Abstract
Nanoparticles possess a wide range of exceptional properties applicable to biology and medicine. In particular, nanodiamonds (NDs) are being studied extensively because they possess unique characteristics that make them suitable as platforms for diagnostics and therapeutics. This carbon-based material (2–8 nm) is medically relevant because it unites several key properties necessary for clinical applications, such as stability and compatibility in biological environments, and scalability in production. Research by the Ho group and others has yielded ND particles with a variety of capabilities ranging from delivery of chemotherapeutic drugs to targeted labeling and uptake studies. In addition, encouraging new findings have demonstrated the ability for NDs to effectively treat chemoresistant tumors in vivo. In this review, we highlight the progress made toward bringing nanodiamonds from the bench to the bedside.
Introduction
Engineering at the micro/nanoscale has contributed greatly to improving applications in diagnostics and therapeutics.1–8 Due to their extraordinary attributes, nanoscale particles have been of interest to researchers in the past decade for applications in biology and medicine.9–19 Nanodiamonds (NDs) in particular offer a unique combination of biologically relevant properties that provide possible advantages over other nanoparticles. As a result, NDs are being explored for use in an array of clinical applications from cancer therapy to gene delivery.20–22
A primary requirement for biologically applicable carriers is biocompatibility. Previous in vitro studies have demonstrated that NDs are not cytotoxic in neuroblastoma and macrophage cell lines. 23 In fact, in a comparative study, NDs have been shown to be the most biocompatible carbon-based nanoscale material, relative to species such as carbon black, multiwalled, and single-walled carbon nanotubes. 24 In vivo biodistribution studies performed in mice and Caenorhabditis elegans further demonstrate the stability and safety of this particle.25–27
To better understand their complex surface chemistry, NDs have been modeled extensively.28–32 It has since been confirmed that NDs exist in the shape of truncated octahedrons, containing two types of electrostatically distinct facets of opposing charges.33–35 Its charge characteristics result in the alignment of water molecules on the ND surface to form a tight hydration layer, which contributes to the stability of ND agglomerates in solution and its ability to potently bind with molecules such as the chemotherapeutic drug, doxorubicin (DOX). Atomistic modeling using molecular dynamics (MD) has provided insight into the role of pH dependence on NDs’ adsorption abilities. 36
An important advantage of NDs is the ease and diversity to which they can be functionalized.37–40 Modification of surface functional groups through covalent and noncovalent methods allows for the attachment of medically relevant agents onto the particle. Due to the surface electrostatics of the ND facets, the particle is able to immobilize a variety of biomolecules, such as the protein cytochrome c, insulin, and therapeutic antibodies.41–43 Covalent attachment of moieties such as biotin and peptide sequences, using linkers such as silane, has also been demonstrated.44–47
NDs can be induced to fluoresce by the addition of nitrogen defects through ion irradiation.48–52 The particle displays low photobleaching with high biocompatibility, resulting in a promising candidate for cellular tracking studies.53–56 NDs are also viable for in vivo tracking as a powerful magnetic resonance imaging (MRI) contrast agent when coupled with gadolinium [Gd] (III). 57 Reported strengths of the signal produced by this ND-Gd(III) complex were over 10 times higher than other clinical Gd(III)-based contrast agents. Moreover, NDs are also being developed for use in cutting-edge detection techniques such as magneto-optic and scanning single-photon microscopy.58,59
The features of the ND platform make it highly suitable as a drug delivery agent. In cancer therapy, NDs have exhibited the ability to support steady release of chemotherapeutic drugs.10,21,60 ND drug platforms have also been embedded into thin-film devices, resulting in an implantable patch that is capable of sustained administration of therapeutic treatment.61–65 In addition, ND platforms, functionalized with the cationic polymer polyethylenimine (PEI), are capable of effectively mediating gene delivery with high efficiency and low toxicity.18,20 Recently, the performance of ND-based drug delivery has been validated in vivo. 9 Tangible clinical potential has emerged as a result of this important demonstration of both safety and efficacy over clinical standards.
Fluorescence Imaging
Fluorescent probes are critical tools for biological applications ranging from fundamental intracellular tracking to diagnosis of diseases, such as the visualization of cancerous tumors. However, current fluorescent labels are limited by photobleaching, photoblinking, and cytotoxicity. NDs offer a nanodisperse, photostable, biocompatible, and scalable alternative.53–55
NDs are capable of fluorescing after the introduction of nitrogen-vacancy centers, by irradiation from a high-energy electron beam followed by high-temperature annealing. Some endogenous cellular biomolecules, such as flavins, collagens, and porphyrins, tend to autofluoresce at low-absorption wavelengths (absorption 300–500 nm, emission 400–450 nm). 66 Fluorescent NDs (FNDs) display useful fluorescence capabilities because their absorbance (~560 nm) and emission (~700 nm) wavelengths are well above endogenous background signals. 66 Furthermore, ND-based probes have presented a constant fluorescence signal for up to 300 s of excitation, a significant improvement compared with an industry standard dye, Alexa Fluor 546, which has exhibited photobleaching after 12 s. The high contrast and stability of the FND fluorescent signal provides significant utility as a biological marker.
