Abstract
Cynomolgus macaques (Macaca fascicularis) are commonly used in safety assessment and as translational models for drug development. Recent supply chain pressures, exportation bans, and increased demand for drug safety assessment studies exacerbated by the COVID-19 pandemic have prompted the investigation of utilizing macaques of different geographic origin in preclinical toxicity studies. This study compares routine hematology, coagulation, and clinical chemistry endpoints of 3 distinct subpopulations of mainland Asia origin (Cambodia, China, and Vietnam) with Mauritius origin macaques compiling results of 3,225 animals from 123 regulatory toxicology studies conducted at North American and European Union contract research organization facilities between 2016 and 2019. Results were generally similar amongst the subpopulations compared in this study. Few notable differences in hematology test results and several minor differences in serum biochemistry and coagulation test results were identified when 3 distinct subpopulations of mainland Asia origin macaques were compared with Mauritius origin macaques. Our findings support the use of different origin macaques in drug development programs; however, emphasizes the importance of maintaining consistency in geographic origin of animals within a study.
Keywords
Introduction
Refinement and innovation in preclinical toxicity assessment of candidate new drugs has greatly reduced the use of animals in pharmaceutical safety assessment; however, preclinical animal studies are still necessary and required by regulatory agencies in the development of safe and effective therapeutics for human medicine. The cynomolgus macaque (M. fascicularis), also known as the crab-eating or long-tailed macaque, is a cercopithecine primate and is the most frequently used nonhuman primate (NHP) species in biopharmaceutical research. Macaques originate from a wide range of geographic locations including India, China, Southeast Asia, and the island of Mauritius located in the Indian Ocean just east of the African continent. 1 Increased demand for cynomolgus macaques for use in biomedical research in North America and Europe has been exacerbated by the increased development of large molecule therapeutics (NHPs are often the only relevant species for assessing these therapeutics) and the most recent increasing need for resources in emerging disease research (eg, SARS-CoV-2 studies). Combined with NHP exportation bans, this increased demand has led to a global shortage of Chinese origin NHPs for preclinical toxicity studies and has impacted the breeding/sourcing practices for NHPs destined for biomedical research. 2 Given this recognized limited availability of mainland Asian origin NHPs and pressures on the supply chain, it is imperative to consider and investigate the utilization of NHPs from different geographical origins and their potential interchangeability, or lack thereof, in pharmaceutical safety assessment programs. Recognition and characterization of origin-related differences can lessen or even eliminate the possibility of erroneous interpretation of preclinical study data in pharmaceutical safety assessments.
Mainland Asian macaques including Cambodian, Chinese, and Vietnamese subpopulations originated in Southeast Asia from common ancestors and share a similar genetic background, which supports using these subpopulations interchangeably for biomedical research. 3 In contrast to Asian macaques, Mauritian macaques are a population of NHP that have been geographically isolated for several centuries and based on mitochondrial DNA analysis, have expanded from a small number of founder animals originating from Indonesia. 4 Genetic diversity between Asian and Mauritius macaques has been observed and described in the literature.3,5,6 Mauritius macaques have a more homogenous genetic pool than mainland Asia origin macaques, are devoid of herpes B virus, respond differently to immune system challenges, and have lower incidences of background pathology based on previously published literature1,3,5-8 although a recent study 9 reports these lower incidences depend on the tissues and/or organs sampled with a higher incidence of inflammatory infiltrates identified in some tissues when Mauritius origin macaques were compared with other origin macaques.
Dedicated breeding colonies in China, the United States, and/or the European Union have led to predictable decreases in genetic diversity within each of the individual colonies, which mimics the decreases in genetic diversity observed in the naturally sequestered Mauritian cynomolgus macaques.1,3,10 Cynomolgus macaques are now purpose-bred in a limited number of breeding centers in different geographical locations across the globe, but most European countries depend on NHP importation due to fewer breeding facilities in Europe compared with other countries.
Evaluation of clinical pathology data is an integral part of pharmaceutical candidate safety assessments and a robust database for standard clinical pathology endpoints is available for the cynomolgus macaque, including some subpopulations. Nevertheless, when interpreting clinical pathology data, it is imperative to consider the variability observed in cynomolgus macaques, not only as individuals but also mindful of the source and geographic background.11-14 Limited information on the potential differences in routinely evaluated clinical pathology endpoints (ie, standard hematology, coagulation, and clinical chemistry parameters) of Mauritian macaques compared with unique Asian origin subpopulations (ie, Cambodia, China, and Vietnam) is available in the literature and many of the previous studies were comprised of data sets containing low animal numbers.5,15,16 This study contributes to the literature with a large scale, well-powered, review of standard clinical pathology endpoints of cynomolgus macaques conducted at 3 contract research organization (CRO) facilities; 2 in North America and 1 in Europe. Comparisons were made to investigate and describe differences of toxicological relevance, if any, between Mauritius macaques and other Asian origin macaques, and to highlight the influence geographic isolation may have on the Mauritius population of macaques. A secondary objective was to determine if any differences would impact animal sourcing for a given study or drug development program.
Material and Methods
Animals
Cynomolgus macaques of mainland Asia (ie, Cambodia, China, and Vietnam), or Mauritius origin were procured from AAALAC-International accredited sources/suppliers and housed at one of 3 geographically distinct AAALAC-International accredited CRO facilities in North America (Madison, WI, USA [MSN], Greenfield, IN, USA [GRN]) or the European Union (Münster, DE [MUE]). Animals underwent required physical examinations, pathogen screenings, and at a minimum were serologically tested and confirmed negative for simian immunodeficiency virus (SIV), Cercopithecine herpesvirus 1 (B virus), simian retroviruses type D, rabies, simian T-cell leukemia virus (STLV), and filoviruses. Additional testing, including tuberculin and bacterial (eg, Shigella, Campylobacter, Yersinia, and Salmonella) and parasitological (eg, malaria smears) screening was performed. Upon receipt to the facility, animals underwent required quarantine and a minimum of 3 to 6 weeks acclimation per local facility standard operating procedures. Animals were pair or group-housed in climate-controlled rooms (a minimum of 8 air changes/hour, 19-26°C, 30%-70% relative humidity, 12-hour artificial light/dark cycles), received tap water ad libitum, and were fed a standardized diet across facilities once or twice daily (Certified lab diet for primates [LabDiet 5048, PMI Nutrition International, Inc.] supplemented by fresh fruit and vegetables). Animals were given a variety of room, cage, and dietary enrichment. Studies were performed in compliance with United States federal regulations and recommendations and institutional ethical committees or in accordance with the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe); see Animal Use Statement for more detail.
Animals were fasted overnight for a minimum of 6 hours prior to nonsedated blood collection from the femoral (North America [NA] facilities) or cephalic or saphenous (European Union [EU] facility) vein. Hematology, coagulation, and serum biochemistry data used in this comparative assessment were obtained from scheduled intervals for blood collection prior to study initiation (naïve animals). Animal origin, study location, regulatory compliance, and animal numbers are summarized in Table 1.
Distribution of 123 Studies (3,225 Animals) Surveyed to Compile Data Used in This Study.

Abbreviations: GLP, Good Laboratory Practice; Greenfield, IN, USA (GRN); Madison, WI, USA (MSN); Muenster, DE, EU (MUE).
Clinical Pathology
Clinical pathology data from 123 preclinical toxicology studies (assembling data from 3,225 animals) conducted predominantly under Good Laboratory Practice (GLP) regulations from the 3 previously described CRO facilities between 2016 and 2019 were tabulated. Clinical pathology parameters assessed for this comparison are listed in Table 2.
