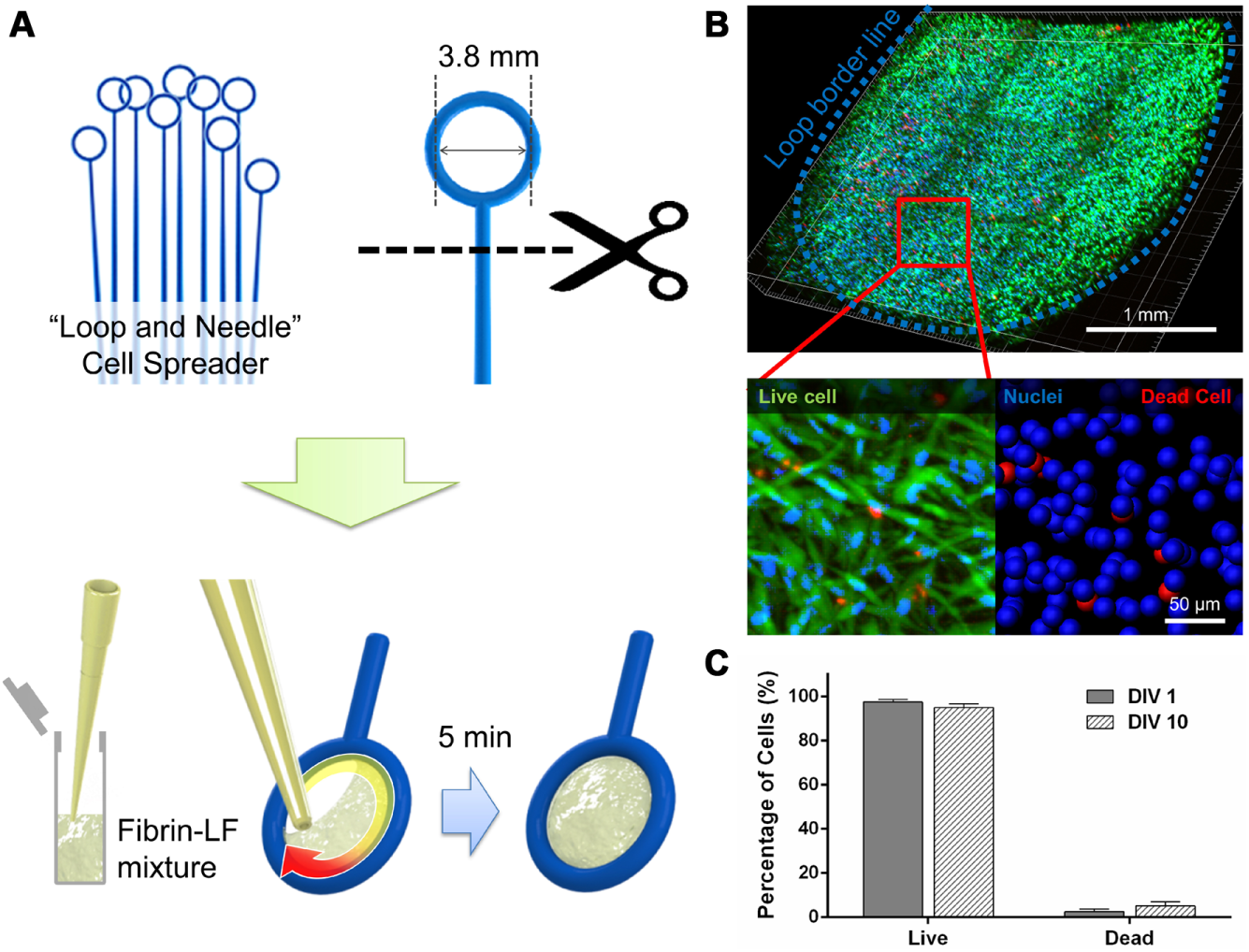
Culture of lung fibroblast (LF) captured in a hydrogel loop. (
Verification of Blood Vessel Formed in µBVM
Blood vessels, the most important conduit for organs, were recently engineered in a microfluidic platform. In the previous research of our group,12,15,16 we have reported that co-culturing of LFs and ECs in a defined hydrogel microenvironment could induce the perfusable blood vessel network. This work was different in a few ways from previous works that used preformed tubes in hydrogel whose inner surface was coated with endothelial cells. First, our vessels showed tighter cell–cell junction and thus lower permeability. When endothelial cells coated the inner surface of a tube, it took some time for the cells to proliferate and eventually form a completely covered monolayer. In this approach, there may be some local areas where the cell–cell junctions do not close completely or do not mature enough to form a tight barrier.10,12 Second, the vessels in this work were formed naturally rather than coated on an artificially formed single