Abstract
Study Design
Retrospective Cohort
Objective
This study utilized a large national database to compare two-year revision rates, in addition to complications and costs, of hybrid surgery (HS) compared to two-level anterior cervical discectomy and fusion (ACDF).
Methods
This study used the PearlDiver Mariner dataset selecting for patients aged 18 and older who had at least 90-day active longitudinal follow-up who underwent two-level ACDF or two-level Hybrid surgery (single level ACDF and single level CDA). Patients with prior spinal trauma, infection, cancer, or posterior fusion were excluded. Primary outcomes measures were 90-day major and minor medical complications, ED visits, readmissions, as well as two-year revisions. Patients were also assessed for postoperative dysphagia, incidental durotomy, vascular injury, 90-day surgical site, and implant complications. Additionally, hospitalization and postoperative costs were evaluated.
Results
There were 4570 two-level ACDF surgeries and 888 hybrid surgeries. After matching the cohorts, no statistical differences in demographics were found. There were no differences in reoperation rates at all measured time points nor 2-year complications. HS had a lower incidence of major (1.6% vs 3.1%,
Conclusion
Hybrid surgery is a safe and effective surgical treatment for cervical disease in appropriately selected patients.
Introduction
Cervical degenerative disc disease (DDD) is a common source of neck pain afflicting those over 60.1,2 Degenerative disc disease can cause compression of the spinal cord, leading to progressive neurologic symptoms, thus decreasing an individual’s quality of life.2,3 With more than 138,000 cervical spine surgeries performed annually, 4 anterior cervical discectomy and fusion (ACDF) is one of the most commonly utilized procedures in the surgical management of cervical DDD. However, adjacent segment disease (ASD) following ACDF remains a concern due to altered biomechanics of the cervical spine.5-8 Furthermore, ACDF for multilevel DDD results in increased cervical stiffness and adjacent segment motion compared to single level fusions and thus the risk of progressive ASD. 9 Cervical disc arthroplasty (CDA) has become a widely accepted motion-sparing alternative to the standard ACDF in appropriately selected patients by preventing excessive load on adjacent discs.10-16
Cervical disc arthroplasty has some advantages over ACDF, yet it is contraindicated in patients with poor bone quality, segment instability, disc height less than 3 mm, and severe spondylosis. 17 Multiple pathological cervical segments can have unique characteristics, necessitating different procedures for subsequent levels. 18 Hybrid surgery (HS) was developed to combine the stability of ACDF and the motion preservation of CDA.19,20 Previous literature has shown that HS leads to improved natural biomechanics, and recovery of range of motion.21,22 In addition, patient reported outcomes have shown that HS leads to decreased pain following surgery compared to two-level cervical fusion. 21
Given the novelty of this procedure, there is limited literature regarding postoperative outcomes following HS
Methods
Data Source
A retrospective database review was performed using the commercially available PearlDiver (PearlDiver Inc., Colorado Springs, Colorado, USA; www.pearldiverinc.com) patient records; this study used the Mariner dataset containing records of over 121 million patients from 2010 to 2020 searchable by International Classification of Diseases, Ninth Revision (ICD-9), International Classification of Diseases, Tenth Revision (ICD-10), and Current Procedural Terminology (CPT) codes. Considering the data is de-identified and Health Insurance Portability and Accountability Act compliant, this study was exempt from institutional review board approval.
Study Population
Inclusion and exclusion CPT, ICD-9, and ICD-10 codes are summarized in (Appendix Table A1). The study population included all patients aged 18 years of age or older who underwent two-level ACDF or two-level Hybrid surgery (single level ACDF and single level CDA) with at least 90-day active longitudinal follow-up. Patients with a history of spinal trauma, infection, malignancy, or posterior fusion were excluded from the population. Basic demographic information and comorbidities of each cohort were queried and used to match the two cohorts including age, gender, obesity, tobacco use, diabetes (DM), chronic pulmonary disease (COPD), hyperlipidemia (HLD), hypertension (HTN), peripheral vascular disease (PVD), chronic kidney disease (CKD), coronary artery disease (CAD), and congestive heart failure (CHF).
