Abstract
Background
To investigate the effect of arthroscopy combined with high tibial osteotomy (HTO) on cartilage regeneration in patients with knee osteoarthritis.
Methods
A retrospective analysis of 50 patients with varus and medial compartment osteoarthritis of the knee treated by arthroscopy combined with HTO. One year after the operation, a second-look arthroscopy was performed to observe the cartilage regeneration. The regeneration of cartilage was evaluated by different pathological staining of some of the new cartilage. Finally, part of the new cartilages (n = 6) were taken for quantitative real-time PCR and western blotting experiments to display the mechanism of cartilage regeneration.
Results
One year after arthroscopy combined with HTO, the results of arthroscopy revealed the formation of new tissue in the defect area of the medial compartment’s cartilage in the knee joint. In addition, different pathological staining results indicated that the new tissue was cartilage-like tissue. Furthermore, HTO potently up-regulated the expression of p-ERK1/2 at the protein level in knee osteoarthritis patients compared with control group. However, there was no significant difference in the relative expression of collagen II at mRNA and protein level between control group and knee osteoarthritis patients.
Conclusion
Arthroscopy combined with HTO can promote cartilage regeneration in patients with knee osteoarthritis.
Introduction
With the advent of global ageing, knee osteoarthritis has become one of the major chronic degenerative diseases seen in orthopedics among middle-aged and elderly patients, which has brought a heavy burden to the society.1,2 Osteoarthritis often results in loss of articular cartilage, which is critical to the function of knee joint. 3 Knee joints commonly affect one compartment, and it is associated with malalignment such that a greater load is placed on the affected compartment. 4 A correlation has been shown between malalignment and longitudinal cartilage loss, in both the medial femur and the tibia.5,6
Currently, high tibial osteotomies (HTO) are commonly used internationally to treat varus genicular combined with unilocular osteoarthritis. Osteotomy can relieve the pressure of the medial compartment of the knee, alleviate pain, delay the further development of osteoarthritis, and delay the time of total knee replacement.7,8 High tibial osteotomy alters the weightbearing axis of the lower limb by realigning the tibia, which changes forces on the articular cartilage within the knee, reducing the load on the affected compartment. 9 Woon-Hwa Jung et al. 10 have found that the degenerated cartilage of the medial femoral condyle and medial tibial plateau could be partially, if not entirely, covered by newly regenerated cartilage at 2 years, after adequate correction of the varus deformity by a medial opening-wedge high tibial osteotomy without cartilage regeneration strategies.
Studies have found that arthroscopy combined with HTO can promote cartilage regeneration in patients with knee osteoarthritis.11–13 However, the detailed molecular mechanism of arthroscopy combined with HTO on cartilage regeneration in knee osteoarthritis patients is unclear. Therefore, this study aimed to uncover the underlying molecular mechanisms of arthroscopy combined with HTO on cartilage regeneration in patients with knee osteoarthritis.
In this study, arthroscopy combined with HTO was used to treat osteoarthritis of the medial compartment combined with varus knee. We hypothesized that arthroscopy combined with HTO would promote cartilage regeneration in patients with osteoarthritis of the knee through a certain biological mechanism.
Patients and methods
Patients
All the patients were treated with arthroscopy combined with HTO. The wounds healed well postoperatively. One year after arthroscopy combined with HTO, no complications, such as infection, fracture nonunion, fracture of the nail plate and so on, were observed in any of the patients. All patients were removed plate and screw 1 year after surgery and underwent a second-look arthroscopy. Flow diagram of patient involvement in the study was presented in Supplementary Figure S1.
Clinical evaluation index
Radiological evaluation
The patients were examined by anterior and lateral knee joints and full-length X-ray films of both lower limbs before and after surgery. The surgical effect was evaluated from the perspective of imaging
Tibiofemoral Angle
The tibial Angle (FTA) after osteotomy was measured and compared with that before osteotomy. The effect of surgery was evaluated by the change of FTA on objective X-ray film
Visual analogue scale of pain
Visual analogue scale (VAS) score was used to compare the pain degree of patients before and after surgery. The surgical effect was evaluated from patients' subjective pain sensation.
Arthroscopic evaluation
The medial tibial plateau and femoral condyle articular cartilage were evaluated by arthroscopy before and again about 1 year after arthroscopy combined with HTO. To evaluate cartilage degeneration, we used the ICRS grading system 14 to grade the degeneration of articular cartilage at the time of the initial HTO. Briefly, according to the ICRS grading system, grade 1 cartilage defect were defined as superficial fissures and cracks and superficial lesions. Lesions extending down to less than 50% of the cartilage depth were defined as grade 2 defects. Lesions extending down to more than 50%, but not involving the subchondral bone were defined as grade 3 defects. Grade 4 defects were defined as lesions involving the subchondral bone.
