Abstract
Objective
To support the preclinical evaluation of therapeutics that target chondrogenesis, our goal was to generate a rat strain that can noninvasively report endogenous chondrogenic activity.
Design
A transgene was constructed in which the dual expression of bioluminescent (firefly luciferase) and fluorescent (mCherry) reporters is controlled by regulatory sequences from rat Col2a1. Candidate lines were established on a Lewis background and characterized by serial bioluminescence imaging as well as ex vivo measurement of molecular reporter levels in several tissues. The sensitivity and specificity of the reporter strain were assessed in models of orthotopic and ectopic chondrogenesis.
Results
Substantial bioluminescence signal was detected from cartilaginous regions, including the appendicular synovial joints, spine, sternum, nose, and pinnae. Bioluminescent radiance was intense at 1 month of age, rapidly declined with continued development, yet remained detectable in 2-year-old animals. Explant imaging and immunohistochemistry confirmed that both molecular reporters were localized to cartilage. Implantation of wild-type bone marrow stromal cells into osteochondral defects made in both young adult and aged reporter rats led to a time-dependent elevation of intra-articular reporter activity concurrent with cartilaginous tissue repair. To stimulate ectopic, endochondral bone formation, bone morphogenetic protein 2 was overexpressed in the gastrocnemius muscle, which led to bioluminescent signal that closely preceded heterotopic ossification.
Conclusions
This strain can help develop strategies to stimulate cartilage repair and endochondral bone formation or to inhibit chondrogenesis associated with heterotopic ossification.
Introduction
Chondrogenesis is essential to the development and regeneration of not only cartilage, but also bone. This process is highly coordinated during skeletal development in such a way that is not replicated fully in response to skeletal injury. Within the knee, for example, articular cartilage and menisci are highly specialized structures that do not mount a sufficient regenerative response when damaged; the resulting loss of tissue integrity contributes to the onset of osteoarthritis (OA). 1 An example of de novo chondrogenesis within the adult skeleton involves the healing of long bone fractures, where mesenchymal progenitor cells recruited to the fracture site form a cartilaginous callus that acts as a template for bone modeling. 2 For critical-sized bone defects, however, such endochondral bone formation is insufficient. 3 Whereas orthotopic chondrogenesis in response to skeletal injury can often be inadequate, trauma can lead to ectopic chondrogenesis and endochondral ossification within musculoskeletal tissues, such as muscle and tendon. 4 New strategies are needed to better stimulate chondrogenesis in bone and cartilage defects as well as to prevent heterotopic chondrogenesis following trauma.
Rodent models of musculoskeletal injury are important for the development of next-generation therapeutics. The application of gene editing tools, such as Cre-Lox recombination, to the well-characterized murine genome introduced the revolutionary advancement of transgenic and knockout mouse strains. These strains have been instrumental in studying mammalian gene function for skeletal development and regeneration and in tracing stem/progenitor cell fate through conditional expression of reporter proteins.5-7 While genetically engineered mice are now used routinely in orthopedic research, similar rat models have been lagging. Given the notable differences in skeletal size and drug metabolism rates between mice and rats, in many cases the latter species would be preferred for initial therapeutic testing in vivo. When genome modification is not essential for hypothesis testing, the rat is routinely used for evaluating new treatments to address critical-sized bone and cartilage defects. 8 For studies of cartilage repair in particular, mouse models are technically feasible9,10 but prohibitively small for testing certain therapeutics, such as biomaterials. For studies that require the assessment of joint pain, rats are more easily habituated to sensory testing apparatuses and less susceptible to handling-induced analgesia than mice. 11 Given these advantages, rat strains engineered to aid the study of skeletal development and/or regeneration would benefit the field.
Here we present a transgenic rat whose chondrogenic cells co-express firefly luciferase (fLuc) and mCherry for reporting chondrogenesis at the tissue and individual cell scales. As demonstrated using models of skeletal repair and heterotopic ossification, this strain can be used to provide noninvasive, longitudinal assessment of chondrogenesis to complement endpoint measures of cartilage or bone formation.
Materials and Methods
Transgenic Construct
To specify reporter expression within chondrogenic cells, we chose to apply a well-characterized transgenic construct, p1757, 12 which was kindly provided by Dr. Yoshihiko Yamada (Molecular Biology Section, National Institute of Dental and Craniofacial Research, Bethesda, MD). This plasmid incorporates nearly 1 kb of the proximal rat Col2a1 promoter (−977 to +110) and part of the first intron containing a known enhancer sequence12,13 ( Fig. 1A ). While these sequences are of rat origin, the construct has been used to generate chondrocyte-conditional expression within multiple transgenic mouse strains.12,14-16 The cDNAs for fLuc and mCherry flanking the Thosea asigna virus 2A (T2A) “self-cleaving” peptide sequence were cloned into pCMV6-XL5 (Origene, Rockville, MD) upstream of the human growth hormone polyadenylation (hGHpolyA) signal site. 2A peptides permit stable, stoichiometric co-expression of 2 cDNAs by the same regulatory elements in transgenic mice 17 and rats. 18 The fLuc-T2A-mCherry-hGHpolyA ORF was then inserted at the BamHI site of the p1757 plasmid, between a rabbit β-globin intronic sequence and the Col2a1 enhancer element ( Fig. 1A ). The accuracy of the final construct, named p1727-fL2AmC, was confirmed by Sanger sequencing prior to use for transgenesis.
