Abstract
Study Design
Retrospective cohort study
Objectives
The criteria for determining completion of intervertebral stability after posterior lumbar interbody fusion (PLIF) remain controversial. Several new radiological indicators of bone growth and osteointegration have been established. We compared computed tomography (CT) findings related to osteointegration after PLIF with interbody cages of two different materials and designs.
Methods
We retrospectively analyzed data from 103 patients who underwent PLIF with three-dimensional porous tantalum (Tn) cages or titanium-coated polyetheretherketone (TiP) cages. CT images obtained 3 months and 1 year after surgery were examined for trabecular bone remodeling (TBR), cancellous condensation (CC), and vertebral endplate cyst (VEC) formation. The incidences of each finding were compared by cage type, and rates of instrument failure and pseudarthrosis were determined.
Results
Three months postoperatively, 87% of the levels with Tn cages exhibited TBR, whereas 96% of those with TiP cages did not (P < .001). Most levels with Tn cages levels exhibited TBR and no CC 3 months (81%) and 1 year (94%) after surgery. Although 78% of levels with TiP cages exhibited CC and no TBR 3 months after surgery, 59% exhibited both CC and TBR 1 year after surgery. Significantly fewer VECs formed around the Tn cages than around the TiP cages both 3 months (P = .002) and 1 year (P < .001) after surgery. Implant-related problems occurred at levels that exhibited neither TBR nor CC.
Conclusions
The porous tantalum cage may enable intervertebral stability that is comparable to bony fusion soon after surgery.
Keywords
Introduction
Posterior lumbar interbody fusion (PLIF) is one of the standard procedures for correcting lumbar spinal stenosis with instability or malalignment. 1 Achieving clinical success with orthopedic implants that support loads such as those provided by PLIF is important 2 because pseudarthrosis after PLIF can lead symptom recurrence, malalignment, poor quality of life, and hardware failure.3-5 Although the rate of radiological bony fusion after PLIF is generally considered favorable, the state of fixation/fusion should be determined as well. Despite the importance of osteointegration, 2 described as “a process whereby clinically asymptomatic rigid fixation of alloplastic materials is achieved, and maintained, in bone during functional loading,” 6 no universal method of assessment has been established.4,7 Therefore, the criteria for determining completion of intervertebral stability in PLIF remain controversial.
Computed tomography (CT) is considered an excellent imaging method to evaluate spinal fusion; the appearance of bony continuity between the vertebrae can indicate bony fusion. Rough metal surface structures designed with titanium-coated polyetheretherketone (PEEK) and three-dimensional (3D) porous metals have been applied to interbody cages because their surfaces are reportedly bioactive in inducing bony fusion.8-13 For these cages, several new radiological indicators have been devised to detect bone growth or osteointegration. Makino et al 14 evaluated bone formation on the cage surface by using color mapping based on Hounsfield unit (HU) values. Kashii et al 15 evaluated cage–vertebral endplate bone growth by calculating vertebral cancellous condensation from HU values. Furthermore, Segi et al 16 proposed that changes on CT images of the inside of the vertebral trabecular bone adjacent to the cage could be visualized as indicators of osteointegration.
Several studies have attempted to develop suitable materials for intervertebral cages. Although titanium has excellent strength and biocompatibility for use in living bodies, 17 its Young’s modulus >100 GPa is much stiffer than that of cortical bone (6-30 GPa); 18 this difference in elastic modulus may lead to failure. PEEK has a low elastic modulus, similar to that of cancellous bone (3 GPa for pure PEEK and 7.7-11.5 GPa for carbon fiber-reinforced PEEK); 18 however, its bioactivity is relatively low due to its hydrophobic nature. Surface coating can improve PEEK bioactivity; 19 in particular, titanium coating can improve the performance of PEEK intervertebral cages. Another solution is 3D porous metal. Currently, most porous metals used in clinical practice are made of tantalum, 20 as porous tantalum has an elastic modulus of 3 GPa, 20 similar to that of cancellous bone. Furthermore, porous metal structures designed to mimic human bone tissue have similar properties to cancellous bone,21,22 have been applied to intervertebral cages. Thus, we chose to focus on titanium-coated PEEK and porous tantalum.
Although cage materials or designs may influence postoperative imaging appearances, this issue has not been fully investigated. Investigation of the effect of cage materials on imaging findings can clarify the properties of these materials in their adapted state in the human body and provide a basis for considering their clinical advantages and disadvantages. The purpose of this study was to compare CT imaging findings related to osteointegration after PLIF with two different types of interbody cages.