tube. Therefore, the diameter was not controlled by the diameter of the tube but by the origin of the endothelial cells themselves. These factors made it more physiologically relevant. In this research, to make a functional unit blood vessel, we introduced transferrable loop-cultured fibroblast instead of a predefined microfluidic device (
Fig. 2A
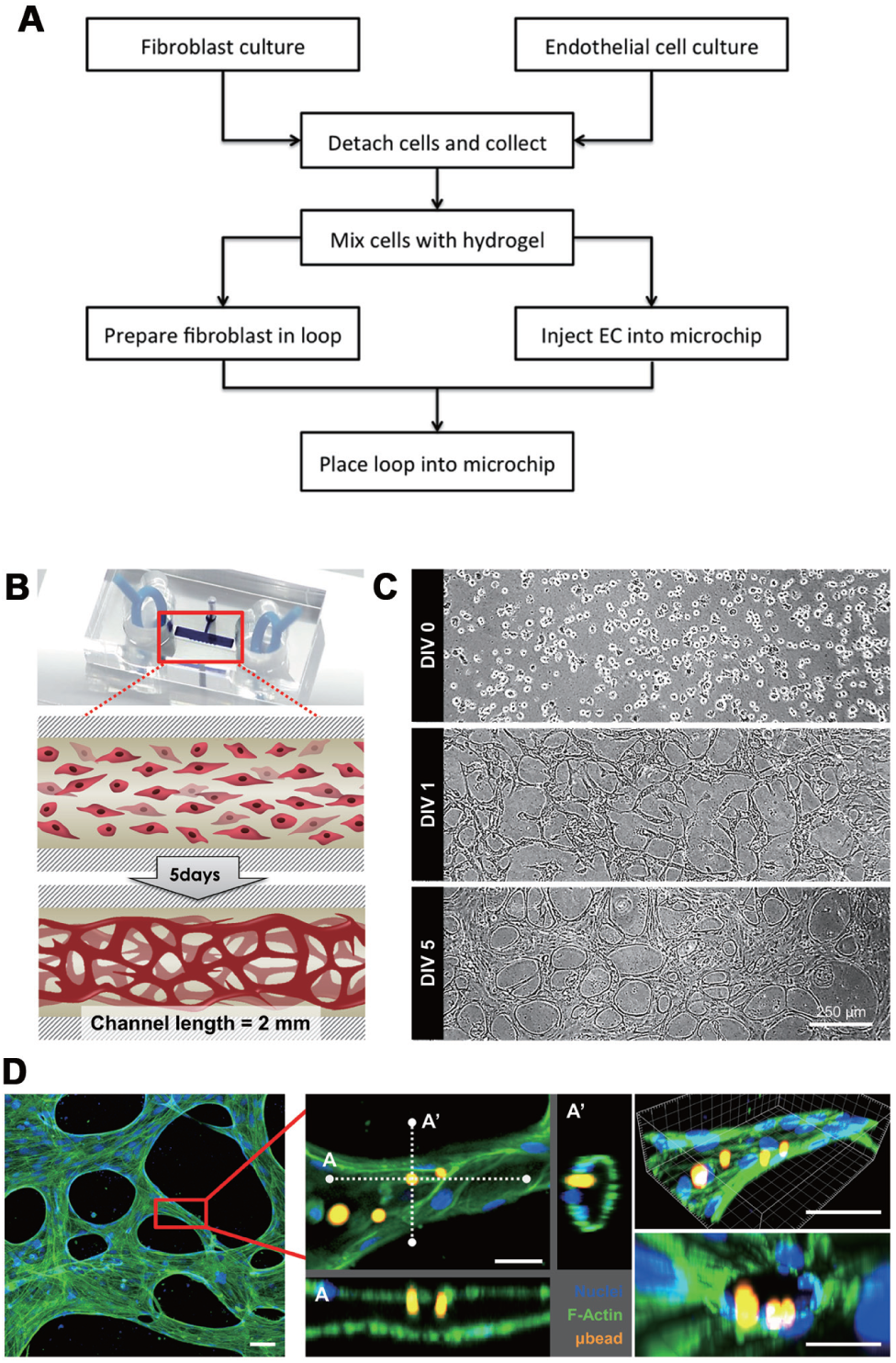
Formation of a microscale blood vessel module (µBVM) in a single microchannel device. (
After the blood vessel formation, we have immunostained µBVM to visualize the lumens. 7 µm of red fluorescent microbead was introduced in the reservoir to visualize the perfusability ( Fig. 2D ). Because the blood vessel was induced by a physically separated hydrogel loop, we were able to decouple the LF loop. However, without growth factors responsible for blood vessel maturation, removal of the hydrogel loop leads to disassembly of the lumen within a day (data not shown). Further experiments to stabilize the blood vessel without LFs can be researched with µBVM because this is the first model supporting transferable culture.
