Abstract
For centuries, animals have been used in research due to their genetic and physiological similarities to humans. However, significant differences exist between humans and animals, which have the potential to confound results obtained from such experiments. These differences result in reduced translatability of animal data to humans, which is a major contributing factor to the 92% failure rate for novel therapies in clinical trials. Advances in scientific research have enabled the development of human-focused New Approach Methodologies (NAMs), which include in silico and 3-D in vitro models. By harnessing these novel approaches, greater predictive power for human biology, human diseases and assessment of novel therapies could be achieved. However, several obstacles remain to their wider adoption, including potential financial constraints, publication bias, and some concerns about the reliability of NAMs due to the novelty of this field, compared to animal studies. Here, we outline the differences between humans and animals used in research, discuss in detail the obstacles to the greater adoption of NAMs in research, and provide recommendations on how to accelerate a shift toward human-focused research.
Introduction
Animals have been used in scientific experiments in the UK since the 17th century, 1 as they share the same vital organs and have a high level of genetic homology with humans. However, there are significant physiological and genetic differences between animals and humans, which impact the translation of research results between species.2–5 One salient consequence of this is the failure of more than 92% of drugs during clinical trials, mostly due to issues with safety and efficacy not predicted by animal data.3,6 These late-stage failures result in an average cost of USD $1300–$4000 million (£980–£3030 million) to bring a new therapy to market,7,8 meanwhile some therapies are shelved which could have shown success in humans. 9 This highlights the importance of progressing towards more predictive, human-relevant research and testing, such as New Approach Methodologies (NAMs). NAMs are defined as in silico, in chemico or in vitro models which could provide greater human relevance than current animal models.10–13
Despite concerns regarding the human relevance of animal experiments,14–16 a high level of animal-based research continues worldwide. In the UK, approximately 2.68 million animals were used in research in 2023: 1.47 million within experimental procedures, and the remainder for producing animal lines with genetic mutations. 17 Frameworks are in place to protect animals in research, including Directive 2010/63/EU, which was put in place by the EU and states that where there is a recognised, validated non-animal alternative, animal experiments should not proceed. 18 Additionally, in countries around the world including the UK, USA, EU and Brazil, researchers are encouraged to consider the Three Rs principles — replacement with alternative methods, reduction in the number of animals used, and refinement of experiments to reduce lasting harm — when applying for animal studies licences. 19 However, some argue that the implementation of Directive 2010/63/EU and the Three Rs principles could be improved. 14 Furthermore, focusing on refinement and reduction can hinder focus on replacement, which impacts and potentially delays fulfilment of Directive 2010/63/EU’s final aim of animal replacement. 20
This primary focus on refinement and reduction by many researchers may be because they believe that full replacement of animal use is not yet feasible. A survey in the Netherlands regarding a goal set by the Dutch Government, which aimed to increase animal-free research, revealed that approximately 71% of researchers who used animals did not believe that replacement of animals was possible in the near future, though 40% of respondents indicated that they would support replacement. 21 A 2013 survey in Portugal revealed that approximately 70% of responders believed that some experimental steps may be replaced by NAMs within 50 years. 22 Furthermore, a survey of US-based researchers showed that 77% of responders did not believe that NAMs would sufficiently replace animals in their research. 23 However, the survey also revealed awareness of reproducibility issues in animal models due to multiple factors, including lack of detail in animal research publications, and variation between the animals used. Taken together, this highlights that reproducibility within animal models is a significant issue, and that some researchers are open to the idea of NAMs, if these could provide more predictive and human-relevant results compared to animal studies. Hence, this review examines key issues with animal studies, including: the differences between humans and animals; factors affecting the extrapolation of animal data to human disease research; and current NAMs which have the potential to reduce or replace animal studies. Furthermore, obstacles to NAM research will be discussed, and a roadmap to accelerated NAM uptake will be outlined.
What are the main scientific issues associated with animal use in research?
Translational issues due to genetic and physiological differences between humans and animals
Animals are commonly used in research as they are complex organisms which share some physiological and genetic features with humans. For example, mice and humans share 85% gene homology. 24 However, although a high proportion of genes are conserved, significant genetic variation exists between the species, and homology within non-coding regions can be lower than 50%. 24 Non-coding DNA regions include sequences that can influence transcription of coding regions, which may affect the level and type of gene product.25,26 Differences in non-coding regions between humans and mice can therefore result in discrepancies in protein structure and function between these species, highlighting that gene regulation and expression are as important as genetic homology. An example of this is the cytochrome P450 (CYP) enzyme superfamily, which is responsible for the metabolism of approximately 80% of all drugs in humans. 27 As these genes are highly conserved, small amino acid changes between species may result in significant differences in structure and/or function. For example, the human enzyme CYP2A6 exhibits different functions compared to the murine orthologue.28,29 Such differences are widespread and negatively impact the translatability of animal data to human safety and toxicity testing.
There are also many physiological differences between humans and mice, including — but not limited to — skin,
30
the gastrointestinal (GI) tract,
31
heart size, output and anatomy,
32
metabolic rate,
2
the immune system,
33
and the nervous system
34
(Figure 1). Taking the nervous system as an example, structural differences are present between human and murine brains,
34
such as the absence of furrows and ridges on murine brains,
42
murine motor neurons functioning differently to human motor neurons,43,44 and differences in pineal and pituitary gland structure and function.34,45 Despite these differences, approximately 1.9 million mice collectively were used in experiments and breeding in the UK in 2023.
