Abstract
Microfabricated organs-on-chips consist of tissue-engineered 3D in vitro models, which rely on engineering design and provide the physiological context of human organs. Recently, significant effort has been devoted to the creation of a biomimetic cardiac system by using microfabrication techniques. By applying allometric scaling laws, microengineered cardiac systems simulating arterial flow, pulse properties, and architectural environments have been implemented, allowing high-throughput pathophysiological experiments and drug screens. In this review, we illustrate the recent trends in cardiac microsystems with emphasis on cardiac pumping and valving functions. We report problems and solutions brought to light by existing organs-on-chip models and discuss future directions of the field. We also describe the needs and desired design features that will enable the control of mechanical, electrical, and chemical environments to generate functional in vitro cardiac disease models.
Introduction
The advent of microfluidics and microfabrication techniques creates the concept of organs-on-chips, enabling high-throughput in vitro biomimetic tissue formation and real-time biomarker screens.1–5 Currently, development of organs-on-a-chip is focused on silicon-supported, layered, and/or micropatterned fluidic systems as an attempt to mimic 3D microenvironments of native organs.6–9 By mimicking organ-specific dynamic conditions in the microscale, organs-on-a-chip allow systematic approaches to discover the effects of biochemical and biomechanical factors. Furthermore, by integrating various sensors and actuators into the chips, organs-on-a-chip provide a tunable biochemical and biomechanical cellular environments.10–22
Among organs, the heart is the most dynamic structure, which consists of electromechanically actuated pumps and multiple passive valves. The macroscopic motion of the heart is generated by microscopic motion of each cardiomyocyte triggered by electrical signals. With the organs-on-a-chip concept, cardiac microsystems (CMSs) have been developed to enable the studies of electrophysiological responses and contractile motion of cardiomyocytes under various biochemical factors and fluidic conditions.23–32 This contractile motion has been used to fabricate a bio-hybrid pump, which is powered by contractile forces of cardiomyocytes with or without electrochemical stimulation in the microfluidic device.28,30,32 To increase throughput and controllability of CMS platforms, parallel microfluidic pumps and valves have been introduced.25,29 Such platforms have eventually heightened interest in the integration of pulsatile flow and structure of the cardiac system within the microfluidic cell culture. Further development of these in vitro CMS platforms has significant roles to find the mechanism of various cardiac diseases with tunable static and dynamic culture environments and improve drug development by generating an engineered disease model.
In this review, we focus on the advancements of tissue- and non-tissue-engineered CMSs mimicking cardiac contractility-based sensors, actuators, and reactors with functional structural designs for cell responses. By introducing tunable mechanical and chemical environments to generate functional in vitro cardiac disease models, we will discuss physiological details of the heart and the required engineering design for CMSs. Furthermore, limitations of existing integrated systems, such as tissue structure, blood flow, multiple cell types, electrical conductivity, contraction, and relaxation, will be highlighted as future challenges for fully integrated biomimetic in vitro CMS.
Cardiac Physiology Guiding Engineering Design
The heart is an electromechanical pump that circulates blood to the whole body. The cardiac pump requires three main functionalities to complete each cycle of blood pumping: cardiac excitation by electrical signal generation, mechanical contraction or dynamic deformation of heart muscle, and electrochemical coupling of excitation-contraction by intracellular calcium transients.
The cardiac cycle is controlled by action potentials from the electrical conduction system of the heart along with a pressure gradient across the different chambers. At the beginning of the action potential, pacemaker cells from the sinoatrial node of the heart are depolarized, which initiates current propagation down the septum and throughout cardiomyocytes down the ventricle walls. Electrical conduction results in the contraction of the ventricles, closure of the mitral and tricuspid valves (MV and TV, respectively), and opening of the aortic and pulmonary valves (AV and PV, respectively). 33 The development of contraction force in the heart depends mostly on calcium ions, known as direct activators of myofilaments. 34 Physiological contraction generates both isometric force (and ventricular pressure) and rapid shortening to eject blood. In simple terms, after a certain wall region is electrically activated to undergo systole, it undergoes transition from radial thinning to thickening and from longitudinal lengthening to shortening, which in effect causes diastolic relaxation and systolic contraction. 35 During the cardiac cycle, the four heart valves are essential components to control hemodynamic environment. The heart valves consist of mainly valvular leaflets and chordae tendineae that exhibit a highly nonlinear stress-strain relationship, large deformations, and complex viscoelastic and axial coupling behavior, with elastic moduli in the range of megapascals.36–39 Under systolic and diastolic pumping cycles, heart valves encounter three different mechanical loading modes such as flexure, shear, and tension, as shown in Figure 1 . Both flexure and shear stress occur during valve opening, while flexure along with tension occurs during valve closing. The tension in leaflets during closing is necessary for coaptation, which prevents leaflet overdeflection and regurgitation. Accurate values of these loads are not clearly known, but estimates for average tension are 50 to 100 N/m, shear stress on the AV leaflet is 10 to 80 dyn/cm2, and flexural stress can vary depending on the leaflet angle. 36 According to a study performed by Mirnajafi et al., 40 at a flexure angle of 30 degrees, moduli of ~43 kPa in the forward direction and ~75 kPa in the reverse direction were found.
