Abstract
A five-year-old boy presented with left tibial swelling; there was a typical radiological feature of osteofibrous dysplasia. He was observed until the age of 14 years; there was development of aggressive radiological features. Biopsy revealed chondroblastic osteosarcoma. It was excised and the histology revealed chondroblastic osteosarcoma. A small lesion distal to the main tumour revealed adamantinoma within a background of feature of osteofibrous dysplasia. Proximally, feature of osteofibrous dysplasia was identified focally. The finding of adamantinoma over the distal part of the lesion supports that feature of osteofibrous dysplasia may be a precursor of adamantinoma. They may be a spectrum of diseases with multistep neoplastic transformation. The osteosarcoma may be a result of de-differentiation from adamantinoma. This case is remarkable as the patient was only 14 years old and the tumour showed typical features of osteosarcoma, with feature of osteofibrous dysplasia and adamantinoma found in the same specimen. Feature of osteofibrous dysplasia may be a precursor of adamantinoma, and adamantinoma may dedifferentiate into osteosarcoma.
Introduction
Osteofibrous dysplasia (OFD) is a rare benign fibro-osseous lesion mainly involving the tibial shaft during childhood. It was first reported by Frangenheim in 1921. 1 Adamantinoma (ADM) is a low-grade malignant primary bone tumour with a strong predilection for the tibial shaft. 2 Young adults are most affected. The relationship between OFD and ADM is not clear yet. Whether the former could progress to the latter remains controversial. 3 However, they have similar location, histology, and radiological appearance.
