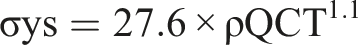Abstract
Study Design
Retrospective study.
Objective
We found that inter-vertebral signal concordance index (ISCI), as a novel factor, was significantly higher in the osteoporotic vertebral compressed fracture (OVCF) patients than non-OVCF patients in our previous retrospective study. In this study, we aim to validate the association between ISCI and OVCF using finite element analysis (FEA).
Methods
From the non-fracture cohort of our previous diagnostic study, 18 female patients (50-75 years) with degenerative lumbar disease were selected. To minimize confounding factors affecting bone strength, paired patients were matched under criteria: 1. The same femoral neck T-score; 2. Age caliper of 4 years. FEA was adopted to predict vertebral bone strength under 500N compression and failure load for the paired patients.
Results
Among three ISCI subtypes (T1, T2, STIR), only STIR-ISCI showed significant biomechanical differences: Compared to lower STIR-ISCI group, the higher STIR-ISCI group exhibited increased vertebral deformation (2.62 ± 0.72 mm vs 1.24 ± 0.55 mm, P = .002) and increased stress distribution (10.92 ± 1.98 MP vs 6.49 ± 2.87 MP, P = .002) under 500N load. Moreover, the yield strength in the higher STIR-ISCI group was lower (4443.11 ± 8.99 N vs 4467.67 ± 9.66, P < .001). Consequently, the deformation at yield strength was higher in the higher STIR-ISCI group (21.25 ± 4.73 mm vs 15.55 ± 2.55 mm, P = .027). No differences were observed in the T1-ISCI or T2-ISCI subgroups.
Conclusions
STIR-ISCI is an independent risk factor for OVCF, strongly associated with compromised vertebral biomechanics independent of bone mineral density. This magnetic resonance imaging-derived biomarker offers a promising tool for clinical fracture risk stratification.
Keywords
Introduction
Osteoporosis is a systemic skeletal disorder characterized by reduced bone mass and microarchitectural deterioration, which compromises bone strength and increases the susceptibility to fragility fractures, particularly in the spine and hip regions.1,2 Osteoporosis is a predominant global contributor to fractures, with vertebral fractures comprising a substantial portion. 3 Osteoporotic vertebral compression fracture (OVCF) is associated with severe outcomes, including chronic pain, spinal deformity, reduced mobility, and notably increased mortality rates, particularly in the elderly population. 4 A significant challenge in managing OVCF is that the majority are asymptomatic (“silent fractures”), frequently resulting in delayed diagnosis and intervention until considerable structural damage has ensued.5,6
Current risk assessment predominantly relies on bone mineral density (BMD) measurements obtained through dual-energy X-ray absorptiometry (DEXA). 7 However, BMD alone accounts for typically below 30% of fracture risk, as it does not encompass critical determinants of bone strength beyond mass, such as the heterogeneity of trabecular microarchitecture.8,9 This diagnostic limitation results in the under-detection of high-risk patients, underscoring the urgent need for additional biomarkers to accurately quantify fracture susceptibility.
Emerging magnetic resonance imaging (MRI) biomarkers, such as proton density fat fraction (PDFF) and vertebral bone quality (VBQ) scores, present promising alternatives for osteoporosis assessment.10,11 PDFF measures bone marrow adiposity using chemical shift imaging sequences and has shown significant correlations with the risk of OVCF, independent of BMD. 12 Similarly, VBQ, derived from T1-weighted signal intensity ratios between lumbar vertebrae and cerebrospinal fluid, is an effective predictor of incident OVCF, offering an alternative or complementary approach to DEXA-derived BMD assessment. 13 Despite their diagnostic value, both techniques encounter significant standardization challenges: PDFF demonstrates considerable variability between scanners due to differences in field strength and coil configuration, whereas VBQ exhibits noticeable discrepancies across platforms owing to inconsistencies in sequence parameters.14,15 These limitations motivate the exploration of alternative MRI-based biomarkers with potentially improved consistency.
