Abstract
Study Design
Retrospective Cohort Study.
Objectives
In this study, we aimed to evaluate the risk factors associated with cancer-specific survival (CSS) of patients with spinal and pelvic diffuse large B-cell lymphoma (SP-DLBCL) and construct a competing risk-based model for predicting 3- and 5-year CSS.
Methods
Data on patients diagnosed between 2010 and 2019 were obtained from the Surveillance, Epidemiology, and End Results (SEER) database. The total dataset was randomly divided into the training and testing datasets at a ratio of 7:3. Factors associated with CSS were identified by competing risk regression for the construction of a nomogram on the training dataset. Further, the testing dataset was used to validate the performance of the nomogram.
Results
Competing risk regression revealed that age, Ann Arbor Stage, and chemotherapy were independent prognostic factors for CSS. The nomogram exhibited satisfactory agreement on calibration plots for the training and testing datasets. The nomogram performed well on the training and testing datasets, exhibiting a good clinical net benefit. Thus, a nomogram-based web calculator was constructed for clinical application.
Conclusion
In this study, we evaluated the risk factors of CSS in patients with SP-DLBCL using competing risk regression analysis. The established nomogram and web-based tool performed well and could be used widely in clinical practice.
Introduction
Primary bone lymphoma (PBL) is a rare extranodal lymphoma, accounting for approximately 5-7% of malignant bone tumors. 1 Non-Hodgkin’s lymphoma (NHL) is the most common type of bone lymphoma, whereas diffuse large B-cell lymphoma (DLBCL) is the most common subtype of NHL.2,3Due to its complex anatomical structure, spinal and pelvic DLBCL (SP-DLBCL) in PBL is a challenging tumor to treat. Different strategies, including chemotherapy, radiotherapy, surgery, and immunotherapy, have been used to treat spinal DLBCL. However, the prognosis is generally poor, and the 5- and 10-year overall survival (OS) rates of patients with SP-DLBCL are approximately 60% and 45%, respectively.4,5