Methods
Patient Population
We retrospectively reviewed the data of 155 consecutive patients who underwent PLIF with 3D porous tantalum (Tn) cages or titanium-coated PEEK (TiP) cages for lumbar degenerative disease between 2017 and 2020. All clinical interventions and radiological examinations were routine procedures. This research has been approved by the IRB of the authors’ affiliated institutions. All participants provided written informed consent to use their data. The indications for PLIF were (1) the appearance of spondylolisthesis with >3 mm slippage or >5° of posterior opening on dynamic lateral radiographs and (2) stenosis that necessitated total facetectomy for decompression (eg, foraminal stenosis caused by severe intervertebral height reduction). Data from patients who did not undergo postoperative CT evaluation both 3 months and 1 year postoperatively were excluded, as were those of patients with severe scoliosis (Cobb angle of >30°), patients with preoperative severe vertebral endplate sclerosis that made the CT findings difficult to interpret, and patients on hemodialysis because of renal failure. We also excluded data of patients who had sustained vertebral fractures at the level included in the PLIF and of patients whose cages were later removed because of surgical site infection. The final sample included 103 patients (mean age of 69.2 ± 11.5 years), 46 of whom were men (Figure 1). Patient selection flowchart. Tn, three-dimensional porous tantalum; TiP, titanium-coated polyetheretherketone.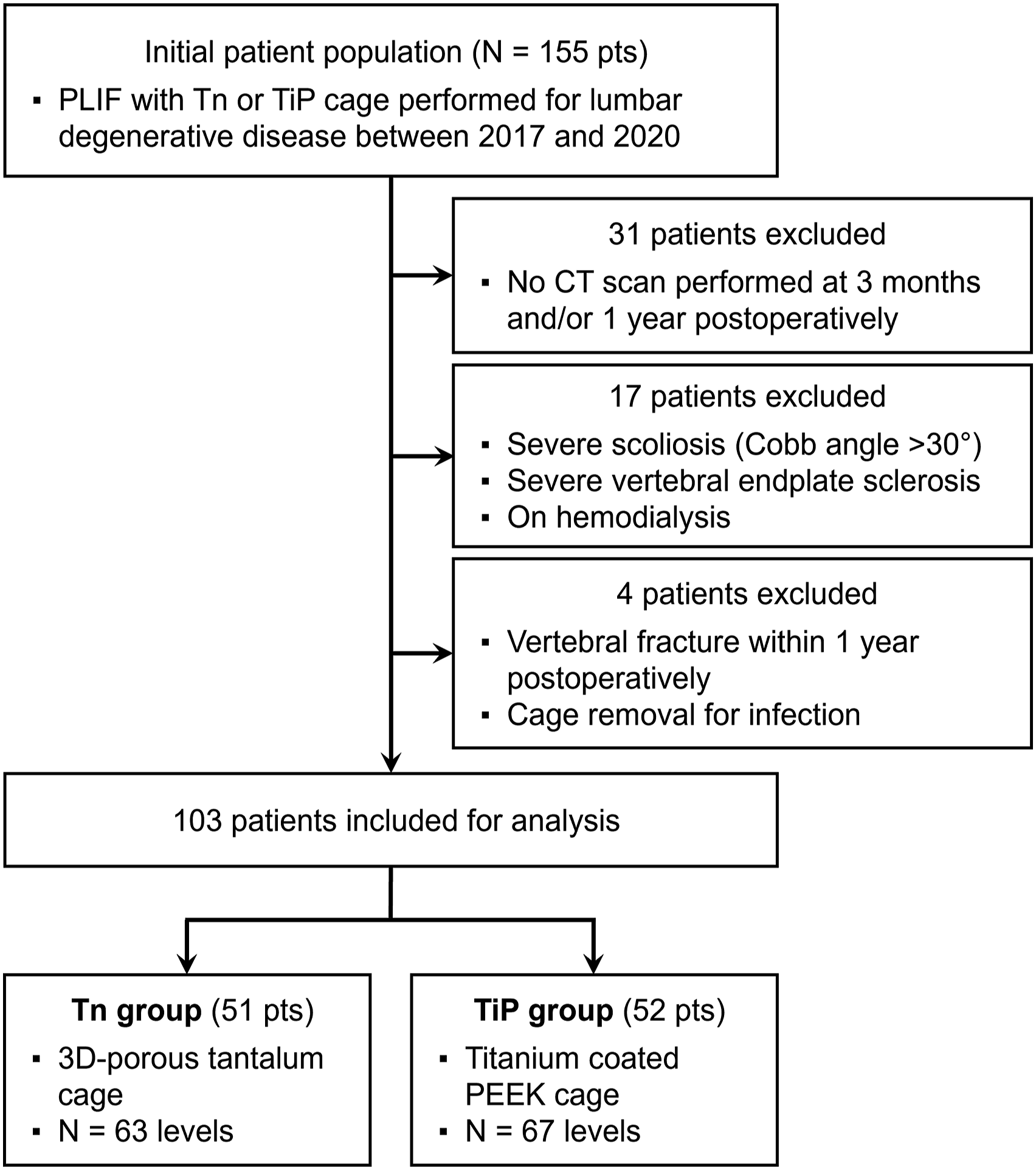
Surgical Procedure
PLIF was performed in a standard manner. 16 A bilateral pedicle screw–rod system and two interbody cages per level were used in all cases. Bilateral facet joints were resected as required, and grade 1 osteotomy 23 was performed in the majority of cases. After the pedicle screw was inserted, temporary fixation was performed, and the disc was resected, with care not to damage the vertebral endplate. The local bone was morselize to form a graft, and at each level, the graft bone was inserted into the intervertebral space before the two cages were inserted. The two cages were always of the same type: either Tn cages (TM Ardis Interbody System; ZimVie, Westminster, CO, USA), in 51 patients at a total of 63 levels, and TiP cages—MectaLIF-TiPEEK (Medacta, Castel San Pietro, Switzerland), Concorde Pro Ti (DePuy Synthes, Raynham, MA, USA), or Capstone PTC (Medtronic Sofamor Danek, Memphis, TN, USA)—in 52 patients at a total of 67 levels. The Tn cage did not have space for bone grafting; the inside of the TiP cage was filled with the same grafted bone used in the intervertebral space. All patients wore a hard brace for 3 months postoperatively.
Patients’ Demographic and Operative Data
We documented patients’ age, sex, body mass index, preoperative bone mineral density (T-score of the proximal femur measured with dual-energy X-ray absorptiometry [Prodigy; GE Healthcare, Chicago, IL, USA]), and PLIF levels. The Japanese Orthopaedic Association 24 scores (range: 0-29 points) were recorded before and 1 year after surgery. In the Japanese Orthopaedic Association scoring system, three subjective symptoms are scored from 0 to 9; three clinical symptoms, such as neurological deficit, are scored from 0 to 6; seven activities of daily living are scored from 0 to 14; and bladder function is scored − 6 to 0. The higher the scores, the better the patient’s condition.
Radiological Assessments
Standing radiographs were obtained preoperatively, postoperatively, and 1 year after surgery. Lateral images were used to measure lumbar lordosis angle (positive values indicated lordosis) and local angle (measured from the rostral endplate of the upper instrumented vertebra to the caudal endplate of the lower instrumented vertebra; positive values indicated lordosis). The correction angle was defined by the difference between the local angle before and immediately after surgery, and the difference in local angle from immediately after surgery to one year later was defined as correction loss. In addition, to measure the range of motion of the local angle, we used standing lateral forward- and backward-bending radiographs. CT images were obtained through standard methods at our institution, and multiplanar reconstruction (MPR) images were produced.
Trabecular Bone Remodeling
TBR was defined as trabeculae placed obliquely in the adjacent intravertebral cage toward pedicle screws or the endplate on the other side, according to previous reports.
