Abstract
Alternating current (AC) electrokinetics is a collection of processes for manipulating bulk fluid mass and embedded objects with AC electric fields. The ability of AC electrokinetics to implement the major microfluidic operations, such as pumping, mixing, concentration, and separation, makes it possible to develop integrated systems for clinical diagnostics in nontraditional health care settings. The high conductivity of physiological fluids presents new challenges and opportunities for AC electrokinetics–based diagnostic systems. In this review, AC electrokinetic phenomena in conductive physiological fluids are described followed by a review of the basic microfluidic operations and the recent biomedical applications of AC electrokinetics. The future prospects of AC electrokinetics for clinical diagnostics are presented.
Introduction
In the past decades, micro–total analysis systems (µ-TAS) have been widely studied for various biological and medical applications.1,2 A prominent advantage of µ-TAS is the ability to integrate multiple components, such as liquid handling, cell and molecule manipulation, and detection into an automated, sample-in-answer-out system. Despite intensive efforts, most developed microfluidic systems have limited applications as automated biomedical analysis tools. Developing µ-TAS for laboratory automation is often constrained by complex sample preparation procedures and the inability to resolve full-system integration.3,4 Numerous microfluidic approaches, such as paper-based microfluidics, lab-on-a-CD, electrowetting-on-dielectric, and multiphase flow,4,5 are therefore being developed to address these challenges.
Among various microfluidic techniques alternating current (AC) electrokinetics represents a promising approach toward the development of a fully integrated µ-TAS. The advantages of AC electrokinetics include rigorous micro- and nano-manipulation methods, low power consumption, cost-effectiveness, simplicity in microelectrode fabrication, and advancement in portable electronics.6,7 AC electrokinetic phenomena include dielectrophoresis (DEP), AC electroosmosis (ACEO), and AC electrothermal flow (ACEF). These techniques are capable of performing most fundamental microfluidic operations, such as sample pumping, mixing, concentration, and separation, to develop automated biomedical analysis systems.
Biomedical applications often require manipulation of physiological samples and biological buffers with high conductivity. Table 1 summarizes the conductivities of typical physiological fluids and media. Nevertheless, most conventional electrokinetic studies were performed in low conductivity buffers to avoid unwanted side effects, such as electrolysis and heating. To design electrokinetics-based µ-TAS for laboratory automation applications, proper electrokinetic phenomena for manipulating conductive fluids should be considered. For example, ACEO is most effective in low-conductivity solution (<0.1 S/m) and has limited applicability in conductive biological buffers and physiological fluids. In contrast, ACEF is effective in a wide range of conductivities, and it is commonly used at frequencies greater than 100 kHz.8,9 Understanding the dominant electrokinetic phenomena and their limitations throughout a wide range of conductivities is essential in the design of electrokinetics-based µ-TAS.