To date, there is no report of OFD with malignant transformation to osteosarcoma, but there are a few papers about adult ADM cases with sarcomatous transformation.4–6
In this article, we present an adolescent case with concomitant OFD, ADM and osteosarcoma of the tibia.
Case report
A 5-year-old boy presenting with a painless left leg swelling for 1 month was referred to our clinic in 2009. On physical examination, there was no local tenderness or skin changes. There was mild anterior bowing of the tibia. Radiographs and computed tomography (CT) scan revealed typical features of OFD of the left tibia (Figures 1 and 2). The child was asymptomatic and radiologically the swelling had the typical appearance of OFD. After discussion with the radiologist, we accepted the clinical diagnosis of OFD without a biopsy. He was managed conservatively with observation. Annual radiographs revealed no significant change for 8 years (Figure 3). Upon follow-up at the age of 14 years, he complained of increasing swelling for 2 months. Radiographs and magnetic resonance imaging (MRI) showed aggressive radiological features (Figure 4). CT-guided needle biopsy revealed chondroblastic osteosarcoma. Positron emission tomography-computed tomography (PET-CT) revealed no evidence of distant metastasis.
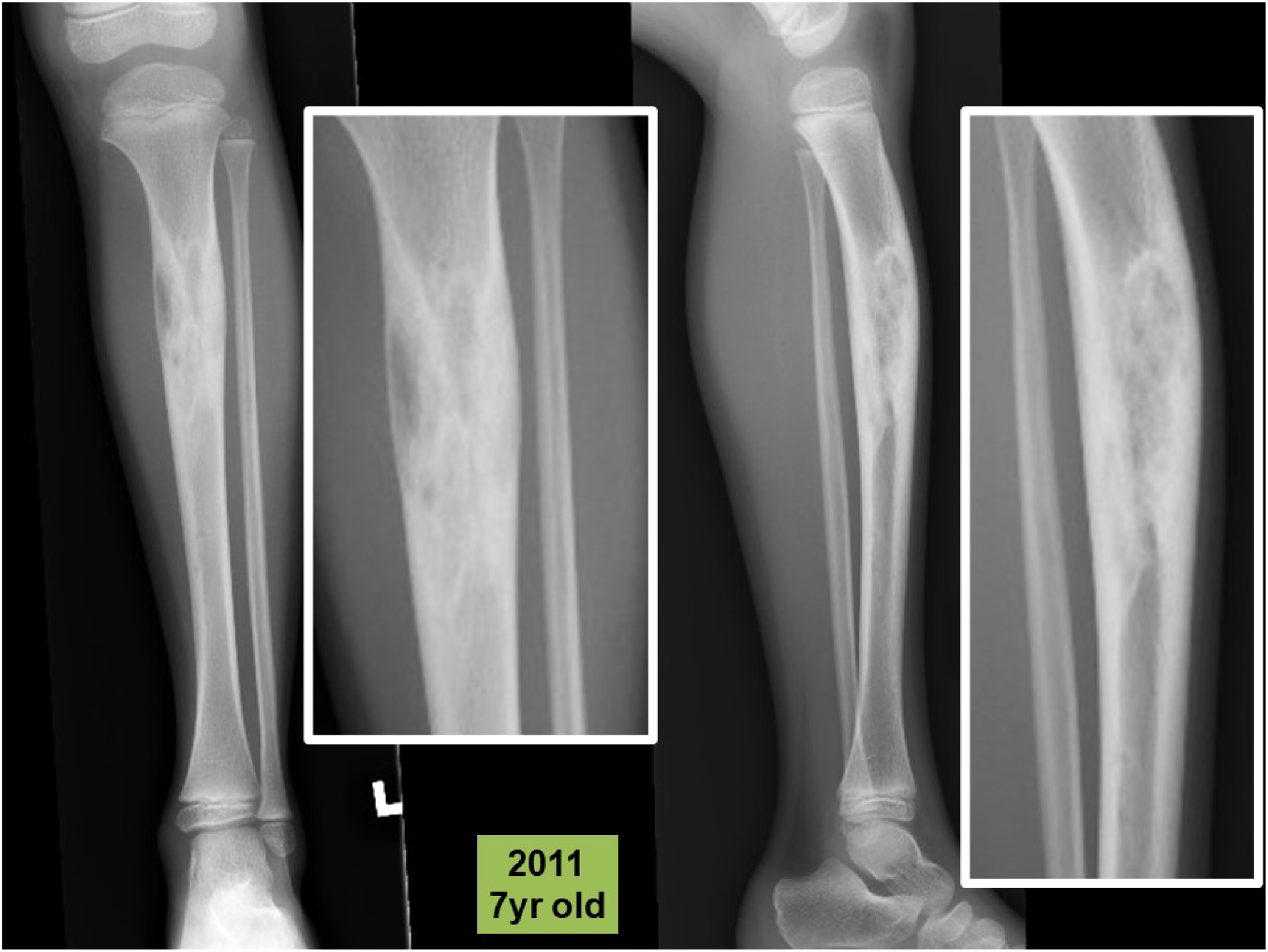
Antero-posterior and lateral radiographs of tibia in 2011 with close-ups (in white frames), showing a mixed lytic and sclerotic lesion of tibia with anterior bowing. It has a narrow zone of transition without periosteal reaction.
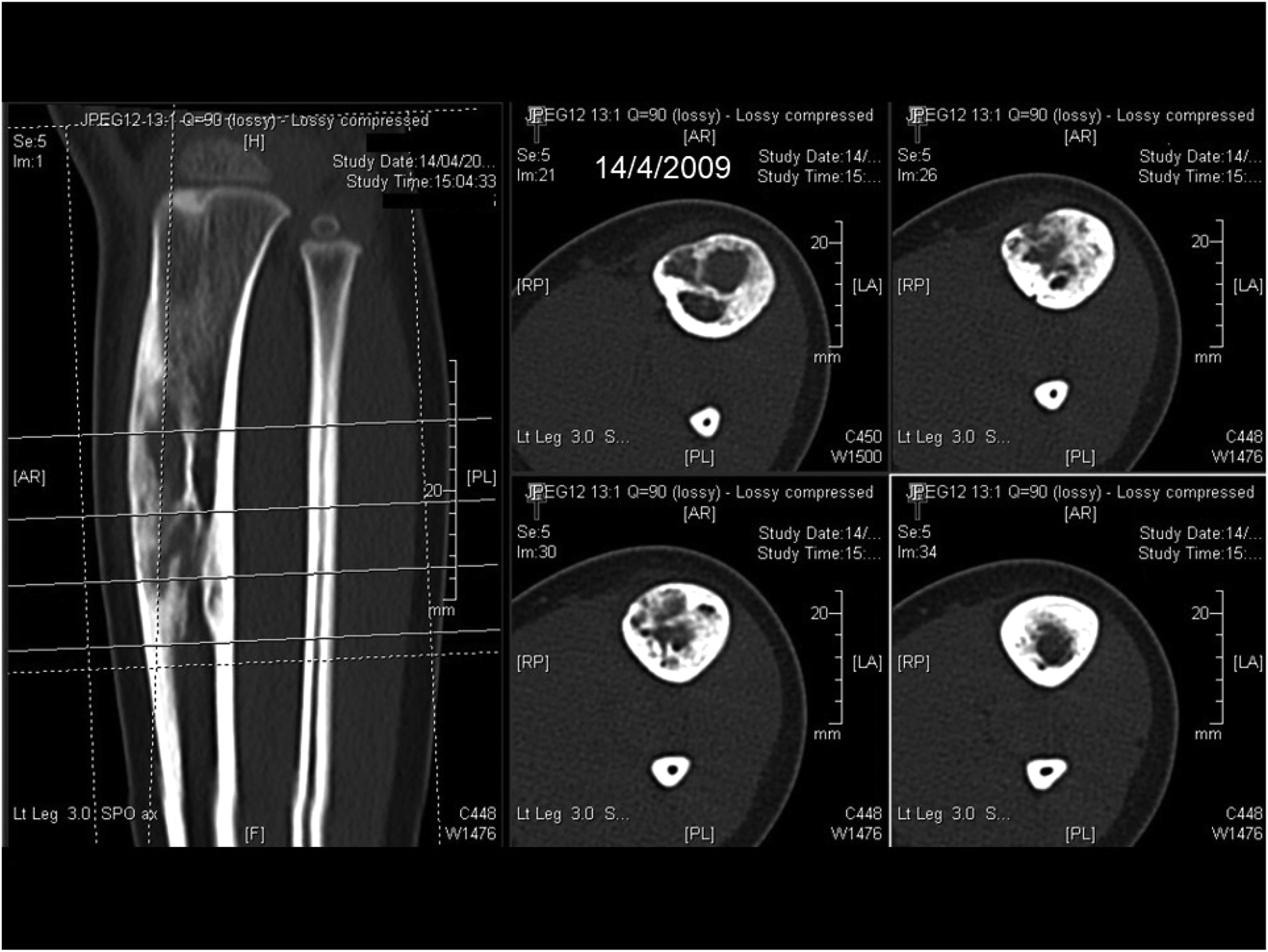
CT of tibia in 2009 showing an expansile intracortical mixed lytic and sclerotic lesion with thickened cortex and marked narrowing of the medullary cavity, suggestive of OFD.
