Abstract
Amphiphilic block copolymers are finding increased potential in biological and medical research due to their innate alternating hydrophilic and hydrophobic blocks/segments that can be used to package therapeutics, or coat a broad array of biological interfaces. Some studies are already directed toward using these copolymers' ability to form micelles or vesicles to develop novel methods of drug delivery to prevent inflammation or pro-cancer activity. Our study, however, aims to investigate the more fundamental cell—block copolymer interaction for use in protective nanofilms to prevent biofouling of non-tissue-based implantable devices. Block copolymers could potentially fill the demand for biologically inert, highly functionalizable biomaterials desirable for this type of application. Two such polymers used in our study include polymethyloxazoline—polydimethylsiloxane—polymethyloxazoline (PMOXA—PDMS—PMOXA) triblock copolymer and polyethylene oxide-poly(methyl methacrylate) (PEO—PMMA) diblock copolymer. Each block copolymer possesses hydrophilic and hydrophobic blocks that enable it to mimic the cell lipid membrane. So far we have shown that triblock copolymer is capable of inhibiting the accumulation of murine macrophages onto glass substrates. Preliminary evidence has suggested that the triblock copolymer has anti-adsorptive and noninflammatory capabilities during short incubation periods (7 days) in vitro. While the diblock copolymer displays minimal anti-adsorptive activities, nanofilms comprising a mixture of the two copolymers were able to significantly reduce macrophage accumulation onto glass substrates. The disparate behavior of macrophages on the different materials may be due to specific inherent properties such as preference for hydrophobic versus hydrophilic surfaces and/or rough versus smooth nanotextures. Furthermore, the specific endgroups of the two polymers may exhibit varying capacities to resisting non-specific protein adsorption. Continued investigation outlining the physical and chemical properties desirable for an anti-adsorptive nanofilm coating will serve as a basis to design durable implant—tissue interfaces that can react to various external stimuli.
Introduction
In recent years, amphiphilic block copolymers have been finding their way into biomedical applications with the prospect of greatly improving the efficiency and effectiveness of modern medicine. Their ability to form structures similar to those of lipid membranes in aqueous solution has rendered these block copolymers as highly useful synthetic versions of natural lipids. 1 -6 However, amphiphilic block copolymers are significantly more stable than conventional lipid systems and the physical and chemical properties can be systematically manipulated to serve a vast range of purposes. 6 -8 For example, although block copolymers are inherently more stable than natural lipids due to their much higher molecular weight, these polymers can further be stabilized by covalent cross-linking across endgroups. 1,9 Although block copolymer membranes are usually two to four times thicker than lipid bilayers, 10,11 work has been done illustrating that transmembrane proteins remain functional after being anchored into a block copolymer membrane. 12 -16 Due to the advantageous qualities presented by block copolymers, these materials are already being pursued as novel drug delivery vesicles, among other applications. Spherical and wormlike micelles have been designed to target specific cells in vivo while simultaneously achieving increased hydrophobic drug solubility and drug elution control. 17 -20 These results prove to be encouraging for continued investigation into nanomedicine with amphiphilic block copolymers.
Another novel application of this class of polymers, and the topic of this study, is the deposition of block copolymer films onto solid substrates. Previous work has been able to synthesize membrane protein-functionalized large area nanofilms capable of being deposited on various solids using the Langmuir–Blodgett method of thin film deposition, which occurs when amphiphiles are transferred to solid substrates at the air–water interface. 13,14 Solid substrate deposition presents the possibility of developing medical implant coatings that may be able to address the current need for bio-fouling prevention of medical devices such as cardiovascular stents and other non-biologically derived implants. It is known that the adsorption of proteins onto implant surfaces is largely responsible for the development of bio-films that significantly decrease performance and lifetime. 21,22 Implants are also known to induce inflammatory responses in vivo, with macrophages playing a main role by infiltrating the implant–tissue interface, biodegrading the material surface, and recruiting other leukocytes to the site of implantation. 23 -29 Because the adsorption of bio-fouling proteins onto implant surfaces seems to be a crucial step for implant degradation, a coating that could prevent this initial reaction would be invaluable. One potential solution for this challenge is based upon the polymethyloxazoline–polydimethylsiloxane–polymethyloxazoline (PMOXA–PDMS–PMOXA) triblock copolymer. The PDMS interior block is hydrophobic and the surrounding PMOXA blocks are hydrophilic. These PMOXA end blocks are biologically inert 30 and have the unique ability to prevent unspecific protein adsorption, 31 thus serving as a potential starting material in designing protective implant coatings. 32 Another potential solution is a second block copolymer being investigated in this study, the polyethylene oxide–poly(methyl methacrylate) (PEO–PMMA) diblock copolymer. This amphiphilic copolymer possesses a hydrophobic backbone (PMMA) and hydrophilic branches (PEO). 33 Diblock copolymers are capable of forming lipid mimetic vesicle assemblies and can facilitate many membrane processes as described previously. 34 -36 Incorporation of this diblock copolymer into protective copolymer films, coupled with the engineering of drug delivery scaffold systems may prove useful in designing anti-fouling coatings.
