Abstract
Study Design:
A prospective, randomized, double-blind, placebo-controlled study.
Objectives:
There are few studies examining the balance between preventing venous thrombus embolism (VTE) and reducing blood loss in posterior/transforaminal lumbar interbody fusion (PLIF/TLIF) surgeries. This study aimed to evaluate the efficacy and safety of the combine application of TXA and rivaroxaban in patients undergoing PLIF/TLIF and explore relevant factors related to blood loss and VTE.
Methods:
Patients in group A which was the control group received 0.9% NaCl solution intravenously. Group B was treated by an intravenous injection of 2 g tranexamic acid (TXA) and the local use of 1 g intraoperatively. Group C was treated the same as group B intraoperatively, and they received 10 mg rivaroxaban qd treatment postoperatively. Eligible patients with an Autar score ≤ 10 were randomly assigned to group A or group B. Patients with an Autar score >10 were allocated into group C.
Results:
The intraoperative blood loss and postoperative drainage were lower in groups B and C than in group A (P < .001). The blood transfusion rate in group B was lower than that in group A (P < .001), while the incidence of VTE in group C was lower (P < .001). Four factors were found to be positively correlated with obvious total blood loss (P < .05). The data showed that 5 factors were correlated with the development of a thrombus (P < .1).
Conclusions:
The combination of TXA and rivaroxaban in PLIF/TLIF patients is safe and effective in reducing D-dimer levels associated with VTE and reducing blood loss.
Introduction
Lumbar spinal disease is 1 of the most common orthopedic diseases, and PLIF/TLIF is an effective and commonly performed treatment. However, the perioperative blood loss due to PLIF/TLIF may be extensive. This may result in epidural hematoma, infections, anemia, etc. and increase the economic burden of medical care. In the field of orthopedics, the topic of blood loss control has become a widely studied topic. TXA, as an antifibrinolytic agent, has been extensively studied recently, and it has been shown to be effective for the control of bleeding in spinal surgery.1,2 However, the optimal dose and route of administration are still unclear. In addition to the postoperative downtime of the patients being longer, TXA may also increase the risk of venous thrombosis.3,4 In terms of venous thrombosis prevention, compared with traditional drugs such as low molecular weight heparin, rivaroxaban has many advantages. Rivaroxaban has been widely used recently to prevent thrombosis. However, it has been reported that patients given rivaroxaban after surgeries have higher incidences of wound bleeding and infections.5,6
Although TXA and rivaroxaban are both effective, when the 2 drugs are used in combination, the effects of 1 drug may reduce the adverse effects of the other drug. Currently, the effects and risks of the combined application of these 2 drugs in patients have not been extensively studied.
In conclusion, this study was to evaluate the efficacy and safety of the combined application of TXA and rivaroxaban in patients undergoing PLIF/TLIF.
Methods and Materials
Study Design
Patients aged over 18 years who were diagnosed as having lumbar spondylolisthesis/intervertebral disc herniation/spinal stenosis, and requiring PLIF/TLIF were enrolled in this prospective, randomized, double-blind, placebo-controlled study (level II) after signing a written informed consent form. This study was registered in the Chinese Clinical Trial Registry (ChiCTR-1900024985) and approved by the Medical Ethics Committee of Qilu Hospital of Shandong University (No.2019105). All patients signed an informed consent form prior to participation in the study.
The sample size was calculated using a formula based on the literature on the incidence of thrombosis after lumbar spine surgery, with a minimum of 63 cases per group. In order to make the results more reliable and to reduce the possible influence of statistical errors, the number of included cases was significantly increased.
Exclusion criteria: 1. patients with deep venous thrombosis; 2. patients with obvious liver disease or kidney disease (such as acute hepatitis, cirrhosis, renal failure, etc.); 3. patients with active bleeding or a high risk of bleeding; 4. pregnant or breastfeeding women; 5. patients in whom a CYP3A4 inhibitor is necessary.; 6. patients with a thrombotic tendency (such as a history of atrial fibrillation); 7. patients who have been on long-term antiplatelet drugs; 8. patients allergic to TXA or rivaroxaban; 9. patients whose religious beliefs limit blood transfusions; 10. any other situation that the researchers considered inappropriate to participate in the study.
Randomization and Concealment
Patients were first assessed for thrombotic risk factors by Autar score after admission to the hospital. The random number method was used to classify eligible patients with an Autar score ≤ 10 into group A or group B. Patients with an Autar score >10 were allocated into group C (Figure 1). The participants and researchers were blinded to the group allocation. However, at any point in the perioperative period, any doctor or nurse could intervene immediately to prevent a medical emergency.
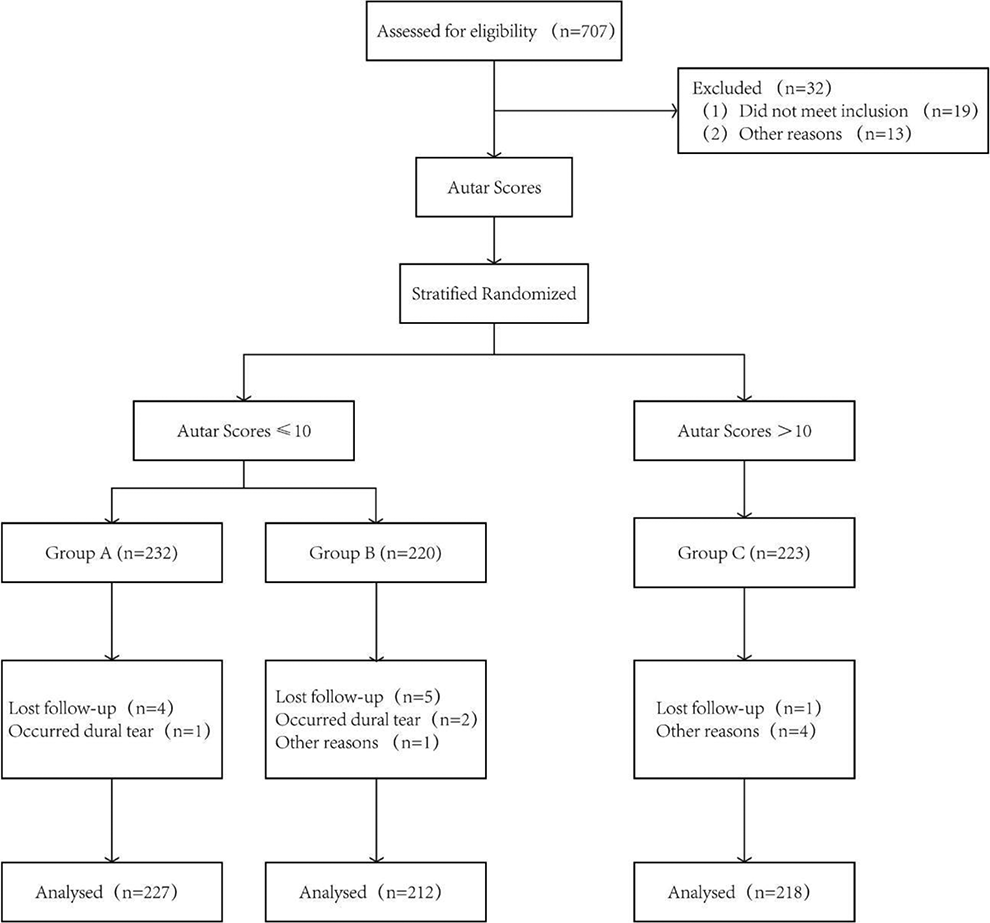
Patient flowchart.
