Abstract
Study Design
Meta-analysis.
Objectives
Spinal tumors and metastases remain a challenge for spine surgeons, with a significant risk of perioperative blood loss, which might require blood transfusions and lead to increased complications. Tranexamic acid (TXA) is an antifibrinolytic agent widely used in various surgical procedures; however, its efficacy in spinal oncology surgery remains unclear. This meta-analysis aims to evaluate the clinical effectiveness of TXA on perioperative outcomes, mainly blood loss, among patients with oncological spines undergoing surgery.
Methods
PubMed, Scopus, and Web of Science were systematically searched from inception for eligible articles. We included studies assessing TXA vs no TXA or placebo on perioperative outcomes among patients with spinal tumors or metastases.
Results
After a comprehensive search, seven studies were included. Blood loss (MD −111.75 mL; 95% CI −217.08 to −6.43;
Conclusion
Retrospective data suggest TXA may reduce blood loss in spinal tumor surgery. However, its effect on hospital stay and complications remains uncertain. Despite appearing safe, evidence remains limited by bias and heterogeneity. High-quality RCTs are needed to confirm its efficacy and define clinical guidelines.
Introduction
Spinal surgery in oncological patients, including those with primary or metastatic tumors, remains a significant challenge due to the anatomical complexity of the spine, its rich vascularity, and the associated risk of substantial intraoperative blood loss. 1 For instance, spinal decompression is frequently performed in patients with metastatic disease to alleviate spinal cord compression, improve neurological function and quality of life, and stabilize the spinal column. 2 These metastases may originate from a variety of primary malignancies, including breast, lung, prostate, endocrine, and hematologic cancers. 3 Despite advances in surgical techniques and hemostatic technologies aimed at minimizing intraoperative bleeding, excessive blood loss continues to be a major concern, potentially leading to longer hospital stays, increased perioperative complications, and greater transfusion requirements. 4
Various antifibrinolytic agents were employed in surgical procedures. One of them, tranexamic acid (TXA), became well-known due to its potential to reduce perioperative bleeding and the need for transfusion. 5 Tranexamic acid (TXA) is a synthetic derivative of the amino acid lysine that acts as an antifibrinolytic agent. It exerts its effect by reversibly binding to lysine receptor sites on plasminogen, thereby inhibiting its conversion to plasmin, the enzyme responsible for fibrin degradation. By blocking this activation, TXA helps stabilize fibrin clots and reduces fibrinolysis, leading to decreased blood loss during and after surgical procedures. 6 The effectiveness of TXA was verified in various fields of medicine, including hepatic, liver, cardiac, or spine surgery, demonstrating a significant reduction in perioperative bleeding and transfusion needs.7-9 However, while the role of TXA has been validated in various spinal procedures, these mostly focused on degenerative, trauma, or deformity populations. 10 Meanwhile, the effectiveness of TXA among spinal tumor and metastasis subpopulations remains relatively unexplored.
Among the patients with spinal tumors, the risk of increased perioperative bleeding is significant due to the high vascularity of the tumors. Excessive bleeding can compromise hemodynamic stability, prolong surgical duration, and increase the risk of postoperative complications such as thromboembolism, delayed recovery, and infection. 11 Therefore, effectively minimizing intraoperative blood loss and reducing transfusion requirements remains a critical priority in this complex spine patient population. The use of antifibrinolytic agents, such as tranexamic acid, may offer a promising strategy to address this challenge.
However, while these benefits of TXA were validated in various fields of medicine including spinal surgery, the use of TXA among oncological spine patients remains controversial. Currently, studies show mixed results on blood loss and transfusion requirements; therefore, there is no clear answer whether TXA significantly impacts bleeding. 12 Moreover, concerns regarding complications are still present.
Therefore, this systematic review and meta-analysis aim to evaluate and compare the effectiveness of TXA on perioperative outcomes among patients undergoing spinal surgery for tumors and metastases. We aim to assess various clinical endpoints, including blood loss, operation time, transfusion parameters, length of hospital stay, and complications. Through this analysis, we aim to answer whether TXA significantly impacts spinal surgery outcomes.
Methods
This systematic review and meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines and the Cochrane Handbook.13,14 This meta-analysis was prospectively registered in the PROSPERO database.
Search Strategy
We performed a systematic search across three major electronic databases: PubMed, Scopus, and Web of Science. The search was performed from inception to February 2025. No filters or restrictions were applied during the database search to maximize the capture of the articles. The search strategy included a combination of keywords related to “tranexamic acid,” “TXA,” “antifibrinolytic agents,” “aprotinin,” “spine surgery,” “spinal surgery,” “spinal decompression,” “spinal fusion,” metastasis, tumor, neoplasm, oncology, or metastatic. Full search string is presented in the Supplemental Table 1.
Eligibility Criteria
We included studies if they met the following criteria: study design was one of: randomized controlled trials (RCTs), case-control, prospective, or observational cohort studies; included patients undergoing spine surgery for spinal tumor or metastases; compared the TXA group to the non-TXA group; reported at least one outcome of interest; provided information on dose and route of TXA.
We excluded case reports, reviews, or animal studies; trials with overlapping patient populations; studies with insufficient data for meta-analysis; and studies with unclear or unstandardized TXA dose.