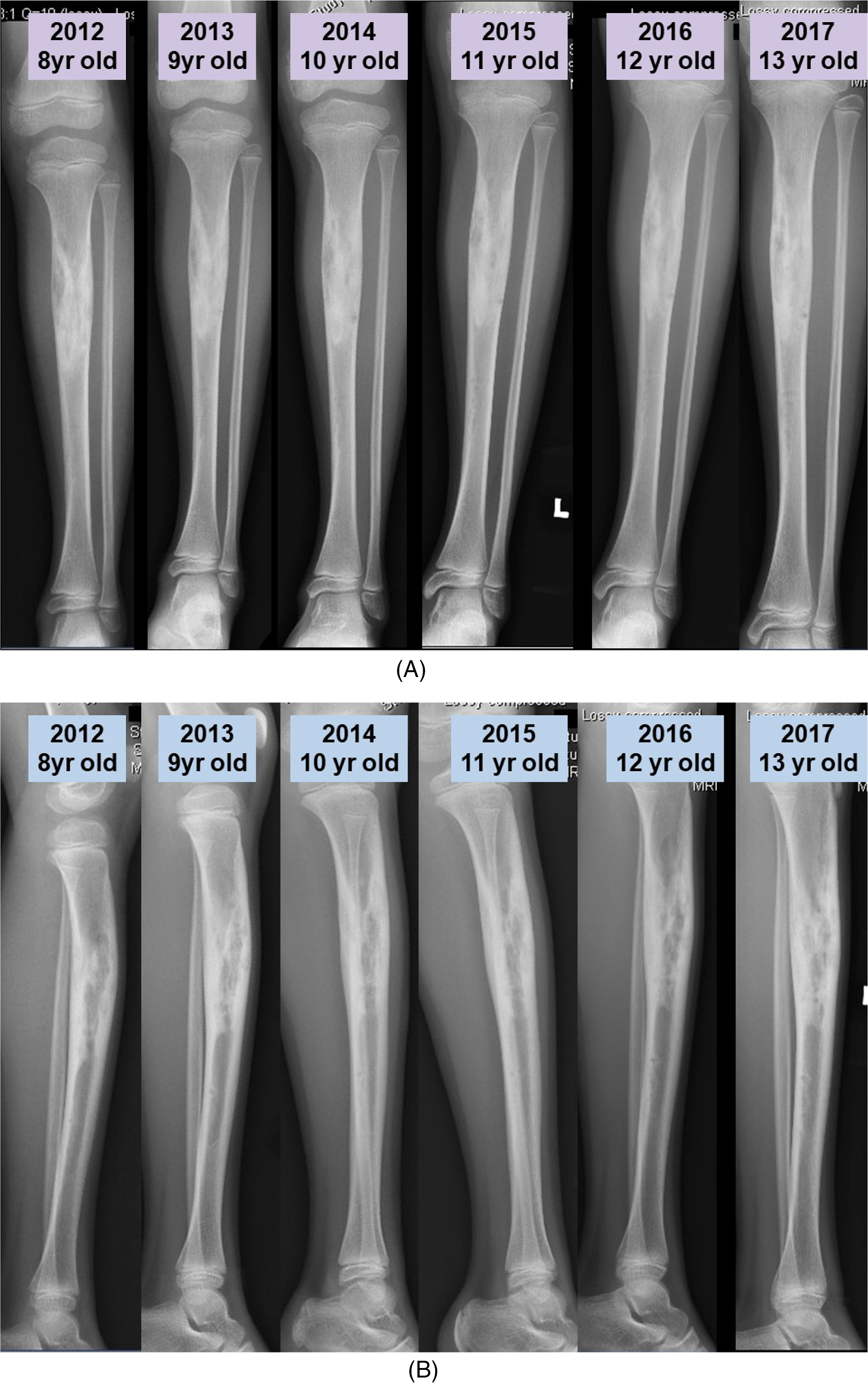
(A) and (B): Annual antero-posterior (AP) and lateral radiographs revealed no significant change before 2018.