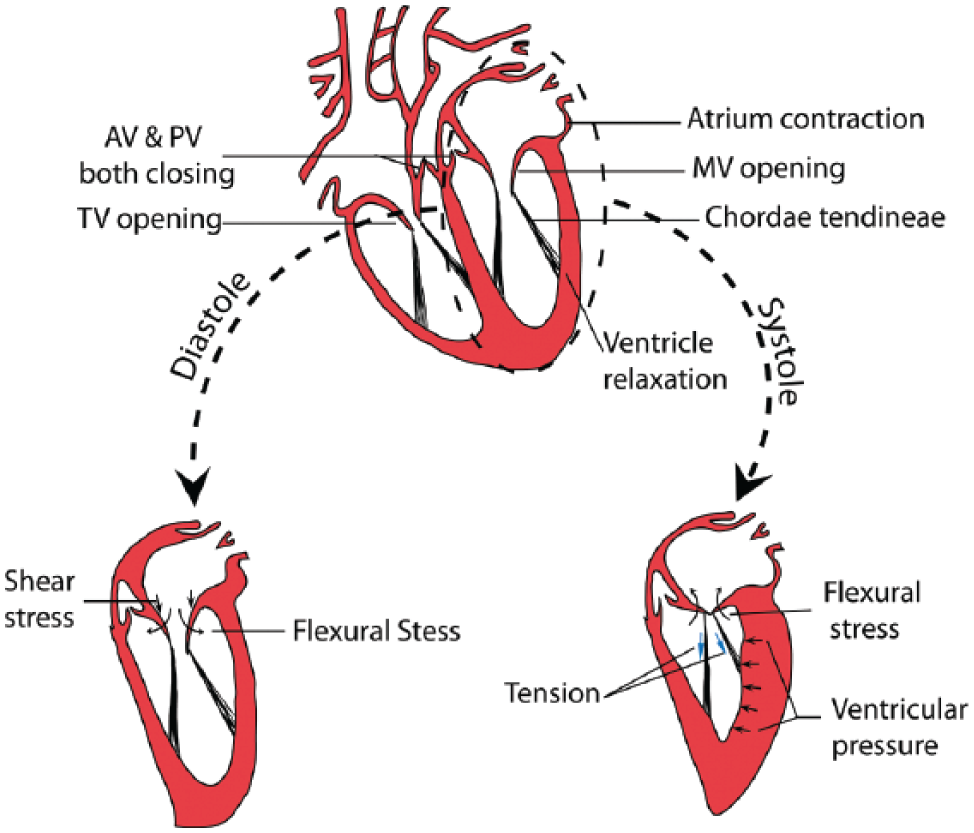
Heart physiology: muscle contraction and relaxation during systolic and diastolic cardiac cycle, fluid structure interaction, and three basic mechanical loading modes—flexure, shear, and tension—encountered by valves. AV, aortic valve; PV, pulmonary valve; MV, mitral valve.
The biomechanical uniqueness of the heart is tightly related with various cell types such as pacemaker cells in the nodal regions, smooth-muscle cells in vessels, fibroblast-like cells in the heart valves, and endothelial cells (ECs) lining various tissues. Physiological responses of the heart response to neurotransmitters and mechanical loads are different from each cell type and need to be distinctly interpreted by individual cell types. As a consequence, it is complicated to re-create such an organized system in vitro. In addition, each cell type mentioned above is responsible for a specific set of physiological properties of the heart. Cardiomyocytes constitute the main striated muscle cells contracting with each heartbeat. Their functional role is verified with the expression of actin, myosins, and troponin, among other sarcomeric contractile proteins.23–26,36,41–44 Their responses to digitizer drugs, such as digoxin, are of interest when studying CMSs as well.6,23,24 Pacemaker cells are difficult to isolate, and so far no CMS can replicate their ability to generate spontaneous action potentials. Smooth-muscle cells are also contractile but respond to neuronal stimuli in a unique manner, due to specific ion channels, which distinguishes them from striated muscle cells. In addition to cellular heterogeneity, the design of CMSs takes into account the extracellular matrix of various cardiac tissues. For example, striated or smooth muscle requires a tight packing of cells that communicate via gap junctions, to allow for efficient electrical conduction. Other tissues, such as heart valves, primarily composed of connective material, present low cell density and a vast network of collagens, elastins, and proteoglycans in their extracellular matrix. The composite layer, consisting of those macromolecules, shows biphasic mechanical properties. The cellular heterogeneity and complicated extracellular components make it very challenging to realize CMSs recapitulating such extracellular network structure and biomechanical properties.
