Abstract
Study Design
Retrospective cohort study.
Objectives
Successful bone fusion is crucial for favorable posterior lumbar interbody fusion (PLIF) outcomes. While trabecular bone remodeling (TBR) indicates osseointegration, its interpretation remains uncertain. This study aimed to clarify the clinical significance of distinct TBR types—fine TBR (fTBR) and sclerotic TBR (sTBR)—as seen on computed tomography (CT) scans and to evaluate their predictive value for segment stabilization and complete bone fusion and their relationship with interbody cage materials.
Methods
Seventy-one patients who underwent one-level PLIF with follow-up exceeding 5 years were included. Interbody cages were made of carbon fiber-reinforced polymer (CFRP; 37 segments) or titanium-coated polyether-ether-ketone (TiPEEK; 34 segments). CT images were obtained at 3 months, 1 year, and incidentally at 5 years or later. TBR was classified morphologically as fTBR or sTBR.
Results
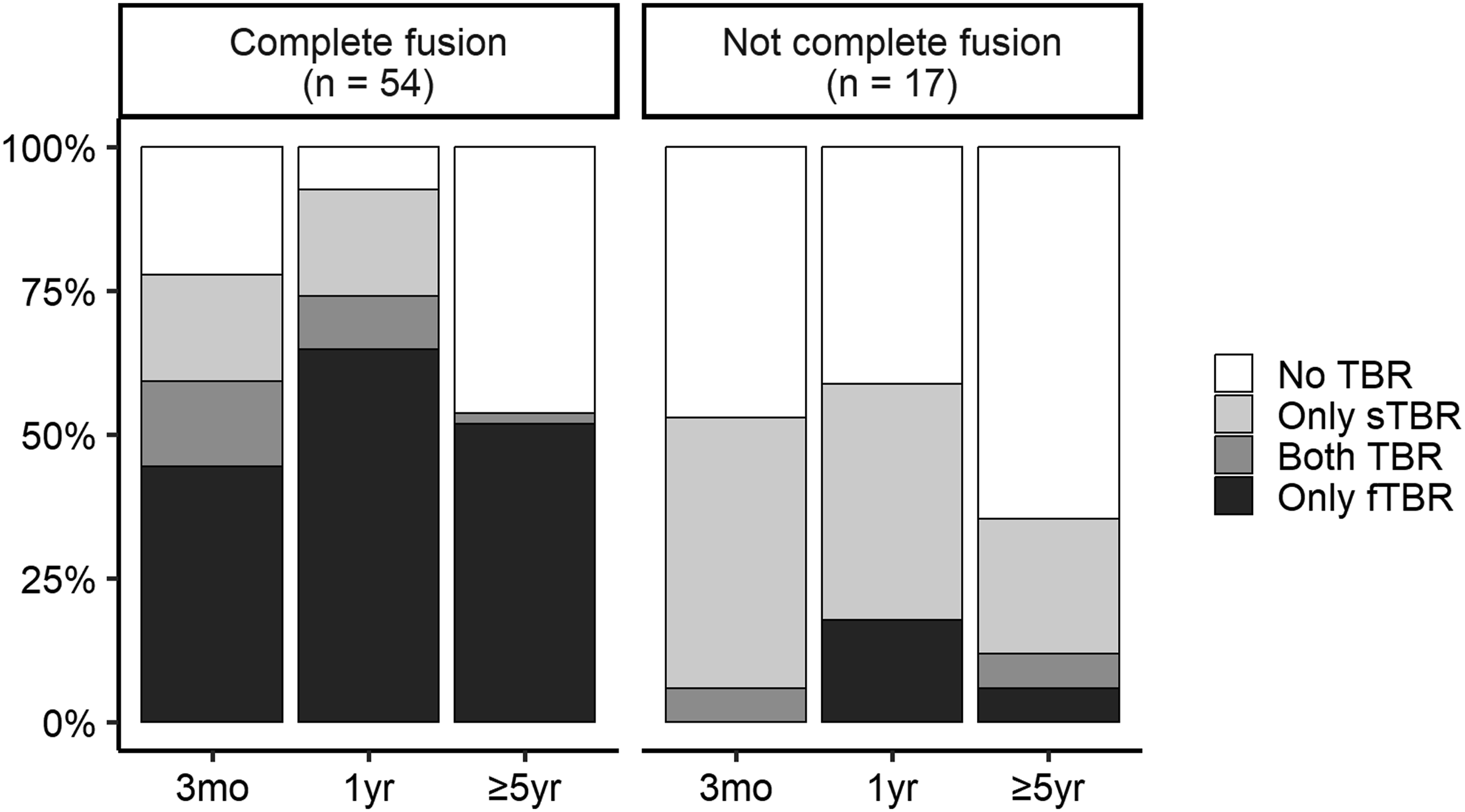
At 1-year postoperatively, the fTBR positivity rate was significantly higher in the TiPEEK group than in the CFRP group (48.6% vs 73.5%, P = .032). The 33 fTBR-positive segments at 3 months had a significantly higher complete fusion rate than 38 fTBR-negative segments (97.0% vs 57.9%, P < .001). There was no significant difference in fusion rates between 27 sTBR-positive segments at 3 months and 44 sTBR-negative segments (66.7% vs 81.8%, P = .15).
Conclusions
fTBR may serve as a positive indicator for predicting future segment stabilization and bone fusion post-PLIF. sTBR, however, is not associated with future segment stability. This refined TBR classification could enhance postoperative assessment and broaden our understanding of implant-bone integration in spinal fusion.
Keywords
Introduction
Posterior lumbar interbody fusion (PLIF) is a widely performed surgical procedure for various spinal pathologies, including degenerative disc disease, spondylolisthesis, and spinal stenosis, and is aimed at achieving spinal stability and alleviating pain via intervertebral bone fusion. Successful bone fusion, leading to solid segmental stability, is paramount for favorable long-term clinical outcomes. Pseudarthrosis (nonunion) can result in persistent pain, neurological deficits, and the potential need for revision surgery, significantly affecting the patient’s quality of life.1,2 However, the assessment of intervertebral bone fusion after PLIF remains a clinical challenge, particularly for accurately determining the status of bone fusion in the early postoperative period. Cage subsidence does not necessarily correlate with nonunion or poor clinical outcomes. 3 Vertebral endplate cyst (VEC) formation, which is characterized by cyst formation adjacent to the endplate, is a significant negative indicator of segmental stability.4,5 VECs can efficiently detect segments at a high risk for nonunion. However, positive indicators are of greater importance to both surgeons and patients. As yet, a reliable positive indicator of ongoing segment stabilization is lacking.
In recent years, “trabecular bone remodeling (TBR)” has emerged as a novel radiological finding in the vertebral body adjacent to the interbody cage after spinal fusion surgery. 6 Before TBR, bone growth on the cage surface was evaluated using visual information obtained through computed tomography (CT) value color mapping 7 or using values derived from calculations of CT values of the vertebral endplates contacting the cage, termed cancellous condensation. 8 While valuable, these findings required precise CT imaging and image processing. Furthermore, while CT values were the focus, the pattern and morphology of bone changes were not examined. In contrast, TBR is characterized by the formation of relatively thick, obliquely oriented trabeculae extending from the cage–endplate interface into the vertebral body, observable on CT images as early as 3 months postoperatively. Initial studies have suggested that the presence of TBR can be a promising indicator of osseointegration and segmental stability, correlating with a lower incidence of pedicle screw loosening and pseudarthrosis. 9
Despite its promise, the interpretation of TBR is complex. Paradoxically, some studies have reported instances in which TBR-positive segments subsequently exhibit instability, indicating that TBR may not always be a straightforward predictor of segmental stability. 10 This suggests that TBR, rather than being a singular phenomenon, may represent diverse types of bone responses that reflect different states of bone reconstruction and implant–bone integration. Therefore, a more nuanced understanding of the morphological variations of TBR and their distinct clinical implications is crucial. Furthermore, the material composition of interbody cages plays an important role in bone integration. In addition to excellent radiolucency and biomechanical properties, which are exemplified by carbon fiber reinforced polymer (CFRP), titanium coatings that promote cell adhesion, proliferation, and osteoconduction, thereby enhancing stability at the bone-implant interface, are clinically applied. The distinct osseointegration characteristics of these materials may influence TBR patterns and timing, thereby affecting its predictive value for long-term fusion outcomes.
