Abstract
Study Design
Retrospective cohort study.
Objectives
Imaging changes in the vertebral body after posterior lumbar interbody fusion (PLIF) are determined to be trabecular bone remodeling (TBR). This study aimed to investigate the influence of cage materials on TBR and segment stabilization in PLIF by studying image changes.
Methods
This was a retrospective study reviewing 101 cases who underwent one-level PLIF with three-dimensional porous titanium (3DTi) cages (53 patients) or polyether-ether-ketone (PEEK) cages (48 patients). Computed tomography images obtained 3 months, 1 year, and 2 years postoperatively were examined for TBR, vertebral endplate cyst formation as an instability sign, cage subsidence, and clear zone around pedicle screw (CZPS).
Results
No significant differences in the TBR-positivity rates were observed between the two cages at 3 months, 1 year, and 2 years postoperatively. However, all 3DTi cage segments that were TBR-positive at 3 months postoperatively showed no CZPS and fewer final instability segments than the TBR-negative segments (0% vs 9%). In contrast, although the PEEK cage segments that were TBR-positive at 3 months postoperatively were not associated with future segmental stabilization, those that were TBR-positive at 1 year postoperatively had fewer final instability segments than the TBR-negative segments (0% vs 33%).
Conclusions
The 3DTi cage segments with TBR 3 months postoperatively showed significant final segmental stabilization, whereas TBR at 1 year rather than 3 months postoperatively was useful in determining final segmental stabilization for the PEEK cage segments. The timing of TBR, a new osseointegration assessment, were associated with the cage material.
Keywords
Introduction
Titanium and polyether-ether-ketone (PEEK) are the most popular materials for interbody cages used in spinal fusion surgeries. Titanium is a popular implant material because of its excellent corrosion resistance and high biocompatibility. 1 However, because solid titanium has a much higher elastic modulus than bone, stress shielding and subsidence problems have been identified for intervertebral cages. 1 Recently, additive manufacturing techniques, such as three-dimensional (3D) printing, 2 have attracted attention because of their ability to improve the biomechanical properties of metal cages. Therefore, 3D porous titanium (3DTi) cages are currently the mainstream titanium cages used in clinical applications. In contrast, PEEK is a hydrophobic polymer with excellent properties, such as in vivo stability, fatigue resistance, and low elastic modulus, similar to bone. 3 Therefore, it is widely used as an interbody cage material. However, PEEK is not a perfect material because it does not make direct contact with bone and has properties that can induce inflammatory tissue reactions. 4 Nevertheless, PEEK cages have been continuously used for many years, providing satisfactory clinical results. Thus, despite their respective advantages and disadvantages, titanium and PEEK are currently popular primary materials for spinal interbody cages.
However, the clinical outcomes of posterior lumbar interbody fusion (PLIF) using 3DTi and PEEK interbody cages remain controversial. Although animal studies have shown that 3DTi achieves better osseointegration than PEEK, 2 several studies clinically comparing 3DTi and PEEK cages have found that segmental fusion and cage subsidence are comparable.5,6 Segmental fusion and cage subsidence are influenced by multiple factors and are consequences observed after the PLIF segment is complete. Therefore, it is difficult to explain the influence of cage materials on PLIF segment stabilization using these parameters because both 3DTi and PEEK cages can usually achieve favorable clinical outcomes.