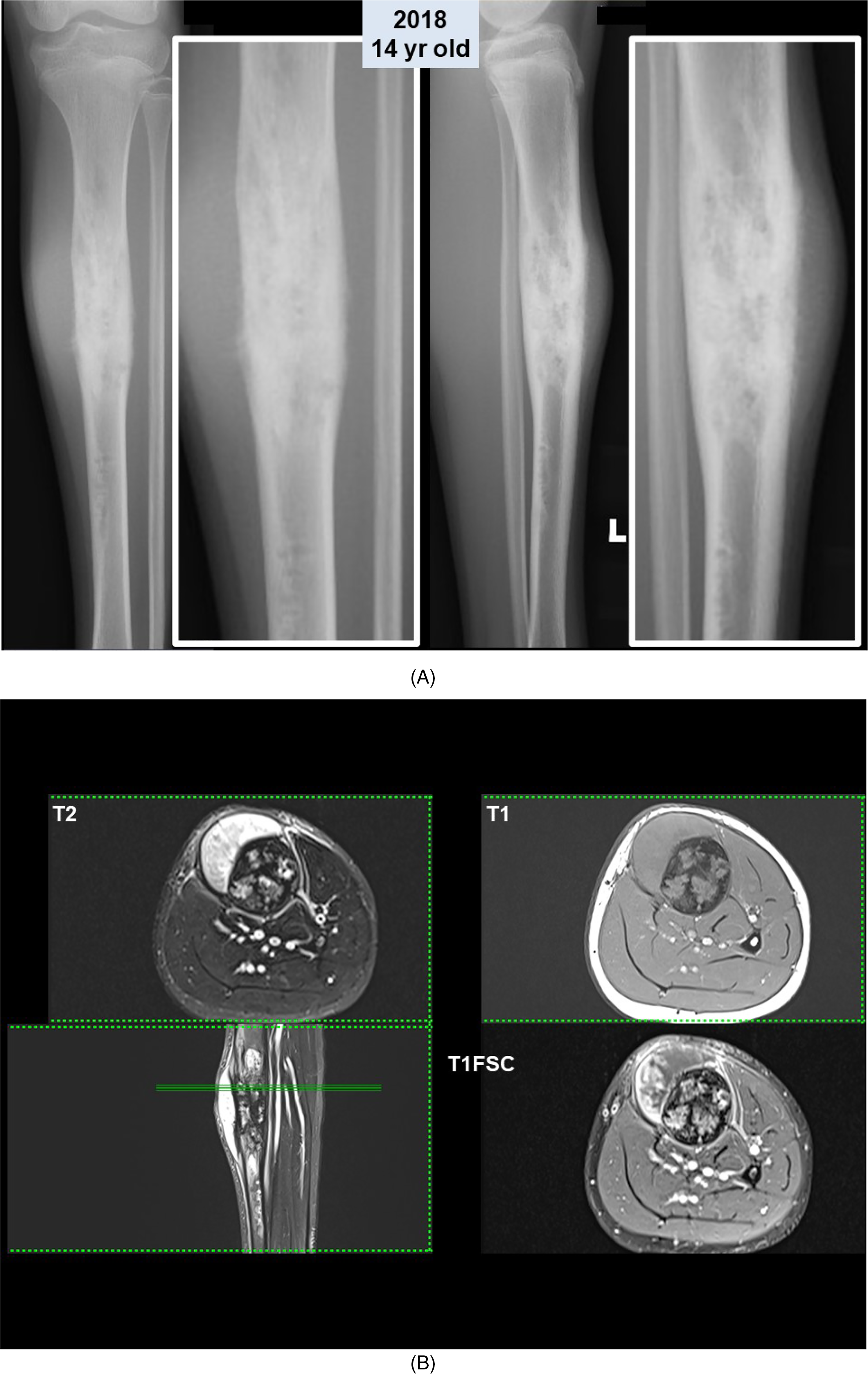
(A): Antero-posterior and lateral radiographs of tibia in 2018 with close-ups (in white frames), revealed aggressive changes with increase in size of the lesion and periosteal reaction. (B) Magnetic resonance imaging (MRI) of the tibial lesion in 2018 revealed a heterogeneously enhancing mass with extraosseous soft tissue components at the proximal tibia. There is irregular cortical thickening with intramedullary contrast enhancement.
The patient was treated with neoadjuvant chemotherapy following the protocol for osteosarcoma by the Hong Kong Paediatric Haematology Oncology Study Group Chemotherapy (Figure 5). Two cycles of high-dose methotrexate, adriamycin and cisplatin were given before the operation. The tumour was excised and reconstructed with a custom-made joint-saver prosthesis (Figure 6). Histologically, the resected specimen is composed of three different areas (Figure 7). The central main tumour bulk revealed high-grade chondroblastic osteosarcoma with 15% of chemo-necrosis (Figure 7B). A small lesion distal to the main tumour revealed features of classic ADM arising within a background of OFD (Figure 7C). Over the proximal part of the lesion, OFD was identified focally (Figure 7A). Immunohistochemically, the tumour cells in osteosarcoma were cytokeratin (MNF116) negative and special AT-rich sequence-binding protein 2 (SATB2) positive, while the tumour cells in classic ADM were cytokeratin positive; isolated cytokeratin positive cells were present in OFD. Postoperatively there was no complication, and the patient completed four cycles of chemotherapy with VP-16, ifosfamide, adriamycin and cisplatin. Follow-up CT thorax at 5 months revealed a new 3 mm nodule in the left lower lobe of the lungs, suspicious for a metastasis. Repeated CT thorax at 6, 8, 11 and 15 months revealed no significant change. He was last seen 1 year 4 months after the operation. There was no evidence of local recurrence. Radiographs revealed the prosthesis in situ. He could walk unaided with an active range of motion of 0–150° of the knee (Figure 8).