Study Selection
Study selection was performed by two reviewers who worked independently. The first phase of screening involved title and abstract selection, and the second phase involved full-text assessment for final eligibility. Discrepancies between the reviewers were resolved through mutual discussion or by consulting a third reviewer. Zotero software was used for screening.
Data Extraction
Data extraction was performed by two authors independently. Extracted information included study characteristics (author, publication year, country of origin), patient demographics (sample size, age, primary diagnosis), surgical details (procedure type, TXA dosage, and administration), and outcomes (intraoperative blood loss, proportion of patients receiving transfusion, transfusion volume, operative time, length of hospitalization, complications, postoperative drain volume and duration).
Risk of Bias Assessment
The risk of bias was assessed using the Risk Of Bias In Non-randomised Studies—of Interventions (ROBINS-I) for observational studies. 15 Two independent reviewers performed a quality assessment. Studies were categorized as having a low, moderate, or serious risk of bias across all seven domains of ROBINS-I. The results of the quality assessment were visualized with the robvis software. 16
Statistical Analysis
We applied a random-effects model meta-analysis. Continuous variables (eg, blood loss, operation time, hospital stay) were reported with mean differences (MD) with 95% confidence intervals (CIs). If different measurement scales were used, standardized mean difference (SMD) was applied. Binary outcomes (eg, complications, transfusion events) were reported with odds ratios (OR) with 95% CIs.
Results
A systematic search was performed from database inception. We retrieved a total of 1645 records from 3 databases. More specifically, we found 461 records in PubMed, 753 in Scopus, and 431 in Web of Science databases. Screening software marked 6 retracted articles and 669 duplicate entries, which were removed before the proper screening process. Title and abstract screening was performed across 970 records by two authors independently. We excluded 955 ineligible records, and 15 studies were assessed in a full-text screening process. We removed studies with overlapping populations (n = 1), wrong control (n = 2) or intervention (n = 2), single-arm studies (n = 2), and those without a clear dose of TXA (n = 2). Furthermore, one study was found in the manual search. Finally, 7 studies were included in this systematic review and meta-analysis.18-24 A detailed search process is available in Figure 1. PRISMA Flow Diagram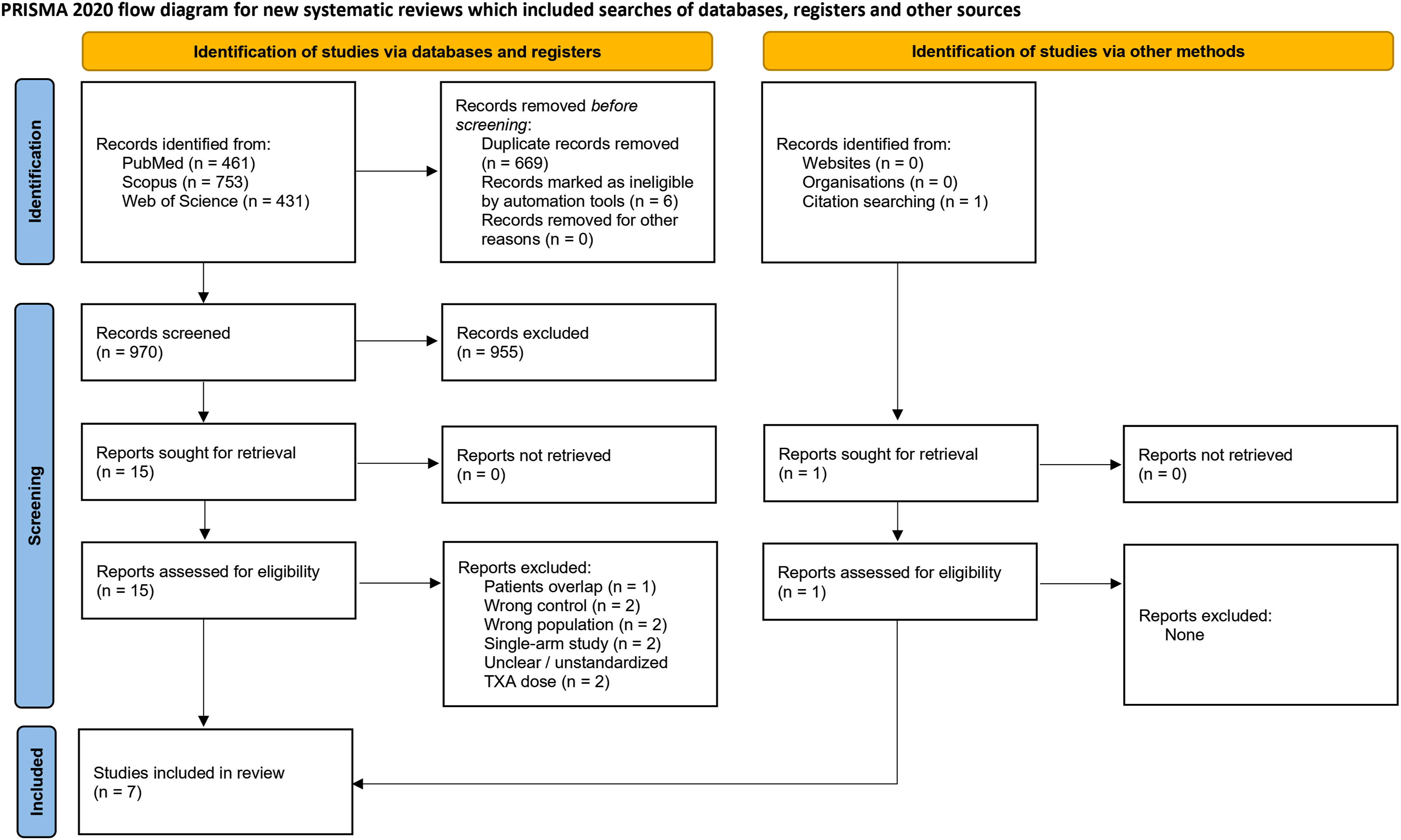
Baseline Demographic Information on Included Studies
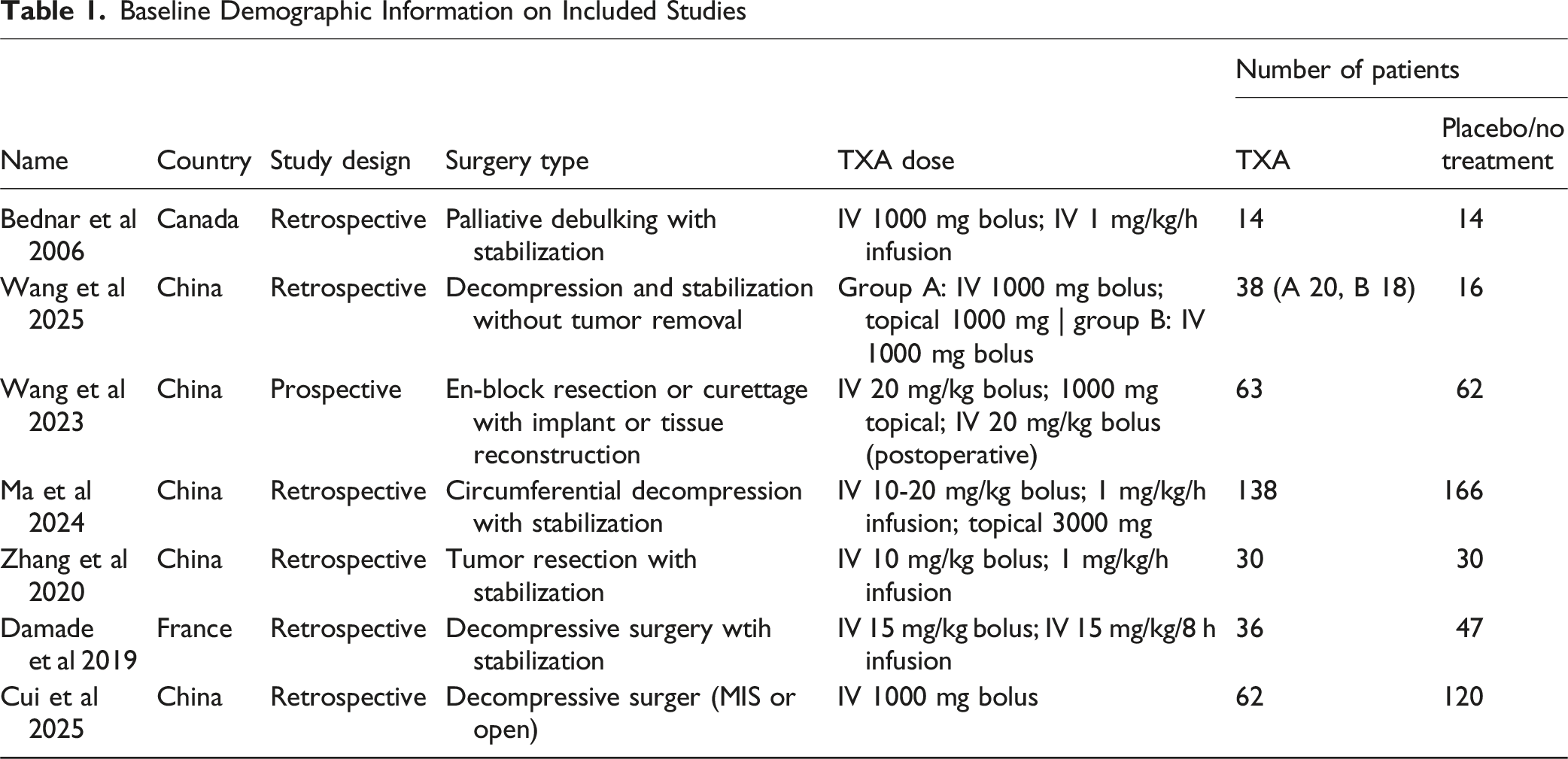
Most common diagnoses included breast, lung, lymphoma, myeloma, or unknown origin; however, diagnosis was not fully reported in all studies (Supplemental Table 3).