In this review, electrokinetic techniques for manipulating conductive physiological fluids are discussed. In particular, ACEF and DEP are focused on due to their effectiveness in manipulating fluids with a wide conductivity range. After a summary of the theory and characteristics of DEP and ACEF, recent device designs and applications using these electrokinetic techniques for pumping, mixing, and bioparticle manipulation are presented. Finally, the potential areas in need of development and application of AC electrokinetics are discussed.
AC Electrothermal Flow
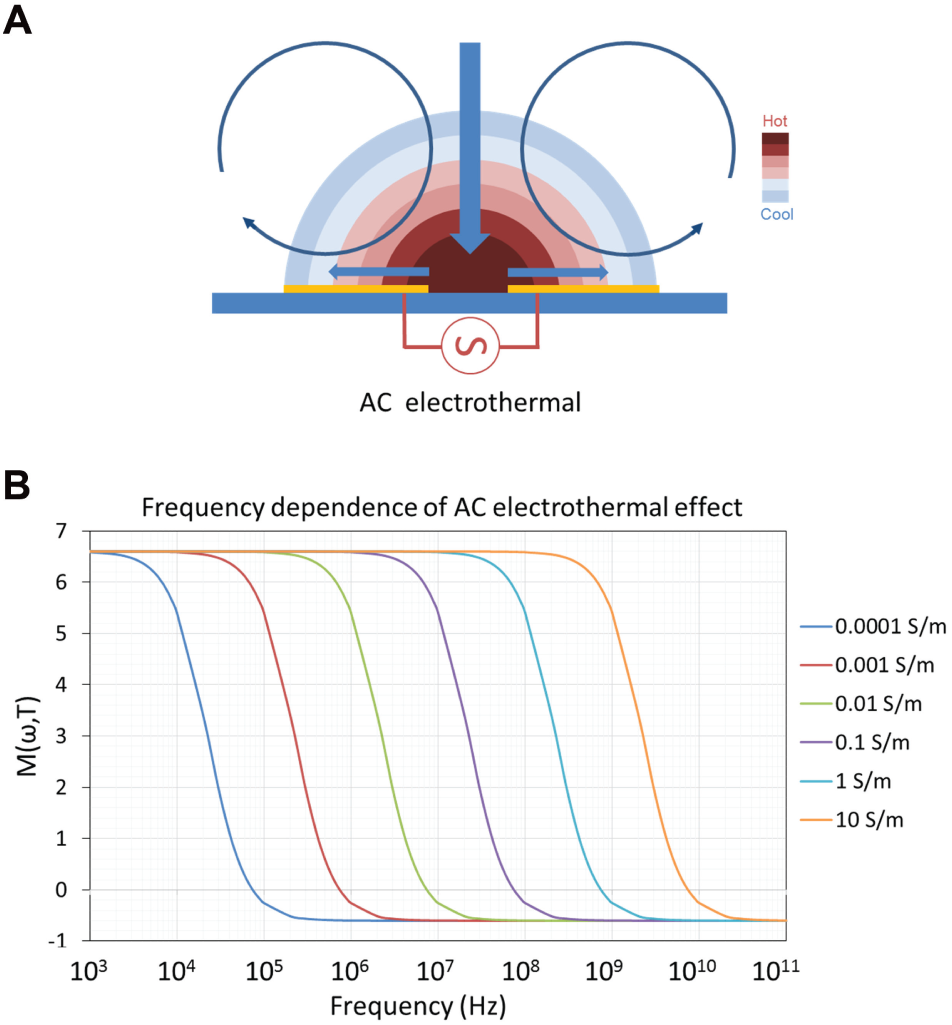
ACEF arises from temperature gradients developed within a fluid when an external electric field is applied. The local temperature gradients create conductivity, permittivity, viscosity, and density gradients in the solution. These gradients and their interactions with the electric field, in turn, create bulk fluid forces and fluid motion ( Figure 1A ). For instance, the interaction between the conductive gradient and the nonuniform electric field induces the Coulomb force. The vertical density gradient generates the buoyancy force. A theoretical model has been developed for estimating the electrothermal force.6,10,11 In this model, the charge density and the electric field are described by Gauss’s law (1) and the charge conservation equation (2):

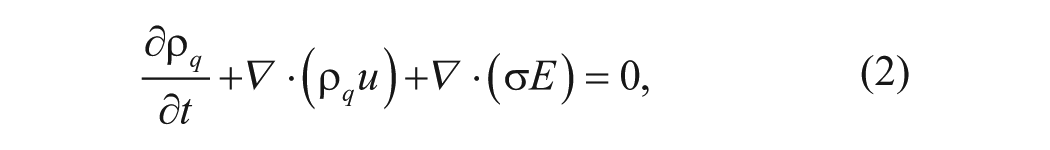
where ϵ, ρ q , σ, and u are the electrical permittivity, the charge density, the solution conductivity, and the fluid velocity. E is the electric field. By using perturbation analysis, the electrical potential φ in the medium can be estimated by
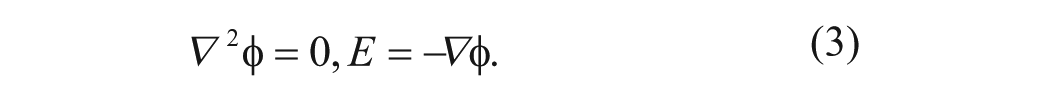
(
Joule heating is induced by the electrical field according to the Ohm’s law, and the temperature field can be determined by considering the following energy equation:
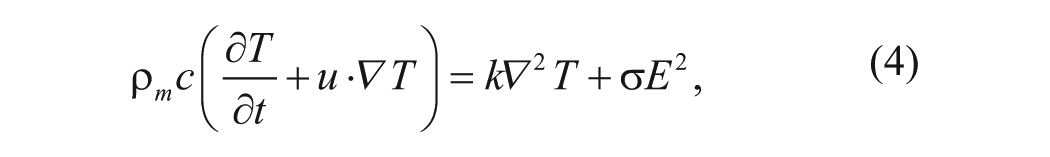
where k,
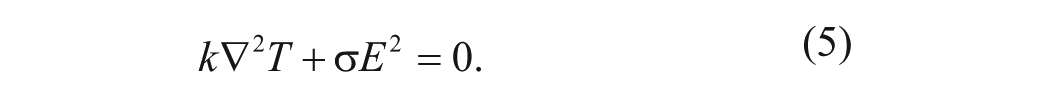
The temperature field can then be determined to estimate the time-averaged electrothermal force:
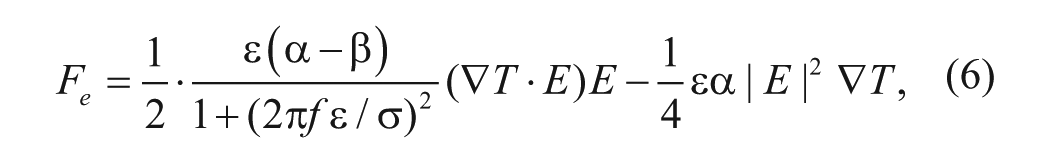
where
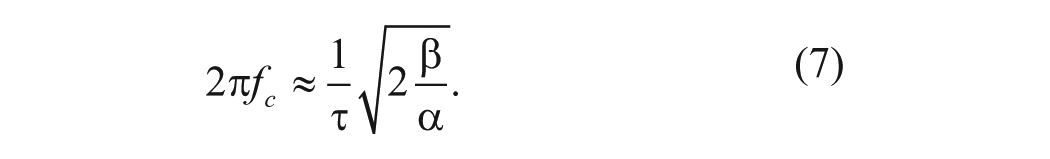
The charge relaxation time of the liquid is given by
For a parallel electrode with a small gap, the gradient of temperature can be estimated by
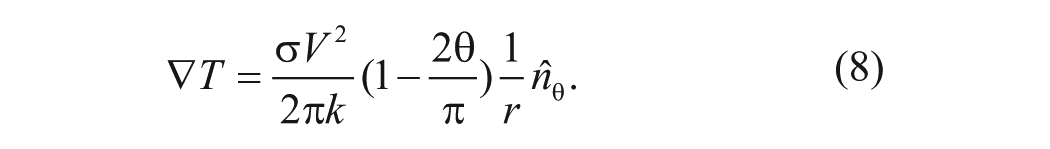
The time-averaged force with an applied alternating potential is
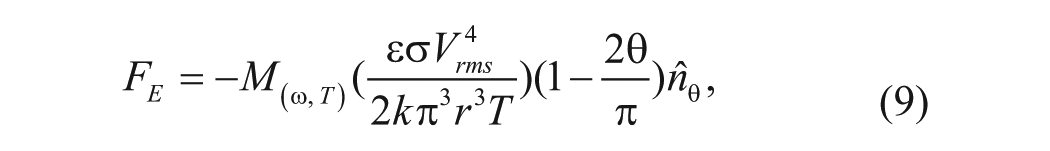
where
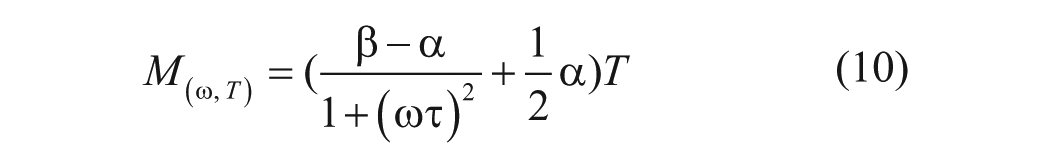
is a dimensionless factor describing the frequency dependence.