17
A comparison of mouse and human anatomy, immunology and biochemistry. This diagram outlines key differences between several key organs in mice (left) and humans (right). These organs include (as listed top to bottom): brain and CNS (blue); heart (red); lungs (pink); gastrointestinal tract (tan/brown); pancreas (yellow); and immune system (leukocyte production by bone marrow and lymphatic system (green).35–41 Image created with BioRender.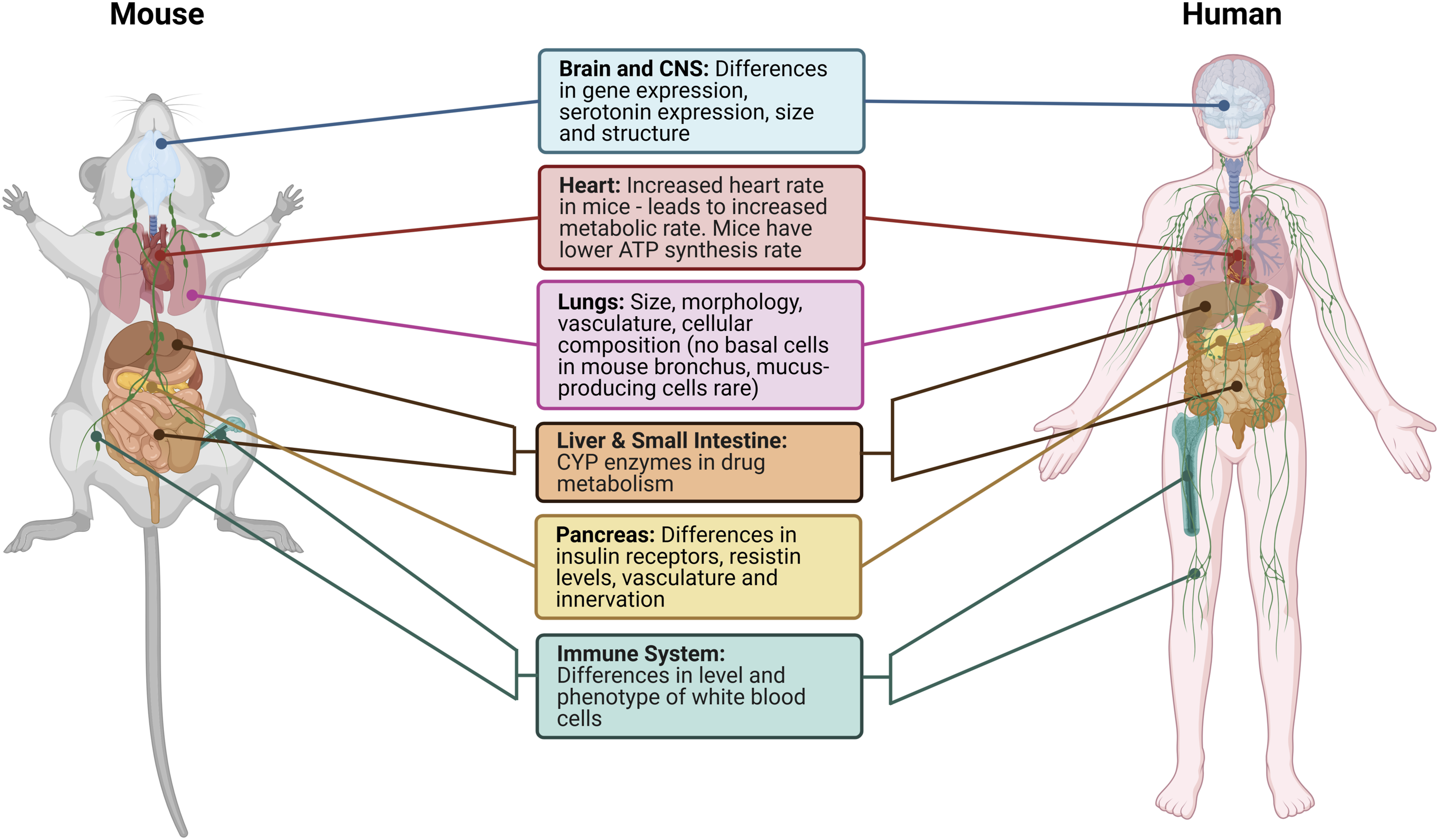
Non-human primates (NHPs), such as rhesus macaques and cynomolgus macaques, are often selected for research due to their high level of genetic and anatomical similarity to humans. In fact, over 1800 NHPs were used in experimental procedures in the UK in 2023. 17 However, genetic homology between humans and rhesus macaques is only 90.76%. 46 Since our evolution from NHPs, human genomes have accumulated many human-specific mutations, including segmental inversions, duplications and translocations,4,47 which can affect gene expression due to the new position of these genes in relation to promoter and enhancer regions,4,5,48 and the evolution of new genes in humans and NHPs, accompanied by the loss of less beneficial genes. 49 This level of genetic variation can lead to significant phenotypic differences, which restricts the reliability of the extrapolation of data from NHP research to humans. An important example of this is the CYP subfamily, as discussed above. CYP amino acid sequences in cynomolgus macaques and marmosets display 68–97% homology to their human orthologues, 46 but small amino acid changes are known to affect substrate specificity and enzyme activity. 5 This, and other differences in metabolism, impacts the translation of data from toxicology testing to humans, and as a result the absence of toxicity in NHPs does not translate to an absence of toxicity in humans.50,51
Furthermore, humans and NHPs display variation with regard to immune cell phenotype, signalling and composition.52,53 For example, significant differences are present between human and NHP immunoglobulin G and their corresponding Fc receptors, which impact the ability of NHPs to model infectious diseases such as HIV. 54 This, along with differences in disease progression, confounds HIV research in NHPs, and whilst HIV/AIDS vaccines have been discovered which are effective in NHPs, this has not yet translated to an effective vaccine in humans. 55 NHPs are also utilised for research into neuroscience and neurological conditions such as Alzheimer’s disease (AD) due to similarities between human and NHP brains and AD-related genes.56,57 However, whilst age-related amyloid-β accumulation can occur within NHPs, differences remain between human and NHP AD, such as a reduction in phosphorylated tau and reduced cognitive decline compared to humans.58–60 Whilst humans and animals share the same vital organs, the differences between these species can affect the application of animal data to human disease.