Intraoperative Blood Loss
Intraoperative blood loss was significantly lower in the TXA group (MD −111.75 mL; 95% CI −217.08 to −6.43; Forest Plot for Intraoperative Blood Loss Volume Showing Significantly Lower in the TXA Group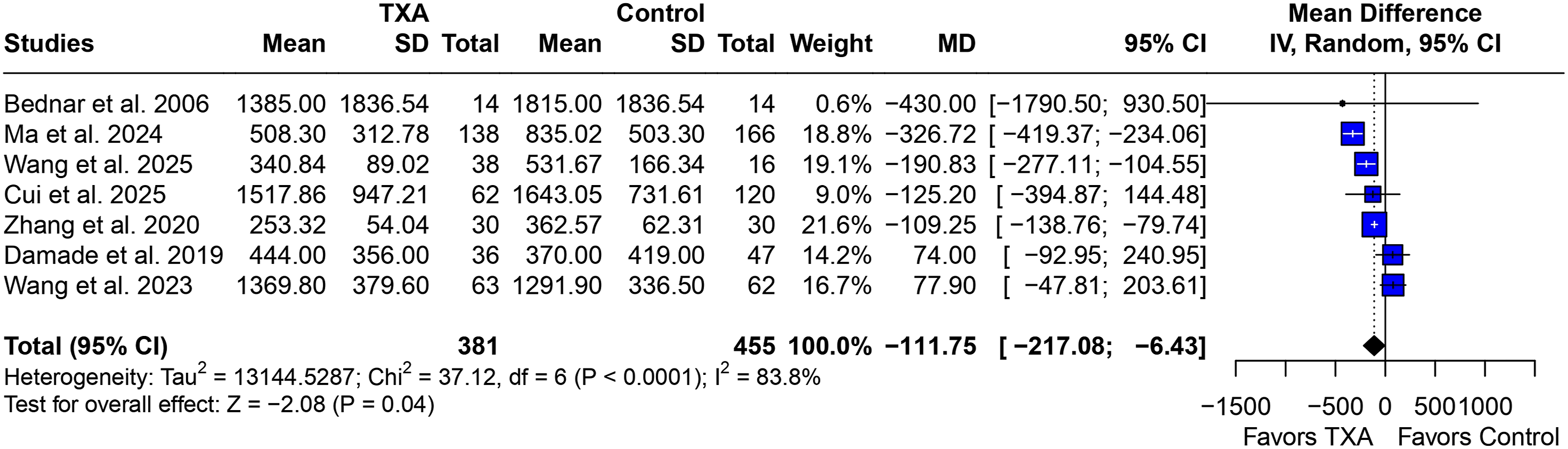
Both surgery subgroups yielded insignificant results, and there was no significant difference between them (
Postoperative Drain Volume and Duration
All analyzed studies were without ERAS. The volume of postoperative drain was significantly reduced in the TXA group (MD −90.49 mL; 95% CI −150.17 to −30.81; Forest Plot for Postoperative Drain Volume. The Volume of Postoperative Drain was Significantly Reduced in the TXA Group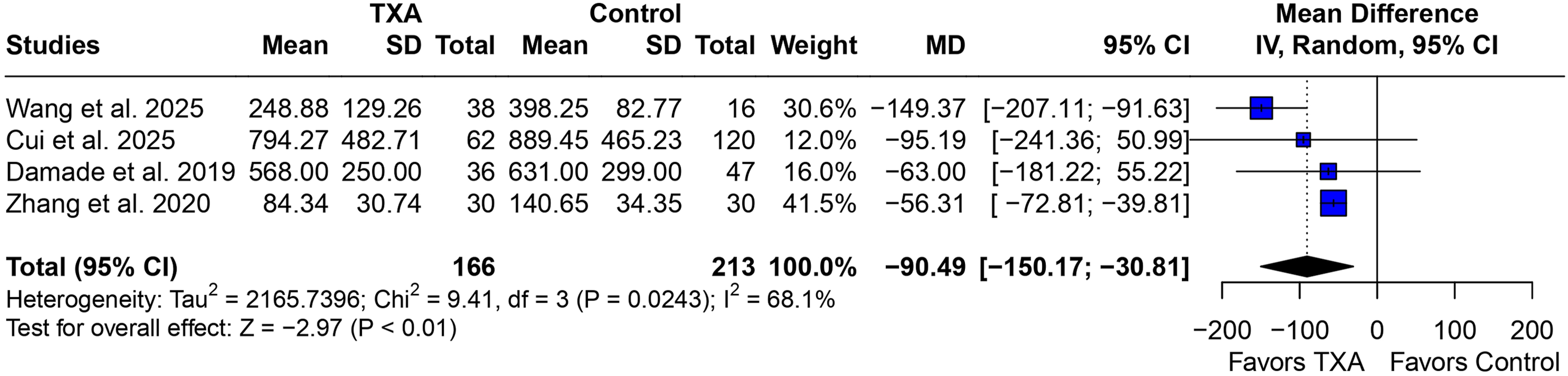
However, there was no significant difference in terms of postoperative drain duration (MD −1.61 days; 95% CI −4.27 to 1.04;
Operation Time
There was no significant difference in terms of operation time (MD −1.23 min; 95% CI −9.29 to 6.83; Forest Plot for Operation Time: There was no Significant Difference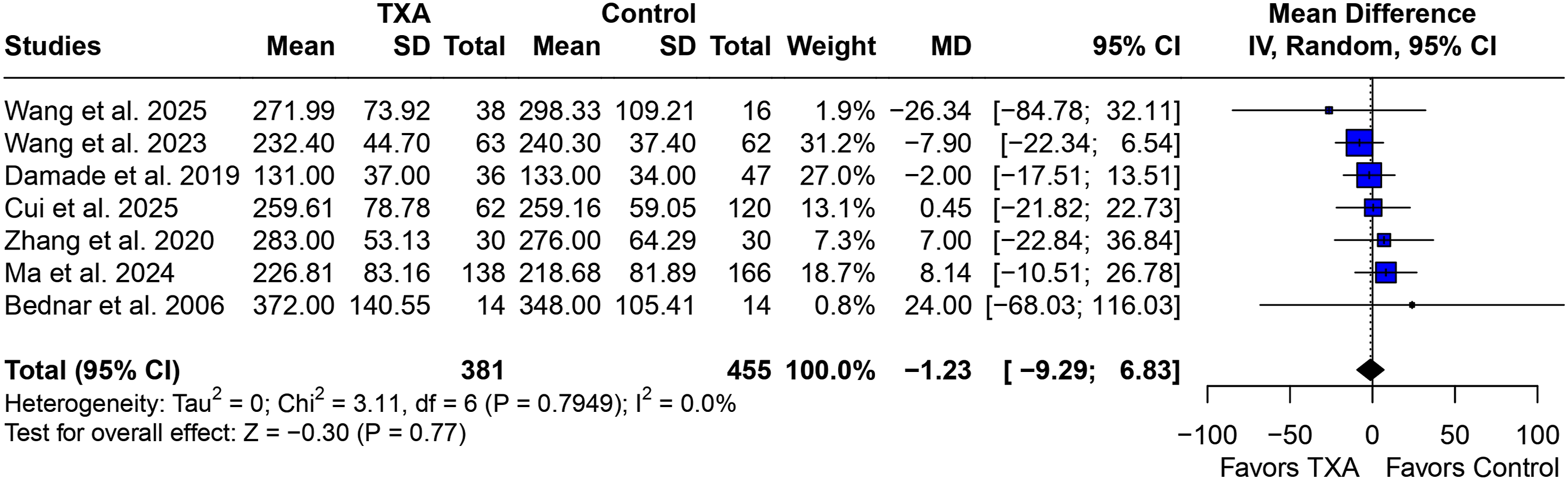
Length of Hospitalization (Days)
Length of hospital stay was significantly shorter in the TXA cohort (MD −1.88 days; 95% CI −3.33 to −0.44; Forest Plot for Length of Hospitalization ERAS Subgroup Showed Significant Difference for Length of Hospitalization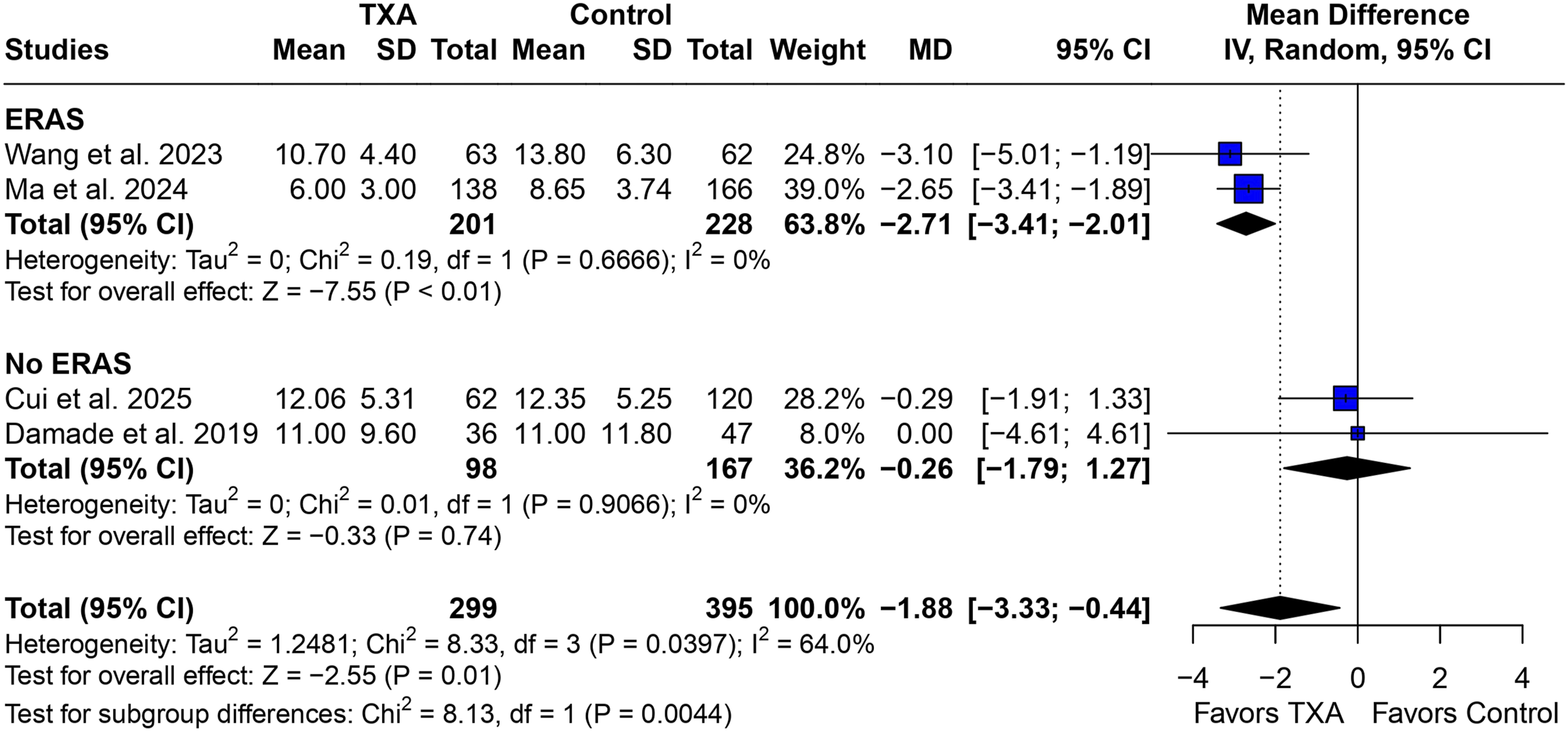
Volume of Intraoperative Transfusion
Only studies without ERAS protocol and with decompression and stabilization surgery reported volume of transfusions. There was no significant difference for volume of intraoperative transfusions (SMD −0.37; 95% CI −0.76 to 0.03;
Number of Transfusions