ACEF creates fluid motion in samples with a wide range of conductivity. 8 Figure 1B shows the frequency dependence of the electrothermal force in different conductivities. For physiological conductivities (~1 S/m), the ACEF velocities are constant and effective at a high frequency range (~1 MHz),10,11 which can minimize electrolysis of the fluid. This presents a great advantage of ACEF flow. From equation (9), the ACEF is proportional to V 4 . For a sample with a higher conductivity, only a low voltage is required for biomedical applications. By properly designing the electrode and microchannel structures, ACEF can be applied to perform necessary microfluidic operations.
Dielectrophoresis
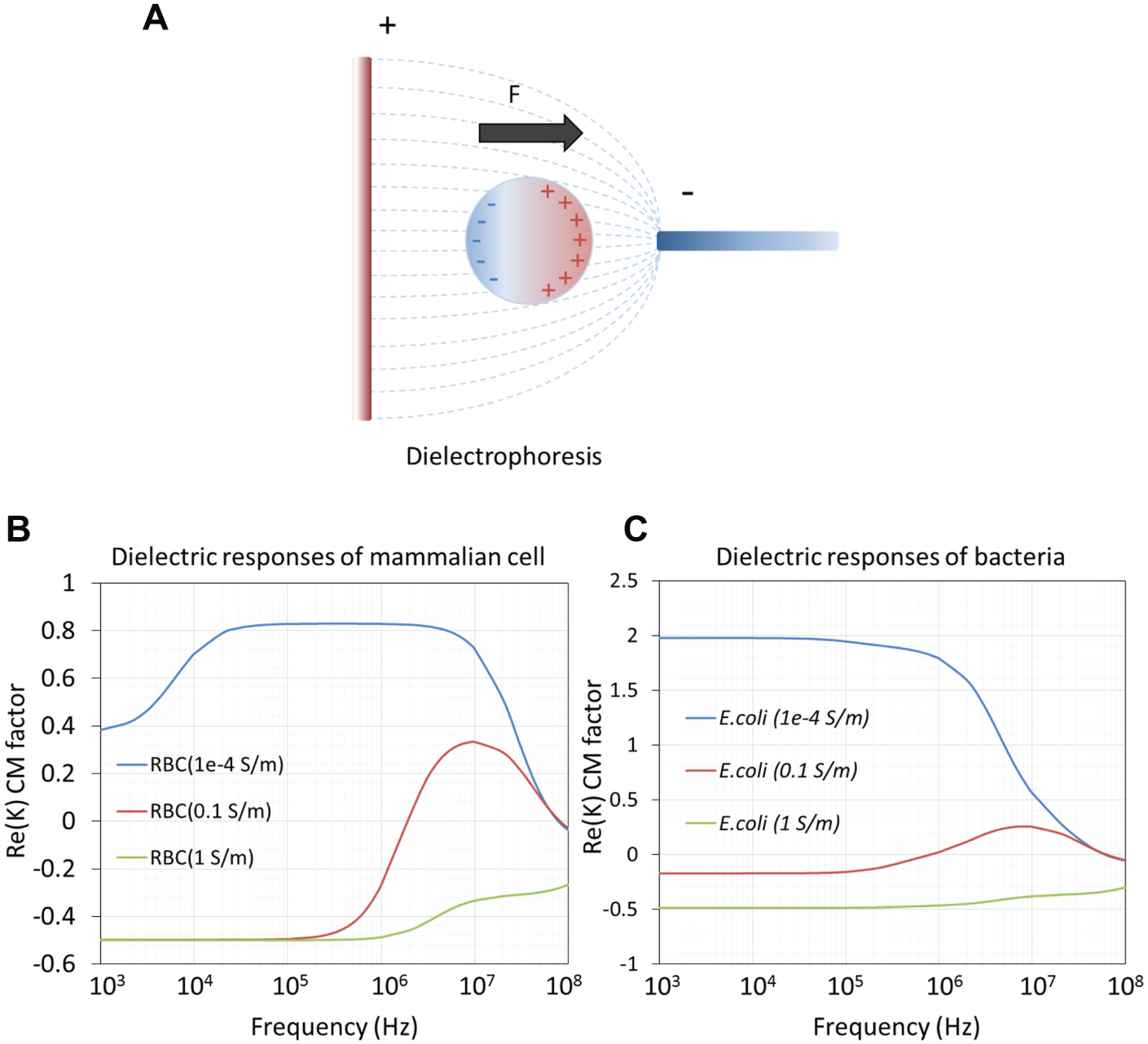
DEP arises from the interaction between a nonuniform electric field and the induced dipole of a polarizable object ( Fig. 2A ). The dielectrophoretic force can move the object toward the high electric field region or low electric field region, depending on the effective polarization between the object and the medium. If the object has a higher polarizability, the force will push the object toward the high electric field strength region (positive DEP); otherwise, the force will point toward the low electric field strength region (negative DEP). DEP has been demonstrated to effectively manipulate various types of molecules, particles, and cells.7,12
(
The time-averaged dielectrophoretic force on a spherical object is given by 13
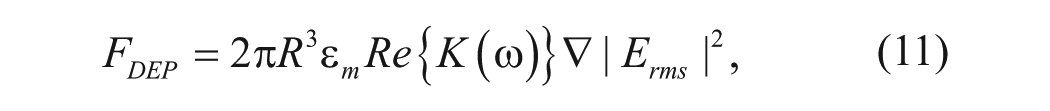
where R is the particle radius, Erms is the root mean square electric field, ω is the angular frequency, and K(ω) is the Clausius-Mossotti factor, which describes the frequency variation of the effective polarizability of the particle in the medium. The Clausius-Mossotti factor is defined by
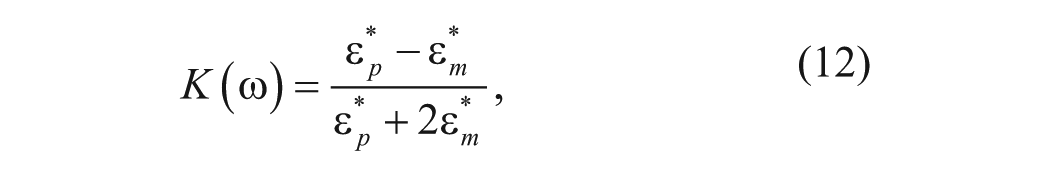
where

where ϵ is the permittivity and σ is the conductivity of the particle and medium.
Several major characteristics of DEP should be noticed. First, DEP is most effective near the edge of the electrode, where the gradient of the electric field is strongest (F ~
Microfluidic Operations
