Abstract
Pathology has developed substantially since the 1990s with the introduction of total laboratory automation (TLA), in response to workloads and the need to improve quality. TLA has enhanced core laboratories, which evolved from discipline-based laboratories. Work practices have changed, with central reception now loading samples onto the Inlet module of the TLA. It is important to continually appraise technology. This study looked at the impact of technology using a self-administered survey to seniors in clinical biochemistry in NATA GX/GY–classified laboratories in Australia. The responses were yes, no, or not applicable and are expressed as percentages of responses. Some of the questions sourced for descriptive answers. Eighty-one laboratories responded, and the locations were 63%, 33%, and 4% in capital cities, regional cities, and country towns, respectively. Forty-two percent were public and 58% private. Clinical biochemistry was in all core laboratories of various sizes, and most performed up to 20 tests per sample. Thirty percent of the 121 surveyed laboratories had plans to install an automated line. Fifty-eight percent had hematology and biochemistry instrumentations in their peripheral laboratory, and 16% had a STAT laboratory on the same site as the core laboratory. There were varied instruments in specialist laboratories, and analyzers with embedded computers were in all laboratories. Medium and large laboratories had workstations with integrated instruments, and some large laboratories had TLA. Technology evolution and rising demand for pathology services make it imperative for laboratories to embrace such changes and reorganize the laboratories to take into account point-of-care testing and the efficiencies of core laboratories and TLA.
Introduction
The clinical laboratory has evolved rapidly since the 1990s, driven mainly by technological advances that focus on automation. 1 Advances in therapy and basic sciences have also contributed to drive clinical laboratory automation (Markin 1996, cited in Zaninotto and Plebani 2010). 1 -8 The late 1970s saw the beginning of a new generation of laboratory automation with developments in robotics and information technology, which are critical elements of automation because these are associated with all the phases of analysis, including before and after as well as quality assurance. 1 The pattern of evolution of the laboratory was tool-machine-automation (Schmidt 1941, cited in Buttner 1982), 9 and the first automated instrument was the Technicon AutoAnalyser introduced in 1956 (Skeggs 1957, cited in Zaninotto and Plebani 2010). 1
As pathology grew, discipline-based laboratories evolved, and then core laboratories appeared, with at least two disciplines. Today, we are in the age of total laboratory automation (TLA). Hawker et al. 10 defined TLA as automation that includes preanalytical and postanalytical functions combined with analytical activities (analyzers) that are interfaced directly with the automation system. Melanson et al. 11 saw automation as a customized process in analysis, ranging from automating a few steps to TLA. The level of automation depends on the needs and resources of laboratories, and the reasons for automation also vary. In Australia, there has been an increase of about 8% per year in pathology testing without a matching rise in funding. 12 Urbis 13 in a survey of the pathology workforce (RFQ 345/0809) reported that reliance on pathology was increasing, and according to an Australian Institute of Health and Welfare General Practice Activity in Australia study, pathology was ordered in 29.7% of all general practitioner encounters in 2000–2001, and this rose to 36.7% in 2004–2005. Advances in communication and information technology have also allowed the development of networks of laboratories that provide a comprehensive pathology service to smaller hospitals and communities. The range of the analytical work is extensive, with large laboratories now performing in excess of 100 different analyses 14 and the number of requests reaching more than 5000 per day.
TLA systems are available for clinical biochemistry, hematology, serology, and urinalysis, and such facilities, among others, accept and sort specimens; decap; transport to a centrifuge, aliquoter, analyzers; and recap and deliver for storage in refrigerated stockyards. 1,10 Analyzers with embedded computers and TLA have created practices that permit loading of analyzers and the input modules for an automated line by technicians and central reception staff. Zaninotto and Plebani 1 called TLA “human-less” robotic laboratories that could allow better operation with less human errors. Peck-Palmer 15 reported that the introduction of TLA was an effort to standardize laboratory testing and improve patient care. Hawker et al. 16 reported that a yearly growth in workload and the competitive cost pressures are factors that lead them to consider some form of TLA. These authors’ laboratory was cognizant of the need to keep up with anticipated growth by increasing the capacity to handle new work, and with the introduced automation, they observed improvements in turnaround time. 17 Melanson et al. 11 noted that although high standards in safety and quality of service were expected from clinical laboratories, satisfying such requirements was problematic because of the reductions in staff and the need to control expenditures. Berman 18 stated that a manual system of 42 steps of sample handling can be reduced to 19 steps with complete automation. This substantially decreases the likelihood of sample errors and therefore improves patient safety. Hence, automation provided a solution, and it is now widely implemented (Tatsumi et al. 1999; Sasaki et al. 1998; Park et al. 2002; and Trevino 2007, cited in Melanson 2007). 11 Melanson et al. 11 also noted that 17% of laboratories in the United States had TLA. Seaberg et al. 19 Sarkozi et al., 20 Holland et al., 21 and Melanson et al. 11 cited some benefits of automation as reductions in errors, increases in productivity, and improvements in turnaround time and safety, as well as allowance of reallocation of staff for growth and expansion.