Intervertebral osseointegration after lumbar fusion surgery produces changes on computed tomography (CT) images that are thought to be trabecular bone remodeling (TBR)
7
within the vertebral body away from the cage (Figure 1). Thus, attempts have been made to evaluate PLIF segment stabilization using TBR.
8
TBR is expected to result from stresses on the vertebral endplates being concentrated in the interbody cages in the PLIF segment, which is partially consistent with the results of finite element models.9,10 Theoretically, TBR is expected to be observed earlier in 3DTi cage segments than in PEEK cage segments. Alternatively, the implications of TBR may differ depending on the cage material. However, the difference in TBR has not yet been investigated between these cage materials. Thus, observing and understanding the changes in segments would provide a better understanding of the impact of cage materials in lumbar interbody fusion procedures. Therefore, this study aimed to investigate the imaging findings of patients who underwent one-level PLIF using 3DTi or PEEK cages up to 2 years postoperatively to understand the impact of cage materials on TBR and segment stabilization. Trabecular bone remodeling (TBR). TBR (white arrow) 1 year postoperatively in a PLIF segment using 3DTi cages. Coronal (A) and sagittal images (B). 3DTi, three-dimensional porous titanium.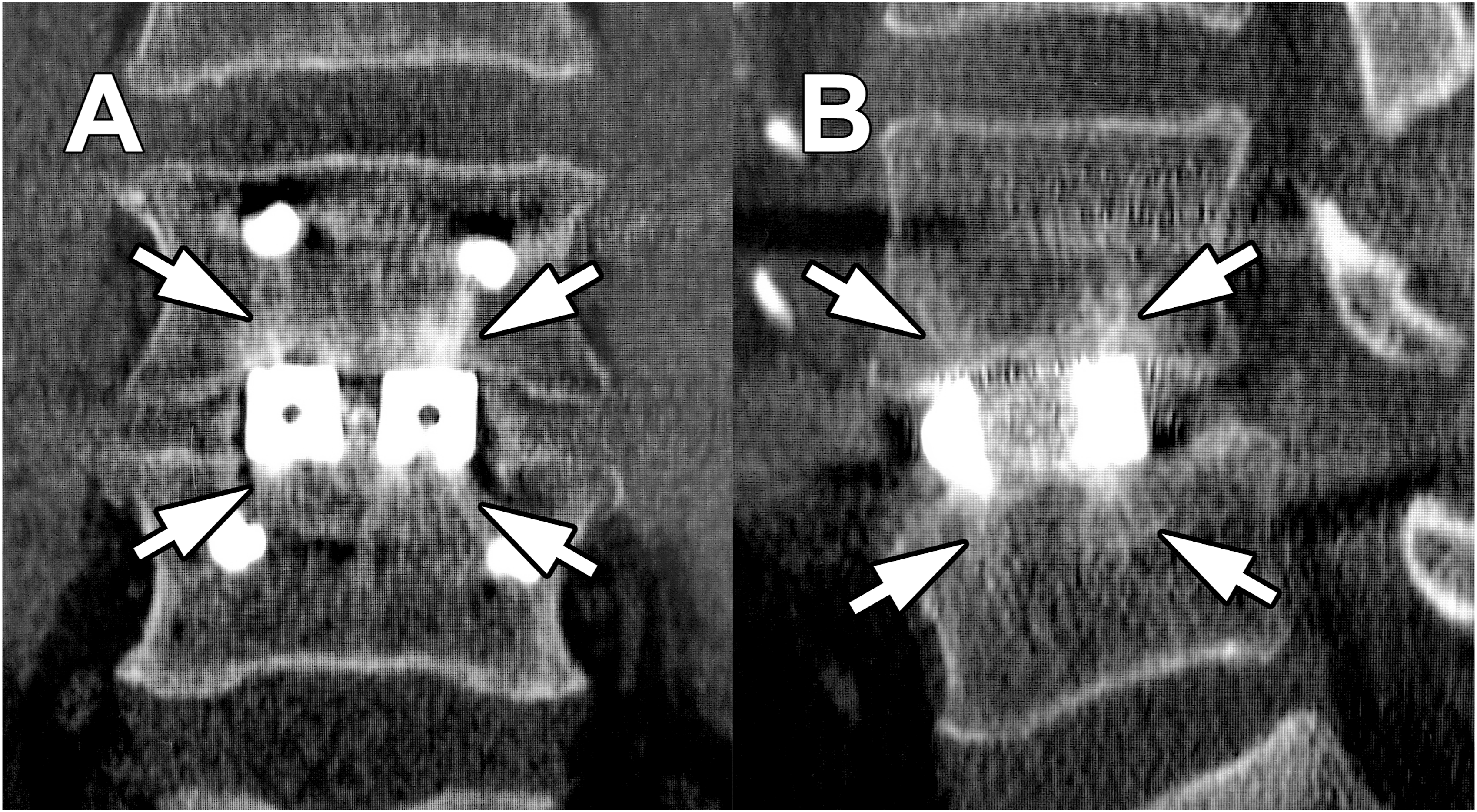
Methods
Patient Population
This study was approved by the Institutional Review Board of the authors’ affiliated institutions. Additional written informed consent was waived because all interventions, including surgical procedures and pre- and postoperative imaging studies, were routinely performed. This was a retrospective study including patients who underwent one-level PLIF at a single institution between July 2016 and June 2018. A total of 151 consecutive patients were selected as the initial patient population. Of the 151 patients, 16 were excluded, including those whose surgery was not the initial surgery, those with severe scoliosis (Cobb angle >30°), those with severe vertebral endplate sclerosis that inhibited obtaining exact imaging findings, and those on hemodialysis. Additionally, 35 patients were excluded because of not performing postoperative CT evaluation. Finally, 101 patients were included in the analysis (Figure 2). The patients were divided into two groups: the 3DTi group (53 patients), which included patients who underwent PLIF using 3DTi cages (Tritanium PL, Stryker, Kalamazoo, MI, USA) from July 2017 to June 2018, and the PEEK group (48 patients), which included patients who underwent PLIF using PEEK cages (OIC PEEK, Stryker) from July 2016 to June 2017. Patient selection flowchart. PLIF, posterior lumbar interbody fusion; CT, computed tomography.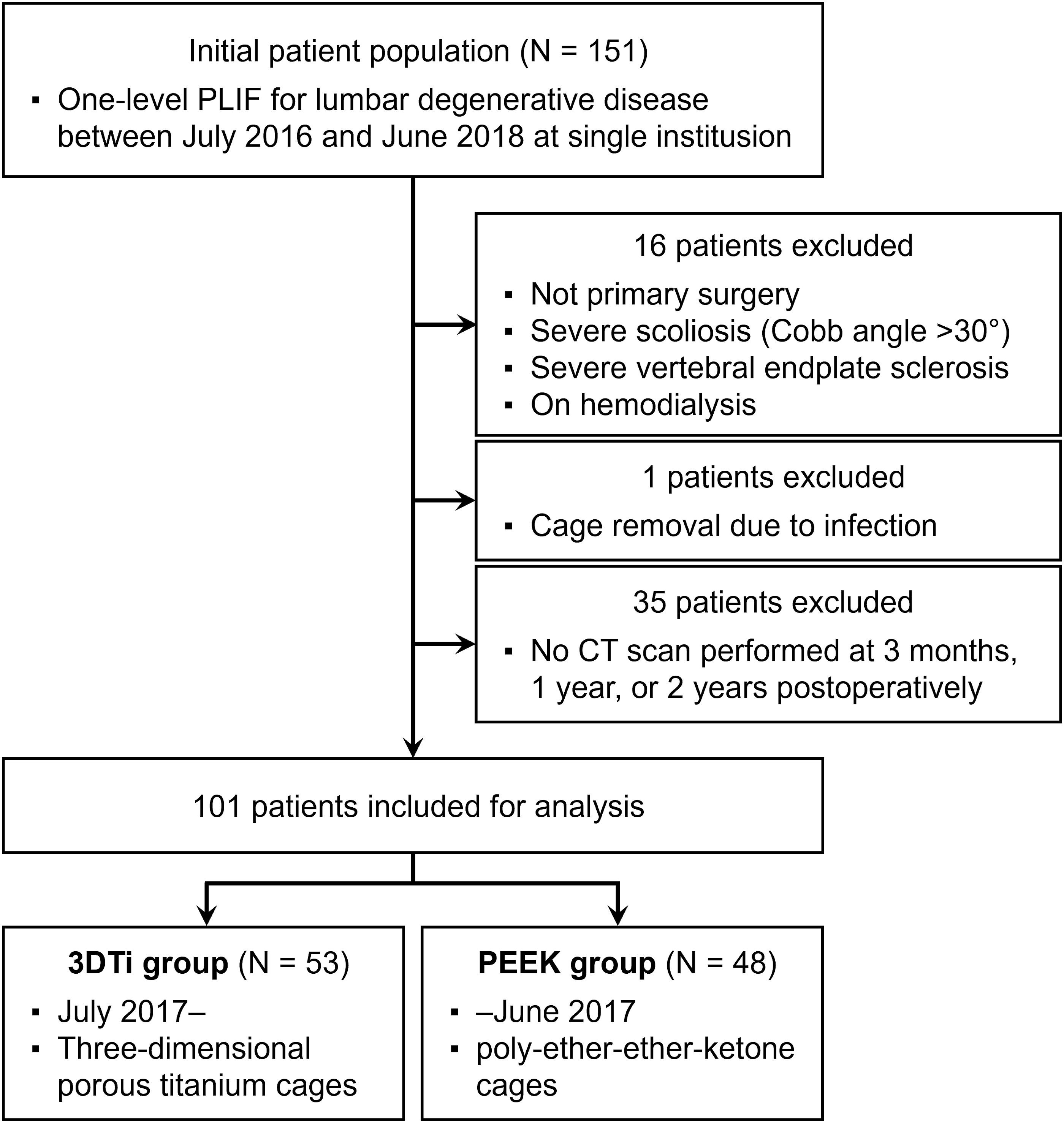
Surgical Procedure
PLIF was performed in a standard fashion. A bilateral pedicle screw (PS)-rod system and two interbody cages per segment were used in all patients. Bilateral or unilateral facet joints were resected as needed. After insertion of the PSs, temporary rod fixation was performed, and the disc was resected, taking care not to damage the vertebral endplates. The local bone was morselized and used as a bone graft before the insertion of the two cages. The two cages used were always of the same type, and the insides of the cages were filled with the same bone graft used in the intervertebral cavity. After cage insertion, compression manipulation was performed between PSs for final fixation. All patients wore a rigid brace for 3 months postoperatively.