Only a few studies have specifically reported the prognosis of SP-DLBCL. A previous study performed multivariate Cox regression analysis to establish a prognostic model that could predict the 3- and 5-year OS of patients with SP-DLBCL. 4 Nevertheless, the study did not account for a clinically relevant outcome, cancer-specific survival (CSS), and other causes of death (OCOD), in which OCOD is considered a competing risk in CSS but is often ignored. 6 This oversight can introduce potential confusion and bias in predicting outcomes for DLBCL.
Fine and Gray regression model is more accurate than conventional Cox analysis when competing risks are present. It has been used to perform survival analysis in many cancers to account for competing risks.7-9 Nevertheless, no study has used this method to predict CSS in SP-DLBCL. Based on this, in the present study, we evaluated the risk factors associated with the CSS of SP-DLBCL and constructed a competing risk-based model for prognostic prediction. Our model can help clinicians precisely and easily evaluate prognosis in the clinical decision making process.
Methods
Study Population
Data of patients with SP-DLBCL (ICD-O-3 histology code 9680/3, primary site code C41.2 and C41.4) between 2010 and 2019 were obtained from the Surveillance, Epidemiology, and End Results (SEER) database. The exclusion criteria were as follows: (1) no positive diagnostic confirmation, (2) cause of death unknown; (3) not primary tumor, (4) only autopsy or death certificate available, and (5) follow-up time less than 5 years (diagnosed after 2016 which could not reach a 5-year follow-up length, as the extracted SEER data was updated to 2011). The process of inclusion and exclusion was presented in Figure 1. The SEER database is a publicly available database that obtains information from population-based cancer registries. Ethical approval was not needed due to the anonymization of patients. Workflow of the Patient Selection and Model Development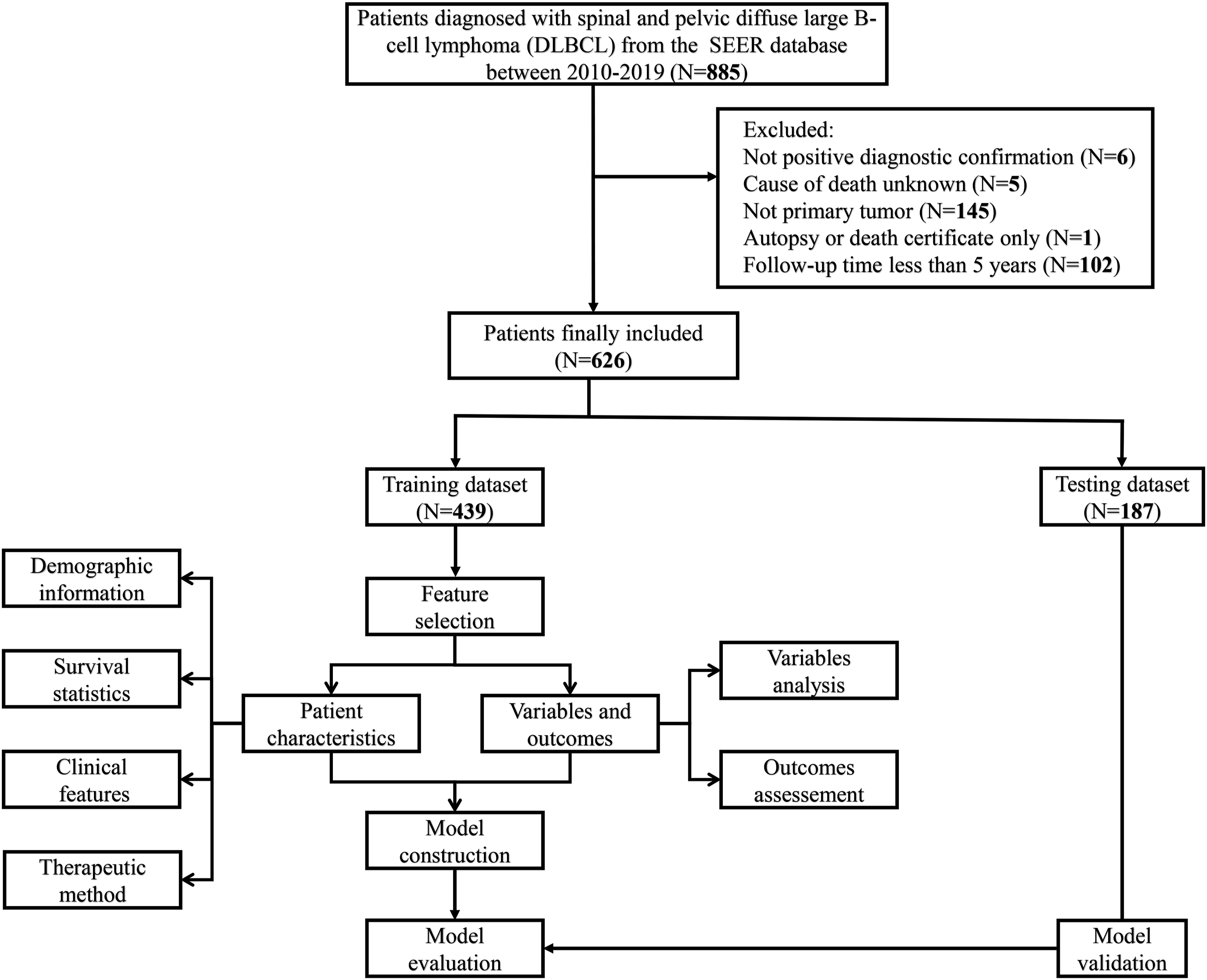
Variables and Outcomes
The clinical characteristics included demographic data, such as age, sex, race, and marital status; tumor diagnosis, including primary site and stage; treatment choice, including primary site surgery, chemotherapy, and radiotherapy; and prognosis, including follow-up time. Similarly, survival outcomes included CSS and other cause-specific survival (OCSS), causes of death (COD) included cancer-specific death (CSD) and OCOD, and OCOD was considered a competing risk for CSD. The primary outcome was the detection of the association between characteristic variables and CSS using the Fine and Gray competing risk-based model, which was expressed by the corresponding subdistribution hazard ratio (SHR) and 95% confidence interval (CI). 