Abstract
Background:
The bioactive cell-free formulation (BIOF2) for cartilage regeneration has shown a major therapeutic response in severe knee osteoarthritis. However, its effect on patients with mild or moderate stages of the disease has not been studied.
Objective:
To evaluate the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score, minimal clinically important improvement (MCII) and sleep disturbances in mild, moderate, and severe stages of knee osteoarthritis (OA) with the novel cell-free formulation treatment (BIOF2).
Methods:
An open-label, nonrandomized, baseline-controlled, parallel group study on patients with mild, moderate, and severe knee OA was conducted to evaluate the effect of intra-articular administration of BIOF2. Clinical improvement was determined through the WOMAC score and MCII, whereas sleep disturbances were measured through a Likert scale questionnaire.
Results:
At 6 months post-treatment, the mean decrease in the total WOMAC score was 16.4 +/- 4.7%, 49.9 +/- 6.4%, and 62.7 +/- 4.5% in the patients with mild, moderate, and severe disease, respectively (p < 0.001, analysis of variance test). MCII at 6 months was 18%, 78%, and 100% for mild, moderate, and severe disease, respectively (p < 0.001, likelihood-ratio χ 2 test). Concerning sleep disturbances, 60% of the patients with severe OA had important sleep problems before beginning treatment, and those difficulties were overcome 6 months after treatment. Only 18% of the patients with mild disease and 16% with moderate disease had serious sleep disturbances at the beginning of the study, and there was slight improvement after treatment. No adverse events were recorded during follow-up.
Conclusion:
BIOF2 generates better patient-reported health outcomes (on pain, stiffness, function, and sleep) in the more severe cases of knee OA.
Introduction
Osteoarthritis (OA) is a chronic degenerative disease of the articular cartilage characterized by the inability of chondrocytes to produce adequate functional matrix in response to continuous damage to the joint. 1 The Centers for Disease Control and Prevention found that 52.5 million adults over the age of 18 years, which is 22.7% of the adult population, were reported to have arthritis. 2,3 Surgical treatment costs are estimated to be at least $185.5 billion per year, 4 and pharmacologic and nonpharmacologic interventions have been shown to be ineffective in preventing OA progression. 5
The latest systematic review and network meta-analysis of long-term pharmacologic intervention trials on knee OA is that we consulted and tested 33 pharmacologic interventions, including analgesics, antioxidants, bone-acting agents, nonsteroidal anti-inflammatory drugs (NSAIDs), intra-articular injection medications (hyaluronic acid and corticosteroids), symptomatic slow-acting drugs for OA, and putative disease-modifying agents for a 12-month follow-up. Their results were inconclusive regarding change in pain control, in all comparisons with placebo, 6 coinciding with the findings of other reviews. 7,8
Therefore, the prevalence of OA and its impact in terms of disability and economics have made it a worldwide health problem, given that the nonrenewable nature of the articular cartilage decreases patient quality of life.
OA has been associated with the risk factors of age, obesity, mechanical injuries, and joint trauma. 9 Symptoms of the disease include neuropathic pain, depression, and sleep disorders. 4 The knee is the most frequently affected joint and close to half of adults 50 years of age and older are estimated to have pain caused by OA.
Early management of OA is currently based on education, exercise, and drug treatments, mainly to alleviate pain and enhance daily activities and quality of life. 10 Advanced stages cause patients to undergo total knee arthroplasty with an artificial joint. However, waiting time for surgery can be long, the procedure is costly, and patients with chronic comorbidities have increased surgical risks. 11 Moreover, there are controversies regarding surgery for patients under 60 years of age. Therefore, therapeutic decisions must be individualized, and conservative treatments must be employed before performing a total knee arthroplasty. 11
The most widely used conservative treatment is the administration of paracetamol and NSAIDs. Effectiveness varies greatly, depending on the drug utilized, the length of time of its use, and disease severity. 12 NSAIDs often do not satisfactorily relieve symptoms and usually produce gastrointestinal adverse effects. 13 When that occurs, other conservative treatments can be added, such as the intra-articular application of hyaluronic acid derivatives or platelet-rich plasma (PRP). The effectiveness of both is comparable and their benefits can last up to 6 months. 14,15 Nevertheless, those treatments cannot reverse the damage. Therefore, new strategies for articular cartilage regeneration are in development. Intra-articular inoculation of mesenchymal stem cells (MSCs) is currently one of the most widely studied therapies for cartilage regeneration 16 and requires an advanced cell culture facility. Those authors also stated that implantation strategies for stem cells need to be improved and deeper knowledge must be gained about the unknown factors that influence stem cell differentiation into chondrocytes.
