Abstract
Background
Knee osteoarthritis (KOA) is the most common degenerative joint disease. Adipose-derived stromal vascular fraction cell therapy slows the progression of knee osteoarthritis and prevents hyaline cartilage degeneration without serious side effects. This study aims to present retrospectively the effectiveness of stromal vascular fraction cells isolated from adipose tissue by the non-enzymatic method applied to 55 osteoarthritis patients of different age groups and Kellgren-Lawrence grades on the recovery of the disease.
Material and Methods
Fifty-five patients with knee osteoarthritis, treated with stromal vascular fraction cells at the International Medical Centre (Azerbaijan) between 2020 and 2021, were included in the study to be evaluated retrospectively. Patients aged 32–67 years, Grades I–IV according to Kellgren-Lawrence classification, were included in this study. Pain and functional limitations were assessed using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) questionnaires before treatment, at the 5th month, and 12th month after stromal vascular fraction injection.
Results
At the fifth and 12th months post-treatment, it was observed that the WOMAC scores were significantly improved compared with pre-treatment scores. It was also observed that stromal vascular fraction therapy was effective for Kellgren-Lawrence Grades I and II at 12 months. The most significant decrease in the WOMAC score after stromal vascular fraction treatment was observed in patients aged 45–60 years, and it was also found that there was no meaningful relationship between stromal vascular fraction efficiency and gender.
Conclusion
According to our results, non-enzymatically stromal vascular fraction treatment is more effective for longer in osteoarthritis patients with early age and low Kellgren-Lawrence grades (I and II). Moreover, our finding is that the stromal vascular fraction cells could be used safely in osteoarthritis treatments and significantly benefit patients’ quality of life.
Introduction
Knee osteoarthritis (KOA) is the most common degenerative joint disease characterized by articular cartilage degeneration, and synovial tissue inflammation that occurs with aging and impairs quality of life.1,2 The latest evidence has been reported that osteoarthritis (OA) is a multifactorial disease induced by extracellular matrix (ECM) degradation as a result of cellular senescence through DNA damage caused by synovial inflammation and oxidative stress.3–7
Various pharmacological and non-pharmacological approaches are applied to treat this pathology. The most commonly used treatment methods are corticosteroid or non-steroid anti-inflammatory drugs, physiotherapy, and hyaluronic acid injection. These treatments could reduce pain and improve joint function, but they are poor at regenerating damaged articular cartilage.7–9
Autologous cellular therapy (ACT) is the use of an individual's cells or tissue as a treatment after processing them outside the body. 10 Mesenchymal stem cells (MSCs), platelet-rich plasma (PRP), and adipose tissue-derived stromal vascular fraction (AD–SVF) cells are types of ACTs that have shown promise in modulating joint inflammation and regenerating articular cartilage.11,12 The stromal vascular fraction (SVF) cell population contains preadipocytes, mesenchymal stem cells, endothelial progenitor cells, T and B cells, and mast cells. These cells slow the progression of KOA and prevent hyaline cartilage degeneration without serious side effects through the immunomodulatory, anti-inflammatory, paracrine, and hematopoietic effects.13–17
SVF cells can be obtained from lipoaspirate samples without cell separation or culturing conditions. 18 There are two, enzymatic and non-enzymatic methods available for adipose tissue-derived SVF cell isolation. Enzymatic isolation is based on digesting lipoaspirate with collagenase after washing. Then centrifuged to separate into three main layers, the adipose tissue layer, the aqueous layer, and the pellet. The SVF cells get separated from the pellet and enzyme. Alternative to enzymatic digestion, non-enzymatic methods focus on using shear force to break down the adipose tissue, then proceed by washing to eliminate fat and blood cell residues.18–21 The enzymatic method is a time-consuming procedure, uses animal-derived enzyme products, and is much more expensive while the non-enzymatic method provides practical, instantaneous, sterile, and minimal manipulation for cell isolation.18,21,22 The potential of the pro-regenerative and anti-inflammatory effects of SVF treatment for osteoarthritis has been presented by an in vitro study where mechanically derived SVF stimulates chondrocyte proliferation while preserving its function. 23 Both methods have different advantages and disadvantages, but it has been reported that there are no significant differences in terms of cell viability, genetic structure, cell proliferation, and differentiation abilities.23–26 Nevertheless, there are more limited studies that show the effectiveness of SVF cells isolated by non-enzymatic methods compared to the enzymatic method.
