Abstract
We evaluated fermentation capabilities of five strains of Scheffersomyces stipitis (WT-2-1, WT-1-11, 14-2-6, 22-1-1, and 22-1-12) that had been produced by UV-C mutagenesis and selection for improved xylose fermentation to ethanol using an integrated automated robotic work cell. They were incubated under both facultative and anaerobic conditions to evaluate ethanol production on glucose, xylose, cellobiose, and a combination of all three sugars. The medium contained 50 g/L total sugar and 5 g/L yeast extract. The strains performed significantly better under facultative compared with anaerobic conditions. As expected, glucose was the most readily fermented sugar with ~100% fermentation efficiency (FE) under facultative conditions but only 5% to 16% FE anaerobically. Xylose utilization was 20% to 40% FE under facultative conditions but 9% to 25% FE anaerobically. Cellobiose was the least fermented sugar, at 18% to 27% FE facultatively and 8% to 11% anaerobically. Similar trends occurred in the sugar mixture. Under facultative conditions, strain 22-1-12 produced 19.6 g/L ethanol on glucose, but strain 14-2-6 performed best on xylose (4.5 g/L ethanol) and the sugar combination (8.0 g/L ethanol). Ethanol titers from glucose under anaerobic conditions were again highest with strain 22-1-12, but none of the strains produced ethanol from xylose. Future trials will evaluate nutrient addition to boost microaerophilic xylose fermentation.
Introduction
High petroleum prices, unstable supplies, and the adverse effects of petroleum-derived greenhouse gases continue to support efforts to develop renewable energy sources. 1 Research over the past 30 years has focused on lignocellulosic biomass for ethanol production. The U.S. Department of Energy (DOE) has estimated that there are 1.3 billion tons of available biomass (corn stover, wheat straw, municipal waste, plant residue, and forestry wastes) for use in ethanol production.2,3 Agriculture and forestry waste alone could possibly account for 60 billion gallons per year by 2030. That would help cut our petroleum uses by 30% and reduce carbon dioxide emissions. 4 The Energy Independence and Security Act of 2007 established the goal of producing 36 billion gallons of ethanol (with 21 billion gallons from lignocellulosic ethanol) by 2022. 5
One challenge for lignocellulosic ethanol is that the hydrolysates of almost all biomass sources contain a diverse mixture of sugars, including glucose, xylose, galactose, mannose, and arabinose, with the first two being the most abundant. Finding or developing microbes that are capable of efficiently fermenting these sugars has been a challenge, especially for xylose. Yeast capable of fermenting xylose were first discovered in 1980, and Jeffries and Kurtzman 6 provided an early review of developments in this field. If an efficient xylose ethanologen can be developed, ethanol yields could rise by 25%.7,8
The most promising organism for lignocellulose hydrolysate fermentation is Scheffersomyces stipitis (formerly Pichia stipitis), as it has the ability to ferment glucose, xylose, cellobiose, mannose, and galactose. 9 It can be found in decaying wood and in the larvae of organisms that inhabit wood. Its optimal temperature is 25 to 33 °C. S. stipitis produces β-glucosidase, allowing it to hydrolyze cellobiose into glucose. 10 S. stipitis metabolizes xylose to xylitol using xylose reductase and then uses xylitol dehydrogenase to produce ethanol. 5 Unfortunately, S. stipitis requires low levels of oxygen to ferment xylose, 11 and this would add a further process cost. Another limitation is that xylitol can accumulate due to an imbalance of cofactors 12 and become toxic, inhibiting further ethanol production. However, the yeast has the salicylhydroxamic acid (SHAM) pathway, which helps regulate cofactor levels, allowing further ethanol production. 5
The purpose of this study was to evaluate the fermentation capabilities of five strains of S. stipitis that had been produced by mutagenesis of wild-type S. stipitis and selected for improved xylose fermentation under anaerobic conditions. 13 They were analyzed for ethanol production on various sugar sources, under both facultative and anaerobic conditions. The goal was to identify a suitable strain to ferment both hexose and pentose sugars from lignocellulose under anaerobic conditions to minimize the requirement (and costs) for aeration. Because xylose can account for up to half of the sugars in hydrolyzed biomass, utilization would dramatically increase the ethanol production, making lignocellulosic ethanol a more feasible renewable fuel source.
Materials and Methods
Mutagenization of S. stipitis NRRL Y-7124
Preparation of isolates 14 and 22
Duplicate 2-L Fernbach flasks were prepared by adding 1 L of YM medium (0.3% yeast extract and 0.3% malt extract, 0.5% peptone [Becton Dickinson, Sparks, MD], 0.1% dextrose [Sigma, St. Louis, MO]) to each flask and inoculating with 20 mL of a 2-day YM 28 °C culture of wild-type S. stipitis NRRL Y-7124 (ARS Culture Collection, Washington, DC) from a 100-mL flask. Before irradiation, a sample of the log phase culture was taken from the Fernbach flask to obtain an estimate of cell numbers using a Reichert Neubauer/Bright-Line Hemacytometer (American Optical Corp., Buffalo, NY). The Fernbach flasks were incubated at 28 °C for 2 days with shaking at 100 rpm. The culture from each flask was divided into two Beckman 500-mL spin bottles and centrifuged in a Beckman Avanti J20 centrifuge (Beckman, Indianapolis, IN) at 20 °C for 20 min at 3000 rpm. Cell pellets were washed with sterile water, and each pellet was resuspended in 50 mL of sterile water. A 25-mL aliquot was taken from each resuspension and placed into a Marsh RR-0014 deep trough plate with baffled bottom (Marsh Biomedical Products, Inc., Rochester, NY). The plates were placed 14 cm below a source of UV-C radiation (234 nm; UPC, LLC Light Table [inverted]; Upland, CA) and irradiated for 1 min ( Fig. 1 , step 1).
