Abstract
Study Design
Case report and literature review.
Objective
To present the first case of metastatic adamantinoma of the spine with immunohistochemical confirmation and an updated literature review.
Summary of Background Data
Spinal metastatic disease could be difficult to diagnose because of the multiple differential diagnoses involved. Spinal surgeons must be aware of unusual primary extra-axial tumors that metastasize to the spine because in certain cases the primary surgery must determine the prognosis of this lesion.
Methods
We present a fully documented case of a middle-aged man with tibial adamantinoma who developed spine metastasis, confirmed by immunohistochemistry. A literature review was done.
Results
Based on clinical, imaging, and pathology findings, we provide evidence for the first proven metastatic adamantinoma of the spine, adding this condition to the long list of differential diagnoses of secondary spinal disease.
Conclusions
Adamantinoma is a very rare bone tumor representing ∼1% of primary bone tumors. Spinal metastatic adamantinoma with immunohistochemical confirmation has not been described previously. Due to the lack of specific image findings or serum markers and multiple differential diagnoses, biopsy with immunohistochemical confirmation is mandatory, because “en block resection” might prove to be curative treatment.
Introduction
We present the first case of metastatic spinal adamantinoma with immunohistochemical confirmation and an updated literature review regarding epidemiology and natural history, differential diagnosis, prognosis, and treatment options.
Adamantinoma is a rare malignant bone neoplasm of epithelial origin. It represents ∼1% of primary bone tumors. 1 It has been observed primarily in long bones and mainly in the tibial shaft (80 to 85%). Multiple osseous locations may represent synchronously or metachronously and may be primary tumors or metastases. 1 , 2
Because the tumor can manifest in so many varied ways, there are no specific findings on plain X-rays or computed tomography (CT) scan. To get a representative sample for study, multiple biopsies are usually performed. Immunohistochemistry is mandatory to confirm the diagnosis.
Adamantinoma is an aggressive tumor that grows very slowly, and it may metastasize. When therapy is inappropriate, it may recur; and with time, the recurring neoplasm may acquire the appearance of sarcoma. Metastases have been observed in 15 to 20% of patients, most frequently in the lungs and local lymph nodes. 3 , 4 , 5 , 6 Occasional liver metastases have been described. 1 Skeletal metastases are rare, and to date, immunohistochemically confirmed metastatic adamantinoma of the spine has not been described in the existing literature.
We present the first report of a metastatic adamantinoma of the spine with immunohistochemically confirmed diagnosis.
Case Report
A 37-year-old previously healthy man presented at another institution complaining of progressive pain in his left leg. Imaging studies showed a lytic lesion in the tibial diaphysis (Fig. 1A and B). Positron emission tomography scan was negative for other lesions. Complete oncologic workup showed no evidence of secondary disease.
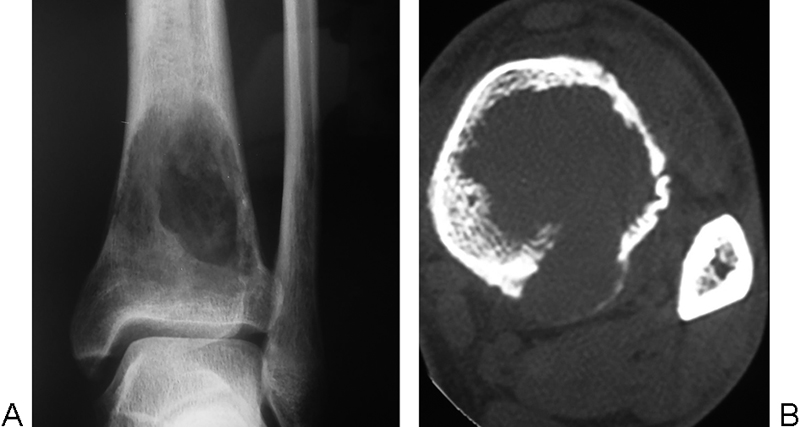
(A) Plain radiograph and (B) multislice computed tomography axial image showing a lytic lesion involving the distal tibial metaphysis, with destruction of the posterior cortex and mild periosteal reaction.