Intervention
Group A was the control group. In this group, patients were treated with 0.9% NaCl solution intravenously 15 minutes before the skin incision. Group B was treated by intravenous injection of 2 g TXA. Before suturing the incision, TXA solution was used to soak the wound for 5 minutes, then aspirated it out, and the gelatine sponge that was pre-soaked in TXA (1 g TXA dissolved in 10 ml 0.9% NaCl solution) was placed on the surface of the spinal dura mater. Group C was treated the same as group B intraoperatively, and then, they received 10 mg rivaroxaban qd treatment for 35 days postoperatively.
During the operation, the patient lay prone with an exposed abdomen. The surgical area was exposed routinely. After evaluating the position of the screw by an X-ray, the pedicle screw was placed. Then, we removed the intervertebral disc and put a PEEK cage into the intervertebral space after routine decompression. During the operation, the dura mater and nerve roots were protected carefully. After complete hemostasis, drainage tubes were placed routinely before closing the incision.
Autologous transfusion was performed when we found that the patient was bleeding more intraoperatively and when the blood count showed that the patient had an Hb of <100g/L or an HCT of <35%. The blood used for autologous transfusion was obtained from intraoperative bleeding recovered through a strict aseptic process.
In addition, the cost of tranexamic acid was about ¥336, while rivaroxaban was ¥965.58. The cost of venous ultrasound of both lower extremities was about ¥260 per visit. Each of the above costs was an initial cost. After reimbursement, most patients could reduce their costs by a certain percentage due to differences in health insurance policies from region to region. Rivaroxaban, for example, was reimbursed at a rate of about 80-90% in some regions. But if a patient was completely at his own expense, he would have to pay a total of ¥1561.58.
All patients received an identical postoperative treatment regimen, including physical prevention of VTE. According to the guidelines by the National Ministry of Health, when the postoperative Hb level was <70 g/L or 70-100 g/L with symptomatic anemia, the patients would receive packed red blood cells.
Outcome Measurements
Primary outcomes included: intraoperative blood loss, postoperative drainage, obvious total blood loss, the ultrasound results of the veins of both lower limbs postoperatively and number of patients receiving blood transfusions.
Demographics, medical history, blood lipid levels and bone density were collected before the surgery. Hemoglobin levels, hematocrit levels, activated partial thromboplastin time (APTT), prothrombin time (PT), liver and kidney function, C-reactive protein (CRP) and D-dimer were tested preoperatively and on postoperative day 1 (POD1), POD3 and POD5.
The amount of blood in the suction bag minus the amount of irrigation fluid and the dry weight of the gauze were used to estimate the amount of intraoperative blood loss. The daily drainage volume after the operation was recorded. The obvious total blood loss = intraoperative blood loss + postoperative drainage. When the drainage volume was less than 100 ml/24 h, the drainage tubes were removed.
Statistical Analysis
The mean and standard deviation are used to express quantitative data, while qualitative data is expressed as numerical values. The difference between the 2 sets of quantitative data was determined via the Welch two-sample t-test. Pearson’s chi-square test or Fisher’s exact test were used to analyze the qualitative data. Correlation analysis was used to analyze relevant factors affecting the obvious total blood loss. Logistic regression was used to analyze the risk factors of VTE. All statistical analyzes were performed using SPSS version 26 (SPSS, Chicago, IL), and the significance level was P < .05 or .1.
Results
Patient Demographics
A total of 657 eligible patients participated in this study. After randomization, there were 227 patients in group A, 212 patients in group B and 218 patients in group C. There were no significant differences in sex, age, BMI or MAP among the 3 groups. No significant difference was detected in hematological parameters among the groups (Table 1).
Characteristics of Patients.