Using AC electrokinetics, multiple microfluidic operations, such as pumping, mixing, and particle manipulation, can potentially be implemented within a single system with the proper microelectrode design and electronic interface.8,15 This renders AC electrokinetics a promising strategy for microfluidic system integration. Recent advancements in microfluidic operations using AC electrokinetics for physiological fluids are summarized in this section.
Pumping
Existing microfluidic systems often require external driving mechanisms (e.g., pumps or pressure sources) to accomplish transportation of reagents or samples within the devices. Incorporation of such mechanisms, however, greatly reduces the portability of the device for point-of-care applications.16,17 This represents a fundamental hurdle for implementing μ-TAS in point-of-care diagnostic applications. Electrokinetic pumps, which do not involve any mechanical moving parts, can be fabricated easily and resolve the need of external driving mechanisms necessary for microfluidic system integration. 18 Various electrokinetic pump designs have been proposed.19,20 The electrolysis pump, for instance, can be applied to generate fluid motion in a microchannel. 16 ACEF flow represents another promising method for fluid pumping in microchannels.21,22 Recently, various ACEF pumps have been designed.
With a proper electrode design, an imbalance of the temperature and electric fields is created to break the symmetric competitive vortices and create a net fluid flow ( Fig. 3A ). The ACEF pump with an array of asymmetric microelectrode pairs has been demonstrated.23,24 The study reported an asymmetric electrode design with low voltages (<15 Vrms) for driving fluids with conductivities between 0.02 and 1 S/m. Fluid velocities of 100 to 1000 µm/s were reported. Furthermore, an asymmetric electrode array with biased AC signals has been reported ( Fig. 3B ). 25 Compared with pure AC signals, the direct current (DC) bias increased the local ionic strength near the electrodes, leading to a substantial conductivity gradient. This gradient could in turn induce a stronger ACEF. The design reached a linear velocity of 2.5 mm/s with 4.42 Vrms AC signal and 1.0 V DC bias. The volumetric flow rate or pumping pressure, however, was not specified in these studies.