Given these unanswered questions, this study aimed to thoroughly investigate the diverse forms of TBR following PLIF. We hypothesized that TBR is not a monolithic finding but comprises distinct morphological types, “fine TBR (fTBR)” and “sclerotic TBR (sTBR),” each with different predictive values for segment stabilization and bone fusion. By analyzing these two distinct types of bone restructuring reactions in a cohort of patients undergoing PLIF with either CFRP or titanium-coated polyether-ether-ketone (TiPEEK) cages and evaluating their relationship with other radiological findings and long-term fusion outcomes, we aimed to (1) establish a refined classification of TBR based on its morphological appearance, (2) elucidate the clinical significance of fTBR and sTBR in predicting segment stability and complete bone fusion, and (3) provide a more comprehensive understanding of bone reconstruction and implant–bone integration after PLIF, potentially redefining the radiological criteria for successful fusion.
Materials and Methods
Study Design and Patient Population
This study was conducted in accordance with the guidelines of the Declaration of Helsinki and was approved by the institutional review boards of the authors’ affiliated institutions. This single-center retrospective study was based on previously acquired imaging studies and medical records. Therefore, all interventions, including surgical techniques and pre- and postoperative examination items, were routine procedures, and the requirement for written informed consent was waived. We retrospectively reviewed the data of patients who underwent one-level PLIF at a single institution. Patients who had previously undergone lumbar surgery were excluded. As the cages used for PLIF varied over time at the institution, the study period was set to 2010 and 2016, when a single cage type was used each year to ensure uniformity of the samples. In 2010, CFRP cages made of PEEK-OPTIMA (Invibio, UK) were used (CFRP group, 37 patients, 37 segments; CALIBER CFR PEEK, Robert Reid Inc., Tokyo, Japan). In 2016, interbody cages composed of PEEK with titanium-coated surfaces were used (TiPEEK group: 34 patients, 34 segments; PROSPACE XP, Aesculap AG, Tuttlingen, Germany).
Thus, 126 consecutive patients were included in the initial patient cohort. Of these, 103 patients had a follow-up period of ≥5 years. Owing to regional characteristics, patients tended to remain in the same area and often visited the same institution for health issues other than spinal disorders. This allowed for the evaluation of CT images taken ≥5 years post-surgery as the imaging-based final outcome. Among the 103 patients, 28 were excluded because CT images were not obtained ≥5 years postoperatively. Additionally, four patients who underwent additional surgery owing to adjacent segment disease were excluded. There were no other reasons for reoperation. The final sample comprised 71 patients (Figure 1). Patient Selection Flowchart. PLIF, Posterior Lumbar Interbody Fusion; CFRP, Carbon Fiber-Reinforced Polymer; TiPEEK, Titanium-Coated Poly-Ether-Ether-Ketone; CT, Computed Tomography
Surgical Procedure
PLIF was performed using standard techniques. Bilateral pedicle screw-rod systems were used in all cases, with two interbody cages placed in each segment, one on each side, resulting in two cages per level. Facet joints were resected as required. Pedicle screws were inserted, and intervertebral expansion was performed. After temporary fixation, the intervertebral disc was carefully removed to avoid damaging the vertebral endplates. A local bone graft (morselized local bone autograft) was transplanted into the interbody before the cage insertion. The same type of cage was used, and the cavity of the cages was filled with the same graft bone. After inserting the cages from both sides, compression was applied between the pedicle screws, and final fixation was achieved. All patients wore hard braces for 3 months postoperatively.
Variables
Patients’ age, sex, comorbidities, and smoking status were assessed. CT values (Hounsfield units) were measured on axial CT images of the cephalic vertebral body of the PLIF segment,11,12 and the average value was recorded. The Japanese Orthopaedic Association (JOA) score 13 (range, 0-29 points) was investigated. The JOA scoring system consisted of three subjective symptoms scored from 0 to 9; three clinical symptoms, such as neurological deficit, scored from 0 to 6; seven activities of daily living scored from 0 to 14; and bladder function scored from −6 to 0. Additionally, preoperative and 1-year postoperative JOA Back Pain Evaluation Questionnaire (JOABPEQ) scores, 14 a patient-reported outcome measure, was investigated. The JOABPEQ aggregates 25 questionnaires results into five domains: low back pain, lumbar function, walking ability, social life function, and mental health. Each domain is scored out of 100 points, with higher scores indicating better outcomes.
Radiological evaluations were performed preoperatively, postoperatively, and 3 months, 1 year, and 5 years postoperatively. Local lordotic angles (measured from the rostral endplate of the upper vertebra to the caudal endplate of the lower vertebra, with positive values indicating lordosis) were measured on the standing lateral images. The difference in the local lordotic angle between the postoperative period and 5 years postoperatively was defined as correction loss. Local range of motion (ROM) was calculated from standing lateral forward- and backward-bending radiographs.
CT images were obtained postoperatively and at 3 months and 1 year postoperatively. Additionally, if a CT scan including the PLIF segment was performed for any reason ≥5 years postoperatively, this was defined as the final CT image. These CT scans were acquired for adjacent segment disease, trauma, or abdominal diseases. The final CT image was obtained an average of 7.0 ± 2.6 years postoperatively. CT images were obtained using standard methods, and multiplanar reconstruction (MPR) images were obtained. Imaging findings were independently assessed by two authors (Y.N. and N.S.) in a blinded manner, and any discrepancies were resolved by establishing consensus between the two authors. CT image findings were recorded on a segmented basis.