Drawing inspiration from entropy theory, which posits that biological disorder is reflected in increased signal heterogeneity, we developed the Inter-Vertebral Signal Concordance Index (ISCI) to quantify marrow signal discordance across lumbar vertebrae (L1–L4) using standard MRI sequences (Supplemental Methods). This innovative metric captures progressive microstructural disorganization in osteoporotic bone by assessing deviations from uniformity. Importantly, our retrospective case-control study demonstrated significantly elevated ISCI values in the fracture cohort (Supplemental Table 1). Multivariate regression analysis confirmed Short Tau Inversion Recovery (STIR)-ISCI as an independent risk factor (Supplemental Table 2), indicating its direct association with fracture susceptibility. However, while these findings establish a clinical correlation, further research is needed to validate these results and explore their broader implications. They are unable to determine whether ISCI-mediated heterogeneity directly affects biomechanical integrity independently of traditional factors such as bone density. To resolve this mechanistic ambiguity, it is necessary to isolate the causal impact of ISCI on vertebral load-bearing capacity through controlled validation that eliminates confounding variables such as age and BMD.
Finite element analysis (FEA) offers a robust computational method for validating the biomechanical impact of ISCI by accurately simulating vertebral responses under physiological loads. 16 Unlike clinical imaging, FEA quantifies critical mechanical parameters including vertebral deformation, stress distribution, and yield strength through patient-specific modeling of bone geometry and material properties derived from quantitative computed tomography (CT). 17 This approach enables direct assessment of fracture susceptibility independent of density metrics. FEA-predicted yield strength demonstrates a strong correlation with experimental bone failure loads in validation studies and suggests enhanced predictive capability for fracture risk beyond conventional BMD measurements.18,19
In this study, we employed FEA within a rigorously matched cohort design to investigate the biomechanical effects of ISCI variation. By pairing subjects with identical femoral neck T-scores and age differences of ≤4 years, we effectively isolated the biomechanical impact of ISCI variation while controlling for major determinants of bone strength. This methodological approach establishes a causal relationship between elevated ISCI and reduced vertebral load-bearing capacity, addressing the mechanistic uncertainties left by previous associative studies.
The study aims to determine whether elevated ISCI, particularly as derived from STIR imaging, is associated with compromised vertebral bone strength independent of BMD. Utilizing FEA in subjects matched by femoral neck T-scores and age, we assess whether higher STIR-ISCI is correlated with increased vertebral deformation and stress under 500 N compression, as well as decreased yield strength, compared to matched low-ISCI controls. This mechanistic validation elucidates the role of ISCI as a BMD-independent risk factor for OVCF.
Materials and Methods
Selecting Study Population
This study received approval from our hospital (Approval No. 2024-255-01), with a waiver of informed consent granted due to its observational design using anonymized imaging data. All procedures strictly adhered to the principles of the Declaration of Helsinki. Patients diagnosed with degenerative lumbar disease underwent screening between January 2022 and July 2024.
The inclusion criteria for this study were as follows: (1) Female patients (50-75 years); (2) Availability of complete lumbar CT scans; (3) Availability of hip DEXA scan; (4) Availability of lumbar MRI scans including T1-weighted, T2-weighted, and STIR sequences.
The exclusion criteria for this study were as follows: (1) Presence of scoliosis with a Cobb angle greater than 10°, spinal tumors or infections, and previous spinal instrumentation; (2) Inadequate CT scan specifications: CT scans with slice thickness not equal to 0.625 mm or 1 mm; (3) Lack of a CT scan performed within 1 month (±1 month) of the MRI scan.
Participants were drawn from the non-fractured cohort of our prior VBQ study, which included patients with degenerative lumbar disease.
