Abstract
Study Design
Retrospective cohort study.
Objective
To evaluate whether undergoing total hip arthroplasty (THA) influences the risk of subsequent lumbar intervention in patients with lumbar degenerative disease (LDD) and hip osteoarthritis (HOA), and to identify patient-specific risk factors and procedural characteristics associated with post-THA lumbar intervention.
Methods
Utilizing the PearlDiver Mariner Database (2010-2023), 1 194 269 patients with LDD and HOA were included, 16.3% of whom underwent THA. Patients with prior lumbar surgeries, trauma, infection and malignancy were excluded. Multivariable logistic regression adjusted for demographics, comorbidities, and scoliosis was performed. Subgroup analyses assessed procedural characteristics, lumbar pathologies, and comorbidities within the THA cohort.
Results
THA was associated with reduced odds of lumbar decompression (OR 0.42, 95% CI [0.41-0.44]) and fusion (OR 0.42, 95% CI [0.40-0.44], both P < 0.001). Predictors of lumbar surgery after THA included preoperative opioid use, obesity, fibromyalgia, depression, diabetes, scoliosis, spondylolisthesis, myelopathy, radiculopathy, stenosis, and spondylolysis. Unilateral THA was associated with significantly higher odds of subsequent lumbar intervention compared with the reference group, whereas bilateral THA was associated with significantly lower odds of subsequent lumbar intervention compared with the same reference group. Risk was highest in patients aged 60-69 years but decreased in those over 69. THA also delayed the average time to lumbar intervention.
Conclusion
THA, particularly bilateral procedures, was protective against future lumbar surgery in patients with HOA and LDD. Unilateral THA was less protective, potentially reflecting spinopelvic imbalance. These findings refine understanding of hip-spine syndrome and support prioritizing timely hip arthroplasty in high-risk patients with severe HOA.
Keywords
Introduction
Degenerative hip and spine pathologies are among the most prevalent musculoskeletal pathologies. 1 While total hip arthroplasty (THA) and lumbar fusion surgery (LFS) effectively address localized pathology, their biomechanical interplay continues to reveal complex relationships. Recent cohort analyses suggest that total hip arthroplasty (THA) has a limited impact on long-term low back pain, challenging prior assumptions about its therapeutic benefit for referred pain between the hip and spine.2-4 This paradox highlights the need to reevaluate the hip-spine relationship through large-scale cohort studies.
The concept of “Hip-Spine Disorder,” first described by Offierski and MacNab (1983), 2 now requires refinement given new evidence from finite element analyses 5 and large-scale cohort studies.2,6 While early classifications emphasized overlapping symptoms, more recent studies have begun to highlight potential mechanical and anatomical links between the hip and spine. Emerging evidence from imaging and cohort data points to complex interdependencies, suggesting that the relationship may extend beyond symptom-based categorizations.5,6
Recent research challenges traditional biomechanical assumptions. Although pelvic parameters like tilt, slope, and incidence are important for surgical planning, target trial emulation studies 2 show no causal link between THA and lumbar degeneration after adjusting for age, BMI, and disease severity.
The clinical imperative now focuses on whether THA-induced alterations in hip-lumbar mobility predispose patients to subsequent lumbar surgeries, such as decompression or fusion in patients with hip osteoarthritis (HOA) and lumbar degenerative disease (LDD). Despite growing recognition of hip-spine syndrome, there remains a gap in understanding whether restoring hip biomechanics through THA can mitigate or delay the need for lumbar interventions, particularly in patients with pre-existing lumbar degenerative disease. Recent cohort studies have offered conflicting evidence.2-4,7,8 However, these studies often lack comprehensive adjustment for confounding factors and do not examine the nuanced roles of unilateral vs bilateral THA, comorbidities, and age-specific risks.
Therefore this study aims to determine whether patients with a history of LDD and THA are more likely to undergo subsequent lumbar spine fusion and/or decompression compared to those with no THA. This study also aims to determine risk factors for lumbar spine surgery in patients with a history of THA.