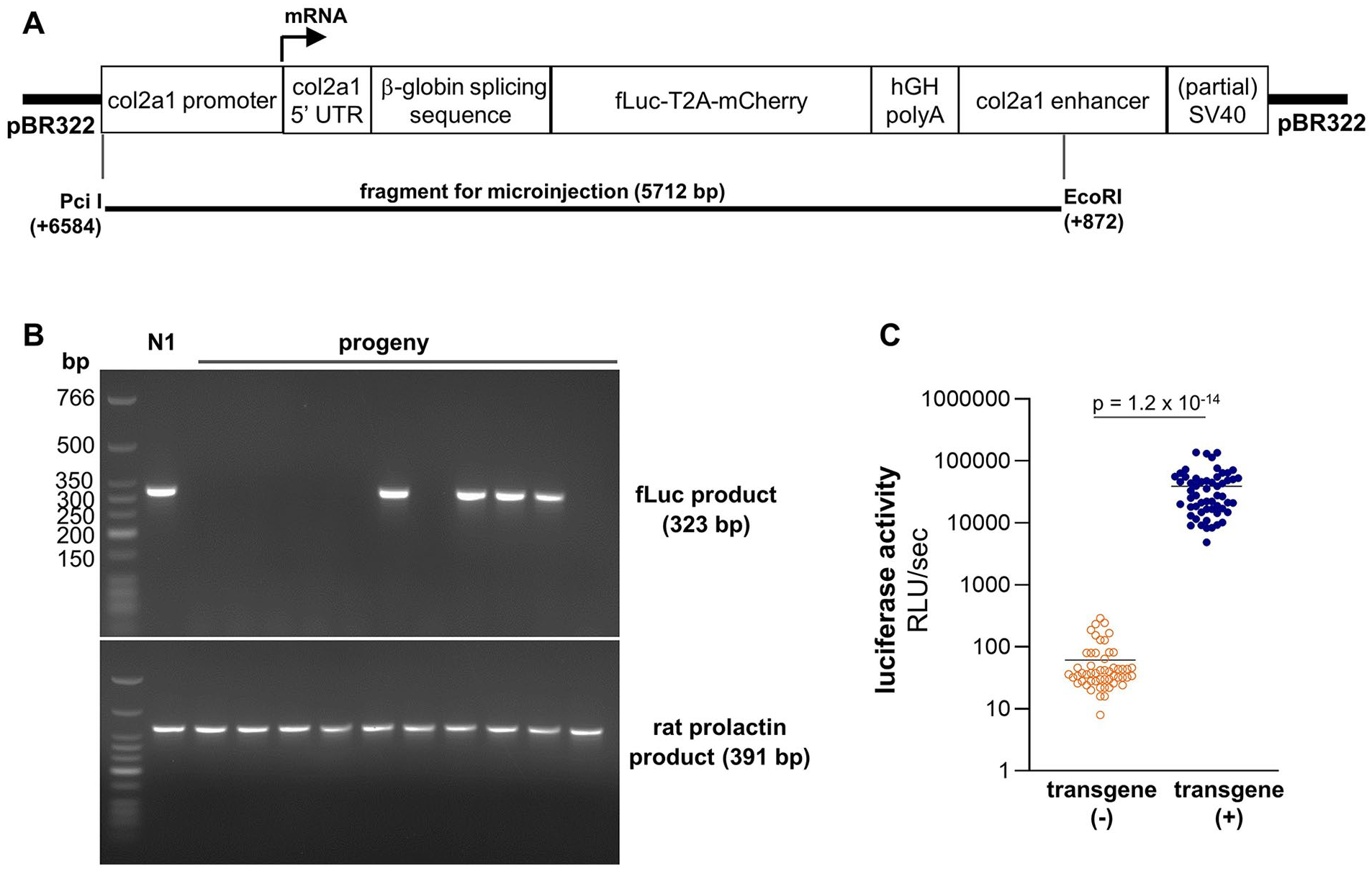
Transgenic construct and genotype identification. (
Generation of Transgenic Line LEW-Tg(Col2a1-luc,-mCherry)Rmpt
All animal studies were conducted as pre-approved by the Institutional Animal Care and Use Committees at the University of Arkansas for Medical Sciences (UAMS) or the University of Michigan (UMich). Construct p1727-fL2AmC was digested by PciI and EcoRI to generate the fragment for pronuclear injection ( Fig. 1A ), verifying complete digestion by agarose gel electrophoresis. The restriction enzyme digest was delivered to the UMich Transgenic Animal Modeling Core, who purified the digest and performed pronuclear injections into Lewis rat embryos (LEW/Crl; Charles River, Wilmington, MA). Tail snips from the resulting pups were sent to UAMS for genotyping, which was performed by PCR (polymerase chain reaction) using the following primers specific for the transgene or for rat prolactin (Prl): For_fLuc: 5′-AGACGCCAAAAACATAAAGAAAGGCCCGGC-3′, Rev_fLuc: 5′-TATAAATGTCGTTCGCGGGCGCAACTGCAA-3′ (amplicon = 323 bp); For_Prl: 5′-GCTTCTGAGCAATGACACCA-3′, Rev_Prl: 5′-ATTCCAGGAGTGCACCAAAC-3′ (amplicon = 391 bp). This genotyping strategy produced distinct bands that were not detected in transgene-negative littermates ( Fig. 1B ).
As a rapid measure of rat genotype, a 2-mm biopsy of the pinna was taken at the time of weaning using an aseptic ear punch. Biopsies were transferred to 2-ml round bottom tubes containing 200 µl GLO lysis buffer (Promega, Madison, WI) and a 5-mm stainless steel bead. The biopsies were homogenized at 25 Hz for 2 minutes using a Tissue Lyser II (Qiagen, Germantown, MD) equipped with microcentrifuge tube adapter, and the resulting lysate was centrifuged at 13,000 rpm for 5 minutes (4°C). A 100-µl aliquot of supernatant was then used to measure luciferase activity (as in Fig. 1C ).
The established line, named LEW-Tg(Col2a1-luc,-mCherry)Rmpt (RRID: RGD_149735895), is in the process of being deposited with the NIH-funded Rat Resource & Research Center at the University of Missouri (RRRC; http://www.rrrc.us/) to facilitate distribution of the strain (RRRC# 00942).
Noninvasive Bioluminescence Imaging
Candidate founders and their progeny were imaged on either an IVIS 200 (Xenogen, Hopkinton, MA) for whole-body imaging or an IVIS Lumina XRMS (PerkinElmer, Waltham, MA) for x-ray co-registration. After anesthetizing using isoflurane, the skin over areas of interest were typically shaved to remove fur and improve signal detection. For most applications, rats were injected intraperitoneally (i.p.) with the fLuc substrate
Ex Vivo Imaging
The following tissues were harvested at 5, 8, 12, and 23 weeks of age: hind paw with skin removed; ear biopsy (4 mm ∅); menisci; femoral condyles; tibial plateau; anterior joint capsule with patella and extensor mechanism; sternum; 2 caudal vertebrae; vastus lateralis muscle; subcutaneous fat; kidney (bisected). Explants were rinsed in PBS then immersed in a bath of 0.5 mg/ml
Molecular Extraction
fLuc mRNA levels and protein activity were measured in tissues collected at the same timepoints as for ex vivo imaging, except that whole knees were analyzed as a single sample. Tissues were flash frozen immediately after dissection, then transferred to stainless steel grinding jars containing 2-cm steel balls (Qiagen) that were precooled in liquid nitrogen. The tissues were pulverized frozen at an oscillation frequency of 25 Hz for 60 seconds using a TissueLyser II. The resulting powder was divided into fractions for RNA extraction or luciferase activity assay. Total RNA was extracted from pulverized tissues using TRI Reagent RT (Molecular Research Center, Cincinnati, OH), purifying RNA within the aqueous fraction using an RNeasy Mini kit with DNase I digestion step (Qiagen). fLuc protein was extracted from tissue homogenates using 500 µL of GLO lysis buffer for 100 mg of pulverized tissue. After sample incubation at room temperature for 1 hour with frequent mixing, lysates were centrifuged at 13,000 rpm for 5 minutes (4°C) and supernatants were used for quantification of luciferase activity.
qRT-PCR
For cDNA synthesis, 1 µg of total RNA was reverse transcribed using a High Capacity Reverse Transcription Kit (Applied Biosystems, Waltham, MA). Real-time, quantitative PCR was performed on a StepOnePlus™ instrument (Applied Biosystems) using predesigned TaqMan assays for fLuc (assay ID# Mr03987587_mr; amplicon = 70 bp) and hypoxanthine phosphoribosyltransferase 1 (Hprt1) (assay ID# Rn01527840_m1; amplicon = 64 bp). Relative expression levels for each gene were normalized to Hprt1 using the -ΔΔCT method.
Luciferase Activity
Luciferase activity was determined using the Bright-GloTM Luciferase Assay System (Promega). After combining lysate supernatant and Bright-Glo substrate into a 96-well white assay plate (Corning, Corning, NY), the plate was mixed for 5 minutes and luciferase activity was measured on a Victor3 microplate reader (PerkinElmer).