In this study, the improvement levels of knee regeneration of KOA patients who were treated with non-enzymatically isolated SVF cells were presented retrospectively. The symptoms of the patient's knee discomfort and diagnosis were evaluated according to the WOMAC score of 55 individuals of different ages, genders, and Kellgren-Lawrence (KL) grades.
Methods
Study population
Fifty-five patients with KOA, treated with autologous SVF cells at the International Medical Centre (Baku, Azerbaijan) between 2020 and 2021, were included in the study to be evaluated retrospectively.
The OA grade of patients was evaluated by the Kellgren-Lawrence (KL) classification. Patients aged 32–67 years, Grades I–IV according to KL classification, and without a history of knee trauma were included in the study. Immunosuppressive patients, patients with autoimmune disease, malignancy, intra-articular infection, receiving chemotherapy, also patients who had surgery on the abdomen and knee joint in the last 6 months were excluded from the study.
The study was conducted according to the Helsinki Declaration. The patients were informed, and their consent was obtained.
Treatment
A non-enzymatic kit called Lipocell® was used (Figure 1). All the procedures were performed under general anesthesia. Firstly, Klein solution (0.1% lidocaine and 1 mg/mL epinephrine in 1000 mL NaCl 0.9% solution 27 ) was injected around the patients’ umbilicus both for better lipoaspirate uptake and to minimize post-operative bleeding or hematoma formation. Then, 200–250 mL lipoaspirate was obtained from the patients and SVF isolation was processed following the instructions of the kit used. As a result of this procedure, ∼ 15–20 mL of autologous SVF cells were obtained, depending on the fat quality of the patient (Figure 2).
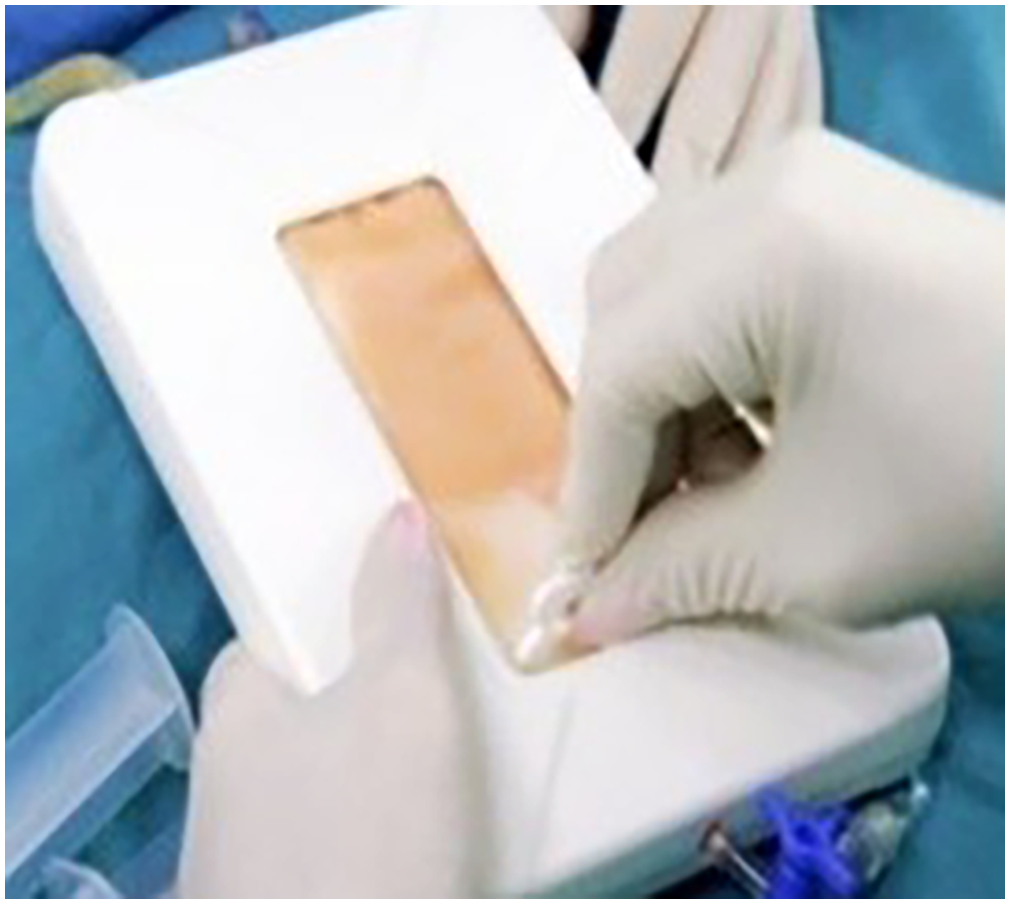
Non-enzymatic stromal vascular fraction (SVF) isolation kits were used in the study. Lipocell.