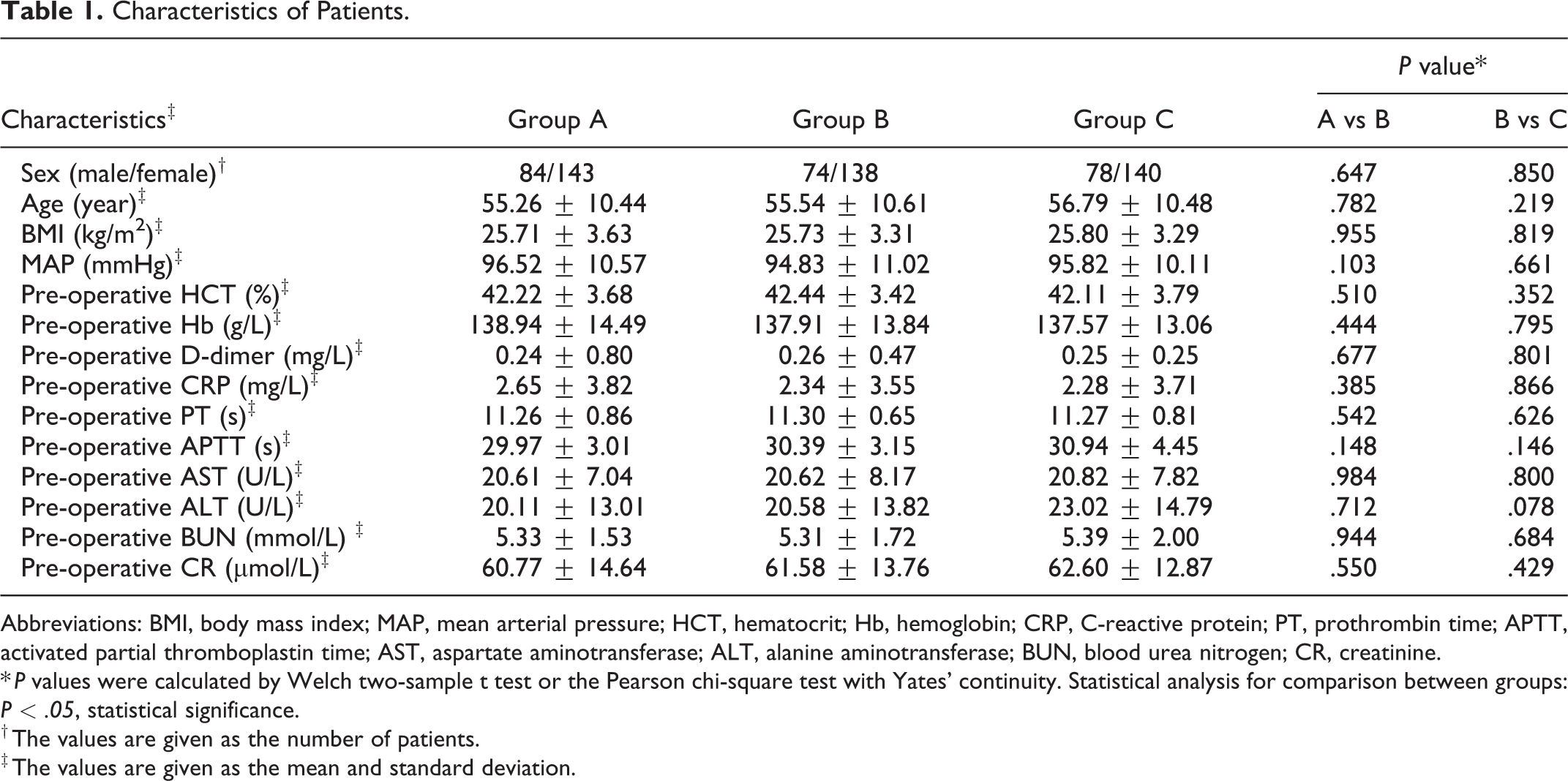
Abbreviations: BMI, body mass index; MAP, mean arterial pressure; HCT, hematocrit; Hb, hemoglobin; CRP, C-reactive protein; PT, prothrombin time; APTT, activated partial thromboplastin time; AST, aspartate aminotransferase; ALT, alanine aminotransferase; BUN, blood urea nitrogen; CR, creatinine.
* P values were calculated by Welch two-sample t test or the Pearson chi-square test with Yates’ continuity. Statistical analysis for comparison between groups: P < .05, statistical significance.
† The values are given as the number of patients.
‡ The values are given as the mean and standard deviation.
Blood Loss and Postoperative Hematological Parameters
The primary outcomes are summarized in Table 2. The amount of intraoperative blood loss and postoperative drainage in group A was significantly greater than that in group B (P < .05). However, no such difference was observed between group B and group C. The levels of HCT and HB in group B were significantly higher than those in group A (P < .05). No significant difference was detected in postoperative hematological parameters between group B and group C. No significant differences in the CRP or D-dimer levels were observed preoperatively among all of the groups. After surgery, the CRP and D-dimer increased in all groups. On POD1, POD3 and POD5, group C (mean and SD 0.81 ± 0.50 mg/L, 1.01 ± 0.27 mg/L, and 1.59 ± 0.30 mg/L) had lower D-dimer levels than group B (mean and SD 1.21 ± 0.16 mg/L, 2.22 ± 0.27 mg/L, 2.08 ± 0.18 mg/L). There was no statistical difference between group A (mean and SD 1.15 ± 2.20 mg/L, 2.17 ± 0.30 mg/L, and 2.04 ± 0.5 mg/L) and group B. At every time point after the surgery, compared with group A (mean and SD 30.17 ± 24.31 mg/L, 35.51 ± 21.28 mg/L, and 36.26 ± 17.12 mg/L), the CRP level was significantly lower in group B (mean and SD 25.42 ± 23.23 mg/L, 23.07 ± 20.31 mg/L, and 29.62 ± 24.45 mg/L) and group C (mean and SD 24.10 ± 15.60 mg/L, 25.80 ± 14.66 mg/L, and 29.46 ± 14.34 mg/L). However, no statistical difference was detected between group B and group C. The data is described in detail in Table 2 and Figure 2.