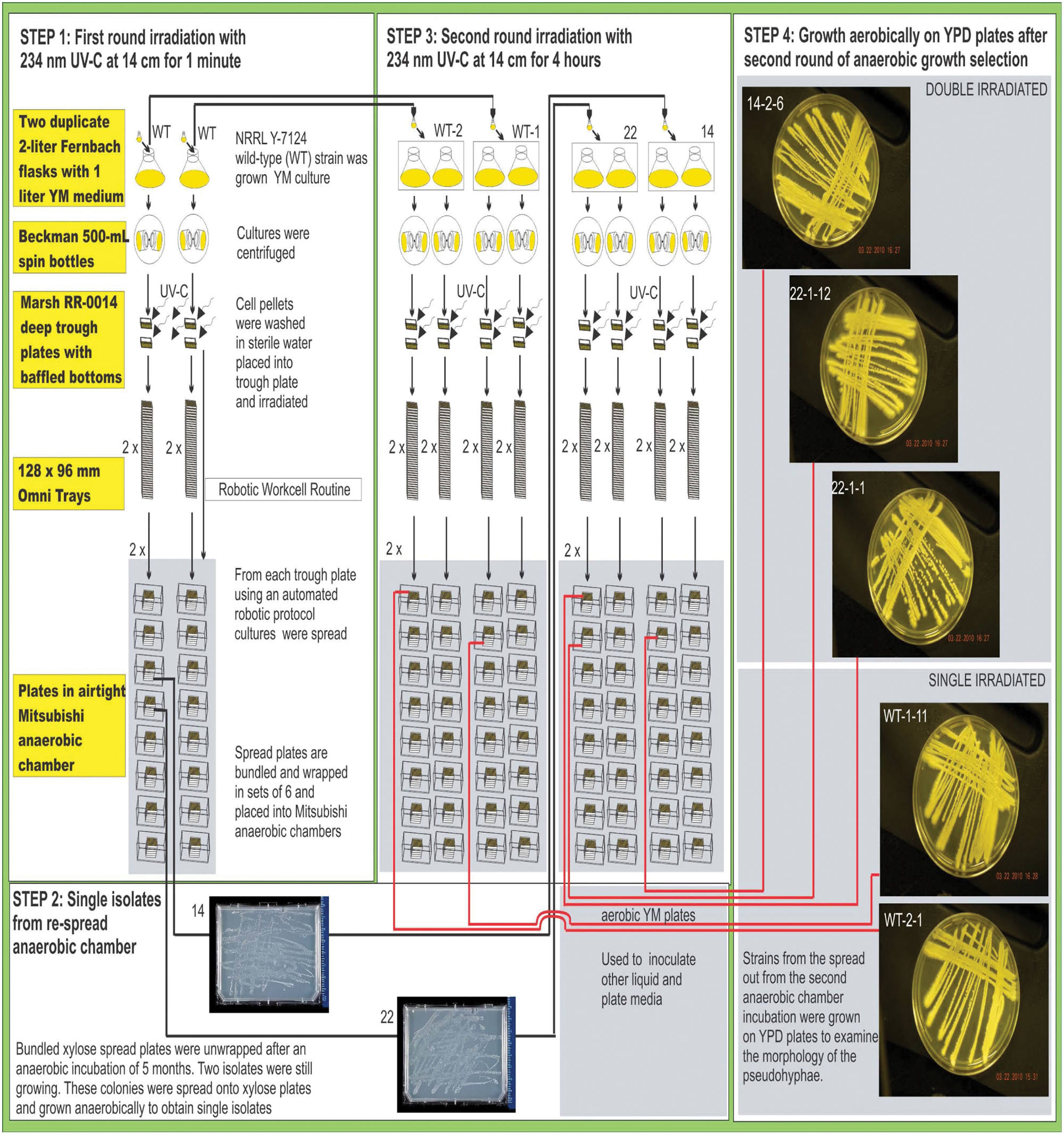
Process for production of Scheffersomyces stipitis NRRL Y-7124 mutants using UV-C irradiation.
