Abstract
Although developments in liquid dispensers have made the use of 1536-well plates for high-throughput screening (HTS) standard, there is still a gap in dispenser technology for performing matrix experiments with several components. Experiments such as those performed during assay development and enzymological studies are therefore still performed by manual pipetting in lower-density plates. We have evaluated a new dispenser, the Certus liquid dispenser (Gyger Fluidics GmbH, Switzerland), that is capable of flexible dispensing in 1536-well format, with a dead volume of less than 200 µL. Taking advantage of the precision of the dispenser for volumes down to 50 nL, we have created concentration gradients on plates by dispensing different volumes of reagent and then backfilling with buffer. Using this method and the flexibility of the dispenser software, we have performed several multidimensional experiments varying two to three components, including an assay development for an HTS, a mode of inhibition study, and a cofactor optimization, in which we determined 32 KM values. Overall, the flexibility of the plate layout for multiple components, the accuracy to dispense volumes ranging 2 log orders, and minimal reagent usage enable this dispenser for complex biochemical experiments.
Keywords
A significant development in early drug discovery has been the high-throughput screen (HTS), in which a large number of compounds can be tested relatively quickly against a particular target/assay. 1 A key factor that enabled such a robust testing of millions of compounds has been the codevelopment of nanoliter liquid dispensers, which dispense reaction components rapidly and precisely to microtiter plates of various densities (standards are 96-, 384-, and 1536-well formats). By reducing the reaction volumes compared with low-throughput experiments, there was an additional benefit that the overall cost per data point was significantly decreased. As the primary use for the liquid dispensers in drug discovery has come from the HTS and profiling, most of the devices on the market have been optimized with respect to precision and speed for dispensing a fixed volume.2,3 In contrast, the development of dispenser technology for performing more complicated experiments in high-density plates has lagged behind.
Two particular cases in which such matrix-type experiments are performed are during assay development and for enzymology studies. Before an HTS, a series of experiments are performed varying the concentration of various components such as enzyme, substrate, buffer salts, detergents, and detection reagent to improve the robustness of the assay, as indicated by the signal-to-background ratio (S/B) and Z′ factor.4,5 In this stage, referred to as assay development, the ability to set up a multidimensional matrix would be of great value. On the other hand, for enzymology studies, there is a need to determine a number of KM values at varying concentrations of the other substrates, inhibitor, or cofactors. Although in this case, the number of dimensions of the matrix would be limited, to determine KM values, at least 2 log units of concentrations are required. Unlike the HTS, however, these experiments are performed manually by pipetting into lower-density (96-/384-well) plates, and the experiments are often tailored to simplify logistics and reduce reagent consumption, rather than performing the full matrix.
Although these types of experiments are performed, they are kept to a minimum because of the time and effort involved. Therefore, both assay development and biochemical studies would benefit from a dispenser that is capable of matrix experiments. The key requirement for such a dispenser would be the capability to set up concentration gradients for multiple reagents without plate layout constraints, which would in turn allow for user-based multiparametric experiments. In addition, such a dispenser should be designed in such manner that the dead volume is minimal so as to minimize reagent consumption. Here, we have evaluated the Certus liquid dispenser (Gyger Fluidics GmbH, Switzerland; Fig. 1 ) by performing progressively more complicated assay developments experiments in 1536-well format. The simple programming allowed us to create various plate layouts for several reagents without difficulty and enabled us to determine a number of assay parameters on the system with minimal effort.

(
Material and Methods
The general designs for a fluorescence intensity–based protease assay 6 and LanthaScreenEu kinase binding assay 7 were as described in the literature. All dispense procedures were performed by Certus liquid dispenser unless otherwise specified.
Enzyme titration
A total of 32 different volumes of stock protease solution (starting at 10 nL and increasing by factors of 1.2) was added to the appropriate well and backfilled to 3 µL with buffer. The reaction was then started by addition of 3 µL substrate, and the fluorescence was measured using an EnVision plate reader (PerkinElmer, Waltham, MA) after a fixed time.
