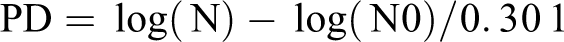Abstract
Background:
Current research in cartilage repair is focusing on the use of soluble factors released by cells during expansion, either as stand-alone therapy or as conditioned media (CM) supplements to optimize cellular phenotype for in-vivo transplantation. The present study aimed at utilizing spent media from cultured human articular cartilage derived chondroprogenitors and assessing their influence on bone marrow mesenchymal stem cell (BM-MSC) growth and phenotype.
Methodology:
CM was collected from passage 2 chondroprogenitors and evaluated for latent TGFβ1 levels. Passage 3 BM-MSCs were divided into two groups and cultured with either a) standard expansion medium (EM-group) or b) EM supplemented with 50% CM (CCM-group). At sub-confluence both groups were assessed for population doubling, migration assay, cell surface markers, gene expression for chondrogenic and hypertrophy markers. Additionally, pellet cultures were subjected to chondrogenic differentiation and analyzed by Alcian blue stain.
Results:
On analysis of proliferation and migration, CCM-group showed comparable results to EM in terms of population doubling and cell movement toward scratched area. Similarly, use of spent medium did not affect the surface protein or gene expression profile of BM-MSCs with similar flow cytometric and mRNA results seen in both groups. Glycosaminoglycan deposition (Alcian blue) was seen in the CCM-group, comparable to the EM-group.
Conclusion:
This preliminary report provided valuable information on the influence of unfractionated CCM on BM-MSC characteristics which may be further optimized for cartilage regeneration by the use of purified components such as exosomes, micro-vesicles, and concentrated trophic factors in future.
Introduction
Hyaline cartilage regeneration still remains a challenge in the field of cartilage repair. Cell-based repair of cartilage related pathologies commonly employs bone marrow derived mesenchymal stromal cells (BM-MSC) and chondrocytes. 1,2 As BM-MSC harvest is minimally invasive and their expansion results in high yields with minimal loss of phenotype, it serves as a versatile cell for cyto-therapeutics. However, a higher propensity for hypertrophy resulting in mixed fibro-hyaline cartilage production leads to inferior biomechanical properties in-vivo. 3 –5
Articular cartilage derived chondroprogenitors have been investigated as a potential cell therapy module for osteoarthritis and osteochondral defect repair. 6 –8 Conforming to the minimal criteria specified by International Society for Cellular therapy 2006, progenitors have been classified as MSCs, while also displaying reduced hypertrophy markers and higher potential for chondrogenesis. 9,10
Recently, much focus has been allotted to soluble factors obtained from stem cell expansion namely exosomes, micro-vesicles, and apoptotic bodies extruded into the culture medium, which can be collected and utilized to influence and enhance the regenerative potential of various tissues. 11 Paracrine mediator like activity has been demonstrated by conditioned media (CM) derived from BM-MSCs and chondrocytes, in studies shown to promote regeneration and retard degeneration in osteochondral injury. 12 –15 Additional advantages of exploring the use of these alternatives to cellular therapy are ease of manufacturing, transport and their acellularity (which renders them potentially hypo-immunogenic). Interestingly, chondroprogenitors have been reported to secrete trophic mediators such as TGFβ when cultured, while another study utilized progenitor derived extracellular vesicles as intraarticular injections to demonstrate attenuation of OA progression. 16,17 Thus far, evaluation of the influence of chondroprogenitor derived CM (CCM) on the biology of BM-MSCs when expanded has not been explored. The aim of the present study was to determine if CCM from cultured chondroprogenitors could promote chondrogenesis and reduce hypertrophy of BM-MSCs when cultured.