Clinical Pathology Parameters Assessed.
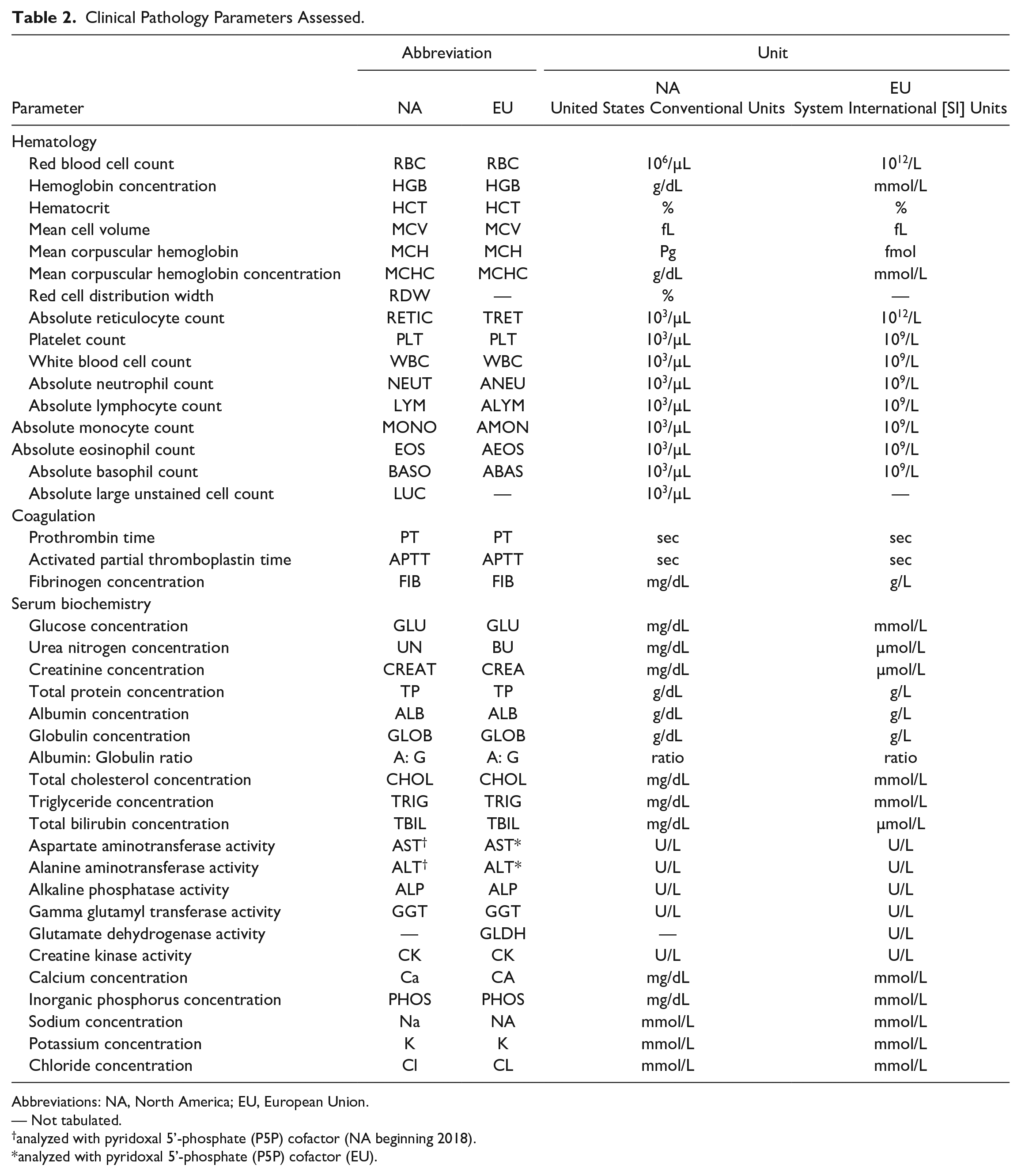
Abbreviations: NA, North America; EU, European Union.
— Not tabulated.
analyzed with pyridoxal 5’-phosphate (P5P) cofactor (NA beginning 2018).
analyzed with pyridoxal 5’-phosphate (P5P) cofactor (EU).
Blood samples were collected into EDTA tubes for hematology tests and analyzed using an Advia 2120i analyzer (Siemens, USA)—(NA) or a Sysmex 2000 analyzer (Sysmex Europe, DE)—(EU). Samples for hematology tests were analyzed on the day of collection. Blood samples were collected into 3.2% sodium citrated tubes for coagulation tests and plasma was harvested following centrifugation (3000 RPM for 10 minutes at room temperature) within 75 minutes of collection. Plasma samples for coagulation tests were analyzed using a Destiny Amax analyzer (Tcoag, Wicklow, Ireland) or ACL TOP 500 analyzer (Instrumentation Laboratory, MA, USA)—(NA) or using a Destiny Max GUI analyzer (Tcoag, Wicklow, IR)—(EU). Plasma samples not analyzed on the day of collection were stored at −20°C for up to 13 days pending analysis. Blood samples were collected into tubes without anticoagulant for serum biochemistry tests. These samples were allowed to clot and serum was harvested following centrifugation (3000 RPM for 10 minutes at room temperature) within 4 hours of collection and analyzed using a Roche Modular Analytics P analyzer (Roche Diagnostics, IN, USA), Cobas 8000 analyzer (Roche Diagnostics, IN, USA)—(NA) or a Konelab Prime analyzer (Thermo Scientific, MA, USA)—(EU). Serum samples not analyzed on the day of collection were stored at −60 to −80°C for up to 7 days pending analysis. Quality Control (QC) was performed with appropriate levels of QC following specific manufacturer’s instructions at a minimum of once daily prior to the first analytical run of each day. Quality control was reviewed to ensure results were within established ranges and results were documented. In addition, prior to analysis, samples were assessed for visible clots, hemolysis, lipemia, and/or icterus.
Statistical Analysis
This dataset consisting of data from Mauritian, Cambodian, Chinese, and Vietnamese origin macaques aged between 2 to 5 years was subjected to statistical evaluation. Due to differences in the clinical pathology instrumentation and methodology between the EU and NA facilities, data availability, and differences in reporting units (NA = United States Conventional Units and EU = System International [SI] Units), comparisons between Mauritian and Vietnamese macaques were carried out using data obtained from the EU facility and comparisons between Mauritian and Cambodian or Chinese macaques were carried out using data obtained from the NA facilities. Separate statistical evaluations were performed for each parameter and sex. To avoid any potentially erroneous or extreme data points from skewing statistical conclusions, outliers were removed using the Tukey box plot method. 17 Only parameters with at least 40 individual values were subjected to statistical evaluation. Differences between origins were evaluated using Analysis of Variance and 2-sample t-tests.18,19 For the purpose of this current investigation, data from the 2 North American laboratories (MSN and GRN) were combined (including coagulation data for the Destiny Amax and ACL TOP 500 analyzers) although direct correlations of the instruments were not performed between the 2 laboratories. The combined data was reviewed and the results were considered similar for the purpose of this investigation based on notable overlap of individual animal results. Differences were felt to have minimal impact on the comparisons and statistical analysis of the overall clinical pathology data. The use of data from a single study analyzed at the GRN location combined with the bulk of the results obtained from the MSN laboratory was felt to have minimal impact on the overall data. Statistical results are reported in Tables 3–6 and describe sample size (“N”), arithmetic group means, observed range of minimum and maximum values, and 95% reference intervals (group mean +/- 1.96 standard deviations).