Materials and Methods
Polymer Isotherms and Deposition on Glass Substrates
The PMOXA–PDMS–PMOXA copolymer with acrylate endgroups and PEO–PMMA copolymer were custom synthesized (Polymer Source, Quebec, Canada) and solubilized to a concentration of 10 mg/mL in chloroform. For isotherm data, the polymer–chloroform solutions were applied to the ethanol and nanopure water pre-treated surface of a Langmuir–Blodgett trough (KSV 2000, KSV Instruments, Finland) at a surface pressure of 5 mN/m. After the chloroform was allowed to evaporate over a period of 25 min, the polymer membranes were compressed at a rate of 5 mm/min until collapse. The abscissa of the isothermal graphs represent the surface area enclosed by the barriers of the LB trough and the ordinate represents the surface pressure. For polymer deposition onto glass substrates, the polymer-chloroform solutions were applied to the Langmuir–Blodgett trough at a surface pressure of 10 mN/m. After a 25-min evaporation period, the polymer films were compressed at a rate of 10 mm/min until the desired surface pressure was reached. The polymer was subsequently deposited onto glass substrates at a rate of 5 mm/min. Due to the nature of Langmuir–Blodgett deposition, it is impossible to coat the entire surface of the glass substrate with polymer films, therefore about 20% of each glass slide remained uncoated.
Macrophage Cell Culture and Plating
RAW 264.7 American Type Culture Collection (ATCC) macrophages were grown until 95% confluency in 75-cm2 laboratory culture flasks with Dulbecco's Modified Eagle's Medium (DMEM) growth media supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin. The macrophages were consequently plated onto 18.75-cm2 substrates of varying character at a density of 25% and further supplemented with the media previously described until a maximum duration of 7 days.
Light and Confocal Microscopy
Macrophage cells grown on glass or polymer substrates were imaged with Brightfield light microscope at time points starting at 4 h and continuing through 24-h intervals for up to 1 week. Confocal microscopy was performed after macrophages had been grown long enough to become confluent, using cellular accumulation on the pure glass portion of each slide as a standard. Each sample was then washed twice with phosphate-buffered solution (PBS), pH 7.4 and fixed in 2% paraformaldehyde in PBS for 15 min. Samples were then incubated with 0.1% Triton X-100 in PBS for 5 min. A 0.16-μM solution of Alexa Fluora 488 phalloidin was applied for 20 min in the absence of light as designated by the manufacturer (Molecular Probes). Each sample was then incubated with 25 μM solution of TOTO-3 dimeric cyanine nucleic acid stain for 15 min as designated by Molecular Probes. Two or more PBS washes were performed after every step during fixing and staining.
Contact Angle Measurements
Contact angle measurements were performed using a Ramé–Hart, Inc. Imaging System and Auto Pipetting System (Mountain Lakes, NJ). An average of 10 stable step-out measurements (25 μL each) taken at 0 °C to the z plane were used to represent the contact angle between the milliQ water and the three substrates used: pure glass, glass coated with diblock, and glass coated with triblock. All block copolymer coatings were deposited at 25 mN/m and measurements were taken at 25 °C.
Quantitative RT-PCR
Total mRNA was obtained and converted to cDNA using conventional methods. After cDNA was obtained, PCR was performed with iCycler Thermocycler (Bio-Rad). Q-PCR was conducted according to the manufacturer's protocol (Bio-Rad). The primers used include IL-1 (sequence: sense 5′-TGCAGAGTTCCCCAACTGGTACATC-3′, antisense 5′-GTGCTGCCTAATGTCCCCTTGAATC-3′), IL-6 (sequence: sense 5′-CACAGAGGATACCACTCCCAACA-3′, antisense 5′-TCCACGATTTCCCAGAGAACA-3′), Type I Interferon (sequence: sense 5′-TGTCTGATGCAGCAGGTGG-3′, antisense 5′-AAGACAGGGCTCTCCAGAC-3′), and CSF (sequence: sense 5′-CATCTCCATTCCCTAAATCAAC-3′, antisense 5′-ACTTGCTGATCCTCCTTCC-3′).
Results and Discussion
Inhibition of Macrophage Accumulation on Triblock Copolymer Substrates
Initial investigation of the ability of murine macrophages to adhere and grow on the PMOXA–PDMS–PMOXA triblock copolymer showed severe differences compared with macrophages grown on pure glass substrates. When triblock copolymer was deposited onto glass at a surface pressure of 20 mN/m or higher, significant inhibition of macrophage adhesion and growth was observed (Fig. 1). Cells were also plated on glass substrates coated with triblock copolymeric films deposited at surface pressures of 5 and 10 mN/m. These samples also showed some early inhibition of macrophage accumulation but within 96 h they began to lose their ability to inhibit accumulation compared with films deposited at higher surface pressures (Fig. 2B-E). An isothermal compression was performed to better observe the deposition surface pressure-dependent ability to inhibit cellular accumulation (Fig. 2A). The isotherm shows that when the polymer is at 10 mN/m or less, the triblock copolymer is in its gaseous or early diffuse liquid phase. In the range of 20–30 mN/m, the polymer is in its compressed liquid phase. These results are similar to previously observed findings and suggest that a more condensed monolayer is more conducive to macrophage accumulation inhibition on glass substrates. 37 The triblock copolymer's anti-adsorptive ability may be due to a weakened cellular ability to adhere to the membrane monolayer, possibly due to certain surface properties such as wettability or topography. It was observed that replacing the media of cells grown on triblock copolymer caused a significant amount of macrophage denudation from the polymer substrates whereas there was almost no effect on macrophage density on glass substrates (Fig. 3). This could be due to the polymer's innate ability to prevent unspecific adhesion as described earlier, but could also be due to the eroding of the polymer itself causing the adherent macrophages to be consequently washed out. The triblock copolymer was also able to maintain inhibition of cellular accumulation for up to 7 days. However, it will be important to find how the polymer reacts in vivo for significantly longer periods of time.