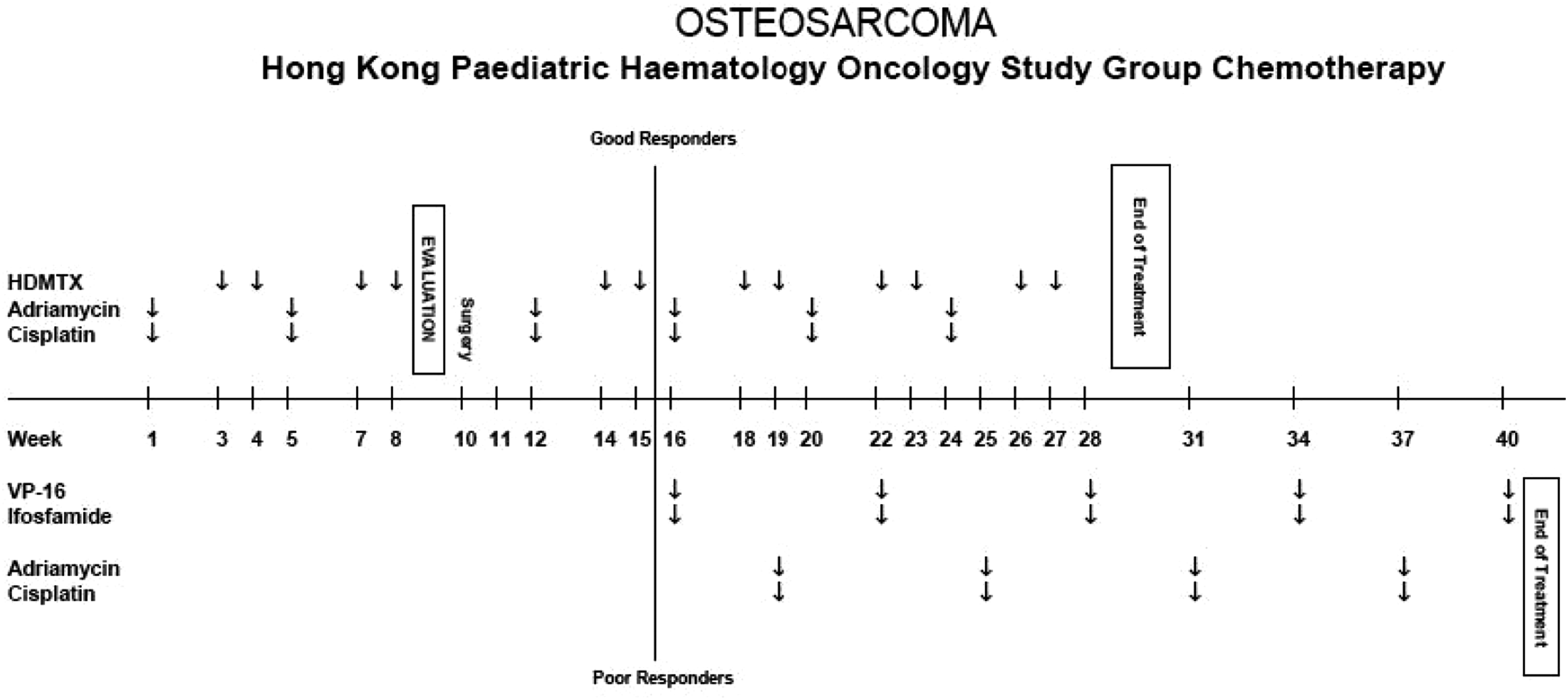
The treatment protocol of osteosarcoma by the Hong Kong Paediatric Haematology Oncology Study Group Chemotherapy.