From each trough plate, using an automated protocol on the robotic work cell ( Fig. 2 ), 600-µL aliquots were spread onto 128 × 96-mm Omni Tray plates (Thermo Fisher Scientific, Waltham MA) containing nitrogen base (6.7 g inositol, CaCl2, NaCl, MgSO4, (NH4)2SO4, and KH2PO4; Sigma) with 2% xylose complete minimal medium plus all amino acids, consisting of 1.4 g/L yeast synthetic dropout medium supplement (minus histidine, leucine, tryptophan, and uracil), 0.06 g/L leucine, 0.04 g/L tryptophan, 0.02 g/L histidine, 0.02 g/L uracil, and 20 g/L D-xylose (Sigma); 15 g/L Bacto Agar (Thermo Fisher Scientific); and 5 g/L ammonium sulfate (Sigma). The spread plates (96 plates per Fernbach flask and therefore a total of 192 plates prepared from the 1-min irradiation) were bundled into sets of six, wrapped in Saran Wrap, sealed with parafilm, and placed into a Mitsubishi anaerobic chamber (Mitsubishi Gas Chemical America, Inc., New York, NY) containing the AnaeroPack dry chemical system (Sigma Fluka, St. Louis, MO) at 28 °C for 5 months ( Fig. 1 , step 1).

Automated routine for spreading aliquots of UV-C–irradiated cells from trough plates onto plates with 2% xylose complete minimal medium plus all amino acids as sole carbon source. The program is run repetitively with groups of 6 plates, with 48 plates in a batch.
Two colonies, designated 14 and 22, were found still growing (above extensive background) when the bundled spread plates were unwrapped after this lengthy anaerobic incubation on xylose medium. Duplicate samples were picked from these colonies and spread onto plates containing rich YM or YPD (1.0% yeast extract, 2.0% Bacto Peptone, 2.0% D-glucose, and 20 g/L Bacto Agar [Thermo Fisher Scientific]) or 2% xylose complete minimal medium plus all amino acids and incubated at 28 °C for 2 weeks anaerobically to check that these isolates were still capable of growth on glucose and to eliminate background ( Fig. 1 , step 2). Five surviving colonies were picked from the respread anaerobic xylose plates onto plates containing YM, YPD, or xylose complete minimal medium plus all amino acids and incubated aerobically at 28 °C for 3 days to verify this growth capability was still present and to provide starter cultures for the second round of irradiation. Two 100-mL flasks were inoculated with samples from isolates 14 and 22 from the YM plates and incubated at 28 °C for 3 days to prepare samples for inoculation into Fernbach flasks for more intensive irradiation.
Preparation of strains WT-2-1, WT-1-11, 14-2-6, 22-1-1, and 22-1-12
Four 2-L Fernbach flasks were prepared in duplicate by adding 1 L of YM medium to each flask and inoculating the first two sets of flasks with wild-type S. stipitis NRRL Y 7124, the third set with isolate 14, and the fourth set with isolate 22 using 50-mL aliquots from the respective 100-mL flasks. Using the same procedures as described above, the Fernbach flasks were incubated, the cultures were centrifuged, and each of the cell pellets was washed and resuspended. A 25-mL aliquot was taken from each resuspension and placed into a Marsh RR-0014 deep trough plate with baffled bottom. The plates were placed 14 cm below a source of UV-C radiation and irradiated at 234 nm for 4 h ( Fig. 1 , step 3). The entire irradiation process was conducted in the dark. Stirring was conducted every hour to make sure all cells were equally exposed to the radiation. The liquid depth in the trough plates was less than 3 mm, and plates were centered under the light source.
Spread plates (192 plates for each of the four strains irradiated and therefore a total of 768 plates) were prepared on the robotic work cell as described above and in Figure 2 , moved to a HEPA-filtered hood, packaged into sets of six, wrapped in Saran Wrap, sealed with parafilm, and placed into a Mitsubishi anaerobic chamber containing the AnaeroPack dry chemical system at 28 °C for 5 months ( Fig. 1 , step 3). One sample from each of the 42 surviving colonies on the 768 anaerobic xylose plates was manually spread onto a plate containing rich YM, YPD, or xylose medium (one sample per plate) and incubated at 28 °C aerobically to check for growth on glucose and xylose and to obtain single isolates. After 3 days of aerobic growth on xylose, the colonies on five of the plates were considerably larger than those on any of the other plates, so these five samples were selected for further characterization and evaluation.
Organism and Culture Maintenance
Five engineered strains of S. stipitis (designated WT-2-1, WT-1-11, 14-2-6, 22-1-1, and 22-1-12) were obtained from the U.S. Department of Agriculture (USDA) ARS National Center for Agricultural Utilization Research. The strains were stored at 4 °C on potato dextrose agar (PDA) plates for short-term storage and in PDA slants covered with sterile mineral oil for long-term maintenance.
Experimental Design
The five S. stipitis strains were evaluated for growth and ethanol production on different sugar sources (glucose, xylose, cellobiose, and a combination), under both facultative and anaerobic conditions. Media were prepared by mixing 50 g/L of each individual sugar with 5 g/L of yeast extract. In the combination medium, we mixed 16.7 g of each sugar with 5 g/L of yeast extract. The pH of the various media was ~6. The optimal pH for S. stipitis fermentation of xylose is reportedly 5.5 to 6.14–16 For facultative trials, media (100 mL) were dispensed into 250-mL Erlenmeyer flasks equipped with a rubber stopper and needle/filter for gas exchange. For anaerobic trials, media (50 mL) were dispensed into 75-mL serum bottles, purged with nitrogen to remove oxygen, and sealed. Media were autoclaved prior to use.