Mode of inhibition studies
For each compound, KM values were determined at six different compound concentrations by setting up a 12 × 6 matrix (in quadruplicate) varying the substrate concentration by column and the compound concentration by row. Initially, the compound titration was set up using the Echo550 compound reformatter (LabCyte Inc., Sunnyvale, CA) by transferring 100 nL of compound solution from a premade dilution series so that each column contained a different concentration of compound (4, 2, 1, 0.5, 0.25 × IC50 value of compound). Substrate was dispensed so that there was a varying volume of substrate in each row (volumes dispensed were 0, 50, 66.7, 100, 133, 200, 266, 333, 500, 1000, 1500, and 2000 nL), followed by a backfill with buffer so that the final volume of substrate and buffer added was 2 µL. Enzyme was added to start the reaction, and the increase in fluorescence was monitored using an Envision plate reader. For each inhibitor concentration, the KM and
Optimization of LanthaScreenEu kinase binding assay
Four 5 × 5 matrices were programmed whereby the volume of the stock solution of labeled antibody dispensed was varied along the row (starting with 300 nL, in increments of 300 nL), the volume of the stock tracer solution dispensed was varied along the column (starting with 29.1 nL, increasing by factors of 2.2), and a different volume of the kinase stock solution was dispensed for each matrix (375 nL, 750 nL, 1125 nL, 1500 nL). To each well, 1.5 µL of buffer with or without staurosporine was added as the inhibitor control ( Fig. 2a ). The reaction was finally backfilled to a total volume of 8 µL with buffer, the components allowed to reach equilibrium, and the time-resolved fluorescence resonance energy transfer (TR-FRET) signal measured on an EnVision plate reader. The S/B was calculated from the ratio of fluorescence from the reaction with and without staurosporine.
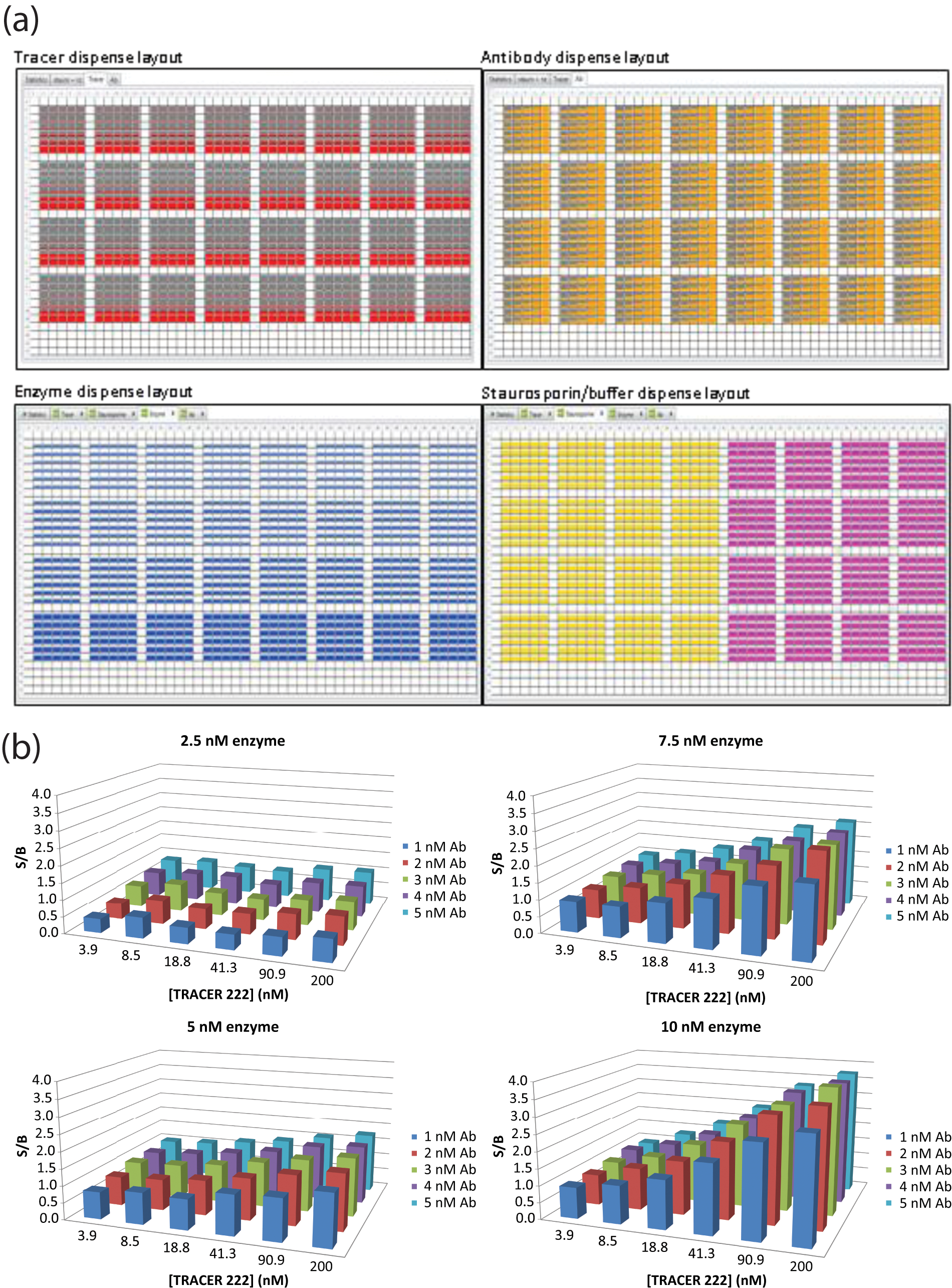
(
Optimization of enzyme with two cofactors
Four 32 × 8 matrices were programmed whereby the substrate volume was varied by column (starting at 10 nL and going up to 2000 nL with varying incrementation), the cofactor A volume was varied by row (starting with 10 nL, increasing by a factor of 2.4), and each matrix had a different volume of cofactor B (volumes dispensed were 0, 10, 400, and 1000 nL). Each well was finally backfilled to 4 µL with buffer. The reaction was started by the addition of 2 µL protease and the fluorescence measured using an EnVision plate reader after a fixed time. Data were fitted to a substrate inhibition model using nonlinear regression in GraphPad Prism 5.0.