BIOF2 (US Patent No. 9089580 B1) is a promising bioactive cell-free formulation for articular cartilage regeneration. 13 It is a mixture primarily composed of a corticosteroid and organic acids with small amounts of an insulin analog. Corticosteroids, when acting alone, can facilitate joint destruction, but when acting in synergy with the other active molecules in BIOF2, they have been shown to have a chondrogenic effect. 13,17 The intra-articular application of BIOF2 in different OA animal models significantly increased cartilage thickness, in addition to producing histological changes that demonstrated a decrease in disease severity. 17 Nonetheless, the formula was tested only on mice. Therefore, a clinical trial on human patients is in place to test whether the histological changes, demonstrating the trend of increasing cartilage thickness, continue.
The results of clinical trials conducted on patients with severe OA of the knee showed that intra-articular application of BIOF2 was well-tolerated and its efficacy was highly superior when compared with paracetamol and other NSAIDs. Outcomes were positive with no significant difference in its success rate versus that of total arthroplasty. Success was correlated with increased articular cartilage. 18 Its therapeutic efficacy was drastically reduced in patients with grade 2 obesity. 19 The novel BIOF2 therapy has only been tested in patients with severe knee OA, and its efficacy in mild and moderate disease stages is unknown. A different therapeutic response, according to OA severity, has been observed in different intra-articular treatments. 20,21 The severity of knee OA can be classified clinically or radiologically, and there is a significant correlation between the two evaluation methods. 22 In general, there is less probability of an important clinical response to intra-articular treatments, the more severe the disease, and patients with early stage disease have had the best therapeutic response. 21,23 However, higher baseline pain or functional impairment associated with greater response to intra-articular steroid injections in knee OA has also been reported. 23
Our proposal is based on the fact that any new therapy for OA must be evaluated in all three stages of the disease to determine whether patient response is maintained across all stages or if there is a specific stage at which the therapy is more beneficial to the patient.
The new BIOF2 treatment has been shown to be superior to NSAIDs in severe knee OA. Therefore, the present study attempts to determine whether BIOF2 therapy improves the WOMAC score, the Knee Function Rasmussen score, minimal clinically important improvement (MCII), and quality of sleep in mild, moderate, and severe stages of the disease.
An open-label, nonrandomized, baseline-controlled study was conducted on patients with mild, moderate, and severe knee OA to determine the limits of the new treatment with BIOF2 and identify the patients that could benefit from its application, according to their clinical disease severity.
Materials and methods
Study design
An open-label, nonrandomized, baseline-controlled, single-blind, parallel group study in patients with mild, moderate, and severe knee OA (phase II clinical trial) was conducted between March 2016 and March 2017.
Variables
The dependent variable was the clinical outcome measure determined through the WOMAC score (quantitative discrete), the MCII response (qualitative dichotomous), and the Knee Function Rasmussen score (quantitative discrete). The independent variable was the grade of knee OA (qualitative ordinal: mild, moderate, and severe). All groups received BIOF2 therapy. The intervening variable was a change in sleep, which was measured through a Likert scale question (quantitative discrete).
Study patients
The inclusion criteria were patients of at least 18 years of age with a body mass index (BMI) <35 kg/m2 and knee OA, according to the diagnostic criteria of the American College of Rheumatology. The target knee was defined as the more symptomatic knee (with a pain score of at least 1 on the 0–10 visual analog scale for at least 6 months before enrollment in the study). The patients had to be under usual medical care based on paracetamol/NSAIDs prescribed by their family physician, and they were stratified into three groups, according to the level of symptom intensity of knee OA determined by the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC). The disease was considered mild with a score of 0–32, moderate with a score of 33–64, and severe with a score of 65–96. 24 The exclusion criteria were having received some type of intra-articular treatment within 12 months before the study, a history of knee surgery, inflammatory polyarthritis, patients with genu varum or genu valgum malalignment greater than 20°, class 2 obesity (BMI of 35 or higher), fibromyalgia, chronic fatigue syndrome, thromboembolic disease, hemorrhagic blood disease, hemoglobin <80 g/L, neuromuscular disease, cancer, metabolic bone disease, alcoholism, or drug addiction. Patients whose BMI increased to 35 or higher during the course of the trial were eliminated from the study. Participants were recruited from primary and secondary health care centers in the State of Colima, Mexico. The efficacy evaluations and intra-articular BIOF2 applications were performed at the Centro Hospitalario Unión (a secondary healthcare center) located in the State of Colima, Mexico.