North America Hematology and Coagulation Results.
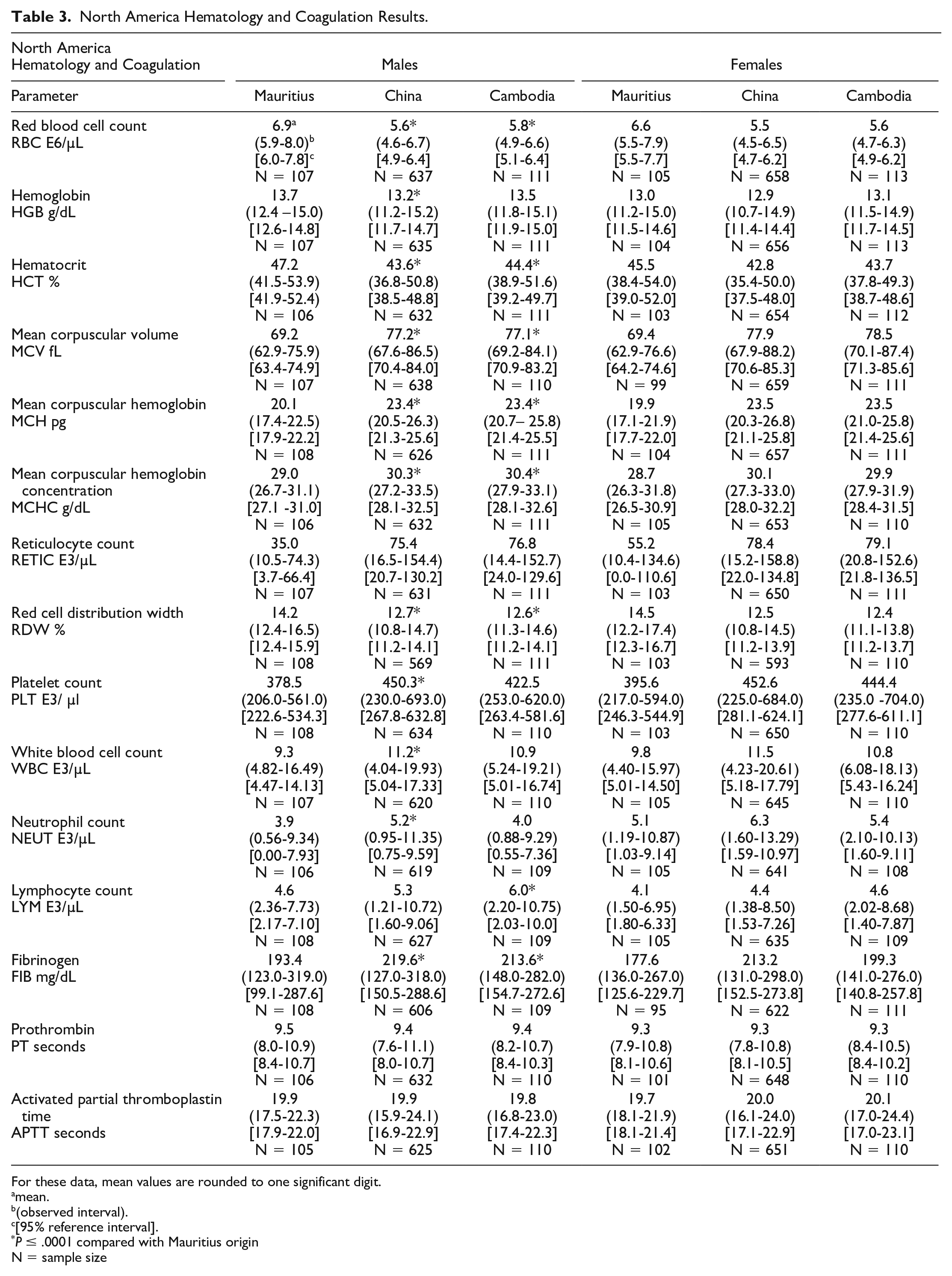
For these data, mean values are rounded to one significant digit.
mean.
(observed interval).
[95% reference interval].
P ≤ .0001 compared with Mauritius origin N = sample size
European Union Hematology and Coagulation Results.
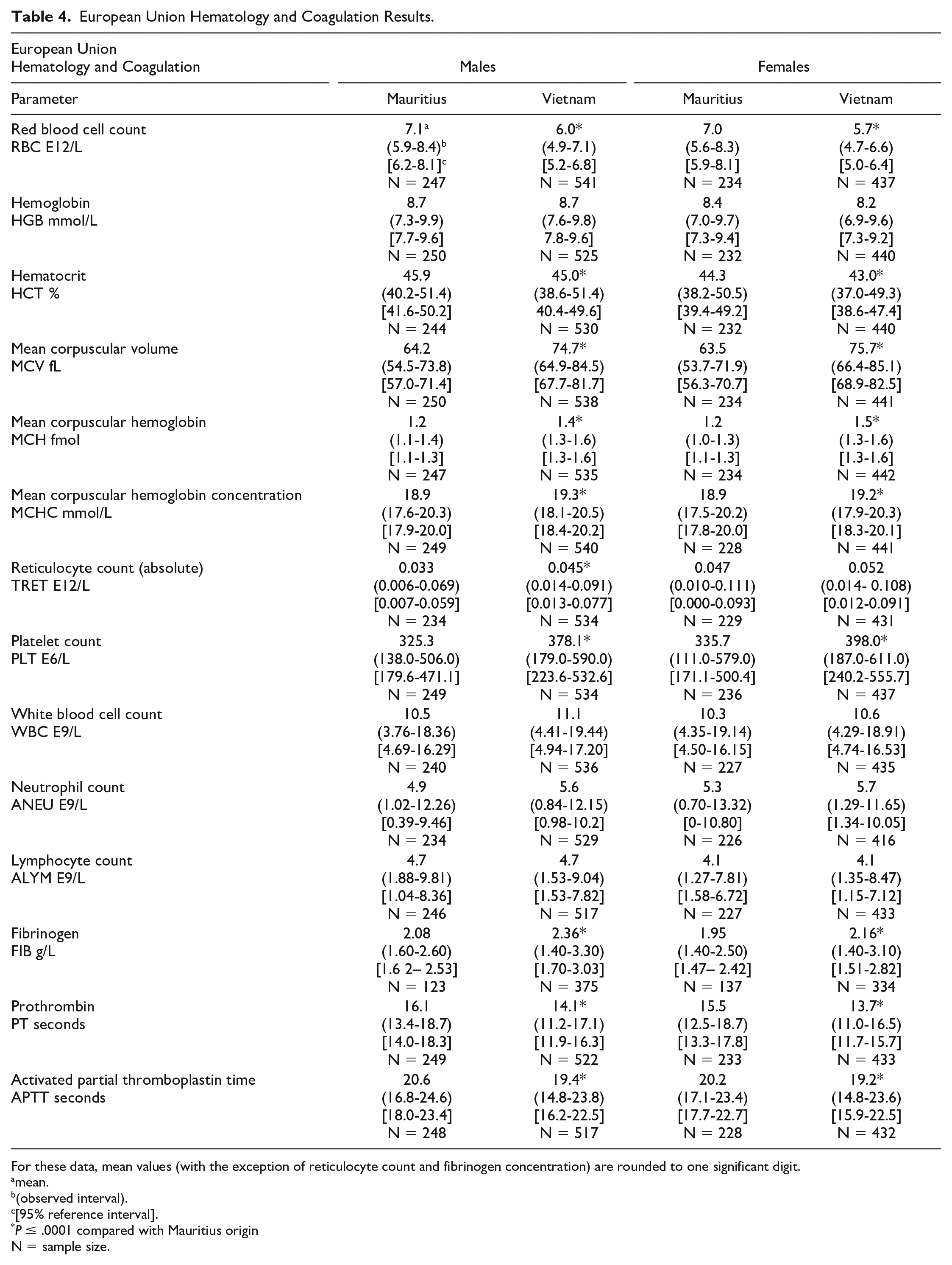
For these data, mean values (with the exception of reticulocyte count and fibrinogen concentration) are rounded to one significant digit.
mean.