Primary Outcomes (I).*
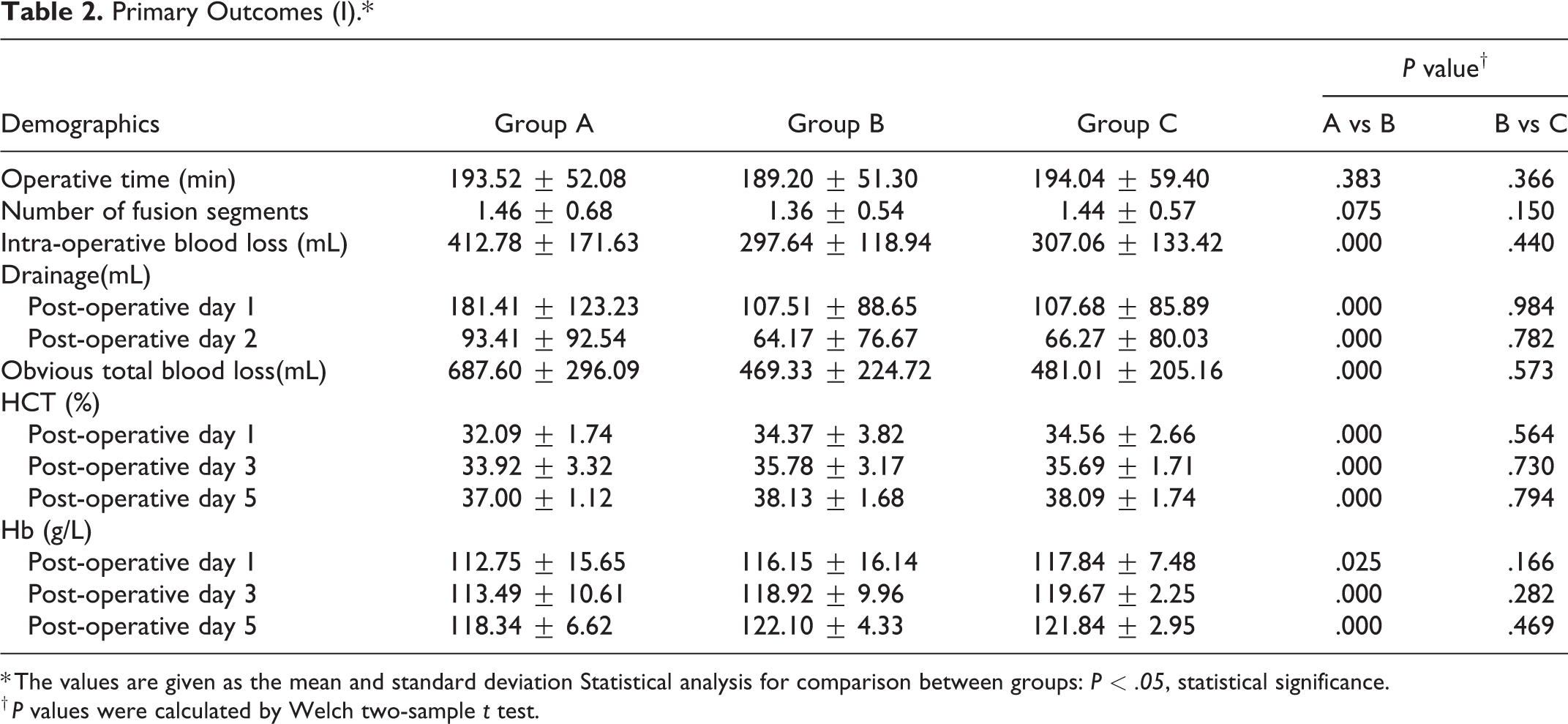
* The values are given as the mean and standard deviation Statistical analysis for comparison between groups: P < .05, statistical significance.
† P values were calculated by Welch two-sample t test.
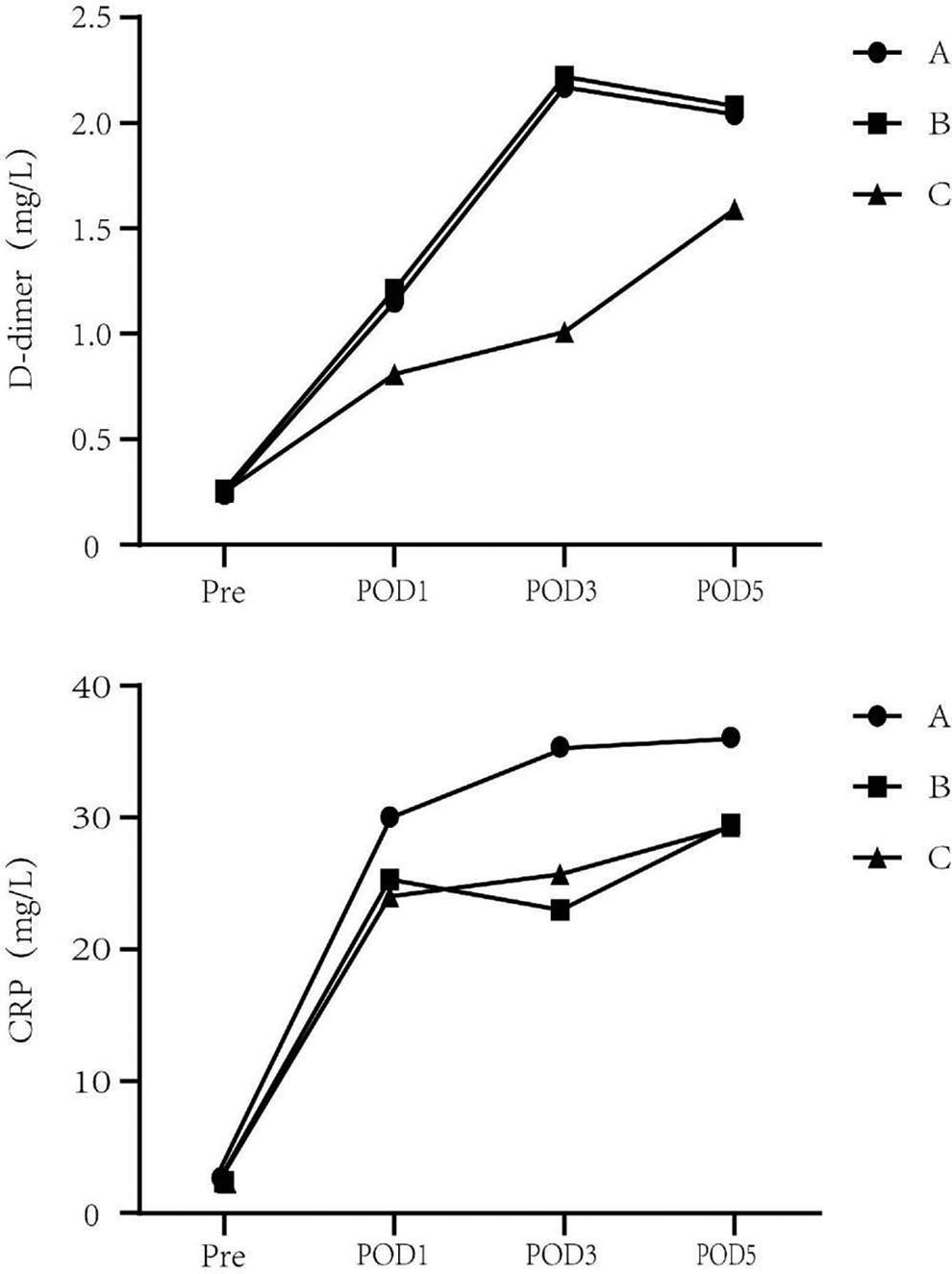
The perioperative serum levels of D-dimer and CRP. Before surgery, no statistical difference in CRP or D-dimer levels was observed between the 3 groups (P < .05). CRP, C-reactive protein; Pre, preoperative; POD1, post-operative days 1; POD3, post-operative days 3; POD5, post-operative days 5.