Chondrogenesis Models
To validate the reporter strain for detecting orthotopic and heterotopic chondrogenesis, 2 in vivo models were employed as outlined in Figure 2 .
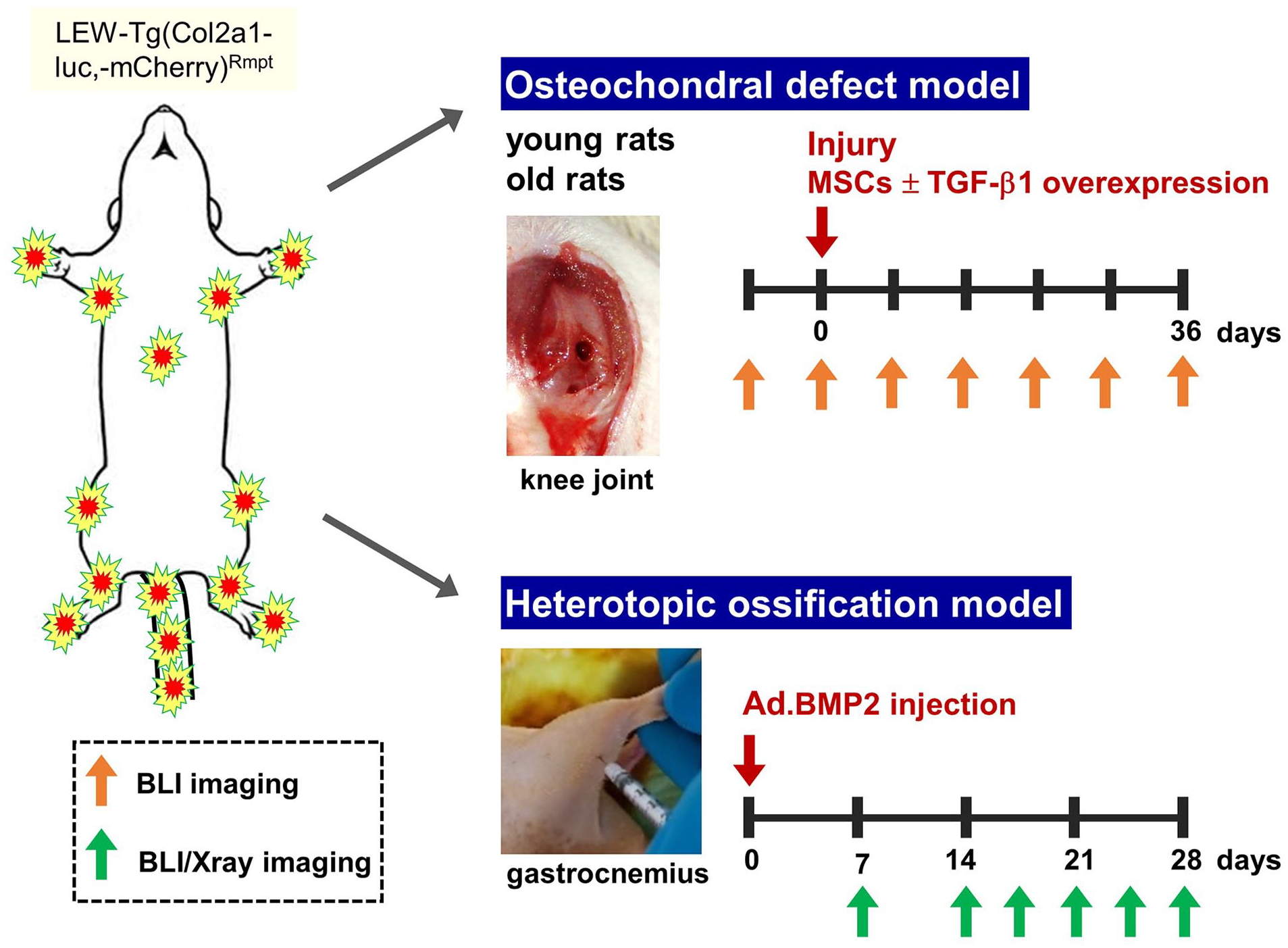
Employed models of orthotopic and heterotopic chondrogenesis. Rats were imaged longitudinally using bioluminescence alone or bioluminescence/x-ray co-registration at the frequency indicated by arrows until the specified endpoints. LEW = Lewis rat embryos; MSC = marrow stromal cell; BLI = bioluminescence imaging; TGF-β1 = transforming growth factor-β1.
Osteochondral defect model
To evaluate chondrogenesis during articular cartilage repair, we applied a bilateral osteochondral defect (OCD) model as previously described. 19 Rats were anesthetized with isoflurane, and both hind limbs were shaved and sanitized by alternating ethanol and povidone iodine scrubs. A 2-cm skin incision was made 1 cm lateral to the knee joint, so that the wound clips used for skin closure would not interfere with bioluminescence signal at early imaging timepoints. The joint capsule was opened by medial parapatellar incision, the extensor mechanism was displaced laterally, and the knee flexed to reveal the patellar (trochlear) groove. Approximately 2 mm proximal to the intercondylar notch, a 1-mm ∅ defect was made to a depth of 1 mm using a sterile 0.75-mm (inner diameter) biopsy punch (Robbins Instruments, Houston, TX). The defect was flushed with saline and either left empty or filled with a pellet of culture-expanded rat bone marrow stromal cells (MSCs; 500,000 cells/pellet). Some rMSCs were genetically modified to overexpress transforming growth factor (TGF)-β1, using adenoviral transduction. 19 Sham controls received arthrotomy without a defect. The joint capsule was closed with 5-0 Vicryl sutures (Ethicon, Raritan, NJ), while the skin incision was closed with 9-mm wound clips, which were removed 10 days after surgery. Rats were imaged by BLI at 3-4 days post-operation, then weekly for 4-5 weeks post-op. Upon euthanasia, the distal femora were processed for histology and IHC.
Heterotopic ossification model
To assess chondrogenesis associated with heterotopic ossification, we overexpressed bone morphogenetic protein (BMP)-2 within the gastrocnemius muscle, based on prior protocols.20,21 Rats were anesthetized and prepped for aseptic surgery, as detailed above. A replication-defective, E1- and E3-gene deleted adenoviral vector encoding BMP-2 under control of the human cytomegalovirus promoter (Ad.BMP-2 22 ; 2×109 viral particles in 50 µl) was injected into the approximate mid-substance of the gastrocnemius muscle. To limiting the adaptive immune response to the adenoviral vector and improve local BMP-2 overexpression, rats were given a daily subcutaneous injection of FK506 (1.0 mg/kg) and SEW2871 (0.5 mg/kg) (both from Cayman Chemical, Ann Arbor, MI) starting the day before adenovirus injection.23,24 Rats underwent noninvasive BLI and x-ray imaging at 7, 14, 17, 21, and 28 days post-injection. After 21 or 28 days, the injected muscle was harvested for histology.