He underwent an intralesional resection and the histology revealed adamantinoma. The patient developed local recurrences, with a second curettage and finally, below-the-knee amputation.
The patient remained asymptomatic and disease-free for the next 5 years, when he developed a pathologic fracture at the proximal femur, which was treated with en bloc resection and hip replacement with a reconstruction prosthesis. The histology confirmed metastatic adamantinoma.
Shortly after this episode, the patient developed a solitary lung nodule, treated with segmental resection, and a pathologic fracture at the proximal humerus, treated with wide resection and reconstruction prosthesis. Confirmation of secondary adamantinoma was made followed by cyclophosphamide chemotherapy. The patient was referred to our hospital 8 years after the initial diagnosis with progressive worsening of lumbar and right sciatic leg pain but no motor deficits or loss of bowel or bladder control.
Imaging revealed a lytic lesion at the sacrum, involving mainly the right sacral ala, extending to the contralateral side and to the L5 vertebra. There was invasion of the canal and lumbosacral nerve root compression.
After discussing with the patient the possible benefits and complications of nonoperative versus operative treatment, he underwent wide posterior decompression, intralesional resection, and cementation of the sacrum, achieving complete relief of pain without neurologic deterioration (Fig. 2A, B, and C).
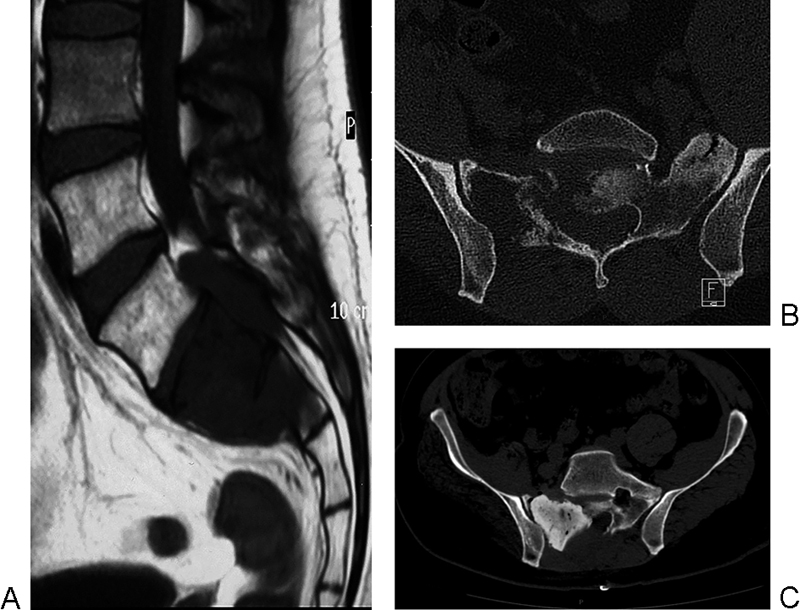
(A) T1-weighed magnetic resonance image (1.5 T) and (B) multislice computed tomography (CT) scan showing extensive lytic involvement of the sacrum, with significant involvement of the spinal canal. (C) Postoperative multislice CT image depicting decompression, debulking, and cement augmentation of the right sacral ala.
Histologic analysis of the tissue samples from the sacrum, as well as those from the previously resected lesions, were reevaluated using immunohistochemistry and PAP standard procedures for cytokeratin, vimentin, and factor VIII. All samples revealed a high-grade spindle cell sarcoma. Given the long-lasting history, the original histology, and the positivity for epithelial cell keratins (CKs) 5, 14, and 19 and vimentin and negativity for factor VIII, all were interpreted as high-grade dedifferentiated adamantinoma (Fig. 3A, B, and C).