Inoculum for each trial was prepared by subculturing into 100 mL of an appropriate medium in a 250-mL Erlenmeyer flask. For trials with glucose, cellobiose, and the combination medium, we used an inoculum medium containing glucose. For trials with xylose, we used an inoculum medium containing xylose. Inoculum cultures were incubated at 35 °C in a rotary shaker (200 rev/min) for 24 h prior to use. An inoculum volume of 1% v/v was used in all trials. Three trials were performed for each sugar or sugar combination. All flasks or serum bottles were placed into a rotary shaker (200 rev/min) set at 35 °C for 96 h. Samples (4 mL) were taken at 0, 3, 6, 9, 12, 24, 36, 48, 72, and 96 h. One milliliter was used for viable cell counts, whereas the remaining 3 mL was filtered through a 0.2-micron filter and placed in high-performance liquid chromatography (HPLC) vials that were frozen (–20 °C) until analysis.
Analytical Methods
Viable cells counts were performed using a hemocytometer. One milliliter of sample was diluted with 9 mL of distilled water and a drop of methylene blue. Samples were analyzed for glucose, xylose, cellobiose, xylitol, lactic acid, acetic acid, and ethanol using a Waters (Milford, MA) HPLC system with a refractive index detector. A Bio-Rad (Hercules, CA) Aminex HPX-87H column was used, and the mobile phase was 0.005 M H2SO4 at a flow rate of 0.6 mL/min.
Results and Discussion
Figures 3 to 6 show the fermentation efficiencies, maximum ethanol titers, ethanol yields, and ethanol productivities of the five engineered S. stipitis strains on various sugars under facultative conditions. Within each sugar trial, there was little variation between the five strains for fermentation efficiency ( Fig. 3 ). The only significant difference was that the WT-2-1 strain performed slightly better than the 22-1-12 strain on xylose, cellobiose, and combined sugar trials. As expected, overall fermentation efficiencies where highest with glucose (~100%), whereas xylose resulted in 20% to 40% fermentation efficiency. Cellobiose utilization efficiency was lower than expected at 18% to 27%. Evidently, these S. stipitis strains had weak β-glucosidase activity. Fermentation efficiency on the sugar combination was 38% to 40%, with complete utilization of glucose and partial utilization of the xylose and cellobiose as observed in the individual sugar trials. The fermentation efficiencies found by Hughes et al. 13 for mutagenized strains 14-2-6, 22-1-1, and 22-1-12 grown on xylose in a fermentor under microaerophilic conditions ranged from 12% to 15%, compared with 18% for wild-type S. stipitis grown under the same conditions measured at the point at which all of the glucose had been consumed (378 h). These fermentation efficiencies were substantially lower than the 96% reported by Nigam7,8 when S. stipitis was grown under microaerophilic conditions on a synthetic medium containing 26.7 g/L xylose, 3 g/L glucose, 6.5 g/L mannose, and 1.5 g/L arabinose.
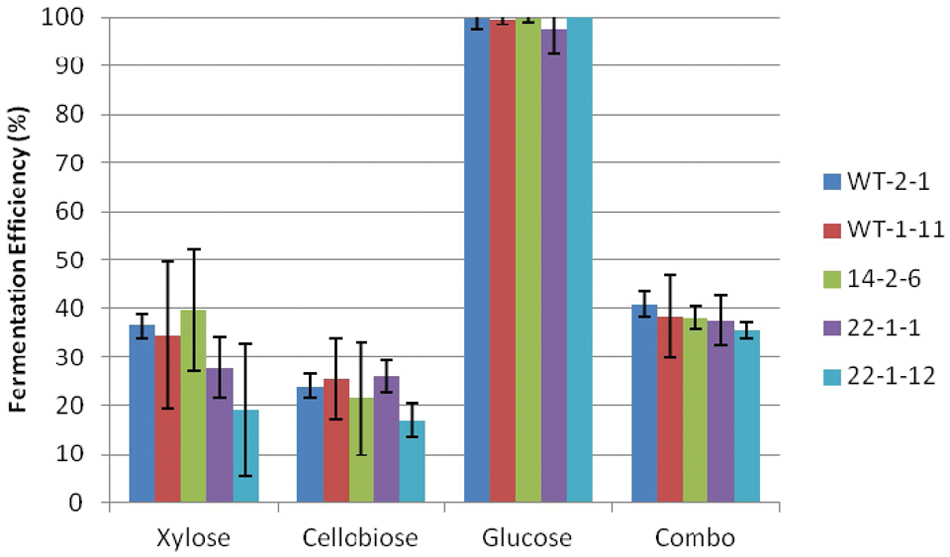
Fermentation efficiency of Scheffersomyces stipitis strains under facultative conditions.