TBR was identified on coronal or sagittal CT-MPR images by comparison with the immediate postoperative images. TBR was relatively thick, with tilted trabeculae inside the vertebra that were not depicted on the immediate postoperative images, according to previous reports.6,9,15 These trabeculae were present in the cage–vertebral endplate contact area and extended into the vertebral body. TBR was classified into two types. fTBR is defined as a high density area (HDA) that appears as lines or threads extending from the cage to the vertebral body (Figure 2). Generally, the fTBR was thinner than the small contact area between the cage frame and endplate. In addition, although spot-like hardening is often observed at points where the cage material contacts the endplate,
9
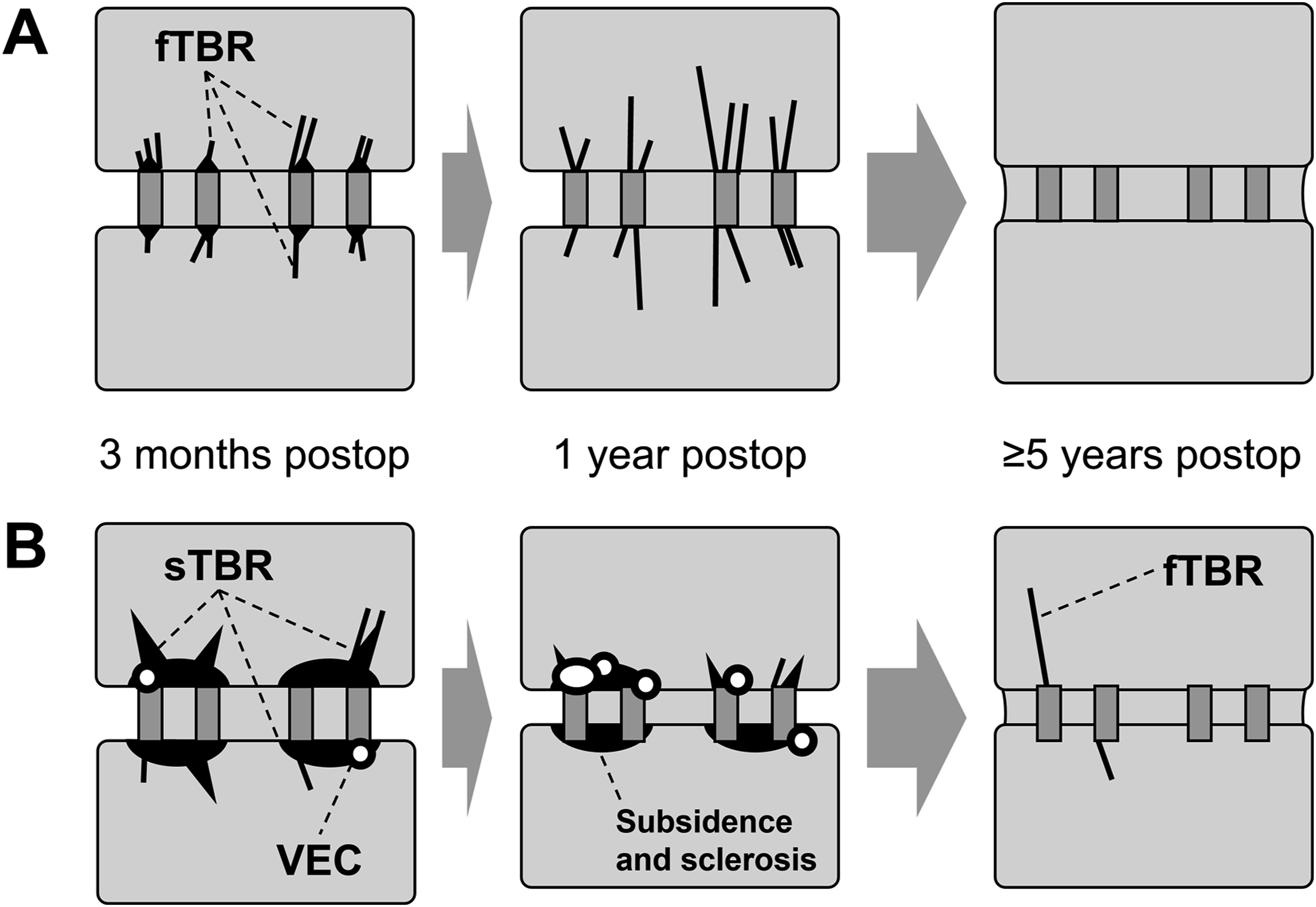
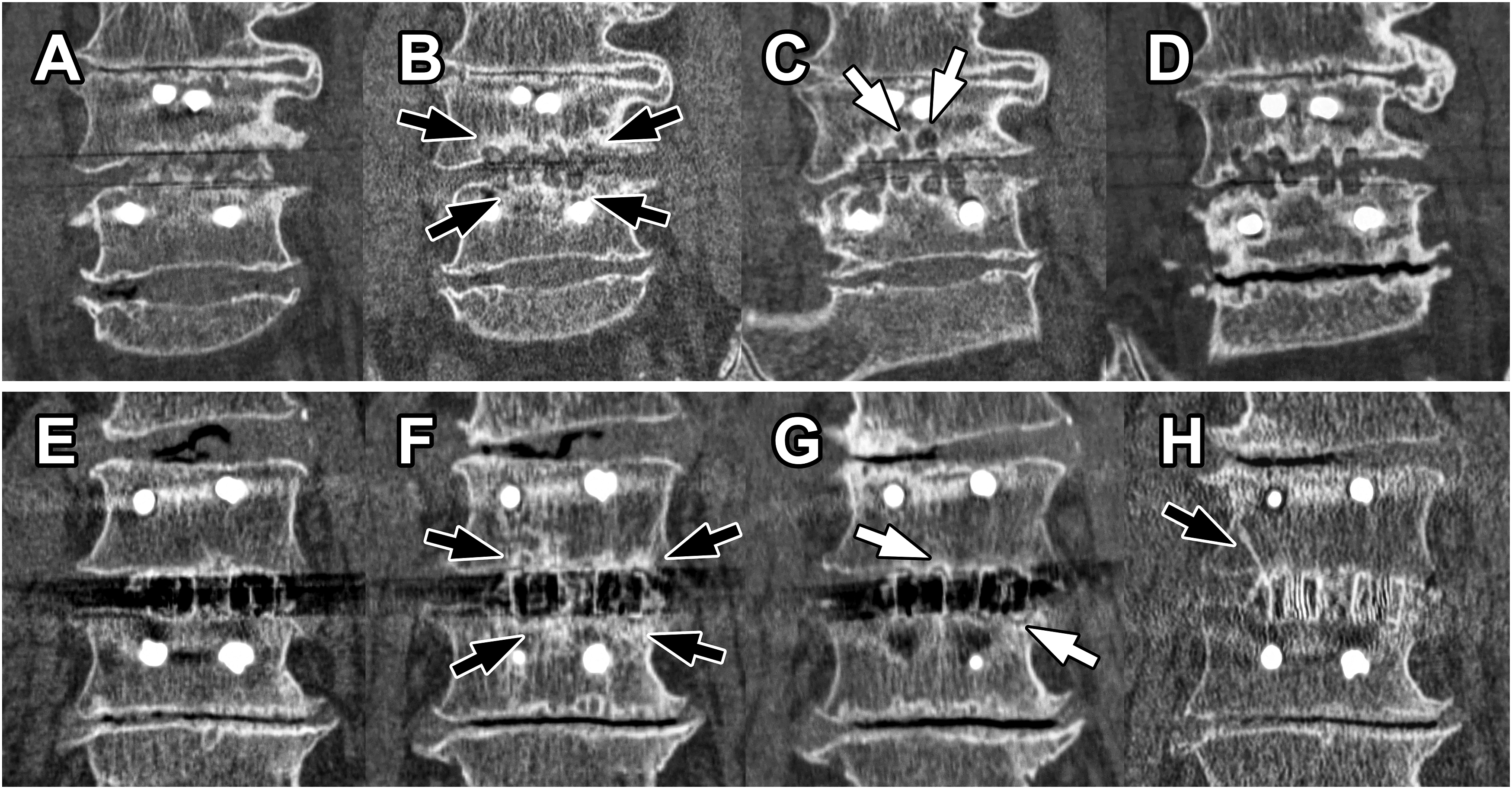
this small hardening was not included in the determination of whether the structure was thread-like. sTBR was an HDA that expands from the cage into the vertebral body or may have a slightly irregular shape accompanied by sclerosis of the endplate over an area equal to or wider than the cage width (Figure 4). The small hardening observed at the cage-endplate contact site in fTBR was equal to or smaller than the cage frame footprint, whereas the sclerosis at the cage-endplate contact site in sTBR extended beyond the cage frame footprint (see also Figure 3). sTBR may also include thread-like portions. Consequently, if both fTBR and sTBR were present, the finding was classified as sTBR. However, because two cages were always inserted into a single segment, if one cage was positive for fTBR and the other for sTBR, the segment was classified as “both positive” for fTBR and sTBR. Fine TBR. (A-D) CT Image Changes in CFRP Cage Segments With fTBR. (E-H) CT Image Changes in TiPEEK Cage Segments With fTBR. (A) Postoperative Coronal Imaging B, 3 Months Postoperatively, Fine TBR (Black Arrows) was Observed. (C) 1 Year Postoperatively, Fine TBR Developed (Black Arrows). (D) 10 Years Postoperatively, the Segment is Completely Fused (White Arrowheads) and TBR Disappears. (E) Postoperative Coronal Image. (F) 3 Months Postoperatively, Fine TBR (Black Arrows) was Observed. (G) 1 Year Postoperatively, Fine TBR Developed (Black Arrows). (H) 5 Years Postoperatively, the Segment was Completely Fused (White Arrowheads) and TBR Disappeared. See Figure 3A. CT, Computed Tomography; CFRP, Carbon Fiber-Reinforced Polymer; TiPEEK, Titanium-Coated Poly-Ether-Ether-Ketone; TBR, Trabecular Bone Remodeling/Restructure; fTBR, Fine TBR Schemas for fTBR and sTBR. (A) Schematic Diagrams Corresponding to Figure 2B-D or F-H. Three Months Postoperatively, the fTBR, a Linear or Thread-Like TBR Extending From the Cage to the Vertebral Body, is Observed. Small Endplate Sclerosis Limited to the Area in Contact With the Cage Frame May be Present (This can be Clearly Distinguished From Sclerosis Associated With sTBR). One Year Postoperatively, the Development of fTBR is Observed. In Segments that