Abstract
Low-cost and quick detection of biotic stresses is critically important for protection of staple food crops such as maize in smallholder farms in developing countries, where access to improved seed varieties, fertilizers, and pesticides is limited due to financial and geographical reasons. Here, we report a new lateral flow detection technology directly integrated in a maize leaf, in which microspheres conjugated with analyte-specific capture antibodies are non-invasively injected. The antibody-conjugated microspheres capture and detect an analyte in a concentration-specific manner. In this study, we optimized microsphere size for effective infiltration and immobilization in the leaf, and further demonstrated detection of a fluorescent mock biomarker, fluorescein, in a live maize plant. This in planta lateral flow biosensor is the first of its kind and is expected to provide a low-cost and user-friendly detection method for biotic stresses in the field.
Introduction
Maize is one of the most widely grown staple crops globally, and it is especially critical in developing regions of sub-Saharan Africa and Mesoamerica, where maize alone comprises more than 20% of food calories and up to 73% of total maize production is used as a food source. 1 Despite high demand, maize growth suffers from significant losses at both pre- and post-harvest stages due to biotic stresses such as viruses, fungi, bacteria, and other pests and pathogens. Numerous methods to improve maize growth and yield, including adoption of improved seed varieties, use of fertilizers, and application of pesticides, have been suggested to alleviate recurrent food shortages; however, these have not been widely used by smallholder farmers due to their high cost and lack of accessibility.2,3
Quick and efficient detection of plant pathogens is desirable for timely intervention of plant diseases. Standard methodologies in plant diagnostics range from physical examination of symptoms to the use of molecular diagnostics such as enzyme-linked immunosorbent assay (ELISA) or polymerase chain reaction (PCR).4–6 ELISA and PCR offer high sensitivity and specificity; however, both these approaches require expensive laboratory facilities and trained operators. In contrast, physical examination can only offer a diagnosis well past the onset of pathogenic infection.
Recently, lateral flow devices have gained much attention as inexpensive, user-friendly, in-field diagnostic tools. 7 Lateral flow devices typically incorporate the use of a nitrocellulose test strip and capture antibodies to detect the presence of an analyte in a two-step sandwich assay. Wicking of an analyte-containing sample solution through a nitrocellulose substrate results in capture of the analyte by the first antibody, conjugated to a colored particle. The analyte–antibody pair then flow downstream to a second analyte-specific antibody that is immobilized on the surface of the test strip (the detection zone). An accumulation of the initial analyte–antibody pairs at the detection zone results in a positive test indicated by an aggregation of colored particles visible to the naked eye. 7 Although lateral flow devices have recently been developed to detect plant pathogens, 5 these devices require the user to initiate testing and are likely used for rapid confirmation of pathogenic infection already suspected from physical examination. An ideal detection platform would provide notification at the first onset of pathogenic infection. Such a platform would benefit from a biodetection strategy that is integrated directly within the host plant itself.
Syringe agroinfiltration is a method of gene delivery into plant hosts using Agrobacterium.8,9 A needleless syringe is filled with an Agrobacterium-containing solution. The tip of the syringe is then placed against the abaxial (lower) side of an intact plant leaf, and the solution is manually injected into the leaf interstitium. A temporary color change from light to dark green indicates successful infiltration of Agrobacterium into the leaf.10,11 Syringe agroinfiltration has been demonstrated successfully in several plant species.11,12 Although there are other methods of transient gene expression in live plant hosts, including biolistics (microprojectile bombardment) 13 and microneedle injection, 14 the need for minimal equipment and simplicity of procedure has made syringe agroinfiltration a popular choice in recent years.
Herein, we report the first development of a biosensing technology that uses a live maize leaf as a lateral flow “test strip.” By modifying the established agroinfiltration procedure, we inject and immobilize capture-antibody-conjugated microspheres in the leaf interstitium. Our one-step lateral flow detection platform aims to target fluorescent biomarkers (e.g., aflatoxins) or to incorporate the use of detection-sensitive, colorimetric polydiacetylene (PDA) vesicles15,16 to eliminate the need for two-step sandwich assays for nonfluorescent biomarkers. In this study, we successfully detect a mock pathogen, fluorescein, using infiltrated microspheres conjugated with anti-fluorescein antibody ( Fig. 1A ).