Methods
Data Source and Study Population
This study utilized the PearlDiver Mariner Database (PearlDiver Technologies, Colorado Springs, CO, USA) to identify patients diagnosed with LDD and HOA who underwent THA between 2010 and April 2023. The PearlDiver database is a nationwide insurance claims repository containing approximately 170 million de-identified patient records, including demographic, diagnostic, and procedural data from various payer types across inpatient and outpatient settings. As the study was retrospective and relied on anonymized data, informed consent was waived, and institutional review board approval was not required.
Identification of Study Cohorts
Adult patients were identified using diagnostic codes from the International Classification of Disease 9th (ICD-9) and 10th (ICD-10) editions, as well as Current Procedural Terminology (CPT) codes. Patients with LDD and HOA were included if they underwent THA after being diagnosed with LDD and HOA. Individuals were excluded if they underwent lumbar decompression or fusion prior to undergoing THA or surgical indications for trauma, malignancy, or infection. A control group comprising 1 000 000 patients with LDD and HOA but no history of THA during the study period was also identified. Supplemental Table 1 provides a detailed list of diagnoses and procedural codes used for cohort identification.
Study Objectives
The primary objective was to evaluate whether undergoing THA in patients with LDD and HOA affected the risk of subsequent lumbar decompression and fusion compared to a control group of patients with LDD and HOA who did not undergo THA.
The secondary objective was to evaluate underlying comorbidities or risk factors for subsequent lumbar decompression and fusion surgeries in the THA cohort, including the time duration between initial diagnosis of LDD and these subsequent surgeries. Risk factors assessed included age, gender, Elixhauser Comorbidity Index (ECI), specific comorbidities, lumbar pathologies (scoliosis, history of spondylolysis, spondylolisthesis, myelopathy and radiculopathy), and unilateral and bilateral THA.
Statistical Analysis
Statistical analyses were performed using RStudio (Version 4.4.2) within the PearlDiver Mariner Database platform. Comparisons of continuous variables were performed using Student’s t-test, whereas categorical variables were analyzed using Pearson’s chi-squared test of independence. Univariate and multivariable logistic regression models adjusted for age, gender, ECI and scoliosis were employed to assess the risk of subsequent lumbar decompression and fusion surgeries in patients with LDD and HOA who underwent THA compared to the control group. Additionally, univariate and multivariable logistic regression analyses identified patient-specific comorbidities and procedural risk factors for subsequent lumbar surgeries within the THA cohort. Statistical significance was defined by a P-value <0.05 for all analyses.
Results
Patient Demographics
Patient Demographics.

Abbreviations: n = number, LDD = Lumbar degenerative disease, HOA = Hip osteoarthritis, THA = Total hip arthroplasty, ECI = Elixhauser Comorbidity Index. Bold P values represent statistically significant result.
Risk of Lumbar Decompression and Fusion Surgery Following THA
Odds Of Subsequent Lumbar Decompression and Lumbar Fusion Surgery in Patients With LDD AND HOA undergoing THA.

Abbreviations: LDD = Lumbar degenerative disease, HOA = Hip osteoarthritis, THA = Total hip arthroplasty. Bold P values represent statistically significant result.
Time to Subsequent Surgery
The average time between initial diagnosis of LDD and subsequent decompression was 6.55 ± 3.17 years in the THA cohort, compared to 3.1 ± 3.2 years in the non-THA cohort. For LFS, the average time between initial diagnosis of LDD and LFS was 6.6 ± 3.0 years in the THA cohort, compared to 3.4 ± 3.2 years in those without THA.
Risk Factor Analysis for Subsequent Decompression in THA Cohort
Odds of Subsequent Lumbar Decompression And Lumbar Fusion Surgery in THA Cohort.