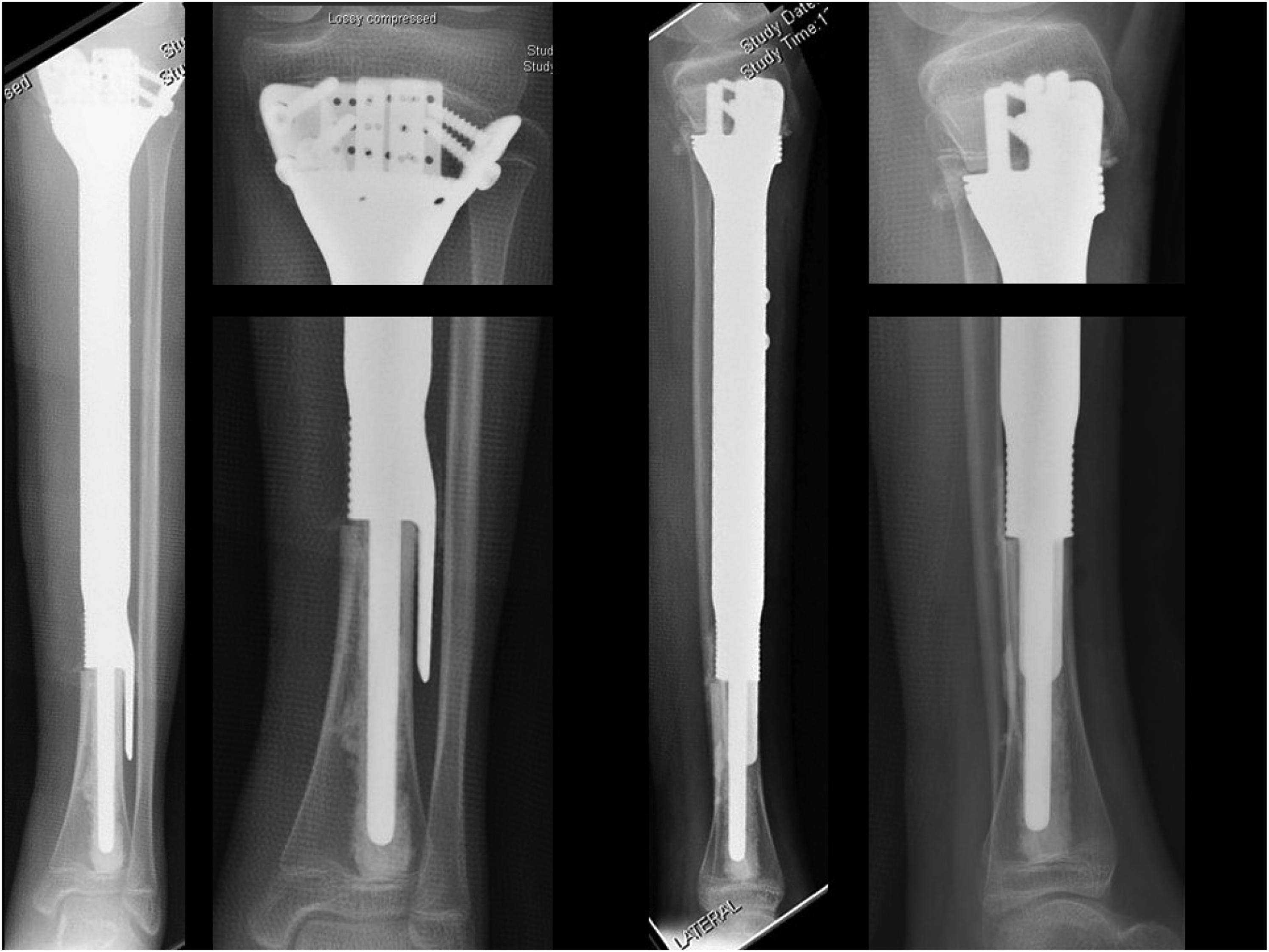
Postoperative radiographs showing the reconstruction of custom-made joint-saver prosthesis.

Photos of the gross specimen, sagittal section after formalin fixation and the sagittal view MRI, showing the corresponding three areas of histology, areas A, B and C, from proximal to distal. (A1) and (A2) Low power and high power views of area A showing OFD. (B1) and (B2) Low power and high power views of area B, showing high-grade osteosarcoma characterised by infiltrative borders with extension into the soft tissue. Most of the tumour shows chondroblastic differentiation, comprising lobules of hyaline cartilage with pleomorphic tumour cells in lacunae. Other areas consist of sheets of plasmacytoid, spindly or stellate-shaped cells, with focal osteoid formation. (C1) and (C2): Low power and high power views of area C showing classic ADM with OFD-like area.