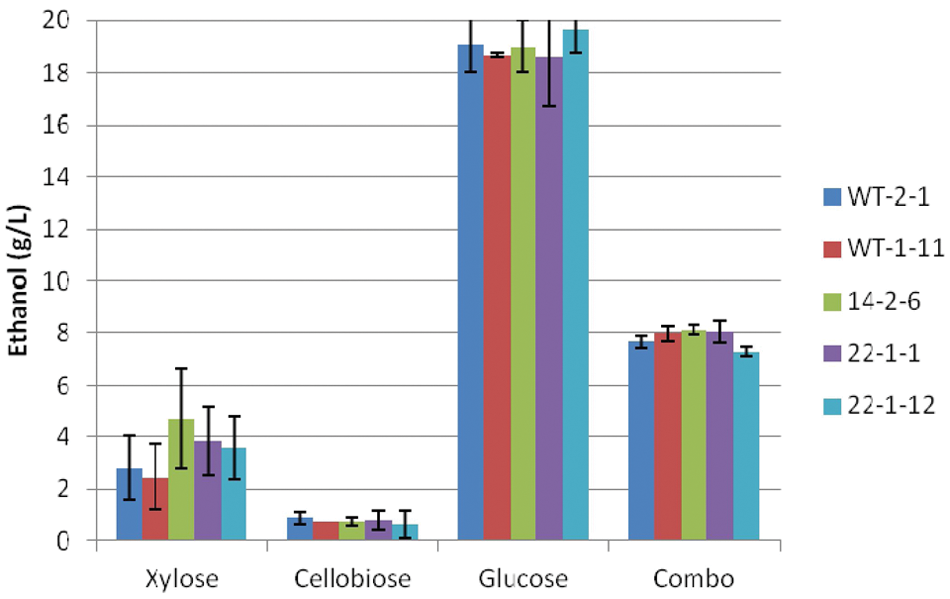
Maximum ethanol titers of Scheffersomyces stipitis strains under facultative conditions.
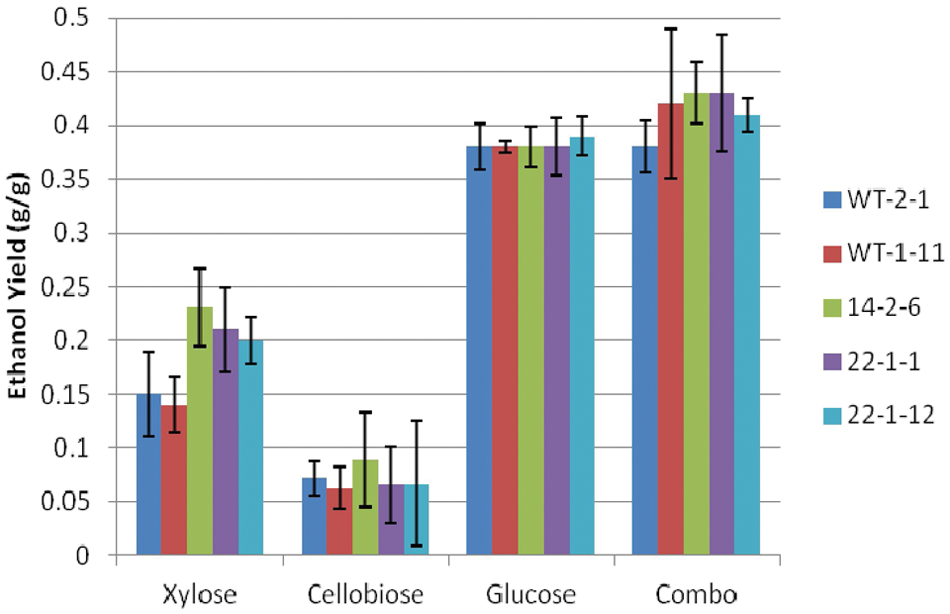
Ethanol yield of Scheffersomyces stipitis strains under facultative conditions.
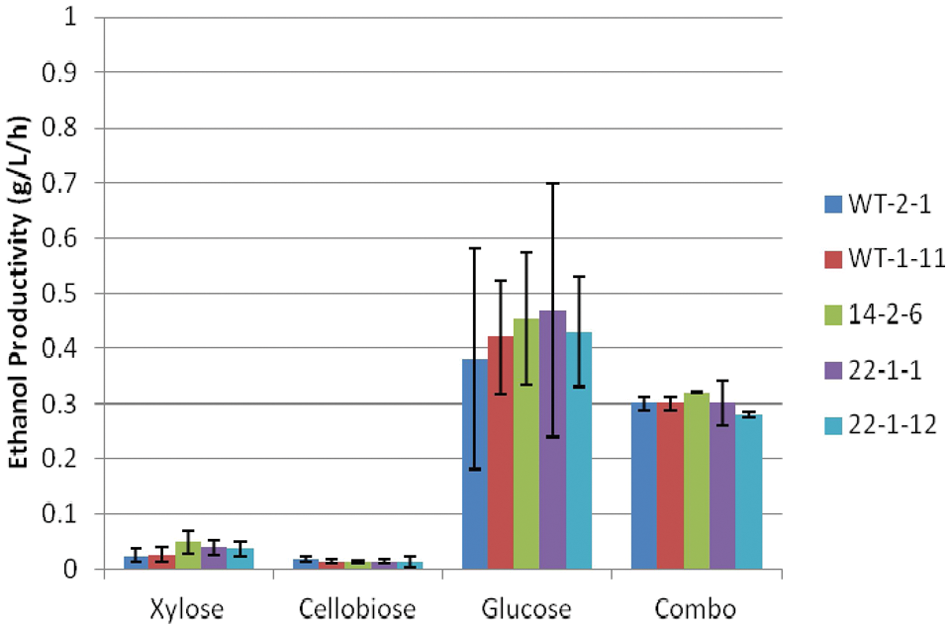
Ethanol productivity of Scheffersomyces stipitis strains under facultative conditions.