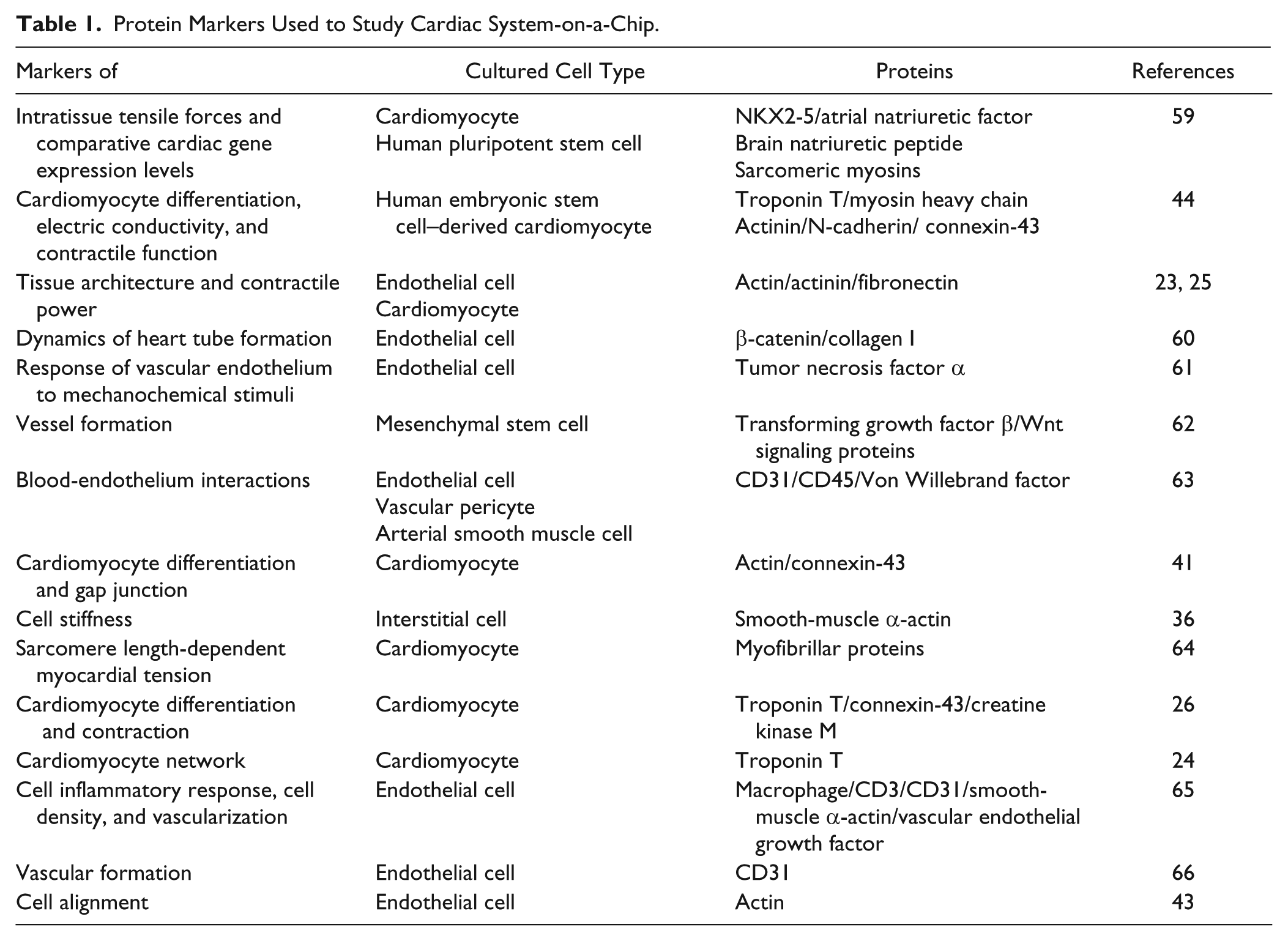
Several attempts of creating biomimetic cardiac microsystems to study disease models were reported, such as a contractile myocardium-on-a-chip mimicking electromechanical interactions of the cardiac pump.23,24,26,27,31 Examples include a microspherical heart-like pump that can generate pulsatile flow, 32 a tissue-engineered pump with valving structure,28,30 and a non-tissue-engineered cardiac pump and valve.25,29 From each CMS, various biomarker proteins as shown in Table 1 have been screened to confirm the effects of distinct chemical and mechanical factors. However, none of the studies incorporates all of the complex characteristics exhibited by heart muscle tissues, including electrical wave generation, calcium-triggered muscle contraction, valve function exhibiting physical loading characteristics, and non-Newtonian blood flow.
Protein Markers Used to Study Cardiac System-on-a-Chip.
Cardiac System-on-a-Chip
As shown in Table 2 , three different types of recently developed CMSs are categorized, including biomimetic contractile myocardium-on-a-chip, tissue-engineered bio-hybrid actuator, and cardiovascular system in vitro with non-tissue-engineered cardiac pump and valve.
Comparison of Current Developed Cardiac Microsystems.
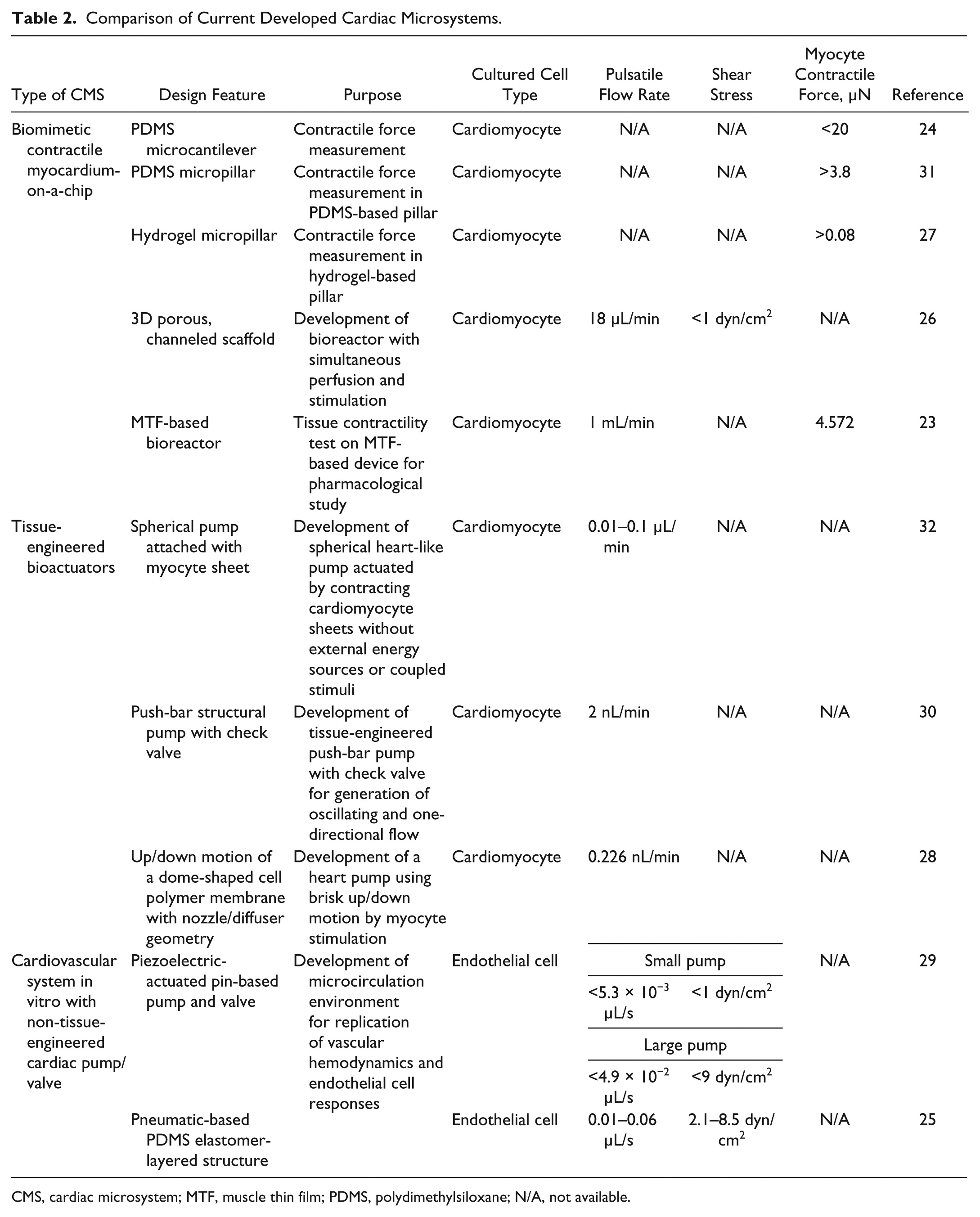
CMS, cardiac microsystem; MTF, muscle thin film; PDMS, polydimethylsiloxane; N/A, not available.
Biomimetic Contractile Myocardium-on-a-Chip
Mechanical contraction of cardiomyocytes is influenced by substrate stiffness in a conventional 2D cell culture technique, while 3D tissue-engineered matrix has improved architecture that can be less affected by substrate stiffness and directly be incorporated with 3D cellular migration. 45 The 3D tissue-engineered matrix also enables mimicking cardiac contraction like in vivo environments by integrating physiological and mechanical stimulations. For investigating contractile capability of cardiomyocytes by mimicking electromechanical interactions of the cardiac pump, various 3D tissue-engineered CMSs have been demonstrated. 24 As a common implementation method, cardiomyocytes were attached onto a 3D micropatterned matrix, which consists of an array of two polydimethylsiloxane (PDMS) microcantilevers, as shown in Figure 2A . Cardiomyocytes cultured on microcantilevers were activated by electrical stimuli, and the resulting contraction forces were measured from deflections of the microcantilever. In addition, contractility and synchronized beating response by triggering isoproterenol and digoxin were evaluated. 