Outcomes
The primary study outcome was rate of reoperation at various time points up to 2 years post procedure. Secondary study outcomes included 90-day major and minor medical complications, emergency department (ED) visits, and readmissions. 90-day major medical complications included pulmonary embolism (PE), pneumonia (PNA), myocardial infarction (MI), cerebrovascular accident (CVA), sepsis. Minor 90-day complications encompassed deep vein thrombosis (DVT), acute kidney injury (AKI), urinary tract infection (UTI), transfusion, and wound complications. Patients were also queried for 90-day surgical complications which included surgical site infection, dysphagia, incidental durotomy, vascular injury, and implant complications. Finally, cost of hospitalization, and 90-day postoperative cost were assessed using reported insurance reimbursement.
Statistical Analysis
Pearson chi-squared test was used to assess differences in demographics and preexisting comorbidities. Multivariable logistic regression was used to determine the odds ratio (OR) and corresponding 95% confidence interval (95% CI) of HS vs ACDF on the postoperative outcomes after adjusting for demographic factors and pertinent comorbidities. A subset of patients with at least 2 years of active longitudinal follow-up were used to assess outcomes at 2-years following index surgery. R software embedded within the PearlDiver database (R Foundation for Statistical Computing, Vienna, Austria) was used for all statistical analysis. Statistical significance was set at
Results
ACDF and Hybrid
Patient demographics and Comorbidities
Hybrid surgery vs Two-Level ACDF Patient Demographics.
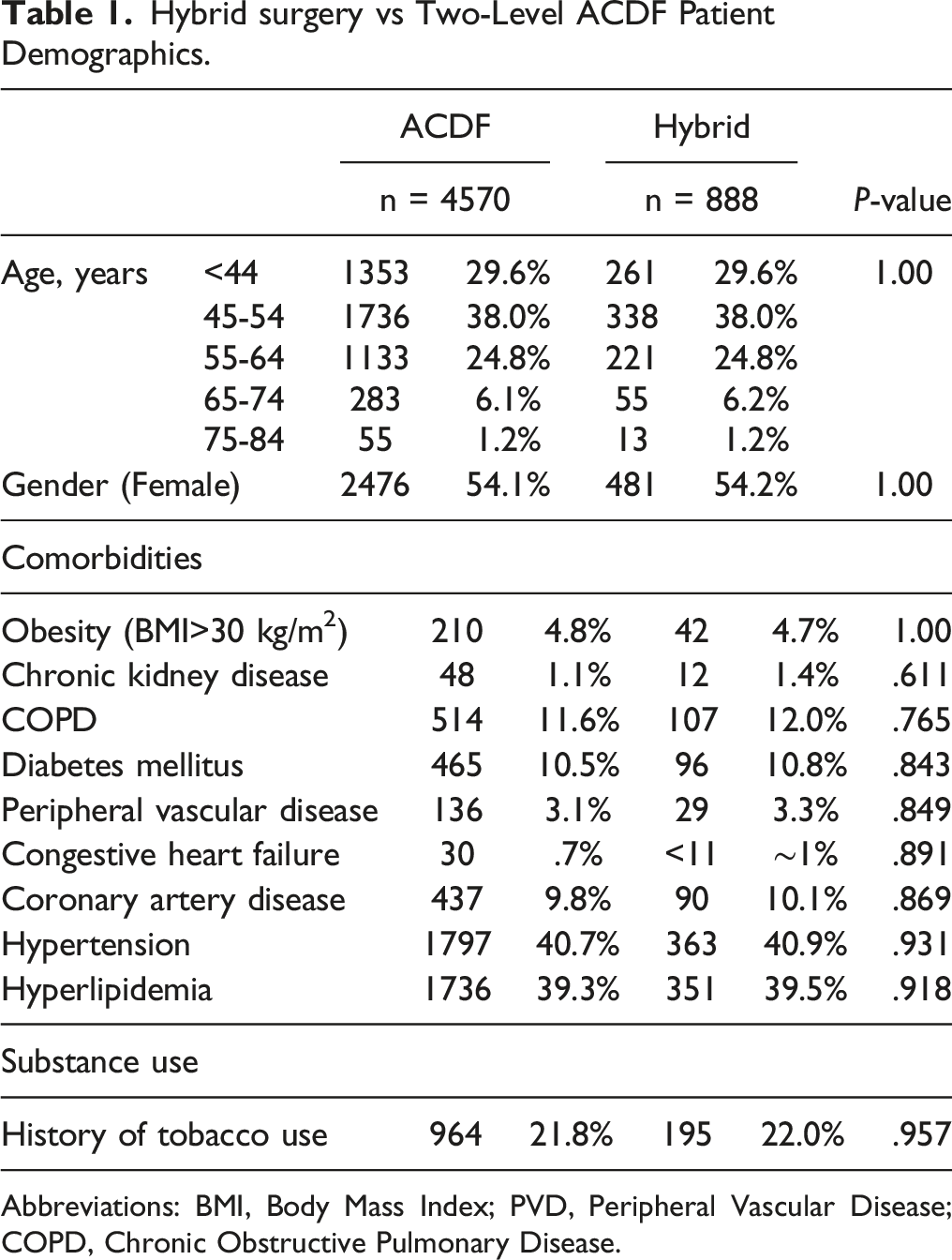
Abbreviations: BMI, Body Mass Index; PVD, Peripheral Vascular Disease; COPD, Chronic Obstructive Pulmonary Disease.