Only studies without ERAS protocol and with decompression and stabilization surgery reported rate of transfusions. There was no significant difference in terms of intraoperative (OR 0.34; 95% 0.08 to 1.47;
Complications
Overall complication rate was significantly reduced in the TXA group (OR 0.54; 95% CI 0.34 to 0.87; Forest Plot for Overall Complication Rate Favoring TXa Group; However, Only ERAS and Aggressive Resection Surgery Subgroups Showed a Significant Reduction of Complications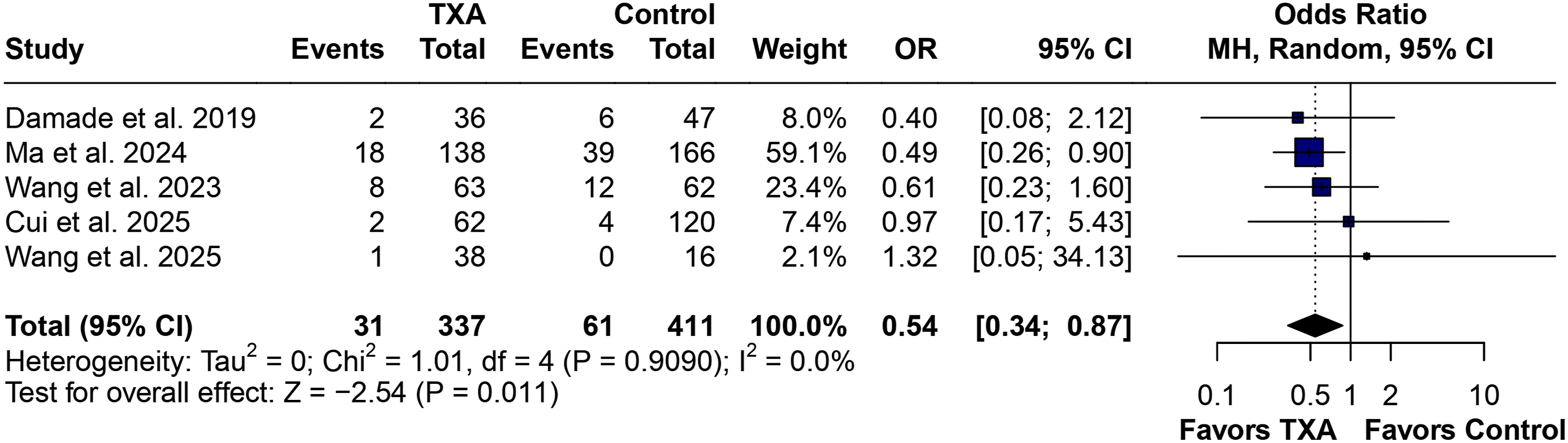
There was no significant difference in respiratory complications (OR 0.58; 95% CI 0.13 to 2.56;
Risk of Bias
Risk of bias was performed by two authors independently. The quality assessment showed moderate concerns. All studies were graded with moderate concerns, mostly due to risk of bias in confounding, measurement of outcomes, participant selection, or classification of the outcomes. Detailed results of the risk of bias are available in Figure 7. Quality Assessment Results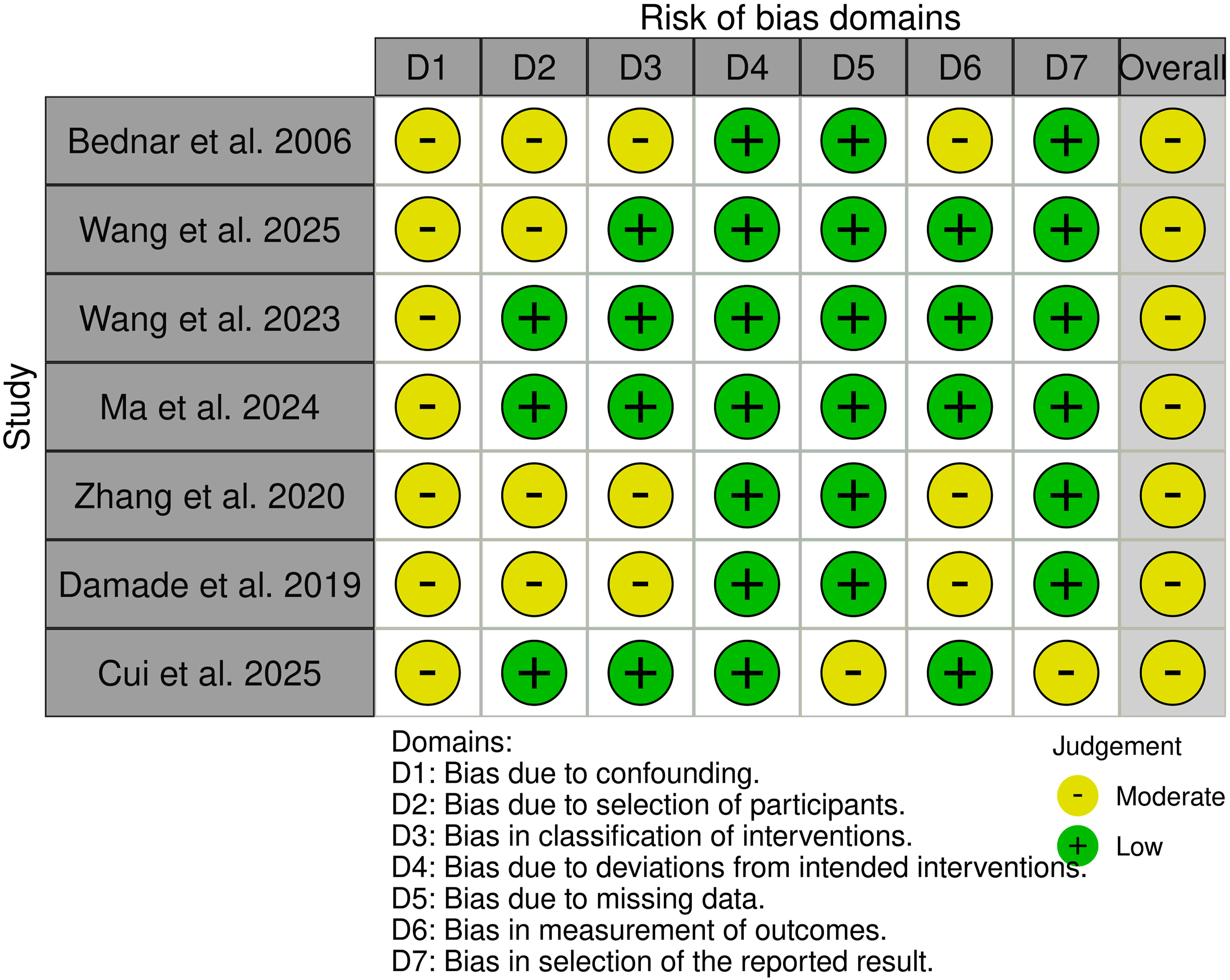
Discussion
This is the first meta-analysis that provides a comprehensive comparison of the use of TXA in spinal tumor and metastasis surgery. This synthesis focused on several crucial clinical outcomes, including blood loss, transfusion requirements, operation time, length of hospital stay, and complication rate.
Increased perioperative bleeding remains a significant challenge in spinal tumor and metastasis surgery, which might lead to high morbidity.2-4 This meta-analysis confirmed the efficacy of the TXA, and showed a significant benefit in terms of bleeding volume reduction. However, significant heterogeneity was observed in the pooled meta-analysis. This could be due to various reasons—for example, surgeons might choose TXA in more complex cases, where the risk of bleeding was higher compared to control groups.