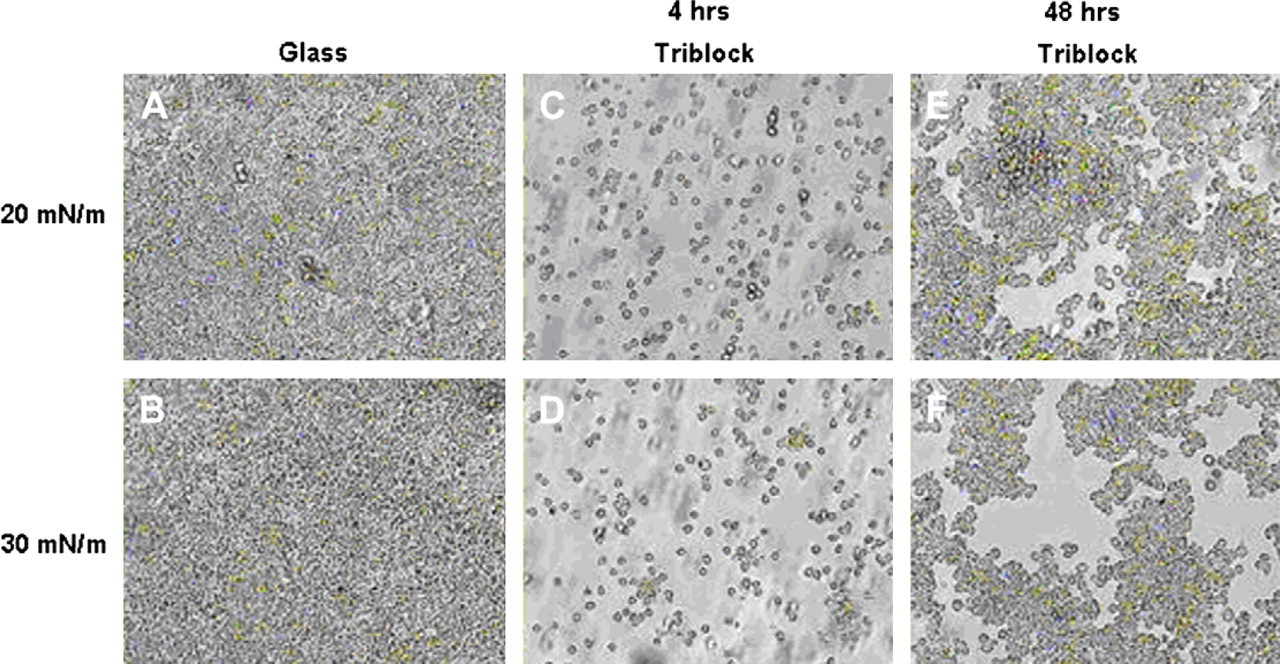
Macrophages cultured on triblock-coated substrates have difficulty in accumulating with respect to glass. (A), (B) Macrophages grown on glass substrates. Cells grown on glass achieved confluence within 4 h and maintained such density through the entire experiment; (C), (D) macrophages grown on triblock deposited at 20 and 30 mN/m, respectively, for 4 h; (E), (F) cell growth after 48 h on polymer substrate deposited at 20 and 30 mN/m, respectively.