Core laboratories that combine chemistry and hematology disciplines can also be automated, and ultimately each laboratory decides when to automate and the level of automation. 11 Asowata 23 reported on implementation of a core laboratory consisting of automated chemistry, urinalysis, coagulation, hematology, and flow cytometry in an effort to create a better and more effective clinical laboratory. In our experience, the core laboratory may be technology based and staff perform assays for a number of disciplines, and the results are verified or validated by staff of the particular disciplines, which retain intellectual ownership of the results. Major chemistry analyzers, today with a large test menu, have discrete analysis, and this facilitates the consolidation of small- and large-volume analyses on the single analyzer with the efficiencies of less sample handling, fewer instruments (and therefore less overhead costs), and less floor space.
The role of central reception in large laboratories has changed along with the change in technology, and manual labeling and recording of samples and requests are required less often. In addition, changes in technology have altered work practices in central reception, with staff now loading samples onto analyzers, either directly or via an automated line. Resource utilization has necessitated the creation of central or core (which may have TLA) and peripheral laboratories. Core laboratories may be defined as those that have a large range of chemistry and immunoassays, which are performed on automated instruments interfaced with a laboratory information system. It is therefore important to continually appraise technology in clinical biochemistry because the environment is continually changing. This study looked at technology and staffing and work practices in clinical biochemistry using a self-administered survey to senior staff in clinical biochemistry. It is recognized that some laboratories have stand-alone clinical biochemistry departments, while in other laboratories, clinical biochemistry is part of the core laboratory. Peripheral (a small range of chemistry and immunoassays required for immediate care), STAT (acute care chemistries only), and specialist (containing instruments that perform a more esoteric range of assays that require high technical skill and have long analytical times) laboratories may have multidisciplinary staff with equipment for clinical biochemistry, hematology, serology, and molecular biology; hence, it is difficult to separate clinical biochemistry from other disciplines.
Materials and Methods
This study had ethical clearance from the Ethics in Human Research Committee of Charles Sturt University, New South Wales, Australia. Self-administered questionnaires were sent to senior staff in charge of clinical biochemistry in NATA GX/GY–classified laboratories. The mode of contact was mail using a four-contact model, which has been demonstrated to be effective in increasing the response rate. 23 –25 The mailed packages followed the method of McCoy and Hargie, 26 which was an adaptation of that attributed to Fox et al. 23 The four-time contact method has also been advocated by a number of researchers. 27 –29 In the current study, the first contact consisted of an introductory letter, questionnaire, and self-addressed stamped reply envelope. The second was a reminder letter, the third contact consisted of another version of the introductory letter and the questionnaire, and the fourth contact was another version of the reminder letter. The time between each contact was approximately 6 wk, and mailing of the 4 contacts was personalized, a format advocated by McCoy and Hargie. 26 Personalization was indicated by a handwritten address, signing of the introductory letter, the use of postage stamps rather than franking, the letter addressed to the person rather than a generic greeting, and inclusion of a business card. Envelopes bearing the crest of Charles Sturt University were used, and the envelopes were individually stamped with the address handwritten.
The survey was sorted to ascertain information on the location and type of the laboratory (private or public) and also investigated the magnitude of workloads; availability of core, peripheral, STAT, and specialist laboratories; and the associated technologies in the laboratories. The responses were yes, no, or not applicable and are expressed as percentages of responses. Some of the questions sourced for descriptive answers to allow capturing of opinions.