24 Both isoproterenol and digoxin stimulate cardiac muscle contraction by activating the conversion of adenosine triphosphate (ATP) to cyclic adenosine monophosphate (cAMP),46,47 along with sarcoplasmic reticulum calcium ion signals, to generate positive inotropic cardiac glycosides. 24 With increased concentration of isoproterenol from 1 to 10 µM, beating frequency was increased up to 1 Hz without significant contractile force change. Digoxin showed up to a 23% increase of contractile force from 1 nM to 1 µM. However, higher than 10 µM of digoxin showed cardiotoxicity with no contractile force change, but beating frequency dropped to under 0.04 Hz from 0.5 Hz.
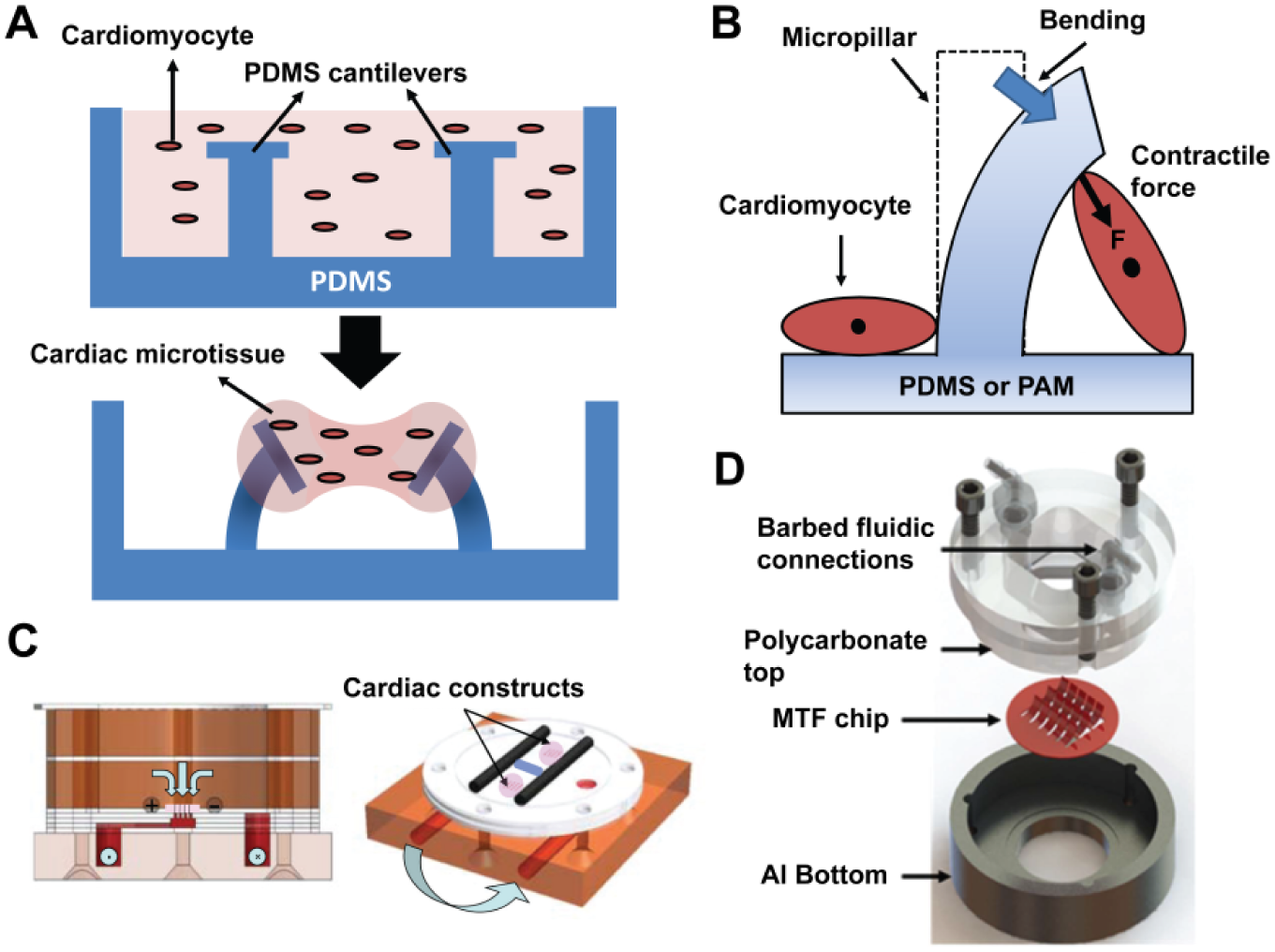
Biomimetic contractile myocardium-on-a-chip: (
Similar devices containing polymeric micropillar structures were fabricated to measure a contractile force without electrical stimuli.27,31 Figure 2B presents the design of polyacrylamide (PAM) hydrogels and PDMS micropillar structures. Cells were grown on the micropillar arrays, where they generated contractions triggered by exogenous glucose. The contractile force was then evaluated by measuring the mechanical bending stress of each micropillar. The values of the contractile forces from cardiomyocytes on PAM and PDMS were >0.08 µN and >3.5 µN, respectively. The difference in contractile force between PAM and PDMS micropillars was due to the number of anchored cells and cell-adhesive promoters on each surface.
In addition to various electrochemical environments of cardiac contractility, myocardial contractile functions are also affected by a perfusion condition enhancing hydrodynamic shear and nutrient delivery.48,49 To provide both nutrient perfusion and electrical stimulation, Maidhof et al. 26 developed a cardiomyocyte-engineered reactor for improving normal myocardial functionality under an in vitro culture system. Figure 2C presents channeled porous elastomer scaffolds for perfusion and uniform cardiac tissue formation. Two parallel carbon rods serve the scaffold as the positive and negative electrodes for electrical stimulation. The elastomers are placed on two bioreactors, while each bioreactor has two tissue constructs. Culture medium is pumped through a tube via an external peristaltic pump to generate a