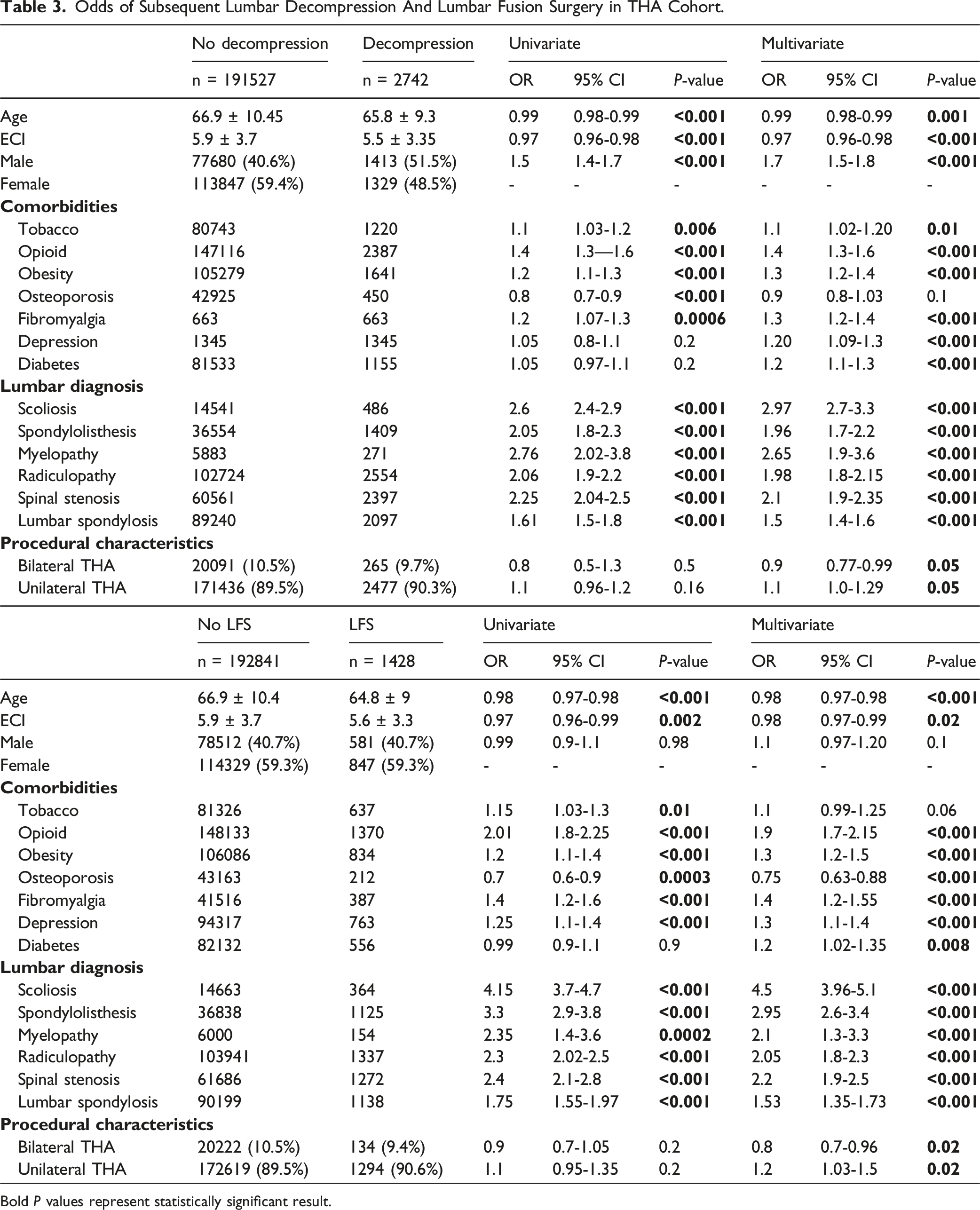
Bold P values represent statistically significant result.