In addition, the production of FNDs is becoming optimized into a high-throughput process, resulting in an increase in the material’s translational relevance.67,68 Chang et al. 48 were able to use lower energy (40 keV) helium (He+) bombardment of NDs to create defects instead of the typical high-energy (2 MeV) electron (e–) bombardment. This new method offers a two orders-of-magnitude increase in efficiency of vacancy production per ion dosage.
The inherent safety of FNDs was confirmed in human HeLa cervical carcinoma and mouse 489-2 osteoprogenitor cells. 69 MTS viability assays, a colorimetric assessment of cellular metabolic activity, demonstrated that there is no significant change in cellular viability after 48-h incubation of concentrations up to 200 µg/mL. The clathrin-specific inhibitor, phenyl arsine oxide, significantly reduced FND uptake while the claveolae-specific inhibitor, filipin, did not. This result suggests that the cellular uptake pathway for FNDs is energy-dependent clathrin-mediated endocytosis.69,70 Moreover, the capacity of progenitor cells to differentiate was not significantly affected by increasing concentrations of FNDs as determined by alkaline phosphatase (ALP) staining. FNDs have even been used safely in tissue engineering, by improving material properties of a scaffold with no negative effects on cell growth. 71
The in vivo imaging capabilities of FNDs were used to evaluate their biocompatibility in the model organism C. elegans. 26 After being fed with FNDs, they were able to excrete the nanoparticles from their gut lumen within an hour. Upon injection of FNDs into the gonads, the fluorescence signal was tracked through embryogenesis. It was subsequently determined, by measuring brood size, longevity, and presence of reactive oxygen species (ROS), that there were no adverse effects on embryonic development. NDs ultimately did not produce significant levels of in vivo toxicity or stress response. The combination of high-throughput production and biocompatibility indicates the viability of ND-based probes in both biology and medicine.
Drug Delivery
At the forefront of NDs’ translational potential is their use in drug delivery, especially in cancer therapy. Nanoparticles have a high surface area to volume ratio, which allows for very efficient small-molecule loading. 12 Nanoparticle carriers also display increased circulation time and promote drug accumulation at the tumor site. NDs, in particular, enhance the efficiency of chemotherapeutic drugs by strongly binding to them and sequestering extra-tumor toxic effects. 9 ND conjugates subsequently mediate sustained release of drugs such as DOX in response to intracellular stimuli, such as changes in the pH environment.10,21,60 As it crucially applies to translational significance, these ND conjugates were produced in a scalable and efficient process.
Drug-loaded NDs have also been incorporated into flexible microfilm patches, capable of implantation and localized drug elution.61–65 ND-DOX (NDX) complexes were embedded into a parylene C structure, with an ultrathin porous cover layer. The combination of the controlled release of NDX complexes from the microfilm, as well as the subsequent stable elution of drug from the ND surface, produces steady, continuous treatment that mitigates harsh side effects associated with burst release.
NDs are also capable of functioning as effective carriers of nucleic acids. Gene therapy is another important treatment modality that is showing increasing promise in cancer therapy. Existing gene delivery vehicles, such as viral-, liposomal-, and polymer-based agents, are hampered by high cellular toxicity and low delivery efficiency, especially in biological environments. In the case of PEI, higher molecular weight (MW) forms of the polymer are more effective, while exhibiting significantly more toxicity. However, NDs functionalized with low MW PEI were able to demonstrate remarkable efficiency while remaining biocompatible. In fact, ND-PEI delivered plasmid DNA to HeLa cells 70 times more efficiently than PEI alone at equivalent concentrations ( Fig. 1 ). 18 At the same time, ND-PEI represented a sixfold improvement in cellular biocompatibility over high MW PEI. Moreover, ND-PEI is also capable of delivering small interfering RNA (siRNA), a promising form of gene therapy based on RNA interference. 20 As determined by in vitro gene knockdown experiments with M4A4 breast cancer cells, which were conducted in a realistic biological environment in the presence of serum proteins, ND-PEI outperformed the liposomal gold standard Lipofectamine.