Results
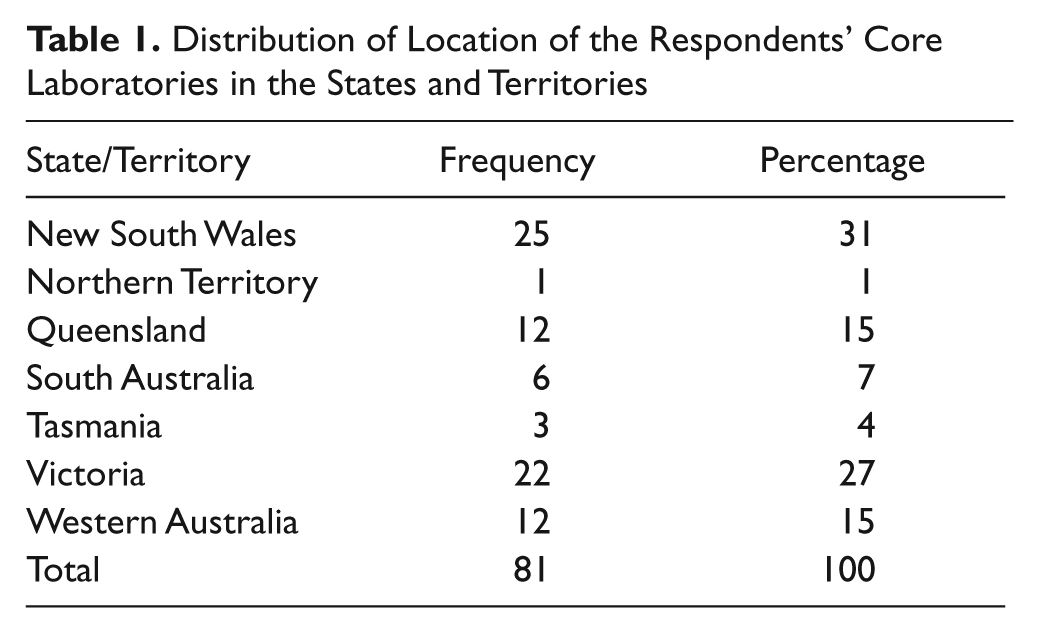
The largest group of the replies were from New South Wales, and no responses were received from the Australian Capital Territory ( Table 1 ). Of the 81 laboratories that responded (121 laboratories were surveyed), 63%, 33%, and 4% were in capital cities, regional cities, and country towns, respectively. Forty-two percent of the laboratories were public and 58% private.
Distribution of Location of the Respondents’ Core Laboratories in the States and Territories
Core Laboratories
Table 2 indicates that the largest group of laboratories were in public hospitals or were stand-alone laboratories, and a small fraction was situated in a teaching hospital (also public hospitals).
Distribution of the Type of Hospitals in Which the Respondents’ Core Laboratories Are Located

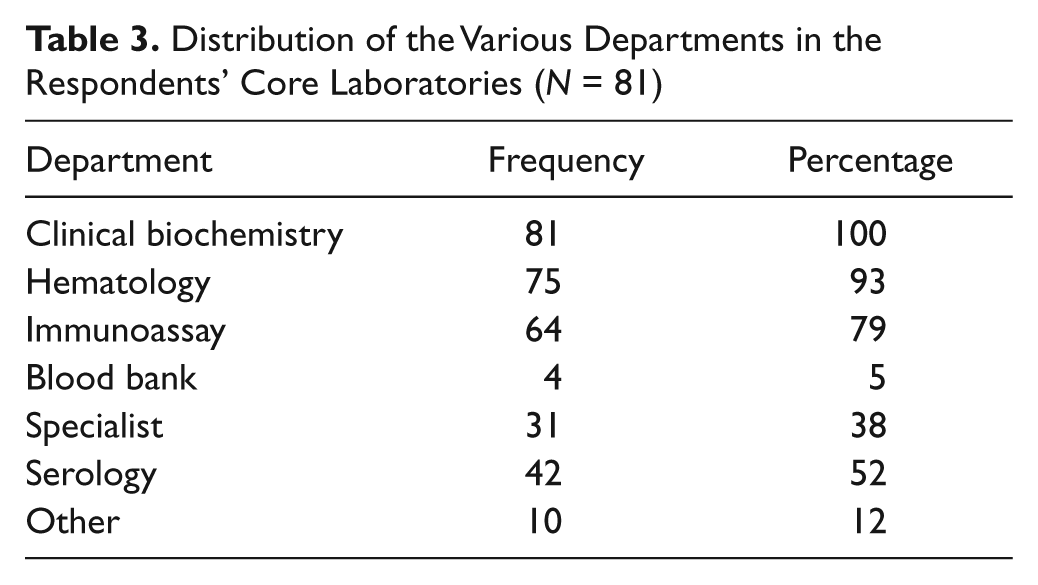
Clinical biochemistry was found in all core laboratories; few core laboratories have a blood bank, and serology and specialist units are sparingly found ( Table 3 ). In 80% of the 81 laboratories, the major analyzers were not linked to the automated line: 16% were linked, while in 1%, this was not applicable and 3% did not respond to this question. The respondents who indicated that an automated line was in operation also stated that the major analytical instruments were linked to it. Fifty-nine percent of the laboratories indicated that they had no plans to install an automated line, 30% had such plans, and in 9% of the responses, this was not applicable. Two percent did not respond to this item.