All patients were removed plate and screw 1 year after arthroscopy combined with HTO and underwent a second-look arthroscopy. The medial femoral condyle and tibial articular cartilage were evaluated by regeneration grade.15,16 For evaluation of cartilage regeneration, the articular cartilage was classified into two stages, as no regenerative change (grade 1) or white scattering with fibrocartilage, partial coverage with fibrocartilage, or even coverage of fibrocartilage (grade 2).10,17
Pathological examination
At the time of second-look arthroscopy, part of new cartilage tissue was reserved for pathological examination. To observe the histology of the new tissue, part of the tissue was fixed, dehydrated, embedded in paraffin and stained with Hematoxylin-Eosin, toluidine blue and safranin O-fast green staining. In order to prove that the new tissue was cartilage-like tissue, we performed the immunohistochemical staining of collagen II.
Quantitative real-time PCR
Normal cartilage tissue was obtained from non-osteoarthritis patients whose cartilage was exfoliated due to tibial plateau fracture of knee joint. Normal cartilage tissue was used as the experimental control group (n = 6). Total RNA was extracted by RNA sample Total RNA Kit (TIANGEN, China). Total RNA of 2 μg each sample was purified and reversely transcribed using FastKing gDNA Dispelling RT Super Mix (TIANGEN, China). Quantitative real-time PCR (qRT-PCR) was performed using SYBR Green (Molecular Probes) on the ABI 7900 HT FAST Real-Time PCR system. The primer sequences were: β-actin, F 5′-AGGGGCCGGACTCGTCATACT-3′, R 5′-GGCGGCACCACCATGTACCCT-3’; Collagen II, F 5′-GCCTGGTGTTCATGGGTTT-3′, R 5′-GTCCCTTCTCACCAGGTTTG-3’. Relative mRNA levels were quantified and normalized to the β-actin expression level. The relative quantification of gene was assessed by the 2-∆∆Ct method.
Western blotting
Total protein was extracted by RIPA reagent according to the manufacturer’s protocol, respectively. Protein concentration was quantified using the BCA protein assay kit (Bio-Rad, USA). Equal amounts of protein from each group were separated using SDS-PAGE and then transferred to NC membranes. After being blocked with 5% milk, the NC membranes were incubated with primary antibodies against β-actin, collagen II, ERK1/2 and p-ERK1/2. This was followed by incubation with the appropriate HRP-conjugated secondary antibodies. The membranes were then visualized with ECL and exposed on film. 18
Statistical analysis
Data were represented as mean ± standard deviation for triplicate specimens, and the statistical significance was determined by paired t-test. All statistical analysis was calculated with SPSS version 21.0. p < 0.05 was considered statistically significant.
Results
Clinical evaluation index
Standard of patients selection.

Changes of FTA and VAS before and after surgery.

*p < 0.05.

Radiological evaluation: the patients were examined by anterior and lateral knee joints and full-length X-ray films of both lower limbs before and after surgery. (a) Preoperative anteroposterior X-ray of the knee joint. (b) Preoperative lateral knee radiographs. (c) Preoperative full-length x-rays of both lowerextremities. (d) Postoperative anteroposterior X-ray of the knee joint. (e) Postoperative lateral knee X-ray. (f) Postoperative full-length X-ray films of both lower extremities.