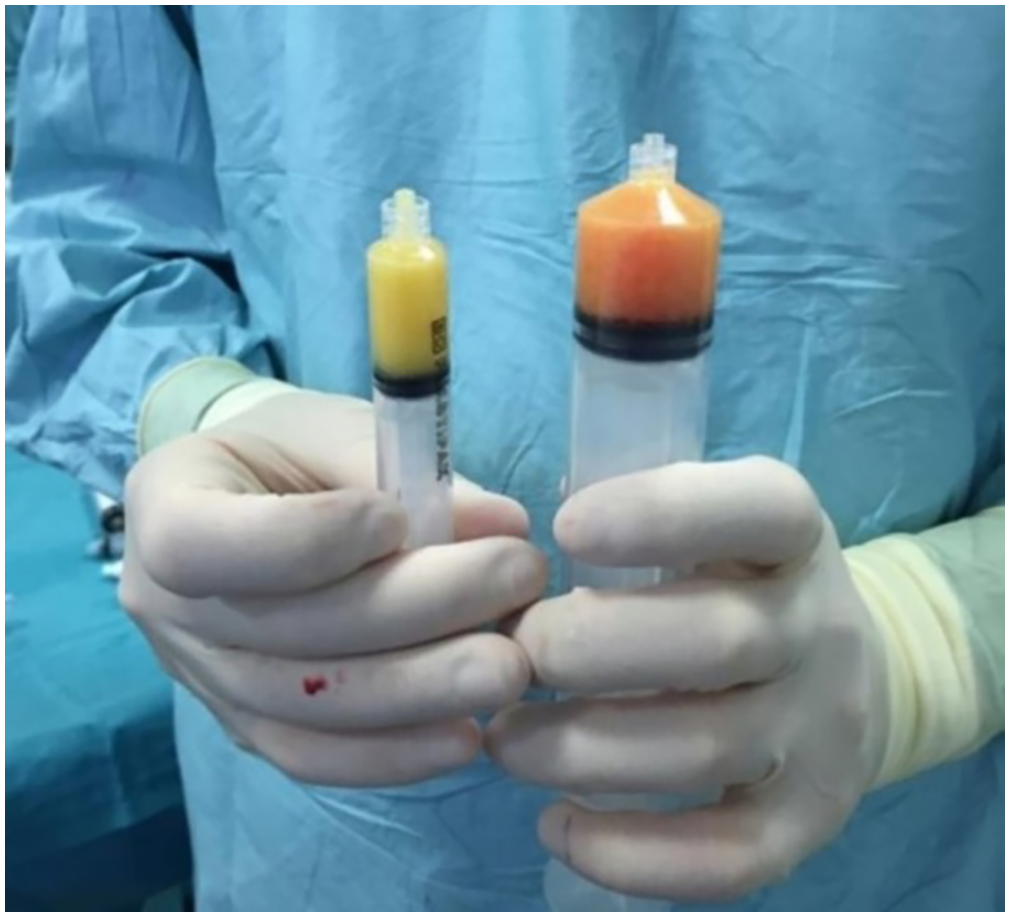
Lipoaspirate (right) and stromal vascular fraction (SVF) cells (left).
Patients were placed in a supine position, and then 7 cc of SVF was injected into each joint from the superolateral pole of the patella when the knee was in full extension or slight flexion. A 16 gauge 1.5-inch needle was used for the injection. The whole process was performed under aseptic conditions and for a total of 30–40 min.
Evaluation
Pain and functional limitations were evaluated using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) questionnaires before treatment, at 5 months, and 12 months after SVF injection.
Furthermore, X-ray imaging of the knee was obtained in all patients before and 5 months after the injection to evaluate joint space status and synovial fluid concentration (Figure 3).