Histology and IHC
Tissues were fixed for 48-72 hours using 4% paraformaldehyde (PFA) in PBS (pH 7.4) at 4°C with constant mixing. For whole knees, the joint capsule was opened near the patella to better expose the intra-articular tissues. Skeletal tissues were then decalcified in 15% EDTA (ethylenediaminetetraacetic acid) in PBS (pH 7.4) for 2-4 weeks at 4°C with constant mixing, bisecting larger tissues midway through decalcification. Tissues were then processed for paraffin embedding, and 5-µm sections were stained by Toluidine Blue or Safranin O with Fast Green, using protocols from the University of Rochester Center for Musculoskeletal Research.
Addition sections were immunostained for mCherry. Antigen unmasking was performed in 10 mM sodium citrate buffer, 0.05% Tween-20 (pH 6.0) at 65°C for 2 hours in a water bath. Endogenous peroxidases were quenched using BLOXALL (Vector Laboratories, Burlingame, CA). After additional blocking using 5% normal goat serum, 1% bovine serum albumin (BSA) in PBS for 1 hour, slides were incubated overnight (4°C) with a 1:300 dilution of polyclonal rabbit anti-mCherry IgG (ab167453; Abcam, Waltham, MA) in blocking buffer. Antigens were visualized using a Vectastain Elite ABC Rabbit IgG kit (Vector Laboratories) with 3,3′-diaminobenzidine as substrate, and sections were counterstained for 10 seconds with 0.5% Methyl Green.
Statistical Analysis
Unless specified elsewhere, experiments were performed using littermates from the same generation of the transgenic line. Subject age(s) and replicate numbers for each experiment are reported within the figure legends. Time-dependent changes in bioluminescence were determined by repeated measures analysis of variance (ANOVA), with any influence of sex or genotype determined by 2-way ANOVA. Specific pairwise comparisons between timepoints or treatments were performed using Tukey’s multiple comparisons test. Outlier animals excluded from analysis were first identified using Grubb’s test. Statistical significance was denoted when P < 0.05, though specific P values are reported where space allows. Statistical analyses were performed using either Microsoft Excel 2016 or GraphPad Prism 8.
Results
The Transgenic Strain Displays Cartilage-Localized Dual Reporter Expression
Pronuclear injections led to 5 transgene-positive pups, with 3 displaying luminescent signal localized near cartilaginous structures (as in Fig. 3B ). To select one founder for establishing a transgenic line, their offspring were characterized by longitudinal bioluminescence imaging (BLI; Fig. 3 ) combined with ex vivo tissue analysis ( Fig. 4 ). To date, 3 Lewis-backcrossed generations have been produced from the chosen founder displaying a stable reporter expression pattern. No abnormalities have been noted in growth rate (Suppl. Fig. S1), skeletal morphology, or longevity compared to wild-type littermates. During strain characterization, we established that a 2-mm biopsy of the pinna taken during notching for identification can rapidly and reliably identify transgene-positive pups, as luciferase activities from heterozygous biopsies were approximately 3 orders of magnitude above background ( Fig. 1C ).
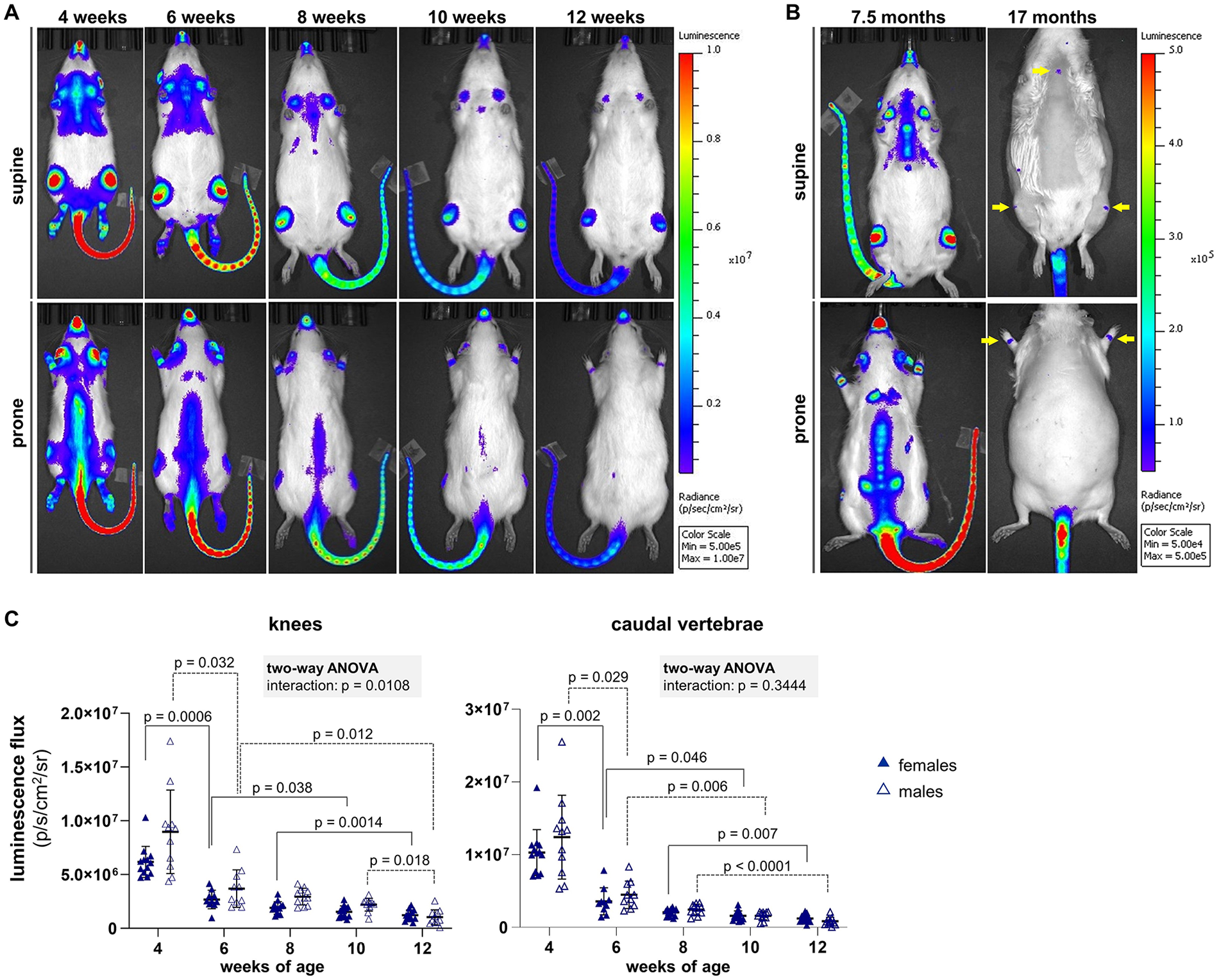
Longitudinal bioluminescence imaging in vivo. (
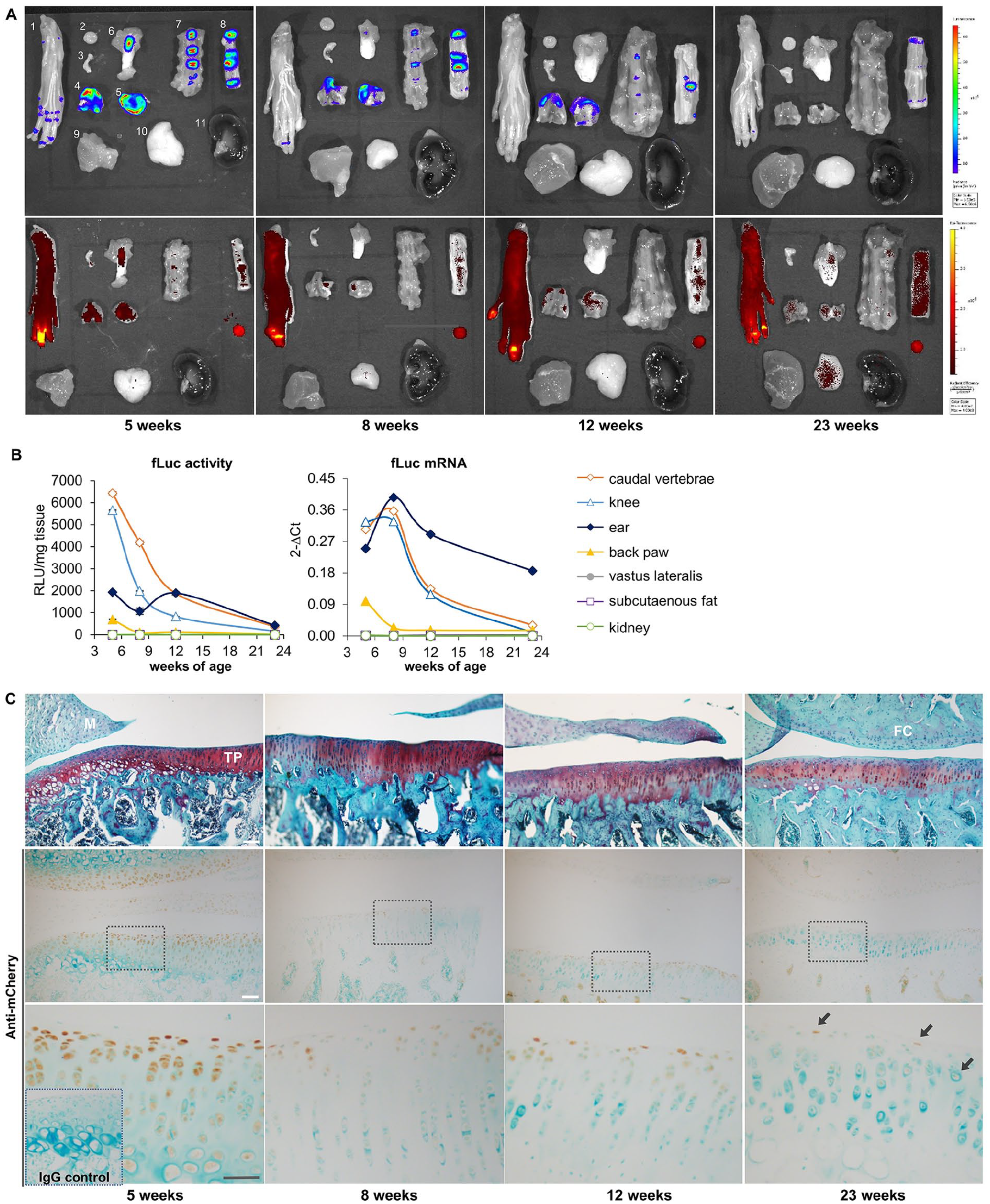