Reoperation and Long-term Outcomes
ACDF vs Hybrid Long-term Outcomes.

90-day Postoperative Complications
Surgical and Postoperative 90-day Outcomes.
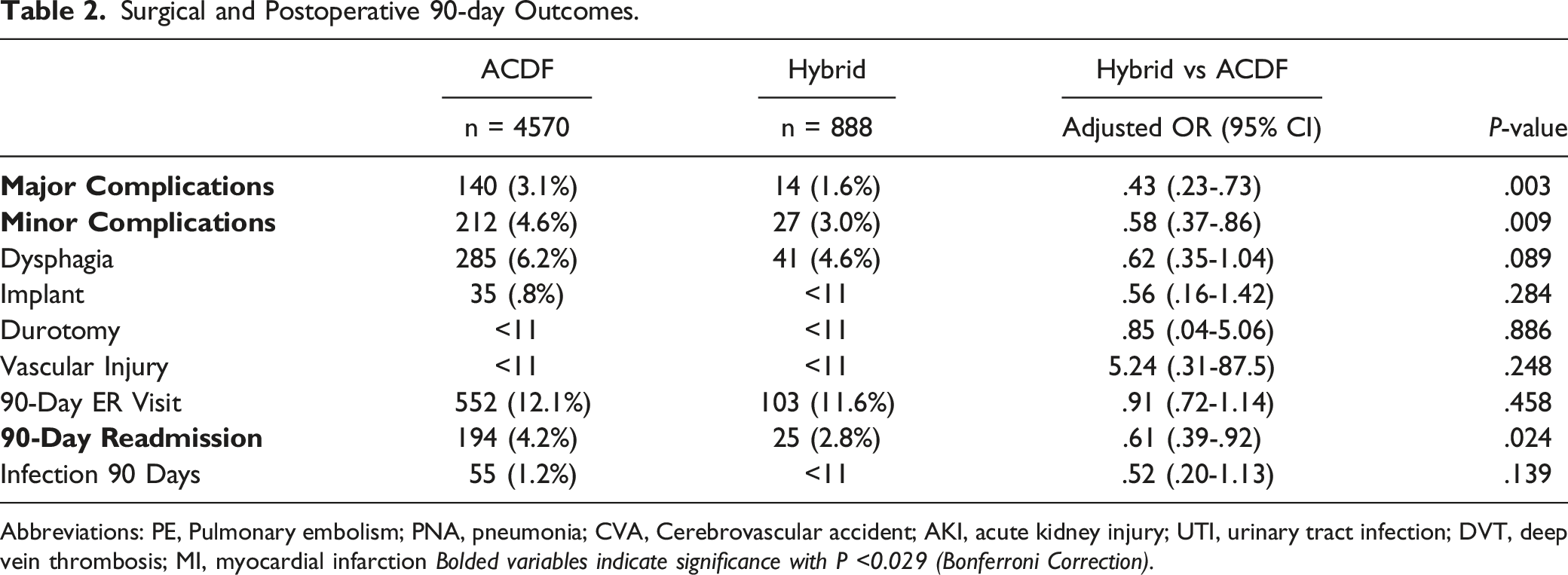
Abbreviations: PE, Pulmonary embolism; PNA, pneumonia; CVA, Cerebrovascular accident; AKI, acute kidney injury; UTI, urinary tract infection; DVT, deep vein thrombosis; MI, myocardial infarction
Surgical and 90-Day Postoperative Costs
Total Hospital and 90-Day Postoperative Cost.