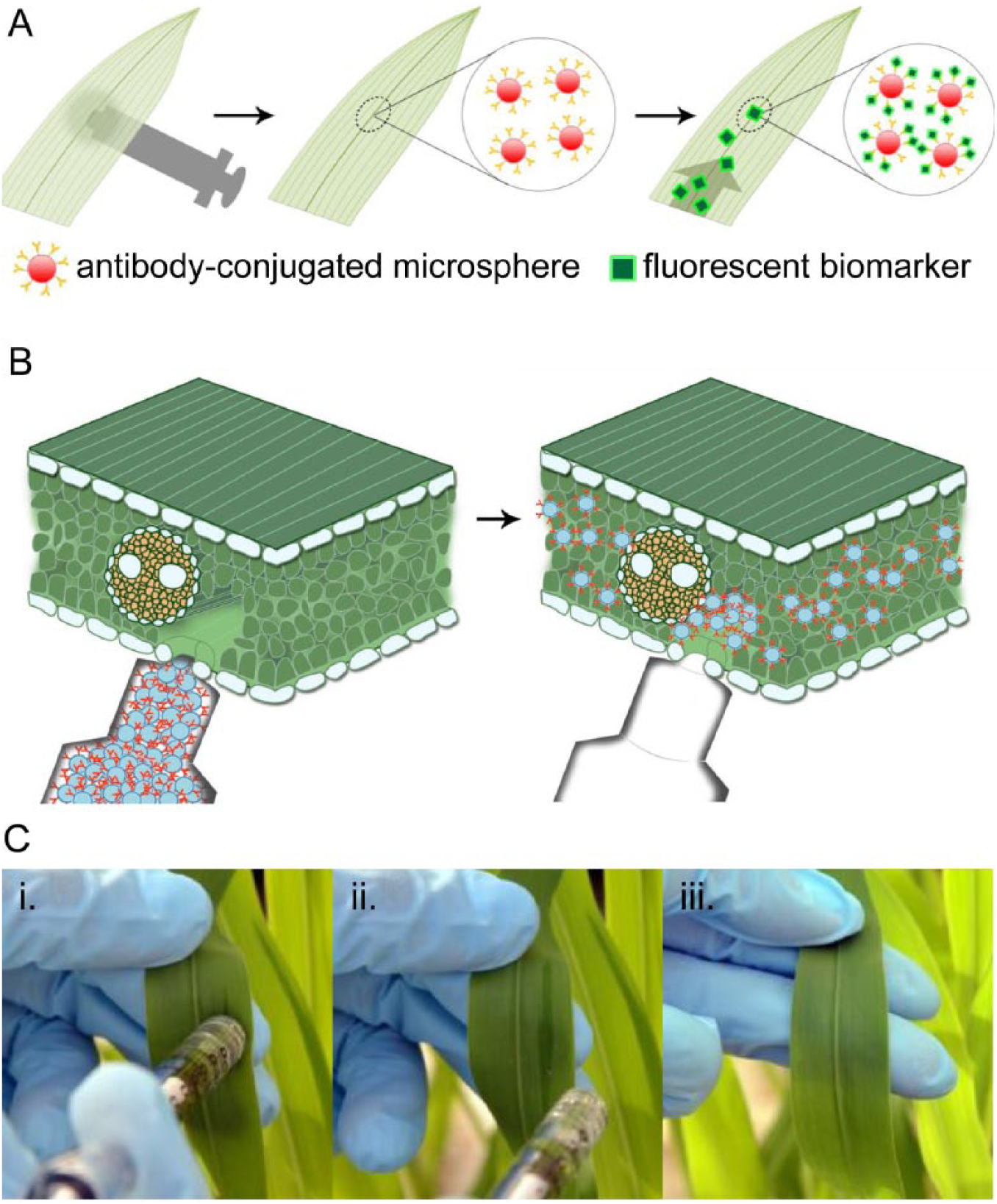
Method of one-step lateral flow detection of pathogen markers in live maize leaves. The present study focuses on detection of a fluorescent biomarker using antibody-conjugated microspheres (
Materials and Methods
Materials
Maize seeds were purchased from Carolina Biological Supply Company (Burlington, NC). One milliliter needleless syringes were purchased from Fisher Scientific (Waltham, MA). Fluorescent microspheres were purchased from Spherotech (Lake Forest, IL). Biotinylated anti-fluorescein antibody was purchased from eBiosciences (San Diego, CA). All other chemicals used were research grade and were purchased from Sigma-Aldrich (St. Louis, MO).