Reporter expression during skeletal maturation. (
Longitudinal, noninvasive BLI revealed a fLuc expression pattern that closely reflected chondrogenic activity during postnatal development (
Fig. 3A
). Bioluminescent signal was intense at 1 month of age, with hotspots overlying the synovial joints of the appendicular skeleton, spine, sternum, nose, and pinnae. The intensity of the signal diminished considerably with continued development up to 3 months of age but remained well above background. Sensitivity was improved by shaving the fur above regions of interest (ROIs;
Fig. 3B
). Luminescence signal further diminished with advanced aging yet was still detected above background in older rats (
Fig. 3B
,
To better define the localization of fLuc and mCherry expression, both skeletal and nonskeletal tissues were harvested at different ages for ex vivo imaging. Overall bioluminescence signal was reduced in explants compared to the same tissues measured noninvasively (
Fig. 4A
,
To evaluate mCherry expression at the individual cell scale, IHC was performed on paraffin-embedded tissues, including sections from the knee ( Fig. 4C ). Abundant immunostaining was detected throughout the full thickness of articular cartilage and the articulating surface of menisci at 5 weeks of age. The fraction of immunopositive cells declined at subsequent timepoints; by 6 months of age, only a small subset of chondrocytes in the superficial zone of articular cartilage were immunopositive. Specific immunostaining was not detected within bone or ligament cells or within any subpopulation in sections of skeletal muscle, subcutaneous adipose tissue, or kidney (not shown). Initial attempts to detect mCherry fluorescence in PFA-fixed, EDTA-decalcified, frozen tissue sections were not successful, but may be possible with further optimization.
The Transgenic Strain Can Report Chondrogenesis in Response to Osteochondral Injury
To determine whether the strain can report de novo chondrogenesis following skeletal injury, an established model of articular cartilage repair was employed, involving an OCD made in the rat patellar groove ( Fig. 2 ). 19 When OCD or sham surgery was performed on 6-month-old reporter rats, time-dependent changes in knee signal were measured for all groups receiving arthrotomy ( Fig. 5A ). Signal trended toward a decrease from baseline at day 3 post-injury, followed by a subsequent elevation in knee signal by day 7 that lasted until at least 4 weeks post-injury, when joints were harvested. To reduce variability among rats (as in Suppl. Fig. S2), signal from an ROI placed over the proximal knee was normalized by that from an ROI placed over the caudal vertebrae ( Fig. 5B ). Using this approach, knees treated with culture-expanded rMSCs displayed higher signal than sham controls as early as day 3 post-surgery (P = 0.026), a difference that persisted until the experimental endpoint ( Fig. 5C ). While empty defects had higher signal than sham knees at day 7 (P = 0.015), differences between these 2 groups did not meet the threshold of significance at later timepoints (P = 0.067 at day 14, P = 0.084 at day 21, and P = 0.256 at day 31). A difference between empty and MSC-treated defects was also noted at day 7 (P = 0.007), with a trend toward higher signal for MSC-treated defects at day 14 (P = 0.074).
