Abstract
Objective
Chondroprogenitors have recently gained prominence due to promising results seen in in vitro and animal studies as a potential contender in cell-based therapy for cartilage repair. Lack of consensus regarding nomenclature, isolation techniques, and expansion protocols create substantial limitations for translational research, especially given the absence of distinct markers of identification. The objective of this systematic review was to identify and collate information pertaining to hyaline cartilage–derived chondroprogenitors, with regard to their isolation, culture, and outcome measures.
Design
As per Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, a web-based search of Scopus and PubMed databases was performed from January 2000 to May 2020, which yielded 509 studies. A total of 65 studies were identified that met the standardized inclusion criteria which comprised of, but was not limited to, progenitors derived from fibronectin adhesion, migrated subpopulation from explant cultures, and single-cell sorting.
Result
Literature search revealed that progenitors demonstrated inherent chondrogenesis and minimal tendency for hypertrophy. Multiple sources also demonstrated significantly better outcomes that bone marrow–derived mesenchymal stem cells and comparable results to chondrocytes. With regard to progenitor subgroups, collated evidence points to better and consistent outcomes with the use of migratory progenitors when compared to fibronectin adhesion assay–derived progenitors, although a direct comparison between the two cell populations is warranted.
Conclusion
Since chondroprogenitors exhibit favorable properties for cartilage repair, efficient characterization of progenitors is imperative, to complete their phenotypic profile, so as to optimize their use in translational research for neocartilage formation.
Introduction
Optimization of cell-based therapy is essential to alleviate the immense global burden of cartilage afflictions. Chondroprogenitors have recently garnered interest as a potential contender for cartilage repair due to their phenotypic tendency toward chondrogenesis and minimal hypertrophy. 1 Additionally, they have been likened to multipotent mesenchymal stromal cells/mesenchymal stem cells (MSCs), demonstrating features as described by the International Society for Cellular Therapy (ISCT) criteria 2006, such as adherence to plastic, specific surface marker expression and multilineage potential. 2 In the sequence of chondrogenesis, MSCs condense and aggregate to form progenitors, which differentiate further to form chondrocytes. 3 This lineage bias exhibited by chondroprogenitors can prove to be useful for both cell-based and tissue engineering strategies for neocartilage formation. However, lack of clarity regarding the definition of said progenitors, coupled with multiple methods of isolation, render categorization imprecise. Previous literature reports chondroprogenitor isolation from various tissue sources such as articular cartilage, synovium, meniscus, and infrapatellar fat pad, among others.4,5 As the end goal remains regeneration of hyaline-like cartilage, accurate characterization, and optimization of chondroprogenitors isolated from hyaline cartilage, is imperative and may prove to be beneficial in the long run since they are naturally primed for chondrogenesis.
Historically, chondroprogenitors were first detected by Hayes et al. as label-retaining cells in the superficial zone of fetal cartilage. 6 Circumstantial evidence demonstrating presence of these cells driving appositional growth led Dowthwaite et al. to isolate and characterize chondroprogenitors, derived from superficial layer of articular cartilage using fibronectin adhesion assay. 7 In addition to these early studies, characterizing of progenitors demonstrated high SRY box transcription factor 9 (SOX9) and Notch homolog 1 (NOTCH1) expression, affinity for CD49e, and higher telomerase activity when compared to mature chondrocytes.8,9 Alternatively, Koelling et al. studied the potential role of chondroprogenitors with respect to homing/migration in response to cartilage injury, thus contributing to tissue repair. 10 This method of isolation of progenitors based on their migratory potential yielded cells that also displayed high chondrogenic potential and features similar to MSCs. Another variation utilized for selection of chondroprogenitors from the general pool of chondrocytes, is based on segregation using potential chondrogenic surface markers. Studies detailing cellular sorting used to obtain progenitors have also reported higher migratory potential and chondrogenic potential in the final isolate.11,12
This systematic review focused on information pertaining to chondroprogenitors isolated from hyaline cartilage, namely nasal septum, and articular surface. All published literature spanning the previous 20 years concerning hyaline cartilage derived chondroprogenitors, isolated via fibronectin adhesion assay, cartilage explants (migratory progenitors), and single cell sorting was assessed and compared. In summarizing available reports, evaluation of cell type most suitable for cartilage repair was performed. This was based on comparison with routinely used cells (MSCs and chondrocytes) and consistency in measured outcomes especially in terms of chondrogenic potential.
Methods
Search Strategy
The literature review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement. An electronic search was performed on “Scopus” and “PubMed” database for the duration spanning January 2000 to May 2020. Due to the diverse nomenclature of the term chondroprogenitors, the search was carried out using the following keywords separately: “chondroprogenitor,” “cartilage-progenitor,” and “chondrogenic-progenitor.” Inclusion criteria comprised all studies published in English language which involved isolation of chondroprogenitors from hyaline cartilaginous tissue using (a) fibronectin adhesion assay, (b) migratory subpopulations derived from cartilage explants, and (c) cell sorting techniques. Reviews, studies involving chondroprogenitors derived from non-cartilaginous tissue (such as differentiated bone marrow mesenchymal stem cells [BM-MSCs], induced pluripotent stem cells [iPSCs], etc.) or using chondroprogenitor cell lines (e.g., ATDC5) were excluded. Two independent observers (EV and UK) conducted the screening process and analysis, following which 2 authors reanalyzed the collated data (RP and BR). First the titles and abstracts were screened using the selection criteria, as mentioned above and further eligibility was confirmed after reviewing the full text. A final screening of the listed articles was also performed to eliminate duplicated publications.
Results
The search database identified a total of 358 articles on Scopus and 151 articles using PubMed. After eliminating 115 paper duplicates, 394 articles were analyzed against the inclusion criteria. After exclusion of 329 articles, the remaining 65 were included for qualitative analysis ( Figure 1 ). A standardized format was used for chronological tabulation of data obtained from the reports analyzed. Categories included source of cartilage sample (species), method of isolation and culture (culture medium used, seeding density, and study timeline), assessment parameters, and study outcomes.