Materials and methods
Study design
Following Institutional Review Board approval, the study was performed according to the Ethics Committee rules. Human knee joint samples and surgical site bone marrow aspirate were obtained from three patients (average age: 58 ± 12 years), admitted for knee replacement surgery for severe osteoarthritis (OA). All samples were acquired subsequent to obtaining a written informed consent. Our consideration for inclusion involved essential degenerative OA with grade 4 Kellgren-Lawrence score. Joints were excluded if pathologies identified with inflammation, infection and post traumatic causes were suspected. BM-MSCs and chondrocytes were isolated from bone marrow aspirate and articular cartilage shavings, respectively. Chondroprogenitors were selected after subjecting chondrocytes to differential fibronectin adhesion assay. Additionally, the expansion media used to grow chondroprogenitors was collected, evaluated for human latent TGFβ1 levels, and used as the CM supplement for BM-MSCs. Thus, the two study groups included passage 3 BM-MSCs, cultured either with standard expansion medium (EM-group) or medium containing CCM at 50% strength and 50% standard EM (CCM-group). At confluence, BM-MSCs were comparatively analyzed for population doubling, migratory potential, cell surface marker expression using fluorescence assisted cell sorting, mRNA expression and chondrogenic differentiation potential (Figure 1).
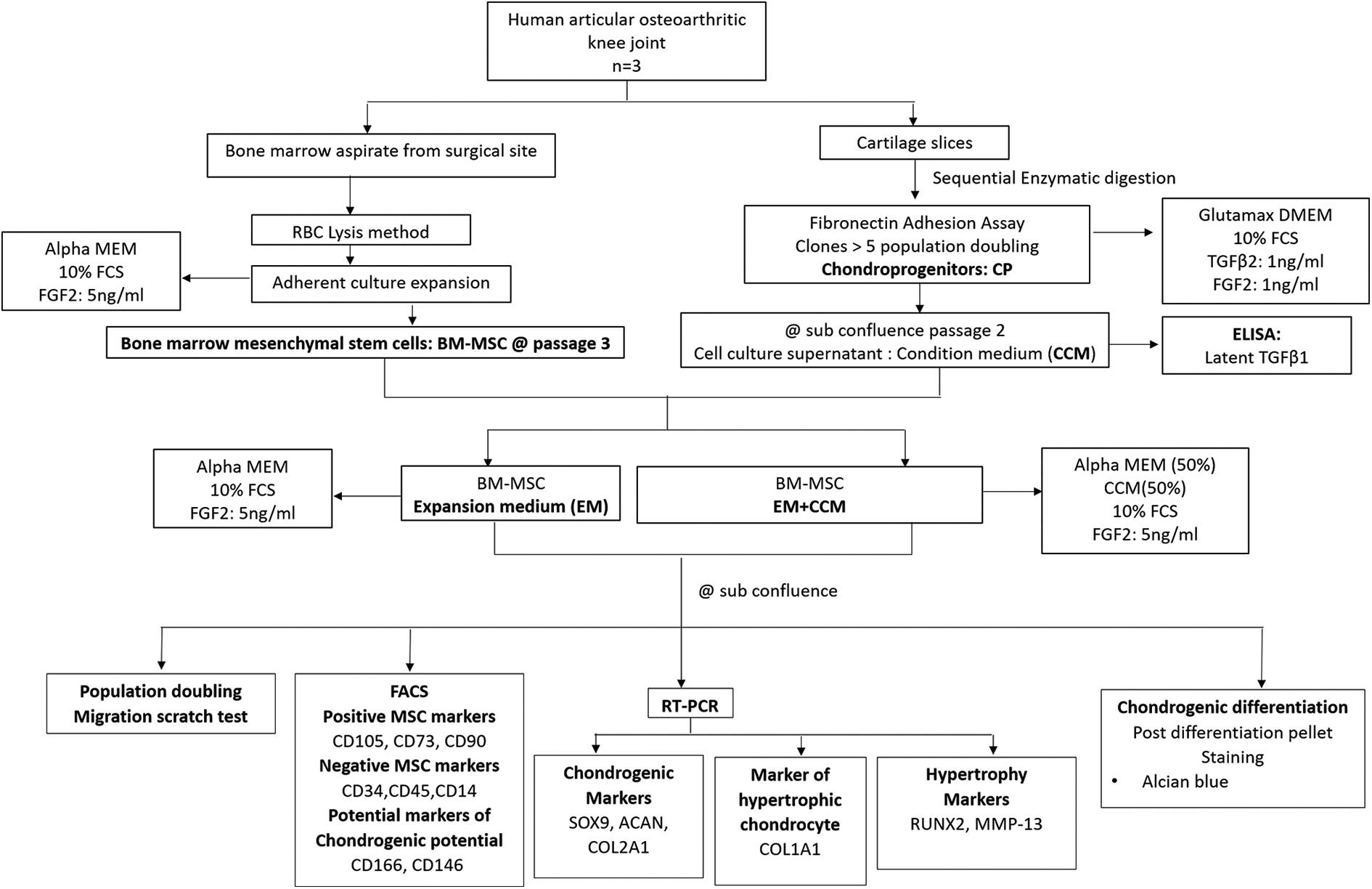
Study algorithm outlining the two culture conditions used for comparison, namely BM-MSCs grown in standard expansion medium (EM) or EM supplemented with 50% condition media (CCM-group). Samples were taken from three donors in each group. BM-MSCs: bone marrow mesenchymal stem cells, CP: chondroprogenitors, FACS: flow cytometric analysis, CD: cluster of differentiation, FCS: fetal calf serum, TGF: transforming growth factor, SOX9: sex determining region Y-box 9, ACAN: Aggrecan and COL2A1: Collagen type 2A1, COLA1A1: Collagen type 1A1, RUNX2: Runt related transcription factor-2 and MMP13: Metalloproteinase type 13.
Cellular isolation and culture
BM-MSC: Red blood cell lysis (RBC) buffer method