Discussion
HS has gained traction in recent years as an effective alternative to ACDF; however existing literature on postoperative outcomes is limited. The present study found overall lower rates of medical complications, as well as readmission in the HS cohort within 90 days postoperatively compared to standard ACDF. In addition, HS had a lower hospitalization cost compared to those undergoing two-level ACDF, but 90-day postoperative cost was similar for both cohorts. Similarly, there were no differences in long-term complications following surgery.
Hybrid surgery was developed to combine the advantages of both ACDF and CDA in the treatment of multi-level cervical DDD. Historically, ACDF has been shown to be a safe, efficacious treatment for cervical DDD; yet it is associated with loss of cervical mobility at fused segments, causing compensatory motion at adjacent segments.5-8 This can lead to degeneration at those adjacent segments, necessitating future procedures. 23 Meanwhile CDA is a safe, motion-preserving alternative to traditional ACDF, yet indications are narrow.24-27 Furthermore, there is still a paucity of studies demonstrating the long-term efficacy of CDA compared to ACDF. HS gives the provider the opportunity to tailor the treatment of multi-level cervical DDD to the specific pathology of each vertebrae, which may expand its range of indications while preserving motion and mitigating adjacent segment disease. 28
Previous literature has demonstrated similar short term perioperative outcomes between HS and ACDF.18,29-31 Studies completed using national claims databases found no significant differences in 30 day perioperative complications between two-level ACDF and two level HS.18,31 The present study, which includes a larger sample size and longer follow-up period found that HS was associated with fewer overall major and minor complications within 90 days postoperatively. Furthermore, there were no increased risk in surgical complications, including dysphagia and surgical site infection following HS. 18,29,30,32 This may be explained by the fact that patients selected in the HS group tend to be younger, and healthier, which the present study attempted to control for by matching.18,31 In addition to age and comorbidities, strict surgical indications are often used to screen patients for HS. Common contraindications are similar to those described for CDA and include advanced osteoporosis, facet arthropathy, or DDD.33,34
Given the novel nature of HS, studies assessing long-term reoperation in HS are limited. Zigler et al. found that in 115 hybrid surgeries followed for a mean of 6.5 years, only 6 required reoperation with 5 due to adjacent segment disease, and 1 due to pseudarthrosis. 35 HS implant failure is also rare, and is not usually the cause of reoperation.32,34,35 Scott-Young et al. reported similarly low revision rates of 1.9% following HS, with the most common perioperative indication for reoperation as drainage of hematoma. 34 This complication appears to be common as previous studies have also highlighted an increased risk of hematomas requiring surgical drainage after HS.31,36 While the present study was unable to assess the reason for reoperation, the rates of two-year reoperation was 4.5%, similar to literature findings with rates ranging from 0 to 8%.30,34,35
To date, few studies exist comparing the cost-effectiveness of HS to ACDF. The majority of previous research has assessed two level CDA and two level ACDF.37-39 These studies have shown that while CDA is a more expensive surgery, ACDF is associated with longer time to recovery, and greater overall cost due to loss of productivity.37,39,40 The present study revealed that patients undergoing HS are associated with decreased cost of hospitalization compared to the ACDF group, but the 90-day postoperative costs were similar across cohorts. While matching was utilized in this study, it remains challenging to account for every covariate. Patient cohorts undergoing ACDF are likely to have a greater comorbidity burden resulting in increased hospital costs.18,31 Further studies are necessary to assess cost-utility of HS compared to two-level ACDF surgeries.