The effects of stress and environment on data from animal experiments
Although animals may experience stressors in their natural environments, the stressors experienced in a laboratory setting are different, unavoidable, and potentially more chronic compared to those experienced in nature. 61 Exposure to these stressors results in prolonged activation of pathways which produce and secrete stress steroid hormones. 62 This can lead to increased blood pressure and heart rate, perturbation of adaptive and innate immune responses, and subsequent cardiovascular diseases, cancers and GI disorders, all of which can affect the validity of animal experiments.61,63
Constitutive stress from living outside of their natural habitat, and living in laboratory conditions, can result in epigenetic alterations in animals in laboratories. Offspring can inherit these epigenetic changes, resulting in increased incidence of heart disease, asthma, autoimmune disorders and diabetes, among other disorders. 61 Environmental stressors experienced by animals in captivity, therefore, impact the results obtained through animal experimentation. A computational analysis study of nociceptive sensitivity in mice showed that environmental stressors were responsible for 42% of experimental variability. 64 Environmental stressors in a laboratory setting include: noise of ventilation systems and other stressed animals; 65 artificial light; 66 being handled (and the sex of the person handling them);67,68 observation of other animals undergoing experimental procedures; the seasons; 69 and restrictive housing environments such as cages, which do not allow for natural behaviours such as foraging and nesting.61,66 This can cause many long-term physiological, psychological and epigenetic changes within these animals, limiting the extrapolation of animal data to human disease.61,64
The application of NAMs to reduce and replace the use of animals in research
Many researchers in academia and industry are developing in silico and human cell-based in vitro models, which could have greater predictive power compared to animal research data, and result in reduced or no animal suffering (Figure 2). In silico techniques use previously generated datasets to predict the outcome of an experiment, utilising mathematical modelling and computer simulations.
70
These can be used to predict metabolites produced after breakdown of a new drug in vivo,
79
screen currently available drugs that could be repurposed,
80
and predict toxicity of chemical compounds.
81
Examples of the types of NAMs currently used in research. This diagram illustrates the human model systems that are discussed in the main text.13,70–78 QSAR = Quantitative Structure–Activity Relationship; ADME = Absorption, Distribution, Metabolism and Excretion; PDMS = polydimethylsiloxane. Image created with BioRender.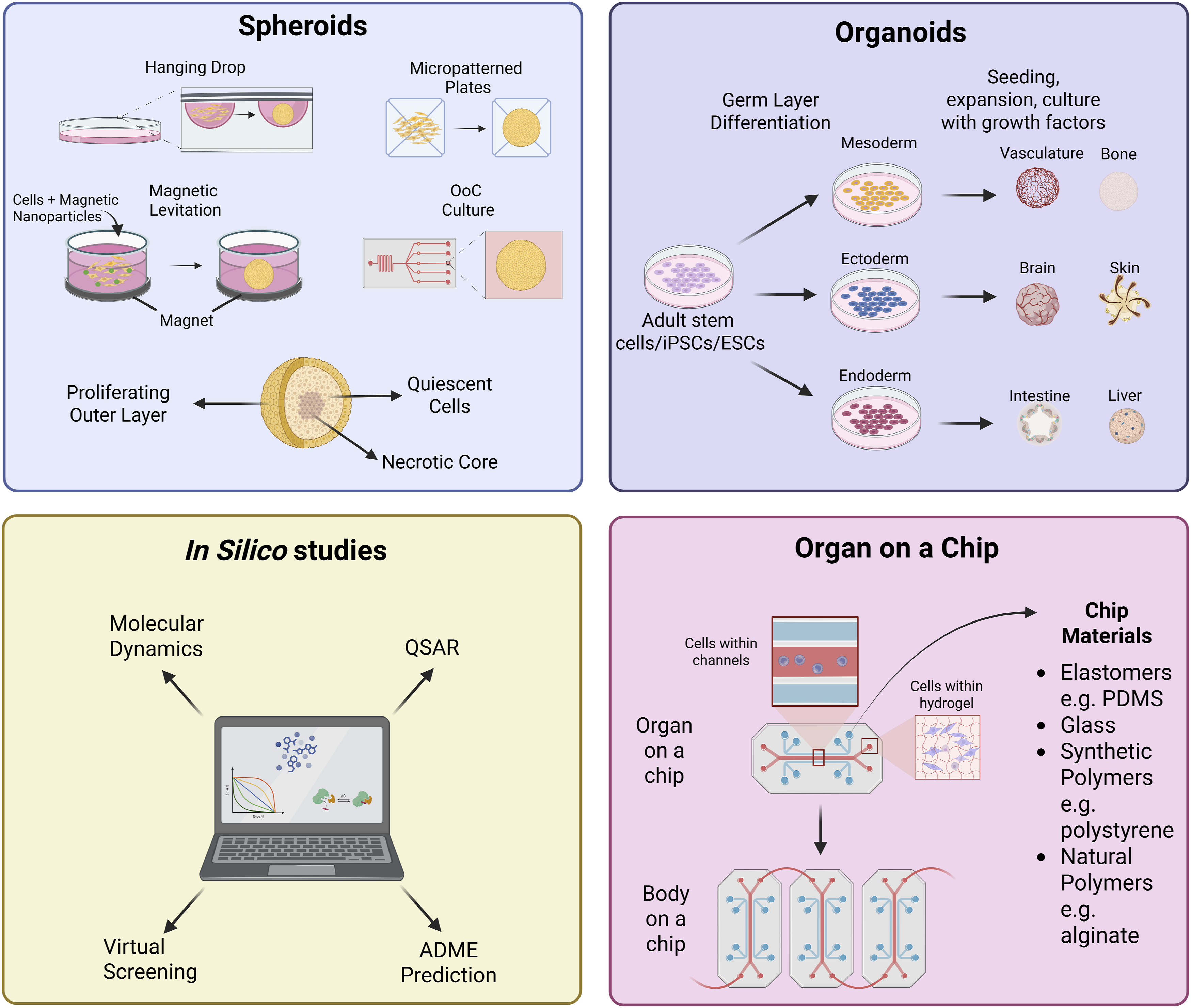