The collected bone marrow aspirate was added to RBC lysis buffer solution (1:5 dilution) and centrifuged for 5 minutes at 480 g. Supernatant containing the lysed RBCs was discarded and the pellet resuspended and loaded on culture-ware. Following a wash at 24 hours, adhered cells were visualized and further expanded using a basal medium which comprised of Minimum essential medium Alpha Eagle(α-MEM, Lonza) containing 10% fetal calf serum (FCS), 0.1 mM ascorbic acid and 5ng/mL human fibroblast growth factor 2 (bFGF-2, Biovision). Cells were grown to confluence (loading density: 5000 cells/cm2 each subsequent passage) under standard culture conditions (humidified chamber with 5% CO2 at 37°C) and expanded to passage 3.
Chondroprogenitors and conditioned media
In order to isolate chondroprogenitors, released chondrocytes were seeded onto pre-coated fibronectin plates (10µg/ml) as described by Nelson et al. In brief, 4000 chondrocytes were loaded on to fibronectin coated plates (9.3 cm2) and allowed to incubate for 20 minutes. Following this, medium containing non-adherent cells was removed, and adherent cells were maintained in culture for a period of 12–14 days (till colony formation was achieved). Subsequently, polyclones from each well were transferred to a T-25 flask and expanded to confluence (seeding density 5000 cells/cm2 at each subsequent passage). The expansion medium comprised of Glutamax DMEM-F12 with 10% FCS, 0.1 mM ascorbic acid, 10 mM HEPES, 1ng/ml TGFβ2 and 5ng/ml FGF2. At passage 2, the medium used to grow the cells was collected every 3 days (on the day of medium change), centrifuged to remove cellular debris, and stored at −80°C till further use. This served as the unfractionated CCM which was used for the present study.
Human latent TGFβ1 levels
To estimate endogenous production of growth factor, the collected CCM was also evaluated for TGFβ1, (regulator of chondrogenic differentiation). Quantitative estimation of TGFβ1 was performed utilizing MABTECH ELISA kit (Cat no: 3550-1H-6). In brief, 96 well plates were coated with the antibody at a concentration of 2 µg, pH 7.4 at 4°C for 12 h. Following this, plates were blocked with the incubation buffer (0.05% Tween 20 containing 0.1% bovine serum albumin). This was followed by addition of standards or samples for 1 h at room temperature. Following a wash, the microplates were incubated with Streptavidin-HRP and TMB substrate. After an appropriate developing time, the optical density was measured (450 nm) by an ELISA reader.