16
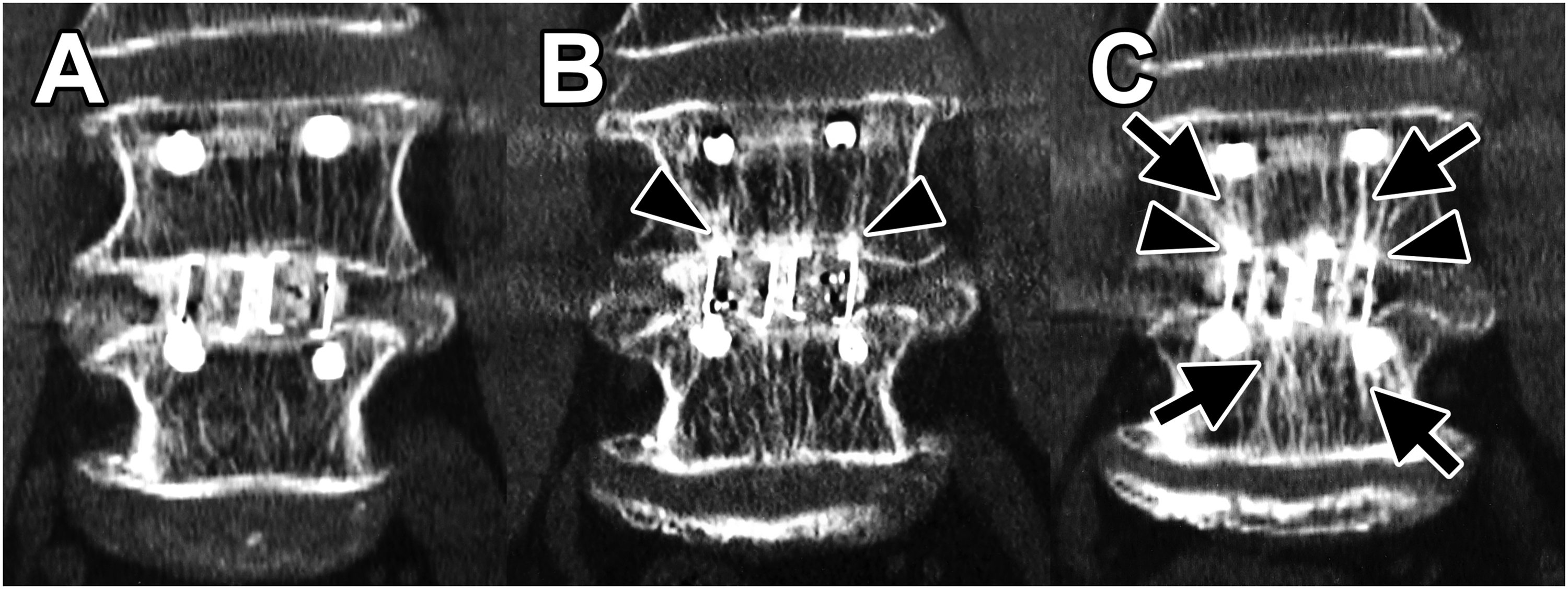
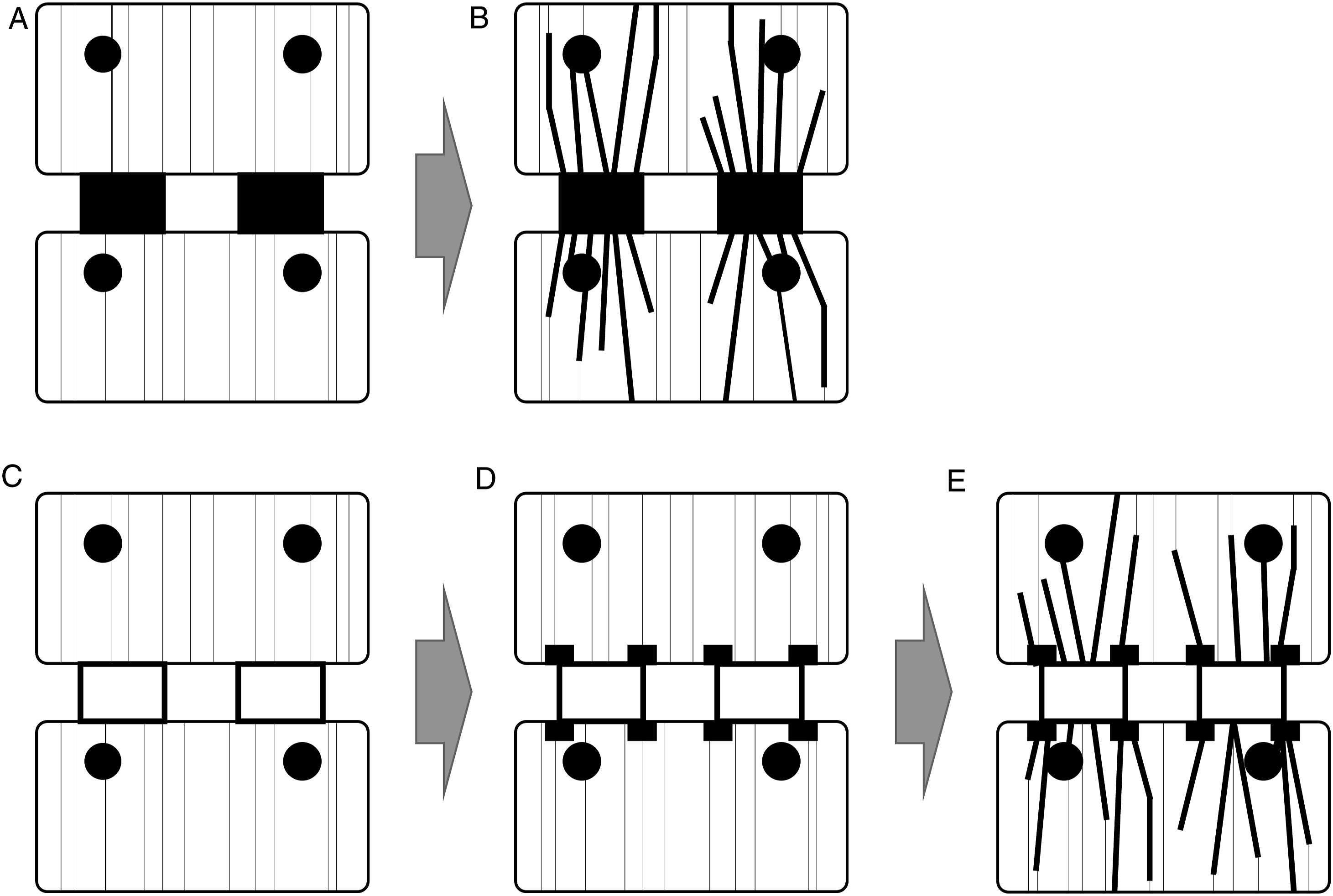
To identify TBR, we compared immediate postoperative coronal CT-MPR images with those obtained 3 months and 1 year later. TBR was distinguished by its obliquity and thickness, being thicker than the normal (preoperative) trabeculae (Figures 2(B), (C) and 3(C)); schematic diagrams of TBR are presented in Figure 4(B), (D), and (E). TBR was identified in the vertebrae on both sides of the cage in most cases. Therefore, the presence of TBR was counted for each cage, and sum of these numbers was recorded as a score, which ranged from 0 to 2 (0, no TBR; 1, TBR was present around one cage; and 2, TBR was present around both cages). Representative images of three-dimensional porous tantalum (Tn) cages (A) Postoperative view. (B) View 3 months after surgery. (C) View 1 year after surgery. Arrows point to areas of trabecular bone remodeling. Representative images of titanium-coated polyetheretherketone (TiP) cages (A) Postoperative view. (B) View 3 months after surgery. Arrowheads point to areas of cancellous condensation. (C) View 1 year after surgery. Arrows point to areas of trabecular bone remodeling. Schematic diagram of TBR and CC. (A) Postoperative schema of the Tn cage. (B) TBR observed around the Tn cage (corresponding to Figure 2(B) and (C)). (C) Postoperative schema of the TiP cage. (D) CC observed around the TiP cage (corresponding to Figure 3(B)). (E) CC and TBR observed around the TiP cage (corresponding to Figure 3(C)).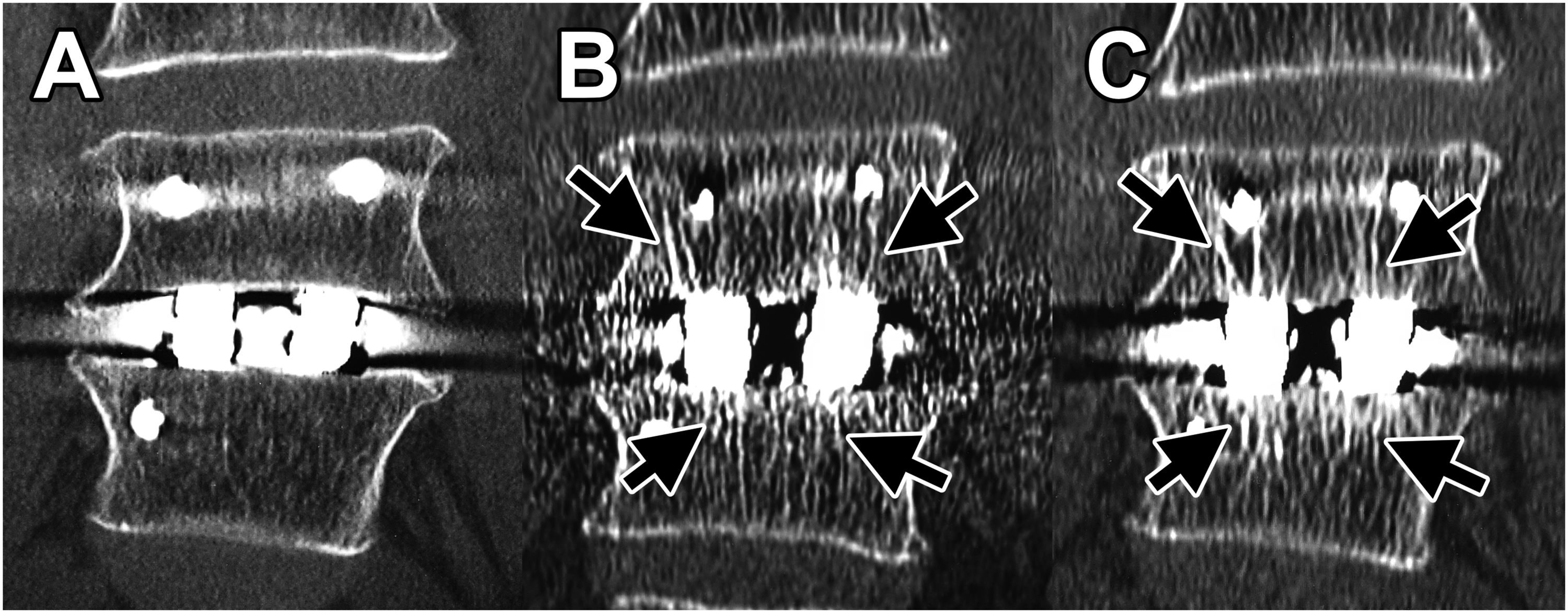
Cancellous Condensation
Regarding a previous report, 15 CC was defined as the presence of locally high radiation density of the vertebral endplate in contact with the edge of the cage (ie, a spot-like appearance of high-density areas in the vertebral endplate; Figure 3(B)); schematic diagrams of CC are shown in Figure 4(D) and (E). CC was identified on CT-MPR coronal images. CC was originally defined by the calculation of HU values, whereas TBR was considered a non-calculated visual change. Accordingly, CC was defined as a visual change in our study. The overall high-density of the vertebral endplate (so-called sclerotic findings) was not considered as CC. Although CC was sometimes observed only on one side of the cage, and not necessarily on both, the two were not distinguished and were evaluated only for the presence of CC. CC evaluation was performed in the same way as that for TBR and scored as 0-2.
Vertebral Endplate Cyst
VEC, a cystic change of the vertebral endplate found in unstable interbodies, was used as an imaging reference that contrasted with osteointegration. VEC formations25,26 were evaluated on sagittal and coronal CT-MPR views. Furthermore, VEC formation was evaluated in the same way as TBR and scored from 0 to 2.