Components of in vitro human-focused models include: hydrogels, which are 3-D hydrophilic polymer networks that can mimic the 3-D tissue environment; spheroids and organoids; and organ-on-a-chip (OoC). Spheroids can be used to model tumours 82 or microenvironments such as bone marrow stroma, 83 due to their cytokine, oxygen and nutrient gradients, which resemble human tissue more accurately compared to 2-D cell culture. 71 Organoids are produced from stem cells, and can self-organise into organotypic structures, resulting in a higher level of structural complexity compared to spheroids. These can be used to model many processes such as organogenesis, genetic disorders and cancer.73,84,85 Organoids and spheroids can be incorporated into more complex models, such as OoC.
A summary of novel organ-on-a-chip (OoC) studies for a range of organs, outlining some of the purposes which the OoC models have been successfully used.
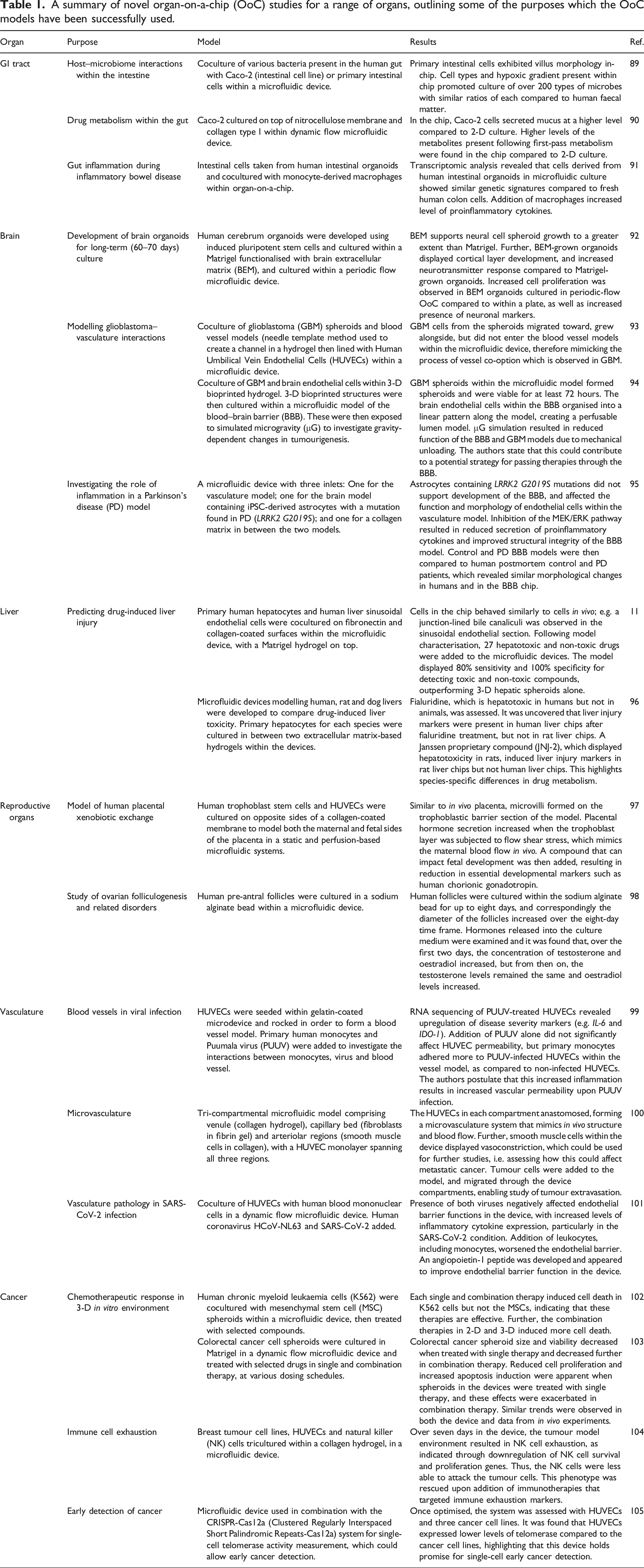
Recently, models of one or more organs have been combined to create a humanised multi-organ-on-a-chip (MOoC).106,107 Many biological processes rely on signalling from other organs, therefore MOoCs can provide more biologically relevant data when multiple organs are included.106,108 Additionally, MOoCs enable cell migration to be visualised, enabling the study of metastatic disease.109,110 The use of MOoCs also permits the assessment of potential off-target effects following treatment with novel therapies, by modelling the organ of interest in combination with other organs.111–113 Furthermore, the combination of multiple methods — such as in silico mathematical prediction models with in vitro simulations in OoCs — has led to the effective prediction of safe drug dosages that eliminate cancer cells with minimal impact on healthy cells. 114 Together, this highlights that novel 3-D models and in silico analysis could have a higher predictive power compared to animal models, and could be used in preclinical testing alongside, or eventually instead of, animals. 10
While NAMs have the potential to reduce or replace animal research and testing, obstacles remain. For example, induced pluripotent stem cells (iPSCs) are frequently selected for in vitro models, as they can differentiate into a myriad of cell types. However, some believe that the process is time-consuming, expensive and has issues with reproducibility. 115 Further challenges include: a lack of standardisation; variation between the NAMs used; potential genetic drift within long-term cell culture; and the fact that NAMs may only be able to recapitulate certain aspects of an organ as opposed to the entire organ.115–117 However, the latter may be overcome by generating several in vitro models, with each model replicating a different aspect. For example, the Organisation for Economic Co-operation and Development (OECD) Test No. 442E utilises three in vitro tests that examine the same aspect of skin sensitisation, but which provide different readouts to determine the effects of the test chemical on the skin. 118