18
Additionally, highly-vascularized metastatic lesions were not analyzed separately, as insufficient data was available in the literature.
25
This aspect was only analyzed in the study conducted by Cui et al. (2025), where authors showed that patients with hypervascular tumors had significantly higher blood loss volume compared to non-hyper vascular lesions (
Moreover, this study found no significant difference in the total transfusion volume between TXA and no TXA groups. This could be caused by high vascularity, and complexity of surgical procedures in both groups, which showed no significant impact on blood transfusion. However, the transfusion endpoints were rarely reported, and future studies should also assess the use of allogenic transfusion rates more extensively.27-29
Prolonged hospital stay is associated with increased healthcare costs and an additional risk of hospital-related infections.30-32 However, this meta-analysis showed no benefit of TXA use in terms of reduction of hospital stay - significant reduction was only achieved with ERAS protocol, which heavily confounded the conclusion. This program was applied by Wang and Ma, successfully leading to shorter hospital stays.20,21 It is important to note that the experimental group involved a significant number of factors, and the use of TXA was only one of them. Therefore, further studies should explore hospitalization more deeply, as non-ERAS studies suggest no significant benefit of TXA use for reduced hospital stay.
A significant concern about using TXA perioperatively, especially among patients with tumors or metastases, is the potential increase in risk for thromboembolic events. 33 Patients with oncological spines possess an elevated risk of thromboembolism caused by various malignancy-associated factors. In our systematic analysis, we found no statistically significant difference in terms of respiratory complications, vein thrombosis events, which suggest TXA use is safe in these patients.
While the meta-analysis indicated a statistically significant reduction in the overall complication rate in the TXA group, this result should be interpreted with caution. First, a significant reduction of complications was only observed in ERAS protocol studies, which involve multiple perioperative factors, and not only TXA use. Only a limited subset of the included studies reported complication outcomes, and the most clinically relevant adverse events—such as thromboembolic events, surgical site infections, and major perioperative complications—occurred at similar rates between groups. This raises doubts regarding the likelihood of a true reduction in complications attributable to TXA. More plausibly, the data support the conclusion that TXA use does not increase the risk of adverse events, reinforcing its safety profile. Nonetheless, the potential influence of selective reporting and short follow-up periods in several studies cannot be overlooked. Given these concerns, the most optimal approach is the balanced individualized risk assessment of given patients, considering thrombotic risk, and the use of anticoagulation protocols. Therefore, TXA should be considered as non-inferioror, rather than a definitive solution to significant reduction of complications among spinal metastatic patients.