Variables
Data, including patients’ age, sex, body mass index, comorbidities, and teriparatide administration, were investigated. CT values (Hounsfield units) were measured on CT axial images of the center of each vertebral body on the cephalic and caudal sides of the PLIF segment, 11 and the average value was recorded. Clinical outcomes were evaluated using the Japanese Orthopaedic Association (JOA) score. 12 The JOA scoring system (0-29 points) assesses three subjective symptoms (0-9 points), three clinical symptoms, such as neurological deficit (0-6 points), seven activities of daily living (0-14 points), and bladder function (−6 to 0 points).
Radiological evaluations were performed preoperatively, postoperatively, and 3 months, 1 year, and 2 years postoperatively. The lumbar lordosis angle and local lordosis angle, measured from the rostral endplate of the upper vertebra to the caudal endplate of the lower vertebra, were measured on standing lateral images, with positive values indicating lordosis. The difference in the local angle between postoperative and 2 years postoperatively was defined as correction loss. The local range of motion (ROM) was calculated from standing lateral forward- and backward-bending radiographs.
CT images were obtained using standard methods, and multiplanar reconstruction (MPR) images were produced. TBR was identified on coronal or sagittal CT-MPR images by comparison with immediate postoperative images. According to a previous report, 7 TBR was defined as relatively thick, tilted trabeculae inside the vertebra that were not depicted on normal (immediate postoperative) images. TBR were present from the cage-vertebral endplate contact area and extended into the vertebral body. TBR was evaluated regardless of the size of the spread. Segmental bridging was defined on CT-MPR images as the presence of an intervertebral bone graft in contact and continuity with both cephalad and caudal vertebral endplates or vertebral body-to-vertebral body bridging by the new bone.
The formation of vertebral endplate cysts (VECs)13,14 appearing in unstable interbodies was used as an imaging reference that contrasted with osseointegration. The appearance of VECs that were not present on previous imaging or an enlargement of VECs was defined as VEC-positive. Additionally, because the vertebral endplate in contact with the cages may show degeneration-like sclerosis, and small depression (local cyst) 15 may be observed, this depression was also defined as a VEC in this study. Cage subsidence was defined as cage migration of more than 2 mm to the vertebral endplate compared with previous imaging studies. The absence of subsidence progression after previous imaging was defined as negative. Of note, no cage dropout from the intervertebral space was observed in this study. Clear zone around PS (CZPS) was defined as a translucent zone of 2 mm or greater width around the PS. CZPS was expressed as the number of PSs that met the definition (0-4). Segmental instability was defined as any of the following findings: CZPS of 4, intervertebral vacuum sign, or local ROM >5°. 16 Of note, no pseudarthrosis that required revision surgery was observed.
Statistical Analysis
Data were presented as mean ± standard deviation for continuous variables or as numbers and percentages for categorical data. Statistical analyses were performed using R version 4.3.1 (The R Foundation; https://www.R-project.org) with Wilcoxon rank-sum and Fisher exact tests. A P value <.05 was considered statistically significant.
Results
Patient Characteristics.
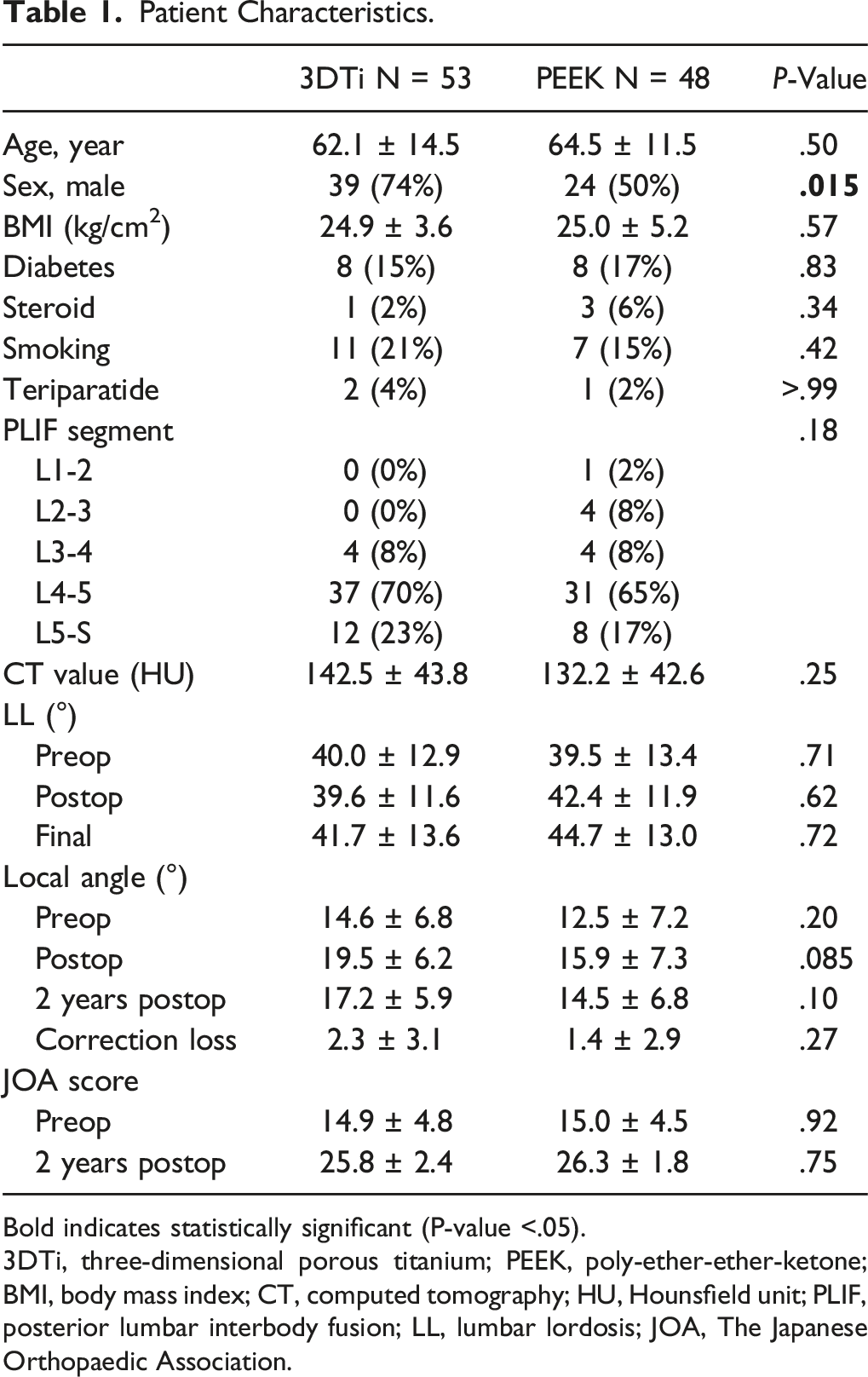
Bold indicates statistically significant (P-value <.05).