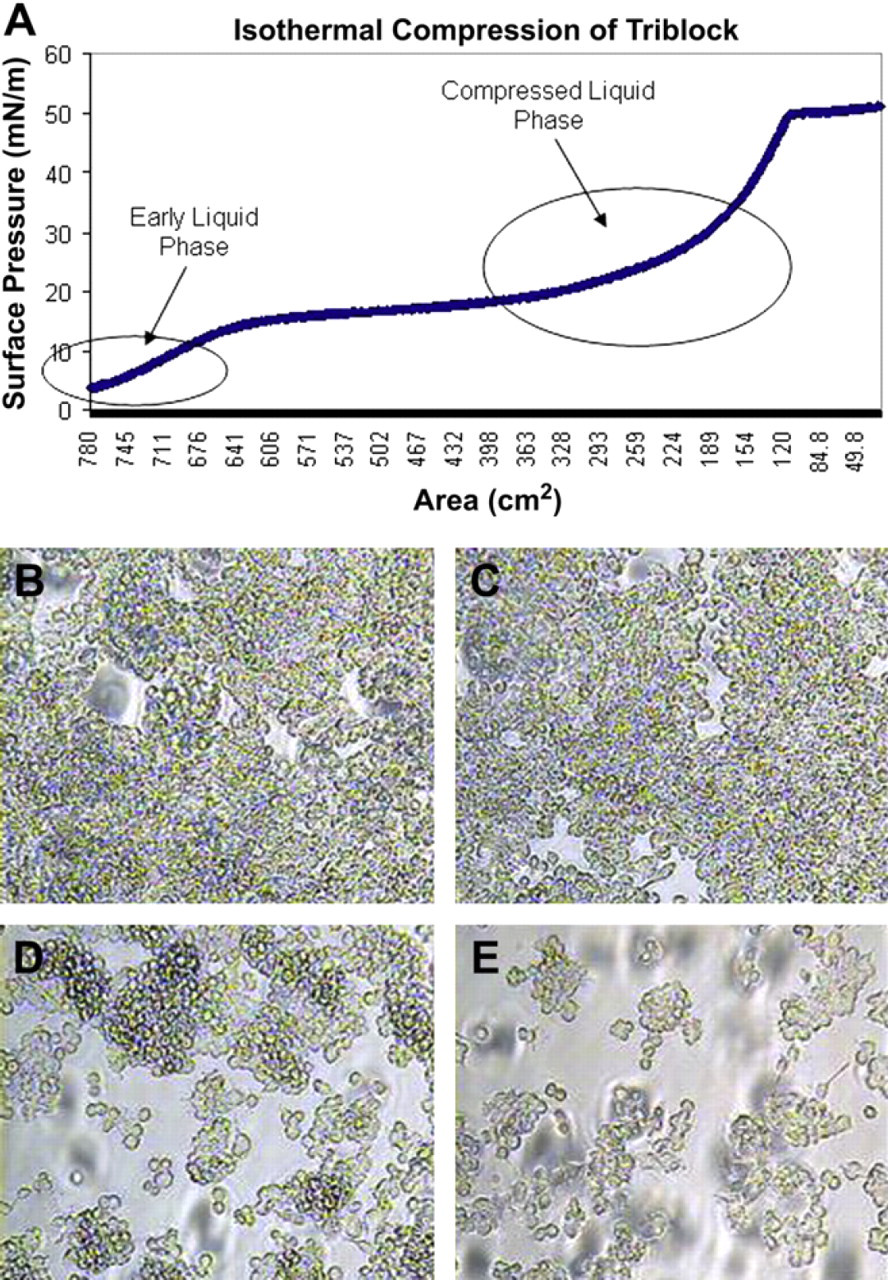
Isotherm of the triblock copolymer and the resulting ability of macrophages to accumulate on glass substrates. (A) An isothermal compression of the triblock copolymer illustrates the different phases exhibited by the polymer on the trough surface. When the surface pressure of the polymer is below approximated 15 mN/m, it exhibits a diffuse liquid phase. Above 15 mN/m, the polymer adopts a compressed liquid phase until 50 mN/m, where it collapses into a solid phase. Macrophages imaged at 96 h on glass coatings with polymer deposited at (B) 5 mN/m, (C) 10 mN/m, (D) 20 mN/m, and (E) 30 mN/m illustrate lower levels of accumulation when high deposition surface pressures are used.
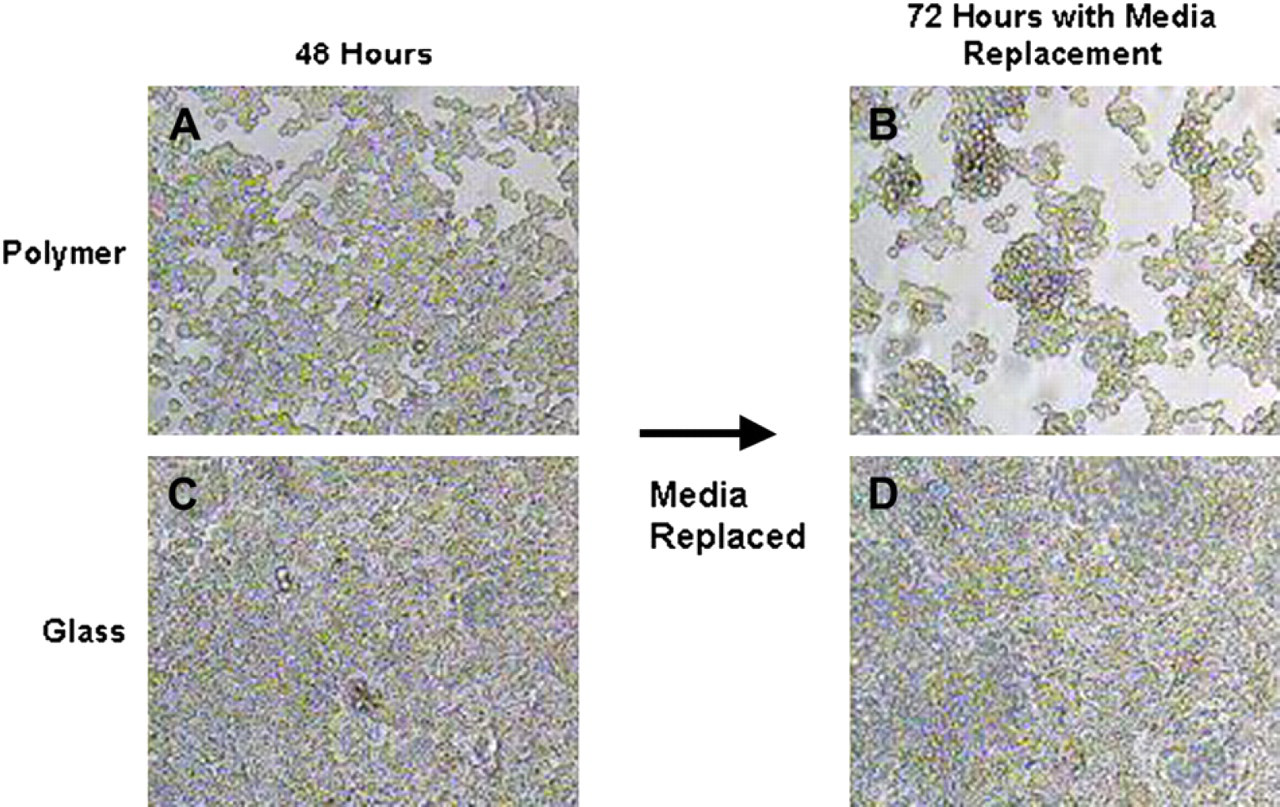
Media replacement causes macrophage denudation on triblock-copolymer substrate. Reduction of cell density occurred on triblock copolymer surfaces after replacement of growth media. Macrophages were imaged on polymer deposited at 20 mN/m after (A) 48 h and (B) 72 h with media replacement; (C), (D) show growth of cells on glass after 48 h and 72 h with media replacement, respectively.
Hydrophobicity Measurements of the Block Copolymers
Monolayer membranes fabricated from the PEO–PMMA diblock copolymer differed from the previously created membranes in two principal ways excluding the variation of polymer endgroups. The surface topography changed due to diblock's shorter chain length and the degree of hydrophobicity increased due to the exposure of different endgroups. The latter change was predicted because the diblock copolymer's hydrophilic end block was likely to adhere to the hydrophilic glass surface during deposition, exposing the hydrophobic end block as a cellular substrate. This prediction was verified using water contact angle measurements (Fig. 4). Glass was found to have an average contact angle of 29.3°. The triblock copolymer deposited on glass at 25 mN/m had a slightly increased degree of hydrophobicity with an average contact angle of 49.2°. Although the triblock used has hydrophilic endgroups, natural formation of three-dimensional structures and bending of the polymer likely caused some of the interior hydrophobic blocks to be exposed. The diblock copolymer also deposited at 25 mN/m on glass had the highest level of hydrophobicity with an average contact angle of 68.9°. Again, this result was probably due to the diblock exposing its hydrophobic PMMA end block.