Cage Subsidence
We defined cage subsidence at 3 months postoperatively as cage entry into the vertebral endplate by 2 mm or more. Additionally, cage subsidence at 1 year was defined as the increased worsening of subsidence, or new cage entry into the vertebral endplate of >2 mm. Subsidence was assessed on CT-MPR images and scored on a scale of 0 to 2, similar to the scale for TBR. We found no cage dropout from the intervertebral space.
Clear Zone Around Pedicle Screw
We defined clear zones around pedicle screws (CZPSs) as translucent zones more than 2 mm wide around a pedicle screw. CZPSs were assessed on axial CT images. For each level, we counted the number of pedicle screws surrounded by such clear zones; the numbers ranged from 0 to 4.
Pseudarthrosis
Pseudarthrosis was considered to have occurred if any of the following findings was present on radiological examination one year after surgery: (1) range of motion of the local angle was >5°, 27 (2) the number of CZPSs was 4, or (3) CT images showed a vacuum sign between the vertebral bodies. In addition, the need for reoperation for these problems was also considered indicative of pseudarthrosis.
Statistical Analysis
Data were calculated as means ± standard deviations for continuous variables and as numbers and percentages for categorical variables. To analyze statistical data, we used R software, version 4.2.1 (The R Foundation, Vienna, Austria; http://www.R-project.org), for the Wilcoxon rank-sum test, Fisher’s exact test, and Pearson’s chi-square test. A P value of <.05 was considered statistically significant.
Results
Demographics by Cage Type.
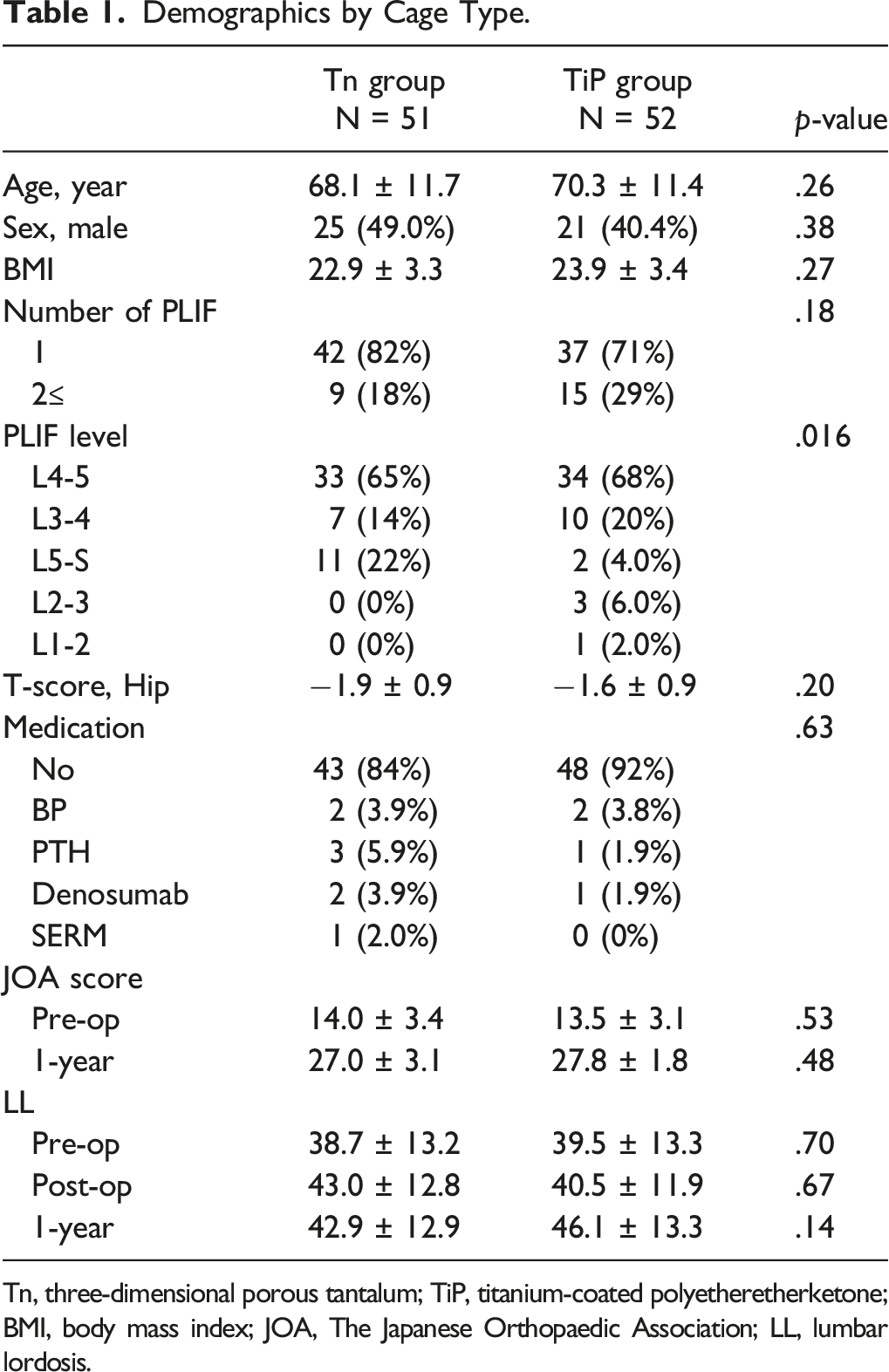
Tn, three-dimensional porous tantalum; TiP, titanium-coated polyetheretherketone; BMI, body mass index; JOA, The Japanese Orthopaedic Association; LL, lumbar lordosis.
Local Radiographic Findings.