BIOF2 administration
BIOF2 is a patented formulation for cartilage regeneration, whose main components are corticosteroid and organic acids. 13 The formulation was produced by Esteripharma Mexico (Mexico City, Mexico), according to the Good Manufacturing Practices for pharmaceutical products for use in clinical trials. BIOF2 was administered on three occasions at 2-month intervals (at months 0, 2, and 4). It was an outpatient procedure performed at a traumatology or orthopedics consultation office, as previously described. 19 The patient was in a seated position with the target knee flexed at 0°. BIOF2 was injected into the knee joint space with a 1.5-in 20-gauge needle under sterile prep conditions. The area of injection was inferior and lateral to the patella at the joint line level. The patient could carry out his/her normal activities (excluding strenuous physical activities) immediately after the procedure with no special indications. All patients continued to be seen by their family physician for general care, healthy lifestyle promotion, and if necessary, continued taking the paracetamol/NSAID-based treatment regimen with no intervention from the researchers, concerning drug prescription or lifestyle indications.
Outcome measures and follow-up
The coprimary endpoints, assessed as change from the baseline, were the differences between the values at enrolment and 3 and 6 months post-treatment.
The primary endpoint was the change in the WOMAC score. The WOMAC instrument has a total score and subscales for pain, stiffness, and physical function. 20 The total score may be used to classify the severity of the disease as mild (0–32 points), moderate (33–64 points), or severe (65–96). 24 Because patients with different disease severity levels (and consequently, a different baseline WOMAC total score) were analyzed, the response criterion was based on the improvement percentage of the total score, per group, as previously postulated. 25 The reviewers and patients could observe the reduction of symptom severity with that approach, and it also provided a straightforward overview of clinical improvement, regardless of the degree of disease severity. 25,26
The second endpoint was the number of patients achieving MCII, defined as the smallest change in measurement that signifies an important improvement in a symptom. 27 It was calculated through a dichotomous score per outcome, based on a 30% improvement of the WOMAC score from the baseline. That percentage of improvement has been described as clinically relevant in different clinical trials. 28 Treatment success was defined as the MCII at months 3 and 6 of the follow-up.
The third endpoint was the change in the Rasmussen clinical score, which provides a record of the functional results of the joint after treatment. A score of 28–30 was considered excellent, a score of 24–27 was good, 20–23 was fair, and a score of <20 was poor. 29
Given that OA can be associated with sleep disturbances, 30 the fourth endpoint was the patient’s quality of sleep, determined by the response to the question: Are you able to get a good night’s sleep? The response options were (1) without any difficulty; (2) with some difficulty; (3) with much difficulty, and (4) unable to sleep. That question is validated in the Routine Assessment of Patient Index Data 3, which is a pooled index of the three patient-reported core data set measures of the American College of Rheumatology and it has previously been used in patients with OA. 18 Finally, during the follow-up period, all adverse events were registered and monitored by the researchers through anamnesis and abnormal routine laboratory test results. The laboratory blood test parameters were evaluated at the baseline and 3 and 6 months after treatment, and included complete blood count, glucose, creatinine, chloride, potassium, total cholesterol, triglycerides, liver enzymes (alanine aminotransferase, aspartate aminotransferase, and lactate dehydrogenase), fibrinogen, C-reactive protein, and the erythrocyte sedimentation rate.
Blinding
Only the researchers that evaluated treatment effectiveness through the WOMAC score and MCII instruments, answered by the patients, as well as those who performed the statistical analyses, were blinded.