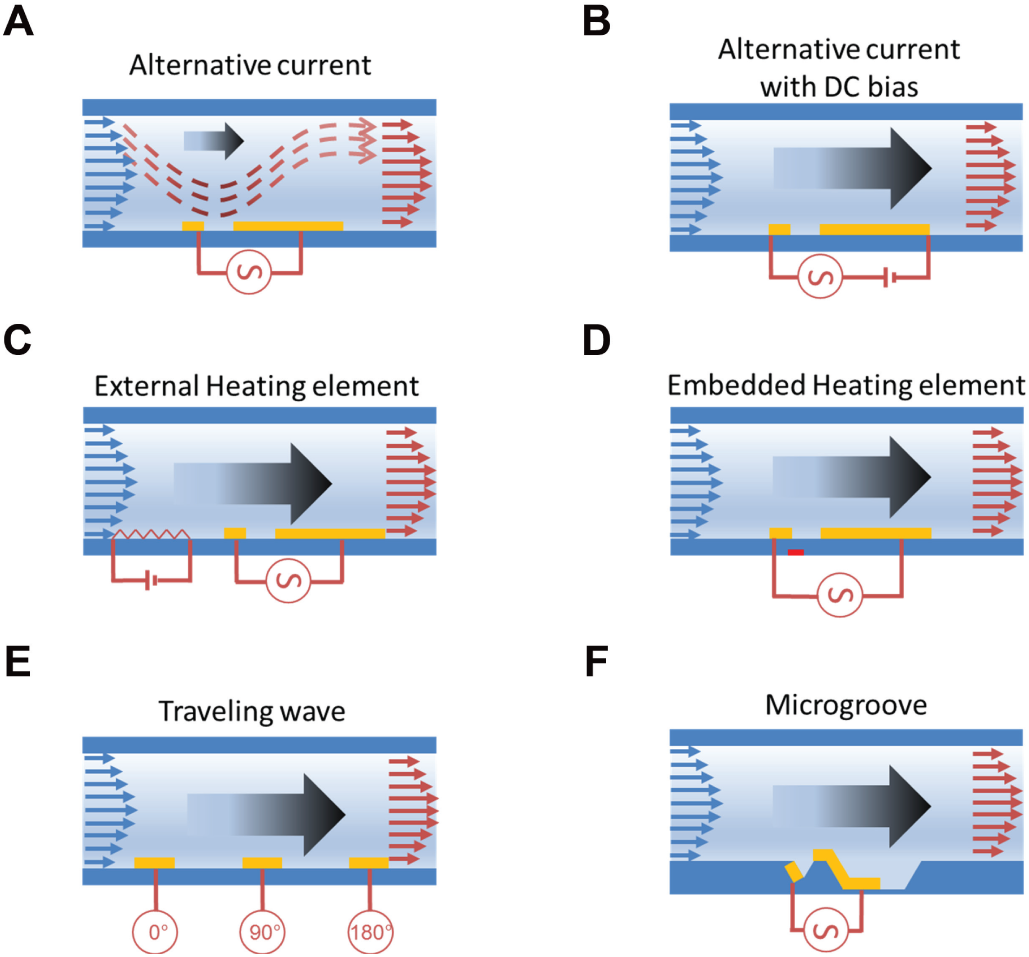
Schematic illustration of six alternating current (AC) electrothermal pump designs. (
An important consideration for ACEF pumps is the creation of the temperature gradient by Joule heating, which depends on the sample conductivity. For a relatively low conductive medium (<0.1 S/m), a high voltage is required to generate a large temperature gradient. This can be resolved via two approaches: (1) external heat source or (2) ACEF pump with voltage phase change. Inclusion of an external heat source can create a longitudinal temperature gradient and allows independent control of the temperature gradient and the electric field. For instance, Stubbe et al 26 incorporated a separate heating electrode, in addition to the asymmetric electrode, in the microchannel ( Fig. 3C ). The pump velocity was measured at conductivities varying from 0.1 S/m to 1.3 S/m and reached 20 µm/s with a voltage of 30 V peak-to-peak (Vpp). The velocities were shown to be relatively independent of the medium conductivity. Another approach of the ACEF pump with external heating was also proposed. 27 For this design, the heating element was embedded in the glass substrate ( Fig. 3D ). Numerical simulation showed that the design provided over two times higher volumetric flow than Joule heating alone for the same input power.
The second type of ACEF pump designed involves a phase change of the electrical signal. A two-phase AC voltage was applied to the asymmetric electrode array in a high-conductivity solution (0.224 S/m). 28 The major advantage of the two-phase ACEF pump, compared with the single-phase pump, is enhanced electric field magnitude due to superposition of the electric fields produced by the different-phased electrodes. Numerically, the two-phase ACEF pump achieved at least 25% faster fluid flow rates than the single-phase ACEF pump. Recently, a multiphase ACEF pump with improved performance shown by theoretical and numerical simulation also has been studied ( Fig. 3E ). 29 These results suggest design information necessary to build micro-pump systems for lab-on-chip applications. However, these concepts have not been verified experimentally.
Theoretically, a net pumping action can be generated as long as the design breaks the symmetric competitive vortices over the electrodes. In the asymmetric electrode design, the large effective vortices provide the major force pumping the fluid forward. However, the existence of small reversal vortices can greatly reduce the overall pumping capacity. 30 Channel geometry modification has proven effective in addressing this issue. This concept was first demonstrated by Du and Manoochehri 30 via incorporation of microgrooved channels in the asymmetric electrode array design ( Fig. 3F ). The microgrooves on the channel floor significantly reduced the effects of the reversal flows, which enhanced the overall pumping efficiency. In the study, the fluid conductivity was 0.1 S/m. The microgrooved structure increased the pumping capacity by 5- to 6-fold compared with a simple asymmetric electrode arrangement with the same effective dimensions.30,31
Mixing
Mixing is essential in most biochemical assays. It is especially important for µ-TAS due to the laminar nature of microfluidic flows. The small characteristic length of a microchannel typically leads to a low Reynolds number (Re < 1), indicative of laminar flow. Without turbulent mixing, molecular advection in the microscale only relies on diffusion. The diffusion time scale can be estimated by T = L2/D, where L is the diffusion length and D is the mass diffusion coefficient. In a biochemical assay (e.g., protein or nucleic acid detection), the reaction time can be greatly limited by the low mass diffusion coefficient of the macromolecules. Therefore, effective mixing in microfluidic devices represents a key component toward the development of µ-TAS. 5
Several methods have been developed for microfluidic mixing and can be categorized to two types: passive or active mixers. For passive mixers, an external actuation mechanism other than fluid pumping is not required. Mixing can be enhanced by split-and-recombine, multilaminating, Dean vortex, or 3D channel geometry dependent on the Reynolds number regime. For active mixers, external driving forces, such as pressure, ultrasound, magnetism, and electrohydrodynamics, are required. 32 However, advantages of active mixers include shorter mixing time and simpler microchannel design. Numerous electrokinetic micromixers have been designed due to the effectiveness of electrokinetic mixing and the simplicity of the microelectrode fabrication compared with other active mixing approaches.33,34 ACEF mixing, in particular, has received extensive attention for biomedical applications. Various ACEF mixers have been studied extensively using numerical approaches, and effective mixing can be accomplished in a shorter period of time compared with diffusion. Two ACEF mixer designs for reagent mixing are shown in Figure 4A , B . The mixer in Figure 4A consists of a pair of coplanar electrodes under two fluid streams. By applying a DC-biased AC voltage, an electrokinetic flow is induced with a flow profile perpendicular to that of incoming laminar streams of liquids to be mixed. 35 An improved design has also been reported recently ( Fig. 4B ). 36 In this design, a pair of coplanar electrodes with a sinusoidal interelectrode gap was used to enhance the mixing in a microchannel.