High tibial osteotomy promoted cartilage regeneration
Before HTO, the patient had obvious knee varus (Figure 2(a)), however, 1 year after HTO, the knee varus disappeared with the appearance of new cartilage tissue. (Figure 2(b)). According to the ICRS grading system, at initial arthroscopy (before HTO), 10 knees (20%) had grade 3 medial femoral condyle or medial tibial plateau articular cartilage findings, 37 knees (74%) had grade 2 findings, and 3 knees (6%) had grade 1 findings. However, at the second-look arthroscopic examination (after HTO), 2 knees (4%) had grade 3 medial femoral condyle or medial tibial plateau articular cartilage findings, 26 (52%) knees had grade 2 findings, and 22 (44%) knees had grade 1 findings. According to the regeneration grade, after HTO, no regeneration of the articular cartilage (grade 1) was found in 6% of knees (3 of 50) and grade 2 regeneration was found in 94% of knees (47 of 50). Overall, HTO might promote cartilage regeneration in patients with knee osteoarthritis. Arthroscopic pictures of arthritis patient. (a) Before HTO. (b) One year after HTO.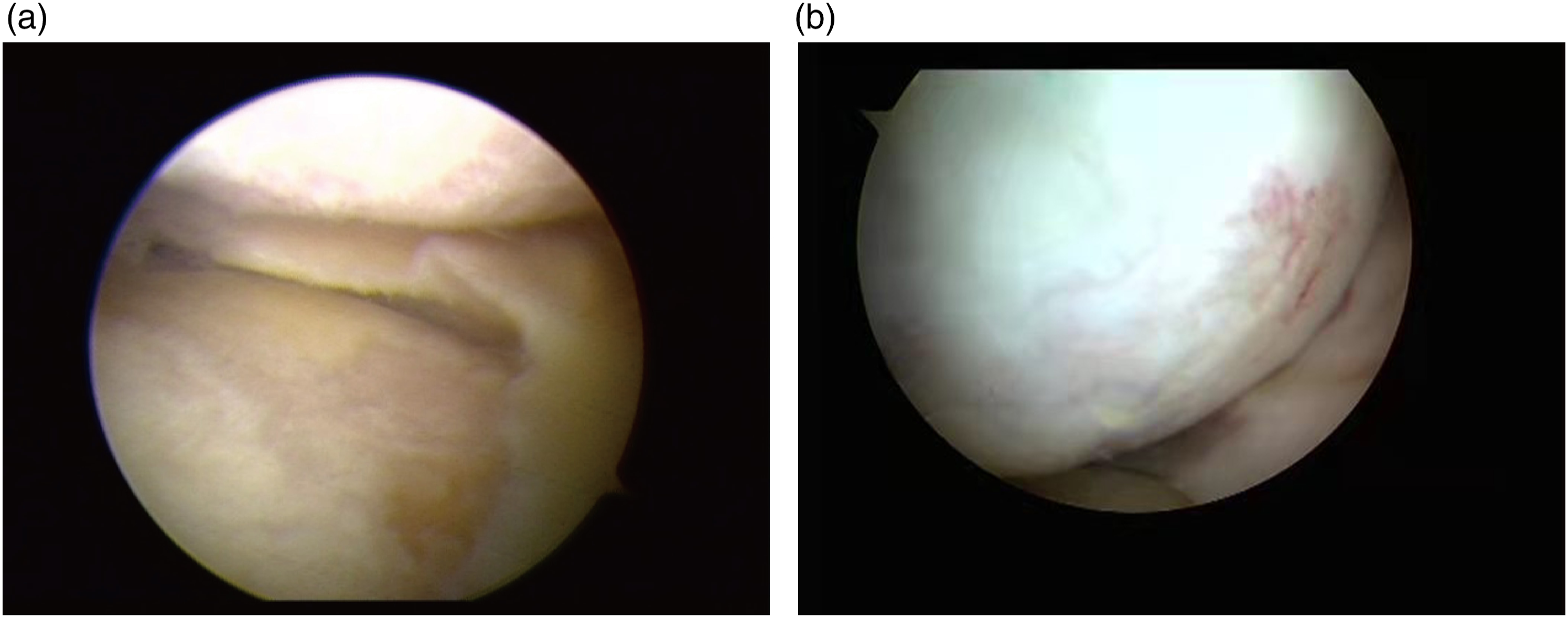
Regenerated cartilage tissues confirmation
In order to observe the regeneration of cartilage, we took some of the new cartilage from 6 knee osteoarthritis patients for pathological staining. Differential staining of the articular new cartilage tissues were shown in Figure 3. Hematoxylin-Eosin (Figure 3(a)) and toluidine blue (Figure 3(b)) staining revealed that the new tissue was bonded and integrated with the perimeter articular cartilage, and there was obvious fibrous cartilage or cartilage dimples. Collagen II immunohistochemical staining revealed that amount of collagen II existed in the tissues (Figure 3(c)). Safranin O-fast green staining results showed that red cartilage tissue, cartilage dimples and green bone tissue