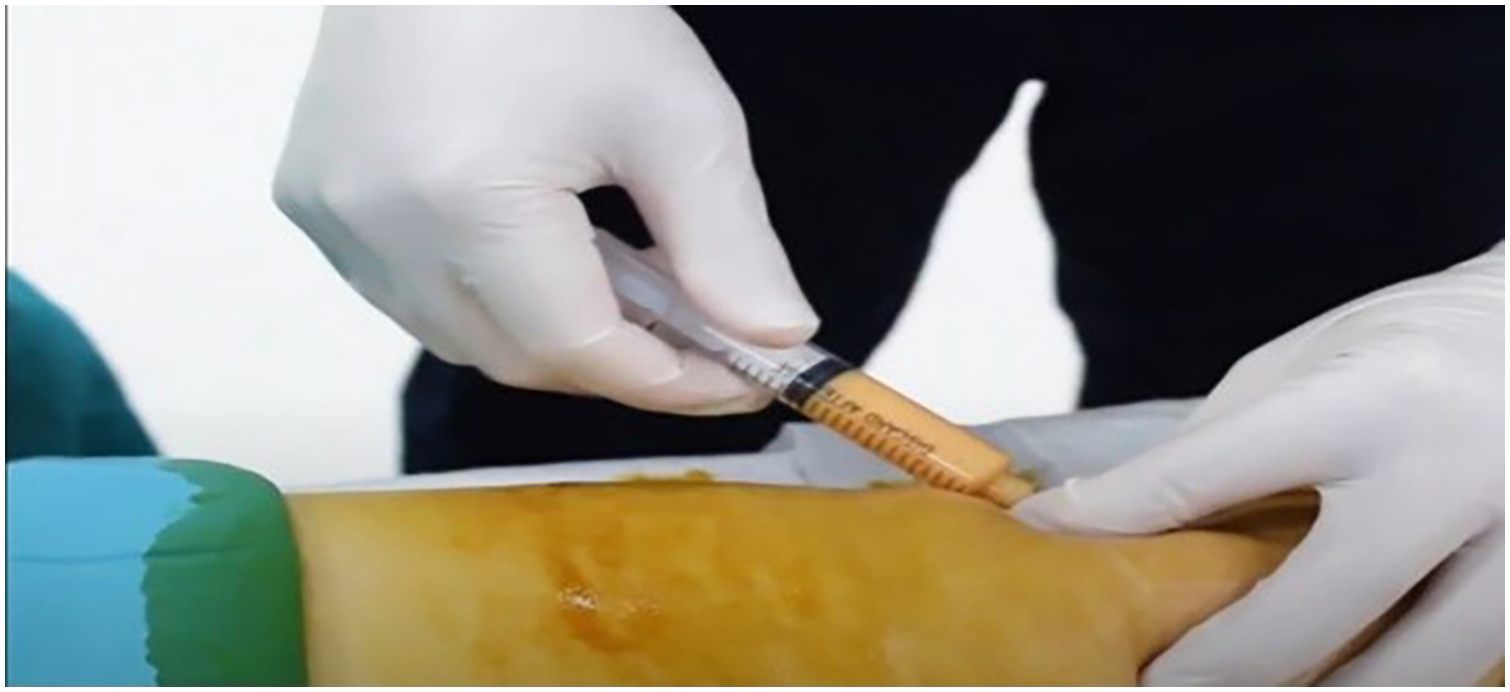
Injecting stromal vascular fraction (SVF) cells.