Maize Growth
Maize seeds were germinated in coarse vermiculite in 1020 half flat trays in a greenhouse maintained within a temperature range of 16–27 °C (61–81 °F). The trays were filled with Peters Excel 21-5-20 Multi Purpose fertilizer solution (125 ppm N) from Everris (Dublin, OH) once a day.
Infiltration of Microspheres
Fluorescent microspheres were washed in 1× phosphate-buffered saline (PBS) and resuspended in an infiltration buffer (10 mM MgCl2, 10 mM MES-potassium, pH 5.6). Microsphere solutions (0.5% v/v solids) were infiltrated into the abaxial side of intact maize leaves. Immediately after infiltration, the abaxial epidermis was gently wiped with deionized water to remove residual uninfiltrated microspheres. Infiltrated leaves were examined from the adaxial (upper) surface under a Leica DM2000 fluorescence microscope following infiltration and every 24 h for 72 h. Fluorescence intensities were determined as a ratio of the signal intensity of the target region to the leaf background. Retention rate was calculated as a ratio of fluorescence intensity at 72 h after infiltration and immediately following infiltration.
Lateral Flow Detection
Anti-fluorescein antibody-coated red fluorescent microspheres (AF microspheres) were prepared by mixing 0.5 µm streptavidin-coated red fluorescent microspheres with biotinylated anti-fluorescein antibodies, washed, and resuspended in the infiltration buffer. The AF microsphere solutions (0.5% v/v solids) were injected into the second leaf from the bottom of 15- to 20-day-old maize plants, examined for initial microsphere content, and transferred to fluorescein solutions (0, 3.2, 8, 20, or 50 µM fluorescein in PBS adjusted to pH 6.8). The plants were grown in the fluorescein solutions for 72 h to allow for sufficient fluorescein uptake by the plants, and the solutions were changed every 12 h. The plants were examined again at the end of the 72 h experiment. For the duration of the experiment, plant roots were fully immersed in the fluorescein solutions and the solutions were protected from light. Fluorescence intensities were determined as a ratio of the mean signal intensity in a region of interest (ROI) to the leaf background. The ROI is defined per plant as a fixed rectangular area that encompasses the spot where red fluorescent microspheres have been infiltrated. The leaf background is defined as a corresponding region lateral to the ROI on the same leaf. Normalized fluorescence intensities for test plants were calculated as a ratio of the fluorescence intensities to the mean intensity of control plants grown in the 0 µM fluorescein solution.
Results and Discussion
Infiltration of Microspheres in Leaf Tissue
A pressure-driven infiltration technique, inspired by agroinfiltration, was developed to noninvasively introduce microspheres into the leaf. A pressure gradient, introduced by the syringe on the leaf surface, propels bio-reagents through pores in the leaf abaxial epidermis and into the interstitial space ( Fig. 1B ). Infiltration in maize leaves is successfully demonstrated without damaging the epidermis ( Fig. 1C ). In the proposed technology, microspheres are used to provide stationary surfaces through which antibodies are effectively immobilized within the leaf tissue. Without such surfaces, antibodies cannot be retained at a fixed spot inside the leaf.