Distribution of the Various Departments in the Respondents’ Core Laboratories (N = 81)
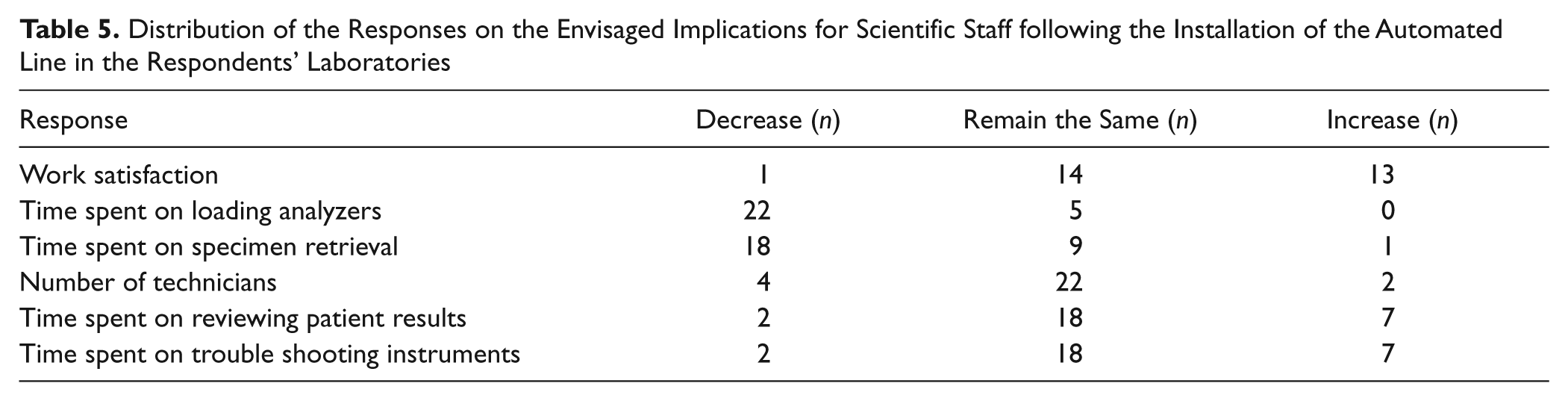
On the implications of installing the automated line, most of the respondents envisaged that work satisfaction among laboratory staff would remain the same or increase after installing the automated line. None of the respondents were of the opinion that time spent on loading analyzers would increase, but the majority thought that time spent on specimen retrieval would decrease. Most of the respondents were of the opinion that time spent on reviewing patients’ results and on troubleshooting instruments would not change after installing the automated line ( Table 5 ). Four laboratories responded to this question, although they did not indicate that they were planning to install an automated line.
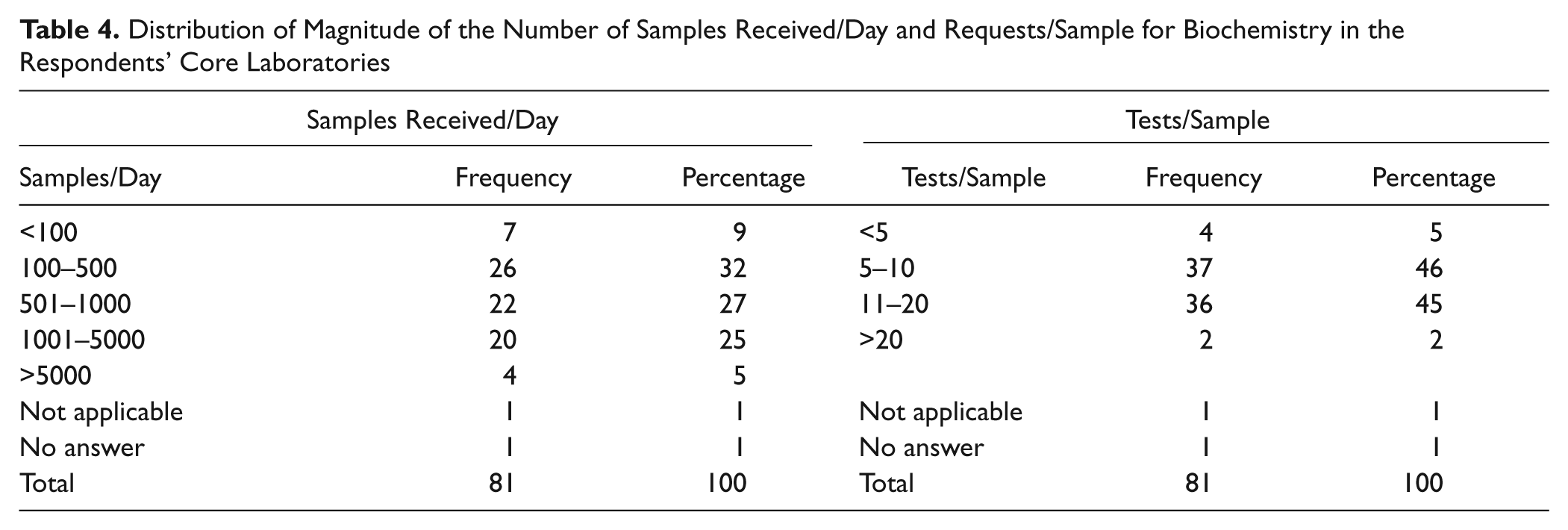
Table 4 shows that the majority of laboratories handle samples that number up to 5000 daily, and most laboratories perform up to 20 tests per sample. The same table also shows that in laboratories that handle a small number of samples, the number of requests/tests per sample is also small.