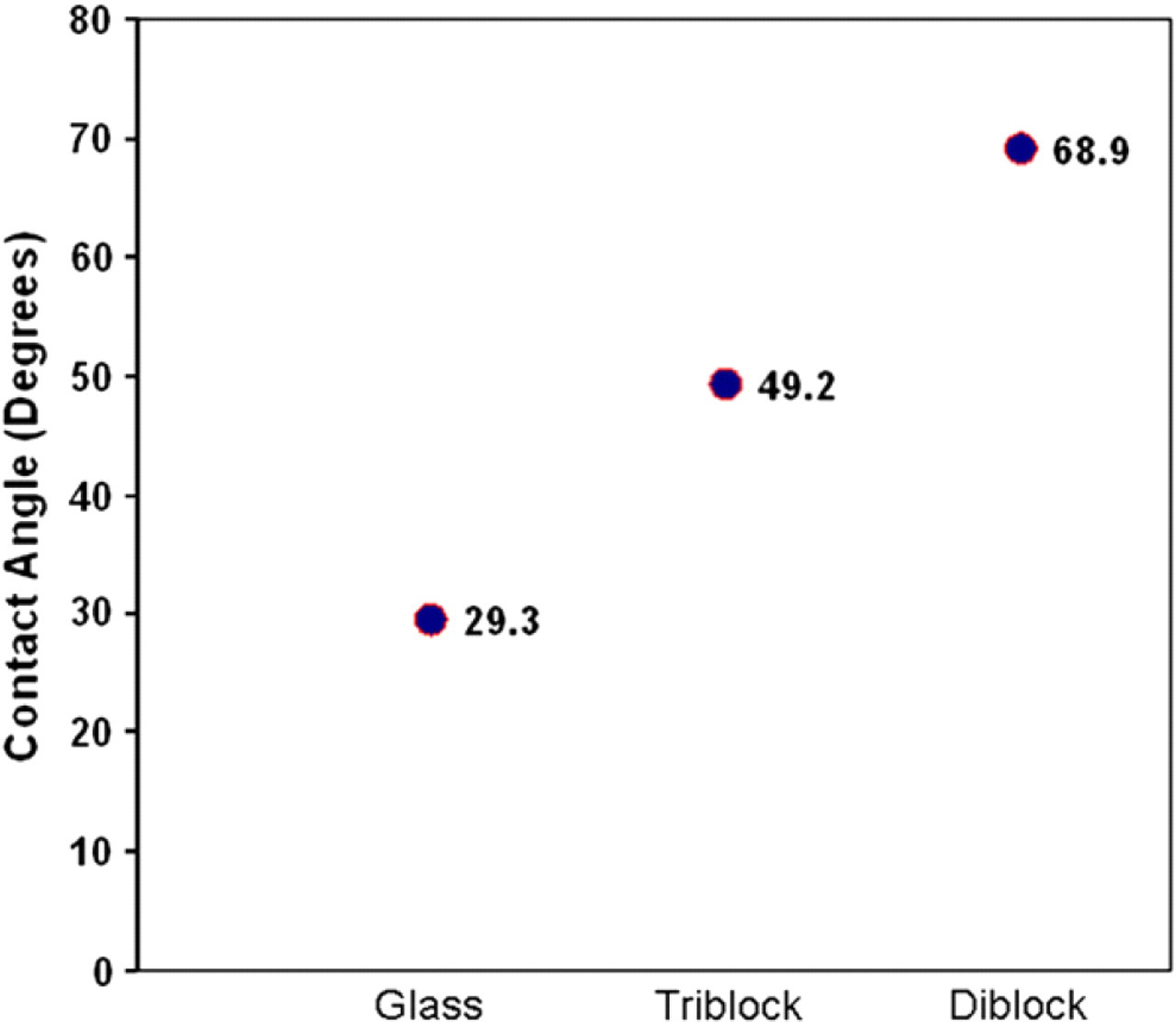
Contact angle measurements of water droplets on polymer-coated surfaces. Water droplets create contact angles with the substrates on which they adhere. Water droplets on pure glass, triblock-coated glass, and diblock-coated glass have contact angles of 29.3°, 49.2°, and 68.9°, respectively.
Incorporation of Diblock Copolymer into the Monolayer Membranes
Glass slides coated at 25 mN/m with pure diblock copolymer, pure triblock copolymer, and several mixtures of the two copolymers in specific ratios showed significantly varying results when macrophages were cultured on these substrates. The ratios used to create the mixed monolayer membranes were Triblock-to-Diblock 9:1, 7:3, 5:5, 3:7, and 1:9 mN/m. After 48 h, all mixed monolayer membrane coatings inhibited cellular accumulation on glass substrates to some extent (Fig. 5D-H), but the coating composed of equal ratios of the triblock and diblock copolymers showed the greatest anti-adsorptive activity (Fig. 5F). In contrast, pure diblock copolymer membrane coatings had little or no ability to inhibit cellular accumulation (Fig. 5C). The dense growth seen on diblock copolymer-coated substrates could be due to the macrophages' preference for hydrophobic substrates, 38 or it could have been due to the PMMA end block lacking the ability to prevent non-specific protein adsorption. These findings suggest that the mere presence of biomimetic copolymers was not enough to cause anti-cell accumulation, but rather an intrinsic property of the triblock copolymer was responsible for inhibition of accumulation early in the cellular adhesion and growth process. The fact that mixed monolayer membrane coatings composed of as little as 10% triblock copolymer could inhibit cellular accumulation indicates several possibilities. The first would be