3DTi, three-dimensional porous titanium; PEEK, poly-ether-ether-ketone; BMI, body mass index; CT, computed tomography; HU, Hounsfield unit; PLIF, posterior lumbar interbody fusion; LL, lumbar lordosis; JOA, The Japanese Orthopaedic Association.
Comparison of Imaging Findings by Cage.
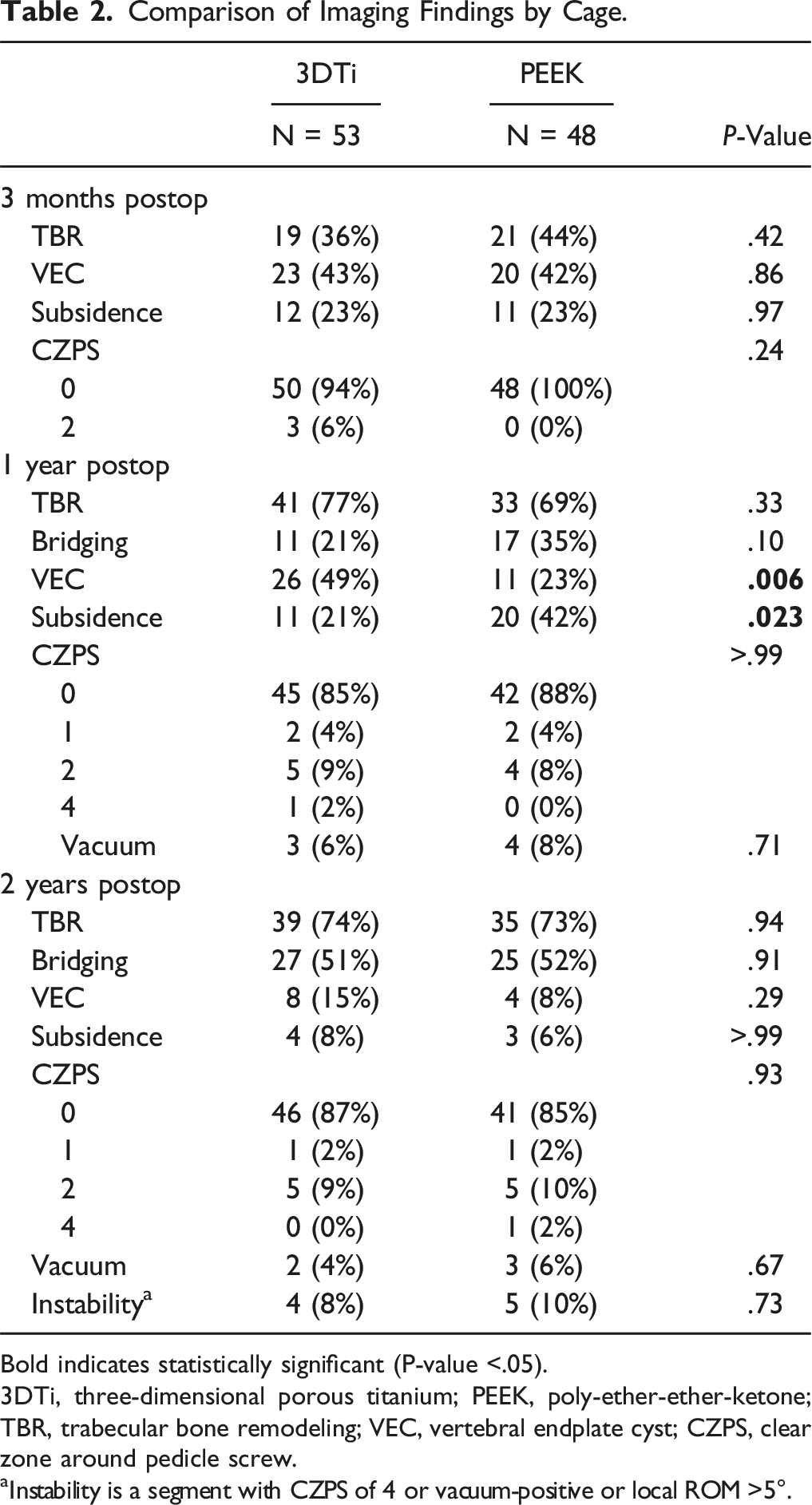
Bold indicates statistically significant (P-value <.05).
3DTi, three-dimensional porous titanium; PEEK, poly-ether-ether-ketone; TBR, trabecular bone remodeling; VEC, vertebral endplate cyst; CZPS, clear zone around pedicle screw.
aInstability is a segment with CZPS of 4 or vacuum-positive or local ROM >5°.