Achieved Complete Fusion for >5 Years Postoperatively, the Cage is Embedded Within the Vertebral Bone Tissue, and TBR is No Longer Observable. (B) Schematic Diagram Corresponding to Figure 4F-H. Three Months Postoperatively, Widespread Endplate Sclerosis was Observed at the Cage–Endplate Contact Site, Often Accompanied by VECs. Some High-Density Areas Extending Into the Vertebral Body Were Observed; However, They Differed From the Linear or Thread-like Structure of fTBR, Being Broader or Irregular in Shape, and Were Therefore Classified as sTBR. One Year Postoperatively, Cage Subsidence and Associated Sclerosis Were Observed Along With VECs. Fortunately, This Segment Achieved Fusion. Consequently, in Segments that Have Completely Fused for >5 Years Postoperatively, the Cage is Embedded Within the Vertebral Bone Tissue, With Some Residual fTBR Believed to Have Formed During the Fusion Process Over the Years. TBR, Trabecular Bone Remodeling/Restructure; fTBR, Fine TBR; sTBR, Sclerotic TBR; VEC, Vertebral Endplate Cyst Sclerotic TBR. (A-D) CT Image Changes in CFRP Cage Segments With sTBR. (E-H) CT Image Changes in the TiPEEK Cage Segments With sTBR. (A) Postoperative Coronal Imaging B, 3 Months Postoperatively, Cage Subsidence and Sclerotic TBR (Black Arrows) are Observed. (C) 1 Year Postoperatively, Progressive Cage Subsidence, VEC Formation (White Arrows), and Disappearance of TBR are Observed. (D) 7 Years Postoperatively, the Segment is in Nonunion With the Intervertebral Vacuum. (E) Postoperative Coronal Image. (F) 3 Months Postoperatively, Sclerotic TBR (Black Arrows) is Observed. VEC Formation (White Arrows) and Disappearance of TBR are Observed. (H) 8 Years Postoperatively, Cage Subsidence is Observed; However, No Signs of Nonunion are Observed in the Segment. (A Fine TBR (Black Arrow) is Observed. See Figure 3B. CT, Computed Tomography; CFRP, Carbon Fiber-Reinforced Polymer; TiPEEK, Titanium-Coated Poly-Ether-Ether-Ketone; TBR, Trabecular Bone Remodeling/Restructure; sTBR, Sclerotic TBR; VEC, Vertebral Endplate Cyst Percentage of Each TBR With and Without Complete Fusion of Segments. The Raw Values are Shown in Supplemental Table 2. TBR, Trabecular Bone Remodeling/Restructure; fTBR, Fine TBR; sTBR, Sclerotic TBR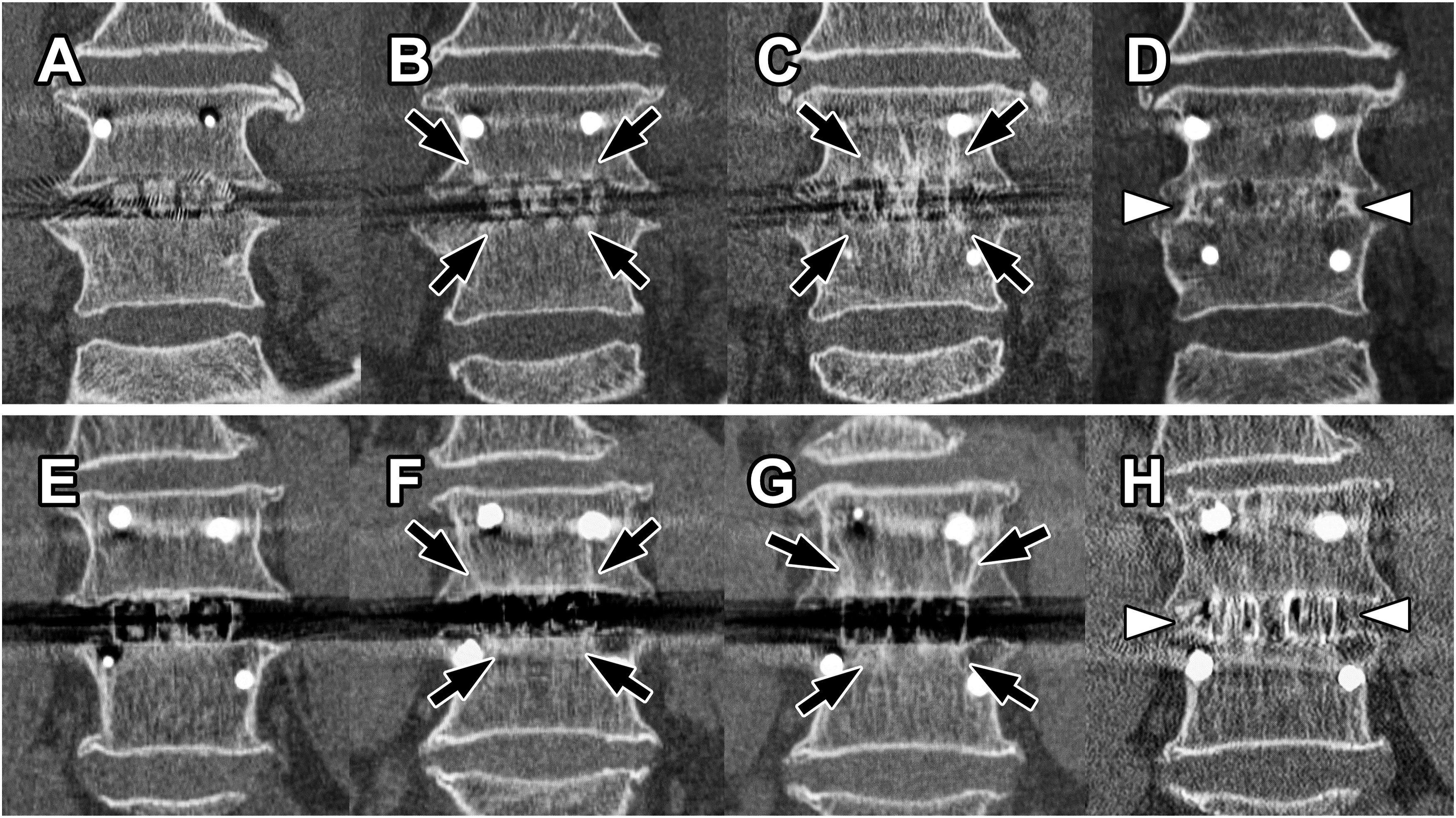
VEC4,5 that was not present on previous imaging or enlarged VEC was defined as VEC-positive. Cage subsidence was defined as cage migration of >2 mm into the vertebral endplate in any part of the cage compared with previous imaging studies. No cage dropouts from the intervertebral space were observed. If air was found in the PLIF segment interbody on sagittal or coronal CT images, it was considered positive for the vacuum sign. Instability was defined as a local ROM >5°. 16
Complete fusion was defined as the complete bony integration of the PLIF segment on the last CT image (Figure 2D and H). Nonunion was defined as any of the following findings: positive vacuum sign or positive instability. None of the pseudarthroses required revision surgery.