Distribution of Magnitude of the Number of Samples Received/Day and Requests/Sample for Biochemistry in the Respondents’ Core Laboratories
Distribution of the Responses on the Envisaged Implications for Scientific Staff following the Installation of the Automated Line in the Respondents’ Laboratories
Peripheral Laboratories
Forty-seven respondents indicated the instrumentation available in their peripheral laboratories. Microbiology had the lowest instrumentation, with clinical biochemistry and hematology having the highest ( Table 6 ).
Distribution of Availability of Instrumentation in the Peripheral Laboratories of Respondents’ Laboratories (n = 47)

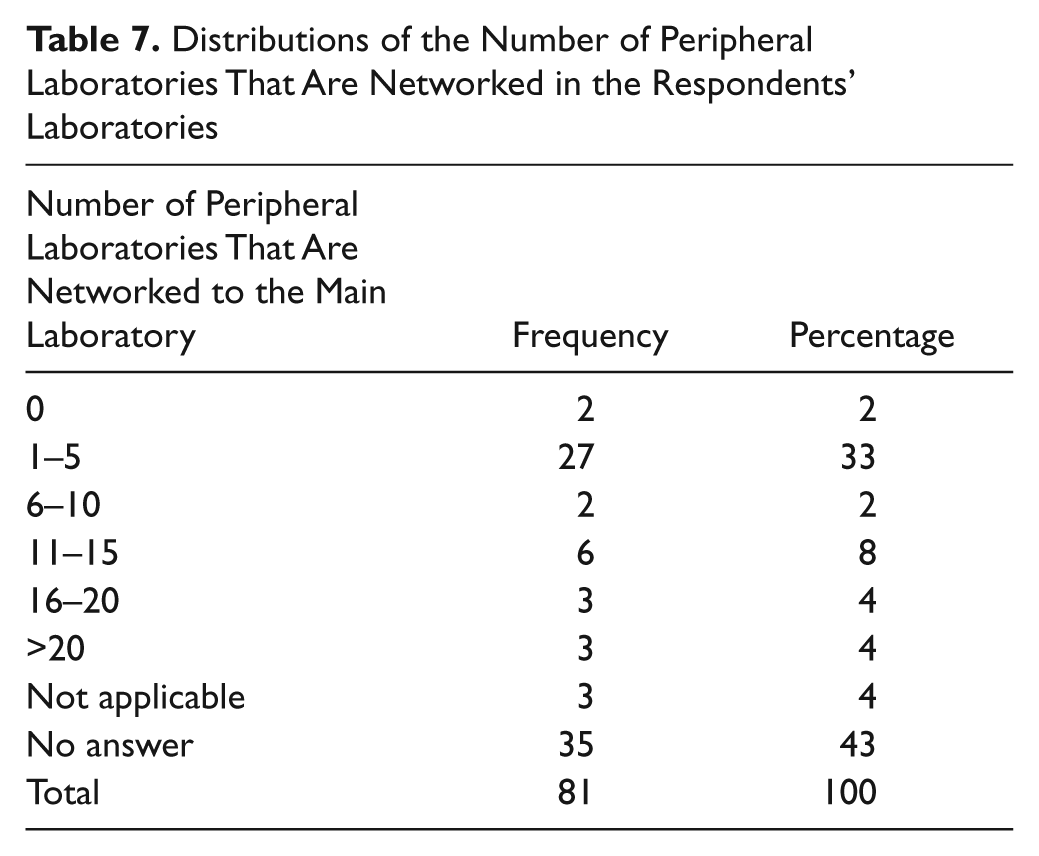
Fifty-five percent of respondents are part of a network of laboratories, and a third have one to five peripheral laboratories that are networked to the main laboratory ( Table 7 ).
Distributions of the Number of Peripheral Laboratories That Are Networked in the Respondents’ Laboratories
STAT Laboratories
Sixteen percent of the 81 laboratories have a STAT laboratory for urgent work on the site of the main laboratory and offer a 24-h service, and 12 of 13 of these operate a 24-h service.