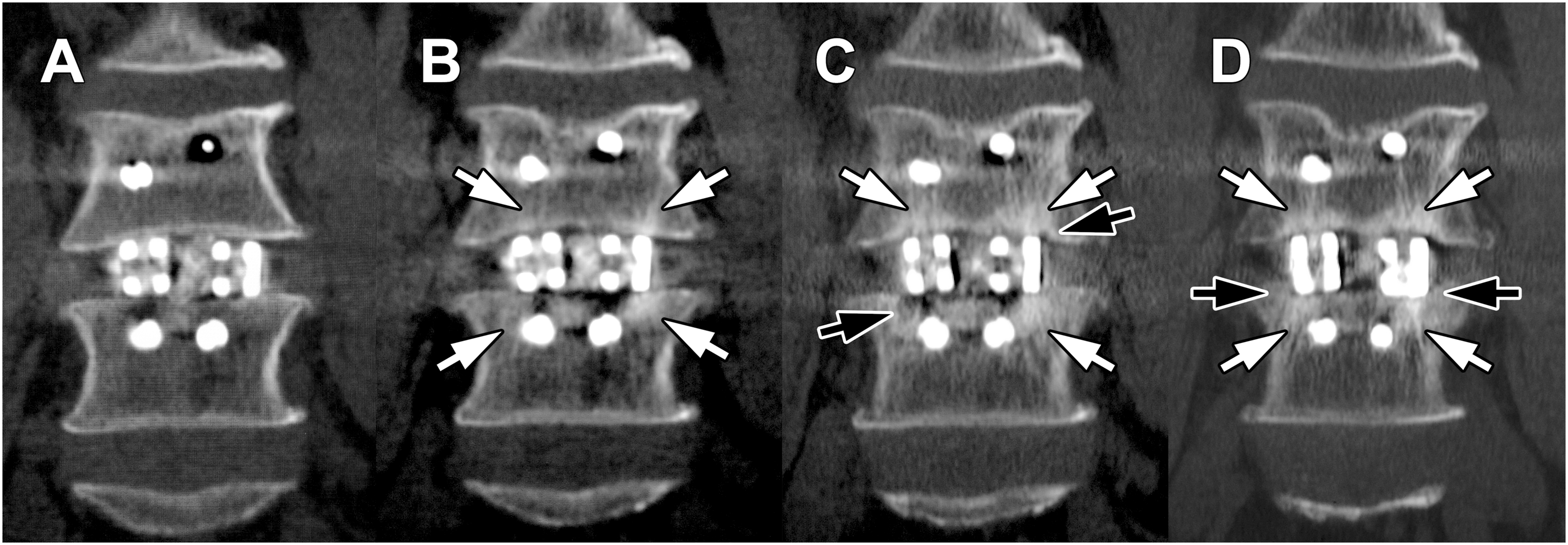
3DTi group. (A) Postoperative coronal image. (B) 3 months postoperatively, TBR was observed (white arrow). (C) 1 year postoperatively, TBR (white arrow) and VEC (black arrow) were observed in both cages. (D) 2 years postoperatively, TBR (white arrow) continued, VEC disappeared, and cage subsidence (black arrow) was observed. 3DTi, three-dimensional porous titanium; TBR, trabecular bone remodeling; VEC, vertebral endplate cyst.
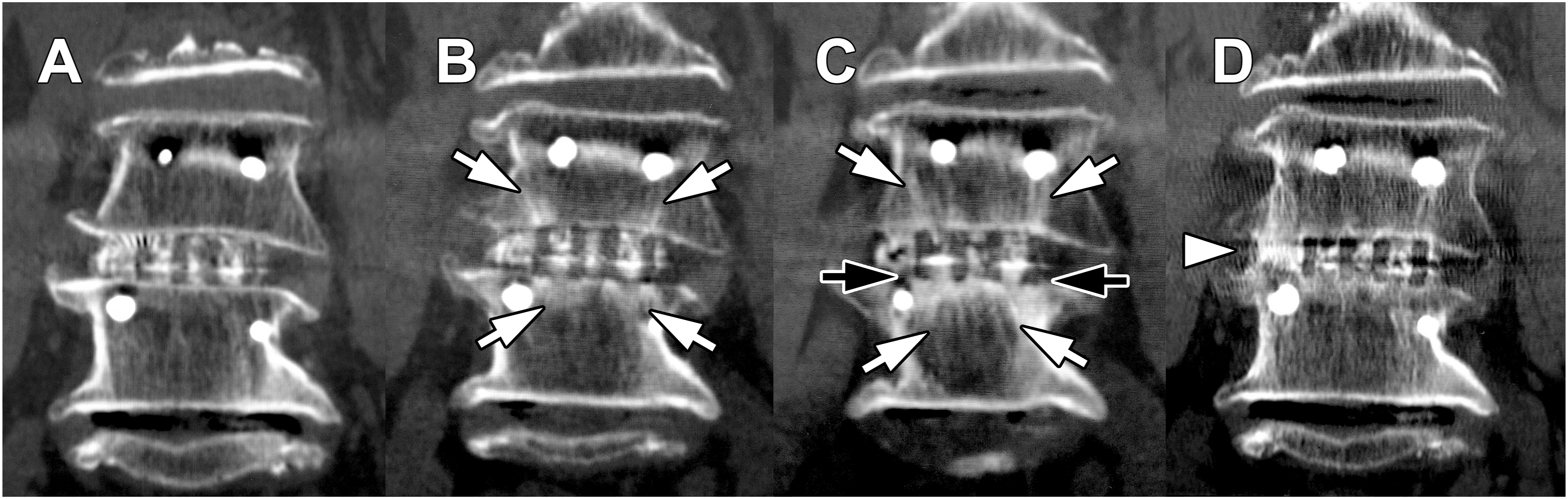
PEEK group. (A) Postoperative coronal image. (B) 3 months postoperatively, TBR was observed (white arrow). (C) 1 year postoperatively, TBR (white arrow) and cage subsidence (black arrow) were observed in both cages. (D) 2 years postoperatively, interbody bridging (white arrowheads) was observed. PEEK, polyether-ether-ketone; TBR, trabecular bone remodeling.
Comparison of Imaging Findings by 3-Month TBR According to Cage.