(observed interval).
[95% reference interval].
P ≤ .0001 compared with Mauritius origin N = sample size.
North America Serum Biochemistry Results.
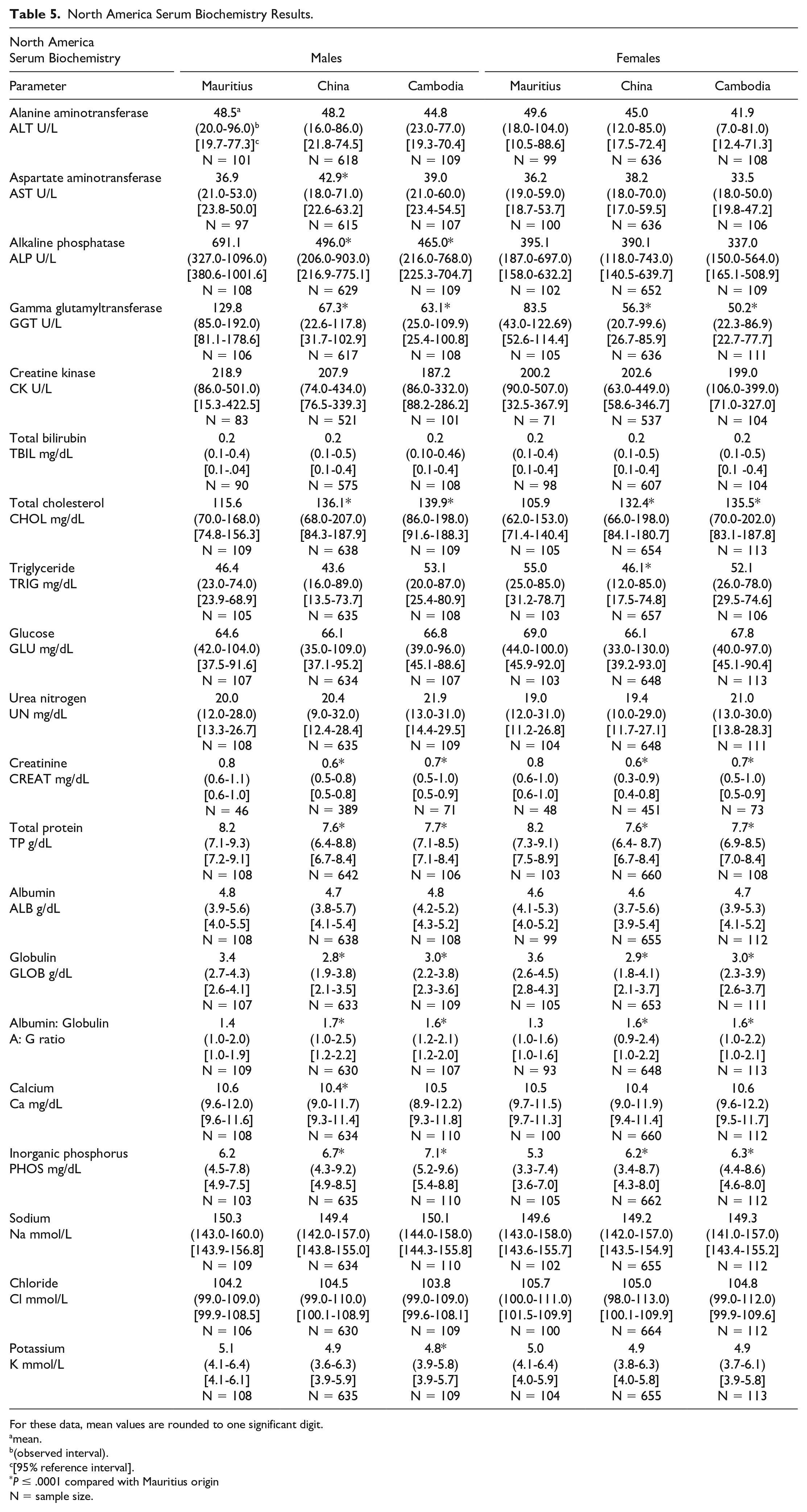
For these data, mean values are rounded to one significant digit.
mean.
(observed interval).
[95% reference interval].
P ≤ .0001 compared with Mauritius origin N = sample size.
European Union Serum Biochemistry Results.
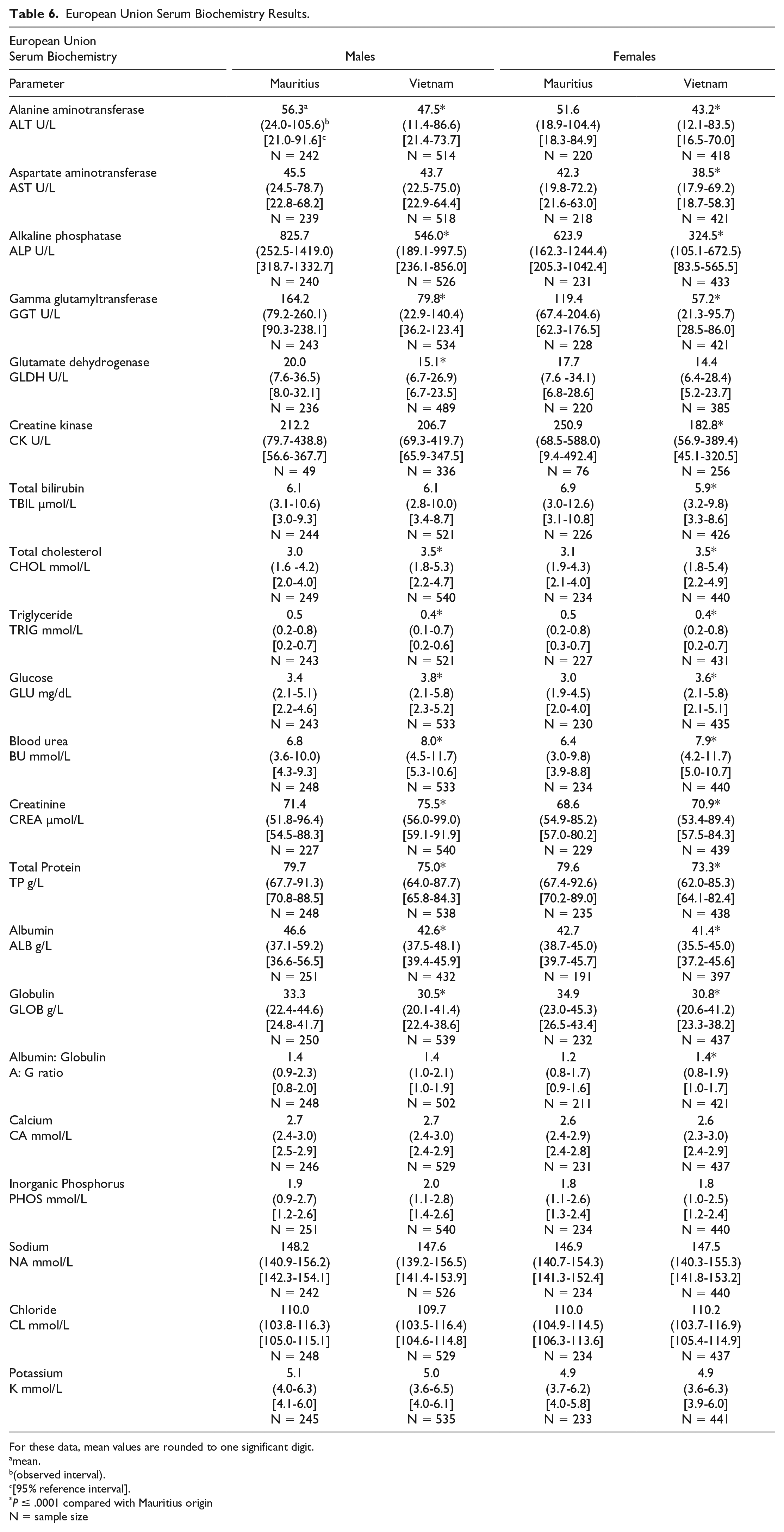
For these data, mean values are rounded to one significant digit.