What are the barriers to NAM adoption, and how can we address them?
Concerns about the reliability of NAMs to model the efficacy and safety of novel therapeutics
Some NAMs are relatively new, and animal researchers often believe that some of these new methods may not be as reliable as the existing animal-based methods. 21 Furthermore, many researchers may be reluctant to change to techniques that they are unfamiliar with. 116 A survey of researchers in the Netherlands revealed that ‘reliability’ was perceived as the main obstacle to NAMs uptake, 21 even though animal-based studies may fail to reliably predict whether novel pharmaceuticals will be effective in humans, resulting in 40–50% of clinical trials failing due to a lack of efficacy,119–121 and 20–30% due to toxicity.119–121 Animal toxicity data indicating that a new pharmaceutical is safe for use does not necessarily predict a lack of toxicity in humans.122–124 This not only constitutes a costly clinical trial failure,125,126 but can result in severe side effects and in some cases human harm.127,128 Furthermore, some animal studies cannot be replicated, and this can be due to incomplete experimental reporting, 129 lack of blinding,130,131 low statistical power and in some cases inadequate statistical analyses.130,132 Due to these pitfalls, more humanised and standardised alternatives must be considered.
As discussed in the introduction, researchers may have doubts about the ability of NAMs to replace animals in academic research.21–23 However, surveys have indicated that researchers would be willing to switch to computer-based alternatives, if these were more scientifically accurate and were recommended by their colleagues. 133 Several in silico model systems are available, such as GLORY, a software that predicts the structure of metabolites produced when human CYP enzymes metabolise drugs.79,134 This software could detect potentially toxic metabolic products that may not be present in animals, due to interspecies variations in metabolism. Additionally, there have been promising in vitro models that could be more reliable and predictive compared to animal studies, including an OoC DILI model 11 and the MatTek EpiDerm™ model, which is included in an OECD Test Guideline. 135
Additionally, whilst animals are complex systems, this does not necessarily make them the most effective model, as reflected in the high clinical trial failure rate. 136 NAMs also have the potential to be scaled up, especially less complex systems that model specific human-focused interactions, which could be useful for producing reproducible models for disease modelling and drug testing. 136 Recently, NAMs have been used to assess novel therapies, and the resulting data have been included within the clinical trial applications, which were subsequently accepted.12,137,138 This is highly promising and highlights the potential of NAMs to predict efficacy and safety in humans without relying on animals.