Statistical Analyses
Data are presented as means ± standard deviations for continuous variables and as numbers and percentages for categorical data. Statistical analyses were performed using R version 4.4.0 (https://www.r-project.org/, 2024-04-24) with packages tidyverse, 17 irr, 18 and gtsummary. 19 Interobserver reliability was assessed using Cohen’s kappa coefficient. Comparisons between groups were performed using the Wilcoxon rank-sum test, Fisher’s exact test, and Pearson’s chi-squared test, and P < .05 was considered statistically significant.
Results
Patients’ Demographics and Radiological Characteristics Based on Cage
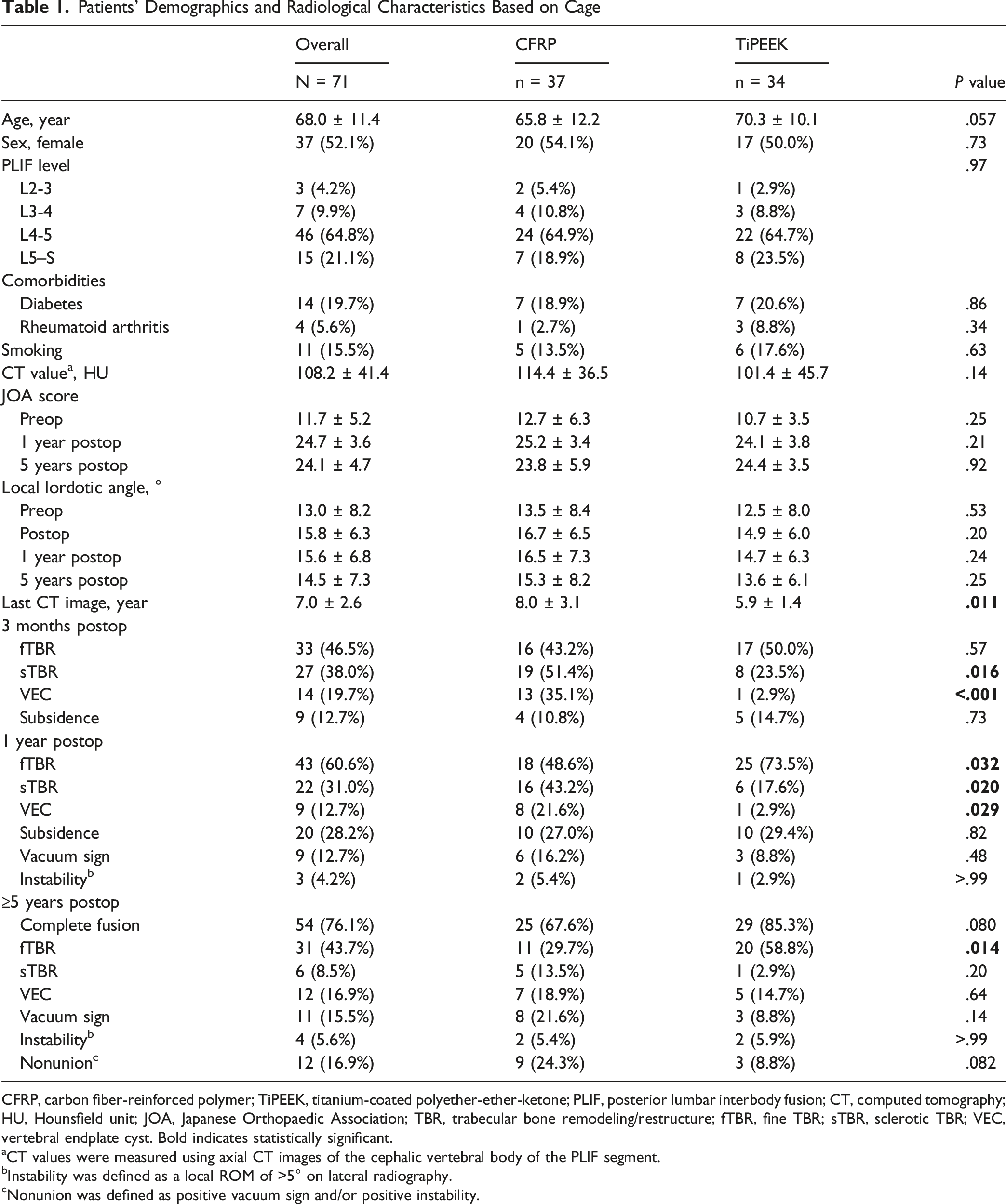
CFRP, carbon fiber-reinforced polymer; TiPEEK, titanium-coated polyether-ether-ketone; PLIF, posterior lumbar interbody fusion; CT, computed tomography; HU, Hounsfield unit; JOA, Japanese Orthopaedic Association; TBR, trabecular bone remodeling/restructure; fTBR, fine TBR; sTBR, sclerotic TBR; VEC, vertebral endplate cyst. Bold indicates statistically significant.
aCT values were measured using axial CT images of the cephalic vertebral body of the PLIF segment.
bInstability was defined as a local ROM of >5° on lateral radiography.
cNonunion was defined as positive vacuum sign and/or positive instability.