10 The secondary outcomes included risk factors for CSD using logistic regression analysis. Additionally, the Cox proportional hazards regression model was also used to investigate the prognostic variables of CSS to calculate hazard ratio (HR) and 95% CI and compare the results with those of the competing risk-based model. Competing risk regression was performed for risk factors for OCSS. The importance of each independent variable was ranked based on their contribution and measured in the final regression model as a partial χ2 statistic minus the variable degrees of freedom (χ2 − df).
Construction and Validation of Nomogram
The total dataset was randomly divided into training and testing datasets at a ratio of 7:3. A visual nomogram was constructed to predict the 3- and 5-year CSS based on the independent factors on the training dataset. The performance of the nomogram was validated using the testing dataset. Calibration curves and Integrated Brier scores (IBS) were used to evaluate calibration. The overall Concordance index (C-index) and time-dependent receiver operating characteristic (ROC) curves were used for discrimination analysis. Clinical utility was evaluated by performing Decision curve analysis (DCA). The performance of the nomogram was compared with the performance of the 3 single variables. Patients were then assigned to high-, medium-, or low-risk groups on the basis of the cutoff values of the total scores calculated using the nomogram. Furthermore, the Cumulative Incidence Function (CIF) curve was constructed to distinguish different risk groups of CSS.
Statistical Analysis
Continuous variables were presented as mean ± standard deviation (SD), whereas categorical variables were presented as frequencies and percentages. One-way analysis of variance (ANOVA) and χ2 tests were used to compare continuous and categorical variables, respectively. The log-rank test was performed for Cox regression, whereas the Fine–Gray test was performed for the competing risk model. Missing values were estimated by performing multiple imputation analyses. Logistic regression and polynomial regression were used for binary and categorical features, respectively. Data were obtained using SEER * Stat (version 8.3.9). Optimal cutoff points were determined by the X-tiles program (version 3.5). Statistical analyses and visualizations were performed using R software (version 3.6.1). A Two-tailed P-value of <0.05 was considered statistically significant.