pulsatile flow and returned to the bath via a separate tube in the bioreactor. This device enables simultaneous culture medium perfusion and electrical conditioning during culture of engineered cardiac constructs, which allows the cultured tissues to freely contract in the reactor. This electromechanically coupled device enhances organization and functionality of engineered cardiac tissue and improves contraction amplitude by actively controlling electrical stimulation with perfusion. 26
To increase throughput of electromechanically coupled devices, cardiac muscle thin films (MTFs)–on–chips were developed for pharmacological response studies of contractility of cardiomyocytes.23,50 The MTF fabrication started with glass coverslips, which were taped with a rectangular-patterned low-adhesion tape. The glass was then coated with a thermosensitive poly (N-isopropylacrylamide) (P-NIPAAm), enabling MTFs to be peeled up from the surface by dissolving the P-NIPAAm below 37 °C. After removing the tape, only P-NIPAAm was left on the glass. By spin coating PDMS on the glass, a 10- to 30-µm-thick PDMS layer was obtained. Using a laser cutter, rectangular shapes were cut to create multiple PDMS thin-film cantilevers. To culture cardiomyocytes on the PDMS, multiple strips with 0.3-mm-wide and 1.2-mm-long fibronectin patterns were printed with a microcontact printing technique.51,52 The first MTF device had eight cantilevers cultured with cardiomyocytes in a temperature-controlled bath. Epinephrine, a standard chronotropic agent, was then tested with the MTF device to investigate pharmacological effects. As the concentration of epinephrine was increased from 10−9 M to 10−6 M, the frequency of contraction of MTF was increased by up to four times. 50 An advanced platform was developed to maintain viable culture conditions and control fluid flow in vivo tissue architecture. 23 By using embedded electrodes connected to the inlet and outlet of channels, various contractility tests were performed to determine stress, electrophysiology, and cellular architecture under diastolic and peak systolic motion. Under the dynamic motion of MTF, agonistic effects of isoproterenol on contractility were successfully demonstrated with the array of MTF. The results showed that MTFs at the range of 10−5 M to 10−4 M isoproterenol concentration could generate significantly higher contractile stress (about 40% to 50%) compared with drug-free MTFs.