Microsphere sizes were characterized for both high infiltration efficiency and post infiltration retention rate. Spheres too large cannot pass through the leaf epidermis, whereas spheres too small are easily removed from the infiltrated spot. Fluorescent microspheres, ranging from 0.1 µm to 1 µm in diameter, were infiltrated into intact maize leaves. Microscopy analysis of the infiltration site during the 72 h period indicates that the 0.5 µm diameter microspheres produce initial signal intensities 2.5, 3, and 4 times higher than those of 0.3, 0.1, and 1.0 µm infiltrated microspheres, respectively (
Fig. 2A
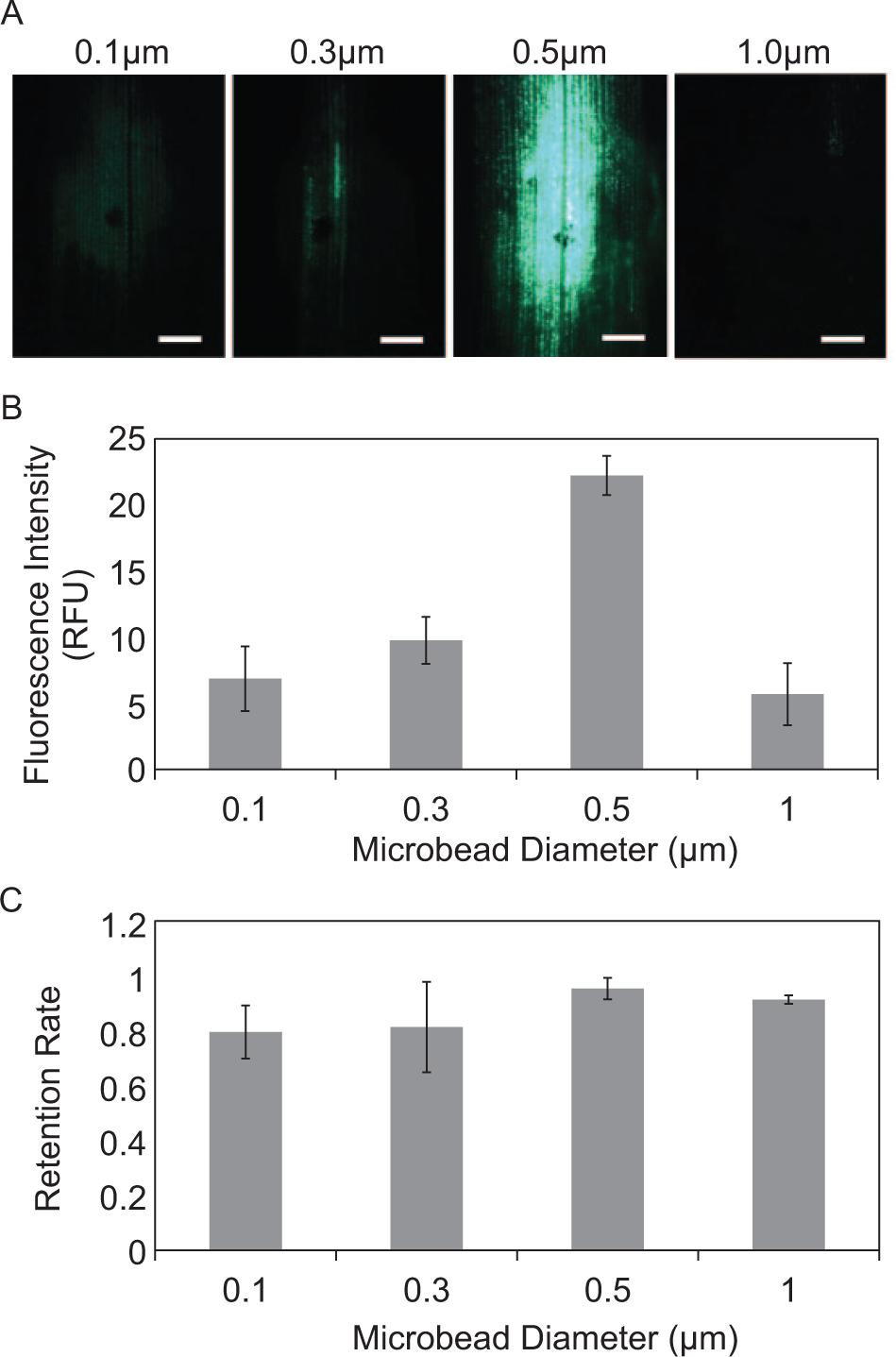
Infiltration of microspheres in leaf. (
In Planta Lateral Flow Detection of Fluorescein by AF Microspheres
Detection of fluorescein by AF microspheres was performed as a proof-of-concept demonstration of the proposed in planta biosensing technology. AF microspheres were prepared ( Fig. 3A ) and injected into leaves of live maize plants as described in the “Materials and Methods” section. Infiltrated plants were grown in the fluorescein solutions for 72 h before optical analysis. AF microspheres themselves do not emit green fluorescence ( Fig. 3B , 0 µM fluorescein concentration) but can effectively capture the trace amount of fluorescein molecules taken up by the maize plant as evidenced by localized accumulation of green fluorescence signals ( Fig. 3B , 3.2–50 µM). Quantitative analysis shows a 22%, 34%, 51%, and 71% increase in normalized fluorescence intensity in plants infiltrated with AF microspheres and grown in the 3.2 µM, 8 µM, 20 µM, and 50 µM fluorescein solution, respectively, as compared to control plants grown in the 0 µM fluorescein solution ( Fig. 3C ). In addition, plants infiltrated with control (nonconjugated) microspheres do not show increased fluorescence intensity with increasing fluorescein concentration, further confirming specific detection in the test plants that are infiltrated with AF microspheres. The limit of detection (LoD), 17 defined as two standard deviations higher than the arithmetic mean of normalized fluorescence intensity of control plants grown in the 0 µM fluorescein solution, is 19 µM. Collectively, these data indicate the feasibility of in planta detection of biomolecules in maize based on the proposed technology.