Notably, while some trends and outcomes were observed in this meta-analysis, several crucial limitations need to be acknowledged before applying these results to clinical practice. First, we observed significant heterogeneity in various outcomes, including blood loss or transfusion volume. We performed robust sensitivity analyses, including leave-one-out, and we identified studies, which could lead to higher inconsistency of the results. Therefore, differences in study design, selection of participants, demographic data, surgical approach, and TXA regimen could significantly impact variability and inconsistency across the outcomes. Moreover, unstandardized reporting of units led to problems in pooling analysis for some outcomes, including total transfusion volume, which remains an essential outcome in such analysis. Future studies need to standardize results and avoid selective reporting, which could prevent making definitive conclusions.
Furthermore, we observed no randomized trials in the current meta-analysis. Most of the studies had a retrospective design; therefore, selection and confounding bias were inevitable. This was confirmed in the quality assessment with the ROBINS-I tool, as all studies had moderate concerns in one or more domains, and no study was given a low bias grade overall. Low quality of included studies along with aforementioned heterogeneity could have highly impacted the results of this review. Differences in baseline characteristics in non-randomized papers lead to presence of patients with more severe conditions in one of the groups, masking the true effect of TXA administration. Therefore, high-quality RCT studies are needed to overcome current methodological limitations and provide an objective, unbiased assessment of effectiveness.
The findings of this study show consistency with previous meta-analyses, which analyzed the use of TXA in spinal surgery. A meta-analysis conducted by Izima and colleagues 34 showed a significant reduction of blood loss during the surgery with the use of topical TXA. Xiao and colleagues 35 conducted a meta-analysis of RCT studies, where TXA was applied for lumbar spine surgery. Their study included 49 articles with a total of 4822 patients. Their meta-analysis also showed the reduction of blood loss and transfusion rates with the use of TXA. Moreover, significant differences were observed in drain time, hematocrit, or hemoglobin levels, which were not analyzed in the current meta-analysis due to a lack of a sufficient number of studies reporting these outcomes. Therefore, the findings of this meta-analysis, especially transfusion outcomes, might be different due to the nature of the oncological spine, where neoplastic vascularity could significantly impact the overall bleeding, and TXA could have a smaller impact on stopping it.
The findings of this meta-analysis indicate several implications for clinical spine practice. TXA remains a safe agent and could reduce intraoperative blood loss. However, while promising, we did not find any significant difference in terms of transfusion requirements. Due to observed heterogeneity, standardization and randomized trials are needed. Moreover, future studies should evaluate the most optimal dosing regimen, TXA administration timing, and perform more detailed subgroups, which could allow for more comprehensive effectiveness exploration. Future studies should aim to stratify outcomes more precisely based on tumor type, methods of spinal stabilization, extent of resection, and other relevant factors. Larger, multicenter trials are necessary to determine whether TXA significantly reduces blood loss in patients at high surgical risk. Additionally, comparing or combining TXA with other antifibrinolytic agents may offer further insights into the most effective strategies for perioperative blood management.
Conclusions
Initial reports from retrospective studies suggest reduced blood loss with TXA. However, the impact on hospitalization duration, or complication rate is unclear. Results of this study are confounded by retrospective design of the studies, risk of bias, heterogeneity, and inclusion of ERAS protocols. TXA is promising, appears safe, but high-quality RCTs are required to establish definitive efficacy and guidelines.
Supplemental Material
Supplemental Material - Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis
Supplemental Material for Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis by Anna Łajczak, Paweł Łajczak, Oguz Kagan Sahin, Ayesha Ayesha, Newton Godoy Pimenta, and Ramon Guerra Barbosa in Global Spine Journal
Supplemental Material
Supplemental Material - Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis
Supplemental Material for Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis by Anna Łajczak, Paweł Łajczak, Oguz Kagan Sahin, Ayesha Ayesha, Newton Godoy Pimenta, and Ramon Guerra Barbosa in Global Spine Journal
Supplemental Material
Supplemental Material - Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis
Supplemental Material for Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis by Anna Łajczak, Paweł Łajczak, Oguz Kagan Sahin, Ayesha Ayesha, Newton Godoy Pimenta, and Ramon Guerra Barbosa in Global Spine Journal
Supplemental Material
Supplemental Material - Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis
Supplemental Material for Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis by Anna Łajczak, Paweł Łajczak, Oguz Kagan Sahin, Ayesha Ayesha, Newton Godoy Pimenta, and Ramon Guerra Barbosa in Global Spine Journal
Supplemental Material
Supplemental Material - Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis
Supplemental Material for Tranexamic Acid Versus no Tranexamic Acid in Spinal Tumor and Metastasis Surgery—Meta-Analysis by Anna Łajczak, Paweł Łajczak, Oguz Kagan Sahin, Ayesha Ayesha, Newton Godoy Pimenta, and Ramon Guerra Barbosa in Global Spine Journal
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data available in respective references.
Use of Large Language Model
Gemini LLM (Google Inc.) was used to enhance manuscript grammar quality and transparency. LLM was not used to generate scientific content, and authors verified all the changes.
Supplemental Material
Supplemental material for this article is available online.