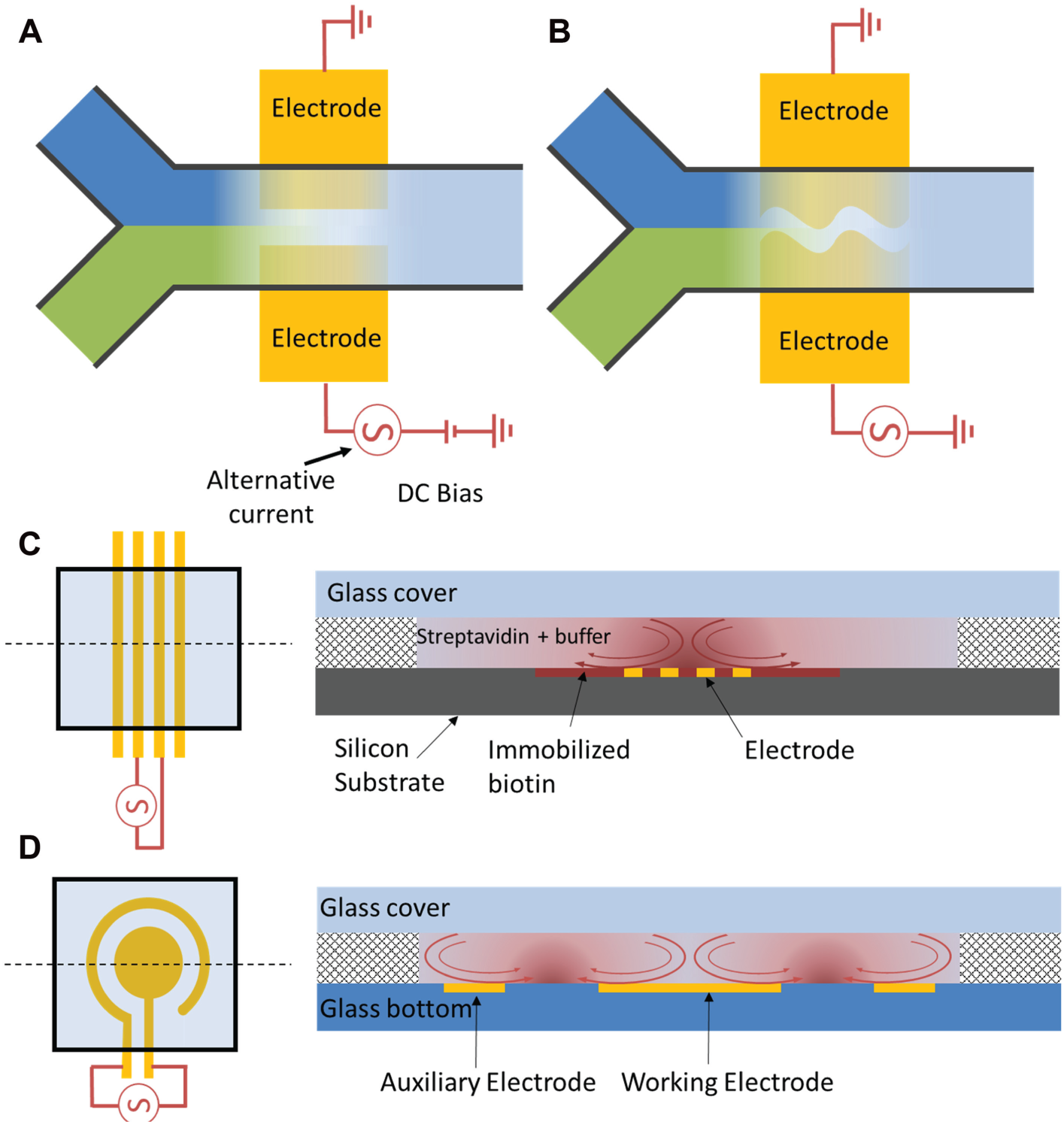
Schematic illustrations of electrode configurations for fluid mixing by alternating current (AC) electrothermal effect with (
In addition to reagent mixing, ACEF can be incorporated for assay enhancement. There are several advantages of ACEF for enhancement of biochemical assays, particularly for electrochemical assays. First, ACEF can be generated by the application of an AC potential to sensing electrodes, which can be easily integrated into the sensing platform.37,38 Second, ACEF can apply on-chip stirring and heating, both of which enhance the sensor signal. Last, the ACEF can also reduce the background noise and enhance the specificity of the assay.