Inherent limitations exist with any database study. A national database such as PearlDiver relies on accurate data entry and is always subject to input error. However, it is estimated that the administrative errors are roughly only 1.1-1.3%. 41 This study was also unable to assess important clinical factors such as changes in functional status using validated scales like the neck disability index or assess postoperative changes in range of motion. Additionally, intraoperative variables, and specific surgical technique for ACDF and hybrid (graft with plating vs anchored or non-anchored cages) could not be assessed. Finally, assessment of longer follow-up past 2 years was limited by adequate sample size. Regardless, the present study provides support that HS is not only cost-effective, but also a safe alternative to ACDF in appropriate candidates.
Conclusion
The present study adds to the growing consensus that HS is at least as safe and effective as other surgical treatments for treating cervical disease in appropriately selected patients. Hybrid surgery provides similar long-term reoperation rates as ACDF, with a lower rate of 90-day complications, readmission and decreased total hospital costs.
Footnotes
Author’s Note
IRB: This study was deemed exempt by the institutional review board
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Level of Evidence
III
Appendix
Inclusion Codes
TDA
CPT-22856
ACDF
CPT-22551, CPT-22554, CPT-22585
ACDF Multi-level
CPT-22585, CPT-22552
Exclusion Codes
Posterior Cervical Procedures
CPT- 22590, CPT-22600, CPT-22840
Spinal Trauma
ICD-9-CM: 805.10, 805.2-5, 805.8-9, 806.00, 806.04-05, 806.4-5, 806.8-9, 905.1
ICD-10-CM: M48.40XA, M48.42XA, M48.42XS, M48.44XA, M48.46XA, M48.47XA, M48.48XA, M48.50XA, M48.52XA, M48.53XA, M48.54XA, M48.55XA, M48.56XA, M80.08XA, M80.88XA, S12.00A, S12.00B, S12.01A, S12.01XA, S12.01XB, S12.02XA, S120.30A, S120.31A, S120.40A, S120.41A, S120.41B, S120.90A, S120.90B, S120.91A, S120.91B, S121.00A, S121.00D, S121.90A, S121.91A, S122.00A, S122.01A, S122.30A, S122.90A, S122.90B, S122.91A, S122.91B, S123.00A, S123.00B, S123.01A, S123.01B, S123.30A, S123.90A, S123.91A, S123.91B, S124.00A, S124.00B, S124.01A, S124.01B, S124.30A, S124.90A, S124.91A, S125.00A, S125.00B, S125.01A, S125.90A, S125.90B, S125.91A, S126.00A, S126.00A, S126.01A, S126.30A, S126.31A, S126.91A, S220.00A, S220.01A, S220.02A, S220.08A, S220.09A, S220.09B, S220.10A, S220.11A, S220.12A, S220.18A, S220.19A, S220.20A, S220.22A, S220.28A, S220.29A, S220.29B, S220.30A, S220.30B, S220.31A, S220.32A,S220.32A, S220.32B, S220.32B, S220.38A, S220.39A, S220.40A, S220.40B, S220.41A, S220.41B, S220.42A, S220.48A, S220.49A, S220.50A, S220.51A, S220.52A, S220.58A, S220.59A, S220.60A, S220.61A, S220.62A, S220.68A, S220.69A, S220.69B, S220.70A, S220.70B, S220.71A, S220.72A, S220.72B, S220.78A, S220.78B, S220.79A, S220.79B, S220.80A, S220.80B, S220.81A, S220.81B, S220.82A, S220.82B, S220.88A, S220.88B, S220.89A, S220.89B, S320.00A, S320.00B, S320.01A, S320.01B, S320.02A, S320.02B, S320.08A, S320.09A, S320.10A, S320.10B, S320.11A, S320.11B, S320.12A, S320.12B, S320.18A, S320.19A, S320.20A, S320.20B, S320.21A, S320.22A, S320.28A, S320.29A, S320.30A, S320.31A, S320.32A, S320.38A, S320.39A, S320.40A, S320.41A, S320.42A, S320.48A, S320.49A, S320.51A, S320.52A, S320.58A, S320.59A, S329.XXA, S329.XXB, S329.XXD, S329.XXG, S329.XXK, S329.XXS
Spinal Neoplasm
ICD-9-CM: 192.2, 198.3, 225.3, 237.5
ICD-10-CM: C41.2, C72.0, D16.6, D33.4, D43.4
Spinal Infection
ICD-9-CM: 324.1, 324.9
ICD-10-CM: M46.39, G06.1, G07