Blood Transfusion and VTE Rates
The autologous blood transfusion rates of group B and C were significantly lower than that of group A (P < .05), while no difference was observed in the rate of allogeneic blood transfusion among all groups. Through postoperative ultrasound examination, 36 patients in group A and 34 in group B were diagnosed with intermuscular venous thrombosis. Compared with group C, there was a higher VTE rate in group B (P < .05). However, there was no significant difference between group A and group B (Table 3).
Primary Outcomes (II).*
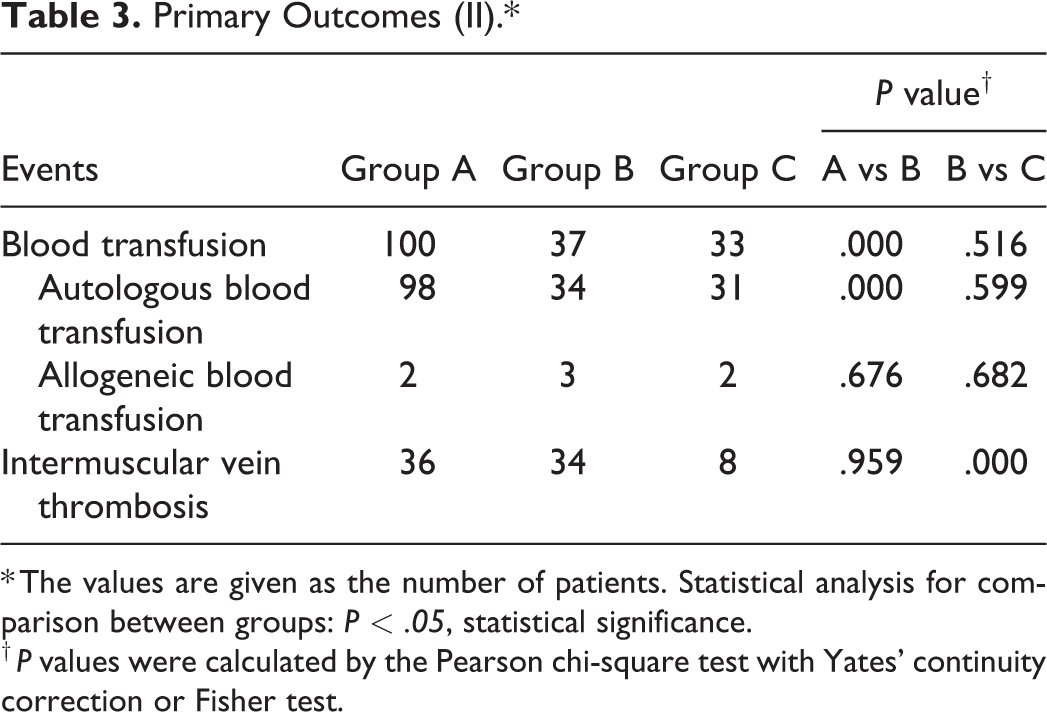
* The values are given as the number of patients. Statistical analysis for comparison between groups: P < .05, statistical significance.
† P values were calculated by the Pearson chi-square test with Yates’ continuity correction or Fisher test.
Safety Events
All patients underwent X-ray and CT examinations after the surgery to determine the position of the endophytes and to check for an epidural hematoma. No fatal complications such as a pulmonary embolism or an epidural hematoma occurred in any of the patients (Table 4). Similarly, there were no clinical symptoms of TXA toxicity, such as epilepsy. Moreover, no significant differences or significant abnormalities were detected in the comparisons of the 3 groups in terms of liver function, kidney function and blood coagulation function. Details of the postoperative demographics are shown in Table 5.
Safety Outcomes (I).*
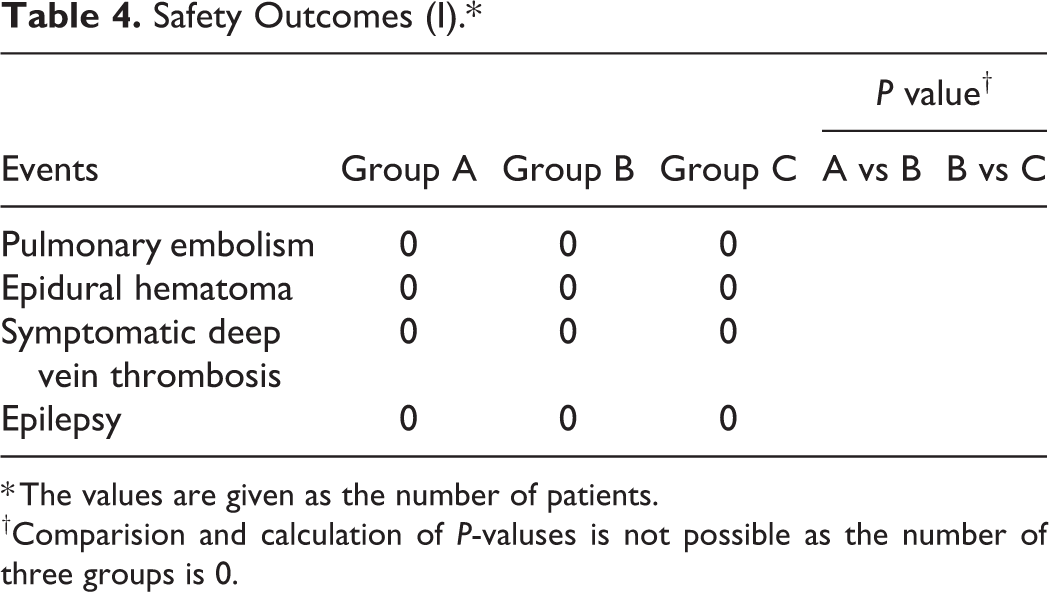
* The values are given as the number of patients.