Sample size
The calculation was based on the difference (60%) in the number of patients with MCII at 6 months, comparing the groups with mild versus severe disease (30% vs. 90%). At least nine patients from each group were needed to reach a statistical power of 80% and a 5% α was used for the statistical analysis as a two-tailed test. The calculation was made using the sample size calculator for two independent study groups with binomial primary endpoints (ClinCalc LLC; http://clincalc.com/stats/samplesize.aspx).
Ethics approval and consent to participate
The study was approved by the ethics committee of the Instituto Estatal de Cancerología of the State of Colima, Mexico (CEICANCL170317-ENM-OSTEOAR-03), and written informed consent was obtained from all the participants. All procedures performed in this protocol were carried out in accordance with the Declaration of Helsinki. The present clinical trial was registered as ARTROTX: RPCEC00000250 in the Cuban Public Registry of Clinical Trials database (Primary Registry of World Health Organization Registry Network).
Statistical analysis
Data were presented as the mean ± standard deviation or error and percentages. For the inferential statistics, normal data distribution was first determined using the Kolmogorov–Smirnov test and the equality of variances was confirmed using the Levene’s test. The numerical data (BMI, age, or clinical scores) were compared between the data of the three subgroups (according to disease severity or point-in-time evaluation) using the one-way analysis of variance (ANOVA) with the Bonferroni post hoc tests. The categorical values were compared between the two and/or three subgroups using the likelihood-ratio χ 2 test. The relative risk (RR) and 95% confidence interval (CI) were calculated to determine the probability of achieving MCII, comparing the severe OA group data versus those patients with mild or moderate level OA. The Pearson correlation coefficient was calculated to evaluate the relation between the baseline WOMAC score and the percentage of clinical improvement at 3 and 6 months from the beginning of treatment with BIOF2. The statistical analysis was carried out using the SPSS, version 20, software (IBM Corp., Armonk, New York, USA), except for the RR, which was calculated using MedCalc v17.7.2 software (MedCalc Software bvba, Ostend, Belgium). A two-sided p <0.05 was considered statistically significant.
Results
Of the 61 patients screened, 52 were included in one of the three study subgroups (number of patients with mild, moderate, or severe disease level: 24, 18, and 10 patients, respectively). Two patients from the mild OA subgroup were eliminated because their BMI increased to more than 35 in the follow-up period, leaving a final total of 22 patients in that subgroup. The clinical characteristics of the patients at the beginning of the clinical trial are presented in Table 1. There were no significant differences in the clinical characteristics of the patients in the three subgroups, except in the variables related to OA severity.