mean.
(observed interval).
[95% reference interval].
P ≤ .0001 compared with Mauritius originN = sample size
Qualitative (eg, minimal, mild, moderate, marked) and quantitative (% difference) descriptors are used in the Results section to give context to the magnitude of change between group means of Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritius origin macaques. Percent difference was calculated by dividing the difference between Cambodian, Chinese, or Vietnamese origin group mean value and Mauritius origin group mean value by the Mauritius origin group mean value and then multiplying that by 100. Data are presented in box plots for select endpoints (Figures 1-4). Box plots partition the data distribution into quartiles. The box indicates the positions of the upper and lower quartiles; the interior of the box indicates the inner quartile range, which is the area between the upper and lower quartiles and consists of 50% of the distribution. Vertical lines extending on either end of the box (referred to as “whiskers”) indicate the distribution, either minimum or maximum values in the dataset. Outliers are represented individually by symbols above or below the whiskers. The crossbar within the box indicates the median of the dataset. The mean value is denoted with a symbol within the box.
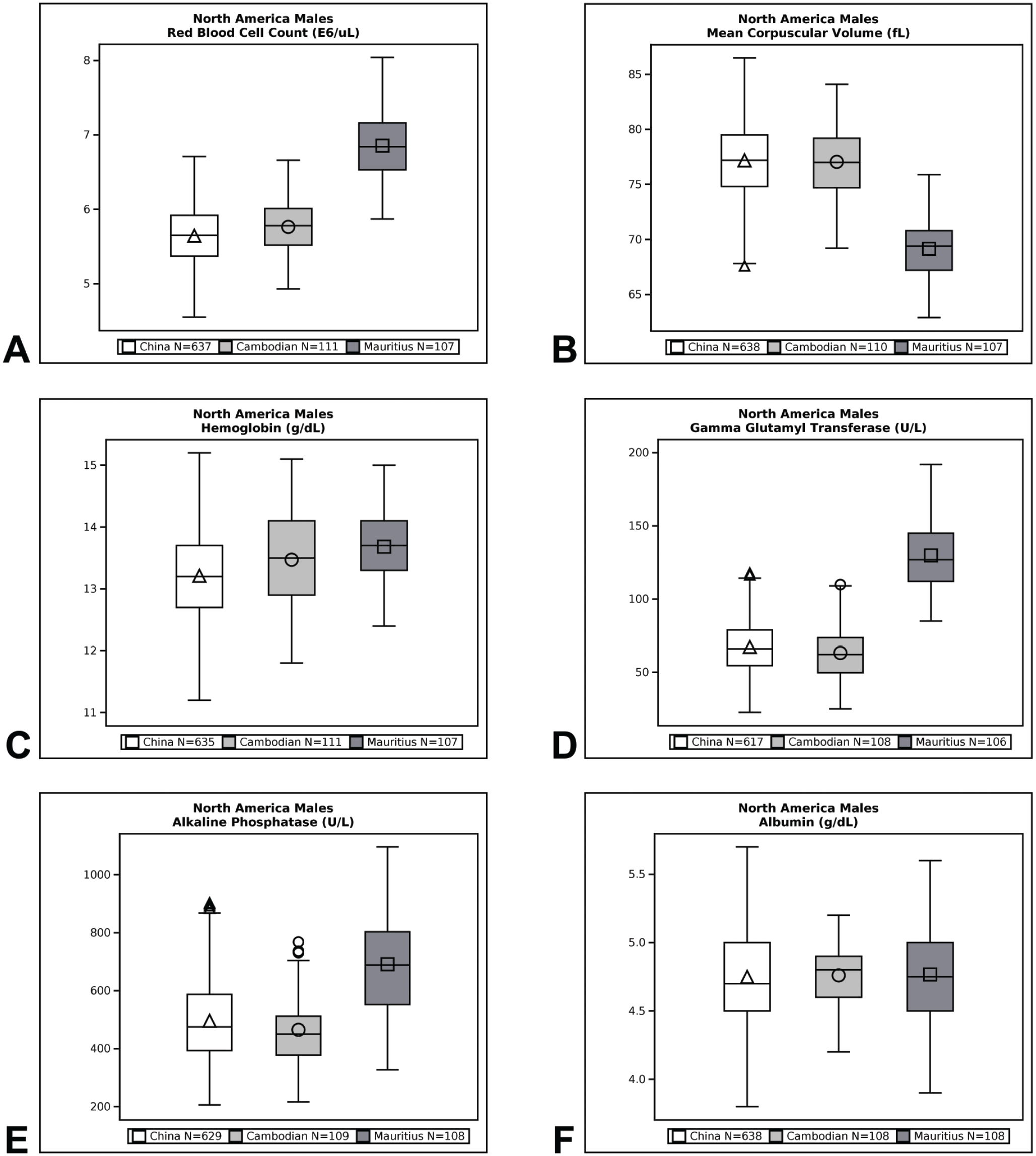
Box plot representations of select endpoints (North America; males).