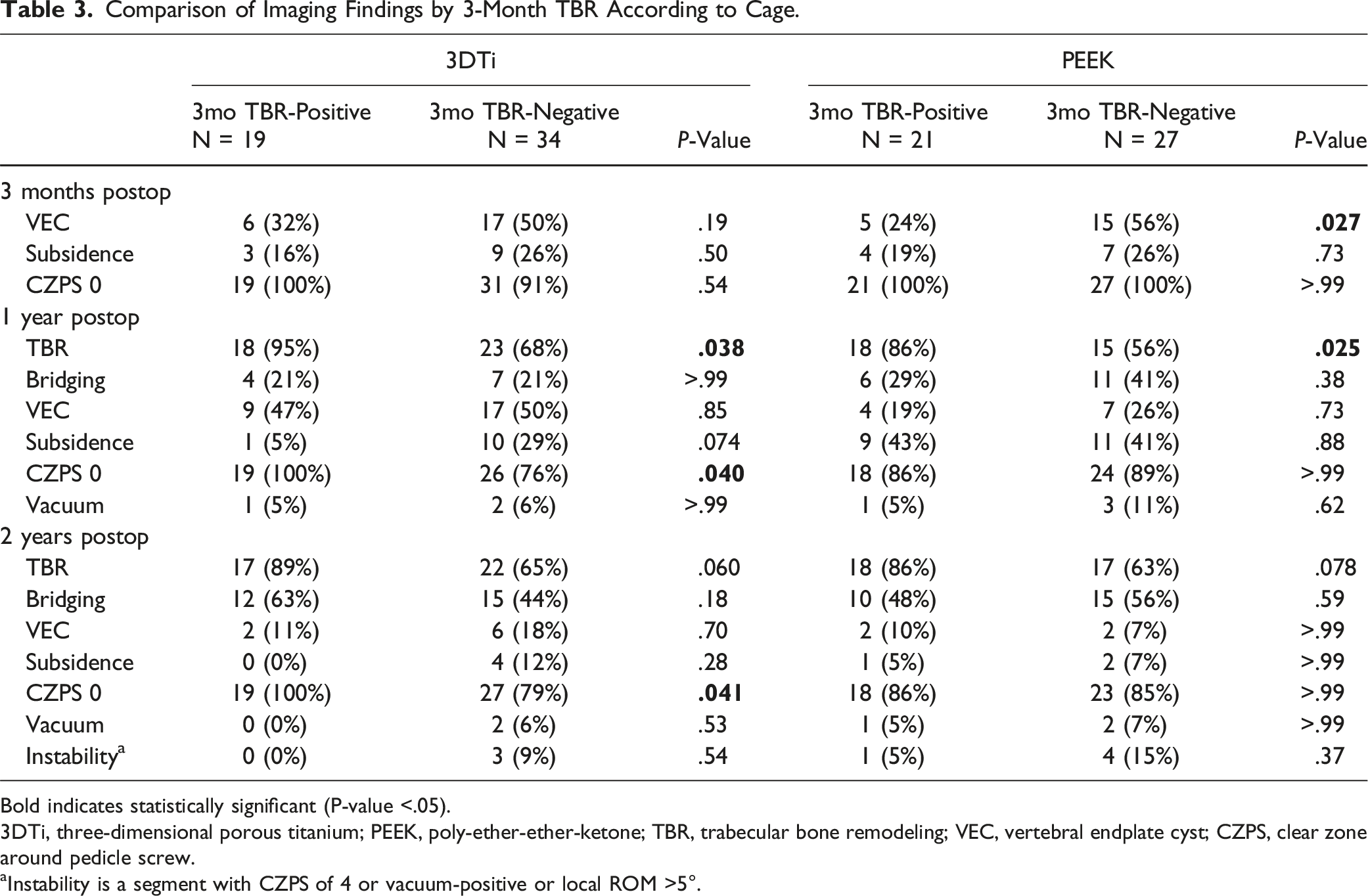
Bold indicates statistically significant (P-value <.05).
3DTi, three-dimensional porous titanium; PEEK, poly-ether-ether-ketone; TBR, trabecular bone remodeling; VEC, vertebral endplate cyst; CZPS, clear zone around pedicle screw.
aInstability is a segment with CZPS of 4 or vacuum-positive or local ROM >5°.
Comparison of Imaging Findings by 1-Year TBR According to Cage.
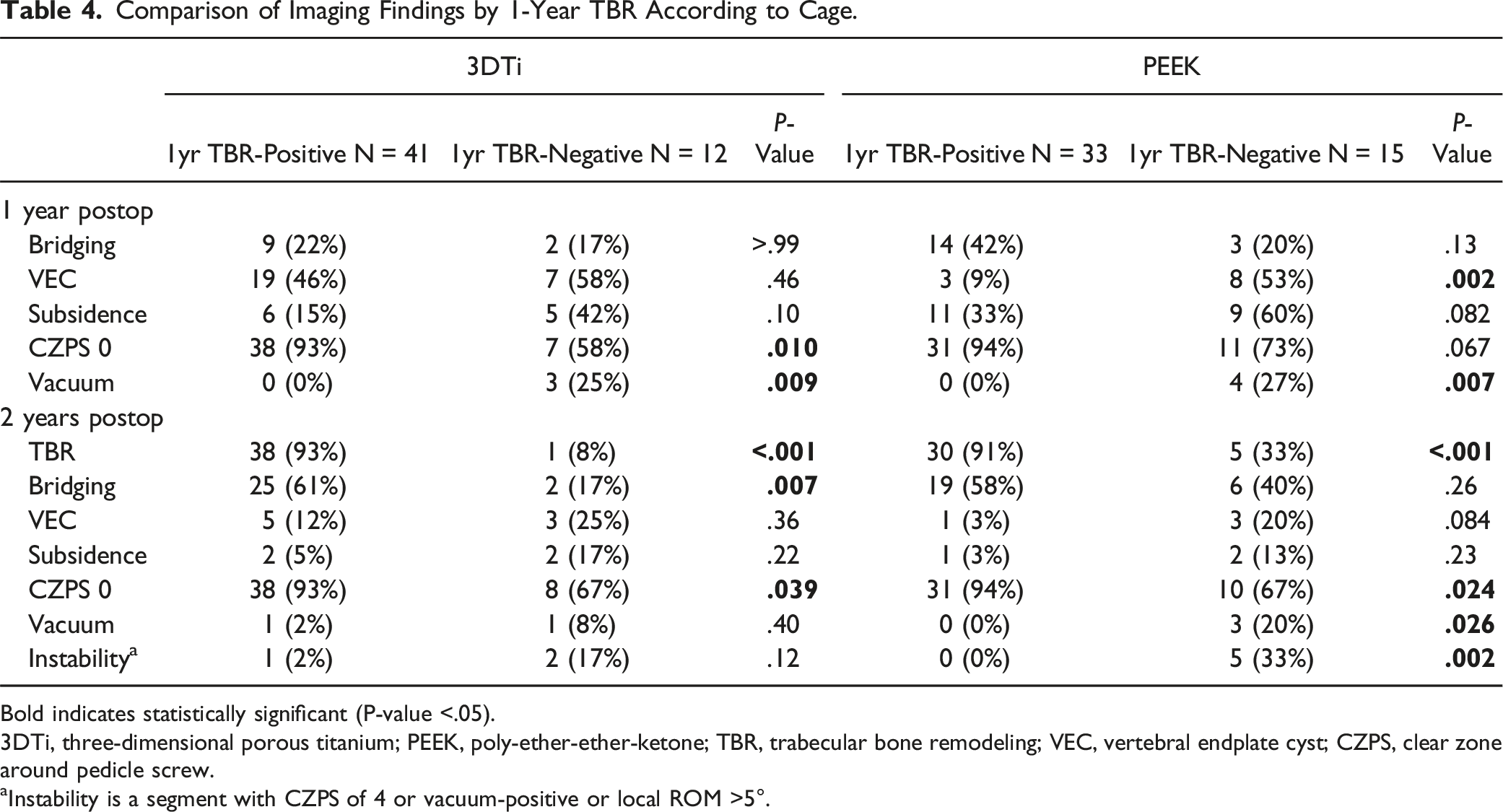
Bold indicates statistically significant (P-value <.05).