existed in the new tissue (Figure 3(d)). The results of staining illustrated that these new tissues were regenerated cartilage. Differential staining of the articular new cartilage tissues. (a) Hematoxylin-Eosin staining. (b) Toluidine blue staining. (c) Collagen II immunohistochemical staining. (d) Safranin O-fast green staining.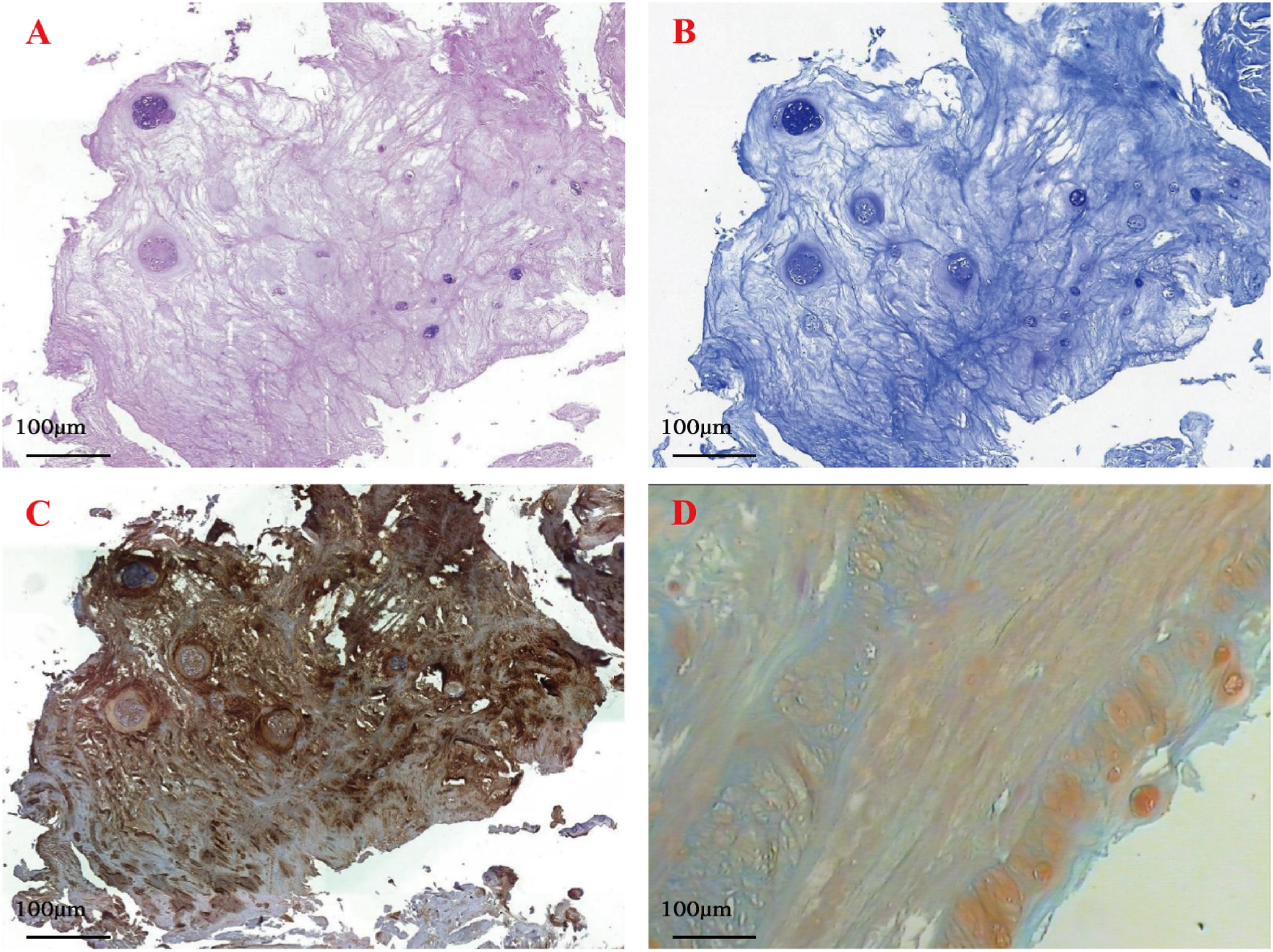
Arthroscopy combined with HTO potently influences p-ERK 1/2 expression in new cartilage tissue
In order to explore the mechanism of promoting new cartilage tissue by arthroscopy combined with HTO, the collagen II mRNA expression in normal and new cartilage tissue was detected using qRT-PCR, and the protein expression of collagen II, ERK 1/2 and phosphorylated ERK 1/2 (p-ERK 1/2) in normal and new cartilage tissue was measured using Western-blot, which were shown in Figure 4. The relative expression of collagen II mRNA in cartilage tissue was not statistically different between control and patients (p > 0.05, Figure 4(a)). In addition, the expression of collagen II, ERK 1/2 and p-ERK 1/2 in cartilage tissue was higher expressed in patients (Figure 4(b)), when compared to the control subjects, p-ERK 1/2 was dramatically upregulated in patients group (p < 0.05, Figure 4(c)), but there was no significant change in the level of protein collagen II expression (Figure 4(d)). These results indicated that the generation of new tissue might be associated with the increased expression of p-ERK 1/2. HTO did not change the level of collagen II but enhanced p-ERK 1/2. (a) The differences of collagen II mRNA expression. (b) Western blot analysis of collagen II, ERK 1/2 and p-ERK 1/2 proteins was taken using β-actin as loading control. (c) The expression of p-ERK 1/2 protein in control groups and patients groups. (d) The expression of collagen II protein in control groups and patients groups. *p < 0.05.