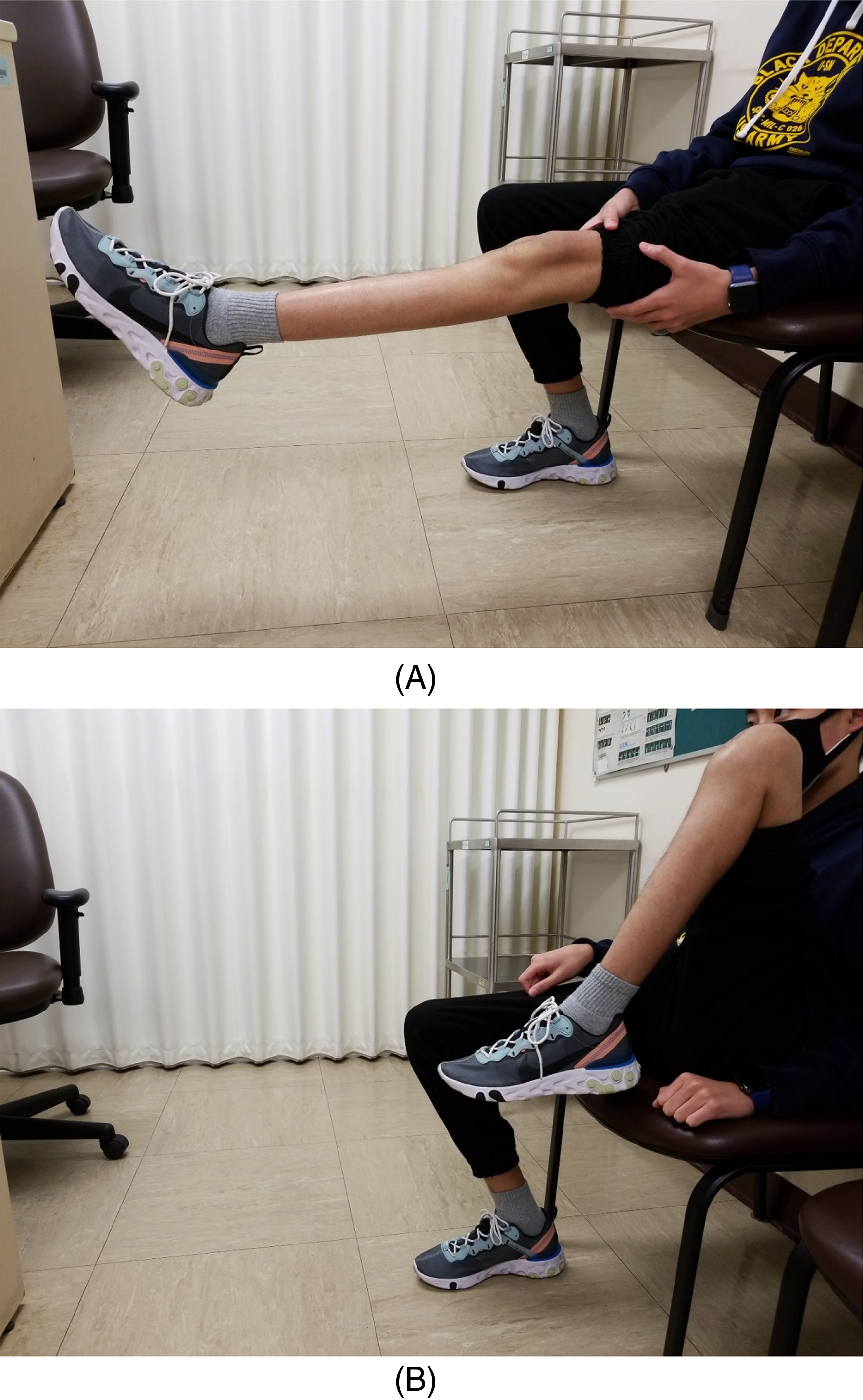
(A) and (B): Clinical photos 6 months after operation showing full extension and flexion of the left knee.
Discussion
OFD generally involves the anterior cortex of the tibial shaft, with osteolysis bordered by sclerotic areas. Patients are usually asymptomatic or present with painless lower leg bony swelling, often with slight anterior bowing. 7 Sometimes, they can present with pathological fracture. 3 Radiologically, it has characteristic bony enlargement, a well-defined intracortical osteolytic lesion often surrounded by sclerosis, with thinning of the cortex or disappearance of the external cortex, and narrowing of the medullary canal. Histologically, it is composed of fibrous tissue enclosing bone trabeculae lined by osteoblasts with a zonal architectural pattern, immature woven trabeculae in the centre and mature lamellar trabeculae at the periphery. Isolated cytokeratin positive cells can be identified in the fibrous stroma immunohistochemically. 8 The management of OFD remains controversial. Some authors suggested no biopsy is required if the radiological findings are typical and the location as well as the age of the patient are considered, 9 while others suggested biopsy is mandatory to differentiate it from ADM. 10 In our case, the patient was asymptomatic and presented at an early age with typical location and radiological features of the lesion; we accepted the clinical diagnosis of OFD without biopsy after discussion with the radiologist. Most authors suggested conservative treatment as OFD seldom progresses and progression halts when patient reaches skeletal maturity.11,12 Scholfield et al. 11 reported 42 cases of OFD diagnosed in childhood, six of which were reclassified as OFD-like ADM after operation or repeat biopsy following a change in symptoms or radiographs. In another case series reported by Lee et al., 1 three out of 16 initially diagnosed OFD cases were confirmed to have ADM after resection; so, they suggested surgical treatment for all OFD cases. Westacott et al. 12 reported the most recent case series in 2019: for 28 cases of OFD in 25 patients, only eight cases required surgery during 8.3 years follow-up. The patients achieved good functional outcome and were satisfied with conservative treatment. There was no transformation to ADM found during the follow-up period. For our case, we closely monitored the clinical symptoms and serial radiographs until there was progression of the lesion.