†Comparision and calculation of P-valuses is not possible as the number of three groups is 0.
Safety Outcomes (II).*
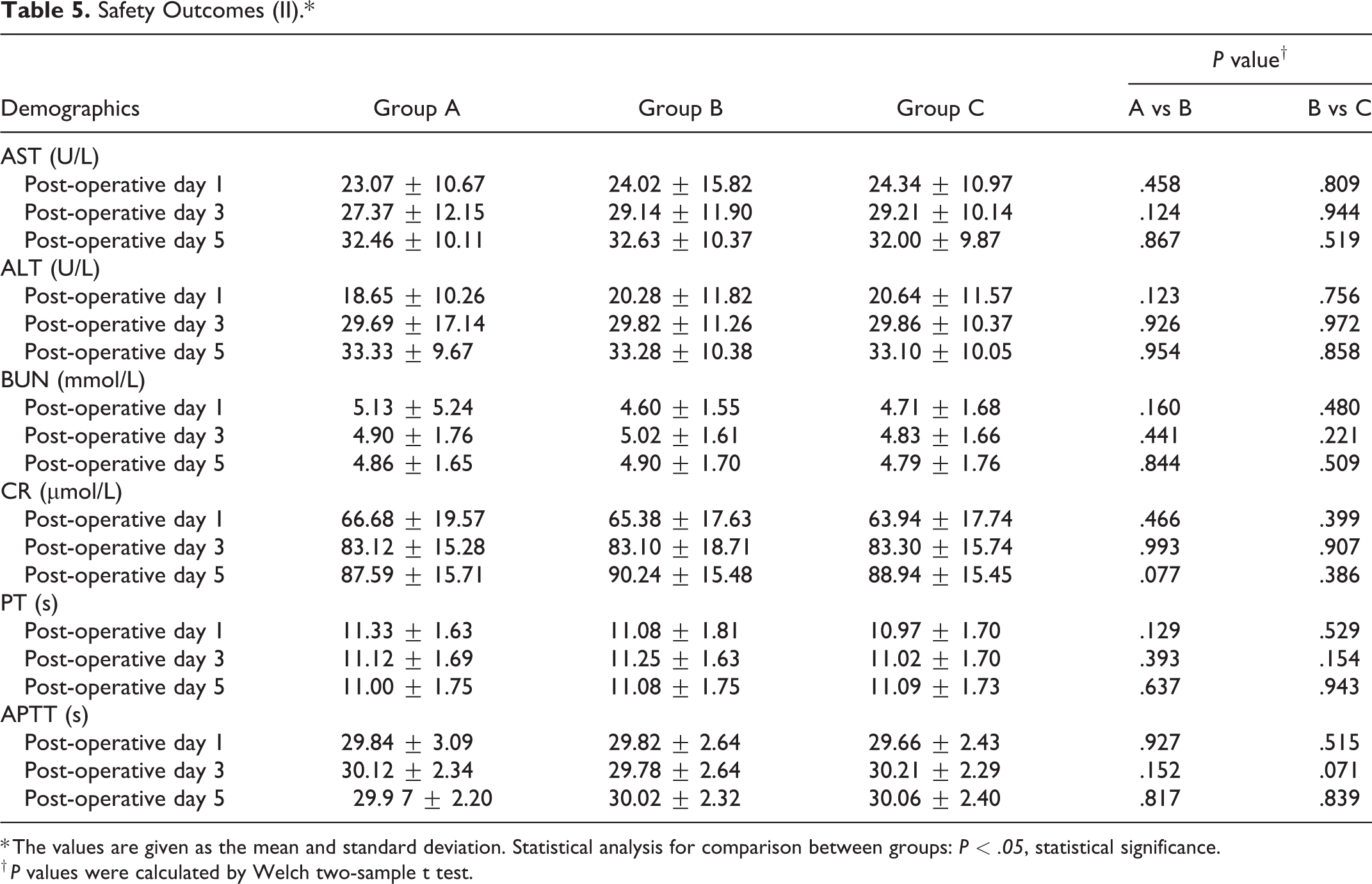
* The values are given as the mean and standard deviation. Statistical analysis for comparison between groups: P < .05, statistical significance.
† P values were calculated by Welch two-sample t test.
Relevant Factors Analysis
Four factors were identified that were positively correlated with the obvious total blood loss via Kendall’s tau-b correlation test (P < .05). Among these 4 factors, the number of fixation segments had the strongest correlation, followed by operative time, and BMI had the weakest correlation. The data obtained by logistic regression showed that BMI, HDL, number of fixation segments, bone density and T-scores of the lumber spine were the risk factors for developing a thrombus (P < .1). Among them, HDL was a negatively correlated factor, and the other factors were positively correlated. Detailed data are located in Tables 6 and 7.