Risk Factor Analysis for Subsequent Lumbar Fusion in THA Cohort
For LFS, preoperative opioid use (OR 1.9, 95% CI [1.7-2.15], P < 0.001), obesity (OR 1.3, 95% CI [1.2-1.5], P < 0.001), fibromyalgia (OR 1.4, 95% CI [1.2-1.55], P < 0.001), diabetes (OR 1.2, 95% CI [1.02-1.35], P = 0.008) and depression (OR 1.3, 95% CI [1.1-1.4], P < 0.001) were significant predictors. Osteoporosis was associated with less subsequent surgery (OR 0.75, 95% CI [0.63-0.88], P < 0.001). Lumbar diagnoses including scoliosis (OR 4.5, 95% CI 3.96-5.1, P < 0.001), history of spondylolisthesis (OR 2.95, 95% CI 2.6-3.4, P < 0.001), myelopathy (OR 2.1, 95% CI 1.3-3.3, P < 0.001), radiculopathy (OR 2.05, 95% CI 1.8-2.3, P < 0.001), spinal stenosis (OR 2.2, 95% CI 1.9-2.5, P < 0.001), and lumbar spondylosis (OR 1.53, 95% CI 1.35-1.73, P < 0.001) were all independently associated with increased odds of fusion. Similar to decompression, bilateral THA showed a protective effect against LFS risk (OR 0.8, 95% CI [0.7-0.96], P = 0.02), when compared to unilateral intervention (OR 1.2, 95% CI [1.03-1.5], P = 0.02) (Table 3).
Stratification by Age in the THA Cohort
Odds Of Subsequent Lumbar Decompression And Lumbar Fusion Surgery Stratified By Age In THA Cohort.
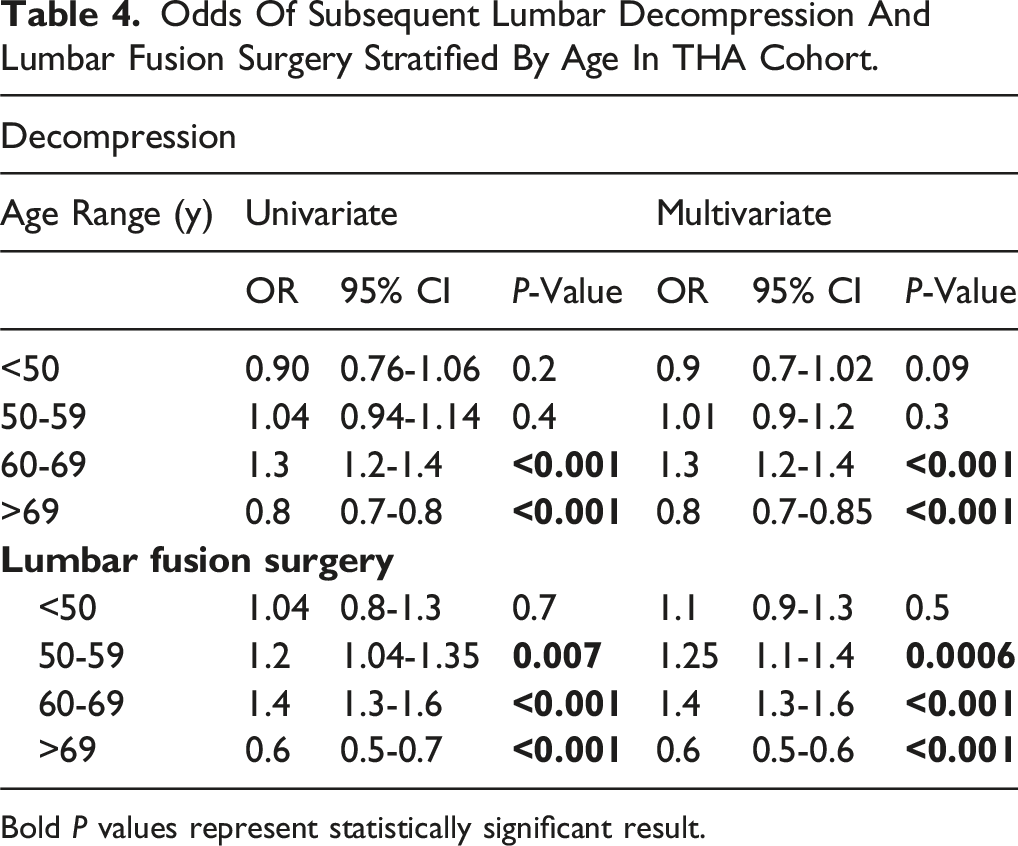
Bold P values represent statistically significant result.