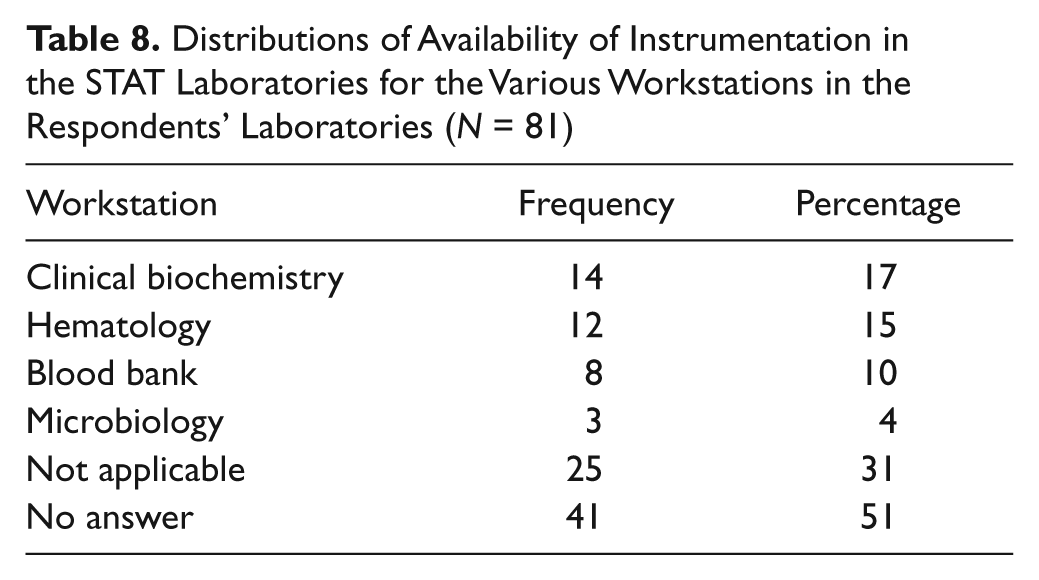
Table 8 shows that microbiology workstations are not usually found in a STAT laboratory. This question had multiple responses, and biochemistry had the highest availability of instrumentation, followed by hematology.
Distributions of Availability of Instrumentation in the STAT Laboratories for the Various Workstations in the Respondents’ Laboratories (N = 81)
Specialist Laboratories
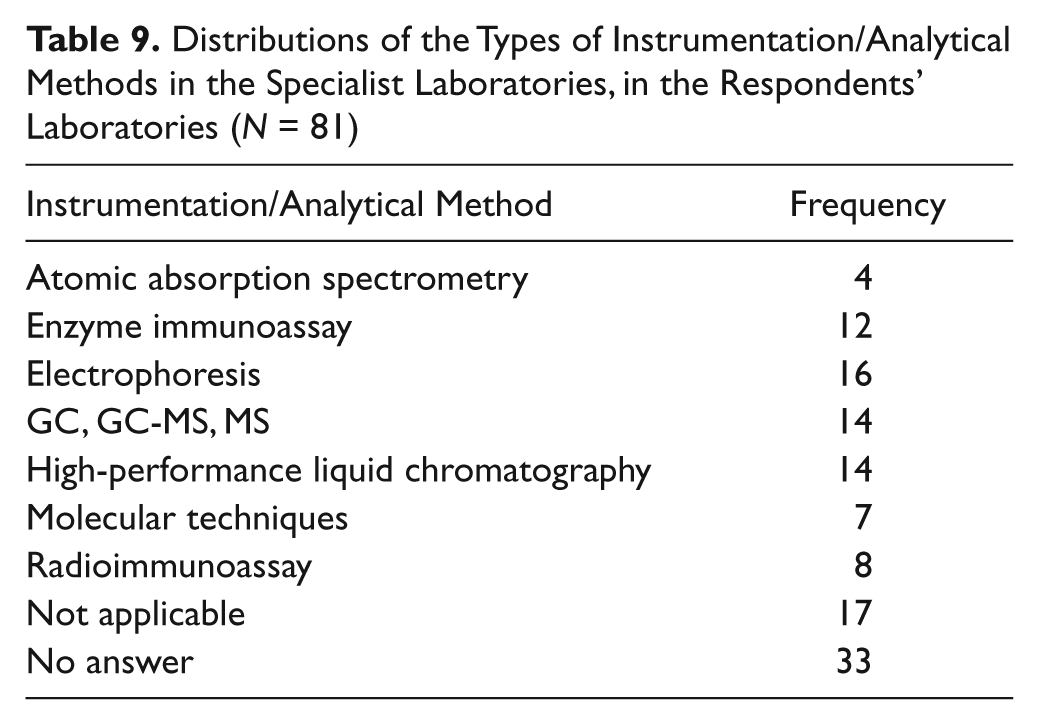
Table 9 shows that a varied range of instruments were used in the specialist laboratories. Atomic absorption spectroscopy, molecular techniques, and radioimmunoassays are used less in comparison with enzyme immunoassays, electrophoresis, and chromatographic techniques.