Maximum ethanol titers under facultative conditions for xylose and cellobiose showed no significant trends among the strains ( Fig. 4 ). Strain 22-1-12 performed slightly better than strain WT-1-11 on glucose, whereas strain 14-2-6 performed slightly better than strains WT-2-1 and 22-1-12 on the sugar combination. Glucose resulted in the highest maximum ethanol titers (18–20 g/L) as expected, whereas ethanol titers in the sugar combination trials were 7.5 to 8 g/L, due primarily to glucose fermentation. Xylose resulted in only 3 to 5 g/L ethanol, with only 1 g/L ethanol for cellobiose. These ethanol titers are well below the maximum levels (46–48 g/L) obtained by Preez et al. 17 on a synthetic xylose medium, as well as those reported by Amartey and Jeffries 18 on a xylose medium supplemented with corn steep liquor (28–30 g/L). The ethanol titer obtained by Hughes et al. 13 for the wild-type strain grown in a fermentor on combined xylose plus glucose medium under microaerophilic conditions was 44 g/L at the completion of the fermentor run (500 h) compared with 39 to 42 g/L for mutagenized strains 14-2-6, 22-1-1, and 22-1-12. Therefore, in the trials described above, ethanol inhibition of S. stipitis was not a limiting factor.
Ethanol yield was calculated as the amount of ethanol produced per gram of sugar consumed ( Fig. 5 ) to differentiate the relative efficiency of ethanol production by the strains. Although there was no difference between strains in the cellobiose, glucose, and sugar combination trials, strains 22-2-12, 22-1-1, and 14-2-6 outperformed strain WT-1-11 on xylose. Trends between the sugars showed an average yield of 0.38 g/g on glucose and 0.38 to 0.43 g/g on the sugar combination. These ethanol yields from glucose are lower than the 0.46- to 0.48-g/g yields common for Saccharomyces spp.19,20 Much lower ethanol yields occurred for xylose (0.14–0.23 g/g) and cellobiose (0.06–0.09 g/g), meaning that significantly more xylose and cellobiose were used for cell growth and maintenance. Numerous authors have reported ethanol yields of 0.34 to 0.42 g/g on synthetic xylose-based media or biomass hydrolysate.7–9,18,21
Ethanol productivity under facultative conditions exhibited little variation between strains for each sugar ( Fig. 6 ). Similar to the ethanol titer data, ethanol productivity was highest on glucose (~0.4 g/L/h) and the sugar combination (0.3 g/L/h). However, these productivities are much lower than the 2- to 3-g/L/h rates achieved by Saccharomyces cerevisiae.19,20 Agbogbo and Coward-Kelly 5 also stated that S. stipitis has a slower sugar consumption rate than S. cerevisiae. Ethanol productivities on xylose and cellobiose were much lower. Lawford and Rousseau22,23 noted that ethanol production from xylose yields less adenosine triphosphate (ATP) than from glucose, resulting in a lower productivity. However, Nigam7,8 reported a productivity of 0.4 g/L/h on a synthetic medium containing primarily xylose, whereas Amartey and Jeffries 18 obtained a productivity of 0.93 g/L/h with Pichia (now Scheffersomyces) grown on xylose supplemented with corn steep liquor.
When grown under anaerobic conditions, we observed generally reduced performance for all four parameters evaluated. In their review of S. stipitis, Agbogbo and Coward-Kelly 5 noted that S. stipitis requires microaerophilic conditions for ethanol production. In our prior work, 13 we observed that the wild-type S. stipitis did not grow anaerobically. However, the mutagenized strains of S. stipitis evaluated in this study were selected on xylose plates incubated under anaerobic conditions, and we had anticipated they would exhibit enhanced fermentation performance under anaerobic conditions. Figure 7 shows that fermentation efficiency was substantially lower for each of the sugars, compared with trials conducted under facultative conditions ( Fig. 3 ). Fermentation efficiencies ranged from 10% to 30%, meaning that some oxygen was required for metabolism. Skoog and Hahn-Hagerdal 11 discuss the role of oxygen during conversion of sugars (especially xylose) to ethanol by P. stipitis and determined that the optimal ethanol yield on xylose (0.48 g/g) was obtained at an oxygen transfer rate below 1 mmol/L/h. There were some significant differences in fermentation efficiency between yeast strains for each sugar. However, we observed no consistent trend.