Perceived decrease in likelihood of publication
Despite the scientific concerns around animal use in research, barriers to publication may remain for researchers who aim to transition toward purely NAM-based research. For instance, many researchers have experienced ‘animal methods bias’ during the publication process, whereby reviewers ask the authors to provide data from animal experiments, despite sufficient results having been obtained through NAMs.139,140 A survey by Krebs et al. 139 found that 31% of researchers surveyed (n = 68) pre-emptively used animal methods, as they expected reviewers would ask for them, and 44% had been requested to include animal data by reviewers. Despite the relatively small sample size, these results indicate that animal publication bias is present, though further research is required to understand the extent of this. The authors of this review subsequently conducted a workshop, attended by representatives from academia, industry, publishing and animal advocacy groups, to discuss how to mitigate animal methods bias. 141 Potential mitigation strategies included amending regulatory requirements, educating reviewers and editors about NAMs, and promoting the validation and publication of NAMs. Following this workshop, animal-free research advocates from various backgrounds formed the Coalition to Illuminate and Address Animal Methods Bias (COLAAB). Together, COLAAB produced an author’s guide to publishing animal-free research. 140 This is a highly useful publication for researchers aiming to publish animal-free research.
Despite the presence of animal methods bias in publishing, research that harnesses in vitro and in silico approaches is becoming increasingly popular. Taylor et al. comprehensively surveyed current trends in animal and NAM studies across seven research areas between 2003 and 2022. 142 They uncovered that the relative number of publications based on the use of NAMs had increased compared to studies that used animals, as well as studies which used both animals and NAMs. These data are highly encouraging for researchers aiming to utilise NAMs in their research, as it indicates that despite animal methods bias, a high volume of NAM-based research has been, and is being, published.
Reduced availability of funding
Some researchers may be hesitant to move away from animal models because they are concerned that less funding is available for NAMs-based research. This is a valid concern. In the UK, the main research funding body, United Kingdom Research and Innovation (UKRI), has invested £50 million over the last five-year period toward NAM research; 143 however, in 2024, the UK government’s entire research and development budget was £20,400 million 144 (highlighting that more funding is provided to animal research compared to NAM research). The Netherlands has increased its funding toward animal alternatives, but even then, a study revealed that NAMs received approximately half of the funding compared to animal research. 145
Whilst NAM research typically receives less funding compared to animal research, the level of NAM funding is increasing, particularly in the UK, the EU and the USA. For example, a 2013 survey disclosed that only seven EU member states had set aside a specific budget for NAM-based projects. 146 However, a more recent assessment of EU-funded studies on Alzheimer’s disease, breast cancer, and prostate cancer found that 60% of studies did not use animals, and that €1200 million (56%) of funding was awarded to animal-free research. 147 This highlights an increase in NAM research funding within these research areas. Furthermore, in 2024, Andrew Griffith (the UK Minister for Science, Research and Innovation) declared that more funding will be designated for the development, validation and dissemination of NAMs. 148 The UK is currently part of a funding programme with the EU known as Horizon Europe, which has pledged €270 million toward NAM research between 2021–2027.149,150 Whilst this is substantial funding, it is approximately 0.28% of Horizon Europe’s total budget of €95,500 million, and more money would aid a greater shift toward NAM development and uptake. 150 One key issue for worldwide adoption of NAMs is that in lower-and-middle income countries, there may not be a specific budget set aside for NAMs by the government, and the support may not be available for researchers working with non-animal methods.151,152 For example, for the four years up to 2020, 0.2% of India’s Department of Biotechnology funding was designated for NAMs. 153 However, in 2019, the Indian Council of Medical Research (ICMR) announced the development of a Centre of Excellence in Human-Pathway-Based Biomedicine and Risk Assessment, which will conduct NAM research. This could then provide useful data for NAM researchers worldwide. 153
State-funded research councils, independent research charities and animal advocacy groups, such as Animal Free Research UK, also fund research that advances NAM use in research.154–157 Recently, the National Centre for the Replacement, Reduction & Refinement of Animals in Research (NC3Rs), the Biotechnology and Biological Sciences Research Council (BBSRC) and UKRI collaborated to provide £4.7 million funding toward NAM development. 158 Further, the Department for Science, Innovation and Technology has provided £4.85 million toward projects that increase uptake and validation of NAMs. 159 A similar increase in funding has been observed in the USA. The National Institutes of Health (NIH) has commenced a programme known as Complement Animal Research In Experimentation (Complement-ARIE), which has pledged approximately USD $18 million per year to accelerate the development, standardisation and validation of NAMs.160–163 However, as the NIH’s budget is almost $48,000 million, with an estimated $19,600 million designated for animal studies, 164 more substantial change could be achieved with greater funding. 165 Further, the Foundation for the National Institutes of Health (FNIH) has created public–private partnerships, which bring together NAM developers and regulatory bodies in order to accelerate NAM acceptance and/or validation. 166 Together, not only will this funding support researchers currently developing NAMs but, if enough funding is available, this could also increase the rate at which NAMs are validated and standardised. In turn, this could motivate more researchers to select NAMs instead of animals.