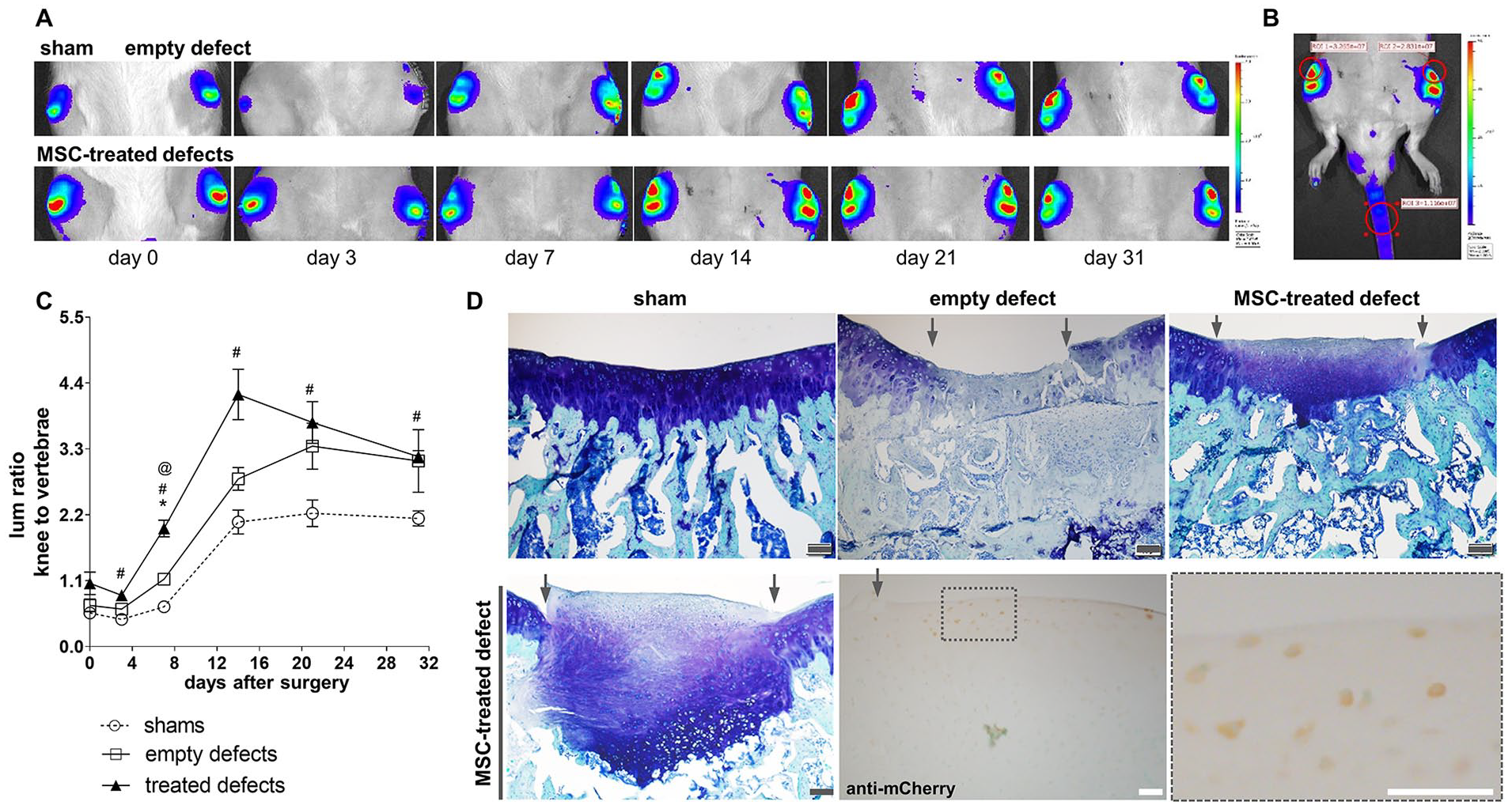
Reporter response to OCD repair: young adult subjects. (
The limited differences in knee bioluminescence between empty defect and MSC-treated groups seemingly contradicted endpoint histological analysis of proteoglycan deposition (
Fig. 5D
,
In addition to chondroprogenitor cells undergoing differentiation within the defects, local chondrocytes may also have contributed to the knee signal shown in
Figure 5
. This includes not only chondrocytes within intact articular cartilage but also those within the nearby growth plates (GPs), which do not close in skeletally mature rats (
Fig. 6A
). In naïve rats at 6 months of age, bioluminescence signal was still strong from the knees following i.p. injection of
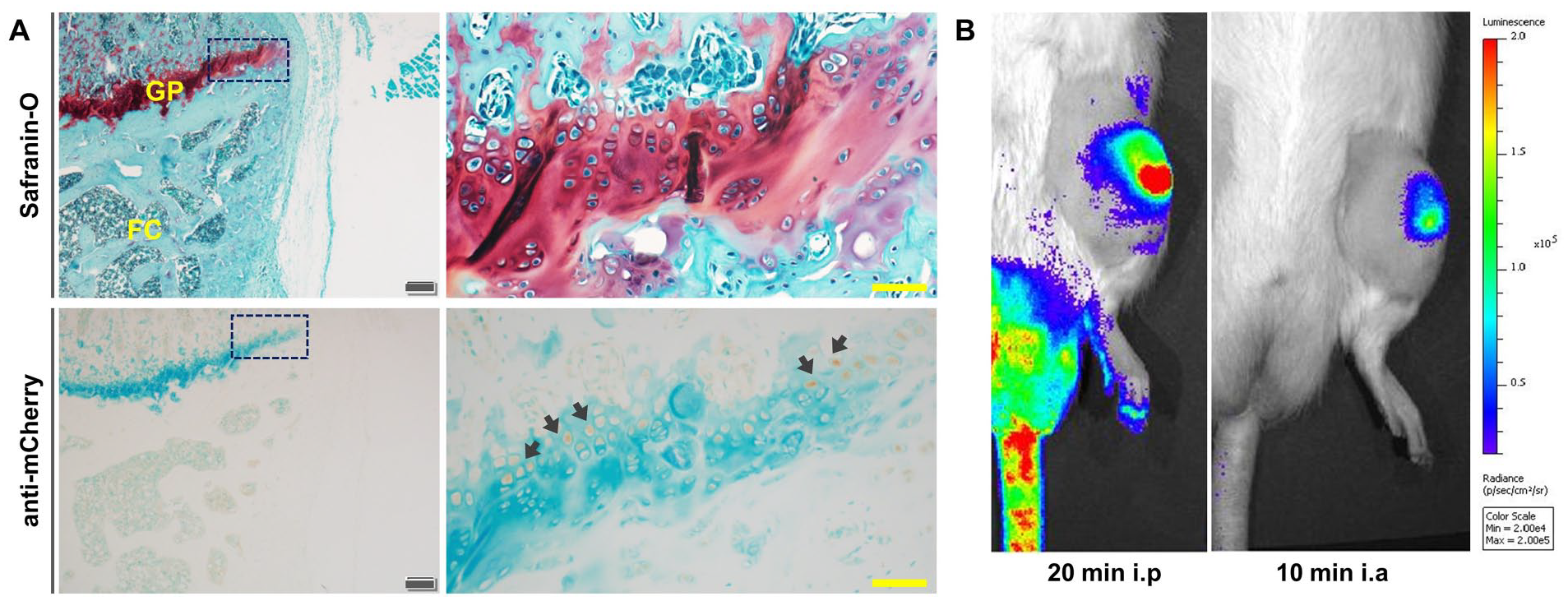
Contribution of epiphyseal plates to reporter levels, substrate delivery route to knee BLI. (
Using the i.a. injection approach, we examined whether a chondrogenic response to MSC treatment could still be produced in aged reporter rats having low basal reporter activity. As demonstrated by representative BLI ( Fig. 7A ) and cohort quantification ( Fig. 7B ), implantation of young adult, wild-type MSCs overexpressing TGF-β1 led to a time-dependent increase in intra-articular bioluminescent signal that plateaued between 2-3 weeks post-injury. Of note, the variance in reporter signal was higher than in these 2-year-old rats than the knee signal without vertebrae normalization from 6-month rats. IHC for mCherry within the repair tissue revealed immunopositive cells within both fibrocartilage repair tissue near the articular surface as well as hypertrophic chondrocytes in the osseous zone undergoing endochondral ossification ( Fig. 7C ).