Results
The Certus liquid dispenser is based on a solenoid valve principle 8 and has five separate dispensers, each supplied by its own reservoir ( Fig. 1a ). This allows a maximum of five assay reagents to be simultaneously prepared for dispensing into a high-density microtiter plate. For two of the five dispensers, the large bottles used for a reservoir can be replaced with a 5 to 10 mL disposable syringe attached directly above the valve. As this requires no tubing, the dead volume using a syringe is less than 200 µL, and we have used this for dispensing some of the expensive reagents for which we had only small volumes. The software is based on a graphical interface that allows for well-by-well control so that it is possible to dispense any of the five reagents in any volume to any well ( Fig. 1b ). Using the preinstalled dilution modules and automated backfill options, the time for designing and programming the three-dimensional matrix described in this article was less than 1 h.
In our group, biochemical assays for HTS are performed in 1536-well plates, with each reagent being dispensed as 1 to 2 µL volumes (total volume of approximate 4–6 µL). To create a concentration gradient for each reagent with a range of 2 log units, we required precise dispensing of reagent between 20 and 2000 nL.
We therefore started by testing the precision of the Certus liquid dispenser for dispensing variable volumes of biological reagent. For this initial experiment, we used a protease assay in which the enzyme activity is monitored by observing the increase of fluorescence due to cleavage of a fluorescently labeled substrate.
6
Enzyme concentration in the reaction was varied by adding different volumes of enzyme stock solution to each well (20–2000 nL) and then backfilling with buffer to a total of 2 µL. The activity with respect to enzyme concentration was fit using linear regression, and the