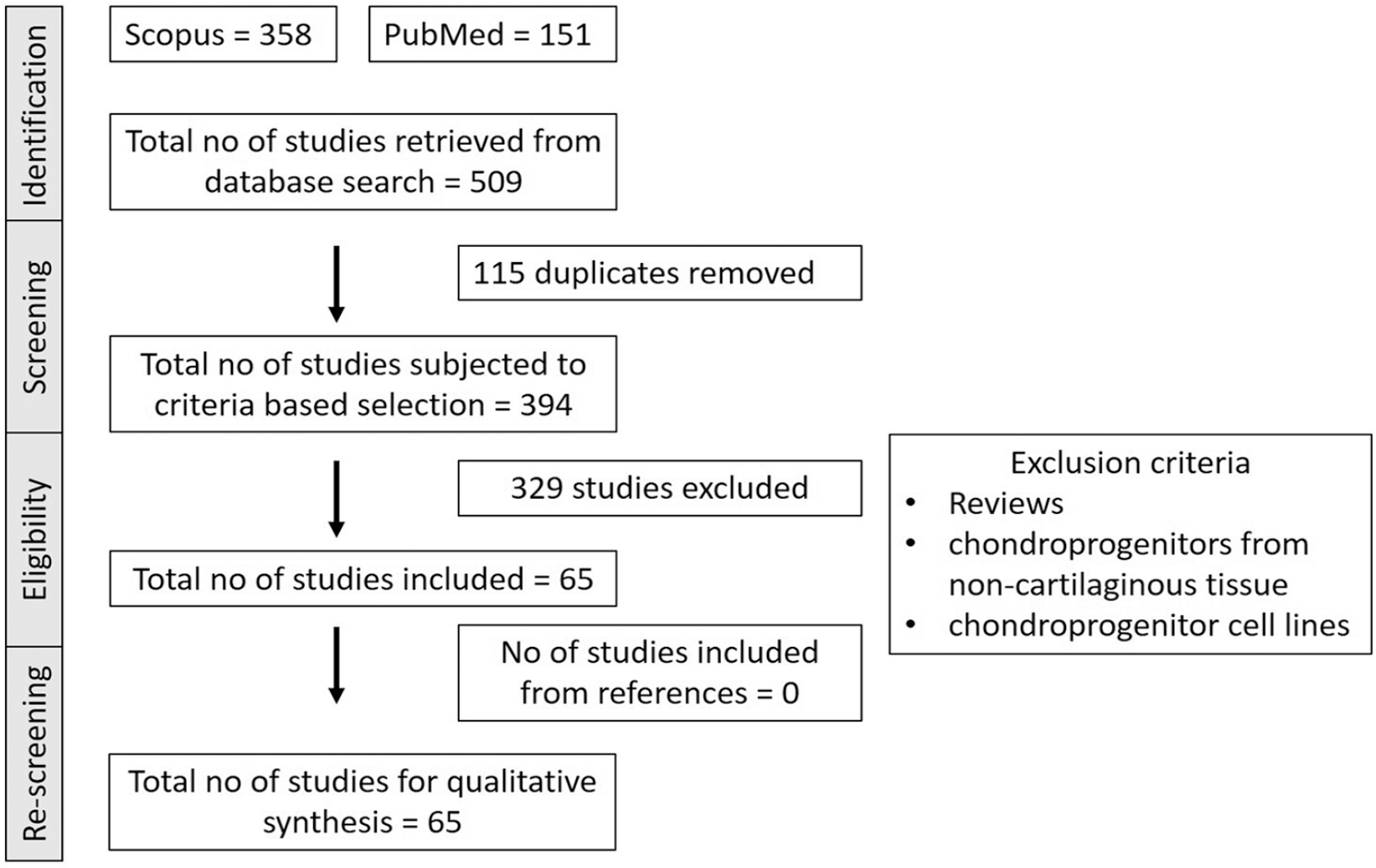
Summary using Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) flow literature search diagram.
Fibronectin Adhesion Assay–Derived Chondroprogenitors
Source of Tissue
A total of 43 reports described chondroprogenitors isolated via fibronectin adhesion assay ( Table 1 ). Cartilage specimens were sourced from both human donors (n = 21) and animals (n = 23). Animal sources included bovine (n = 8), equine (n = 6), swine (n = 2), leporine (n = 2), and murine (n = 5). Literature search revealed that the cartilage samples were collected from the following anatomical sites: knee joint (n = 21), metacarpophalangeal (n = 9), metatarsophalangeal (n = 3), and nasal septum (n = 2), whereas in 8 reports the site of origin was not mentioned. Human cells were derived from either osteoarthritic (n = 10) or nondiseased (n = 7) articular cartilage. Eight reports went on to perform in vivo experiments with chondroprogenitors, while the rest were limited to in vitro investigations.
Summary of Cartilage–Derived Chondroprogenitors Using Fibronectin Adhesion Assay from Articular Hyaline Cartilage.