Population doubling time (PDT)
Passage 3 BM-MSCs grown with either EM (Figure 2A, F) or substituted with 50% CCM (Figure 2B, G) were expanded in monolayer cultures at a density of 5000 cells/cm2 and passaged till sub-confluence. Following trypsinization, cell count was performed using Countess (Invitrogen) by tryphan blue exclusion technique. The PDT for the groups, was calculated using the formula:
where N: number of cells obtained at confluence and N0: initial number of cells seeded.

(a) Representative BM-MSC expanded with either EM or CCM at day 1 and day 6 (A–B, F–G), (b) scratch assay showing migration of cells after 45 hrs in culture (C–D, H–I) and (c) chondrogenic differentiation of BM-MSCs. Images of formed pellets post chondrogenic differentiation. Representative microscopic images of Alcian Blue staining of formed pellets (E, J). Magnification: 20×. EM: BM-MSCs cultured in standard expansion medium, CCM: EM supplemented with 50% condition media.
Cell migration: Scratch test
In order to assess the influence of CCM on migratory potential, cells were loaded on to a 6 well plate and expanded till sub-confluence. A scratch was made at the center of the well (marked for reference) and cellular migration toward the scratch from the surrounding cells was observed intermittently by image capture.
Fluorescence assisted cell sorting (FACS)
Acquisition and analysis of the two groups was performed using FACS Celesta and BD FACS Diva v 5.0.2 software. In brief, isolated cells were washed and stained according to manufacturer’s guidelines for each antibody. Rundown of conjugated antibodies used was a) CD105-FITC, CD73-PE, C90-PE (positive MSC markers) b) CD34-PE, CD45-FITC (negative MSC markers) and c) CD166-VioBright FITC, CD146-PE (potential markers of chondrogenesis). Both unstained and isotope controls for individual conjugates were also run.
Reverse transcriptase polymerase chain reaction (RT-PCR)
Total RNA was isolated from 0.5 × 106 cells using Qiagen RNeasy Mini Kit as per manufacturer’s instructions. RNA concentration and absorbance (A260/A280) were determined using Nanodrop spectrophotometer. For cDNA construction, 14 ng of RNA was reverse transcribed using Bio First-Strand synthesis system (Takara). Quantitative RT-PCR (7 ng of cDNA/10 µl reaction) was performed using Eurogentec Takyon™ Low Rox SYBR Master Mix Dttp Blue run on QuantStudio 6 K Flex thermocycler (Applied Biosystem). All analyses from the three samples (n = 3) were performed in duplicates. Relative expression for each of the target genes was normalized to the house keeping gene (ΔCt), glyceraldehyde 3-phosphate dehydrogenase (GAPDH). The expression of genes for chondrogenesis (SOX9: sex determining region Y-box 9, ACAN: Aggrecan and COL2A1: Collagen type 2A1); and hypertrophy (COLA1A1: Collagen type 1A1, RUNX2: Runt related transcription factor-2 and MMP13: Metalloproteinase type 13) was analyzed. Specific primer sequence for each gene utilized is recorded in Table S1.
Chondrogenic differentiation and staining
Induction toward chondrogenic lineage was performed using StemProTM (ThermoFischer) differentiation kit. For three-dimensional suspension pellet cultures, 2.0 × 105cells were loaded into microcentrifuge tube, centrifuged at 1200 rpm for 5 minutes and left undisturbed for a day. Following this, chondrogenic medium was added to the pellets and changed every 3 days for a period of 21 days. To confirm differentiation, pellets were subjected to short fixation (4% paraformaldehyde), embedded in low melting agarose, and subjected to paraffin embedding. Processed sections were stained with Alcian blue (pH 2.5) and counter-stained with Neutral red to assess glycosaminoglycan deposition.
Statistical analysis
Graphical representation was performed via IGOR Pro Version 5.0.4.8 (Wave metrics Inc.) and data analysis utilized SPSS version 21.0. In order to compare all parameters between the two groups, Mann Whitney U test was used. Numerical values were expressed as mean ± standard deviation. A P value of <0.05 was considered as significant.
Results
Latent human TGFβ1 levels
The conditioned media showed a concentration of 0.25 ng/ml of latent human TGFβ1 when quantified using ELISA.
Migration scratch test
The in-vitro migration assay showed that BM-MSCs of both the groups displayed comparable migration toward the marked scratch area (Figure 2: C–D, H–I).