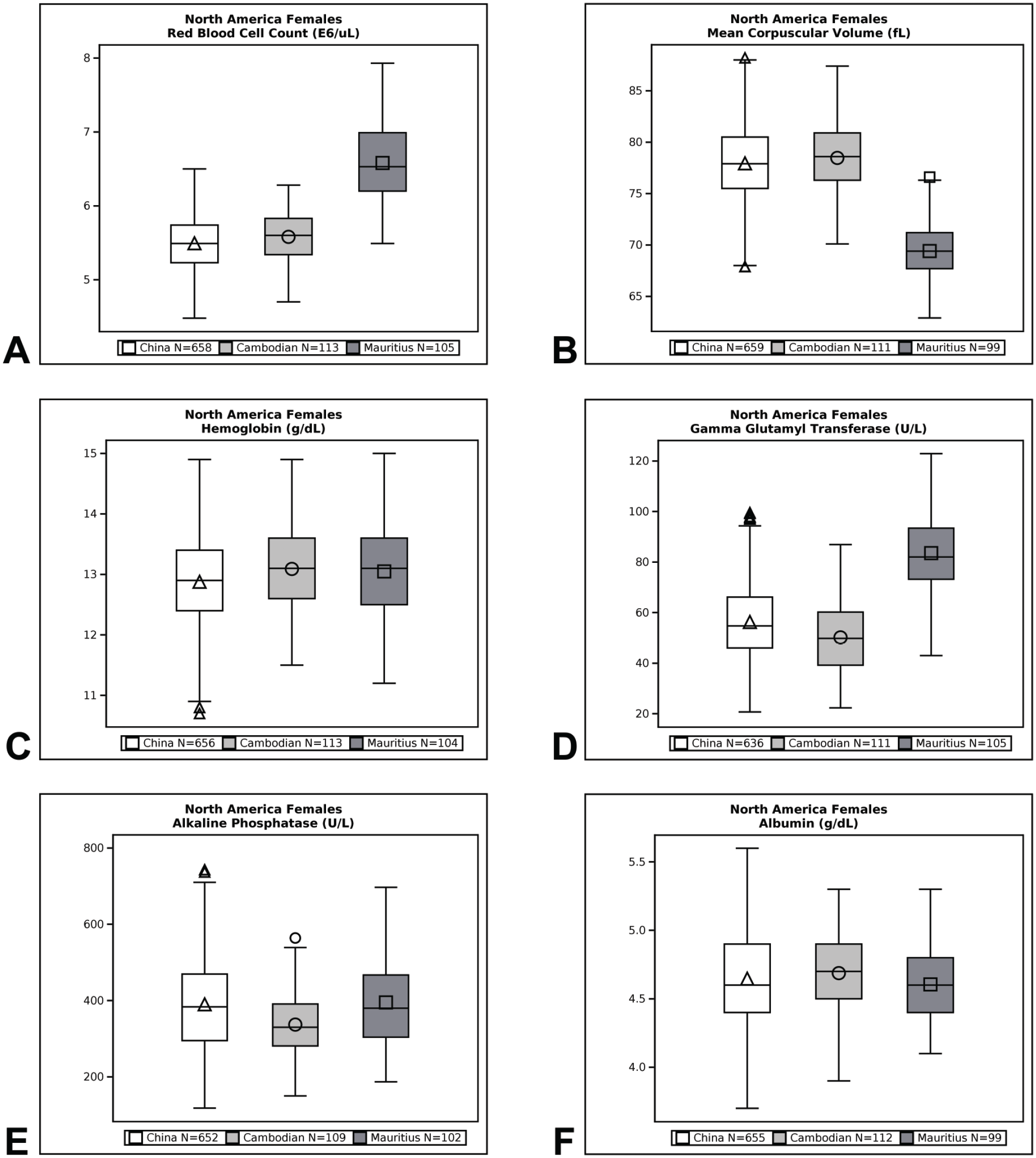
Box plot representations of select endpoints (North America; females).
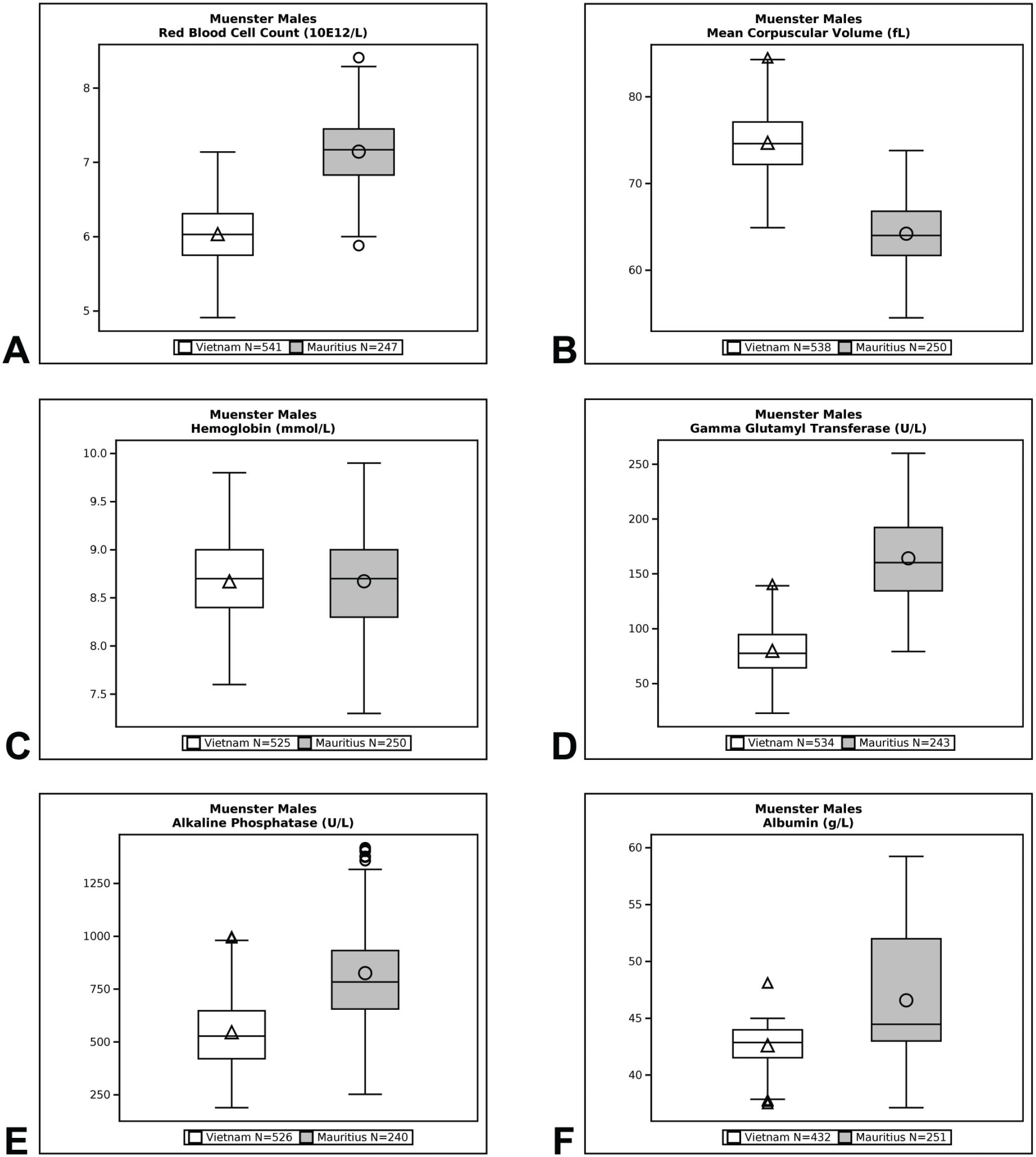
Box plot representations of select endpoints (European Union [Münster/Muenster]; males).