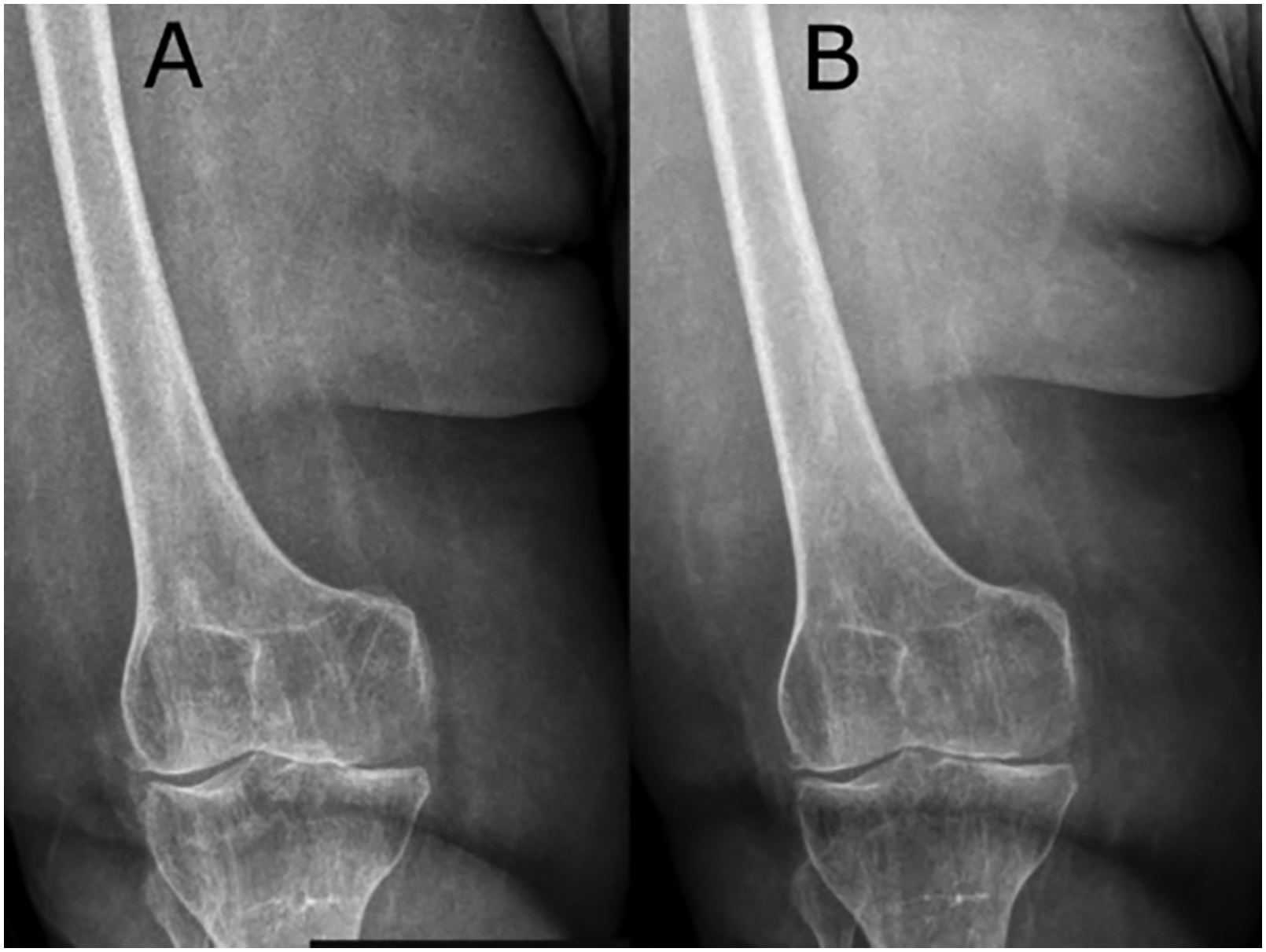
X-ray image pre-treatment (a) and after the fifth month (b).
Statistical analyzes
Statistical analyses were carried out in the SPSS program. The type I error was accepted as 0.05. The distribution of the WOMAC scores of the pre-treatment, 5 and 12 months, and the independent variables age, gender, and KL stages were evaluated using by Shapiro Wilk normality test, Q-Q plotgraphic, and histogram. It was determined that the data did not comply with the normal distribution (p < 0.05). For this reason, non-parametric methods: the Friedman F test (comparison of repeated measurements over time) and the Kruskal-Wallis test (comparisons made in terms of independent variables at the time point) were used to compare group medians. Wilcoxon test was used for pairwise comparison of repeated measurements (pre-treatment, fifth month and 12th month). Differences between groups were analyzed according to their median values.
Results
A total of 55 patients aged between 32 and 67 years (average age 57.7 years) were treated with SVF cells isolated from adipose tissue. Five of the patients were males (9.1%) and 50 were females (90.9%). KL grades of each patient were determined according to the examination of X-ray images: seven patients (12.7%) Grade I, 23 patients (41.8%) Grade II, 20 patients (36.4%) Grade III, and five patients (9.1%) Grade IV. No serious side effects were observed, except for pain and swelling at the injection and fat collection sites, which lasted for a few days.
There were significant differences between the pre-treatment, 5 and 12-month WOMAC scores changes (Table 1) (p < 0.05). The median of WOMAC scores; was 73.96 pre-treatment, 38.54 in the fifth month, and 25.00 in the 12th month. Patients’ median WOMAC score in the 12th month was lower than the fifth month after the treatment (Table 1) (p < 0.05).