Comparison of Segments With and Without 3-Month Fine TBR
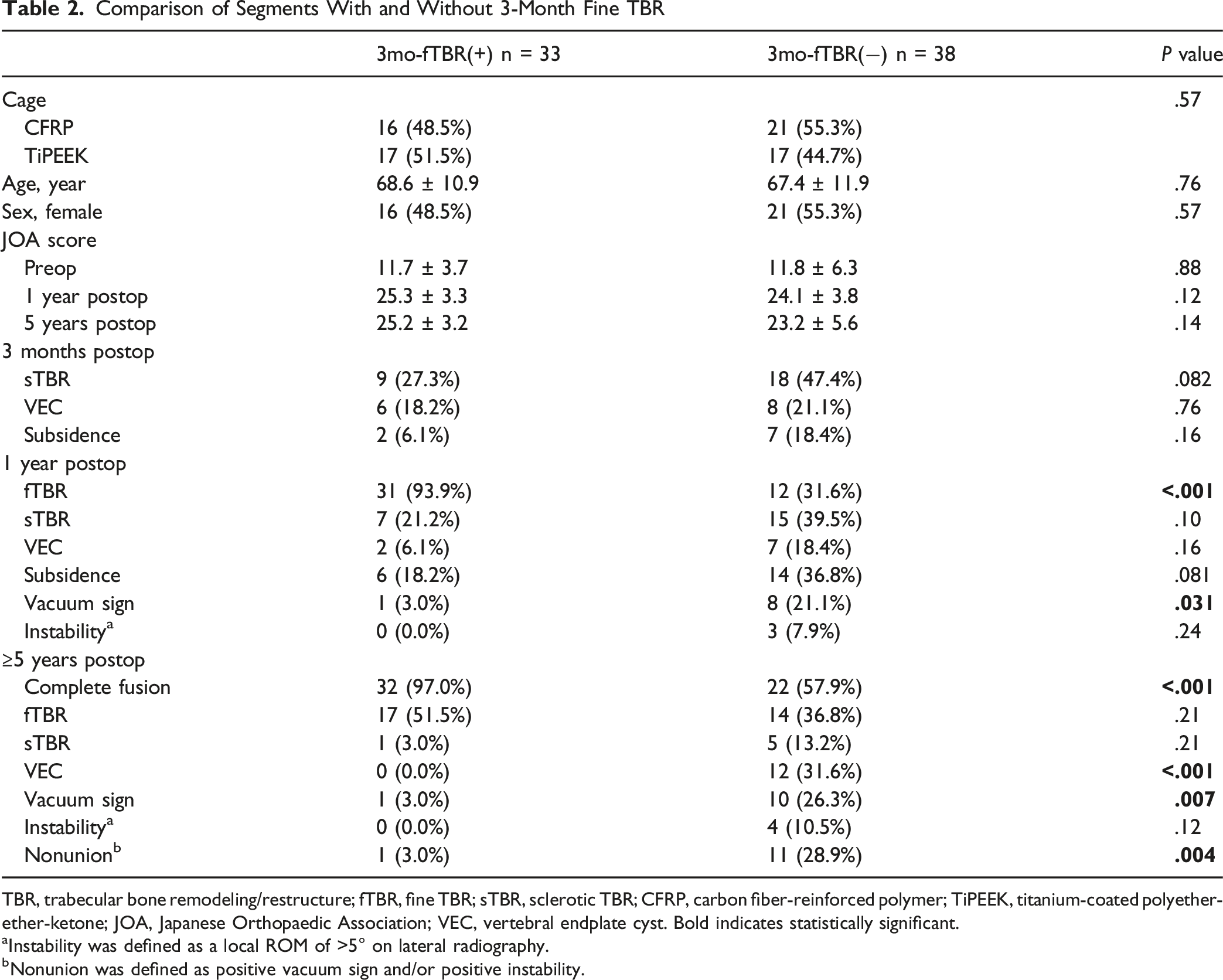
TBR, trabecular bone remodeling/restructure; fTBR, fine TBR; sTBR, sclerotic TBR; CFRP, carbon fiber-reinforced polymer; TiPEEK, titanium-coated polyether-ether-ketone; JOA, Japanese Orthopaedic Association; VEC, vertebral endplate cyst. Bold indicates statistically significant.
aInstability was defined as a local ROM of >5° on lateral radiography.
bNonunion was defined as positive vacuum sign and/or positive instability.
Comparison of Segments With and Without 3-Month Sclerotic TBR
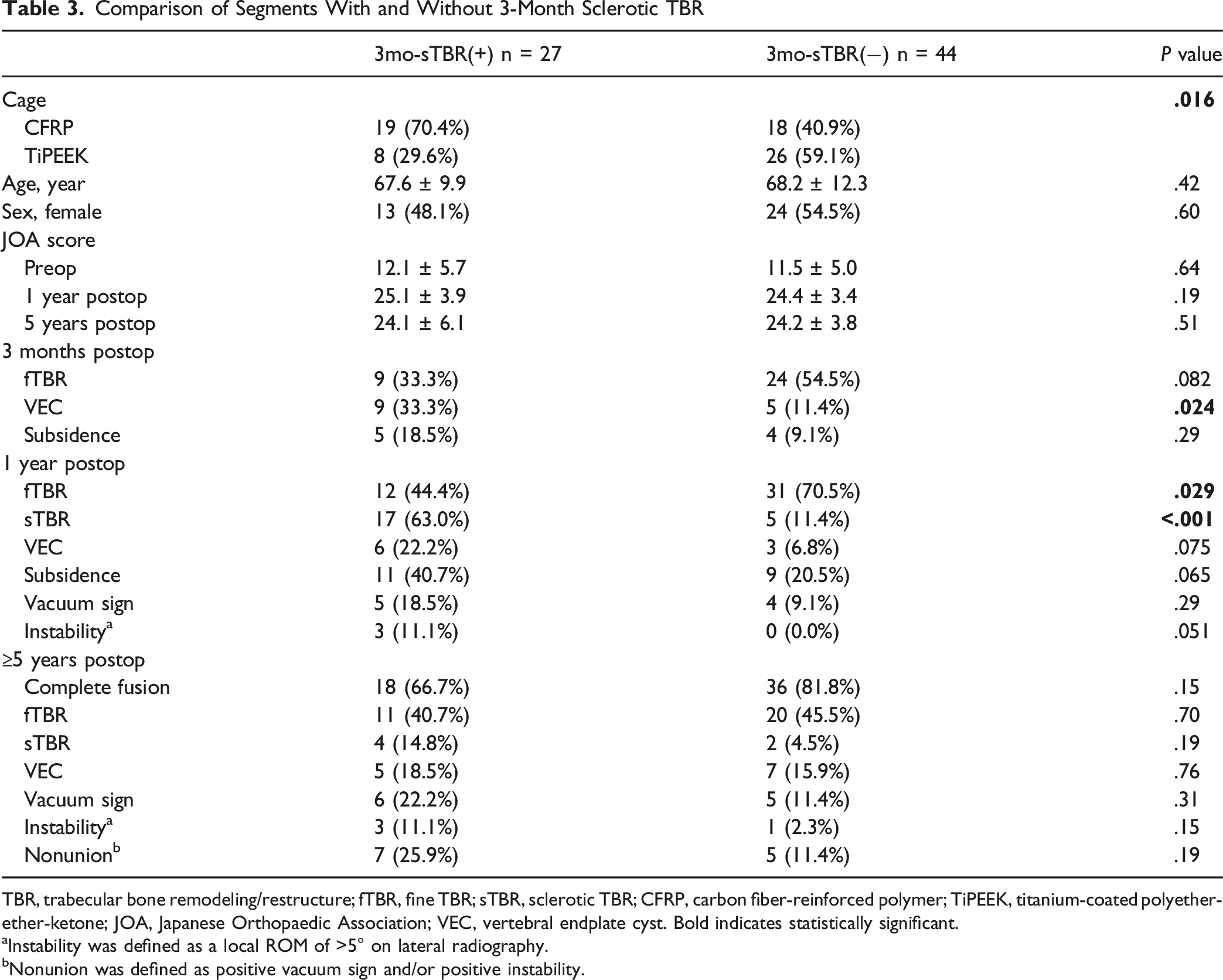
TBR, trabecular bone remodeling/restructure; fTBR, fine TBR; sTBR, sclerotic TBR; CFRP, carbon fiber-reinforced polymer; TiPEEK, titanium-coated polyether-ether-ketone; JOA, Japanese Orthopaedic Association; VEC, vertebral endplate cyst. Bold indicates statistically significant.
aInstability was defined as a local ROM of >5° on lateral radiography.
bNonunion was defined as positive vacuum sign and/or positive instability.