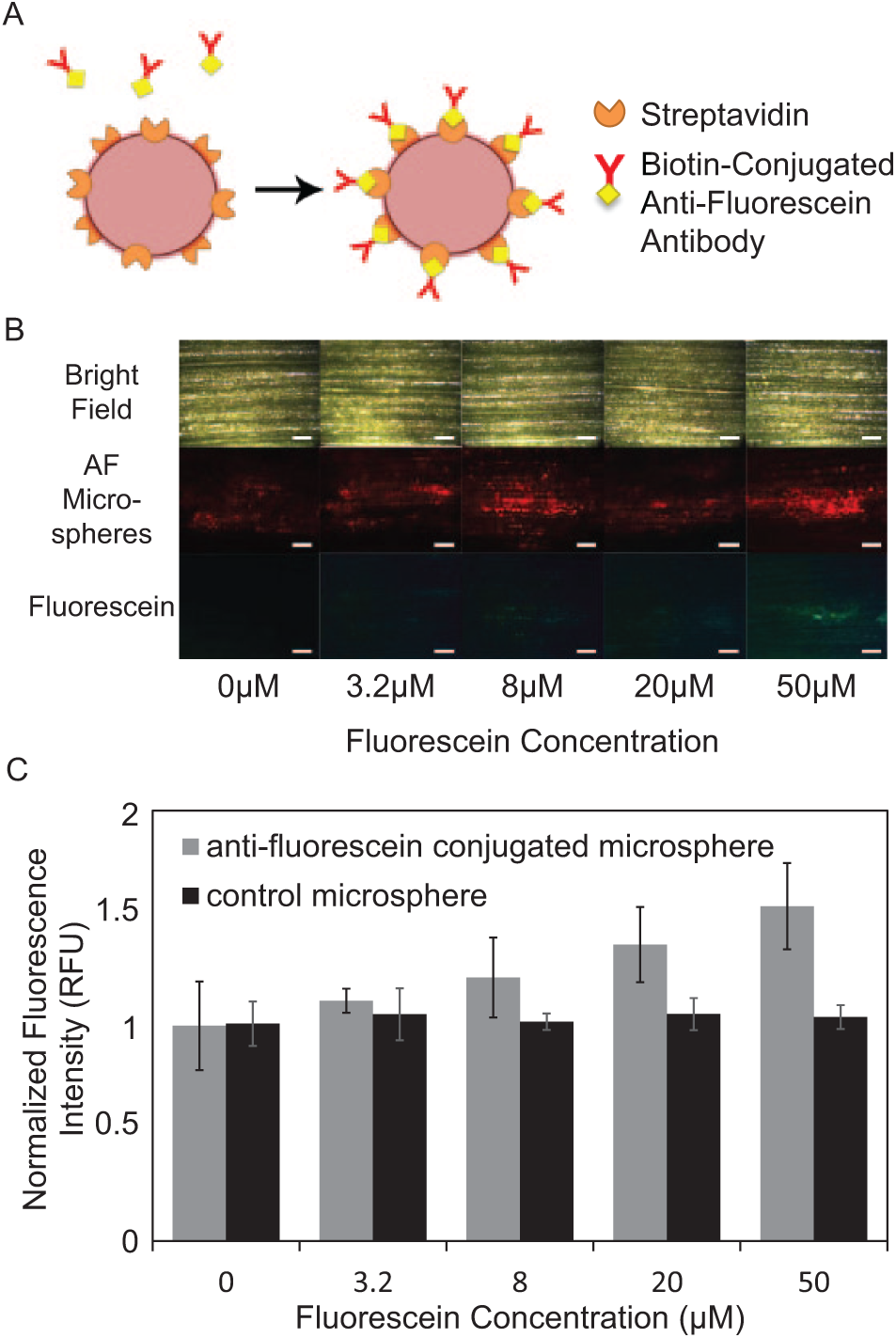
Lateral flow detection of fluorescein in live maize plants after 72 h of growth in fluorescein solutions. (
The anticipated use of this technology is for the early detection of a disease onset during a plant’s lifetime. The conjugated microspheres would therefore be injected into plant leaves at a young and healthy age. Because capture antibodies are likely to degrade and become inactive over time in plant leaves, especially in the field (e.g., in high temperatures and sunlight), more stable capture probes such as DNA aptamers are a desirable alternative for long-term applications. In addition, DNA aptamers are more economical to produce than monoclonal antibodies, thereby allowing for further cost reduction of the proposed low-cost diagnostic method.