3DTi, three-dimensional porous titanium; PEEK, poly-ether-ether-ketone; TBR, trabecular bone remodeling; VEC, vertebral endplate cyst; CZPS, clear zone around pedicle screw.
aInstability is a segment with CZPS of 4 or vacuum-positive or local ROM >5°.
Discussion
In this study, we investigated changes in CT imaging findings up to 2 years postoperatively in PLIF segments with 3DTi or PEEK cages. At 2 years, the incidence of TBR, VEC, and cage subsidence did not significantly differ by cage. Therefore, the imaging findings and clinical outcomes were comparable. However, 1 year postoperatively, a slight but definitive difference was observed between the 3DTi and PEEK cage segments. The 3DTi cage segments had a higher incidence of VEC formation, and the PEEK cage segments had a higher incidence of cage subsidence. Furthermore, the 3DTi cage segments with TBR 3 months postoperatively showed significant final segmental stabilization, whereas TBR at 1 year rather than 3 months postoperatively was useful in determining future segmental stabilization for the PEEK cage segments. Thus, the new measure of TBR allowed us to explore the relationship between cage material and osseointegration over time.
The clinical differences between the 3DTi and PEEK cages may be slight. No differences were observed in CT imaging findings between the cages at 2 years postoperatively and in spinal alignment or clinical assessment. The results of this study showed that the 3DTi cage segments that were TBR-positive in the early postoperative period (3 months) had significantly more findings suggestive of subsequent segmental stability, but not in the PEEK cages. This indicates that the early TBR-positive 3DTi cage segments establish osseointegration, whereas the early TBR in the PEEK cage segments may not. Furthermore, the positivity or negativity of TBR at 1 year postoperatively is a significant predictor of the fate of the segment at 2 years, regardless of cage material. Therefore, it may be too early to determine the establishment of PLIF stability for PEEK cage segments by TBR determination at 3 months postoperatively. A previous study that analyzed data from patients who underwent PLIF with 3D porous tantalum or titanium-coated PEEK cages showed an association between TBR positivity at 3 months postoperatively and segment stability at 1 year.
7
However, early postoperative TBR positivity was significantly more common in porous tantalum cage segments.
8
Thus, TBR positivity at “3 months postoperatively” was associated with final PLIF stability only in segments with “3D porous metal” cages. Additionally, a significant association was observed between TBR at 1 year and segment stability at 2 years, regardless of cage material. Thus, it is more reasonable to assume that TBR itself is a universal phenomenon and that the cage material influences the timing of osseointegration establishment (Figure 5). Illustration of vertebral bone responses to cage material. In the 3DTi cage segments (A–C), the contact between the vertebral endplate and titanium caused osseointegration and TBR. However, TBR and VEC were observed simultaneously during the conflict between forces that attempted to stabilize and destabilize the segment (B). As segment stabilization was established, the TBR developed, and the VEC shrank (C). In the PEEK cage segments (D–E), direct osseointegration did not occur because of the nonbioactivity of PEEK. TBR at 3 months postoperatively did not appear to be associated with osseointegration, but TBR at 1 year suggested osseointegration (E). Once segmental stabilization was established, TBR developed as in 3DTi (F). 3DTi, three-dimensional porous titanium; TBR, trabecular bone remodeling; VEC, vertebral endplate cyst; PEEK, polyether-ether-ketone.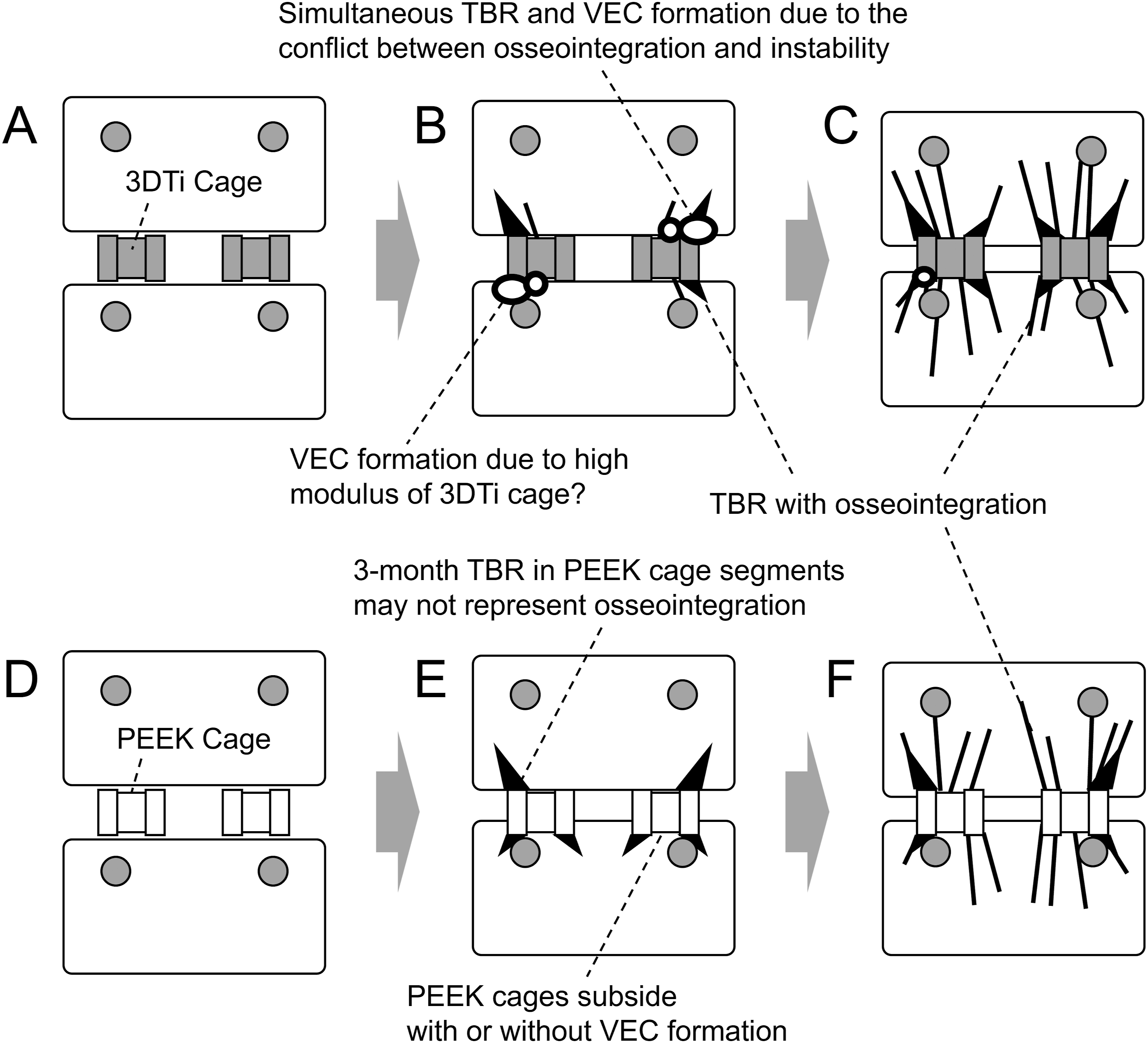
In this study, VEC formation, a recognized predictor of delayed union and nonunion, 13 was more common in 3DTi cage segments than in PEEK 1 year postoperatively. This finding contrasts with a previous report that found no significant difference in VEC formation between 3DTi and PEEK cage segments. 17 This may be due to the expanded definition of VEC in this study. Stress-induced microfractures of vertebral endplates due to cage instability may be a possible cause of VEC formation. 13 Therefore, the small depression with degenerative-like endplate sclerosis was treated as a VEC. Additionally, PEEK cage segments showed opposing rates of TBR (suggesting stability) and VEC (suggesting instability), as previously reported, 7 whereas 3DTi cage segments showed no difference in VEC formation with or without TBR. Therefore, VECs in 3DTi cage segments may not only be suggestive of instability but also include findings specific to 3DTi cages. The 3DTi cages have an elastic modulus of 6.2 GPa, 18 which is much smaller than that of solid titanium but still higher than that of pure PEEK (about 3-4 GPa) 19 . The difference in the elastic modulus between the bone and cage may be associated with cage subsidence. 1 It is possible to hypothesize that the higher elastic modulus of 3DTi cages than vertebral trabecular bone is a risk factor for endplate microfracture and induces VEC formation compared with PEEK. Therefore, the 3DTi cage segments may promote VEC formation with or without TBR. Therefore, further studies are needed to investigate the relationship between these imaging findings and eventual segmental stabilization.