Although patients with fTBR-positive segments at 1 year postoperatively had relatively worse preoperative JOABPEQ low-back pain domain scores than patients without fTBR segments (32.1 ± 30.3 vs 49.4 ± 35.0, P = .060), no significant difference was observed at 1 year postoperatively (66.0 ± 34.1 vs 67.3 ± 31.7, P = .98) (Supplemental Table 2). Regardless of the presence or absence of fTBR or sTBR at 1 year, all JOABPEQ domains showed improvement at 1 year postoperatively (Supplemental Tables 2 and 3).
Image Findings Associated With Complete Fusion at ≥5 Years Postoperatively
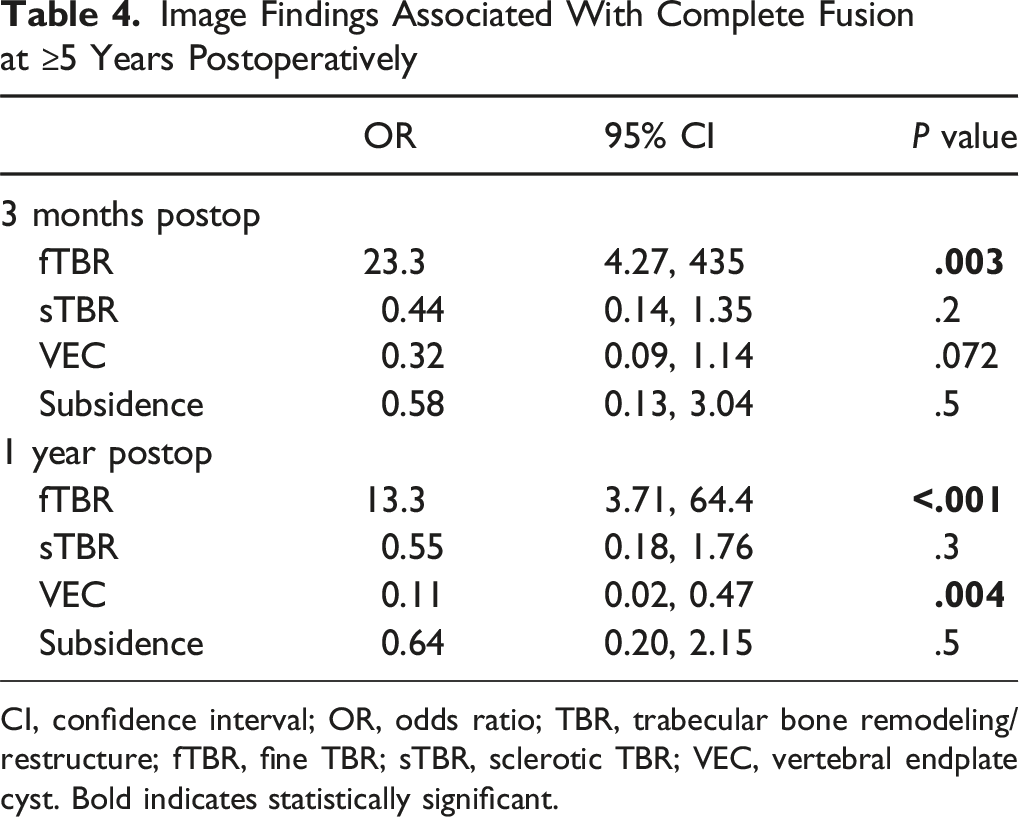
CI, confidence interval; OR, odds ratio; TBR, trabecular bone remodeling/restructure; fTBR, fine TBR; sTBR, sclerotic TBR; VEC, vertebral endplate cyst. Bold indicates statistically significant.
Discussion
In the present study, we introduced a novel classification for TBR observed after PLIF: fTBR and sTBR. Our findings demonstrate that these distinct morphological patterns of TBR have differential predictive values for long-term segment stabilization and complete bone fusion. Specifically, we found that fTBR was a reliable positive indicator of future segment stability and successful intervertebral fusion, which is characterized by significantly lower rates of VEC, vacuum phenomena, and nonunion. Conversely, sTBR was not associated with future segment stability. These results address a critical unmet need in spinal fusion assessment: the identification of a positive radiological sign for segment stabilization, complementing existing markers of instability, such as VEC. Although previous studies have highlighted the potential of TBR as a marker of osseointegration, its interpretation remains unclear. Our refined TBR classification resolved this ambiguity, suggesting that fTBR represents effective load transmission and bone integration, whereas sTBR reflects an inadequate or reactive bone response to mechanical stress. Furthermore, our comparative analysis of CFRP and TiPEEK cages revealed that TiPEEK cages induced fTBR more frequently and sTBR less frequently than CFRP cages, underscoring the influence of implant characteristics on specific TBR patterns and their prognostic implications.
As mentioned in the Methods section, fTBR and sTBR are not always clearly distinguishable, and there have been cases in which both fTBR and sTBR were considered to coexist in a single cage. Here, as TBR and VEC, which are strong indicators of osseointegration and powerful predictors of instability, can coexist, 9 we believe that fTBR and sTBR can also coexist (note that in this analysis, when fTBR and sTBR coexist, we classified them as sTBR). After surgery, changes in load transmission may lead to the formation of sTBR, which may change to fTBR. Alternatively, the fate of the segment may be largely determined relatively early after surgery, leading to the formation of TBR appropriate to the local conditions, fTBR or sTBR. Thus, fTBR, sTBR, and VEC are predictive factors that may appear in this order as positive osseointegration indicators and may form a gradient. In other words, fTBR is the strongest positive indicator of osseointegration, whereas VEC is the strongest negative indicator.
The histological differences between fTBR and sTBR are of interest; however, as there are currently no methods available to observe them, we attempt an indirect interpretation. Following the hypothesis that fTBR corresponds to the established load transfer, the mechanical properties of the environment where fTBR exists are presumed to be uniform. Therefore, fTBR needs to only respond to stresses in specific directions 20 and may exhibit an appearance similar to background trabeculae. In contrast, assuming that load transmission in sTBR segments is not yet fully established, mechanical properties of the environment wherein sTBR exists are expected to be changing. Therefore, sTBR is predicted to exhibit varied mechanical properties and a relatively thick, irregular structure. Some may argue that sTBR is simply the densification of the underlying bone due to cage subsidence. 21 While sTBR often accompanies cage subsidence, we argue that the overall size of the sTBR is significantly large to be considered a hardening pattern resulting from the compression of the vertebral cancellous bone due to cage subsidence. Therefore, it is likely more reasonable to consider sTBR an active response of the bone rather than a phenomenon caused by cage subsidence. To address these issues, histological approaches using animal models and micro-CT analysis are considered effective and represent future research topics.
Other biological mechanisms may also contribute to different types of TBR. Animal models have demonstrated that the mechanical behavior of each microstructure within the vertebral endplate differs. 22 The cartilaginous endplate behaves to distribute compressive loads uniformly to the vertebral body. 23 In PLIF, the removal of the cartilaginous endplate eliminates the original stress-distribution structure, resulting in direct interaction between the bone tissue and the cage. Furthermore, while vascularization is typically absent in the adult intervertebral disc region, a capillary network emerges owing to degeneration. 24 Thus, increased vascular network may influence cage-endplate interactions.