Discussion
The relationship between THA and subsequent lumbar decompression and fusion surgery in patients with HOA and LDD remains a critical area of investigation in hip-spine syndrome research. Our findings reveal a protective association between THA and subsequent LFS and decompression surgeries. Specifically, THA was associated with significantly lower odds of undergoing LFS (OR 0.42, 95% CI 0.40-0.44, P < 0.001) and decompression (OR 0.42, 95% CI 0.41-0.44, P < 0.001) after adjusting for relevant confounders. These findings contrast with earlier studies suggesting an increased risk of complications post-THA, 8 highlights the importance of robust multivariable analysis and careful adjustment for patient-level factors.
Interestingly, these protective findings align closely with the studies by Zhang et al. (2023) and Wu et al (2023).3,4 Zhang et al found that patients undergoing THA first had a significantly lower risk of subsequent lumbar spine surgery within 2 and 5 years (5.7% vs 23.7% at 2 years, P < 0.001). 3 Similarly, Wu et al. demonstrated that patients who had THA first had a significantly lower risk of subsequent lumbar surgery (6.8% vs 62% in those who had lumbar surgery first, P < 0.001), with a 5-year survivorship free of lumbar spine surgery of 93.9% in the THA-first group vs 44.7% in the lumbar-first group (OR 0.61, CI: 0.52-0.70, P < 0.001). 4 Both studies emphasize the advantage of addressing hip pathology first to reduce future lumbar surgical needs. Our findings extend these observations by offering a more refined quantitative risk reduction (OR 0.42) and exploring additional factors such as age-specific risk, comorbidities, influence of spinal pathologies and THA laterality (unilateral vs bilateral), which previous research did not examine. Similarly, our findings align in part with a study by Emily et al (2024), which reported a significantly increased risk of spinal reoperation at three and five years (OR 1.61 and 1.87, respectively) in patients with severe hip OA who underwent lumbar fusion first. 7 The observed difference likely arises from differences in surgical sequencing and patient selection. Emily et al studied patients who had lumbar fusion first while their hip OA remained untreated, leading to persistent hip stiffness and compensatory lumbar stress during the critical recovery period. In contrast, our cohort and those of Zhang et al and Wu et al comprised patients who had already undergone THA, restoring hip mobility before evaluating lumbar spine surgery risk. This sequencing difference likely explains why our study and these of others observed a protective effect of THA against subsequent lumbar procedures.
Importantly, unlike prior studies, our analysis included common spinal pathologies such as scoliosis, spondylolisthesis, stenosis, myelopathy, and radiculopathy. Each of these diagnoses independently increased the odds of subsequent LFS or decompression, with scoliosis demonstrating the strongest association (OR 2.97 for decompression and OR 4.5 for fusion, both P < 0.001). This suggests that while THA may mitigate risk by restoring hip biomechanics, coexisting structural spinal deformities remain dominant drivers of surgical progression. Clinically, this highlights the need for individualized risk counseling in elderly patients with combined HOA and complex spinal pathology.