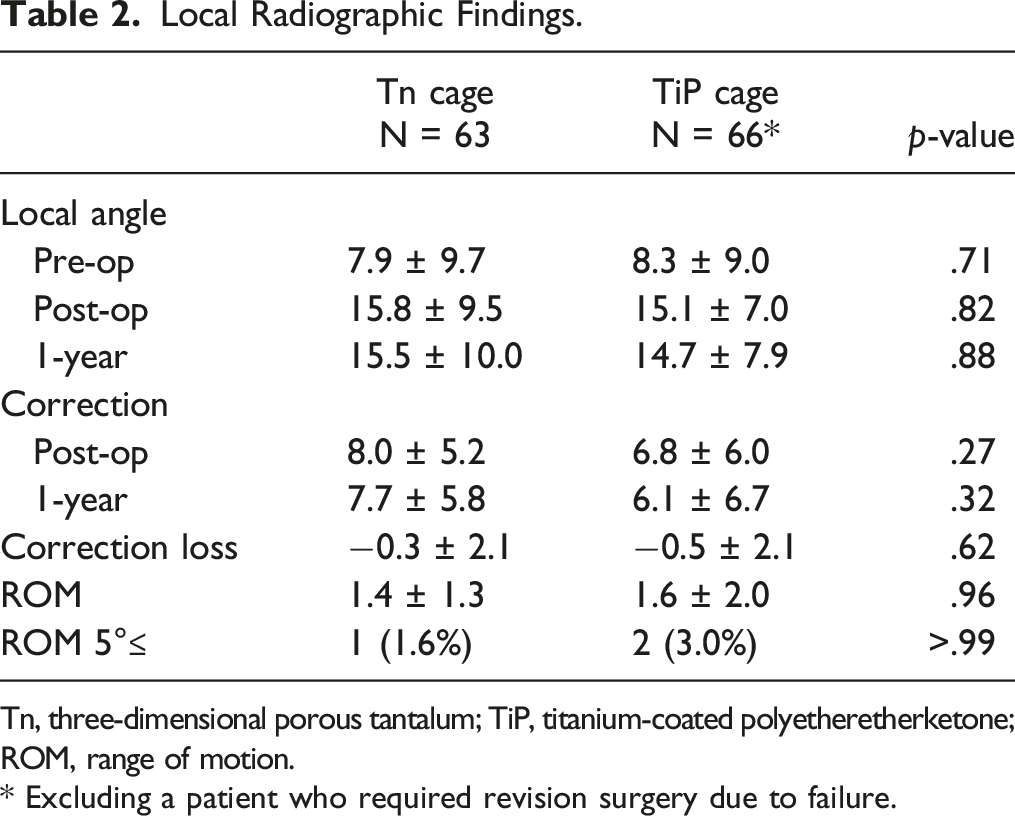
Tn, three-dimensional porous tantalum; TiP, titanium-coated polyetheretherketone; ROM, range of motion.
* Excluding a patient who required revision surgery due to failure.
Computed Tomography Findings.
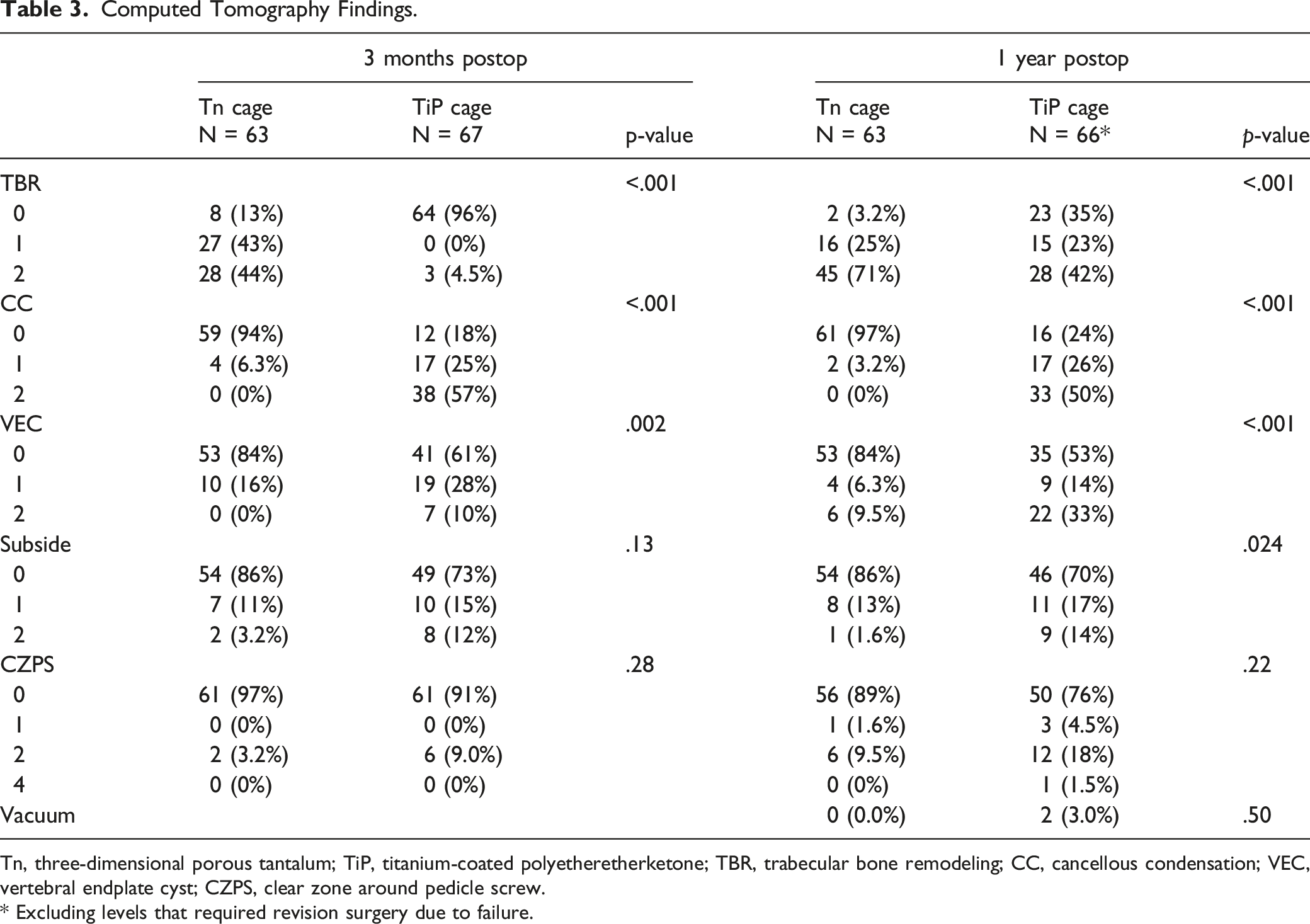
Tn, three-dimensional porous tantalum; TiP, titanium-coated polyetheretherketone; TBR, trabecular bone remodeling; CC, cancellous condensation; VEC, vertebral endplate cyst; CZPS, clear zone around pedicle screw.