The statistical description of the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score changes in pre-treatment, fifth month, and 12th month.
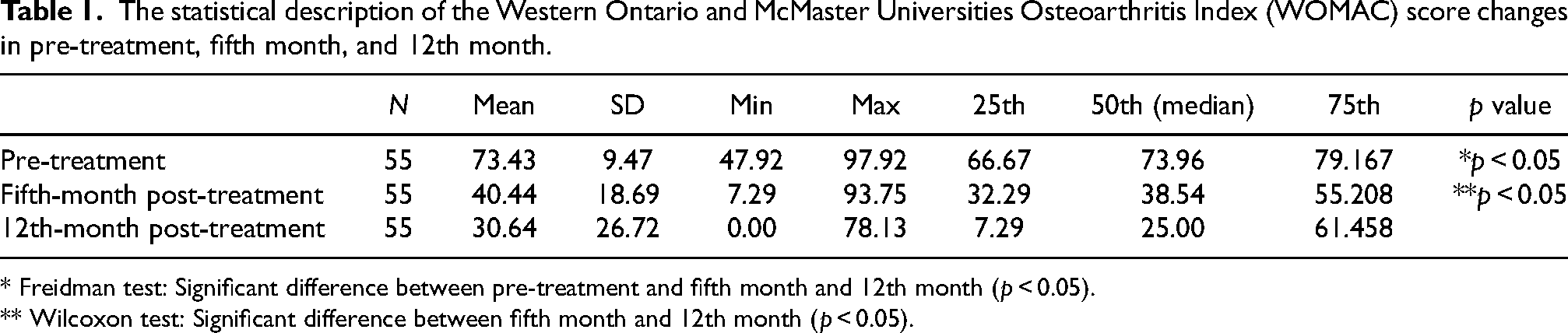
* Freidman test: Significant difference between pre-treatment and fifth month and 12th month (p < 0.05).
** Wilcoxon test: Significant difference between fifth month and 12th month (p < 0.05).
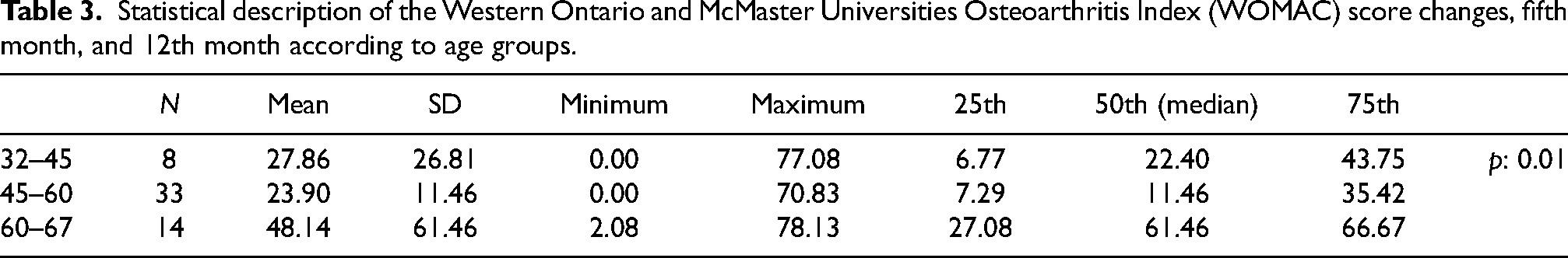
It was analyzed how the effect of SVF treatment changed over time according to KL grades. There were no significant differences between the treatment effect and KL grades in the fifth month (p > 0.05), but significant differences were found in the 12th month (p < 0.05). WOMAC scores of patients with Grades I and II were lower than in Grades III and IV when the treatment effects were compared at the 12th month according to KL grades. In addition, it was observed that the efficiency of SVF decreased after the fifth month in patients with Grades III and IV (Table 2). Also, the effect of SVF treatment was evaluated according to age, who benefited most from the treatment in the 12th month were patients aged 45–60 years. (p < 0.05) (Table 3). There was no significant relationship between SVF efficiency and gender (p > 0.05) (Table 4).
WOMAC score changes in pre-treatment, fifth month, and 12th months according to KL grades.

WOMAC: the Western Ontario and McMaster Universities Osteoarthritis Index; KL: Kellgren-Lawrence.
Statistical description of the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score changes, fifth month, and 12th month according to age groups.