As this dispenser is able to create concentration gradients with a wide range, we reasoned that it would be possible to determine a large number of KM values in 1536-well format. Therefore, we tested whether we could determine the mode of inhibition for several compounds by determining KM values at four to five compound concentrations. As in the previous experiment, we set up a concentration gradient for the substrate by using increasing volumes of substrate stock and buffer backfilling. The compounds at various concentrations were transferred directly to the appropriate wells using the Echo550 compound reformatter. In this way, for each compound, we determined the KM and
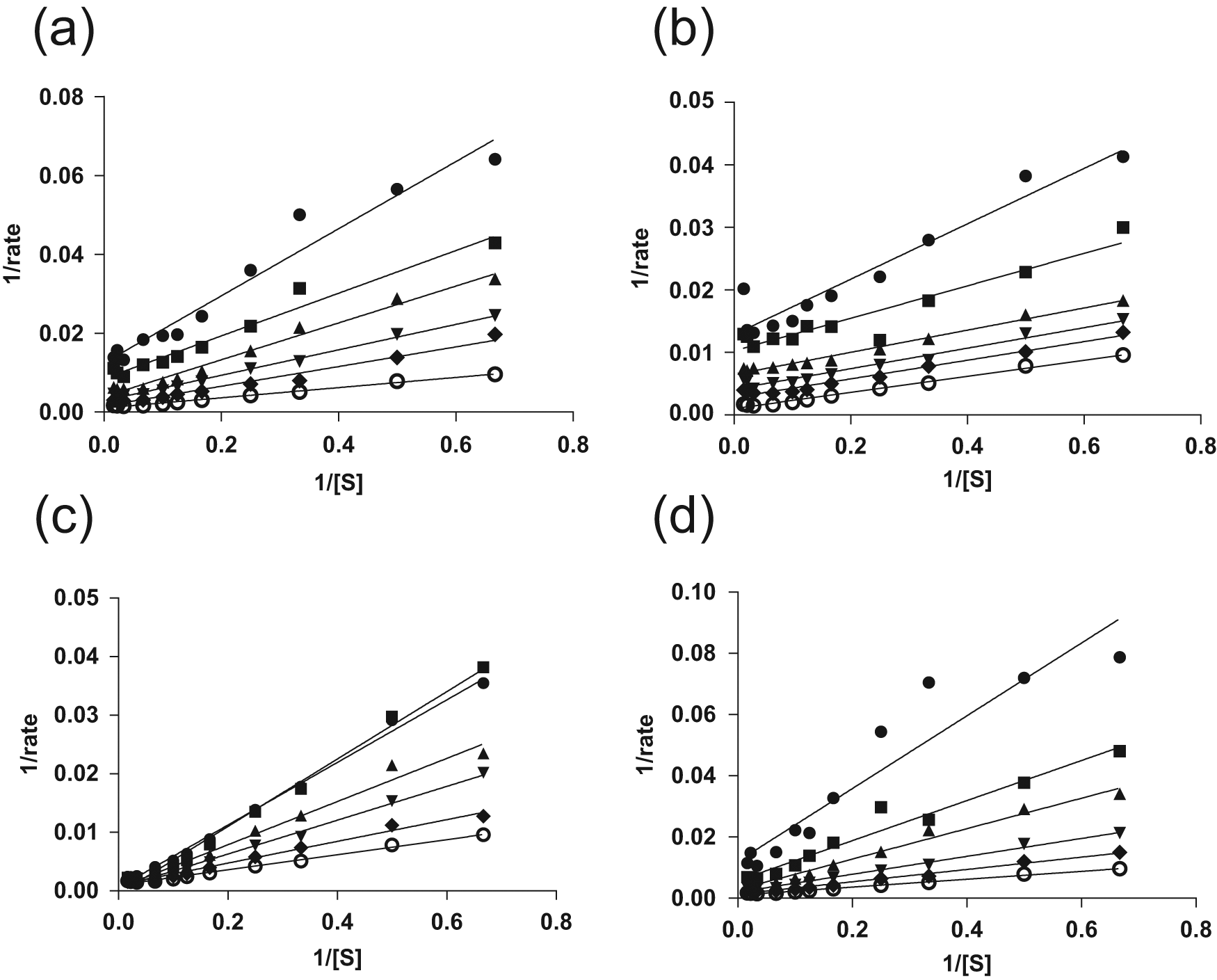
Lineweaver-Burk plot analysis for compounds tested against a protease. Michaelis-Menten plots were generated for six different concentrations of compounds, and four compounds were tested on a single 1536-well plate. The rate was calculated by subtracting the fluorescence intensity at time = 0 min from the fluorescence intensity t = 60 min. Compounds showing (
During assay development, often the assay or detection components are optimized one after another. Although logistically convenient, this method cannot look at interactions between the components and therefore may lead not to the true optimum but only a local optimum. Ideally, a combination of more than one parameter should be varied as in a matrix experiment. However, performing such experiments manually is very time-consuming and can be error prone.
We then performed a three-dimensional matrix experiment on the LanthaScreen Eu kinase binding assay. 7 Briefly, in this assay, a tracer molecule with a fluorophore binds specifically to the kinase active site and is brought in close proximity to a kinase-specific antibody labeled with another fluorophore, so that TR-FRET can occur. Upon compound binding, the tracer is displaced from the kinase, and this is observed by monitoring a decrease in TR-FRET signal. To find suitable assay conditions for this assay, we varied the concentration of the three components (tracer, antibody, and kinase) against each other in a 1536-well format (see Fig. 2a for the plate layout). Comparing the S/B for each concentration combination, we were able to identify minimal concentrations of reagents with a good S/B ( Fig. 2b ). As the experiment was performed in 1536-well format, the assay could be developed with a small amount of reagents, and the conditions were directly applicable for the screen.