Distributions of the Types of Instrumentation/Analytical Methods in the Specialist Laboratories, in the Respondents’ Laboratories (N = 81)
Discussion
This study sought to investigate the extent of automation in laboratories and is part of a large study on the impact of technology on training and staffing in clinical biochemistry. Advances in technology have resulted in reorganization of most laboratories into, for example, core laboratory, periphery laboratory, STAT laboratory, and central reception.
Core Laboratories
The responses to the survey were from all states and territories in Australia except the Australian Capital Territory ( Table 1 ). The distribution of location of the laboratories was 63%, 33%, and 4% for capital cities, regional cities, and country towns, respectively, and it is assumed this was sufficient representation of the state of technology clinical biochemistry in Australia. The location of the majority of the laboratories in capital cities is perhaps a reflection of population distribution, with the majority of Australians living in major cities. A report for 2008–2009 on the population growth rates in Australian capital cities showed continued rise that was faster than the remainder of the country. 30 Legg and associates 12 also reported on maldistribution of health professionals, including pathology, with preferences to live in cities.
In this study, 58% of laboratories were private, while 42% were public; this is in agreement with the findings of Legg and associates, 12 who reported that the volume split between public and private pathology was not clear but was estimated at 60% private and 40% public. This split is augmented and reinforced by the spread of the location of the laboratories in private and public hospitals, with a significant number of laboratories operating as stand-alone laboratories, and all were private, apart from one ( Table 2 ). Such distribution between private and public may also be a reflection of the increases in private health provision in Australia.
The core laboratories surveyed consisted predominantly of biochemistry, hematology, and immunoassay ( Table 3 ). Automated serology was present in 52%, and specialist analytical work was performed in 38% of the laboratories. In our experience, automated serology is normally performed in core laboratories, but the acceptance of the results often remains within the microbiology or serology discipline, where the expertise in interpretation resides. The training of staff in the core laboratory for these assays usually comprises the practical aspects of analysis, calibration, and quality control.
Clinical biochemistry was present is all core laboratories surveyed, and this is perhaps indicative of its central role in pathology and its amenability to automation, which is a feature of the core laboratory. The majority of analyzers in the surveyed laboratories were not linked to an automated line. TLA occurs when the analytical instruments are linked to an automated processor and other linked modules that process samples. TLA configurations include automated centrifugation, aliquot module, and sample storage. This facility removes many of the work practices from reception (e.g., centrifuging of samples and making aliquot tubes as well as manual movement of samples between instruments). The removal of many manual steps assists greatly in preventing preanalytical errors, which are the largest source of error in the clinical biochemistry laboratory. 18 Some TLA configurations with sample storage connected to the automated line and bidirectional control of the automated line allow retrospective testing, and the analyses are performed without human intervention. TLA frees the scientific staff to perform higher duties such as results validation and troubleshooting instruments, rather than carrying out the lower-level duties of looking for specimens, loading analyzers, making aliquots, recording appearance of samples (e.g., hemolysis), and storing samples. It is possible that if more time is devoted to challenging tasks by scientific staff, job satisfaction will improve.
Studies show that most laboratories acquired TLA to cope with increasing workload and to improve efficiency. For instance, Peck-Palmer 15 reported on the introduction of TLA for two large hospitals in an effort to standardize laboratory testing and improve patient care and observed that performance was increased 9 mo after conclusion of the project. A third of the surveyed laboratories had immediate plans to install an automated line. As technology becomes more accessible and associated costs decrease, more laboratories will be in a position to acquire such a facility. There will therefore be significant implications for the deployment of staff and work practices, and this requires significant input into the laboratory from diagnostic companies (e.g., training on instruments and on the associated soft and middle wares as well as advice on modification of work procedures). On the implications of installing the automated line, most of the respondents envisaged that work satisfaction among laboratory staff will remain the same or increase after installing the automated line ( Table 5 ). Peck-Palmer 15 reported that introduction of TLA improved performance 9 mo after conclusion of the project, but the first 6 mo had challenges for the operators as they became familiar with the technology, while at the same time, the hospital community became compliant with the new sample processing procedures.