The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score changes in pre-treatment, fifth month, and 12th months according to gender.
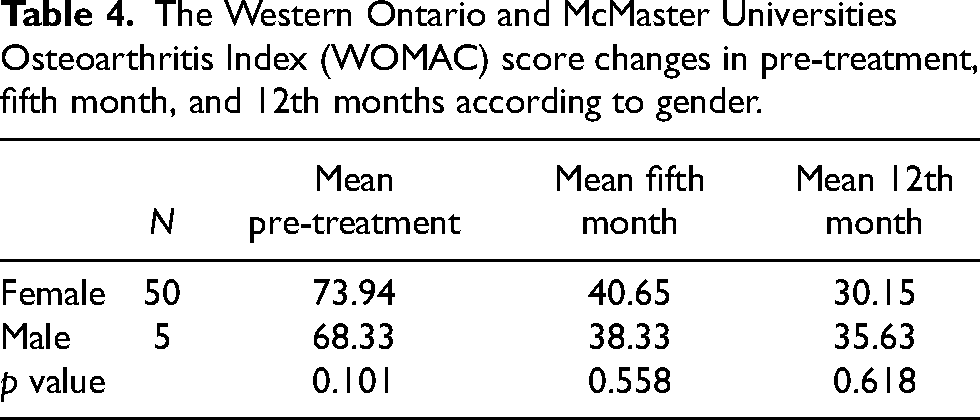
Discussion
Autologous SVF cell therapy has been commonly used in KOA. This therapy is an effective and safe method that significantly improves the life quality of OA patients. MSCs and other regenerative cells included in the SVF can produce many reparative factors with immunomodulatory abilities and migrate to the damaged area.2,28–30
Enzymatic and non-enzymatic methods are used for isolating SVF cells. The most commonly used SVF isolation method, enzymatic isolation is based on digesting with collagenase, however, it is expensive and complicated. Alternative to this, mechanical methods focus on using shear force to break down the adipose tissue so the non-enzymatic methods are cost-effective, faster, and minimally manipulated.19–21 The Lipocell mechanical isolation device offers a simple dialysis mechanism that reduces the possibility of fat obstruction, depends on using less strength of the operator for mechanical forces, and the isolated cells have been displayed to exhibit preserved structural integrity.24,31 In most studies, the average number of cells obtained by the group isolated by the enzymatic method was found to be significantly higher than that obtained by the mechanically isolated group, however, the mechanically isolated SVF group was reported to show greater potential for stemness and differentiation.31–34 It was also reported that the mechanically isolated SVF group had two times greater levels of surface markers CD34, CD13, CD73, CD90, and CD146 for mesenchymal, endothelial, and hematopoietic cells, compared to the enzymatically isolated SVF group.31,33 Mechanically isolated SVF cell culture has been reported to show greater growth rate and confluency than enzymatically isolated SVF cells.33,34 Bianchi et al. 31 demonstrated by histological staining analysis that mechanically isolated SVF cells maintained the vascular stroma better than enzymatically isolated cells. They also showed that vascular endothelial growth factor (VEGF), kinase insert domain receptor (KDR), and hepatocyte growth factor (HGF) genes involved in the regulation of vasculogenesis and capillary formation had higher expression levels. These results indicate better intercellular communication, more vascularisation potential, and higher levels of growth factors meaning consequently better cell growth and regeneration. In addition, it was reported that the enzymatic method disrupts cell–cell interactions, degrades the extracellular matrix (ECM), and cell surface environment. ECM stiffness is known to regulate homeostatic and regenerative processes and the primary function of chondrocytes is to maintain or restore the ECM.23,25 A study by van Boxtel et al. 23 reported that SVF cells isolated from adipose tissue with a mechanical method co-cultured with chondrocyte cells stimulate chondrocyte proliferation. Undisrupted ECM is able to act as a scaffold to protect the ECM niche while guiding the release of regenerative cells, cytokines, and growth factors, so the slow release of stromal and stem cells is thought to prolong their regenerative effects.21,23,35 Desando et al. 36 reported in rabbit experiments that enzymatically derived SVF cells targeted the synovial tissue but mechanically isolated SVF cells target the joint cartilage and provide greater regeneration. A maintained natural environment also supports higher adaptation and adhesion properties and exosome secretion. Exosomes secreted by SVF exhibit important paracrine factors affecting the therapeutic potential of tissue and cellular products.20,31,37,38
In recent years, commercial non-enzymatic isolation kits have been used popularly. The sterile closed isolation kits minimize the risk of bacterial contamination, gentle processing of the tissue limits cell stress, preserve ECM, present a lower amount of hematopoietic cells, and a higher percentage of exosomes.20,24,39 We evaluated the efficiency of non-enzymatic adipose tissue-derived SVF treatment in patients with KOA and presented the study's results retrospectively.