Our study is the first that uniquely highlights the nuanced role of unilateral vs bilateral THA in modulating lumbar surgery risk. Compared with bilateral THA, we found that unilateral THA was associated with a statistically reduced beneficial effect on subsequent lumbar outcomes, showing higher risks of lumbar decompression and fusion surgery (OR 1.1 and 1.2 respectively). This finding may potentially be explained by temporary spinopelvic imbalances in unilateral THA that lead to asymmetrical hip mobility during recovery; further investigation is warranted as the underlying mechanism remains unclear. In contrast, bilateral THA was associated with a statistically significant decrease in decompression and LFS risk (OR 0.9 and 0.8 respectively) on multivariable analysis, suggesting that symmetrical hip restoration may eliminate compensatory lumbar overload. This post-THA restoration of hip flexion leads to changes in spinopelvic alignment, including normalization of pelvic tilt and sacral slope dynamics. 9
While Wu et al identified progressive neurologic deficits and neurogenic claudication as predictors of subsequent lumbar surgery, 4 our study broadens the scope by identifying opioid use, fibromyalgia and depression as the strongest independent predictors of LFS and decompression risk, paralleling findings that centralized pain syndromes increase elective surgery likelihood.10,11 Clinically, they highlight an opportunity for preoperative optimization; addressing chronic opioid dependence and untreated depression may meaningfully alter the trajectory of surgical care. We also observed osteoporosis was associated with lower odds of LFS (OR 0.75, P < 0.001) and a moderate risk from obesity for future lumbar decompression and fusion. This aligns with studies linking obesity to adjacent segment disease (ASD) 12 and supports clinical observations that surgeons avoid fusion in osteoporotic patients due to instrumentation failure concerns.13,14 Diabetes emerged as an independent predictor of both decompression (OR 1.2, P < 0.001) and fusion (OR 1.2, P = 0.008). This finding is clinically relevant, as diabetes has been linked to impaired bone healing, microvascular changes, and increased perioperative complication rates. 15
Age-stratified analyses further contextualize these findings, with the highest risk for LFS and decompression occurring in the 60-69-year age group (OR 1.3 for LFS and 1.4 for decompression), reflecting the intersection of advanced degeneration and maintained surgical fitness. The protective effect in patients >69 years of age (OR 0.8 for decompression and 0.6 for LFS) mirrors national trends showing declining elective intervention rates due to underlying comorbidity burden, 16 as evidenced by increased ECI in THA cohort vs control. The observed delay in time to LFS (6.6 ± 3.0 years) and decompression (6.55 ± 3.17 years) in patients with THA also supports the idea that THA may delay but not fully eliminate the need for subsequent spine surgery, a temporal relationship not explicitly explored by Zhang et al or Emily et al, and only partially addressed by Wu et al.3,4,7 For clinicians and surgeons, this underscores the importance of timing: addressing hip disease in the 60-69 window may not only improve mobility but also delay or reduce the need for subsequent lumbar interventions.
From a clinical perspective, these findings offer several actionable insights. In elderly patients with dual pathology, prioritizing THA; particularly bilateral; may provide both symptomatic relief and biomechanical stabilization that reduces or delays the need for lumbar surgery. This supports prior work by Zhang et al and Wu et al, and emphasizes the biomechanical advantage of treating hip pathology first. Our study also highlights important nuances: unilateral THA carries a modestly reduced beneficial effect compared with bilateral THA, and structural spinal pathologies such as scoliosis and spondylolisthesis can attenuate the protective effect. Age-stratified analyses further reinforce the importance of surgical timing, as performing THA earlier in the degenerative cascade may maximize its protective effect against downstream lumbar procedures. Patient counseling should therefore integrate comorbidity and psychosocial risk profiles, as factors like opioid use, depression, obesity, and diabetes emerged as significant drivers of future surgery. Collectively, these findings underscore the need for a multidisciplinary approach that combines hip mobility restoration with optimization of medical and psychosocial comorbidities to improve long-term outcomes. Future studies should incorporate radiographic spinopelvic alignment metrics, hip osteoarthritis staging with the Kellgren–Lawrence classification, and patient-reported outcomes to clarify the biomechanical and functional impacts of unilateral vs bilateral THA in this complex population.