Extended µBVM for Organ-on-a Chip
By changing the cell types captured in a hydrogel loop, the source of paracrine factors exposed to the blood vessel could be changed. This process may lead to the design of further study to reveal interactions between blood vessels and other distinct cells or tissues. A hydrogel loop can be used as a method to decouple the presence of other cell types regarding the paracrine effect, which would be helpful for tissue and organ models in microfluidics research. We have described the connected reservoirs of the µBVM in serial ( Fig. 3A ). With this strategy, we could handle multiple culture dishes connecting through the perfusable blood vessel. This extended µBVM could connect multiple in vitro human tissues or organ models with biologically induced blood capillaries. As a body-on-a chip platform, previous research had suggested the endothelial-coated conduit. 17 However, after creating a naturally induced blood vessel, we expect to observe the feedback interaction with the blood conduit in more in-vivo-like circumstances, especially for perspectives on permeability.10,12
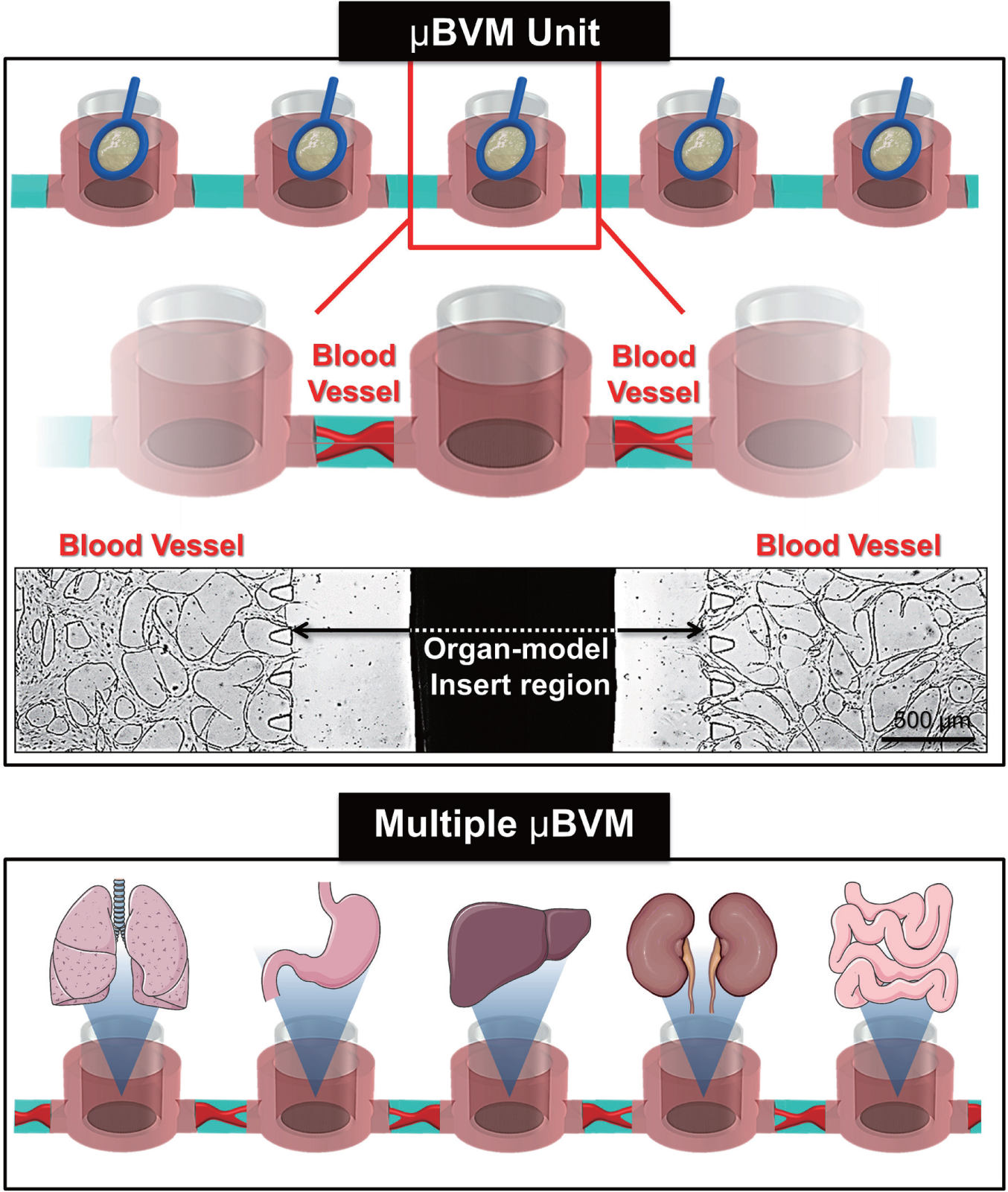
Schematics of the extended microscale blood vessel module (µBVM). Connected µBVM could function as a conduit of multiple in vitro organ models. Tissues or cells cultured in transwells or hydrogel loops could be transferred to each organ region chamber.
Conclusion
We propose a modular blood vessel platform with a simple microchannel design that can be engineered with a paracrine factor secreting hydrogel loop. LFs were captured within a hydrogel loop by surface tension and placed around the HUVEC-laden microchannel, inducing blood vessel formation between the loops. By loading a high density of LFs into the removable loop, “long” blood vessels on the order of a few millimeters were formed between the loops by paracrine factors. When placed in series, the cell-laden loops can form blood vessels that can further function as modular connectors between different compartments. After the vessels are formed, the µBVM loops can be replaced with organ-on-a-chip devices, thus forming a conduit between different organs on a body-on-a-chip device.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Brain Korea 21 Plus Project in 2014 and the Pioneer Project grant (NRF-2013M3C1A3041859) through the National Research Foundation funded by the Ministry of Education.