Distribution of the main clinical characteristics of the patients at the beginning of the clinical trial.a
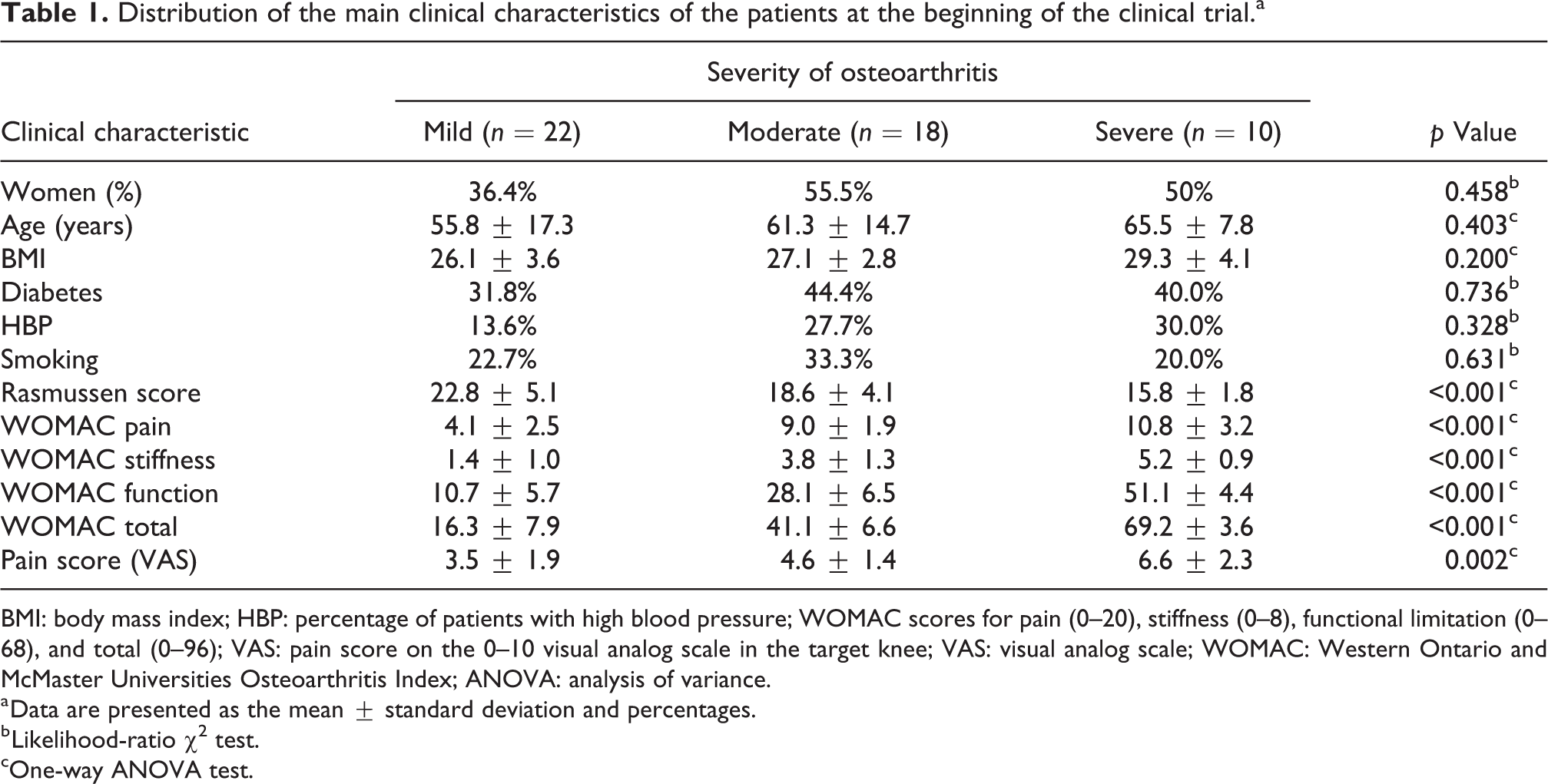
BMI: body mass index; HBP: percentage of patients with high blood pressure; WOMAC scores for pain (0–20), stiffness (0–8), functional limitation (0–68), and total (0–96); VAS: pain score on the 0–10 visual analog scale in the target knee; VAS: visual analog scale; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index; ANOVA: analysis of variance.
a Data are presented as the mean ± standard deviation and percentages.
b Likelihood-ratio χ 2 test.
cOne-way ANOVA test.
The patients with severe OA had the fastest and greatest degree of improvement, whereas patients with mild disease had limited improvement (see Figure 1 and Supplemental Material). The clinical improvement of the patients was greater (higher percentage of reduction in the total WOMAC score) when there was a higher total WOMAC score at the beginning of the study (greater disease severity) with a correlation coefficient of 0.63 (p < 0.001) and 0.57 (p < 0.001) at 3 and 6 months, respectively. Figure 1 shows the different levels of OA severity. At 6 months of follow-up, clinical improvement, or the mean decrease in the total WOMAC score, was 16.4 +/- 4.7%, 49.9 +/- 6.4%, and 62.7 +/- 4.5%, in the patients with mild, moderate, and severe disease, respectively (p < 0.001, ANOVA test). At 6 months, the percentage of reduction in the WOMAC score was lower for mild disease, compared with moderate (p < 0.001) or severe (p < 0.001) disease. There were no differences between moderate disease and severe disease (p = 0.406). The percentage of patients who achieved MCII at 6 months was 18%, 78%, and 100% for mild, moderate, and severe disease, respectively (p < 0.001, likelihood-ratio χ 2 test). It is striking that the patients with severe disease almost completely achieved their maximal clinical improvement starting at 3 months, whereas the patients with moderate disease, despite showing improvement at 3 months, did not achieve their maximal effect until 6 months (see Figure 1 and Supplemental Material). The best response to treatment with BIOF2 was in cases of severe disease. There was a 1.7- and 3.5-fold greater probability of achieving MCII at 6 months for moderate disease (RR 1.7, 95% CI 1.2–2.4) and mild disease (RR 3.5, 95% CI 1.5–8.0), respectively. The therapeutic efficacy of BIOF2 in patients with moderate knee OA can be considered adequate, given that 78% of the patients reached MCII at 6 months and had a 3.6-fold greater probability of achieving clinical improvement, compared with the patients with mild disease (RR 3.6, 95% CI 1.5–8.9).