Results
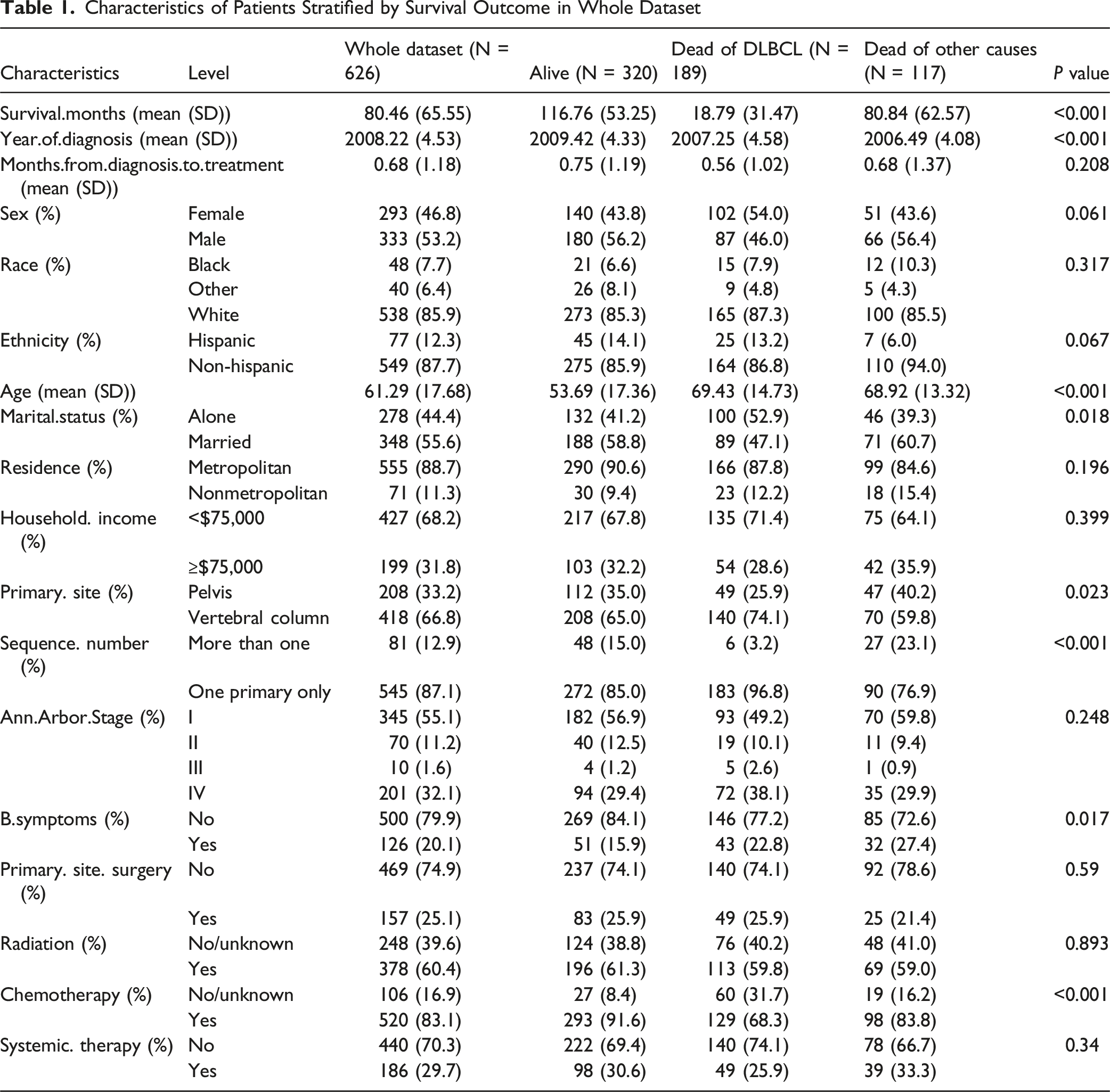
Characteristics of Patients
Characteristics of Patients Stratified by Survival Outcome in Whole Dataset
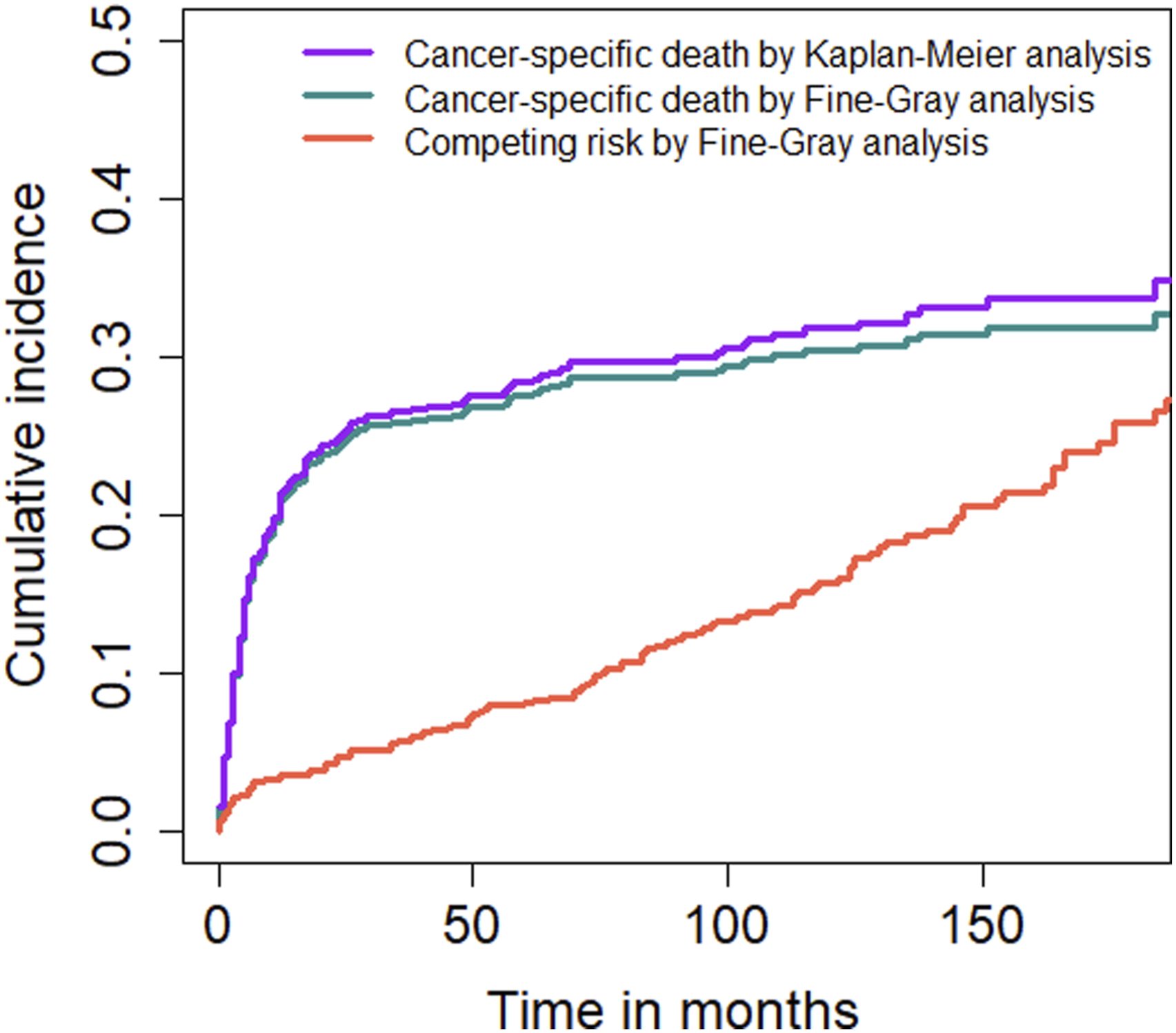
Cumulative Incidences Curves of Cancer and Noncancer Death in Whole Dataset
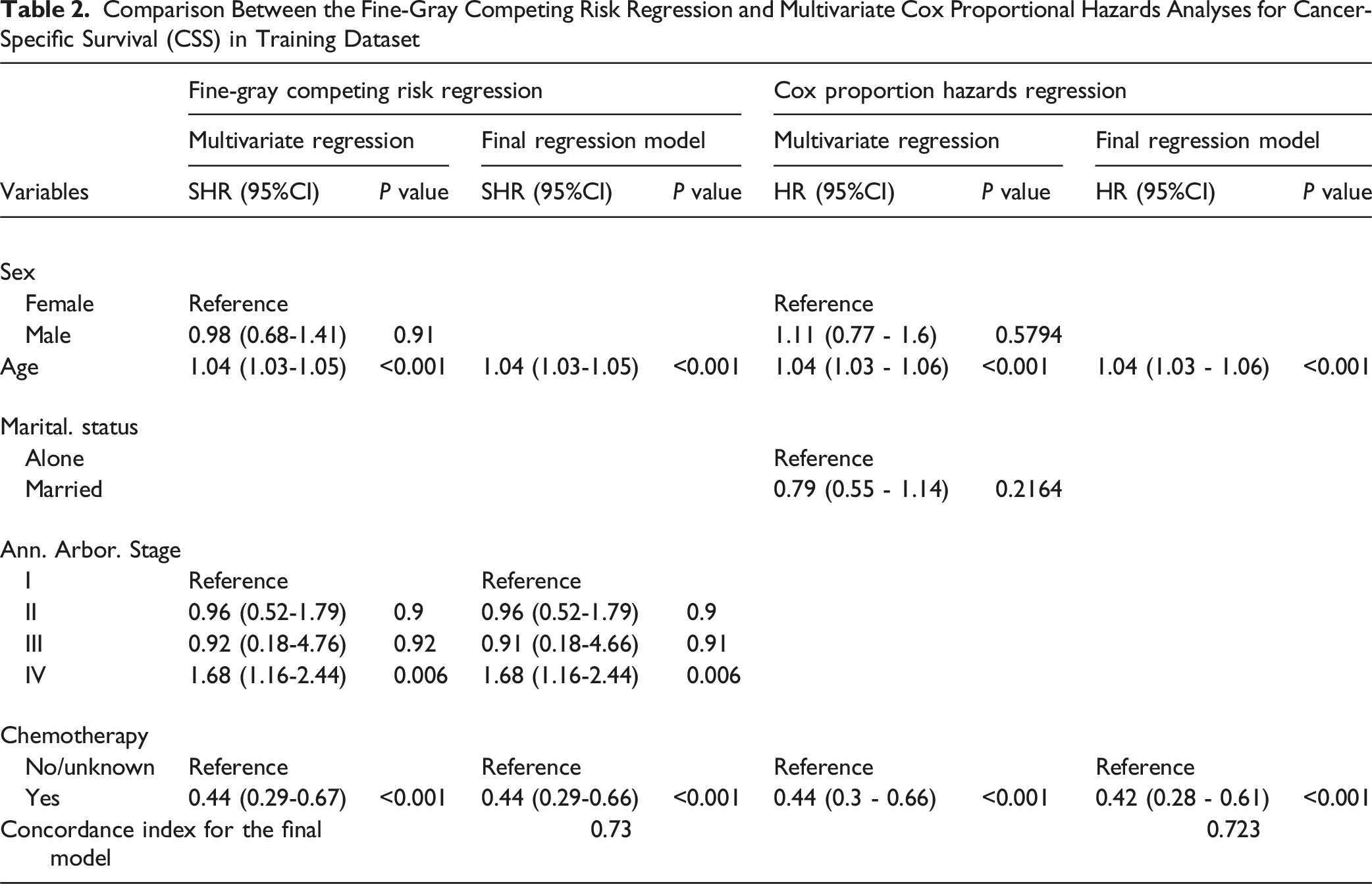
Analyzing Factors Associated with CSS
Comparison Between the Fine-Gray Competing Risk Regression and Multivariate Cox Proportional Hazards Analyses for Cancer-Specific Survival (CSS) in Training Dataset
Detailed Analysis for COD
We had detailed COD for the entire dataset in Online Resource Supplemental Table 6, highlighting heart disease, infectious or parasitic diseases, and pneumonia/influenza as the top three causes of OCOD. In Online Resource Supplementary Table 7, we performed further analysis of the risk factors associated with CSD using binary logistic regression. The multivariate models indicated that the following factors were significantly associated with CSD (P = 0.016 and P = 0.012, respectively): primary site (spine vs pelvis: OR: 1.85, 95% CI: 1.12-3.06), and chemotherapy (yes vs no/unknown: OR: 0.46, 95% CI: 0.26-0.84).
Construction and Validation of Nomogram
The statistically significant factors, such as age, Ann Arbor Stage, and chemotherapy, identified in the multivariate Fine–Gray regression were used to develop a prediction model for 3- and 5-year CSS in patients with SP-DLBCL (Figure 3 and Online Resource Supplemental Figure 2). Age was the most crucial variable for CSS, whereas chemotherapy and Ann Arbor Stage were sequentially given decreasing importance (Online Resource Supplemental Figure 1). The Interactive Nomogram (Produced by R Package ‘Regplot’: https://www.r-project.org) to Predict the 3-, and 5-Year Cancer-specific Survival (CSS) of Patients With Spinal and Pelvic Diffuse Large B-cell Lymphoma (DLBCL), which was Showed by an Example for the Application Based on a Hypothetical Patient (61 Years Old, Stage II, Chemotherapy Performed)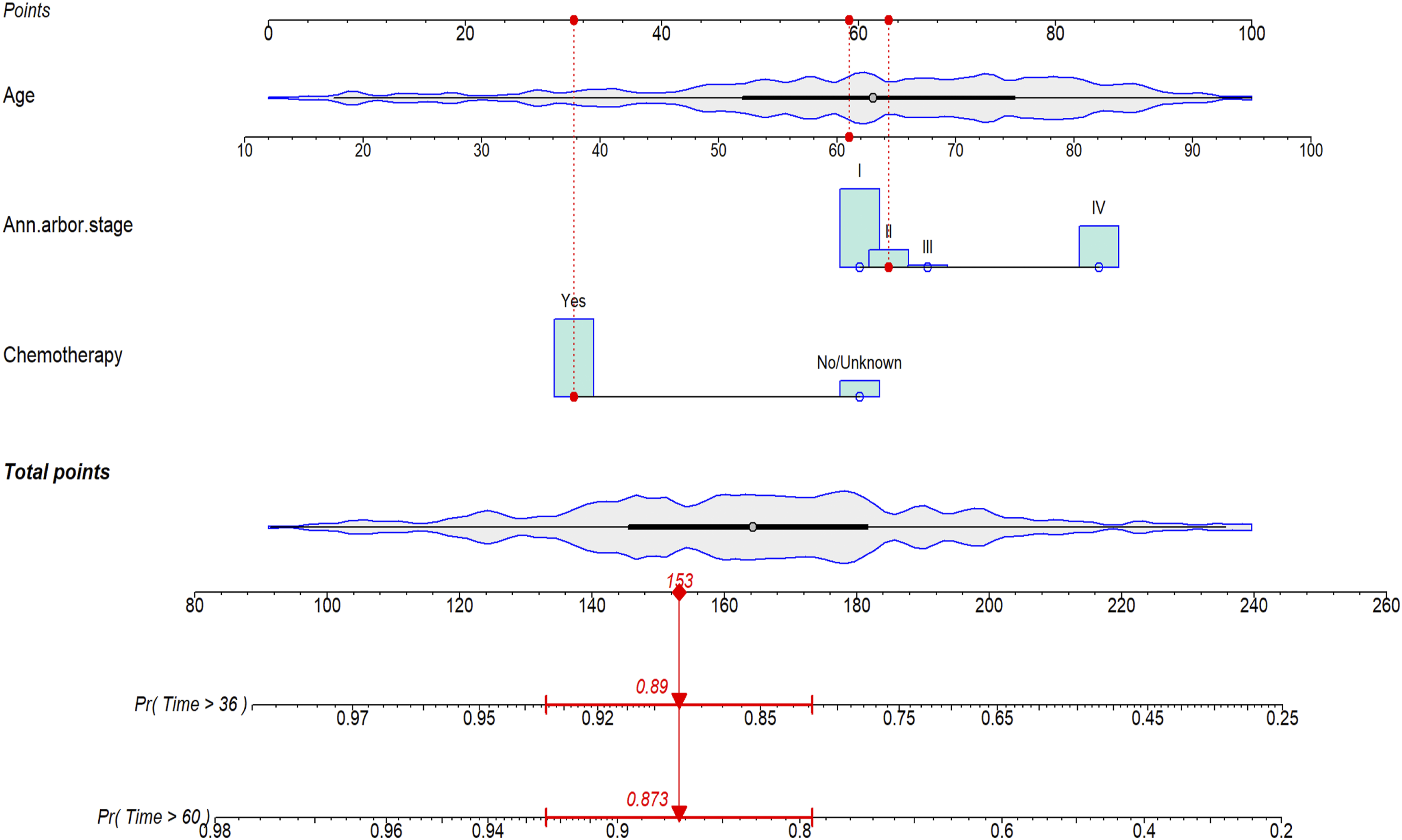