that triblock was solely responsible for inhibiting macrophage accumulation, and was capable of doing so even in small amounts. This is a possibility because we observed earlier that triblock copolymers deposited as low as 5 mN/m onto glass provided some degree of adsorptive inhibition. This conclusion, however, is not likely because we should then have expected to see a triblock-dependent trend of anti-adsorptive activity, namely, as the percentage of triblock copolymer increased within a film, less cellular accumulation should have occurred. Figure 5 clearly shows that this was not true, as mixed monolayers composed of Triblock-to-Diblock 1:9, and 9:1, respectively, exhibited similar degrees of accumulation inhibition. A second and more likely possibility is that mixing the two types of polymers provided a variation in nanoscale texture that the cells were capable of detecting. This is a likely possibility because several studies reported that macrophages preferentially migrated toward specific textures when presented with rough and smooth substrata. 38,39 The specific preferences of macrophage adherence in regard to the nanofilm surface textures are still to be investigated. Furthermore, it was also possible that the mixed polymers interacted in an unpredictable way, forming three-dimensional structures or deformations that were also conducive to cell adsorption inhibition. It is unclear why polymer films with an equal ratio of Triblock-to-Diblock exhibited the most inhibition, but it may have been due to this film composition providing the optimized nanosubstrate texture for highly effective prevention of macrophage accumulation. This possibility can further be investigated with atomic force microscopy (AFM). Fluorescent imaging further illustrated the vast difference between cellular accumulation on uncoated glass and glass coated with properly designed polymer nanofilms (Fig. 6). These junctions were created because it is a challenge to completely coat glass slides with polymer films due to the nature of Langmuir deposition, but they serve as distinct markers of where the polymer coating ends and pure glass begins.