ADM is a rare low-grade malignant primary bone tumour with an epithelial-like component, sometimes reminiscent of ameloblastoma of the jawbone. It can be found in any age, but more commonly in young adults. It is located in the tibia in 80–90% of the cases2); other locations are the fibula, femur, humerus, ulna and radius. Patients usually present with a painful swelling, sometimes with a history of trauma. Radiologically, it is very similar to OFD, with more aggressive features like significant destruction of the cortex and extension to the medullary cavity and/or soft tissues. Histologically, it is characterised by islands of epithelial cells in a spindle cell stroma. There are different variants depending on the patterns of the neoplastic epithelial cells. These include basaloid, tubular, squamous and spindle cell variants. The epithelial nature can be confirmed by positive cytokeratin in immunohistochemical staining. The treatment is excision with clear margins. Metastasis is found in 15–30% of the cases.13–15 Hazelbag et al. 14 reported 32 cases of ADM with a mean follow-up of 122 months, and an overall rate of metastasis of 29%. They found that intralesional or marginal excision was the most significant risk factor for local recurrence or metastasis (p < 0.001). A large multi-centre international study by Qureshi et al. 15 of 60 cases of ADM reported that wide operative margins were associated with a lower rate of local recurrence than marginal or intralesional margins (p < 0.00005). The 10-year local recurrence rate was 18.6% and the survival rate was 87.2%. In addition to OFD and ADM, an intermediate lesion, OFD-like ADM, or differentiated ADM, has been described. 16 This subtype of ADM is characterised with early presentation within the first two decades, an entirely intracortical location, predominance of an OFD-like stroma and only scattered positivity of epithelial elements for cytokeratin in form of small nests of epithelial cells and tubular structures in the stromal tissue.
The relationship between OFD and ADM remains controversial. 13 They have similarities in location, radiographic appearance and certain histologic features.(17) Czerniak et al. 16 introduced the term ‘OFD-like ADM’ and postulated that it may be an intermediate form between OFD and ADM in a spectrum of diseases with reparative process and regression of tumour tissue. Instead, more authors suggested that OFD may be a precursor of ADM. Hazelbag et al. 14 and Hatori et al. 18 reported cases of progression from OFD-like ADM to classic ADM. The mean age at the time of diagnosis of OFD and OFD-like ADM was lower than that for ADM.7,14 Cytokeratin positive cells are found in OFD, OFD-like ADM and classic ADM, with only isolated cells seen in OFD.8,16 Maki and Athanasou 19 demonstrated that several proto-oncogenes and bone matrix proteins were expressed in both OFD and ADM; so, they believed that OFD and ADM are closely related and OFD could represent a precursor lesion to ADM. Cytogenetic studies also suggested that OFD and ADM are likely related. Clonal chromosomal abnormalities, with trisomies of chromosomes 7, 8, 12, 21 and/or 22, were found in OFD.20,21 Kanamori et al. 22 reported that extra copies of chromosomes 7, 8, 12, 19 and/or 21 were found in seven of eight cases of classic ADM and in two of three cases of OFD-like ADM.
An important objection to the precursor hypothesis is sampling error, as the presence of ADM may be missed initially due to biopsy of non-representative areas of the lesion, especially the periphery of the lesion. 14 Another objection is that the initial histological diagnosis of ADM may be missed, and this is reflected by the change of diagnosis in some cases after complete excision1,11 or upon histological review. 23 Lastly, in a few follow-up studies, there was no progression of disease found.7,8,11
Our case presented at a young age of 5 years. Although no biopsy was taken at that time, the radiological features were typical of OFD. There had been no significant change until the age of 14 years. The finding of areas of classic ADM in the distal lesion supports that OFD may be a precursor of ADM. OFD, OFD-like ADM and classic ADM are closely related lesions, and they appear to show progressive complexity in cytogenetic aberrations, perhaps indicative of a spectrum of diseases with multistep neoplastic transformation from mesenchyme to epithelium..3
Osteosarcoma has been reported to arise in fibrous dysplasia24–26 but is not known to be related to OFD. There is no report of cases with concomitant OFD, ADM and osteosarcoma, but there are 11 cases of ADM with sarcomatous change reported in the literature.4–6,27 Hazelbag et al. 5 first described three ADM cases with loss of epithelial differentiation of tumour cells as ‘dedifferentiated’ ADM in 2003, with deposition of chondroid and osteoid matrix observed. The term ‘dedifferentiation’ means loss of epithelial differentiation and replacement by a sarcomatous component, similar to an epithelial–mesenchymal transition. They believed that it was a subtype of ADM. All these dedifferentiated ADM cases were adults. Our case is the youngest case reported so far. The diagnosis was made when he was fourteen, and there was presence of OFD, ADM and osteosarcoma in the same specimen. The young age of presentation means less time available for the development of the dedifferentiation process. The precise prognosis of this disease is unknown because there are only few cases reported in the literature. Of these 11 reported cases, six died of the disease. 6 For our case, upon follow-up at 1 year 4 months, there was no evidence of local recurrence, and postoperative CT thorax revealed a new small lung nodule with no progression.
The relationship between OFD, ADM and dedifferentiated ADM is not clear yet. In our case, this co-existence of the diseases may support the hypothesis of OFD being a precursor of ADM, and they may be part of a spectrum of diseases with multistep neoplastic transformation. ADM may dedifferentiate into osteosarcoma. OFD not only may progress to a low-grade malignancy like ADM, but may further transform to a high-grade one like osteosarcoma. While these related conditions are similar clinically, radiologically and histologically, their disease courses and treatments are different. It is important to carefully differentiate them for appropriate treatments, and to closely monitor the patients with OFD and educate them about the possibility of malignant transformation.