The CSS nomogram exhibited accuracy and effectiveness in predicting 3- and 5-year survival. Calibration curves showed close consistency between predicted and observed CSS at various time points, suggesting good agreement in the training and testing datasets (Figure 4A and 4D). Evaluation metrics, such as Brier’s score and C-index showed superior performance of the nomogram compared with individual predictors (age, Ann Arbor Stage, and chemotherapy) in terms of calibration and discrimination. The mean IBS and C-index values for the nomogram were 0.167/0.171 and 0.728/0.712 for training and testing datasets, respectively (Online Resource Supplemental Figure 3A-B and 3D-E). Evaluation of the Nomogram for Cancer-specific Survival (CSS). (A-C, and G) Evaluation on Training Dataset. (D-F, and H) Evaluation on Testing Dataset. (A, D) 3- and 5-Year Calibration Plots of Nomogram. (B, E) 3-Year and (C, F) 5-Year Area Under the Curve (AUC) for Receiver Operating Characteristic (ROC) Curves of Nomogram, Age, Chemotherapy, Ann Arbor Stage. (G, H) 3- and 5-Year Decision Curve Analysis (DCA) of Nomogram, Age, Chemotherapy, Ann Arbor Stage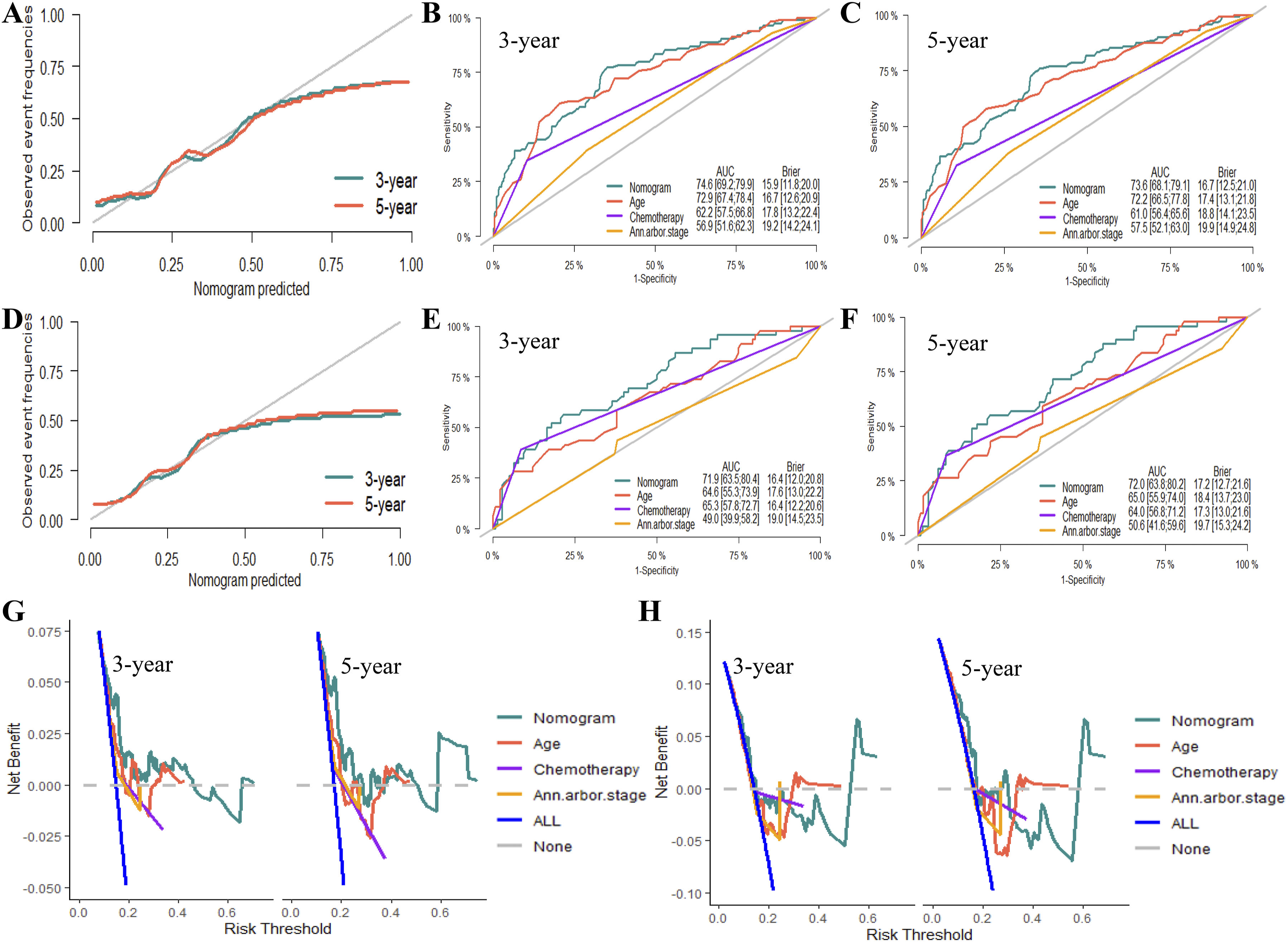
Comparison of the Advantages Between Nomogram and the 3 Single Factors (Age, Chemotherapy, Ann Arbor Stage)
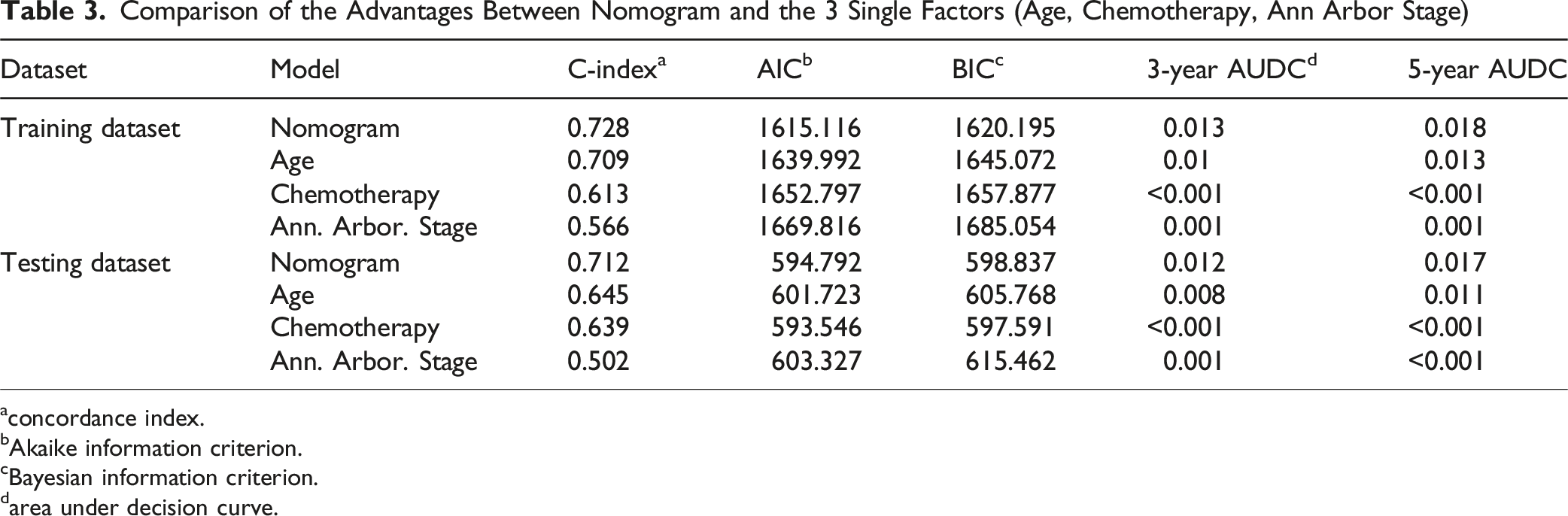
aconcordance index.
bAkaike information criterion.
cBayesian information criterion.
darea under decision curve.
Risk Discrimination and Online Calculator
Patients were categorized into three risk groups based on optimal cutoff values derived from the nomogram total points determined using X-tile analysis (Online Resource Supplemental Figure 4). The discriminatory efficacy of risk groups was illustrated using CIF curves (Figure 5A-B), revealing significant differences in both training and testing datasets. To improve clinical usability, a web-based CSS calculator was developed (Figure 5C, https://vincent--y.shinyapps.io/Online_calculator_for_SP-DLBCL_CSS_prediction/). This tool allows clinicians to input patient clinical characteristics and receive estimated survival projections, as shown in the example of a hypothetical patient (Online Resource Supplemental Figure 5). Prognostic Classification System Stratified by Risk Score and Web-Based Survival Calculator for Cancer-specific Survival (CSS). (A) Prognostic Classification Performance on Training Dataset. (B) Prognostic Classification Performance on Testing Dataset. (C) Web-Based Survival Calculator (https://vincent--y.shinyapps.io/Online_calculator_for_SP-DLBCL_CSS_prediction/)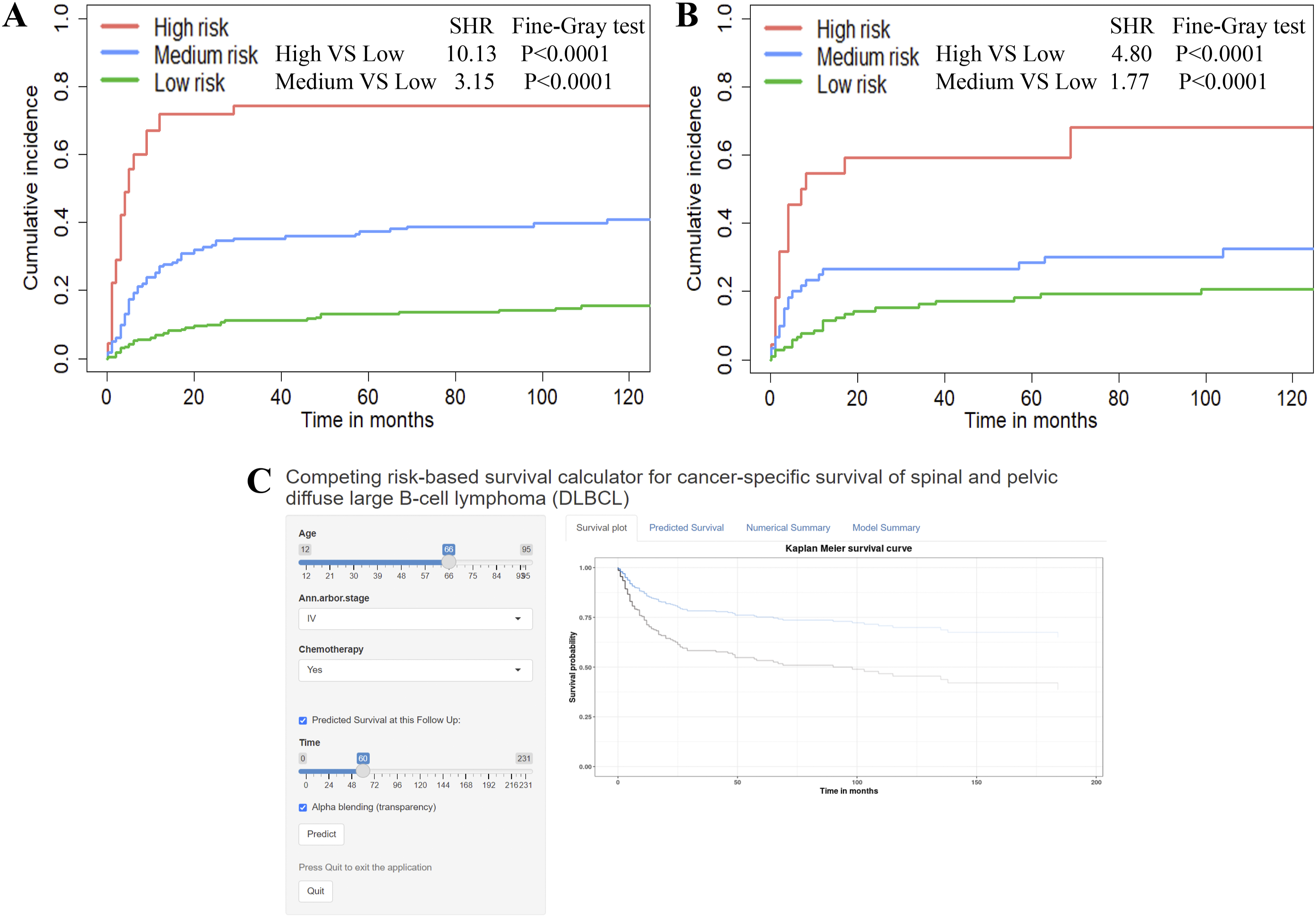