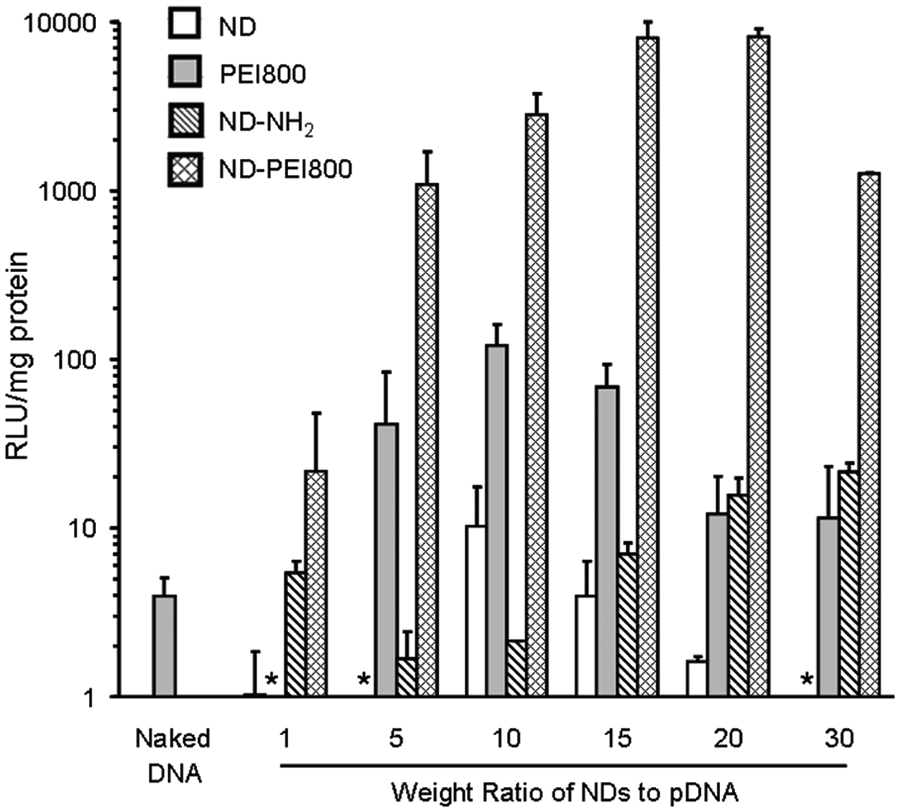
The transfection efficiency of nanodiamond-polyethylenimine (ND-PEI) is quantified using the gene expression of plasmid DNA delivered to HeLa cells. Across various conditions of carrier to DNA weight ratios, ND-PEI is able to outperform PEI alone. Data are represented as means ± SD (n = 2). Reprinted with permission from Zhang et al., 18 copyright 2009, American Chemical Society.
The potent drug delivery capacity of NDs has recently been demonstrated in vivo, further validating NDs’ clinical relevance. 9 The NDX platform effectively treated drug-resistant liver and mammary cancer tumors in mouse models. The adsorption of DOX is facile and scalable, owing to powerful electrostatic interactions between DOX and ND facets. Acquired drug resistance, after repeated administration of chemotherapeutics, is a critical challenge in treating metastatic cancer. Drug efflux proteins that pump out chemotherapeutics from the cell display elevated levels of expression in drug-resistant cancers. However, NDX is able to overcome efflux pumps by entering the cell through endocytosis instead of diffusion. Furthermore, the high drug loading on the NDX platform increases local drug concentrations to enhance therapeutic potency. Testing of the in vivo biodistribution of NDs resulted in clearance times from major organs within 10 days, along with stable levels of serum alanine transferase (ALT), indicating no adverse effects on liver function. Mice bearing resistant tumors were treated weekly through tail vein injections of NDX, DOX, NDs, or phosphate-buffered saline (PBS). Higher doses of DOX were required to significantly reduce tumor growth, but the severe toxicity of this treatment was evident from the abrupt survival curves from DOX-treated mice ( Fig. 2 ). Conversely, NDX was able to considerably reduce tumor size while preserving mice survival. At equivalent concentrations of 200 µg, DOX treatment alone resulted in no mice survival after day 15, whereas NDX demonstrated mice survival until day 60. The substantial promise of ND-based platforms is evident from the wide spectrum of therapeutic modalities that they are capable of supporting.