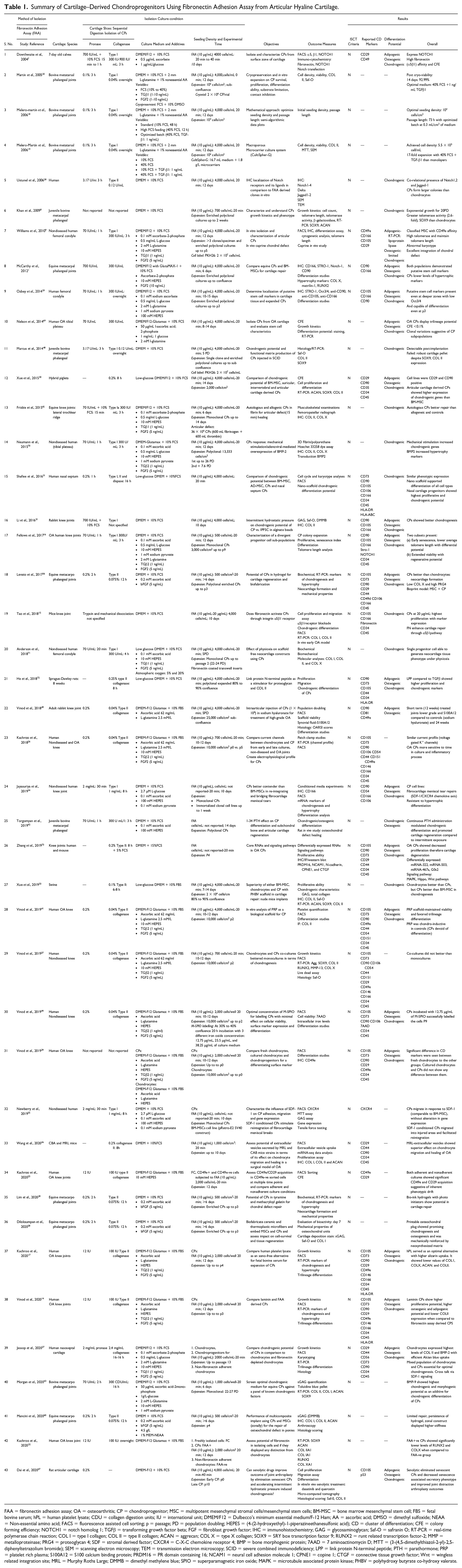
FAA = fibronectin adhesion assay; OA = osteoarthritis; CP = chondroprogenitor; MSC = multipotent mesenchymal stromal cells/mesenchymal stem cells; BM-MSC = bone marrow mesenchymal stem cell; FBS = fetal bovine serum; hPL = human platelet lysate; CDU = collagen digestion units; IU = international unit; DMEM/F12 = Dulbecco’s minimum essential medium/F-12 Ham; AA = ascorbic acid; DMSO = dimethyl sulfoxide; NEAA = Non-essential amino acid; FACS = fluorescence assisted cell sorting; p = passage; PD = population doubling; HEPES = (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid); CD = cluster of differentiation; CFE = colony forming efficiency; NOTCH1 = notch homolog 1; TGFβ = transforming growth factor beta; FGF = fibroblast growth factor; IHC = immunohistochemistry; GAG = glycosaminoglycan; Saf-O = safranin O; RT-PCR = real-time polymerase chain reaction; COL I = type I collagen; COL II = type II collagen; ACAN = aggrecan; COL X = type X collagen; SOX9 = SRY box transcription factor 9; RUNX2 = runt related transcription factor-2; MMP = metalloproteinase; PRG4 = proteoglycan 4; SDF = stromal derived factor; CXCR4 = C-X-C chemokine receptor 4; BMP = bone morphogenic protein; 7AAD = 7 aminoactinomycin D; MTT = (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide); SEM = scanning electron microscopy; TEM = transmission electron microscopy; SCID = severe combined immunodeficiency; LPP = link protein N-terminal peptide; PTH = parathormone; PRP = platelet rich plasma; S100A12 = S100 calcium binding protein; PRDM16 = PR domain containing 16; NCAM1 = neural cell adhesion molecule 1; CPNE1 = copine 1; CTGF = connective tissue growth factor; Wnt = wingless related integration site; MRL = Murphy Roths Large; DMMB = dimethyl methylene blue; SPIO = superparamagnetic iron oxide; MAPK = microtubule associated protein kinase; PHBV = polyhydroxy butyrate co-hydroxy valerate.
Isolation Protocols
For tissue digestion and cellular dissociation, varying durations and enzymatic concentrations were reported. Types of enzymes employed for cartilage digestion included collagenase I (n = 13), collagenase II (n = 18), collagenase (type unspecified, n = 6), a combination of collagenase I, II, and dispase (n = 1), and trypsin (with mechanical dissociation, n = 1). From the data analyzed, most studies (n = 38), reported the use of pronase followed by collagenase digestion of cartilage tissue for cell isolation. Since cellular dissociation and expansion in culture can cause a change in cellular phenotype, the collated data were further analyzed for specific culture conditions such as medium used, seeding density, and additives/growth factors utilized for monolayer expansion. 13
Culture Medium/Additives
All the studies used either DMEM (Dulbecco’s minimum essential medium) or DMEM-F12 as the basal medium, supplemented with either fetal bovine serum (FBS; 10%, n = 38; 15%, n = 1; 40%, n = 3) or human platelet lysate (10%, n = 1). Except for 7 studies, all others reported the use of additional supplements and growth factors. The additives included were ascorbic acid (0.0028 mM to 28 mM, n = 23), L-glutamine (2-2.5 mM, n = 15), sodium pyruvate (0.1-1 mM, n = 6), and HEPES (10-100 mM, n = 19). With respect to growth factors, most commonly used additions were of either transforming growth factor beta 1 (TGFβ1, 1 ng/mL, n = 3), fibroblast growth factor 2 (FGF2, 5 ng/mL, n = 5), or combinations such as TGFβ1 + FGF2 (n = 5) and TGFβ2 + FGF2 (n = 8). Other unique modifications to the culture medium, intended to maintain chondrogenic phenotype included, use of low glucose (0.0027-2.78 mM, n = 14) and hypoxic conditions (5% O2, n = 1).