Although PEEK can reduce the problem of cage subsidence compared with “solid” titanium cages because of its low elastic modulus, 1 the potential of PEEK and 3DTi cages to subside remains controversial. Comparisons of the two materials have shown either no significant difference in subsidence5,6 or a greater risk of subsidence for PEEK.20-22 Because these results may be qualified by the definition of subsidence and various biases, the results of more subsidence for PEEK cages than for 3DTi cages in this study should also be carefully considered. In contrast to the subsidence rate, the PEEK cage segments had a relatively lower incidence of VEC formation than the 3DTi cage segments. Therefore, the mechanism of PEEK cage subsidence may be different from that of the metal cage. A straightforward interpretation of the imaging findings indicates that PEEK cages can migrate to vertebral endplates without vertebral endplate damage that would form VECs. PEEK has low biocompatibility and does not allow direct osseointegration. 2 Therefore, even if the PEEK cage and vertebral endplate are in contact on CT imaging, an intervening tissue exists between them histologically. This form of connection not only does not have robust stability like the bridging state but also causes foreign body responses and chronic inflammation that promote fibrous encapsulation instead of bone binding.19,23 Although how the cage will behave in response to this biological reaction is unknown, the potential risk of cage migration to the vertebral body may be triggered. Thus, PEEK cages, despite their advantage in material elastic modulus, are potentially more at risk of subsidence than 3DTi cages.
Paradoxically, TBR is not a finding that corresponds one-to-one with the establishment of intervertebral stability. As evidence, this series included a segment that was TBR-positive but also showed instability. It has been clinically shown that a metal ball insertion between vertebrae results in a high-density area on CT images. 24 Thus, some of the increased CT values of the trabecular bone adjacent to the intervertebral implant may be due to densification of the underlying bone due to subsidence. 24 This finding may clarify the coexistence of TBR and VEC at a higher rate in 3DTi cage segments. In other words, some findings determined to be TBR probably included densification of the underlying bone by the cage. However, because descriptors to accurately distinguish these findings are currently lacking, this confusion is a problem to be solved in the future. Furthermore, PLIF segments, like other bone tissues, are naturally subject to constant remodeling. Thus, no matter how strong the interbody stabilization findings are at the time of image acquisition, they are not likely to remain the same indefinitely. In short, the status of the PLIF segment should be judged comprehensively in conjunction with the other findings. Despite these challenges, TBR is a promising imaging finding for determining PLIF segment stability.
This study has some limitations. First, the number of cases was relatively small. Therefore, judgments based on P values may be underestimated because of the lack of statistical power. Second, the interpretation of TBR and other imaging findings may be subjective in its determination. Despite these limitations, the results of this study provide valuable insight into the interpretation of imaging results and evaluation of vertebral cage osseointegration for each of the two distinctive and universally used cage materials.
Conclusion
This study investigated changes in CT imaging findings up to 2 years postoperatively in one-level PLIF with 3DTi or PEEK cages. At 2 years, no significant differences were observed in imaging findings by cage. However, the 3DTi cage segments that were TBR-positive at 3 months postoperatively showed significant signs of segmental stabilization. The PEEK cage segments that were TBR-positive at 3 months postoperatively were not associated with future segmental stabilization. However, the PEEK cage segments that were TBR-positive at 1 year postoperatively were associated with future segmental stabilization. Thus, the timing and status of TBR, a new osseointegration assessment, were associated with cage material, which indicates a relationship between cage material and osseointegration over time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