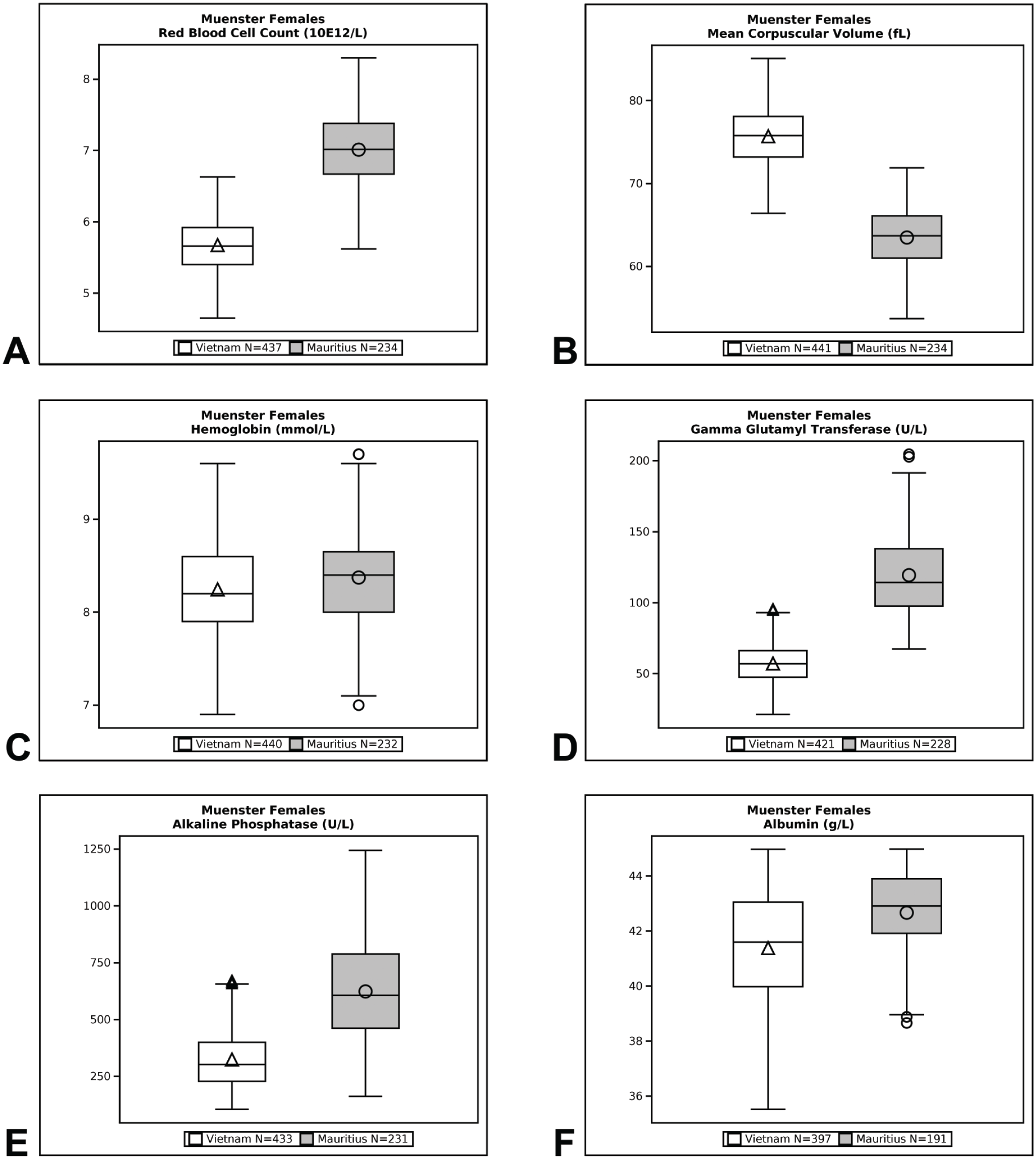
Box plot representations of select endpoints (European Union [Münster/Muenster]; females).
Results
Hematology and Coagulation
Differences in hematology values were identified between Mauritius origin macaques and those from Cambodian, Chinese, and Vietnamese origin (Tables 3 and 4, Figures 1–4). One of the most notable and well documented differences in hematology parameters is that Mauritius origin macaques have smaller red blood cell size (lower mean corpuscular volume [MCV]) and higher red blood cell (RBC) count compared with mainland Asian macaques.5,20 Similar differences in the RBC population were demonstrated in our data showing minimally to mildly lower RBC count (up to −19%) and red cell distribution width (RDW)—(up to −14%) and minimally to mildly higher absolute reticulocyte count (up to +119%), MCV (up to +19%), mean corpuscular hemoglobin (MCH)—(up to +25%), and mean corpuscular hemoglobin concentration (MCHC)—(up to +5%), when macaques of Cambodian, Chinese, and Vietnamese origin were compared with Mauritius origin macaques. These differences in red blood cell indices were consistent and similar in NA and EU data, but as RDW was not reported in the EU data, an RDW comparison of Vietnamese with Mauritian origin was not made. Inter-animal variability was noted in total and differential white blood cell (leukocyte) counts in animals of the origins compared, but no discernible/remarkable group differences were identified due to overlapping individual results amongst the groups. The only notable difference in standard coagulation test results was minimally to mildly shorter prothrombin time (PT)—(up to 2 seconds) in Vietnam origin macaques compared with Mauritius origin macaques.
Serum Biochemistry
Serum biochemistry differences were noted between macaques of Mauritius origin compared with the other origins (Tables 5 and 6, Figures 1–4). Small differences in hepatocellular/hepatobiliary enzyme activities were noted. Vietnam origin macaques were noted with minimally to mildly lower glutamate dehydrogenase (GLDH)—(up to −25%) activity compared with Mauritius origin macaques; minimally to mildly lower alkaline phosphatase (ALP)—(up to −48%) activity was noted in Cambodian (males only), Chinese (males only), and Vietnamese (both sexes) origin macaques compared with Mauritius origin macaques; and minimally to mildly lower gamma glutamyl transferase (GGT)—(up to −51%) activity was noted in Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritius origin macaques. Serum total protein (up to −8%) and globulin (up to −19%) concentrations were minimally to mildly lower in Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritius origin macaques. Minimally to mildly lower serum albumin (up to -9%) concentration was noted in Vietnamese origin macaques compared with Mauritius origin macaques. Serum urea nitrogen (up to +23%) and creatinine (up to +6%) concentrations were minimally to mildly higher in Vietnam origin macaques compared with Mauritius origin macaques. Minimally to mildly lower creatinine (up to −25%) concentration was also noted in macaques of Cambodian or Chinese origin when compared with Mauritian macaques. Total cholesterol (up to +28%) concentration was mildly higher in Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritian macaques, whereas triglyceride (up to −20%) concentration was minimally to mildly lower in Vietnamese origin macaques when compared with Mauritian macaques.
Discussion
Cynomolgus macaques are the most widely used nonhuman primate species in preclinical pharmaceutical safety assessment studies and are well suited and established as translational models for drug development. 21 Given the increased demand for these animals in the development of safe and effective therapeutics for human medicine coupled with a global shortage of Chinese origin NHPs exacerbated by exportation bans, the supply chain has been impacted and macaques of non-mainland Asia origin have been considered for use, and macaques of Mauritius, Vietnamese, and Cambodian origin are being used in increasing numbers in preclinical drug development programs. The high level of genetic diversity between subpopulations of different geographic origin, such as between cynomolgus macaques from Mauritius and those from mainland Asia, is an important consideration for any scientific study with cynomolgus macaques.21-23 Genetic divergence also affects physiological traits and knowing the geographic origin and genetic background of NHPs before beginning any study is a well-recognized advantage. 21 This current investigation of clinical pathology data supports the published literature and in-press work and helps to better characterize differences, or lack thereof, between subpopulations of cynomolgus macaques.