There were variations in staff numbers in the core laboratories (from 5 to more than 40), and the variations are most likely associated with the magnitude of workloads ( Table 4 ). TLA does allow laboratories to cope with high workloads, and the researcher’s laboratory has experienced a growth rate of ~7% per year in workload for the past 7 y, with no additional staff in the core laboratory. In addition, work has been referred from other networked laboratories to the core laboratory. Reports show that in Australia, there has been an increase of about 8% per year in pathology testing, with no corresponding addition of resources. 12 Other studies have reported that the development of information technology and automation in laboratory medicine has allowed laboratory professionals to keep pace with the growth in workload. 31 Sasaki et al., 32 pioneers of total laboratory automation, reported that an automated laboratory system was developed in their laboratory as an alternative to accepting an intolerable shortage of technicians.
Small, medium, and large laboratories took part in the survey ( Table 4 ), and automation in the form of analyzers with embedded computers was present in all laboratories. These analyzers have programs to control the analytical process (e.g., pipetting, mixing and spectral measurements, calibration, quality control) and have bidirectional interfaces with the LIS. The medium and large laboratories may have workstations consisting of integrated instruments (i.e., chemistry and immunoassay), and large laboratories may have TLA. Large laboratories would have an extensive range of analytes, and the number of tests per sample reflects the range of tests available in the laboratory ( Table 4 ). The number of staff and their range of competencies can be restricted in the small laboratories with regard to any pathology discipline, but small laboratories may have multidisciplinary staff.
Peripheral/Branch Laboratories
Hematology analyzers were present in all peripheral laboratories, as shown in Table 6 . The peripheral laboratories may be classified as rapid response laboratories for immediate patient management but with a limited range of assays. Acute care analyses are provided in peripheral laboratories, and most of these analyses involve clinical biochemistry, hematology, and blood bank. Microbiology was provided in only 13 of the laboratories, indicating the large degree of centralization of microbiology and perhaps that most of the tests carried out in this discipline may not be required for immediate patient management.
Forty-one responses indicated a network of laboratories ranging from 1 to greater than 20 ( Table 7 ). Over the past decade, each Australian state has created health care networks and statewide pathology networks to increase efficiency and effectiveness. There were a few stand-alone laboratories, and these were most likely specialist laboratories in children and cancer hospitals or private laboratories. Networked laboratories are usually set up so that supervision comes from the central laboratory with instrumentation and choice of methods, quality control procedures, quality documentation, staff, and budget directed from the central laboratory.
STAT Laboratories
High-throughput main analyzers with small sample volume, bidirectional interface, autoverification, and access to pathology results from remote terminals have enabled fast turnaround of results, which has made the establishment of a STAT laboratory unnecessary in most hospitals. Today, many instruments have programs for random access and can perform STAT analyses. The laboratory LIS may also have the ability to designate a request as STAT and ensure priority processing, and such attributes have reduced the need for STAT laboratories.
Point-of-care instruments, which are now widely used, may also have decreased the need for the central laboratory to establish a STAT laboratory. Many hospitals have a pneumatic tube system that enables immediate dispatch of samples to the laboratory, further obviating the need for a STAT laboratory. Most STAT laboratories have instruments for clinical biochemistry and hematology ( Table 8 ). STAT laboratories on the same site as a main core laboratory imply that the instrumentation of the main laboratory is not suitable for the type of specimens handled by the STAT laboratory. The overhead for pathology with regard to staff and instrumentation in the STAT laboratory calls for additional staff with consequent training and competency assessment and integration with the core laboratory. The necessity for extra inventory and different instrumentation is an additional overhead to pathology. Endeavour is therefore made to choose instruments that are suitable for the range of samples received (i.e., small samples [pediatric, geriatric, difficult bleeds] and normal bleeds). Other specifications that must be taken into consideration are the range of analytes, upper and lower limit of detection, imprecision, interferents, and throughput.
Specialist Laboratories
From Table 9 it can be seen that the most common analytical principles employed are electrophoresis, GC/GC-MS/MS, high-performance liquid chromatography, and enzyme immunoassay (EIA). Molecular biology is used by seven laboratories, which appears to be a low number, but these analyzes may be placed in a separate department. Radioim- munoassay has low utilization because of many assays being reformulated to EIA or chemiluminescence or mass spectrometry. Atomic absorption has low utilization in clinical biochemistry laboratories because of available commercial colorimetric assays for common requests such as calcium, magnesium, lithium, copper, and zinc.
Conclusion
Technology will continue to evolve, and laboratories need to position themselves for such changes as demand for pathology services and consumer expectations will continue to rise. Reorganization of laboratories in the face of increased use of point-of-care testing, evolution of core laboratories, and introduction of TLA is therefore a necessity.
Footnotes
Acknowledgements
The authors acknowledge the help of Ms Sue Wind, Medical Library, Monash Medical Centre, Clayton, Victoria, Australia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