Discussion
SP-DLBCL, the most prevalent type of spinal lymphoma, exhibits an aggressive course and non-specific clinical manifestations, posing challenges for early diagnosis. 11 In this study, based on the Fine–Gray competing risk model, treating OCOD as a competing risk, the relationship between characteristic variables and CSS in patients with SP-DLBCL was analyzed and their role in survival analysis was compared with those in the Cox regression model. Based on the regression analysis outcomes, we constructed a prognostic model visualized as a nomogram to predict CSS in patients with SP-DLBCL. This nomogram showed good calibration and discrimination for objective CSS prediction and exhibited strong clinical utility, enabling digital representation of life expectancy for patients with SP-DLBCL. Both Cox regression and the competing risk model highlighted age and chemotherapy as related factors for CSS, whereas Ann Arbor Stage emerged as another independent prognostic factor identified by the competing risk model. The c-index for the final competing risk regression model surpassed that of Cox regression.
Among all the variables analyzed in this study, age exhibited the most significant effect on CSS (Online Resource Supplemental Figure 1). Advanced age was considered a negative prognostic factor and younger patients with DLBCL had a higher survival rate, which was consistent with the findings of previous studies.2-5,12-15 Older patients often face suboptimal treatment outcomes due to a combination of factors, including age-related comorbidities, reduced tolerance to toxicities of therapies toxicities. For instance, several analyses have shown that older adults are less likely to complete standard chemotherapy regimens like R-CHOP due to treatment-related toxicities, which contributes to worse outcomes.16,17 Further complicating the issue, elderly patients often present with multiple comorbidities, such as cardiovascular or pulmonary conditions, that limit their ability to undergo full-dose treatment regimens. Moreover, even when treatment is administered, the physiological decline associated with aging can reduce the efficacy of therapies. 18 Therefore, despite advances in DLBCL management, ideal treatment outcomes remain difficult to achieve in older patients, and their overall survival prognosis remains poor.