Additionally, many industries choose to invest in NAMs. Collaborations between industry and academia could lead to the scaling up and commercialisation of NAMs, 167 potentially resulting in more standardised and cost-effective preclinical research. Market research has highlighted a shift in the market toward 3-D cell culture models, estimating that it will grow at a mid-teen compound annual growth rate from 2021–2028.168,169 Further projections estimate that the global 3-D cell culture market will be worth USD $3600 million (£2860 million) by 2030, 169 with the organ-on-chip sector worth $303.6 million (£238.8 million) in 2026, 170 rising to $630 million (£495.6 million) by 2029. 171 This further highlights the increasing popularity of 3-D in vitro models, particularly organ-on-chip, which could promote greater adoption within research.
Future directions
Replacing animal studies with NAMs has the potential to increase the rate of progress within research, and could provide economic benefits.
172
However, barriers remain: many NAMs are in early stages of research, and it remains that there are few standardised, validated NAMs. Here, we suggest some solutions to overcome existing barriers for academic and regulatory NAM use (Figure 3). Four main considerations for NAM adoption are knowledge, funding, collaboration, and standardisation and regulatory approval. An outline of the necessary steps toward establishing and validating NAMs for use in basic research and regulation. (a) A schematic of the four main themes identified in this review, and ways to promote NAMs under the respective themes. (b) A tri-phasic approach to improving and disseminating NAMs, with some aspects that would be important throughout these phases.115,116,173–176 Image created with BioRender.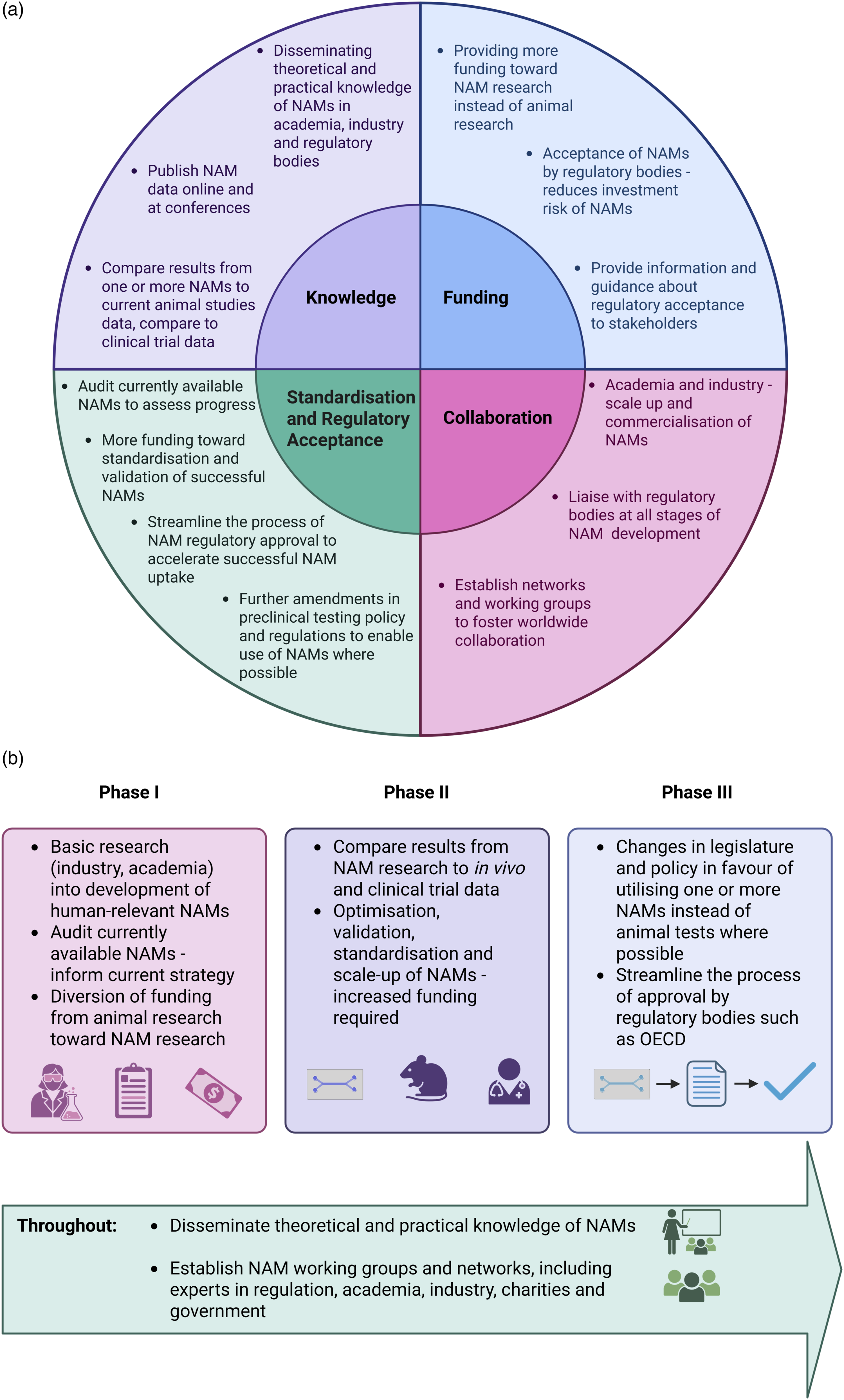
Knowledge
To promote confidence in NAMs, further education into NAMs and their benefits must be provided. Acceptance of NAMs may occur through publication of successful methods and presenting these findings at conferences. More widespread uptake of NAMs could be driven by educating current and new researchers. An interdisciplinary workshop in the Collegium Helveticum in Switzerland discussed four aspects which could further promote education about NAMs: statements from universities about NAMs; knowledge transfer through teaching about NAMs; a push toward NAMs in scientific research; and implementation of ‘transition units’, to ensure smooth transition toward NAMs. 173 One of these areas, knowledge transfer, is currently being addressed by the Karolinska Institutet in Sweden. Here, a ‘ToxMaster’ programme is offered, which covers topics including in silico toxicology techniques and the Three Rs. 177 Universities worldwide could organise similar courses, or assimilate this knowledge into pre-existing courses, allowing the next generation of researchers to drive NAM research. Further familiarisation with NAMs could include organising workshops, webinars, and in-person training sessions such as ‘helpathons’. For example, Animal Free Research UK ran an organ-on-chip Masterclass, which enabled researchers in academia and industry to gain relevant knowledge and experience. 178 People for the Ethical Treatment of Animals (PETA) ran successful workshops and webinars that focused on animal alternatives. 179 Furthermore, in the USA, the 2024 Summer Immersion on Innovative Approaches in Science gathered early career researchers across the globe to provide education and training in NAMs. 180 These learning opportunities could inform current NAM researchers and encourage researchers who do not use NAMs to consider them in their research.
Funding
Greater funding of NAMs development and validation, in both academia and industry, would facilitate the shift away from animal research, as it would enable researchers to develop a greater volume of human-based non-animal methods. Funding programmes (such as Complement-ARIE and public–private partnerships) will help to establish collaborations between academia, industry and regulatory bodies, which could then accelerate development and validation of NAMs in research. However, further NAM-specific funding incentives could drive researchers to incorporate NAMs into their research.