Population doubling time
PD was assessed from cells of passage 2 to passage 3 subjected to the different culture conditions (EM and CCM). It was noted that both the groups showed similar PD without any statistical difference between them (EM: 1.3 ± 0.3 and CCM: −0.3 ± 0.5) (Figure 3).
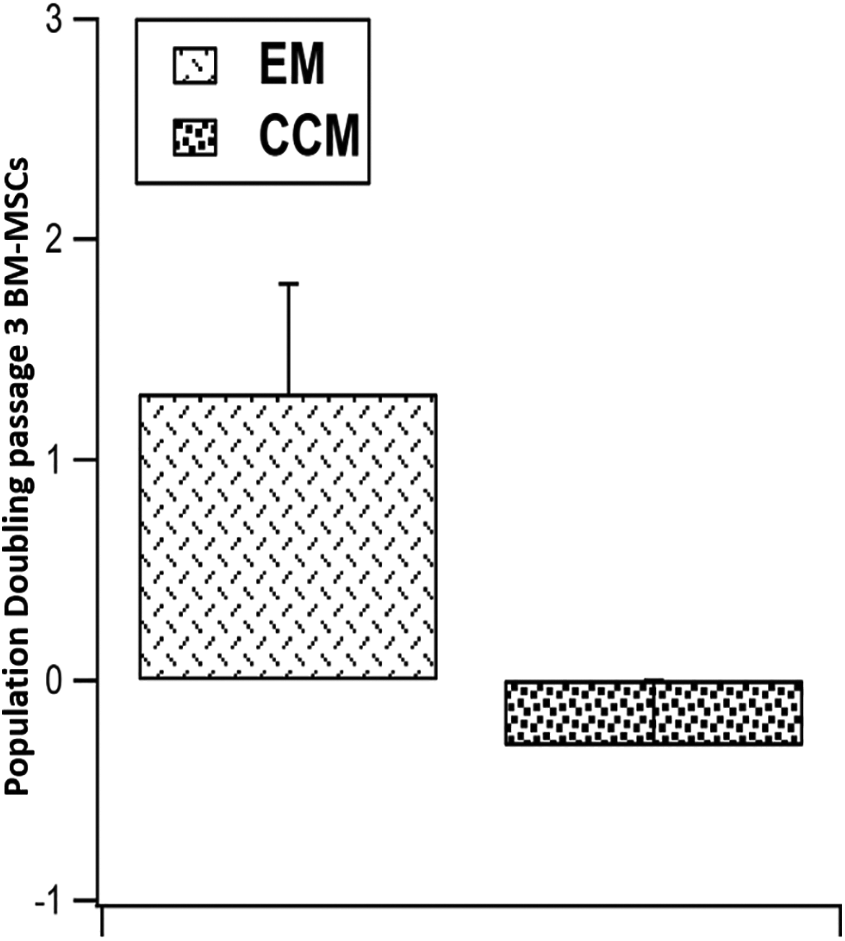
Data representing population doubling values for two subgroups (n = 3, human samples) at passage 3. EM: BM-MSCs cultured in standard expansion medium, CCM: EM supplemented with 50% condition media.
FACS
FACS was performed at P3 between BM-MSCs grown with EM and CCM and analysis revealed a comparable expression. Both groups displayed a high expression of CD105, CD73, CD90, CD166, moderate expression of CD146 and low expression of CD34 and CD45 (Figure 4, Figure S1).