Various CMS platforms to measure contractile forces under tunable electrophysiological, biochemical, and mechanical environments have been introduced in this section. Such cell-based in vitro force measurement concepts offer advantages of investigating quantitative contractile properties of single cells and multicells without nerve or blood supply. Since evaluation of mechano-physiological responses of cardiomyocytes is significant for the development of drugs and toxicity studies, those in vitro systems can be used to assess contractile function of myocytes for the case of hypoxia or heart failure. Furthermore, the engineered cardiac tissues discussed in this section can also be used as an artificial cardiac patch to replace or repair damaged cardiac cells.
Tissue-Engineered Bioactuators
Contractility of in vitro cultured cardiac tissue has been used by Tanaka and colleagues 32 to create a tissue-engineered biohybrid actuator with the concept of tissue contraction and relaxation in a 3D ventricle-like structure. This pump was powered by spontaneously contracting cardiomyocyte sheets without a need for an external energy source or stimulation. After culturing cardiomyocytes on the P-NIPAAm–grafted surface at 37 °C, the cardiomyocyte sheets were detached from grafted surfaces without any damage to cells by a temperature change to 20 °C due to thermally switchable surface wettability of P-NIPAAm. The sheets were then transplanted to the surface of an elastomeric device to create a tissue-engineered pump. The pump was fabricated by wrapping a beating cardiomyocyte sheet exhibiting large contractile forces around a fabricated hollow elastomeric sphere (5-mm diameter, 250-µm thickness polymer), which was fixed with inlet and outlet ports, as shown in Figure 3A . The tissue-engineered pump was immersed in the cell culture medium, and fluid oscillations in the capillary connected to the hollow sphere were observed at 37 °C, which was considered a key culture environment for myocyte self-actuation. This device was unique because it mimicked the ventricular environment rather than just generating contractile force. In addition, unlike other electric, piezoelectric, pneumatic, and thermal force–regulated micropumps, the tissue-engineered pump can generate a flow in the range of 0.01 to 0.1 µL/min without external power sources.
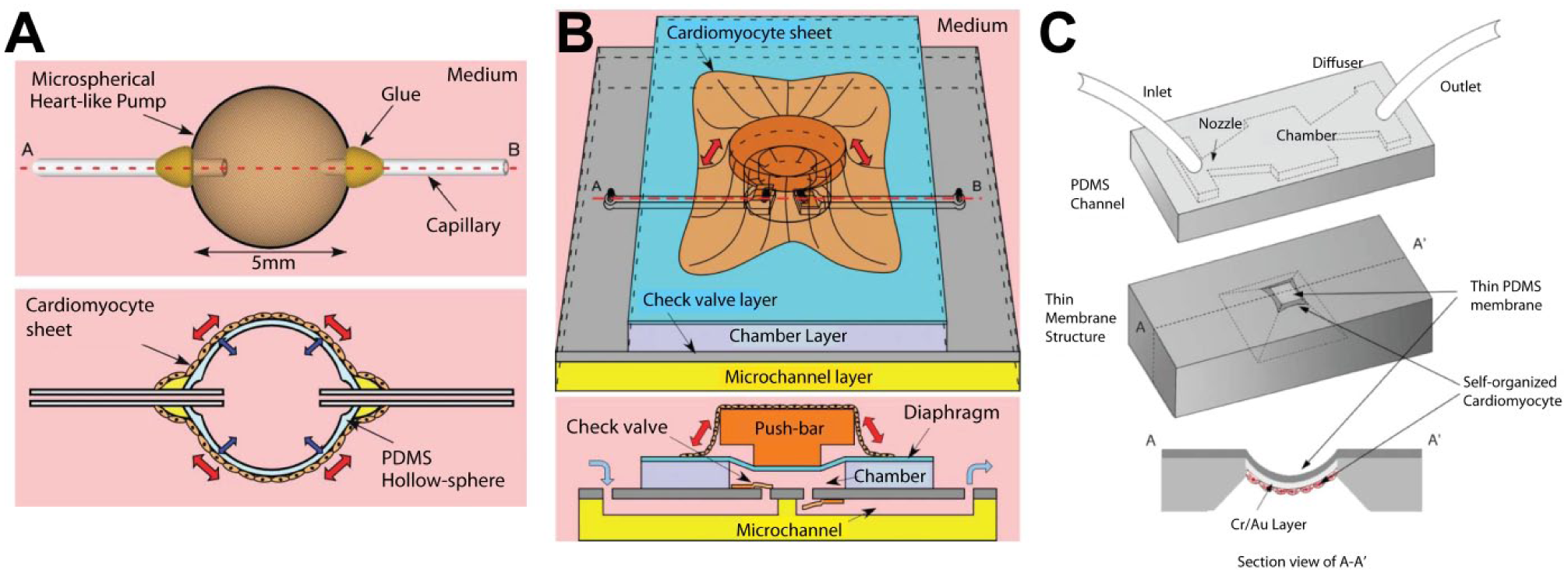
Contractile tissue-engineered bioactuated pumps. (
A follow-up study introduced a tissue-engineered pump integrated with a valving structure that generated a few micro Newtons of force per cell. 30 Figure 3B shows a tissue-integrated hybrid push-bar structure that is actuated by a responding cultured cardiomyocyte cell layer based on temperature change. Similar to a previous study, 32 an increase of incubation temperature increases the frequency of cardiomyocyte contraction. A diaphragm and a push-bar structure were designed in the microchip to transfer contractile forces of the myocyte sheet to the fluid to produce an oscillating microchannel flow. A valve layer was placed between the chamber and microfluidic channel, while 25-µm-thick rectangular cantilever-type micro check valves (700 × 800 µm) were fabricated to generate one-way fluid within the microchannel. The device was immersed in aqueous cell culture medium to supply both glucose and oxygen, which are the energy sources for cardiomyocytes. Fluid movement in the microchannel was visualized by a particle-tracking method using phase-contrast microscopy. For device verification, both fluid displacement and frequency were tested by pump actuation with temperature control under valve and valveless conditions. Culture temperature was increased from 30 to 40 °C using a thermoplate at a rate of about 0.3 °C/min. As the culture temperature increased, cellular contraction frequency was increased while stroke displacement was reduced. With the valve structure, 2 nL/min of directional flow rate was generated. This integrated platform demonstrated the possibility of a tissue-engineered pump enabling flow direction control.