Seeding Density
Fibronectin adhesion assay protocol described by Dowthwaite et al. was commonly followed in all the published studies with a few variations. 7 All the reported studies involved fibronectin coating at a concentration of 10 µg/mL (n = 41). Additionally, one study compared fibronectin assay–derived chondroprogenitors to laminin adhesion assay (10 µg/mL), 14 while another study compared the standard protocol to a higher fibronectin concentration (20 µg/mL), which yielded cells displaying higher proliferation and marker expression. 15 Seeding densities mentioned for FAA varied between 1,000 and 4,000 cells/well (n = 32) or 700 cells/mL (n = 3) or 500 cells/cm2 (n = 5), for a period of 20 minutes (n = 40), following which the nonadherent cells were removed. The adherent cells were further expanded for 6 to 12 days, ensuring 5 population doublings. Clonally expanded progenitors were grown as polyclonal enriched cultures (n = 23) or as monoclonal cultures (n = 6). A report also exhibited the ability of progenitors to expand in culture up to passage 9 while retaining good viability and regenerative potential. 16
Characteristics of Chondroprogenitors
Growth Kinetics
With regard to growth kinetics, in the two studies that compared chondroprogenitors to chondrocytes, one reported higher colony-forming efficiency and 2.5-fold greater telomerase activity in the former, along with exponential growth recorded up to 20 population doublings, 9 while the other demonstrated comparable results between the two cell types. 24 Characterization by Fellows et al. demonstrated the presence of two progenitor subsets, one exhibiting early senescence and lower average telomere length with higher differentiation potential while the other showed extended viability with greater regenerative potential. 16 In another study, chondroprogenitors isolated using a laminin adhesion assay showed significantly higher proliferative potential but lower chondrogenic potential. 14 It was also seen that for creation of xeno-free culture conditions, when human platelet lysate was used in place of FBS, progenitors displayed greater proliferation with lower expression of type I and X collagen (fibrocartilage and hypertrophy markers). 17 With regard to culture conditions producing maximum growth of progenitors, Melero et al. report an optimal seeding density of 10,000 cells/cm2 and medium volume of 0.3 mL/cm2.18,19 The authors further demonstrated an efficient post-cryoviability and 17-fold expansion potential using macroporous microcarrier supplemented with 40% FCS and 1 ng/mL of TGFβ1. 20
Surface Markers
Multiple reports examined chondroprogenitors for a gamut of cell surface markers, investigating positive, negative MSC marker expression and differentiation potential to assess conformation to the ISCT criteria. Of the 41 reports, 25 studies assessed cell surface marker expression, while 30 evaluated potential for differentiation (either monolineage or trilineage) and none of the studies reported evaluation based on the complete minimum criteria laid down for MSCs by ISCT 2006. Regarding differentiation potential, it was seen that intermittent hydrostatic pressure (n = 1) 21 and growth under physioxia (n = 1), 22 demonstrated better chondrogenesis. In another study, higher adipogenesis and osteogenesis was observed in chondroprogenitors derived from laminin adhesion compared to fibronectin adhesion. 14 Supplementation with platelet lysate as opposed to FBS caused chondroprogenitors to show comparatively higher alizarin red uptake. 17 Commonly reported positive MSC markers included CD105 (n = 17), CD90 (n = 20), CD73 (n = 12), while negative MSC markers included CD34 (n = 15) and CD45 (n = 15). Numerous reports evaluated the following cell surface proteins as potential markers of chondrogenesis namely CD29 (n = 12) in combination with CD49e (n = 19), CD146 (n = 4), and CD166 (n = 13). Fibronectin is a classical ligand for the α5β1 integrin receptor, for which CD49e is the α5 component. 23 CD49e was shown to be highly expressed by superficial layer chondroprogenitors when compared to freshly isolated chondrocytes. However, when expression was compared to cultured chondrocytes, no significant difference was seen between the 2 groups. 24 In another report, when CD49e negative cells were assessed in monolayer and suspension culture systems, dramatic upregulation of CD49e expression was noted. 25 Similarly, with regard to CD146 and CD166, no consensus has been found to suggest that sorting based on aforementioned markers will yield cells with enhanced chondrogenic potential.26,27
Chondrogenic Potential: Chondroprogenitors versus Other Cell Types