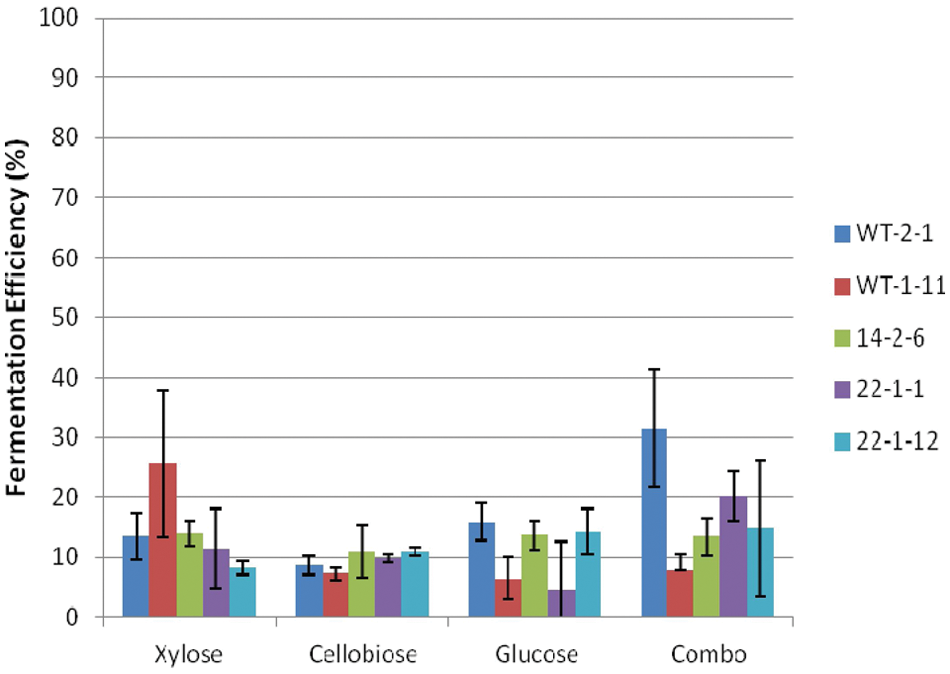
Fermentation efficiency of Scheffersomyces stipitis strains under anaerobic conditions.
Maximum ethanol titers observed under anaerobic conditions on cellobiose ( Fig. 8 ) were similar to that observed under facultative conditions ( Fig. 4 ) but were significantly lower for all other sugars and the sugar combination. In fact, on xylose, no ethanol was produced under anaerobic conditions, which is consistent with the known requirement of S. stipitis for trace amounts of oxygen. 11 Ethanol titers from glucose were ~1 g/L, which is much lower than typically reported with S. stipitis and S. cerevisiae. The sugar combination trials actually produced higher ethanol titers of 2 to 4 g/L, which was unexpected compared with the facultative trails. No significant trends were observed between yeast strains in individual sugar trials.
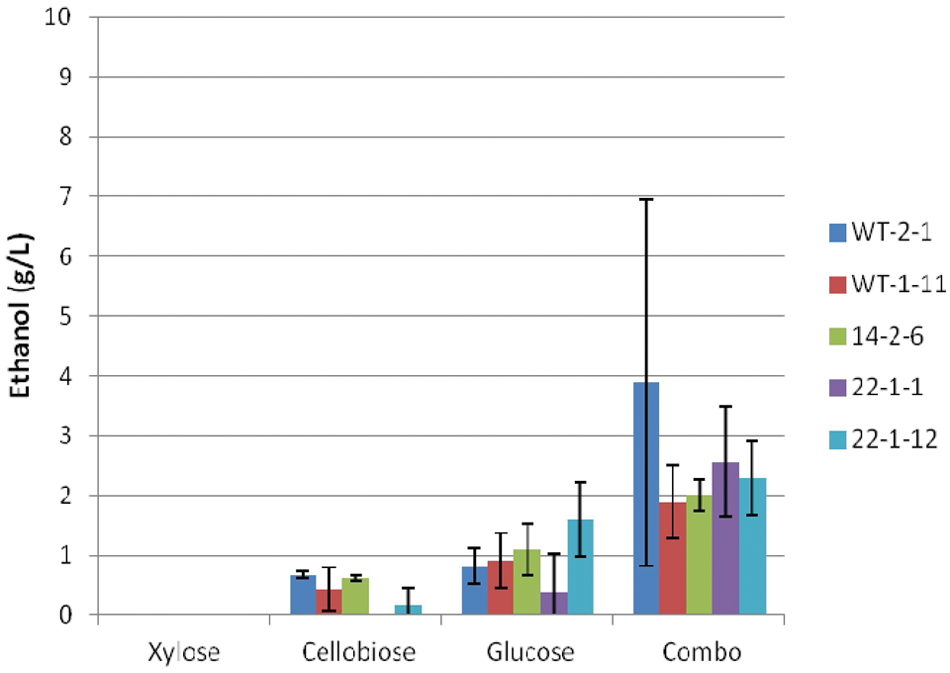
Maximum ethanol titers of Scheffersomyces stipitis strains under anaerobic conditions.