Another integrated form of a tissue-engineered diaphragm pump and valve structure in fluidic device was introduced by Park et al. 28 A dome-shaped cardiomyocyte-polymer diaphragm was devised to create a vertical motion of a diaphragm. The device enables the generation of a brisk vertical motion of cells on the surface to mimic the movement of a cardiac chamber. The outside of the diaphragm was coated with fibronectin and gelatin, onto which cardiac myocytes were seeded to create the monolayer of cardiac cells. When the diaphragm was actuated by the contractile pulsation of spontaneously and autonomously beating cardiac myocytes, the volume of the microchannel chamber shrank and relaxed periodically, resulting in a fluid flow. The top layer of this device is a PDMS-based microchannel layer supporting two passive microvalves—namely, a nozzle and a diffuser. Unidirectional flow was controlled by the fluid resistance generated by the nozzle/diffuser combination, as shown in Figure 3C . Both the push-bar and the dome-shaped pump commonly employ the cardiomyocyte-engineered pump with valve structure without an external energy source.
These tissue-engineered bioactuators provide a great tool to study electrochemical signaling pathways related to cardiac contraction. Utility of these bioactuating platforms can be expanded to medical implant devices such as implanted hybrid bio/artificial tissue-engineered organs and advanced drug delivery devices without any external power sources. Furthermore, dynamic responses of the cardiac cells to various environments such as pH change, drug effects, and pathogenic infection can also be studied systematically by controlling all variables precisely.
Cardiovascular System In Vitro with Non-Tissue-Engineered Cardiac Pump/Valve
Previously described tissue-engineered contractile pumps demonstrated pumping capabilities without an external energy source. However, for a tissue-engineered pump, the generation of force and pump size can be restricted by the cell number: single cells can generate only contractile force on the scale of micro-Newtons or less. 30 In addition, myocytes are required to be contained in media under a controlled environment. Adapting a microfluidic pump into microscale culture platforms can resolve these drawbacks and provide highly parallel fluidic control to investigate the effect of vasculature formation under a reconfigurable hemodynamic condition.
A mechanically actuated microfluidic actuator is the most common system, which can be easily fabricated by multilayer PDMS to support the microscale culture platforms. A multiple elastomeric microfluidic pump actuated by piezoelectric pins was developed to generate the pulsatile flow profile in cultured microchannels. 29 A computer-controlled three-pin peristaltic pump (sequenced by push-up and push-down motion) actuated media through the cultured microchannels. By controlling the size of the actuating chambers, this device could generate a range of 1 to 12 dyn/cm2 shear stress under various pumping frequencies to vascular ECs in vitro. Under the ~12-dyn/cm2 shear stress condition, the vascular ECs aligned and elongated in the direction of flow. However, to encompass physiological shear stress levels (5–20 dyn/cm2), more versatile and reliable pumps are required to investigate the effect of shear stress widely.
To mimic a more versatile range of shear stress level in a microfluidic network, pneumatically actuated microfluidic pumps were used. 25 In this study, a high-throughput multilayered pneumatic actuator creates the cardiac pumping profile connected to microcirculation for replication of vascular hemodynamics and vascular EC responses in a 3D microfluidic environment. Figure 4B shows a three-layered device structure, including the pneumatic layer, PDMS membrane with semicircular valve layer, and rectangular fluidic channel layer. To eject fluid similarly to a ventricle, 16 parallel rectangular channels called “ventricles” were designed. Valve movement and ventricular contractions were actuated by microprocessor-controlled pneumatics to mimic the cardiac cycle. By creating closed-loop microchannel networks, media were pumped to recirculate cultured channels to provide a biomimetic microcirculatory network. For pneumatic actuation, a normally open valve structure was adapted, as shown in Figure 4B . An 80-µm thin membrane was deflected by pressure, and the deflected membrane prevented the flow. Three sequential steps allowed generating an arterial-like pulse waveform to apply a proper range of shear stress. Under high shear stress (τ = 0.85 Pa), both elongation and alignment of the cell were correlated with the direction of flow. Under low shear stress (τ = 0.33 Pa), only cellular elongation was investigated to the direction of flow. The results showed that transient shear stress is critical for vascular physiology and remodeling. 25 However, there are significant discrepancies between actual shear stress pulse wave in arteries (~3 Pa) 53 and the tested shear stress (~0.85 Pa). To overcome the difference between shear stresses in vivo and in vitro, fluid volume, fluid resistance scaling, and blood surrogate/media viscosity must be considered. Only then can replication of shear stress and miniaturization of the cardiovascular environment in microfluidic device be achieved.