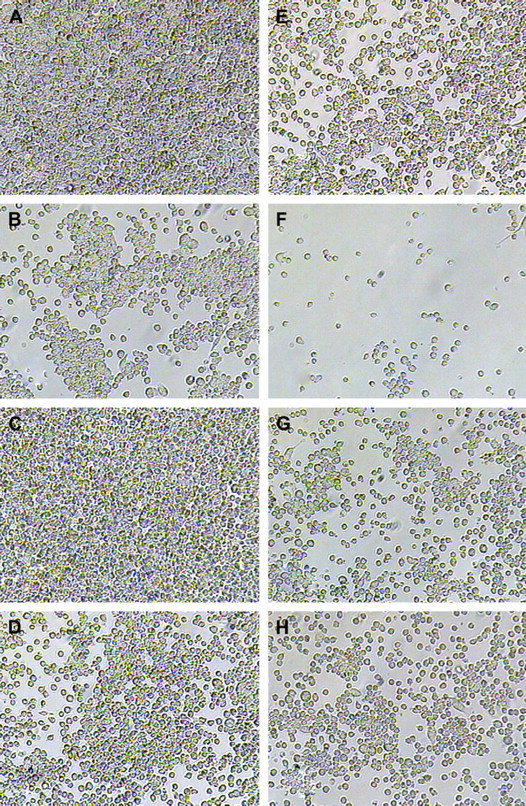
Macrophage accumulation on nanofilms composed of a mixture of diblock and triblock copolymers. Macrophages grown on (A) pure glass, (B) pure triblock, (C) pure diblock, and mixed membrane monolayers of composition Triblock-to-Diblock (D) 1:9, (E) 3:7, (F) 5:5, (G) 7:3, and (H) 9:1 for 96 h. Notes of interest include the total lack of anti-adsorptive activities shown by diblock copolymer films (C) and the ability of films composed of equal quantity of triblock and diblock copolymers to significantly inhibit macrophage accumulation.
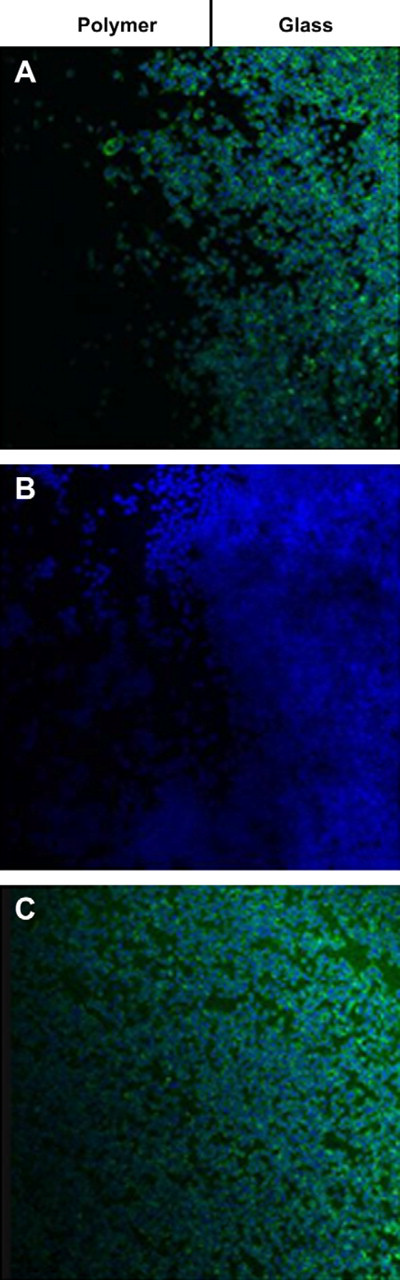
Fluorescent microscopy of macrophages grown on glass–polymer junctions. Flourescent images of the glass–polymer junctions of glass slides coated with (A) 1:1 triblock:diblock film, (B) pure triblock film, and (C) pure diblock film. The green stain represents actin and the blue stain represents cell nuclei. The left half of each image is the glass area coated with polymer and the right half is pure glass.
Assessment of the Inflammatory Response of Macrophages on Copolymer Substrates
When in contact with polymer implants in vivo, macrophages activate and release inflammatory cytokines that directly lead to fibrous capsule formation. 40,41 Therefore, it is of key interest to investigate whether potential nanofilm coatings are able to suppress activation and the inflammatory response of macrophages upon adherence. Cytokines such as interleukin 1 (IL-1), colony-stimulating factor (CSF), and interferon 1 are known major chemical signals that recruit and activate other leukocytes near the site of implantation. 40 -43 Interleukin 6 (IL-6) has also been identified as a useful indicator of inflammation in vivo. 44 -46 By monitoring these cytokines, we may be able to indicate whether our block copolymer films are capable of inhibiting aspects of the inflammatory response of macrophages in vitro.
We have found that coating glass substrates with the triblock copolymer may be able to suppress the inflammatory response of macrophages in vitro to some extent. Consistent results have been found for two cytokines in RT-PCR experiments. First, we showed that IL-1 expression levels were decreased on triblock nanofilms and increased on diblock nanofilms (positive indicator for assay performance) with respect to glass (Fig. 7A). Second, IL-6 seemed to be unaffected with the use of polymer nanofilms (Fig. 7B), indicating that these films alone do not elicit an inflammatory response which further confirms their translational applicability. The likely reason for the absence of pro-inflammatory cytokine gene expression is that the triblock is a biologically inert material due to its PMOXA endgroups which further inhibit unspecific protein adsorption. These serve as important parameters that mediate amenable responses of the macrophages toward synthetic materials. These properties may also have significant downstream benefits/effects in regard to leukocyte activation and recruitment, signifying the translational relevance of the polymeric nanofilm platform.