Collaboration
Fostering collaborations between academia, industry, regulatory and government bodies would enable greater transfer of knowledge and resources between each sector, therefore driving the progress of NAMs. For example, the NIH in collaboration with the FNIH is constructing a Validation and Qualification Network, which will allow public and private sector partners to collaborate, with the goal of accelerating the validation and adoption of NAMs. 181 Furthermore, the Indian Council of Medical Research has established the Collaborating Centre of Excellence, which promotes collaboration of research institutions and organisations to advance biomedical research. 182 Through these collaborations, validated human-relevant non-animal models could be developed, which could promote changes in policy to enable these to be used in basic research and regulatory testing.
Standardisation and regulatory approval
Standardising and validating NAMs may encourage more researchers to switch to these methods, as it could increase their perceived reliability. These could then be scaled up and used in drug and chemical toxicity testing if successful. An example of this is organ-on-chip technology, which demonstrates great potential for drug screening and testing, but its ability to reach this potential is hampered by the absence of a clear regulatory pathway in basic research.13,183 There is a lack of standardised NAMs for use in medical research, which could be because after a new method has been developed, the funding and time required for its validation may be unavailable. 184 Choosing to fund NAM-based studies over animal studies may promote the uptake of NAMs in research and regulation. An audit of all available NAMs would enable a full assessment of NAM progress and inform decisions on the optimal strategy to proceed. 174 This has already been completed to an extent in Belgium, where the ‘RE-place’ project has collected all NAMs in one open access database. 185 Ideally, NAM research worldwide could be included on an online, open access database, which would either be free or low-cost to access. This would not only aid the standardisation of NAMs due to the transparency of research, but would also inform low- and medium-income countries on the current available NAMs, therefore promoting international collaborations.
Conclusions
For decades, the ‘gold standard’ in research was considered to be animal studies. However, due to significant differences between humans and animals in genetics, physiology, pathophysiology and more, animal models fail to accurately recapitulate human disease, which could hinder our understanding of these diseases and our ability to develop effective therapies for them. This is further reflected in the high proportion of drugs that fail in clinical trials, having appeared promising in terms of efficacy and safety in animal tests. Human-specific NAMs, including in vitro and in silico models, are gaining popularity as researchers and funders realise that they have the potential to provide cheaper and more accurate data compared to animal studies. 9 However, more must be done to develop and promote these technologies. There remain several barriers to wide utilisation of NAMs, such as lack of validation and standardisation, reduced levels of funding compared to animal studies and some concerns about their reliability. More research must be completed to optimise NAMs, and more funding and greater collaboration within and between countries is essential for their widespread uptake. If the actions outlined in this review are completed, then we can increase the reliability and use of NAMs, which could provide more human-relevant data at an overall cheaper cost compared to animal data.
Footnotes
Acknowledgments
We thank Dr Ross Dobie and Oscar Lavery for their insightful feedback on this review article.
Funding
This work was commissioned and funded by Animal Free Research UK.
Declaration of conflicting interests
We have no conflicts of interest to disclose.
Data Availability Statement
Data sharing is not applicable to this article as no new data were created or analysed in this study.