Initial characterization of superficial layer chondroprogenitors revealed expression of notch receptors, a gene known to be involved in cellular colony-forming ability.7,28 Later these receptors were also confirmed to be present in progenitors embedded at deeper cartilage zones. 29 With respect to chondrogenic potential, Xue et al. reported the superiority of articular cartilage derived chondroprogenitors to progenitors isolated from other sources such as auricle and intervertebral disc. 30 However, a study employing human nasal septum–derived chondroprogenitors reported better results when compared to articular cartilage–derived chondroprogenitors, adipose tissue–derived MSCs, and BM-MSCs. 31 In addition to this, many studies exhibited superiority of chondroprogenitors to BM-MSCs as a potential candidate for cartilage repair. Literature by McCarthy et al. and Kachroo et al. demonstrated significantly low levels of hypertrophy markers in chondroprogenitors,1,32 while Xue et al. reported higher expression of chondrogenic genes when compared to BM-MSCs.30,33 Similarly, Jayasuriya et al. and Newberry et al. showed that chondroprogenitors bettered BM-MSCs for meniscal tear repairs, demonstrating higher migratory potential in response to SDF-1/CXCR4 (SDF, stromal derived factor; CXCR4, C-X-C chemokine receptor 4) with improved resistance to hypertrophy.34,35 On the same line, Khan et al. and Levato et al. reported superiority of chondroprogenitors to chondrocytes, due to higher SOX9, lubricin (PRG4) levels, and lower type X collagen expression, thus indicating improved potential for neocartilage formation.8,36 Moreover, Vinod et al. demonstrated comparable results in an in vitro human study, 27 while Jessop et al. and Xue et al. reported chondrocyte superiority with regard to type II collagen expression and glycosaminoglycan production.33,37
Scaffold Use, Specific Additives, and In Vivo Studies for Cartilage Repair
Scaffold-based studies (n = 9) using chondroprogenitors showed variable results ( Table 1 ). An in vitro study employing a bio-fabricate made of ceramic and thermoplastic microfibers, further embedded with chondroprogenitors and BM-MSCs exhibited favorable neosynthesized matrix upon mechanical stimulation. 38 Similarly, Lim et al. reported chondroprogenitors embedding in light visible bioink hydrogel made up of tyramine and methacryloyl gelatin and displayed potential for integration and intraoperative bioprinting. 39 However, in an in vivo assessment performed by Mancini et al., cell-laden multicomposite implants showed limited repair and persistence of the hydrogel with zonal construct displaying higher stiffness. 40 In a report by Vinod et al., platelet-rich plasma (PRP), a biological scaffold was used and resulted in favorable glycosaminoglycan deposition even in the absence of chondro-induction. 41 With regard to the effect of specific supplements, added in order to enhance chondroprogenitor phenotype, three different studies were reported. Torgomyon showed that continuous (rather than intermittent) administration of parathormone (PTH) modulated chondrogenic differentiation and enhanced cartilage regeneration in an in vivo osteochondral defect model. 42 In a study by Jayasuriya et al., SDF1 conditioned chondroprogenitors displayed, higher migratory potential into areas of injury with promotion of regeneration, in an in vitro meniscal defect model. 34 Additionally, Morgan et al. showed that addition of bone morphogenic protein (BMP-9, 100 ng/mL) to the chondrogenic differentiation medium, displayed the highest chondrogenic and morphogenic potential when compared against a panel of known chondrogenic factors. 43
In the literature search, only one study by Vinod et al. reported on the successful labelling of chondroprogenitors with 12.75 µg/mL of micron-size superparamagnetic iron-oxide that had minimal effect on viability, surface marker expression, and differentiation potential. 44
The first in vivo study to use chondroprogenitors was a pilot by Williams et al., performed utilizing a caprine chondral defect model. 9 It reported excellent integration, although no difference in the quality of repair tissue was observed when compared to full-depth chondrocytes. In another equine study employing a chondral defect model, autologous chondroprogenitors embedded in a fibrin scaffold displayed superior repair to allogeneic chondroprogenitors and also to the scaffold itself. 45 A study performed by Wang et al. demonstrated the use of intraarticular injections of chondroprogenitor extracellular vesicles secreted by MRL mice, showing attenuation of osteoarthritis, thus providing new insight in their role as exosomal injections. 46 Additionally, another study by Dai et al. reported novel use of Dasatinib and Quercetin in eliminating senescent progenitors, thereby decreasing senescence-associated secretory phenotype, thus improving joint distraction arthroplasty outcomes. 47 The literature search also revealed that a few studies involving fibronectin adhesion–derived chondroprogenitors exhibited variable results. One study which utilized labelled chondroprogenitors, implanted into severe combined immunodeficiency (SCID) mouse thigh muscle, showed maintenance of chondrogenic potential but failure to produce extracellular matrix (ECM). 48 Similarly, another study employing polyhydroxy butyrate co hydroxy valerate (PHBV)-chondroprogenitor constructs as subcutaneous implants demonstrated better chondrogenesis than BM-MSCs but inferior results to chondrocytes. 33 Moreover, another study revealed that chondroprogenitors embedded with hyaluronic acid also failed to show superiority to plain hyaluronic acid when injected intraarticularly in a rabbit-based monosodium iodoacetate–induced high-grade osteoarthritic model. 49 Another study by Mancini et al. using a long-term equine model reported limited production of cartilage-like tissue in osteochondral defects when implanted with a 3D multicomposite construct containing chondroprogenitors and MSCs. 40
Migratory Chondroprogenitors
A total of 18 reports describing chondroprogenitors isolated from cartilage explants by virtue of their migratory properties were published ( Table 2 ).
Summary of Cartilage Explant–Derived Migratory Chondroprogenitors from Articular Hyaline Cartilage.