Other than a slightly higher ethanol yield with cellobiose, ethanol yields for the other sugars were much lower anaerobically ( Fig. 9 ) than under facultative conditions ( Fig. 5 ). This corresponds with the lower ethanol titers. Similarly, we observed much lower ethanol productivity under anaerobic conditions ( Fig. 10 ). Again, there were no significant trends for either ethanol yield or productivity among the strains grown on each individual sugar. Microbial stress from lack of oxygen may explain the large variability observed. 11
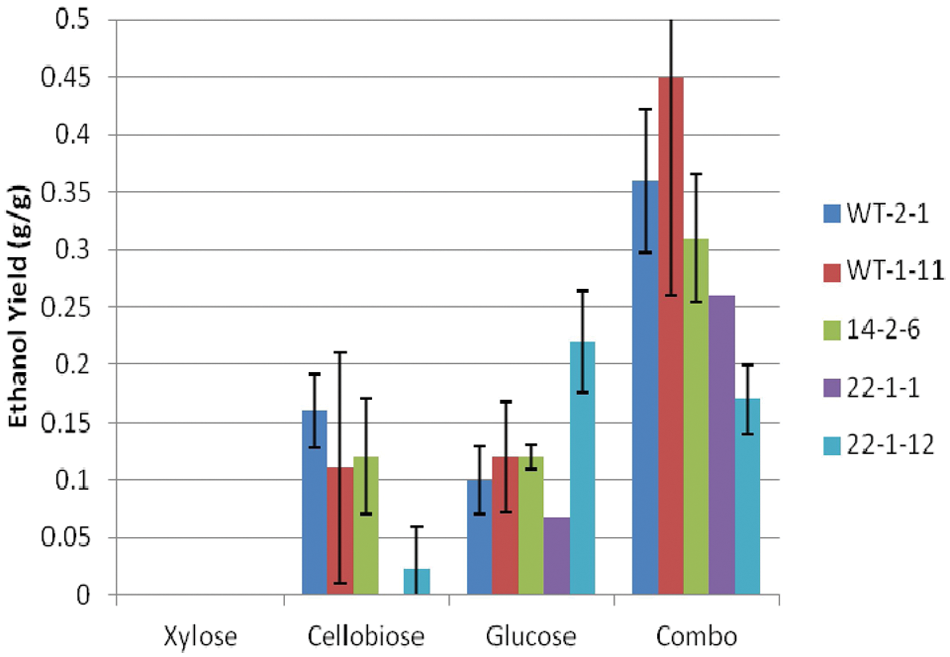
Ethanol yield of Scheffersomyces stipitis strains under anaerobic conditions.
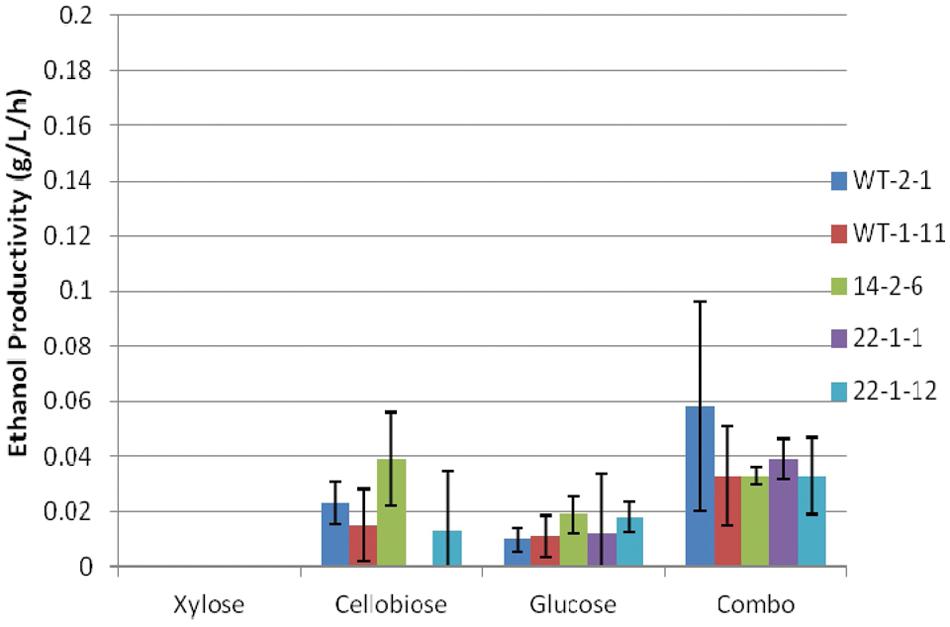
Ethanol productivity of Scheffersomyces stipitis strains under anaerobic conditions.
From the results, it is clear that S. stipitis requires some oxygen for growth on xylose. Microaerophilic conditions provide the optimal environment for this organism to convert xylose to ethanol. 11 The organism evidently also benefits from oxygen when grown on glucose and cellobiose since fermentation efficiency and ethanol titers were much higher for the strains grown in aerobic conditions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors acknowledge the financial support received from the North Central Regional Sun Grant Center at South Dakota State University through a grant provided by the US Department of Transportation, Office of the Secretary, Grant No. DTOS59-07-G-00054.