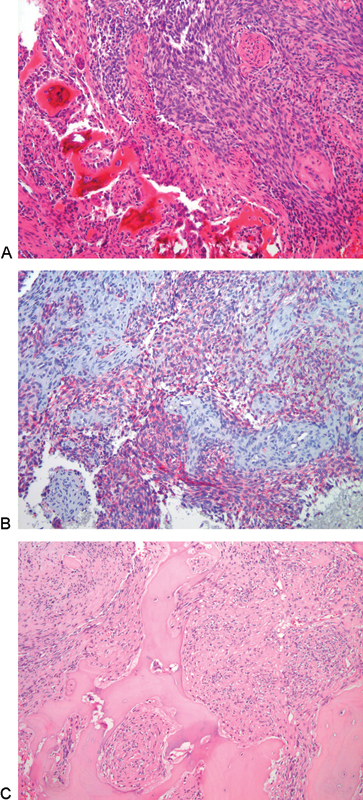
(A) Photomicrograph of the original tibial tumor. Fascicles of spindle cells with elongated slightly pleomorphic nuclei infiltrating host bony trabeculae. The pattern resembles features seen in monophasic synovial sarcoma and fibrosarcoma (hematoxylin-eosin, 10×). (B) Nests of keratin-positive cells in a fibrous stroma (cell keratins 5,14,19; 10×). (C) Photomicrograph of the sacral lesion. High-grade spindle cell sarcoma diffusely infiltrating the host bony trabeculae (hematoxylin-eosin, 10×).
Fluorescence in situ hybridization (FISH) on formalin-fixed, paraffin-embedded tissue sections was performed for all samples. They were negative for TLE1 expression and for CD99, excluding synovial sarcoma and Ewing sarcoma.
At 24-month follow-up after having undergone two cycles of adriamycin-cisplatin chemotherapy, the patient is clinically asymptomatic, and imaging reveals no further spread of the disease.
Discussion
Epidemiology
This tumor was named “primary adamantinoma of the tibia” by Fischerl in 1913 only after exclusion, for one or more reasons, of several known entities. 4 Although Szendroi et al published a review about the management of adamantinoma, 7 because the tumor is rare, most of the literature is composed of retrospective case reports and small case series, which have limited follow-up 8 , 9 , 10 and small numbers of patients, 11 retrospective studies, or case report presentations.
The tumor occurs in patients between their second and fifth decades of age, and the ratio tends to be higher in women (5:4). 12 , 13 Preexisting trauma is related in ∼60% of cases. 14 According to Naji et al, “The most common presentation is a gradually evolving mass associated with dull, insidious, aching pain located in the mid-tibial shaft.” 15
As stated previously, the tumor has prevalence for long bones with the tibia being the most common one involved (80 to 85%). All other major limbs can be involved, including (in order of decreasing frequency) the humerus, ulna, femur, fibula, radius, innominate bones, ribs, and rarely small bones of the hands and feet, with only a few cases reported in short bones. 16 , 17 , 18
Exceptional cases of skeletal metastasis from adamantinoma have been reported. Kanakaraddi et al reported a patient who developed a skeletal metastasis 4 years after resection of the primary tumor. Yet, there was no evidence of recurrence at the primary site or secondary metastasis to the lungs. 2
Although there have been six cases of the so-called primary spinal adamantinoma reported in the literature, all lack immunohistochemical confirmation. And until now, there have been no reports of any case of metastatic adamantinoma to the spine.
Nerubay et al described a case of primary adamantinoma of the spine that developed humerus metastasis. The diagnosis was confirmed by three independent pathologists, but there was no immunochemistry to confirm the diagnosis. 3
Mandibular adamantinoma metastasis to the lung has been described in the literature. Dini et al first described a metastatic adamantinoma of the spine arising from a mandibular location. 18 Immunohistochemistry ruled out a Ewing sarcoma, but the article does not show any specific immunohistochemical reaction for adamantinoma.
In the literature, there are even descriptions of cases of “adamantinoma” with negative cytokeratin or vimentin markers, adding confusion to the presentation and diagnosis of this unusual tumor.