We finally looked into a protease that requires two cofactors, to see the influence of the cofactors on both the KM and
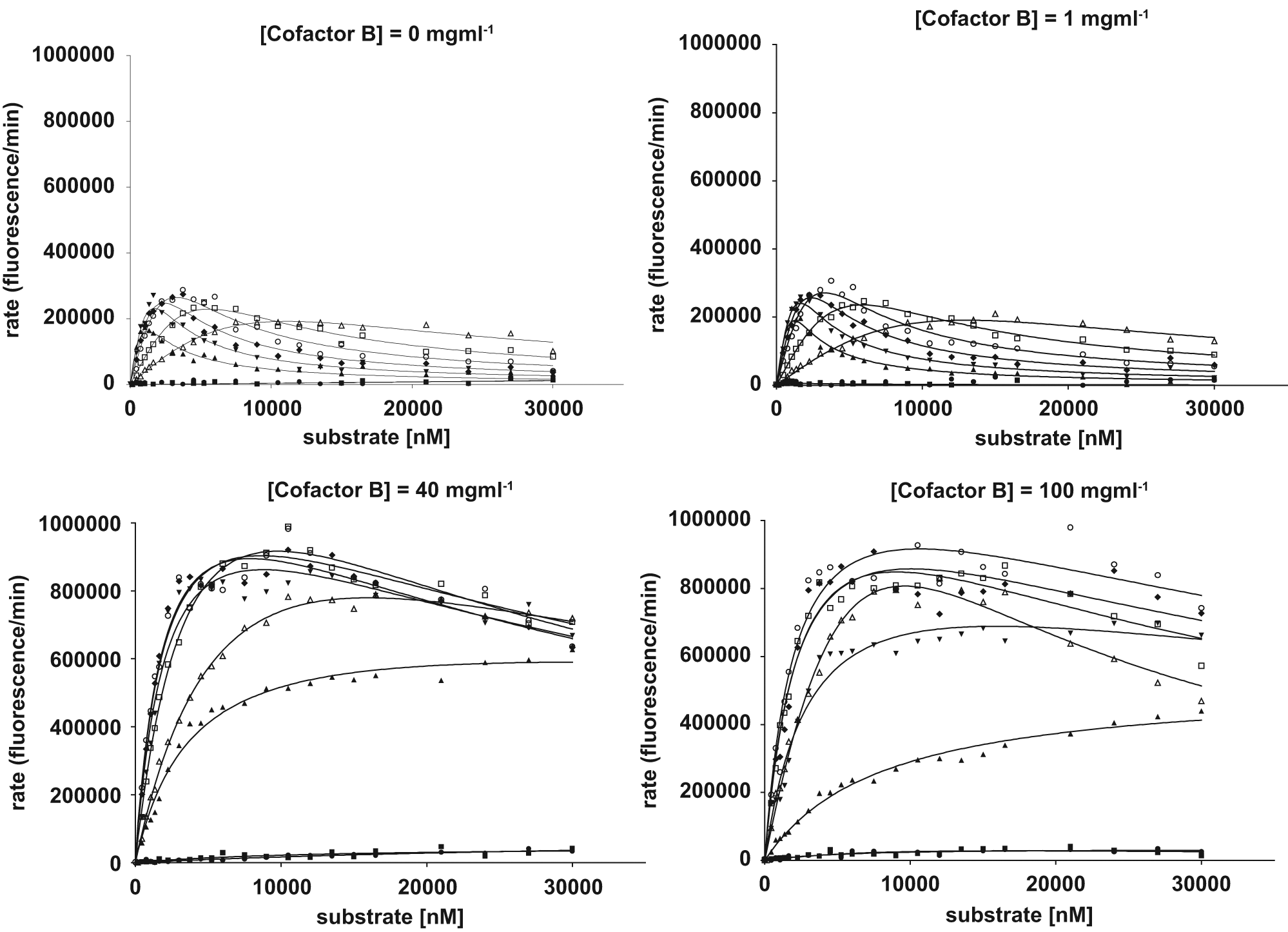
The Michaelis-Menten plots generated for varying concentrations of two cofactors. Each graph shows Michaelis-Menten plots for six concentrations of cofactor A at a single concentration of cofactor B. The data were then fit to a substrate inhibition model.
Conclusions
The Certus liquid dispenser is a precise liquid dispenser with flexible software that allows creation of concentration gradients in 1536-well format by dispensing variable volumes of reagent and backfilling with buffer. Using the method, we were able to perform matrix experiments looking at interactions between two and three components at a time, which would otherwise not have been performed because of time or reagent constraints. Although we have shown a few examples of where such dispenser could be of use, we believe that precise dispensers with a flexible programming and low dead volumes will allow biochemists to perform more complex experiments on a routine basis without the hindrance of logistics.
Footnotes
Acknowledgements
We would like to thank M. Biegger and R. Gyger from Fritz Gyger AG, Switzerland, for their support with the Certus software. We would also like to thank J. Blank and J. Ottl for the critical reading of the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