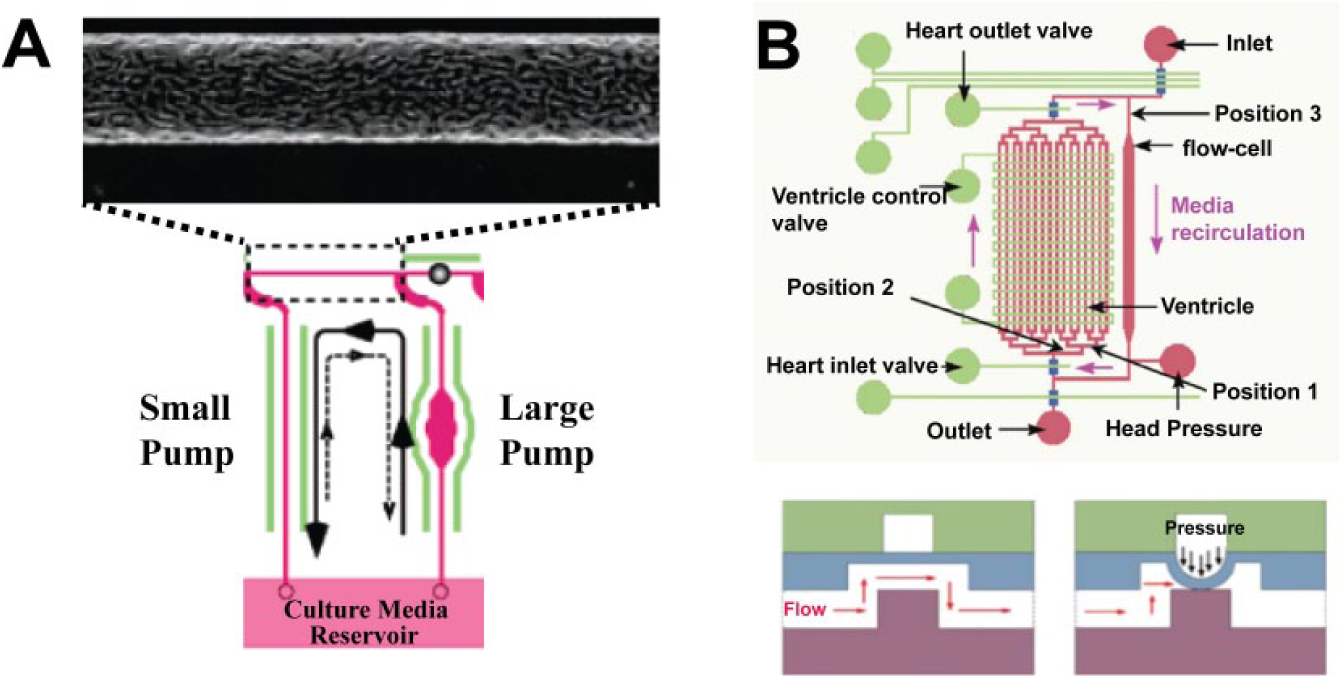
Microcirculatory system for cardiac-like shear stress effect for response of endothelial cells (ECs). (
In summary, CMSs with microfabricated cardiac pumps and valves have been developed to control mechanical shear stress under a pulsatile flow regime. By culturing EC cells in the devices, the effect of shear stress has been investigated to generate various disease models related to shear stress. Further integration of microvalve and pump technologies54,55,56 allows replication of a microcirculatory pulsatile hemodynamic model enabling the mechanism studies of cellular phenotypic alternations and cardiovascular diseases. With the integrated in vitro model, the effectiveness of cardiovascular medications under well-controlled environments can be evaluated.
Outlook
The field of cardiac systems-on-chips is still in its infancy due to highly dynamic environments. Much work had been done before coining the term and not necessarily incorporating microfluidic devices. Many questions remain unanswered, primarily with regard to integration of multilevel systems. The heart alone needs levels of integration of pacemaking cells, as well as major artery and vein systems, before one can study functional diseases on-a-chip.
In this review, we discussed CMSs, including cardiac excitation-contraction coupling with pulsatile pumping in a perfusive 3D microfluidic environment. For replication of additional heart functions, an engineered heart valve is essential. Since heart valves are composed of noncontractile connective tissue and function passively due to pressure, on-chip systems including valve/pump and fluidic chambers need to be integrated with the mechanical environment, primarily shear flow and flexure. A valve-on-a-chip model is innovative as a heart valve is a functional organ but cannot be modeled as a standalone system. This system needs to account for turbulent flow patterns as well as potential exchange of cells and small molecules within the bloodstream. The mechanochemical interactions of valvular interstitial cells and ECs with the bloodstream constitute novel hypotheses to be pursued in the investigation of valvular disease pathobiology. Fluid flow and other biomechanical stimulation of cardiac tissues are integral to a variety of the heart’s intrinsic control mechanisms. Moreover, the hemodynamic environment, along with viscoelastic behavior and a nonlinear stress-strain relationship of valve leaflets, requires more attention in selecting material and media for an in vitro disease model. Blood, a non-Newtonian fluid, exhibits more different fluid flow characteristics than normally used media in the disease models mentioned in this review. The viscosity of blood is determined by plasma viscosity, hematocrit, and mechanical properties of red blood cells. As such, the viscosity of blood varies inversely with shear rate. As a result, heart valves encounter drastic changes from highly viscous to less viscous fluid. This phenomenon should be addressed in the in vitro drug testing models by using blood or similar fluid; otherwise, drug kinetics and dynamics can be misleading. Despite all of the challenges, biomimetic organs in the microscale will lead the way into studies of new drugs, simulations of physicochemical processes in the body, signaling mechanisms leading to diseases, metabolic responses, and synthetic biology approaches for personalized medicine. The ultimate goal of designing a personalized body-on-a-chip4,57,58 is tangible, and the race toward integration of multiple organs exists.
Footnotes
Acknowledgements
This work was supported by new investigator start-up funds from Texas Tech University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