The biomedical applications for ACEF assay enhancement have been demonstrated.37,39–42 Electrothermal assay enhancement was first demonstrated with biotin-streptavidin binding by optical detection ( Fig. 4C ). 39 More recently, an ACEF-enhanced electrochemical biosensor was confirmed for pathogen identification and antimicrobial susceptibility testing.37,43 In this design, ACEF enhancement was directly performed on a self-assembled, monolayer-based electrochemical sensor. The same set of electrodes was used for both electrochemical signal enhancement and electrochemical sensing, leading to enhanced results. In particular, the sensitivity of the bacterial 16S ribosomal RNA (rRNA) hybridization assay was improved at least one order of magnitude, and the incubation time was reduced 6-fold. The ACEF-enhanced biosensor can be implemented within an enclosed microfluidic system, leading to automated molecular diagnostics at the point of care ( Fig. 4D ).16,38
Another challenge of clinical diagnostics is the matrix effects of the physiological fluids, which can affect the assay performance. 44 ACEF was shown to mitigate the matrix effect in an electrochemical biosensor.43,45 ACEF enhancement was able to not only increase the specific bonding efficiency but also reduce the background noise due to the matrix effect in clinical urine and blood samples. The study also showed multiplex detection of three uropathogenic clinical isolates with similar 16S rRNA sequences, illustrating the enhanced specificity of the assay. These results demonstrated that ACEF can significantly improve the signal-to-noise ratio of the biosensor for multiplex urinary tract infection diagnosis.
Bioparticles Manipulation
The ability to selectively manipulate bioparticles, such as cells, DNA, and proteins, is important for various biological applications. Physical positioning of bioparticles through techniques such as trapping, focusing, and isolation is required to perform the biomedical analysis. Various manipulation techniques, such as optical tweezers, 46 acoustic forces, 47 and surface modification, 48 have been reported. For clinical diagnostics at the point of care, the advantages of electrokinetics include simple system requirements, label-free manipulation, and cost-effectiveness. Particle manipulation can be applied to a wide spectrum of biomedical applications, such as rapid bacteria separation for antimicrobial susceptibility testing, 49 DNA extraction for pathogen identification, 50 and proteins concentration for biomarker discovery, 51 to name a few.
DEP is one effective strategy for manipulation of bioparticles, such as mammalian cells, bacteria, yeast, DNA, and proteins. DEP-based cell manipulation has been demonstrated for different applications.49,52–54 Figure 5A shows a schematic of using positive DEP to concentrate bacteria.55,56 Positive DEP is typically performed in media with low conductivity to ensure that the cells are more polarizable than the media. In contrast, negative DEP is more commonly observed in media with high conductivity. For instance, negative DEP force has been shown for cell manipulation, such as separation,14,57,58 concentration,14,59 and patterning.60,61 One of the designs of negative DEP patterning was proposed by Mittal et al. 60 ( Fig. 5B ). The device was demonstrated to effectively position HeLa cells and 3T3 fibroblasts in conductive media by using negative DEP. The biocompatibility of their method was demonstrated by showing that the patterned cells could proliferate and express a normal morphology. Cell separation can also be achieved by negative DEP.14,57 One example is using DEP continuously to separate and to concentrate bacteria in physiological samples ( Fig. 5C , D ). 14 The device separates bacteria from diluted blood and cerebrospinal fluid. The separation efficiency of the device reached 90% at 30 µL/h. The bacteria were further captured and concentrated into a collection chamber using positive DEP. The bacteria capture efficiency was around 95% at 800 µL/h.
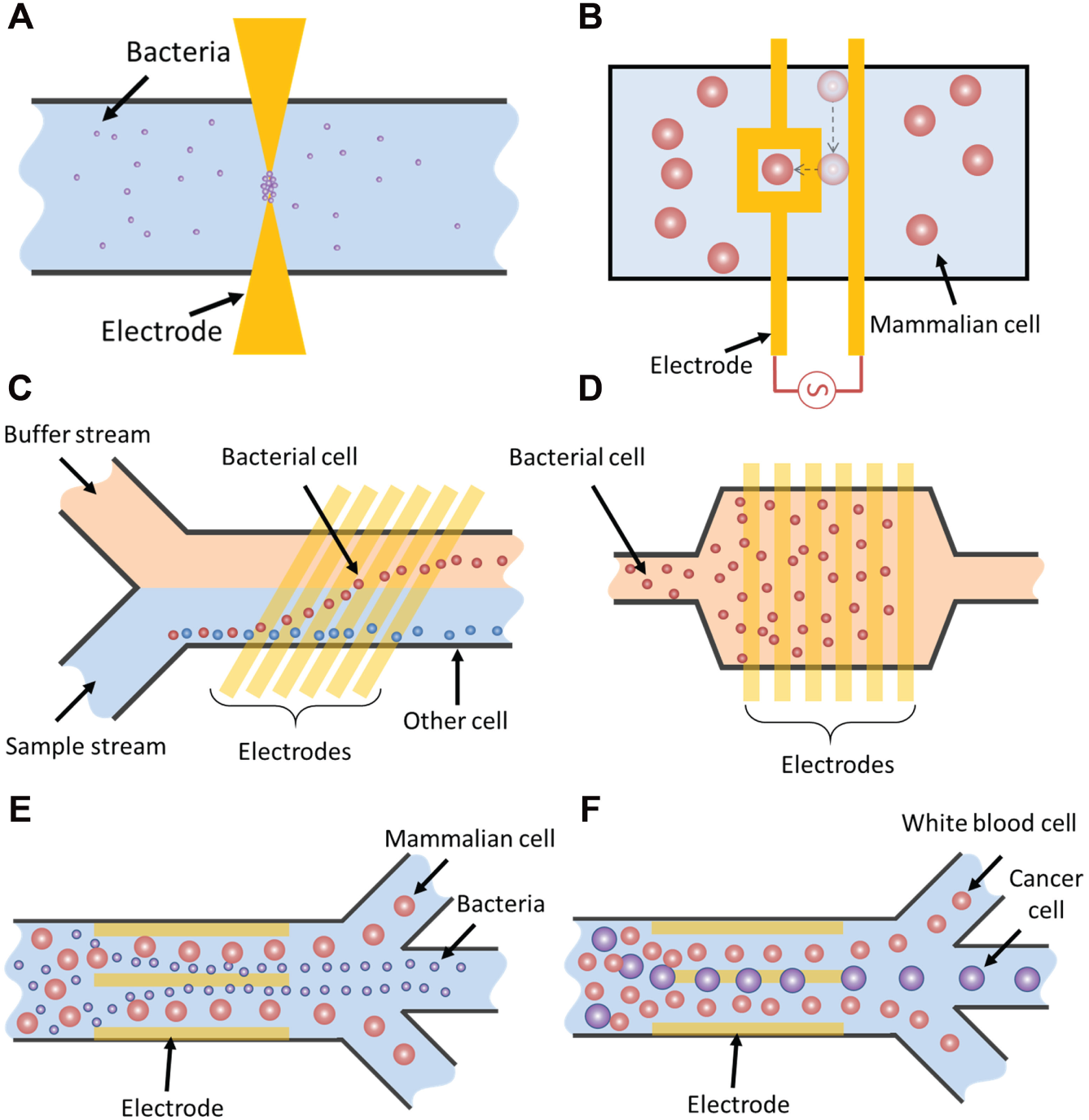
Bioparticle manipulation: schematic illustrations of (