This study is not without limitations. As a retrospective database study, causality cannot be established, and findings are subject to coding inaccuracies and unmeasured confounders such as radiographic alignment, symptom severity, and surgical technique. Because PearlDiver relies on CPT and ICD-9/10 coding, radiographic confirmation of degenerative lumbar conditions (DLK) and clinical assessment of symptom severity were not available, and the stage of hip osteoarthritis could not be determined. Similarly, functional measures such as patients ability to walk independently, use assistive devices, or activity level could not be assessed. Although we included spinal pathologies such as scoliosis, spondylolisthesis, radiculopathy, and stenosis in our multivariate models, we could not characterize their radiographic severity or progression, which may confound associations between THA and subsequent lumbar surgery. Selection bias may also influence which patients undergo THA or lumbar fusion, potentially affecting observed relationships. A major strength of this study is the use of a large, nationally representative claims database (PearlDiver), allowing for robust statistical power and generalizability across diverse patient populations. The study also adjusted for multiple confounders using multivariable regression, enhancing the validity of the findings. Additionally, the inclusion of a large control cohort and comprehensive comorbidity analysis provided insight into patient-specific risk factors for lumbar surgeries.
Conclusion
This study found that THA in patients with LDD and HOA is associated with a significantly reduced risk of subsequent lumbar decompression and fusion surgeries. Bilateral THA demonstrated a protective effect against future lumbar fusion, whereas unilateral THA was linked to a comparatively reduced beneficial effect, likely due to transient biomechanical imbalances. Beyond procedural characteristics, underlying lumbar diagnoses such as scoliosis, spondylolisthesis, myelopathy, radiculopathy, and spinal stenosis were among the strongest predictors of future lumbar surgery, often outweighing the protective benefit of THA. Importantly, comorbidities such as opioid use, fibromyalgia, depression, diabetes and obesity emerged as significant predictors of lumbar intervention risk within the THA cohort, underscoring the importance of comprehensive preoperative risk stratification. Our findings also reveal that THA delays the need for future lumbar surgeries, suggesting that timely hip intervention may play a role in modulating spinopelvic biomechanics and disease progression. Collectively, these insights highlight the importance of a multidisciplinary approach to hip–spine syndrome, integrating surgical sequencing with careful consideration of lumbar pathology and patient-specific comorbidities to optimize long-term outcomes.
Supplemental Material
Supplemental Material - Long-Term Impact of Total Hip Arthroplasty on Subsequent Lumbar Spine Surgery iIn Patients wWith Hip-Spine Disease: A Nationwide Analysis of Risk Factors and Outcomes
Supplemental Material for Long-Term Impact of Total Hip Arthroplasty on Subsequent Lumbar Spine Surgery iIn Patients wWith Hip-Spine Disease: A Nationwide Analysis of Risk Factors and Outcomes by Ali Issani, William J. Karakash, Aidan Lindgren, Daniel Rusu, Henry Avetisian, MirBahadur Athari, Dil Patel, Raymond J. Hah, Ram K. Alluri, John C. Liu, Jeffrey C. Wang in Global Spine Journal
Mnemonic
THA: Total hip arthroplasty
LFS: Lumbar fusion surgery
LDDD: Lumbar degenerative disease
HOA: Hip osteoarthritis
ICD: International Classification of Disease
CPT: Current Procedural Terminology
ECI: Elixhauser Comorbidity Index
ASD: Adjacent segment disease
DLK: Degenerative lumbar conditions
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Jeffrey C. Wang has received intellectual property royalties from Zimmer Biomet, NovApproach, SeaSpine, and DePuy Synthes, and stock options from Bone Biologics, Electrocore, PearlDiver, and Surgitech. Raymond J. Hah has received grant funding from SI bone, consulting fees from NuVasive, and support from the North American Spine Society to attend meetings. Ram K. Alluri has received grant funding from NIH, consulting fees and stock options from HIA Technologies, and payment from Eccential Robotics for lectures and presentations. John C. Liu has received consulting fees from vision, illuminant surgical, Nov approach and Spark spine, and support for attending meetings and/or travel from Global spine congress and NASS summer meeting.
Data Availability Statement
Data is not publicly available but can be available upon request.
Supplemental Material
Supplemental material for this article is available online.