Differential Diagnosis
Due to the rare nature of this tumor, prognosis based on wide or en bloc resection, differential diagnosis, and limited experience with this tumor, immunohistochemistry has become a standard procedure in analyzing bone biopsies. 19 The absence of previous diagnosis or consistent history, emergency cases with spine instability or neurologic compromise, the absence of specific radiologic pattern, and the limited experience with adamantinoma all make its treatment challenging, with en bloc resection the gold-standard treatment.
Intraosseous synovial sarcoma, although rare, is a well-documented entity, and metastatic disease would not be unexpected. According to Hazelbag and Hogendoorn, “TLE1 is a highly specific biomarker for synovial sarcoma in the setting of differential diagnosis of unclassified high-grade sarcomas.” 20
The identifying characteristics of the Ewing sarcoma family of tumors are malignant, small, round, and blue cell tumors of the bone and soft tissue and membranous expression of the CD99. 21 Alternative diagnoses include aneurysmal bone cyst, chondromyxoid fibroma, chondrosarcoma, chordoma, fibrous dysplasia, hemangioendothelioma, osteofibrous dysplasia, eosinophilic granuloma, nonossifying fibromas, epithelial metastases, or simple bone cyst. 15
There is continuous evolution of the connection among classic adamantinoma, differentiated adamantinoma (osteofibrous dysplasia–like adamantinoma), and osteofibrous dysplasia (OFD). The three lesions show proclivity for tibial diaphysis, express cytokeratin (at least focally), and show similar cytogenetic findings. 22 The origin of these entities continues to be controversial. Along with reports of progression from OFD and differentiated adamantinoma to classic adamantinoma, some authors suggest that in fact these are precursor lesions. 14 This suggestion may be truer to the clinical presentation because the lesions caused by both OFD and differentiated adamantinoma are smaller and intracortical and can be observed in a patient population younger than that of classic adamantinoma.
Sarcomatous dedifferentiation of the epithelial component has been described by Hazelbag and Hogendoorn. 22 The rare complication can be detected as areas of frank sarcomatous change in the setting or clinical history of classic adamantinoma.
The usual appearance of chordoma is that of a midline axial lesion in adults over 50; 90% of cases have been observed in the sacrococcygeal area or base of the skull. On imaging studies, the appearance is that of a lytic lesion. Chordoma typically expresses cytokeratins AE1/AE3, CK8, CK19, and CK5, even if they are, at best, only focally positive for CK7 and CK20. 21
Both carcinomatous and noncarcinomatous metastases are relevant at the diagnostic stage in epithelioid bone lesions. It is well known that common metastases include tumors in breast, prostate, lung, kidney, and thyroid.
Image Findings
Plain X-rays, CT, and magnetic resonance imaging (MRI) may all be used to help evaluate a suspected adamantinoma. However, there are no specific findings in the differentiation of an adamantinoma from any other lytic lesion. Soft tissue involvement is usually associated with advanced or recurrent lesions. 21 , 23 , 24
According to Van Geel et al, 5 the most common radiographic appearance is a central or eccentric multilocular mass, sharply or poorly demarcated radiolucent lesions surrounded by areas of dense, and sclerotic bone that can cause fractures. In long bones, the mass is found in a diaphyseal location, although metaphyseal extension of lesions or isolated involvement of a metaphysis may be seen occasionally. The lesion is commonly intracortical but destroys cortex and invades extracortical soft tissues in ∼15% of cases.
Imaging findings using CT and MRI are usually not specific. According to Szendroi et al, “CT scan shows cortical involvement and soft tissue extension when it exists. However, it does not show intraosseous extension of the tumor. CT does play a role in detecting pulmonary metastases and, as such, plays a role in the routine work-up.” 7
When planning reconstructive surgery, MRI becomes a key study in determining tumor-free margins and intramedullary extension of the tumor, as well in detecting distant cortical foci.