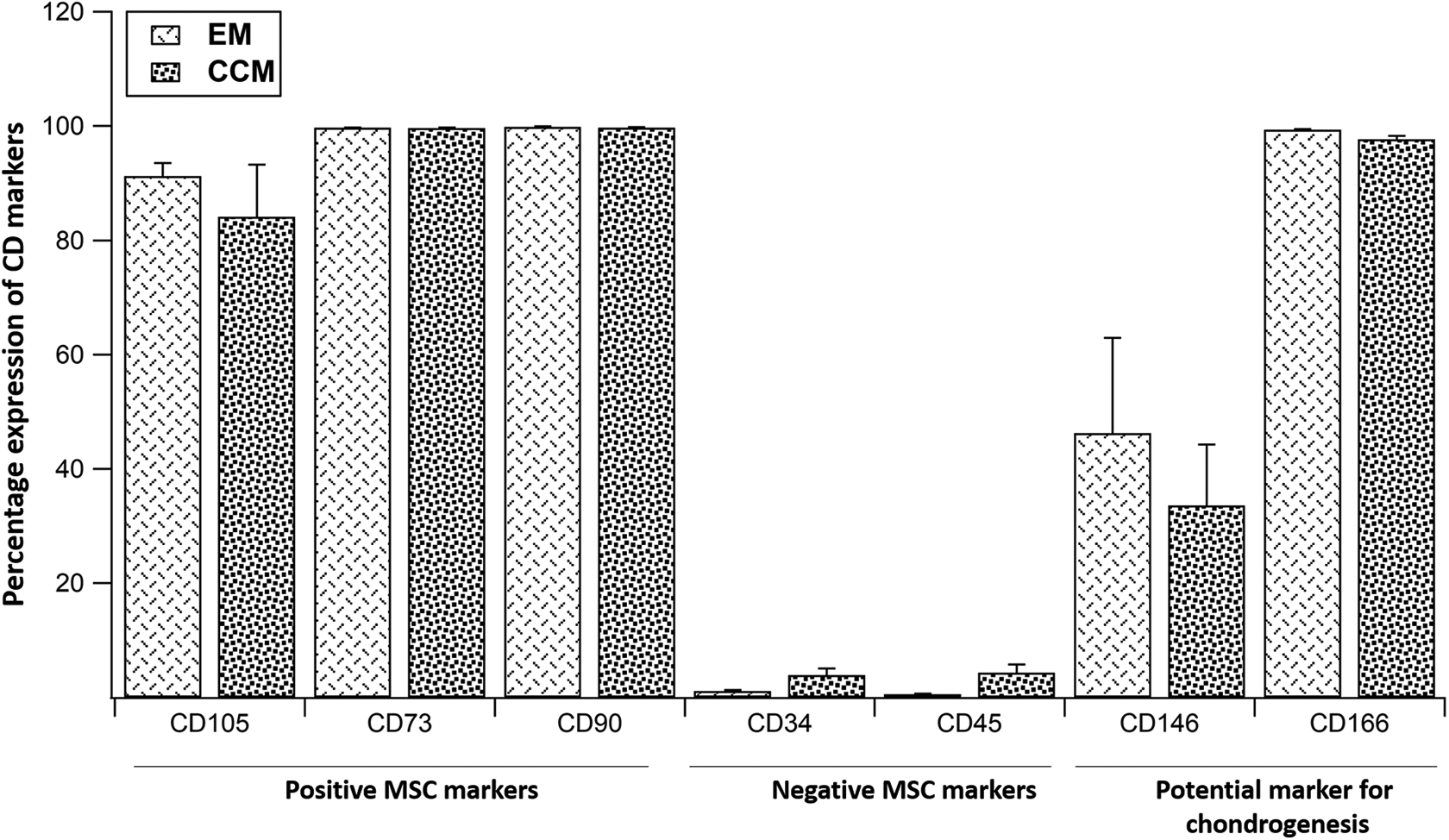
Comparison of percentage expression of CD105, CD73, CD90, CD34, CD45, CD146 and CD146 between the EM and CCM groups. Data expressed as mean ± SD; n = 3. CD: Cluster of differentiation, EM: BM-MSCs cultured in standard expansion medium, CCM: EM supplemented with 50% condition media.
RT-PCR
Comparison of gene expression between the two groups revealed high expression of ACAN and COL1A1, moderate expression of SOX9 and RUNX2 and low expression of COL2A1 and MMP-13 with no significant difference between the two groups (Figure 5).