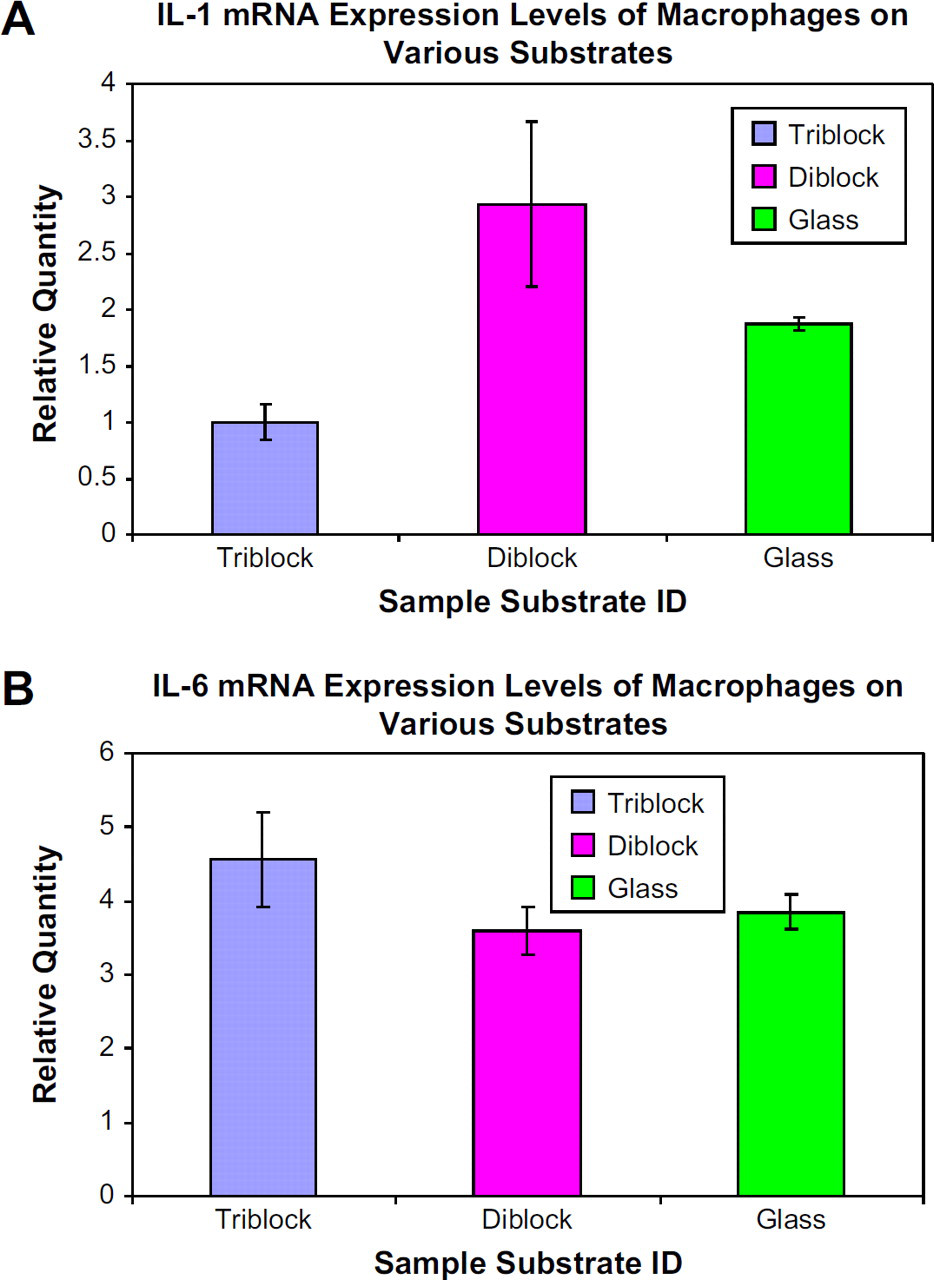
mRNA expression levels in macrophages grown on various substrates. (A) Macrophages grown on glass coated with triblock copolymer express reduced levels of IL-1 and glass coated with diblock copolymer express elevated levels of IL-1 with respect to pure glass. (B) IL-6 expression levels in macrophages are unaffected by substrates in this study.
Concluding Remarks
This work demonstrated the fabrication of hybrid diblock and triblock copolymeric nanofilms for the interrogation of cyto-adhesion behavior. Our findings have demonstrated the potential translational significance of hybrid membranes composed of equal ratios of diblock and triblock copolymers and their ability to potently inhibit cellular adhesion. This functionality may be applicable toward the coating of implants to suppress cellular accumulation at the implant-tissue interface, which is a typical precursor of implant breakdown. Additionally, quantitative RT-PCR measurements demonstrated the innate biocompatibility of the nanopolymer material at the gene expression level, signifying the impact that copolymeric nanomaterials can have toward nanoengineered medicine.
Acknowledgments
DH gratefully acknowledges support from National Institutes of Health grant U54 A1065359. ES gratefully acknowledges support from a McCormick Office of Corporate Relations Partners Grant.