Differences between cynomolgus macaque origins were identified and described in our data comparison of clinical pathology test results. These differences were of small magnitude and considered minor which supports the inclusion of different origin of macaques for use in preclinical toxicity studies. Distinctions in hematology parameters were most notable, whereas smaller differences were identified in coagulation and serum biochemistry parameters. Differences in red blood cell parameters in Cambodian, Chinese, and/or Vietnamese origin macaques compared with Mauritius origin macaques consisted of minimally to mildly lower RBC counts and RDW (NA only), and minimally to mildly higher absolute reticulocyte counts, MCV, MCH, and MCHC which were consistent with the literature.5,16 These origin-specific differences reflect the genetic diversity of different origin subpopulations of cynomolgus macaques. Despite lower numbers of red blood cells of larger size in Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritius origin macaques (indicated by lower RBC count and higher MCV noted in Cambodian, Chinese, and Vietnamese origin), red cell mass was considered similar amongst the 4 origins assessed because hemoglobin concentration and hematocrit remained generally comparable amongst the subpopulations which is also consistent with the literature. 16
Inter-animal variability, which occasionally resulted in statistically significant differences between groups, was noted in total and differential white blood cell counts in animals of all origins; however, no biologically important group differences were identified in macaques of Cambodia, China, or Vietnam origin when compared with those originating from Mauritius, which is consistent with findings in the published literature. 5 The shorter PT in macaques of Vietnam origin reached statistical significance when compared with those of Mauritius origin, although a similar difference was not identified when Chinese or Cambodian origin were compared with Mauritian origin animals. Minimally to mildly shorter PT has been previously described in Mauritius origin compared with Asian origin macaques but that study only compared mixed Asian (ie, animals of Cambodian, Vietnamese, and Indonesian origin), Cambodian, and Mauritian whereas our current study clearly defined animal origin. 5 In our data, no impactful difference in activated partial thromboplastin time (APTT) or fibrinogen concentration was identified in the 3 Asian origin subpopulation origins when compared with Mauritius origin macaques.
Minor differences were identified in serum chemistry results from macaques of Cambodia, China, or Vietnam origin when compared with Mauritius origin macaques. When compared with Mauritius origin macaques, small differences (minimal to mild in magnitude) were identified in hepatocellular/hepatobiliary enzyme activities which consisted of lower GLDH activity in Vietnam origin macaques; lower ALP activity in Cambodia and China (males only) and Vietnam (both sexes) origin macaques; and lower GGT activity in Cambodian, Chinese, and Vietnamese origin animals. Additional clinical chemistry differences were minimal to mild in magnitude and somewhat inconsistent between Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritius origin macaques. Serum total protein and globulin concentrations were lower in Cambodian, Chinese, and Vietnamese origin macaques whereas serum albumin concentration was lower only in Vietnamese origin macaques. Serum urea nitrogen and creatinine concentrations were higher in Vietnam origin macaques whereas lower creatinine concentration was noted in macaques of Cambodian or Chinese origin when compared with Mauritian macaques. Total cholesterol concentration was higher in Cambodian, Chinese, and Vietnamese origin macaques compared with Mauritian macaques while triglyceride concentration was lower in Vietnamese origin macaques when compared with Mauritian macaques. Although not an objective of this current study, appreciable differences were not noted between macaques of different mainland Asia origin in the NA dataset (China compared with Cambodia), which correlated with the literature.11,24,25
Some of the differences between macaques of different origins previously reported in the literature were attributed to age, animal condition, and/or background findings.5,16,24,25 In our current study, many variables were standardized (eg, age, sex, husbandry, and health status) to prevent such biases in the identification of origin-related differences. For example, higher ALP activity has been reported associated with younger aged animals, and therefore animals under 2 years old were excluded from the current study to prevent the influence of young age on clinical pathology results. 5 A potential source of bias or variability in our current investigations was recognized becasue the data from the MSN and GRN lab were combined to make up the NA data set despite the lack of correlation of instruments between the 2 sites. Any impact was considered negligible to minimal given data from a single study analyzed at the GRN location was combined with data analyzed at the MSN laboratory (MSN data made up most of the NA data). In addition, the effect of animal source/supplier has been identified in other research species and should also be considered as a variable in assessing clinical pathology data of NHPs in biomedical research. 20 Future work should be considered to identify any effect of source/supplier of NHPs of different origins and investigate the impact of genetics and in-breeding within source/supplier colonies on clinical pathology parameters assessed for NHP used in preclinical studies.
Numerical differences were present and recognized in our data. These numerical differences occasionally reached statistical significance, but were considered not toxicologically relevant because the differences were small in magnitude, overlap was appreciated in the individual results and observed ranges, and such differences are not expected to impact the interpretation of preclinical toxicology data in the context of a well-designed study (ie, appropriate predose/baseline data and control group comparators).26,27 Statistical significance can also be influenced by seemingly small differences in the assessment of clinical pathology data given the often tightly regulated parameters (eg, electrolytes and minerals) and/or parameters with narrow “physiologic ranges” (eg, total bilirubin, creatinine), large sample size “n”, and results easily impacted by preanalytical/analytical variability (eg, coagulation times or minor leukocyte populations impacted by sample quality or anticoagulant ratio). Statistical analysis of toxicological clinical pathology data should not be solely relied upon, or used in isolation, to interpret differences in data sets. Interpretations should be based on an understanding of each analyte/parameter employing a weight of evidence approach rather than to substitute statistical analysis for the scientific process of appropriate data interpretation.26,27
Based on this study and published literature characterizing differences in clinical pathology data, macaques of different origin (eg, Cambodian, Chinese, Vietnamese, and Mauritian) can be used to support drug development programs.5,24,25 While Mauritius origin macaques are genetically and physiologically different from mainland Asia origin macaques, the herein described differences in clinical pathology endpoints should not preclude their use in preclinical pharmaceutical safety programs. Given this, differences in clinical pathology data based on macaque origin should be considered when interpreting preclinical safety assessment toxicology data to avoid erroneous clinical pathology data interpretation. Same origin macaques should be used for individual studies, and ideally (but not always possible) across a drug development program, to minimize impacts on clinical pathology data. Furthermore, CRO facilities should maintain origin specific (separate macaque subpopulation) clinical pathology reference intervals, even though controls and predose/baseline data are best used, and preferred, for the interpretation of preclincial study data.1,26,27 Finally, the importance of well-designed preclinical toxicity studies with predose/baseline clinical pathology assessments and concurrent controls for proper interpretation of study data cannot be overemphasized.16,26,27
Animal Use Statement
Studies performed in US-based facilities were conducted at AAALAC International-accredited contract research organization (CRO) facilities, and in compliance with United States federal regulations and recommendations detailed in The Guide for the Care and Use of Laboratory Animals (National Research Council 1996), and in accordance with Labcorp (formerly Covance) policies on the care, welfare, and treatment of laboratory animals. In addition, all procedures involving the animals were reviewed and approved by the respective facilities Institutional Animal Care and Use Committees (IACUC) prior to dose administration. Studies performed in the EU were conducted in accordance with the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe), in addition to institutional ethical committees.
Footnotes
Acknowledgements
The authors would like to recognize and thank the following for their contribution to this manuscript. Laura Boone and Niraj Tripathi for their critical scientific review; Scott Templeton, Andrew Newell, and Kelly Ashcroft-Hawley for data management; and Gerhard Weinbauer for his passionate approach to NHP data projects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