Relevant Factors to Obvious Total Blood Loss.*
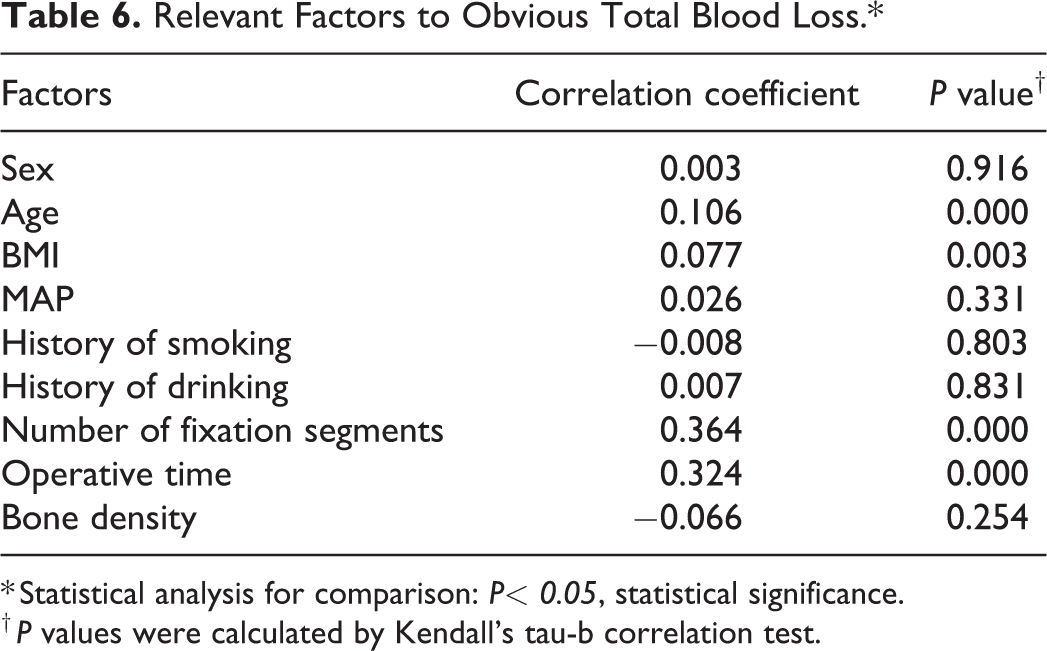
* Statistical analysis for comparison: P< 0.05, statistical significance.
† P values were calculated by Kendall’s tau-b correlation test.
Relevant Factors to VTE.*
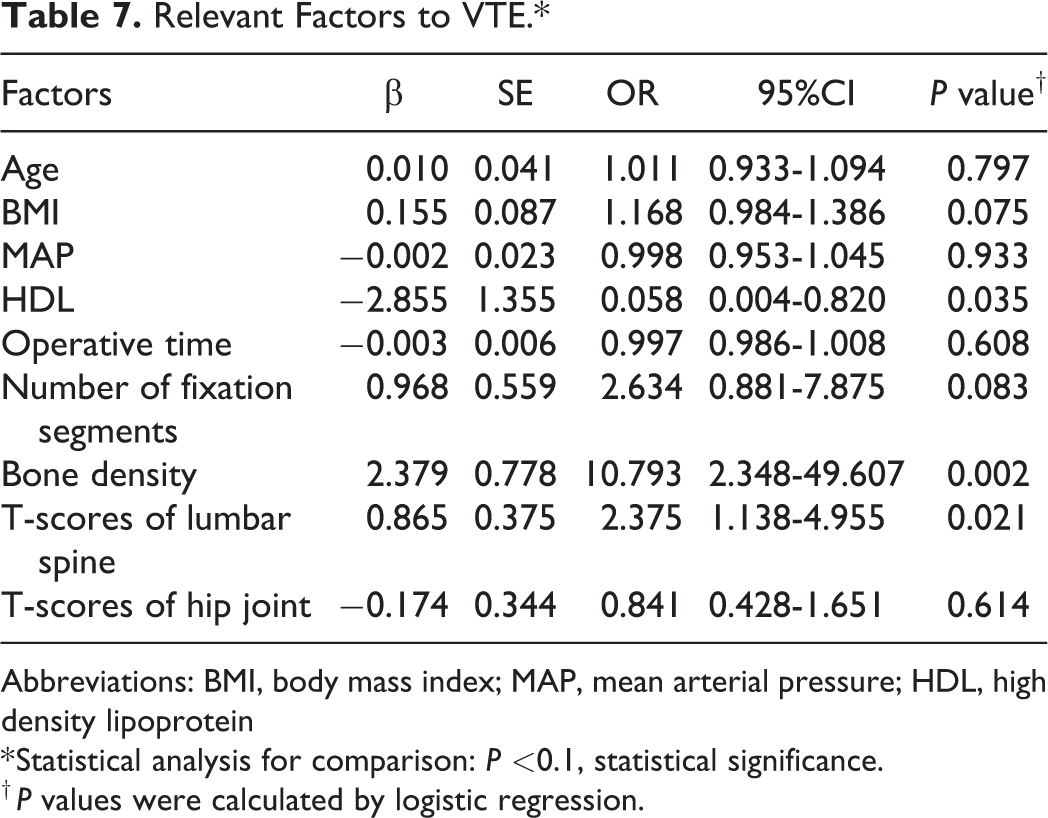
Abbreviations: BMI, body mass index; MAP, mean arterial pressure; HDL, high density lipoprotein
* Statistical analysis for comparison: P <0.1, statistical significance.
† P values were calculated by logistic regression.
Discussion
Blood loss during spinal surgery is still 1 of the most important problems faced by doctors. Allogeneic blood transfusion is the main method for the treatment of postoperative acute anemia, but it may cause complications such as allergic reactions, acute hemolytic reactions and viral infections. Moreover, the shortage of blood resources is also a practical problem.
Recently, studies have proven the hemostatic effect of TXA. The meta-analysis conducted by Thomas Cheriyan et al found that TXA could significantly reduce blood loss during spinal surgery. 7 The study by Hariharan et al also confirmed this. 1 Sun et al believed that intravenous infusion of 15 mg/kg of TXA before making the skin incision could reduce blood loss intraoperatively. 8 Ker et al found that the blood loss was significantly reduced after local application of TXA. 9 Due to the different approaches of applying TXA, its hemostatic effect varies greatly across these studies. We chose an intravenous infusion of 2 g TXA + local application of 1 g TXA, and the results of the study proved that this program was effective. However, we excluded certain types of patients, so our conclusions cannot be applied to these patients.