Conclusions
We have developed a minimally invasive in planta biomolecular detection platform for maize. The design of our detection system incorporates the immobilization of antibody-conjugated microspheres in the leaf to capture analytes and produce a detection signal. Unlike conventional lateral flow assays, which require sample processing (e.g., grinding of plants and extraction of analyte-containing solutions) and are single-use, the proposed, directly integrated technology does not require processing of plants post infiltration and is intended for continuous monitoring of biomarker levels over an extended period of time, potentially enabling early notification of an onset of a disease. This eliminates not only the need for multiple tests per plant but also an external substrate for the lateral flow assay, thereby reducing the costs and resources needed for disease detection. This simple method is minimally invasive, inexpensive, and, to our best knowledge, the first report of a lateral flow biodetection technology that is integrated directly within living plant hosts. Because syringe infiltration is easily adapted to a variety of plants,10,11 this proposed technology is expected to be widely applicable. Although this initial report focuses on the demonstration of in planta detection of a fluorescent mock pathogen (i.e., fluorescein) only, with further development, particularly with the incorporation of colorimetric, stimuli-responsive PDA vesicles ( Fig. 4 ),15,16 this technology is likely to yield an equipment-free and colorimetric in planta diagnosis. We anticipate that such an innovative tool will make a significant impact on improving the yield of healthy food crops by smallholder farmers in developing countries worldwide.
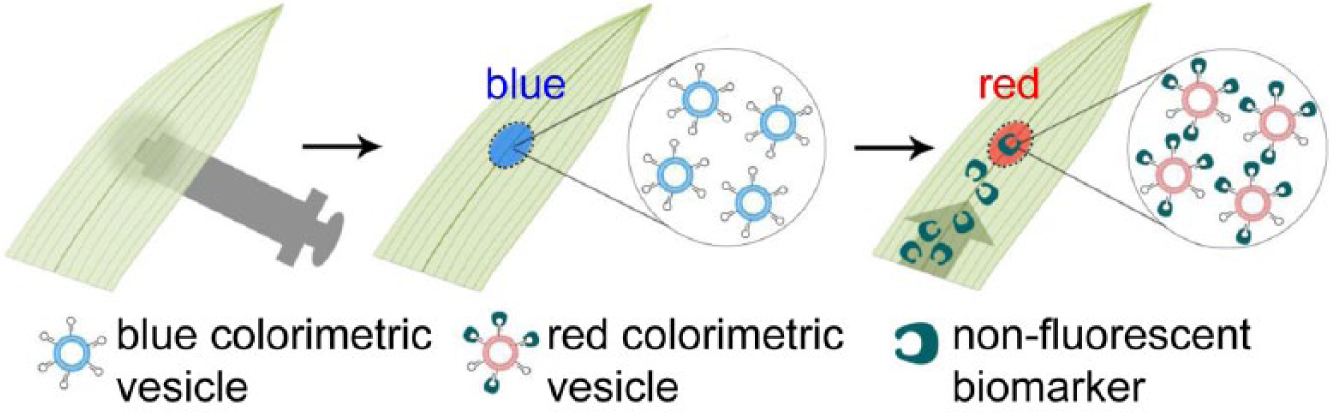
Proposed method of colorimetric, one-step lateral flow detection of pathogens in maize leaves for future development. Infiltrated, stimuli-sensitive, colorimetric vesicles respond to target biomarkers binding to aptamers or antibodies conjugated on the vesicle surface via a color shift (e.g., from blue to red) visible to the naked eye.
Footnotes
Acknowledgements
We thank Mr. Douglas Holt for providing help and training in growing maize plants, and Dr. Patricia Springer and Ms. Elizabeth Luscher for helpful advice and discussion. Access to the greenhouse space was kindly provided by UC Riverside Agricultural Operations.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Bill and Melinda Gates Foundation through the Grand Challenges Explorations program (grant no. OPP1059995).