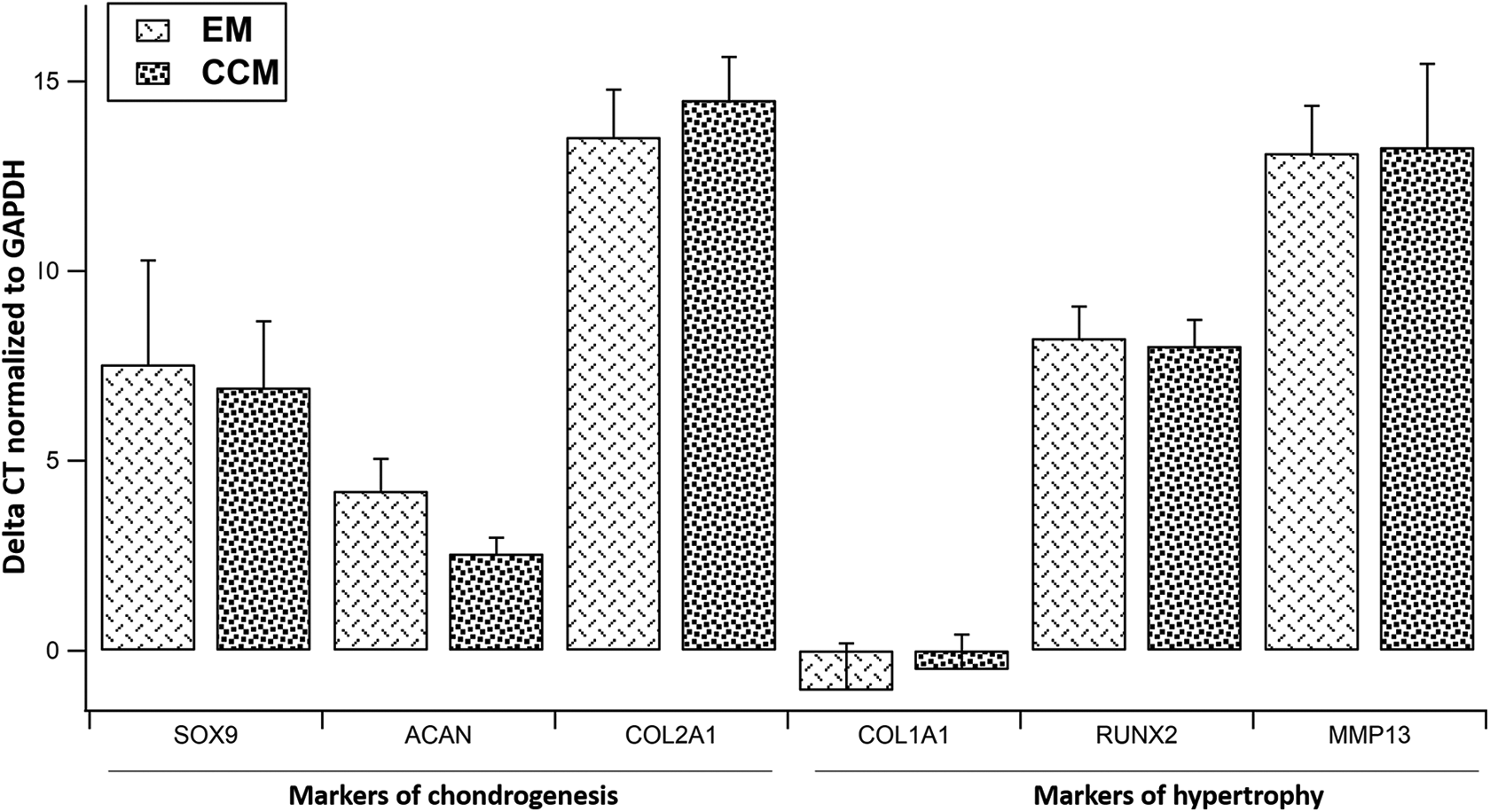
Relative expression of (a) markers of chondrogenesis: SOX9, ACAN, COL2A1 and (b) COL1A1, RUNX2 and MMP13. ΔCt values normalized to GAPDH are expressed as Mean ± SD. Samples taken from n = 3 donors, each sample (n) was run in duplicate. SOX9: sex determining region Y-box 9, ACAN: Aggrecan and COL2A1: Collagen type 2A1, COLA1A1: Collagen type 1A1, RUNX2: Runt related transcription factor-2 and MMP13: Metalloproteinase type 13. EM: BM-MSCs cultured in standard expansion medium, CCM: EM supplemented with 50% condition media.