Because of the high dose of TXA applied in this study, it is necessary to pay close attention to the safety of the medication. Moreover, some studies have found that large doses of TXA may cause nervous system toxicity or tissue toxicity. Lecker et al believed that TXA may cause seizures, especially in patients who had taken high doses (80-100 mg/kg) or had undergone heart-related surgery. 10 A study by Mclean et al found that TXA at a concentration of 100 mg/ml was significantly toxic to tissues around the joints. 11 Jacob et al found that TXA was toxic to human cartilage tissue in vitro. 12 In the course of this study, no evidence of TXA toxicity such as epilepsy was found in the patients during the perioperative and follow-up period, and no obvious abnormalities of liver and kidney function or coagulation function were found in any of the groups. Therefore, we believe that the dose of TXA applied in this study is safe enough for patients.
Studies have found that postoperative blood loss is related to inflammatory response indicators (IL-6 and IL-1β), but the correlation was not significant. 13 Our data showed that the postoperative CRP increase in group B was significantly lower than that in group A. Fuxing Pei et al found that repeated use of TXA could inhibit the increase in IL-6 and CRP after surgery.14-16 These results are consistent with our findings. Blood loss can be seen as a sign of tissue damage that may have a positive effect on inflammatory markers, so it can be inferred that high doses of TXA have an inhibitory effect on postoperative CRP elevation.
TXA strongly inhibits fibrinolysis caused by fibrinolytic enzymes. Therefore, theoretically, TXA should not increase the risk of deep vein thrombosis. TXA has been shown to reduce bleeding in surgery without increasing the risk of complications. 3 Nishihara conducted a study using only mechanical thromboprophylaxis measures and showed a significantly higher incidence of distal DVT in the static TXA group than in the control group. 17 However, in our study, there was no statistically significant difference in the incidence of thrombosis between group A and group B, whereas significant differences were observed between group B and group C. We applied the combined regimen of TXA and rivaroxaban to mitigate both the effect of rivaroxaban on the surgical area and the possible systemic effect of TXA in increasing the risk of DVT.
The drugs traditionally applied to prevent thrombosis have limitations, and in recent years, new drugs have been developed, including Xa factor inhibitors and thrombin inhibitors. Gomez-Outes et al compared these drugs with enoxaparin and found that rivaroxaban was the most effective drug. 18 Although rivaroxaban is a powerful anticoagulant, it may induce complications related to bleeding. The effectiveness and safety of rivaroxaban in patients undergoing spinal surgery has not been extensively studied.
An epidural hematoma and its neurological sequelae are among the most serious complications associated with anticoagulants in the perioperative period of spine surgery. A study by Kirchhof et al found that 20 mg RXB qd was effective in reducing baseline levels of D-dimer and CRP. 19 Our data showed no differences in infection or bleeding rates between group B and group C. Furthermore, there was no difference in CRP levels between group B and group C. This may be because we only collected data from patients in the short-term postoperative period and that a lower dose of rivaroxaban was administered.
The analysis of the factors correlated with an obvious total blood loss showed a positive correlation between age, BMI, time of surgery and the number of fixation segments. Among these, the correlations for the operation time and the number of segments is very strong. It is not difficult to understand that a greater extent and duration of a surgical procedure causes more bleeding. The correlation between age and obvious total blood loss suggests to us that older patients have worse coagulation, are prone to early hemodynamic instability and are less tolerant of blood loss than younger patients. Therefore, adequate evaluation and preparation should be carried out before surgery to prevent complications caused by excessive blood loss from surgery.
In addition, the association between BMI and the obvious total blood loss is also noteworthy. Patients with a high BMI generally have a higher body fat percentage, which is detrimental to revealing the surgical area during surgery (more damaged adipose tissue and more bleeding will occur). However, there seems to be some contradictions in the findings that adipose tissue contributes to the formation of blood hypercoagulability through multiple pathways.20,21 Our data showed that patients with a higher BMI were more likely to develop VTE. Ma et al found that in patients with gynecologic malignancies, BMI >26 kg/m2 was a risk factor for VTE. 22 However, the BMI threshold that increases the risk of VTE cannot be determined at this time.
We also noted a strong correlation between bone density and VTE, with patients with osteoporosis being 10.793 times more likely to develop VTE (P < .05). Osteoporosis indicates to some extent a lack of activity by the patient, and a lack of activity may lead to slow venous blood flow and increase the likelihood of VTE.
However, some limitations are inevitable in our study. The vast majority of the patients we included had a low number of fixation segments, and insufficient data was collected for patients with more fixation segments, so our conclusions do not apply to patients with multiple segmental surgeries (the number of fixation segments >2). Moreover, given the low incidence of DVT and PE23-26 and the fact that the number of cases included in this study was not sufficiently large, the strength of the data demonstrating the prevention of VTE in this study was not robust. Due to the limited number of researchers involved and the high number of patients enrolled, only a short follow-up was feasible for us. The safety of long-term follow-up medication needs to be further studied.
Conclusion
In patients undergoing posterior/transforaminal lumbar interbody fusion surgery (PLIF/TLIF), the combination of TXA and rivaroxaban in PLIF/TLIF patients is safe and effective in reducing D-dimer levels associated with VTE and reducing blood loss. Our study provides evidence for this conclusion.
Supplemental Material
Supplemental Material, sj-jpg-1-gsj-10.1177_21925682211024556 - Combined Use of Tranexamic Acid and Rivaroxaban in Posterior/Transforaminal Lumbar Interbody Fusion Surgeries Safely Reduces Blood Loss and Incidence of Thrombosis: Evidence From a Prospective, Randomized, Double-Blind, Placebo-Controlled Study
Supplemental Material, sj-jpg-1-gsj-10.1177_21925682211024556 for Combined Use of Tranexamic Acid and Rivaroxaban in Posterior/Transforaminal Lumbar Interbody Fusion Surgeries Safely Reduces Blood Loss and Incidence of Thrombosis: Evidence From a Prospective, Randomized, Double-Blind, Placebo-Controlled Study by Xiang Li, Guangjun Jiao, Jingyi Li, Weibin Ji, Zhiwei Hao, Fangli Gong and Yunzhen Chen in Global Spine Journal
Footnotes
Authors’ Note
Xiang Li and Guangjun Jiao contributed equally as first authors.
Acknowledgments
No funds were received in support of this work. No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