As predicted in the analysis of TBR in LLIF, 15 TBR disappeared once segment stabilization was complete and the vertebrae of the fused segment became integrated. This likely indicates that the load transmission “concentrated on the cage” disappears with integration. To achieve complete fusion, the PLIF cage alone should probably be insufficient, and grafted bone outside the cage is necessary to bridge the vertebrae. Consequently, the cage becomes completely embedded within a single bone, allowing loads to be distributed throughout the structure rather than concentrated through the cage, thereby eliminating TBR. In contrast, the stability of segments in which TBR persists over time warrants further investigation. If the failure to achieve clear bone continuity on imaging is solely due to the relative insufficiency of the grafted bone, the segment is likely to remain solid in the future. However, if the TBR persists because of an ongoing interplay between the stabilizing and destabilizing forces, the segment may become unstable under altered conditions (eg, removal of the pedicle screw-rod system due to complications). This issue requires further verification; however, based on the results of this study, fTBR alone is considered to suggest clinical stability even after a prolonged period. In other words, the “bone fusion” of segments can potentially be redefined as a state where “TBR is not detected or only fTBR is detected,” rather than merely recognizing clear bone continuity on imaging.
The influence of the cage surface bone-integration ability on these TBR findings was also clarified. Compared with CFRP, 25 which lacks bioactive properties, TiPEEK,26,27 which enhances bone integration ability by adding titanium to the PEEK surface, demonstrated superiority, a lower sTBR positivity rate at 3 months postoperatively and a higher fTBR positivity rate at 1 year postoperatively. The lower frequency of VEC formation on TiPEEK further supports this point, suggesting that TiPEEK cages promote more favorable bone response than CFRP cages. At the final follow-up, the TiPEEK group had a higher complete fusion rate, although the difference did not reach statistical significance, likely owing to the sample size. The newly introduced parameters fTBR and sTBR allowed us to evaluate the effectiveness and limitations of the TiPEEK osteoconductive surface in promoting segmental stabilization compared to the inert CFRP surface.
Additionally, without waiting for the lengthy confirmation of radiographic complete fusion, the TBR metric allows for a semi-quantitative assessment of osseointegration capabilities of TiPEEK and CFRP in terms of early stability (low sTBR positivity rate) and long-term stability (high fTBR positivity rate). From this perspective, the improvement in bone integration capability owing to surface titanium is limited. 28 For example, surface titanium may reduce sTBR through early bone–tissue bonding 26 ; however, it does not increase fTBR, and the feasibility of segment stabilization appears to be governed by larger mechanical laws rather than the microenvironment of the cage surface. Thus, TBR does not appear to be governed by the bone integration ability of the cage surface, but rather by other mechanical laws. The two types of cages differed not only in their surface bone integration ability but also in various parameters. For example, the shape and elastic modulus (the elastic modulus of the CFRP cage made from PEEK-OPTIMA is approximately 10 GPa, 29 which is higher than that of pure PEEK [approximately 3-4 GPa] 30 ). Because cages were not randomly assigned and a confounding factor (implant era) existed, the influence of these parameters on TBR or osseointegration remains unclear. Our position is that TBR findings can be universally interpreted even if the bone integration ability of the cage differs.
One of the key findings of this study is that TBR is not necessarily a result of bone remodeling. fTBR may reflect the actual remodeling that occurs as a result of the vertebral bone responding to stress transmission through cage–vertebral bone osseointegration. However, sTBR does not necessarily include such findings and may only represent the response of the vertebral bone in relatively unstable segments. Furthermore, even fTBR may be bone “modeling” rather than remodeling.31,32 The original study that used the term “TBR” likely focused on the result of trabecular bone structure being restructured and applied the term “remodeling.” However, based on the current results, we propose that “trabecular bone restructure” is more suitable than trabecular bone remodeling for TBR.
Although we were able to obtain a large number of mid-to long-term CT images, we do not recommend repeatedly exposing patients to CT for the evaluation of segments, including TBR. Such evaluations are unwise—they result in unnecessary radiation exposure for patients and unnecessary cost increases—and should be avoided, although CT radiation exposure has been reduced at the hardware level.33,34 Our intention was to show that it is possible to infer the status of fixed segments from a mechanical perspective using snapshot CT images. Unless further clinical questions arise, such a study design will be unnecessary.
While various intriguing associations between TBR and radiographic outcomes have been demonstrated, associations between TBR and patient symptoms remain unclear. In this study, owing to limitations of the retrospective study design, JOABPEQ data collection was relatively incomplete, resulting in insufficient statistical power. An analysis based on the presence or absence of fTBR at 1 year postoperatively revealed that the low-back pain domain in the fTBR-positive group, which was relatively poor preoperatively, improved to a comparable level. This finding is noteworthy but should not be emphasized at this stage. We consider the analysis of TBR and patient-reported symptoms to be a future research task.
Although we were able to obtain many interesting findings by carefully observing the CT images after PLIF using the two types of cages, this study has some limitations. First, in studies that evaluate imaging findings, intra- and interobserver errors are unavoidable. Second, this was a retrospective study. Establishing causality is impossible; as discussed, fTBR should be understood as a predictor rather than a factor that causes fusion. Additionally, cage-type assignment was not randomized but based on the year. Furthermore, this was a single-center study, and the relatively small sample size and exclusion of multi-level fusion may limit generalizability. Third, a temporal bias may exist owing to the presence of two distinct surgical periods. However, there was no change in the ratio of spine surgery specialists to fellows between 2010 and 2016, and there were no clear differences in the duration of hard brace wear, rehabilitation, discharge criteria, or post-discharge management. Additionally, no replacement of the CT scanner occurred. Fourth, the 5-year follow-up rate was limited to approximately 82%, resulting in the exclusion of 28 cases. Therefore, selection bias may have occurred. Despite these limitations, we identified new findings worthy of being named fTBR and sTBR, further advancing our understanding of the TBR phenomenon in vertebral cancellous bone following interbody fusion and proposing a more clinically relevant interpretation of TBR.
Conclusion
This study introduces a novel classification of TBR into fTBR and sTBR, each with distinct clinical implications for PLIF outcomes. Early fTBR may predict long-term segment stabilization and complete bone fusion, potentially serving as a positive indicator previously lacking in radiological assessments. In contrast, sTBR is not associated with future segment stability. Furthermore, the type of interbody cage material influences the TBR patterns, with the TiPEEK cages promoting more fTBR than the CFRP cages. Our findings suggest that successful PLIF fusion should be re-evaluated to include fTBR as a key sign of effective osseointegration, refining current radiological interpretations and aiding in better postoperative management and implant design. In the future, incorporating routine early postoperative CT or advanced imaging could help identify fTBR, thereby guiding postoperative management. However, caution is advised in interpreting TBR types; thus, further research should explore underlying biological mechanisms and correlation with clinical outcomes.
Supplemental Material
Supplemental Material - Fine versus Sclerotic Trabecular Bone Remodeling/Restructure: A Refined Radiographic Predictor for Long-Term Segment Stability after Posterior Lumbar Interbody Fusion
Supplemental Material for Fine versus Sclerotic Trabecular Bone Remodeling/Restructure: A Refined Radiographic Predictor for Long-Term Segment Stability after Posterior Lumbar Interbody Fusion by Yasuhiro Nagatani, Naoki Segi, Sadayuki Ito, Jun Ouchida, Ippei Yamauchi, Yukihito Ode, Yuya Okada, Yujiro Kagami, Mikito Tsushima, Hiroyuki Tomita, Kazuaki Morishita, Hiroki Oyama, Tokumi Kanemura, Shiro Imagama, and Hiroaki Nakashima in Global Spine Journal
Footnotes
Ethical Considerations
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the institutional review board of the authors’ affiliated institutions (2019-006 [0339]).
Consent to Participate
The requirement for informed consent was waived owing to the retrospective nature of the study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