In addition, we found that chemotherapy was the second most important prognostic factor, which was an independent positive prognostic factor in CSS, with significantly better CSS in patients undergoing chemotherapy. Chemotherapy is the preferred treatment strategy for patients with SP-DLBCL, and rituximab along with cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP regimen) is currently recommended13,19 with standard chemotherapy, which can cure approximately 50% to 70% of patients.20,21 Ann Arbor Stage was the third most important prognostic factor for CSS. We identified stage IV as a risk factor for poor survival prognosis in patients with SP-DLBCL, which was consistent with previous studies on DLBCL that found a worse prognosis in patients with advanced disease than in those with early disease.3-5,22,23
Clinical practice gives immense importance to prognostication and anticipated survival, which allow clinical decision-making, allocation of medical resources, and long-term management of patients. 24 The nomogram we constructed and survival prognosis prediction was visualized as a numerical description, which showed good performance and high accuracy for CSS prediction in patients with SP-DLBCL in terms of calibration curve, IBS, C-index, and ROC curve. Additionally, the DCA curves showed that our nomogram exhibited good net clinical benefit and predicted CSS better than any other single predictor. It exhibited high clinical utility and was suitable for cancer prognosis research. Based on the nomogram, we calculated the total score of patients with SP-DLBCL and divided them by risk group to identify high-risk patients. It can help in clinical decision-making and follow-up management, thus improving the quality of life of patients. Furthermore, the web-based tool can greatly facilitate clinical applications. Such interactive tools facilitate shared decision-making by making complex statistical predictions more accessible and interpretable for both physicians and patients, thereby supporting clearer communication and informed collaborative choices.