Discussion
Knee osteoarthritis is a common joint disease that can cause severe pain and limited activities of daily living. 19 Knee osteoarthritis is caused by an imbalance between biological resistance and mechanical stress. Relieving the stress of articular cartilage is one of the key ways for the treatment of knee osteoarthritis. 20 High tibial osteotomy has been reported to be effective in young and active medial knee osteoarthritis with varus deformity. 21 High tibial osteotomy can relieve the excessive load in the medial compartment caused by the abnormality of the lower limb line, transfer the excessive load in the medial compartment of the knee to the lateral compartment, and then promote the regeneration of cartilage on the surface of the medial compartment to relieve the symptoms of osteoarthritis.22,23 Some studies have reported that arthroscopy combined with HTO can promote cartilage regeneration in patients with knee osteoarthritis.11–13 However, the detailed molecular mechanism of arthroscopy combined with HTO on cartilage regeneration in patients with knee osteoarthritis is still unknown.
The objective of this study was to investigate the effect and possible mechanism of arthroscopy combined with HTO on cartilage regeneration in patients with knee osteoarthritis. Here, the results of arthroscopy revealed the formation of new tissue in the defect area of the medial compartment’s cartilage in the knee joint in 1 year after arthroscopy combined with HTO. In addition, different pathological staining results indicated that the new tissue was cartilage-like tissue. The results illustrated that there was no significant difference of collagen II in the new cartilage tissue, in addition, the p-ERK 1/2 was up-regulated significantly in the patients’ new cartilage tissue.
The mitogen activated protein kinases (MAPK) signal pathway is composed of molecules such as ERK, JNK and p38, 24 which is an important signaling system mediating the cellular response. Among which, Ras-Raf-MEK1/2-ERK1/2 is one of the classical MAPK signal transduction pathway. 25 p-ERK1/2 is the active form of ERK, which can enter the nucleus and activate the transcription factor through phosphorylation. p-ERK1/2 regulates biological function, such cell proliferation and differentiation, by affecting the expression of target genes.26–28 Shakibaei et al. 29 have found that curcumin and resveratrol maintain the differentiation and survival of chondrocytes by activating the ERK1/2 signaling pathway and inhibit interleukin-1β-induced apoptosis. Activation of the ERK1/2 signaling pathway promotes the proliferation of human osteoarthritis chondrocytes, while ERK1/2 specific inhibitors inhibit proliferation of human osteoarthritis chondrocytes. 30 So far, it is unclear whether the ERK signaling pathway is involved in the regulation of cartilage regeneration in knee osteoarthritis patients treated by arthroscopy combined with HTO. In this study collagen II and p-ERK1/2 were highly expressed in the newborn tissue. Our results indicated that arthroscopy combined with HTO significantly increased the expression of p-ERK 1/2 in the new cartilage tissue of patients group. Therefore, we hypothesized that the formation of new tissue may be related to the increased expression of P-ERK1/2. There are still limitations in this study. Firstly, The sample size included in this study was too small. Secondly, the study lacked detailed clinical information and description of patients. Thirdly, only six samples performed western blot and PCR, respectively. Finally, the effect of arthroscopy combined with HTO on cartilage regeneration in patients with knee osteoarthritis was preliminatively investigated, but the potential mechanism was not studied. Considering the key issue, we are collecting cases of osteoarthritis to uncover the effect of arthroscopy combined with HTO on cartilage regeneration in patients with knee osteoarthritis, further function analysis will be performed in our following research.
Conclusion
High tibial osteotomy promote cartilage regeneration by promoting the chondrocyte proliferation induced by activation of the ERK1/2 signaling pathway.
Supplemental Material
Supplemental Material - Arthroscopy combined with high tibial osteotomy promotes cartilage regeneration in osteoarthritis
Supplemental Material for Arthroscopy combined with high tibial osteotomy promotes cartilage regeneration in osteoarthritis by Jiang Wu, Bin Zhao, Wei Luo, Xiao Chen, Qian Zhao, Fuji Ren, Huifeng Zheng and Jingmin Huang in Journal of Orthopaedic Surgery
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This research work was financially supported by the National Natural Science Foundation of China (Grant No. 81501061) and Project of Tianjin Municipal Health Commission (Grant No. ZC20086).
Ethical approval
This study has been approved by the ethics institute of Tianjin Hospital. The signed informed consents of all the participants were obtained.
Supplemental Material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