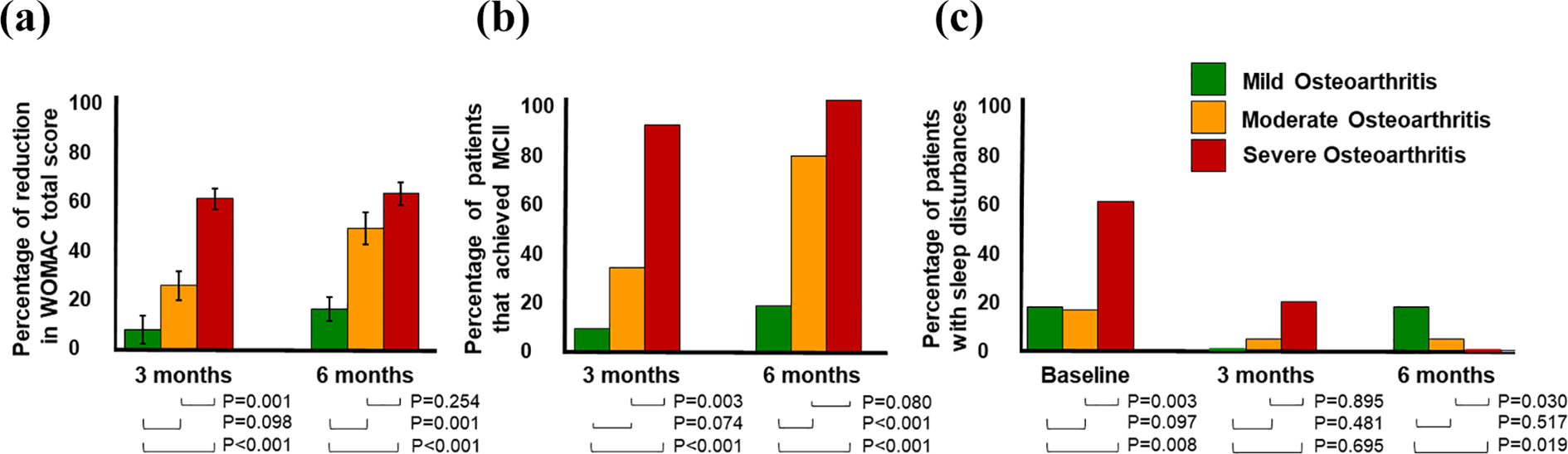
Comparison of PRO per group, at 3 and 6 months, post-BIOF2 treatment. (a) Percentage of reduction in the WOMAC total score (average + SE). (b) Percentage of patients that achieved MCII. (c) Percentage of patients with great difficulty falling asleep/staying asleep at night, or unable to fall asleep/stay asleep. The p values were calculated using Bonferroni test (a) or likelihood-ratio χ 2 test (b and c). PRO: patient-reported outcome; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index; MCII: minimal clinically important improvement.
Patient progression, according to the Rasmussen clinical score, was similar to the results observed with the WOMAC score. Patient clinical improvement at 6 months (higher percentage of increase in that parameter) was greater when there was a lower Rasmussen clinical score at the beginning of the study (greater disease severity) with a coefficient correlation of −0.66 (p < 0.001). At the end of the follow-up period, the mean increase in the Rasmussen clinical score was 28.6 +/- 7.3%, 44.5 +/- 10.4%, and 63.6 +/- 5.7% in the patients with mild, moderate, and severe disease, respectively (p = 0.048, ANOVA test, see Supplemental Material). Table 2 presents the total WOMAC score and Rasmussen clinical score, per subgroup, according to OA severity.
Total WOMAC score and Rasmussen clinical score, per subgroup, according to osteoarthritis severity.

ANOVA: analysis of variance; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index.
aOne-way ANOVA test, the three time periods were compared in patients with the same severity.