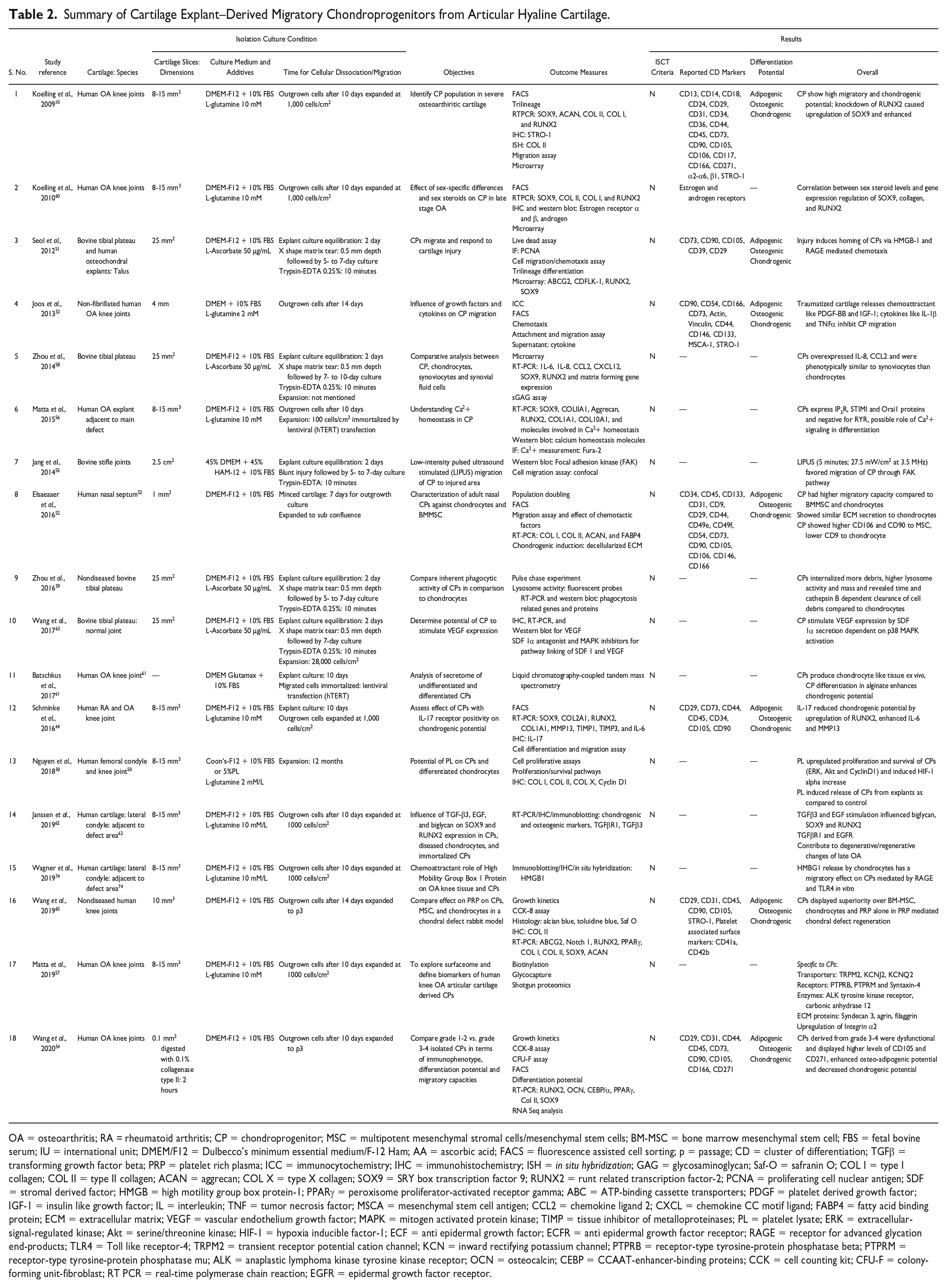
OA = osteoarthritis; RA = rheumatoid arthritis; CP = chondroprogenitor; MSC = multipotent mesenchymal stromal cells/mesenchymal stem cells; BM-MSC = bone marrow mesenchymal stem cell; FBS = fetal bovine serum; IU = international unit; DMEM/F12 = Dulbecco’s minimum essential medium/F-12 Ham; AA = ascorbic acid; FACS = fluorescence assisted cell sorting; p = passage; CD = cluster of differentiation; TGFβ = transforming growth factor beta; PRP = platelet rich plasma; ICC = immunocytochemistry; IHC = immunohistochemistry; ISH = in situ hybridization; GAG = glycosaminoglycan; Saf-O = safranin O; COL I = type I collagen; COL II = type II collagen; ACAN = aggrecan; COL X = type X collagen; SOX9 = SRY box transcription factor 9; RUNX2 = runt related transcription factor-2; PCNA = proliferating cell nuclear antigen; SDF = stromal derived factor; HMGB = high motility group box protein-1; PPARγ = peroxisome proliferator-activated receptor gamma; ABC = ATP-binding cassette transporters; PDGF = platelet derived growth factor; IGF-1 = insulin like growth factor; IL = interleukin; TNF = tumor necrosis factor; MSCA = mesenchymal stem cell antigen; CCL2 = chemokine ligand 2; CXCL = chemokine CC motif ligand; FABP4 = fatty acid binding protein; ECM = extracellular matrix; VEGF = vascular endothelium growth factor; MAPK = mitogen activated protein kinase; TIMP = tissue inhibitor of metalloproteinases; PL = platelet lysate; ERK = extracellular-signal-regulated kinase; Akt = serine/threonine kinase; HIF-1 = hypoxia inducible factor-1; ECF = anti epidermal growth factor; ECFR = anti epidermal growth factor receptor; RAGE = receptor for advanced glycation end-products; TLR4 = Toll like receptor-4; TRPM2 = transient receptor potential cation channel; KCN = inward rectifying potassium channel; PTPRB = receptor-type tyrosine-protein phosphatase beta; PTPRM = receptor-type tyrosine-protein phosphatase mu; ALK = anaplastic lymphoma kinase tyrosine kinase receptor; OCN = osteocalcin; CEBP = CCAAT-enhancer-binding proteins; CCK = cell counting kit; CFU-F = colony-forming unit-fibroblast; RT PCR = real-time polymerase chain reaction; EGFR = epidermal growth factor receptor.
Source of Tissue
Studies involving human subjects (n = 13), utilized hyaline cartilage either sourced from articular areas (n = 12) or the nasal septum (n = 1). Human samples were derived from diseased (osteoarthritis: n = 13, rheumatoid arthritis: n = 1) or nondiseased (n = 1) joints. Five studies used healthy bovine articular cartilage for the isolation of migratory chondroprogenitors.
Cell Isolation and Culture Conditions
To isolate migratory chondroprogenitors, cartilage slices of varying sizes were obtained and cultured as explants for 7 to 14 days. With respect to the size of cartilage explants, most sliced specimens had a size of 8 to 15 mm2 (n = 8), while others were 25 mm2 (n = 6), 10 mm3 (n = 1), 4 mm (n = 1), 1 mm2 (n = 1), 0.1 mm3 (n = 1), and unspecified (n = 1). In a few studies (n = 4), following 2 days of culture, explants were scratched to create a matrix tear (X-shaped) of 0.5 mm depth, and further cultured for 7 days, following which they were treated with 0.25% Trypsin-EDTA (10 minutes) to facilitate easier release of migrated progenitors from the surface. Thereafter, outgrowing or migrated cells were reloaded at a seeding density of 1,000 cells/cm2 and further expanded to confluence. All the studies used either DMEM (Dulbecco’s minimum essential medium) or DMEM-F12, further supplemented with FBS (10%, n = 18) or platelet lysate (5%, n = 1). Addition of ascorbic acid (0.28 mM, n = 4) and L-glutamine (2-10 mM, n = 9) was also reported. Use of additional growth factors for proliferation was not reported in any of the studies. Concerning proliferative capacity, one study reported the ability of progenitors to expand when cultured for a period of 12 months, maintaining its viability and inherent characteristics. 50
Characteristics of Chondroprogenitors
Growth Kinetics: Migratory Potential
In 2009, Koelling et al. were the first to identify, characterize, and report migratory chondroprogenitors isolated from osteoarthritic cartilage. 10 They demonstrated presence of high migratory and chondrogenic potential. Additionally, Seol et al. showed that progenitors in response to cartilage injury displayed higher migration via High Mobility Group Box 1 Protein (HMGB) and receptor for Advanced Glycation End Products (RAGE) mediated chemotaxis. 51 Elsaesser et al. also reported superiority of nasal chondroprogenitors when compared to chondrocytes and BM-MSC with respect to migratory capacity. 52 A study by Joos et al. reported that release of platelet derived growth factor (PDGF-BB) and insulin-like growth factor (IGF-1) enhanced chondroprogenitor migration, whereas interleukin-1β (IL-1β) and tumor necrosis factor-α (TNFα) displayed an inhibitory influence on progenitor movement, following injury. 53 Recently, Wagner et al. identified the vital role of chondrocytes in the release of HMGB-1 protein which influenced the migratory potential of chondroprogenitors via RAGE and toll like receptor 4 (TLR4). 54 Another study reported the use of low-intensity pulsed ultrasound stimulated (LIPUS) migration of chondroprogenitors to the area of injury via focal adhesion kinase pathway. 55
Surface Markers