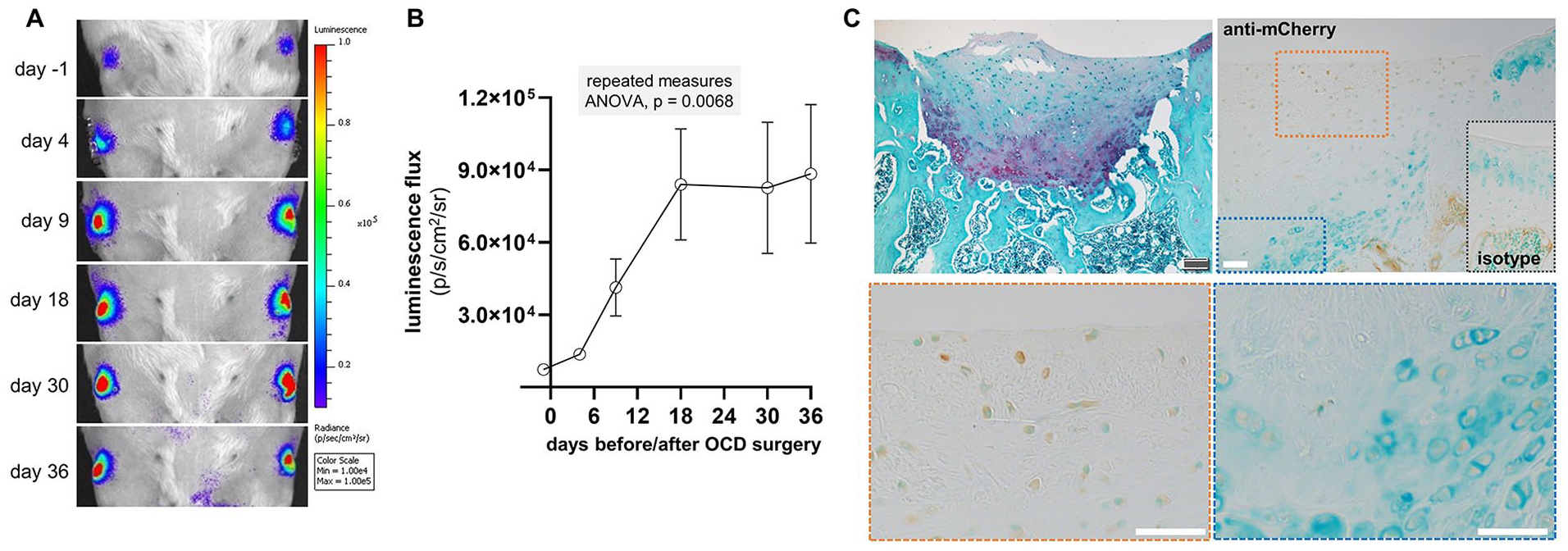
Reporter response to OCD repair: aged subjects. (
The Transgenic Strain Can Report Chondrogenesis Associated With Heterotopic Ossification
To determine whether the strain can report ectopic chondrogenesis known to be associated with heterotopic ossification in skeletal muscle, we exploited the published observation that such ossification can be induced by local overexpression of BMPs.20,21,27 Delivery of Ad.BMP-2 to the gastrocnemius muscle of reporter rats, when combined with suppression of an adaptive immune response to the viral vector using FK506 and SEW2871, consistently led to ossification at the injection site within 4 weeks of adenovector delivery, as revealed by longitudinal x-ray imaging (
Fig. 8A
,
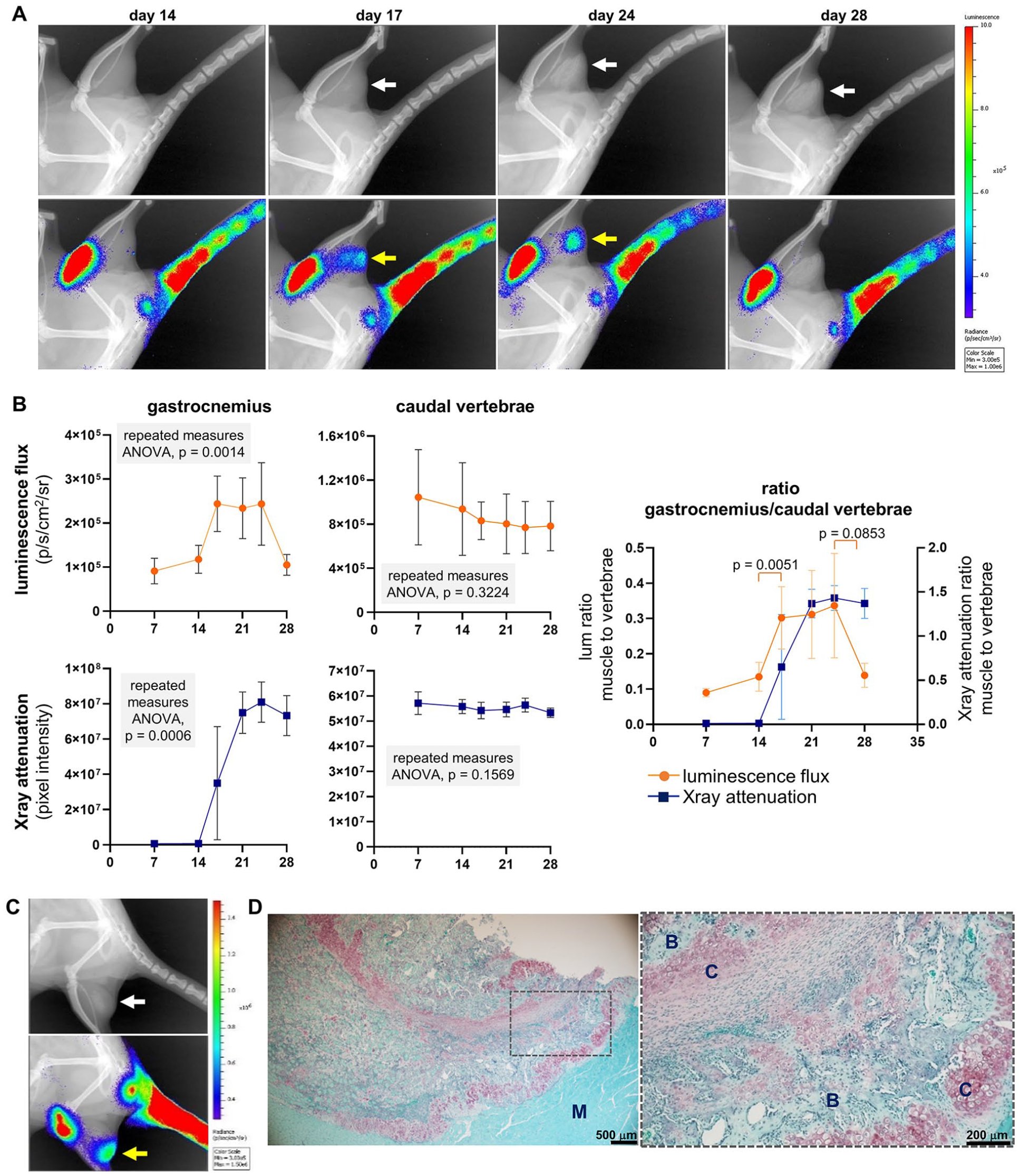
Reporter response to induced heterotopic ossification. (
Discussion
Here we present a chondrogenesis reporter rat that can complement ongoing efforts within the skeletal tissue engineering and regenerative medicine (TERM) community. Rather than reporting the presence of relatively quiescent chondrocytes within mature cartilage tissue, our data demonstrate that this strain predominantly reports areas of active chondrogenesis. Accordingly, the strain can be used in the preclinical evaluation of treatments that may alter chondrogenesis to stimulate or enhance cartilage regeneration, to catalyze endochondral ossification for the purpose of bone regeneration, or to inhibit ectopic chondrogenesis for the purpose of preventing heterotopic ossification. An important advantage of this animal model is the ability to nondestructively evaluate chondrogenic activity over time in the same subject animals, allowing correlation of short-term chondrogenic effects with long-term regenerative outcomes. While mouse models displaying chondrocyte-specific expression of fluorescent or bioluminescent reporter genes have been reported previously,7,28-30 to the best of our knowledge, this is the first such rat model available to the field.