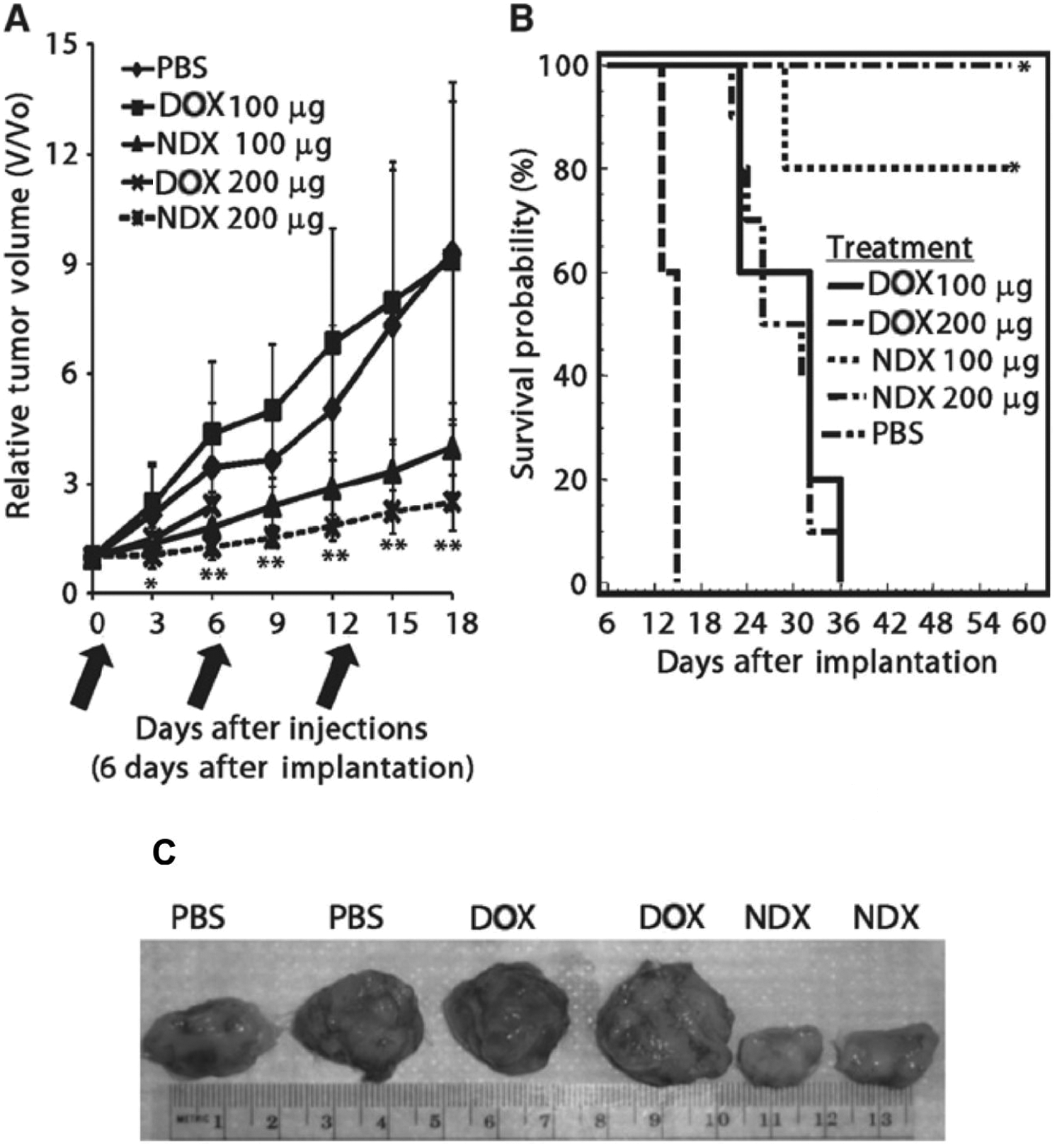
The treatment efficacy of nanodiamond-doxorubicin (NDX) is measured by observing tumor response in murine models. (
Future Outlook
NDs have demonstrated a diverse array of biologically applicable features, from robust nitrogen-vacancy fluorescence to tunable drug loading and delivery. Continued research into controlling surface modifications of NDs will allow for more effective loading and release of relevant moieties. To produce increasingly stable ND carriers, it is important to design innovative ND platforms based on proper primary particle diameters.72–74 The next step is additional testing in larger animals, which is required to address challenges such as optimizing ND platforms for potential use in clinical settings. Dosing levels need to be properly adjusted with continued in vivo experimentation, as well as in-depth clearance studies to validate long-term compatibility of NDs. Although further progress will be required for this material to reach its therapeutic potential, the versatility of the ND platform toward a multitude of medical applications represents an effective tool in nanomedicine.
Footnotes
Acknowledgements
D.H. gratefully acknowledges support from the National Science Foundation CAREER Award (CMMI-0846323), Mechanics of Materials grant CMMI-0856492, Center for Scalable and Integrated NanoManufacturing (DMI-0327077), DMR-1105060, V Foundation for Cancer Research Scholars Award, Wallace H. Coulter Foundation Translational Research Award, American Chemical Society Petroleum Research Fund Grant 47121-G10, National Cancer Institute grant U54CA151880, and National and European Commission funding program FP7-KBBE-2009-3. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute or the National Institutes of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