From the literature analyzed, 7 reports investigated cell surface proteins to look for positive, negative MSC marker expression and differentiation potential. Commonly reported positive MSC markers included CD105 (n = 6), CD90 (n = 7), CD73 (n = 6), while negative markers reported were CD34 (n = 3) and CD45 (n = 5). Numerous reports also evaluated the following cell surface proteins as potential markers of chondrogenesis, namely, STRO-1 (n = 3), CD29 (n = 6), CD49e (n = 2), CD146 (n = 2), and CD166 (n = 4). With regard to trilineage differentiation, chondroprogenitors derived from grade 3 to 4 osteoarthritis (as opposed to grade 1-2) demonstrated higher dysfunction with greater osteo-adipogenic potential and decreased chondrogenic potential. 54 With regard to cellular differentiation, a study reporting on proteins involved in Calcium signaling demonstrated chondroprogenitors to express IP3, STIM1, and ORAI-1 receptors and absence of ryanodine receptors. 56 A recent study by Matta et al. using biotinylation, glycocapture and shotgun proteomics, surfaceome of chondroprogenitors were analyzed in comparison to BM-MSCs. 57 It was reported that chondroprogenitors demonstrate significantly higher levels of ECM proteins such as Syndecan, Agrin, and Filaggrin. They also reported notable upregulation of CD49b, an integrin marker and provided data pertaining to chondroprogenitor specific channels, including those belonging to the transient receptor potential melastatin (TRPM) family and potassium channel subfamily. 57
Chondrogenic Potential: Chondroprogenitors versus Other Cell Types
Zhou et al. were the first to compare chondroprogenitors to chondrocytes, synoviocytes, and synovial fluid cells. 58 It was seen that chondroprogenitors displayed more similarity to synoviocytes than chondrocytes, with overexpression of IL-8 and chemokine ligand-2 (CCL2). In other studies, progenitors showed higher phagocytosis and lysosomal activity dependent on cathepsin B mediated clearance 59 but similar ECM production when compared to chondrocytes. 52
Effects of Interventions
Koelling et al. reported a correlation between sex steroids and chondroprogenitors, that is, hormone replacement therapy provided in combination with physiological levels of testosterone demonstrated a positive influence on chondrogenic potential. 60 In another study, human platelet lysate showed activation and proliferation of quiescent cells, thereby contributing to new cartilage formation. 50 Additionally, differentiation of chondroprogenitors in alginate demonstrated favorable chondrogenesis. 61 When chondroprogenitors were stimulated with TGFβ3 and epidermal growth factor in vitro, upregulation of both SOX9 and runt related transcription factor-2 (RUNX2) levels was observed. 62 A few reports also described the variable influence of chondroprogenitors in a pathological setting. Wang et al. reported that chondroprogenitors might contribute to the pathogenic accumulation of vascular endothelial growth factor in an osteoarthritic environment, 63 while another study found that chondroprogenitors isolated from rheumatoid arthritis joints displayed high levels of IL-17, thus contributing to reduced chondrogenic potential (via RUNX2, IL-6, and matrix metalloproteinase-13). 64
In Vivo Studies for Cartilage Repair
Only one in vivo study involving chondroprogenitors derived from nondiseased human tissue was reported. 65 In this rabbit chondral defect model by Wang et al., chondroprogenitors suspended in PRP showed higher chondrogenic predisposition with superior healing of critical defects when compared to BM-MSCs in PRP, chondrocytes in PRP, and PRP alone.
Chondroprogenitors Derived Following Cell Sorting
Following literature search, 4 studies were found to use cellular sorting, to isolate either single cells or obtain a subset based on cell surface marker expression ( Table 3 ). In a report by Su et al., human chondroprogenitors were isolated based on CD146 positivity, and when compared to unsorted chondrocytes, exhibited higher levels of MSC specific antigens and chondrogenic capacity in a 3-dimensional pellet culture. 11 In another study by Yu et al., bovine chondrocytes were subjected to flow assisted single-cell sorting, seeded on gelatin-coated plates, and expanded in culture using 10% knock out serum replacement (KOSR). Following clonogenicity screening, cells with high-efficiency colonies were labelled as progenitors, wherein superficial layer derived cells displayed improved clonality, whereas deeper zone cells displayed higher chondrogenesis and osteogenesis. 12 Furthermore, these cells were used in another study as a proof of concept, where a bioprinting model used progenitors to generate vessel-like channels. 66 One report also assessed the effect of leptin on etiology and development of osteoarthritis using murine progenitors derived following single-cell sorting. 67 It was seen that leptin caused senescence in the progenitors via the p53/p21 pathway and increased terminal differentiation.
Summary of Chondroprogenitors Derived Following Cell Sorting.
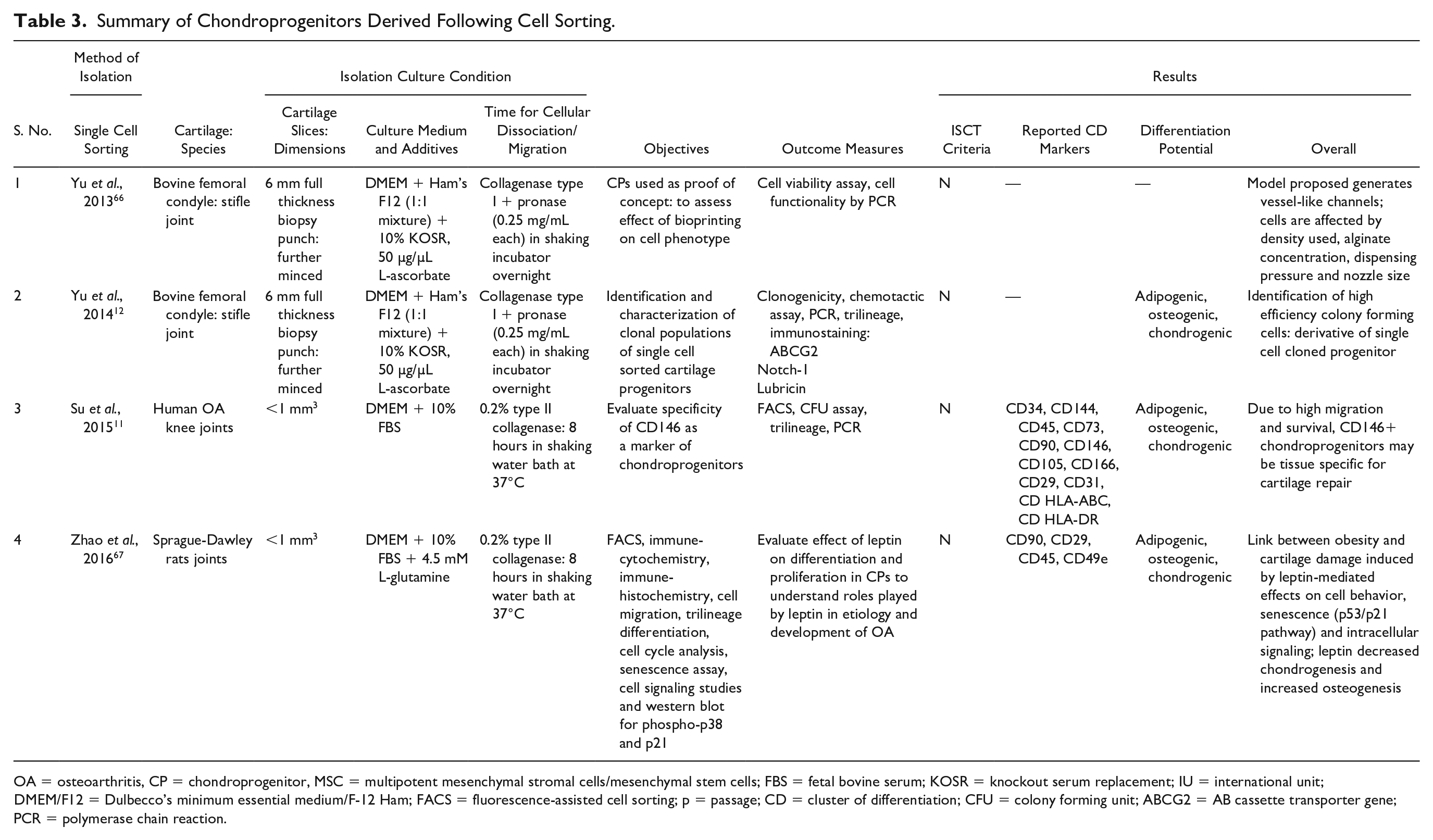
OA = osteoarthritis, CP = chondroprogenitor, MSC = multipotent mesenchymal stromal cells/mesenchymal stem cells; FBS = fetal bovine serum; KOSR = knockout serum replacement; IU = international unit; DMEM/F12 = Dulbecco’s minimum essential medium/F-12 Ham; FACS = fluorescence-assisted cell sorting; p = passage; CD = cluster of differentiation; CFU = colony forming unit; ABCG2 = AB cassette transporter gene; PCR = polymerase chain reaction.