Concerning sleep disturbances, 60% of the patients with severe OA had important sleep problems before beginning BIOF2 treatment, and those difficulties were overcome at 6 months after treatment (Figure 1 and Supplemental Material). Only 18% of the patients with mild disease and 16% with moderate disease had serious sleep disturbances at the beginning of the study, and there was slight improvement after treatment (see Figure 1). No adverse events were recorded during the follow-up, except intense pain in one patient that lasted for 1–3 min at the intra-articular injection site and ceded spontaneously. No differences were found in any of the parameters of the laboratory blood tests at 3 and 6 months, regarding the baseline values (Supplemental Material).
Discussion
Treatment with BIOF2 produced a therapeutic response according to knee OA severity. The patients with severe disease had the greatest clinical improvement, whereas patients with mild OA perceived a limited therapeutic effect. The patients with moderate and severe disease had a 3.5-fold greater probability of achieving MCII at 6 months, compared with the patients with mild disease. Previous reports of clinical trials described a great therapeutic effect of BIOF2 in patients with severe knee OA, 18,19 just as we found in our study, but its effect on patients with mild and moderate disease had not been evaluated. A limitation of the present study is that cartilage thickness was not assessed by ultrasound or magnetic resonance and biochemical markers, such as cartilage oligomeric matrix protein (COMP), were not evaluated.
There are clinical and molecular differences with respect to OA severity. Disease progression and loss of cartilage are slower in early OA 31 than in advanced-stage disease. 9,32 Differences at the molecular level have also been observed that are dependent on the degree of cartilage damage or OA symptom severity. 33 –35 The results of the present study indicate that BIOF2 had a different influence on the intra-articular microenvironment. BIOF2 was previously shown to have the capacity to regenerate articular cartilage in animal models and in humans. 18 In patients with severe OA, there was a better clinical result with BIOF2, producing articular cartilage regeneration. 18
Trials conducted on gene expression in human synovial cells have shown that one of the most important molecular effects produced by BIOF2 is increased SRY-related HMG box 9 (SOX9) expression. 17 SOX9 is a transcription factor that is essential for chondrocyte differentiation and cartilage formation with a notable role in the maintenance of the chondrogenic phenotype. 17 Effective chondrogenesis and inhibition of endochondral ossification have additionally been demonstrated to be achieved by directing MSCs toward the chondrocyte lineage with SOX9. 36 That information has resulted in the proposal that chondrogenesis stimulation due to the elevated levels of SOX9 in the MSCs present in articulation is one of the mechanisms of BIOF2 involved in articular cartilage regeneration. 17,18
SOX9 has recently been shown to vary greatly, according to the stage of OA: it is upregulated in the early stage but downregulated in the later stage of the pathology. 37 Compared with normal cartilage, there is an approximately 80% increase in SOX9 expression in mildly damaged cartilage. In intermediate cartilage damage, the percentage decreases to approximately 60%. There is a three- to eightfold decrease in SOX9 expression in severely damaged cartilage. 37,38 SOX9 upregulation in early stages of OA is a proposed mechanism that attempts to compensate for the damage to the cartilage through a certain regenerative capacity, suppressing extracellular matrix proteins (ADAMTS—a disintegrin and metalloproteinase with thrombospondin motifs—family proteins) and stimulating the production of the structural proteins of the extracellular matrix, such as proteoglycan aggrecan, COMP, and type II collagen. 37 However, compensatory capacity is limited and cartilage damage often results in dysfunction and OA progression, suggesting that moderate and severe stages are the lost generation period with SOX9 downregulation. 37 SOX9 expression has been detected not only in chondrocytes but also in chondrogenic progenitor cells, synovial fibroblasts (synoviocytes), and cells isolated from synovial fluid. 39 Those variations in the molecular microenvironment could be the reason why BIOF2 does not have the same effect on the different stages of OA. In joints with early stage disease, SOX9 expression may be naturally elevated, and thus, the application of BIOF2 at that stage of disease would not produce great molecular changes for SOX9 levels, resulting in a very limited therapeutic effect. As the pathology progresses, SOX9 expression in the joint cells begins to decrease and the application of BIOF2, whose mechanism of action is the elevation of SOX9, could begin to produce important changes that have a therapeutic effect through cartilage regeneration. 18 The results of the present study are congruent with that explanation, given that a highly significant correlation was shown between disease severity and the therapeutic effect: BIOF2 had a greater therapeutic effect, the greater the severity of the disease.