Chondrogenic differentiation
Both EM and CCM groups displayed comparable glycosaminoglycan deposition as indicated by Alcian blue uptake (Figure 2: E, J).
Discussion
Due to the restricted ability of cartilage to self-renew, extensive research has gone into exploring cell-based strategies for repair of osteochondral degeneration and trauma. Optimization of prominent cell types such as BM-MSCs and chondrocytes, so as to improve in-vivo outcomes following implantation still remains a challenge. Toward this, studies reporting the use of conditioned media rich in soluble factors, likely to reduce hypertrophic tendency and enhance the chondrogenic profile both in-vitro and in-vivo have garnered interest. 18
With respect to chondroprogenitors, numerous reports have successfully demonstrated a reduced tendency for terminal differentiation. 10,19 Due to this inherent capacity, this cell type can function as a possible source of soluble factors for cultivating directly injectable therapeutic molecules. Moreover, trophic factors can act as signaling molecules to drive chondrogenesis and restrict propensity for fibro-cartilagenous phenotype during expansion and following implantation. In this study we attempted to utilize CCM and compare its influence on growth of BM-MSCs with that of standard expansion medium.
When PDT and migratory potential were assessed, use of CCM did not alter the growth or efficiency of migration of BM-MSCs. Similarly, expansion of BM-MSCs with CCM did not affect their surface marker profile as no significant difference was observed when positive and negative MSC markers were compared with the group grown in standard media. The average expression of CD146 and CD166 was lower in the CCM group, though not statistically significant. This difference could be attributed to use of a small sample size thus limiting generalizability of the results or due to the use of semi-spent medium which may require further optimization. To analyze the influence of CCM on BM-MSCs with respect to gene expression and differentiation, it was noted that similar results were seen in both MSC groups in terms of genes for chondrogenesis and hypertrophy with an equivalent profile on pellet analysis demonstrating similar Alcian blue uptake.
Conclusion
In this preliminary report, we utilized CCM at 50% strength without fractionation and observed that it allows BM-MSC growth comparable to standard expansion media, although not positively influencing its chondrogenic profile. It is noteworthy since BM-MSC growth requirements are considerable and at 50% strength, this non-fractionated CCM was a semi-spent medium. Admittedly, purification of CCM to concentrate specific paracrine factors, assay quantification and targeted analysis following dose titration will be pre-requisites to optimize outcomes for re-directing cellular phenotype toward true hyaline cartilage regeneration.
Supplemental material
Supplemental Material, sj-pdf-1-otr-10.1177_22104917211006885 - Effect of human articular chondroprogenitor derived conditioned media on chondrogenic potential of bone marrow derived mesenchymal stromal cells
Supplemental Material, sj-pdf-1-otr-10.1177_22104917211006885 for Effect of human articular chondroprogenitor derived conditioned media on chondrogenic potential of bone marrow derived mesenchymal stromal cells by Elizabeth Vinod, Kawin Padmaja and Upasana Kachroo in Journal of Orthopaedics, Trauma and Rehabilitation
Supplemental material
Supplemental Material, sj-tif-1-otr-10.1177_22104917211006885 - Effect of human articular chondroprogenitor derived conditioned media on chondrogenic potential of bone marrow derived mesenchymal stromal cells
Supplemental Material, sj-tif-1-otr-10.1177_22104917211006885 for Effect of human articular chondroprogenitor derived conditioned media on chondrogenic potential of bone marrow derived mesenchymal stromal cells by Elizabeth Vinod, Kawin Padmaja and Upasana Kachroo in Journal of Orthopaedics, Trauma and Rehabilitation
Footnotes
Acknowledgment
The authors would like to acknowledge the Centre for Stem Cell Research (a unit of inStem Bengaluru), Christian Medical College, Vellore, for infrastructural support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by Institutional Fluid Research Grant, Christian Medical College, Vellore (IRB Min no: 11066 dated 20.12.2017)..
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.