Discussion
In the field of cartilage repair, cell-based therapy primarily involves MSCs and chondrocytes as regenerative interventions. Due to effective but suboptimal healing observed with their use, search for a cellular substitute led to work on chondroprogenitors. Since the nomenclature for this cell type is diverse, this review was restricted to studies focusing on hyaline cartilage–derived progenitors and their properties. An assessment of data summary led to categorization of all studies into 3 groups based on technique of progenitor isolation.
With regard to growth kinetics, though all chondroprogenitor subgroups displayed clonal growth, migratory potential was consistently assessed and demonstrated in reports using cartilage explant derived progenitors, given that this parameter was a prerequisite to obtain this cell type ( Table 2 ). In contrast, little data are available regarding fibronectin derived chondroprogenitors’ inherent ability to migrate, with studies only examining the influence of chemokines on efficiency of migration. 35 When cell surface markers were assessed, all chondroprogenitor subgroups exhibited high positive and minimal negative MSC marker expression. In terms of a discrete marker, a study reported significantly high levels of CD49b in migratory chondroprogenitors as compared to BM-MSCs, on evaluation of cell surfaceome. 57 However, no studies reported notably high expression of any specific marker in fibronectin chondroprogenitors when compared to other cell types, with 2 studies showing similar marker profile between fibronectin progenitors and cultured chondrocytes.24,27 The challenge with comparing progenitors and chondrocytes lies in that, they are derived from the same source and may inherently exhibit similar phenotypic profiles. Any minor differences that may exist would be altered or lost upon their release from their microenvironment or due to expansion in culture.24,25,68
On further assessment of chondroprogenitor subgroups, another area of potential conflict could be with regard to culture conditions required for their propagation. There is an apparent species variability wherein progenitors isolated from animal sources do not require additional growth factors in the concentration, as reported for the human subtype. Furthermore, concerning human cells, reports on migratory chondroprogenitors do not employ superadded growth factors, whereas adhesion assay derived progenitors are commonly grown with additives such as TGFβ, FGF, among others ( Tables 1 and 2 ). This observation favors explant derived progenitors over fibronectin assay-derived cells as the use of growth factors to sustain proliferation in culture may also result in additional changes to cell biology, as many additives in common use are important cell signaling molecules.
Although multiple studies have assessed influence of varying culture conditions and interventions on fibronectin progenitor chondrogenesis in vitro, data pertaining to explant derived chondroprogenitors is limited. However, a noteworthy finding reported in studies on both subgroups, was the beneficial effect of PRP or platelet lysate on chondroprogenitor phenotype, as either a biocompatible scaffold or an additive.17,41,65 Use of autologous/allogeneic platelet products may be investigated in the future as xeno free alternative for chondroprogenitor proliferation and differentiation. With respect to in vivo studies involving chondroprogenitors, fibronectin derived cells demonstrated results superior to MSCs but equivalent or inferior results to chondrocytes in other reports.1,33 Fibronectin progenitors embedded in scaffolds also, did not exhibit improved healing over the use of plain scaffold. 49 Although a single report showed that migratory progenitors encapsulated in PRP displayed better results than scaffolded BM-MSCs and chondrocytes, 65 lack of other in vivo data makes it difficult to infer superiority conclusively.
Since multiple studies have demonstrated that hyaline cartilage–derived chondroprogenitors display properties of inherent chondrogenesis and minimal propensity for terminal differentiation, the future direction with translational research involving progenitors should include enhancement of their chondrogenic potential while maintaining their lower hypertrophic preponderance. Following data analysis, it is evident that chondroprogenitors display characteristics akin to MSCs and are a contender for cell-based therapy in cartilage repair. The complications involved in their use stem from the lacunae still present in their biological profile and the myriad ways of isolation and expansion. In particular, no literature was available that elaborated on immunogenic and immunomodulatory properties of chondroprogenitors, which is essential with respect to predicting in vivo behavior of the cell following transplantation. With regard to progenitor subgroups, current evidence points to improved outcomes with the use of migratory progenitors, although a direct comparison between the two cell populations is warranted. Any inferences made from comparisons done on the collated studies also suffer due to the ambiguity in results seen with fibronectin chondroprogenitors, arising due to the culture conditions established for their use and also secondary to limited sample size.
In conclusion, this review reiterates that chondroprogenitors can serve as a useful tool in cell-based therapy for cartilage repair after appropriate optimization. Understanding and further clarification of nomenclature, isolation methodology, along with refinement of culture techniques can yield true chondroprogenitors having multiple applications in the treatment of osteochondral afflictions.
Footnotes
Acknowledgment and Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