The influence of primary site and chemotherapy on COD was statistically significant in the multivariable logistic regression analysis (P < 0.05). The incidence of CSD was higher in DLBCL originating in the spine than in the pelvis. This could be attributed to two main factors. Firstly, the spine has a unique anatomical location. It is more likely to give rise to central nervous system complications, which can significantly impact the prognosis. Secondly, there are treatment limitations. For example, achieving complete resection of spinal lesions is often challenging, and there are restrictions on the doses of radiotherapy and chemotherapy that can be administered, which may affect the effectiveness of treatment and ultimately contribute to the relatively higher mortality rate. As for chemotherapy, the incidence of CSD was lower in patients undergoing chemotherapy. For OCSS, age was considered a significant prognostic factor in competing risk regression. Heart disease, infectious or parasitic diseases, and pneumonia or influenza were found to be the three most common causes of OCOD; therefore, they should be appropriately addressed when predicting CSS. Furthermore, we found that patients undergoing chemotherapy were more likely to die due to heart disease and infectious or parasitic diseases. This might be attributed to the better clinical benefit and longer survival of patients with SP-DLBCL undergoing chemotherapy. Furthermore, the risk of cardiovascular disease or other diseases increased with age. Altogether, these findings suggested that the prevention of OCOD could improve the survival and prognostic outcomes of patients with SP-DLBCL.
The results revealed that OCOD accounted for 38.2% (117/306) of the total COD in patients with SP-DLBCL. We found that the cumulative incidence of CSD estimated by Kaplan–Meier analysis was higher than that by competing risk regression, accounting for competing risks throughout the follow-up period, and the gap widened with survival. Additionally, the cumulative incidence of OCOD gradually exceeded half that of CSD (Figure 2), indicating that OCOD should be appropriately considered when assessing survival in patients with SP-BLDCL. Nevertheless, in conventional Cox regression analyses, patients who died of OCOD are frequently not included and consider to not affect the outcome of interest. 25 Hence, Fine–Gray regression models that include competing risks are more suitable for survival analysis of patients with clinical SP-DLBCL. Additionally, CSS can be a better choice for evaluating clinical survival. To the best of our knowledge, previous studies on the prognosis of SP-DLBCL did not explicitly target CSS and OCOD, especially for DLBCL with a higher competing risk incidence.
Our study has some limitations, which are mainly due to the limitations of the SEER database. Other potential prognostic factors, such as International Prognostic Index, serologic examination (lactate dehydrogenase and immunoglobulin), genomic information,13,26-28 patient’s neurological status at presentation (eg, presence of myelopathy/radiculopathy, American Spinal Injury Association (ASIA) score), degree of epidural spinal cord compression (ESCC), and presence of mechanical instability (eg, as assessed by the Spinal Instability Neoplastic Score (SINS)), are not included in the SEER database. The details on treatment modalities such as the rationale for surgery (biopsy, decompression, stabilization), radiation doses and fields, and specific chemotherapy regimens (eg, R-CHOP) also can not be obtained from the extracted data. Hence, we were unable to evaluate the predictive value of these factors in CSS prediction. Future research should endeavor to incorporate these variables, which might potentially enhance the predictive performance of the model, as the c-index in this study (0.728/0.712 for training and testing datasets) indicated a moderate level of predictive accuracy, and a significant proportion of prognostic variability remained unaccounted for. Further, this study was based on data over a large period and improvements in treatment over time could potentially bias predictions even though the year of diagnosis was not found to be an independent factor. Lastly, subsequent multicenter validation is warranted to evaluate the accuracy and generalization of the model.
Conclusion
To conclude, we evaluated the risk factors of CSS in patients with SP-DLBCL using a competing risk regression model and constructed a nomogram prediction model. Our findings identified age as the most significant predictor influencing future CSS outcomes, followed by chemotherapy and Ann Arbor stage. The nomogram and web-based tool demonstrated robust predictive accuracy and clinical utility, indicating that these tools could be valuable for personalized risk assessment and treatment planning in patients with SP-DLBCL. To ensure the widespread application of this technology, we provide some perspectives on future research directions. First of all, expanding the application of the nomogram to more diverse patient populations and different healthcare settings will help to ensure its generalizability and accuracy. Multicenter studies with larger sample sizes are essential to confirm its predictive performance across various demographic and clinical profiles. Furthermore, incorporating the web-based tool into electronic health record (EHR) systems could facilitate real-time decision-making. This would enable healthcare providers to access risk predictions seamlessly during routine patient care. Lastly, as new treatment modalities and diagnostic tools emerge, the nomogram must be periodically updated to reflect the latest advancements. Continuous monitoring of outcomes and feedback from clinical use would be critical for maintaining its relevance and accuracy.
Supplemental Material
Supplemental Material - A Competing Risk-Based Online Calculator for Cancer-Specific Survival Prediction of Patients With Spinal and Pelvic Diffuse Large B-cell Lymphoma
Supplemental Material for A Competing Risk-Based Online Calculator for Cancer-Specific Survival Prediction of Patients With Spinal and Pelvic Diffuse Large B-cell Lymphoma by Jun Shang, MD, Zhixin Yuan, MD, Xiao Xiao, MD, Ge Jiang, MSc, Sheng Yang, MD, Chuanfeng Wang, MD, Guoxin Fan in Global Spine Journal.
Footnotes
Acknowledgements
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Ethical Approval
The SEER database is a publicly available database that obtains information from population-based cancer registries. Ethical approval was not needed due to the anonymization of patients.
Author Contributions
Conceptualization, SY, CW, and GF; Project Administration, JS; Supervision, JS; Funding Acquisition, GF; Resources, SY, CW, and GF; Data Curation, ZY; Methodology, SY, CW, and GF; Software, ZY and XX; Validation, GJ; Visualization, GJ; Formal Analysis, ZY; Investigation, XX; Writing – Original Draft Preparation, JS; Writing – Review & Editing, JS, ZY, XX, GJ, SY, CW, and GF.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was funded by the Nanshan District Health Science and Technology Major project (Grant No. NSZD2023023), and the Nanshan District Health Science and Technology Project (Grant No. NS2023044) to GF. The funders had no role in study design, data collection, data analysis, interpretation, writing of this report and in the decision to submit the paper for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting the conclusions of this study are accessible from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