During DEP manipulation, electrokinetics-induced fluid motion should be considered as it can change the efficiency of DEP. The fluid motion introduces hydrodynamic drag force that washes away the DEP trapped particles or, on the other hand, the fluid motion could enhance the performance of the DEP manipulation by introducing long-range fluid motion. A hybrid electrokinetic manipulation system was proposed by Sin et al. 34 In this hybrid electrokinetic device, the combination of DEP and ACEF allows separation, mixing, and concentration of colloidal particles ranging from nanometers to micrometers. Furthermore, Gao et al.62,63 demonstrated a three-parallel electrode configuration for continuous isolation of various bacteria and mammalian cells ( Fig. 5E , F ). By properly designing the channel and operating condition, they demonstrated ACEF constrains target cells (Escherichia coli) far away from the bulk solution toward the electrode surface, where DEP is most effective. Target cells can be further trapped on the edges of the central electrode by DEP and electrophoresis ( Fig. 5E ). 62 Using a similar electrode design, the combination of DEP and ACEF resulted in different equilibrium positions of cancer cells and white blood cells in the flow channel, effectively separating the cells ( Fig. 5F ).
Future Direction
AC electrokinetics has been demonstrated as a promising technique for fluid and particle manipulation, including pumping, mixing, concentration, and separation, in microfluidic systems. Although the potential of AC electrokinetics is established in numerous biomedical applications, little work has been done to integrate multiple electrokinetic sample preparation and sensing modules for laboratory automation. Since only microelectrodes and electronic interfaces are required, systems integration with AC electrokinetics can be potentially cost-effective. The development of fully integrated electrokinetic systems is an important step to realize the potential of electrokinetics-based µ-TAS for infectious disease diagnostics, early stage cancer detection, cell biology, and other applications.
ACEF often occurs in electrokinetic manipulation of high-conductivity media. By properly applying knowledge of the phenomenon, ACEF could be an effective AC electrokinetic technique for system integration, leading toward point-of-care diagnostics because of its effectiveness at physiological conductivity. 17 The technique is further applicable to various types of samples and applications. Since molecular advection and molecular binding efficiency are the fundamental barriers that are commonly observed in various biomedical assays, ACEF enhancement has a great potential to benefit other sensing platforms found in clinical and biochemical applications.
Another challenge for current electrokinetics-based µ-TAS is the volume mismatch between the microfluidic system and the physiological sample. For infectious disease diagnostics, samples from a microliter to tens of milliliters are collected from patients. However, present microfluidic systems can typically handle on the order of a 10- to 100-µL microliter solution. It is a fundamental obstacle that is necessary to surpass to move the technology toward clinical applications. In future research, it is important to develop a better strategy to handle a large volume of physiological samples within a short period of time.
Efforts should also be devoted to the fundamental understanding of electrokinetics. Most of the classic theories of electrokinetics are derived based on simplified assumptions, such as small temperature elevation and constant electrical properties of the fluids and the bioparticles. As the techniques and applications are expanding, these classic assumptions may not be valid and should be reconsidered. For instance, as the length scale of the system increases, natural convection, such as buoyancy, should be considered in ACEF design. Furthermore, heat convection has to be considered in systems handling a large volume. The effects of DC bias and hybrid electrokinetics should also be studied systematically.
In conclusion, the characteristics of two AC electrokinetic phenomena, DEP and ACEF, for manipulation of conductive physiological fluids have been discussed. These phenomena are particularly important in clinical diagnostics, as they relate to conductive samples. AC electrokinetics-based operation, such as pumping, mixing, and bioparticle manipulation, in microfluidics are reviewed. Future studies will lead to a fully integrated platform, which can quickly handle a large volume of physiological samples. It is important that a fully integrated ACEF model be created as it is lacking currently. Further understanding of the ACEF model would be useful to predict the electrohydrodynamic phenomenon in novel electrokinetic devices. In addition, new ACEF techniques hold promise as a better tool for developing new biotechnology applications.
Footnotes
Acknowledgements
The authors thank Jose Miguel Valdez for his helpful suggestions and technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH Health Director’s New Innovator Award (DP2OD007161) and NIAID (U01AI082457 and R44A-I088756).