Our results show that SVFs are effective in patients with Grades I and II at 12 months. Although SVF therapy was effective for all grades up to the fifth month, its efficacy decreased after the fifth month in patients with Grades III and IV (Table 2). There are many studies showing that SVF effectiveness is maintained for a longer period of time. In a study by Michalek et al., 40 91% of patients showed significant improvement in KL scoring after 12 months. Another study by Aletto et al., 41 stated that SVF treatment was beneficial for Grades I, II, and III up to the sixth month but excluded Grade IV patients. In a 24-month follow-up study, Tran et al. 42 reported that the KL Grade III group showed significantly greater improvement. According to the results of our study and previous studies, it was shown that SVF treatment is effective in the long term. However, further research may be required to understand the role of KL grades on the efficacy of SVF treatment.
Aging has the most major impact among the several risk factors related to OA. 43 Age-related changes in cells, such as epigenetic alterations, mitochondrial dysfunction, senescence, and altered intercellular communication, cause an inadequate response to joint stress and injury, thus causing cartilage and surrounding joint tissue damage.44,45 In a prospective study of 1128 adult OA patients by Michalek et al., it was reported that older patients had a higher score (KOOS/HOOS) before cell therapy and remained at a higher OA score compared to younger patients throughout follow-up after SVF cell therapy. 46 In this study, the most significant decrease in the WOMAC score after SVF treatment in patients aged 45–60 was observed. The highest WOMAC score was found in OA patients aged 60–67 (Table 3). Aging is a factor affecting SVF cell yield. 47 Mesenchymal stem cells in the SVF content decrease their tissue regeneration abilities with aging. Since the proliferation rate and anti-inflammatory effects are reduced, SVF treatment has a lower effect in older individuals than younger individuals.48,49 The lower SVF treatment efficacy in individuals aged 60–67 years compared to younger groups can be explained by the loss in the regeneration ability of the cells in the SVF content due to aging.
Biomechanical factors, which depend on the different anatomical structures of the female and male bodies, have an essential role in the development of KOA. Females have a more significant mechanical load on their knees due to their wider pelvis and much narrower steps compared to males.50,51 Therefore, OA is generally more common in women than men. Various studies have shown that the effectiveness of SVF is not related to gender.52,53 In our research, it was also found that there was no significant relationship between SVF efficiency and gender (Table 4). This may be because the number of male and female patients is not close to each other.
The limitation of this study was the absence of another conservative treatment control group to compare with. Another limitation is the long-term change in radiography was not evaluated since the fifth month of X-ray imaging may not be sufficient to detect changes in joint space. It should also be considered that the study was observed within a single ethnic group and population size. Several ethnic differences might manifest different symptom severity of OA, depending on genetics, various living styles, or cultural factors. 54
Conclusion
SVF treatment was found to be effective for all age groups, genders, and grades of KL, except KL-III and KL-IV, for up to 12 months. In this study, The Lipocell mechanical isolation kit has been used to isolate SVF cells. In comparison to the conventional enzymatic method, the non-enzymatic method provides cost-effective, faster, sterile cell isolation and less manipulation of the cells. SVF treatment was found to be more effective, especially in the 45–60 age range. It can maintain its effectiveness for longer in OA patients with early age and KL-I and KL-II grades. To sum up, the results demonstrated that a non-enzymatic SVF isolation kit potentially offers an affordable, easily accessible treatment for early KOA patients to conveniently improve physical function, joint pain, stiffness, and life quality.
Footnotes
Availability of data and materials
Apart from the personal information of the patients included in the study, the data and analysis sets created within the scope of this study can be accessed from MD Aytan Safarli (
Consent to publish
Consent to publish their images (Figures 3 and ![]() ) was obtained from the patients.
) was obtained from the patients.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All patients gave their informed consent for SVF treatment. Ethics committee approval is not required for retrospective studies by the International Medical Centre-Baku Hospital legislation. For the retrospective use of patient data, permission was obtained from the chief physician department of the International Medical Centre-Baku Hospital.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