* Excluding levels that required revision surgery due to failure.
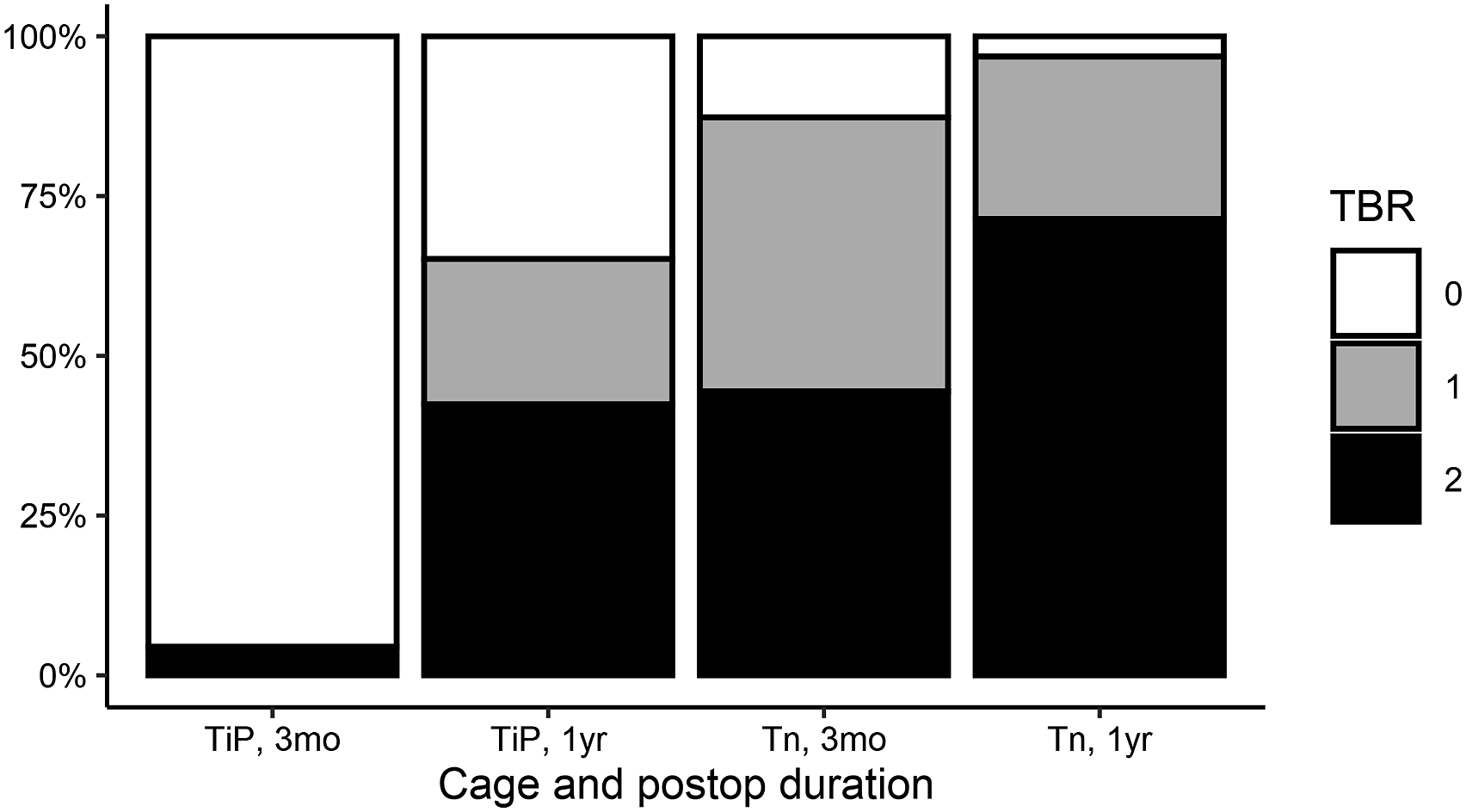
Positive rates of trabecular bone remodeling (TBR) by cage and duration. TiP, titanium-coated polyetheretherketone; Tn, three-dimensional porous tantalum.
We examined the CT images for TBR and CC 3 months and 1 year after surgery for each cage type. Most levels with Tn cages exhibited TBR but not CC both 3 months (81%) and 1 year (94%) after surgery; none exhibited CC but not TBR. Once present, TBR did not disappear, but CC occasionally disappeared. At one of the levels that exhibited no TBR both 3 months and 1 year after surgery, pseudarthrosis occurred. Although the TiP cages were characterized more often by the absence of TBR and the presence of CC 3 months after surgery (78%), the majority (59%) exhibited both 1 year later. In contrast to the Tn cages, none of the TiP cages exhibited TBR without CC 3 months after surgery, and only 6.1% did so 1 year later. Of the TiP cages that exhibited CC 3 months after surgery, 76% exhibited TBR 1 year later; in comparison, of those that did not exhibit CC, 9.1% exhibited TBR (P = .02). In two levels of TiP cages that exhibited no TBR both 3 months and 1 year after surgery, pseudarthrosis occurred, and for one level that exhibited no TBR 3 months after surgery, revision surgery was necessary because of device failure.
Discussion
Three months postoperatively, the majority of the Tn cages exhibited TBR, whereas almost all the TiP cages did not. The Tn cages predominantly exhibited TBR but not CC both 3 months and 1 year after surgery. Of the TiP cages, the majority exhibited CC but not TBR 3 months after surgery, but 1 year later, more exhibited both. Implant-related problems occurred at levels that exhibited neither TBR nor CC.