Pathology and Immunohistochemistry
Due to the various manifestations of the tumor, a sample study is obtained only after multiple biopsies. Some adamantinomas show Ewing sarcoma–like tumor cells, and in others, peripheral zones are characterized by tissue similar to that of fibrous dysplasia or ossifying fibroma. 20 , 23
Hazelbag et al showed that “classic adamantinoma is a biphasic tumor characterized by epithelial and osteofibrous components that may be intermingled with each other in various proportions and differentiating patterns.” 24 In some cases, nuclear atypia has been observed. 25 “Mitotic figures are usually infrequent, most reporting 0–2 mitoses per 10 high power fields.” 2
Sarita-Reyes et al affirm that “regardless of histologic subtypes, all adamantinomas are uniformly positive for keratins 14 and 19. Immunohistochemically, the epithelial cells show co-expression of keratin, especially basal epithelial cell keratins (CKs 5, 14 and 19) and vimentin.” 27
Laboratory Findings
Adamantinoma does not have any specific blood markers. Serum calcium, phosphorus, and alkaline phosphatase are usually normal. However, “severe paraneoplastic, humorally mediated hypercalcaemia, hypercalcaemic coma, and pancreatitis have also been reported.” 6
Recommended Treatment Techniques and Outcomes of Treatment
Adamantinoma is an aggressive tumor that grows very slowly and may metastatize. 28 When therapy is inappropriate, it may recur; and with time, the recurring neoplasm may acquire the appearance of sarcoma. 3 , 4 The main risk of local recurrence or metastasis is directly dependent on the success of the initial index surgical excision of the adamantinoma. Even if it is difficult to determine exact mortality statistics due to the rarity of this tumor, the expected 10-year mortality rate is 10 to 18%. 29 Pulmonary metastasis is the most common cause of death. 5 There have not been probed relevant relationships between survival time and stage (p = 0.058), duration of symptoms, gender, biopsy type (open incisional versus open excisional), wide operative margins, or reconstructive surgery performed. Late metastasis has been observed in 10 to 30% of patients, usually in the lungs, the regional lymph nodes, or the bones. 30 , 31 Rarely, liver metastasis has been described.
Metastatic lesions may be composed of sarcomatous spindle cells or malignant epithelial cells. 7 Lung metastases are reported in 10 to 20% of cases after many years of curative treatment, 32 and most include only the epithelial component of the tumor. Cohn et al reported a patient who had lung metastasis of adamantinoma 16 years after disarticulation of the knee, indicating that the aggressiveness of this tumor may be greater than has been thought. 30 The local recurrence ratio is ∼18%, 9 , 13 , 33 even for en bloc resections with wide margins. Unfortunately, neither radiotherapy nor chemotherapy have proved to be effective in the treatment of this tumor; the only two reports in the literature of lung metastases that responded to treatment used tyrosine kinase inhibitor or chemo- and radiotherapy. 34 .
Among the reported risk factors connected to metastases, suggestions include male gender, pain, symptoms over a period shorter than 5 years, and histologic traits of squamous differentiation. Yet, these factors have not been proved. Even if some neoplasms may metastasize after years of primary therapy, it is still hard to determine clinical, radiographic, or histologic traits that might be useful in predicting reliably the ultimate behavior of any type of tumor. 35
Conclusion
To date, the data published in the literature about adamantinoma of the spine lack immunohistochemical confirmation for a former diagnosis. We present the first documented case of metastatic spinal adamantinoma, adding this condition to the long list of differential diagnoses in metastatic spinal disease.
Spinal metastatic adamantinoma looks like a very unusual differential diagnosis; however, in the presence of clinical history or image findings, it must be considered because of the possibility of curative en bloc resection.
We strongly recommend bone biopsy with immunohistochemistry and FISH analysis to assess the diagnosis and, based on that, to proceed to definitive surgical treatment.
Disclosures
Ruben A. Morales Ciancio, none
Alessandro Gasbarrini, none
Stefano Boriani, none
Marco Gambarotti, none