Another effect demonstrated by BIOF2 application was the threefold decrease in osteoglycin (OGN) gene expression in joint cells. Also known as mimecan, or the osteoinductive factor, OGN has been related to bone formation, and elevated levels have been found in patients with OA in both synovial fluid and damaged cartilage. 40,41 OGN is highly correlated with knee OA severity. 38 The more severe the symptomatology, or the greater the cartilage damage, the higher the OGN expression (Pearson r 0.69, p = 0.0002 and Pearson r 0.70, p = 0.0001, respectively). 35 Therefore, the inhibitory effect of BIOF2 on OGN can have greater relevance, the more severe the OA, which could be another explanation for the varied therapeutic effect of BIOF2, according to the microenvironment found at the different stages of OA.
The therapeutic responses of PRP application or hyaluronic acid viscosupplementation are better in patients with early stage disease. From the radiologic perspective, an important clinical response in the patient is less likely, the more severe the disease. 21,23 Hyaluronic acid has recently been considered inappropriate treatment, or to have uncertain effectiveness, in patients with severe OA. 42 Thus, BIOF2 is a new conservative therapeutic option for patients with severe disease, in whom other conservative therapeutic options have not been satisfactorily effective.
The evaluation of sleep disturbances was another important aspect of the present study. Insomnia is frequently experienced by patients suffering from chronic musculoskeletal disorders 30 but is often seen as simply a symptom of pain or depression. However, insomnia has been postulated to be a significant and pervasive problem in chronic musculoskeletal diseases that is a construct that is relatively independent of both pain and depression, 43 and so, specific insomnia assessment and treatment is recommended. 43 The present study showed that 60% of the patients with severe OA had serious sleep problems, whereas only 16–18% of the patients with mild or moderate disease suffered from sleep disturbances. Treatment with BIOF2 helped all insomniac patients with severe disease to sleep better. The insomniac patients with moderate disease experienced a slight effect and it was practically null in the patients with mild disease. Those results concurred with the clinical improvement of knee OA evaluated by the WOMAC and Rasmussen clinical scores, positioning BIOF2 as an effective therapy for sleep disturbances in patients with severe OA.
Treatment with BIOF2 has previously been shown to have several advantages in severe knee OA: its effectiveness is similar to that of knee arthroplasty. 18 It significantly reduces NSAID use, and it can be applied in the office of the specialist with experience in intra-articular applications, such as traumatologists, orthopedic physicians, and rheumatologists. 19 BIOF2 is easily included within a regimen of usual medical care. 19 The present and previous reports have found no important adverse effects after BIOF2 application. 18,19 The present report also showed that the main biochemical or blood cell markers underwent no significant changes during the study period. Finally, it is important to mention that the majority of patients stated that between 1 and 2 weeks before the second and third BIOF2 applications, knee symptomatology slightly increased (data not quantitatively evaluated), suggesting that future studies could evaluate applications at shorter time intervals.
Conclusion
The intra-articular application of BIOF2 produced clinical improvement in pain, stiffness, function, and sleep in patients with advanced knee OA in moderate and severe stages. Its therapeutic effect was limited in patients with early stage disease. BIOF2 is an easily implemented therapeutic alternative in patients receiving usual medical care for advanced knee OA.
Supplemental material
suplemental_material2 - A novel cell-free formulation for the treatment of knee osteoarthritis generates better patient-reported health outcomes in more severe cases
suplemental_material2 for A novel cell-free formulation for the treatment of knee osteoarthritis generates better patient-reported health outcomes in more severe cases by Ivan Delgado-Enciso, Juan Paz-Garcia, Jose Valtierra-Alvarez, Jorge Preciado-Ramirez, Bertha A Olmedo-Buenrostro, Josuel Delgado-Enciso, Jose Guzman-Esquivel, Carlos E Barajas-Saucedo, Gabriel Ceja-Espiritu, Iram P Rodriguez-Sanchez, Margarita L Martinez-Fierro, Sergio A Zaizar-Fregoso, Daniel Tiburcio-Jimenez, Jorge E Plata-Florenzano and Brenda Paz-Michel in Journal of Orthopaedic Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