TBR is a finding suggestive of osteointegration and has been shown to be a possible radiological marker of good initial fixation after PLIF. 16 TBR can be observed on CT-MPR coronal images and is defined by the appearance of new oblique bone trabeculae from the cages. 16 The specific orientation of the extracellular matrix, such as collagen and biological apatite, are characteristic features of anisotropic bone microstructure and are important determinants of mechanical function and performance of bone under anisotropic stress. 28 Bone can exhibit appropriate mechanical function by developing material anisotropy in response to applied stress. 29 According to one study, subchondral bone sclerosis in osteoarthritis occurred as a result of stress concentration, caused both by an increased degree of preferential normal alignment of the biological apatite and by proliferation of bone tissue. 30 Part of intervertebral stress after PLIF is distributed to the instrumentation (pedicle screws and rods); the remaining stress is distributed to the cages. Proper orientation of extracellular matrix is important for the mechanical performance of bone, 31 and trabecular architecture is important for the mechanical properties of cancellous bone.29,32 Therefore, the extracellular matrix must be aligned in the same orientation in response to the stresses concentrated in the cage, which must produce the bone trabecular architecture that can be visualized on imaging tests. Thus, TBR is not just simple bone ongrowth or ingrowth, but a finding that suggests osteointegration.
Osteointegration after PLIF has been assessed on CT images with various methods. Makino et al 14 and Kashii et al 15 focused on vertebral HU values at the contact surface of the TiP cage. A high HU value was defined as vertebral CC and reflected favorable fixation of the cage and bone. We visually determined the CC as well as the TBR. TBR did not disappear during the year after appearance, whereas CC could disappear. The CC may have become unrecognizable because the TBR appeared consistent with the CC. However, in some cases, TBR did not appear, and CC disappeared. In fractures, the healing process of cancellous bone differs from that of shaft bone. 33 When cancellous bone is damaged, surrounding bone density increases temporarily because of rapid new bone formation; it subsequently decreases and returns to baseline. 34 This suggests that CC reflects a temporary hypertrophy of bone tissue as a result of bone growth, whereas TBR is a remodeled bone internal environment that reflects completed new load transfer.
The physical properties of porous tantalum include high porosity (75%–85%) and pore size in the range of 400-600 μm, which contribute to bone and soft tissue engraftment that result from the 3D internal space and high pore interconnectivity.10,12,35 In addition, porous tantalum has an elastic modulus and compressive strength similar to those of cancellous bone, which properly distributes load stress to adjacent bone tissue, thereby minimizing stress-shielding effects. 36 Furthermore, because clinically applied Tn cages do not have a window for bone grafting, no point of stress alteration exists on the surface where the cage and vertebral endplate come into contact. In contrast, TiP cages have a window, so not only is contact between titanium and vertebral bone limited to the outer periphery on the surface where the cage and endplate contact but also the stress that is transferred through the cage changes between the periphery and the center window. However, the Tn cage is homogeneous throughout, which approximates the structure of cancellous bone.
Because the Tn cage has such a uniform cancellous bone–like structure, it is possible that once bone ongrowth is achieved above and below, a state approximating cancellous bone bridging is achieved. Not only was TBR more prevalent with Tn cages than with TiP cages soon after surgery (3 months postoperatively) but also the number of VECs was significantly lower. In contrast, with TiP cages, CC appeared first as a result of bone growth on the cage surface titanium,37,38 followed by the appearance of TBR with the progression of load transfer through bone fusion. With TiP cages, TBR increased over time from 3 months to 1 year. However, despite the expectation that bone ongrowth would occur with Tn cages, 12 CC did not appear; TBR appeared from the beginning instead. This may be a result of the immediate initiation of bone ingrowth 12 in the Tn cage. Although the TiP cage has a surface titanium coating that promotes bone ongrowth, bone ingrowth into the TiP cage is limited because the titanium coating is 100-200 μm thick and bone cannot enter solid polyetheretherketone. Therefore, to achieve intervertebral stability at the location of the TiP cage, bone bridging by grafted bone must occur. In contrast, the Tn cage level may provide intervertebral stability with bone growth on and slightly into the cage.
The materials, shapes, and surface structures of Tn cages differ from those of TiP cages. Although porous tantalum is a relatively new biomaterial, it has osteoconductive and possibly osteoinductive properties and one of the highest coefficients of friction of any biomaterial.9,39 In contrast, titanium has been used since the 1960s because of its excellent combination of strength, low modulus, and biocompatibility, and because it bonds to bone. 17 However, titanium implants are usually much stiffer than natural bone, which can cause implant failure. 40 TiP combines bone-bonding properties with a low modulus. Therefore, both materials are attractive as biomaterials. More recently, surface porosity was suggested to influence bone ongrowth more than material differences do.2,40 Therefore, the porosity of the Tn cage surface may be more favorable for osteointegration than the rough surface with titanium coating of the TiP cage, which might have been associated with early bone integration after spinal interbody fusion surgery in this study.
Because the purpose of using devices in the treatment of spinal disease is the maintenance and rapid recovery of spinal function, interbody cages should facilitate rapid recovery of spinal function without secondary surgery to harvest autogenous bone. 41 The Tn cage not only meets this goal but also can potentially enable earlier osteointegration than does the TiP cage. Such osteointegration may differ from the bone continuity –solid bony fusion– obtained with conventional interbody cages, but it can indicate intervertebral stability. This finding may change the goal of PLIF from “bony fusion” to “intervertebral osteointegration.”
This study had several limitations. First, it was not a randomized controlled study. Second, the shapes of the different TiP cages varied. Third, the sample size was relatively small. Fourth, because the current study only examined imaging findings, we have not yet identified any association with intrinsically important clinical outcomes such as patient-reported outcome measures or revision rates. Finally, findings such as TBR and CC could be subjective. Despite these limitations, this study revealed information about the development of TBR and CC, which indicated that differences in cages can lead to differences in osteointegration after PLIF and that these imaging findings may have new potential in the evaluation of osteointegration after PLIF.
Conclusion
We examined intervertebral osteointegration after PLIF for cages of two different materials and designs. After 3 months, the majority of Tn cages exhibited TBR, the finding suggestive of osteointegration. TiP cages exhibited mainly CC but not TBR 3 months after PLIF; 1 year later, the majority exhibited TBR. The 3D porous tantalum cages may enable intervertebral stability that is comparable to bony fusion soon after surgery.
Footnotes
Declaration of Conflicting Interests
The authors have no conflicts of interest to declare concerning the materials or methods used in this study or the findings specified in this paper.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Institutional Review Board of Nagoya Spine Group (2005-0354-5) and all involved institutions were part of the Nagoya Spine Group.