To generate a rat strain that reports chondrogenic activity, we chose to employ regulatory elements from Col2a1: this key component of the cartilage extracellular matrix is expressed specifically by chondrocyte-lineage cells in the postnatal skeleton, as demonstrated by prior transgenic mouse strains.28,31 However, the exact choice of regulatory elements, particularly the length of the Col2a1 promoter, has been reported to dictate transgene expression during chondrogenesis within adult versus embryonic tissues. 32 Here we used approximately 1 kb of the rat promoter, similar to the length of the murine promoter used to generate a Col2a1-CreERT2 mouse 31 that was capable of identifying chondrocytes in the fracture callus of adult mice. 30 In contrast, transgenes using longer sequences of the Col2a1 promoter were only expressed by chondrogenic cells during development. 32 As demonstrated above, fLuc and mCherry were expressed not only during skeletal development but also within hyaline cartilage formed in response to skeletal injury.
Characterization of the reporter strain confirmed observations from prior mechanistic studies of chondrogenesis during development, aging, and musculoskeletal injury. For example, longitudinal BLI demonstrated the abundance of chondrogenic activity during skeletal development relative to skeletal maintenance upon maturation
33
: luciferase activity was more than an order-of-magnitude higher in weanlings than young adult rats (
Fig. 1
). Results from the cartilage repair model confirm that exogenous MSCs delivered to a defect stimulate healing at least in part through activation of chondrogenesis by endogenous progenitors recruited to these defects (
Figs. 5
and
7
). This observation is consistent with the current consensus on the regenerative actions of culture-expanded MSCs.34,35 The initial, transient decrease in bioluminescent signal (
Fig. 5A
There are important limitations to the reporter strain for certain applications. Differences in signal between the lumbar and caudal vertebrae demonstrate the influence of target tissue depth on BLI sensitivity. For assessment of chondrogenesis within synovial joints, the signal from any nearby epiphyseal plates can contribute to the total knee signal, potentially masking effects specific to the intra-articular tissues. To circumvent this issue, intra-articular injection of substrate can be used (as in Fig. 6 ). However, frequent i.a. injections for the purpose of longitudinal imaging may have independent effects on joint response. While use of fine needles should mitigate trauma to the joint capsule, this potential limitation should be considered during study design. The epiphyseal plate signal should also be considered when using the strain to study the chondrogenic phase of endochondral bone repair. Accordingly, bone injury models may be limited to the diaphysis of long bones or to the cranium, which develops by intramembranous bone formation but can repair through an endochondral pathway. 45 Finally, while imaging tissues ex vivo can help localize bioluminescent signal, this choice can also limit signal strength and, thereby, assay sensitivity. The presence of atmospheric oxygen has been demonstrated as important for ex vivo BLI 26 ; if tissues cannot be explanted and imaged quickly after substrate injection performed prior to euthanasia, preliminary experiments are required to optimize substrate exposure prior to imaging in open air (as in Fig. 4A ).
There are additional applications for this strain that were not considered here. Beyond their use as the subject of chondrogenesis-modifying interventions, these rats have the potential to serve as a source of cells or tissues for transplantation into nonreporter subjects, to study the direct contribution of donor cells to chondrogenesis. Promising cell types for skeletal TERM include chondrocytes, tissue-resident stem/progenitor cells (e.g., bone marrow, adipose, synovium, ligament/tendon), and induced pluripotent stem cells derived from transgene-positive somatic cells. For studies using the reporter rats as subjects, it may be possible to isolate chondrogenic cells from tissue digests by fluorescence-activated cell sorting, providing a more focused cell population for analysis; however, we have not yet confirmed whether mCherry expression is sufficient to identify chondrogenic cells using fluorescence. Ongoing studies are addressing these possibilities.
Supplemental Material
sj-jpg-1-car-10.1177_19476035211057243 – Supplemental material for A Transgenic Rat for Noninvasive Assessment of Chondrogenesis in Vivo
Supplemental material, sj-jpg-1-car-10.1177_19476035211057243 for A Transgenic Rat for Noninvasive Assessment of Chondrogenesis in Vivo by Elisabeth Ferreira, Landon B. Gatrell, Luke Childress, Hong Wu and Ryan M. Porter in CARTILAGE
Supplemental Material
sj-jpg-2-car-10.1177_19476035211057243 – Supplemental material for A Transgenic Rat for Noninvasive Assessment of Chondrogenesis in Vivo
Supplemental material, sj-jpg-2-car-10.1177_19476035211057243 for A Transgenic Rat for Noninvasive Assessment of Chondrogenesis in Vivo by Elisabeth Ferreira, Landon B. Gatrell, Luke Childress, Hong Wu and Ryan M. Porter in CARTILAGE
Footnotes
Acknowledgements and Funding
We are most grateful to the late Yoshihiko Yamada (Molecular Biology Section, National Institute of Dental and Craniofacial Research, Bethesda, MD) for providing the rat Col2a1 construct p1727. We thank Thomas L. Saunders and colleagues at the UMich Transgenic Animal Modeling Core for their technical expertise in generating the candidate founders. Charles A. O’Brien (UAMS) provided valuable advice on managing and characterizing the transgenic strain.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded in part by the National Institute of Arthritis Musculoskeletal and Skin Diseases (AR069253, AR071560), the National Institute of General Medical Sciences (GM125503), the Arkansas Biosciences Institute, and the UAMS Bone & Joint Initiative.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All animal use was conducted as pre-approved by the Institutional Animal Care and Use Committees at the University of Arkansas for Medical Sciences or the University of Michigan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