20
From this database, 42 individuals met current eligibility criteria. Subsequently, 9 matched pairs (n = 18) were selected through novel triple-stratified matching (identical femoral neck T-scores, age ≤4 years, CT voltage ≤20 kV) (Figure 1). This meticulous design aimed to isolate the biomechanical impact of ISCI by controlling for major determinants of bone strength, including BMD, aging, and scan parameters. The standardized workflow for ISCI quantification is fully documented in Supplemental Methods and the schematic diagram of ROI selection is displayed in Figure 2. Flow Chart of Patient Selection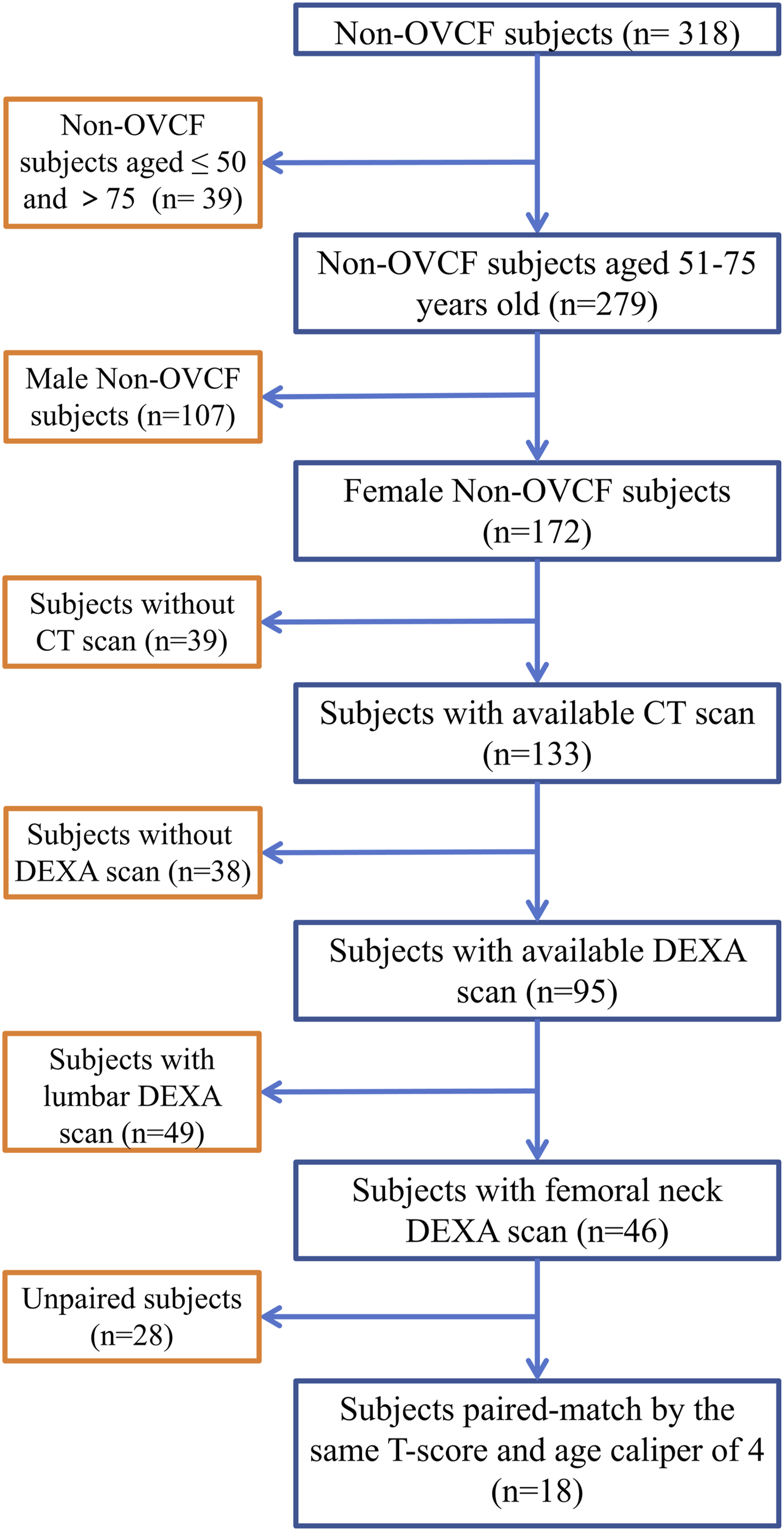
Quantitative Assessment of Vertebral Bone Strength via Finite Element Modeling
Lumbar CT scans were conducted using Toshiba Medical Systems (Tokyo, Japan) at voltages of 100, 120, or 140 kV with automatic tube current modulation, producing isotropic voxels measuring 0.98 × 0.98 × 1.00 mm3 and slice thicknesses of either 0.625 mm or 1 mm. The DICOM data underwent a biomechanical processing workflow, where the L1–L5 vertebrae were segmented using Mimics 21.0 (Materialise, Belgium) to create initial 3D models. The STL files were exported to Geomagic Wrap 2021 (3D Systems, USA) for surface optimization and mesh refinement. Subsequently, the optimized individual vertebral models were imported into SolidWorks (Dassault Systèmes, France) and assembled into a complete L1-L5 segment. Schematic Diagrams of the ROI Selection. The Mid-Sagittal Slice of Non-Contrast Lumbosacral Spinal MRI was Chosen to Obtain Vertebral SI. SIs Were Obtained From (a) T1-Weighted, (b) T2-Weighted, and (C) STIR-Weighted Sequences to Calculate T1-ISCI, T2-ISCI, and STIR-ISCI, Respectively. ROI, Region of Interest; MRI, Magnetic Resonance Imaging; SI, Signal Intensity; STIR, Short Tau Inversion Recovery; ISCI, Inter-Vertebral Signal Concordance Index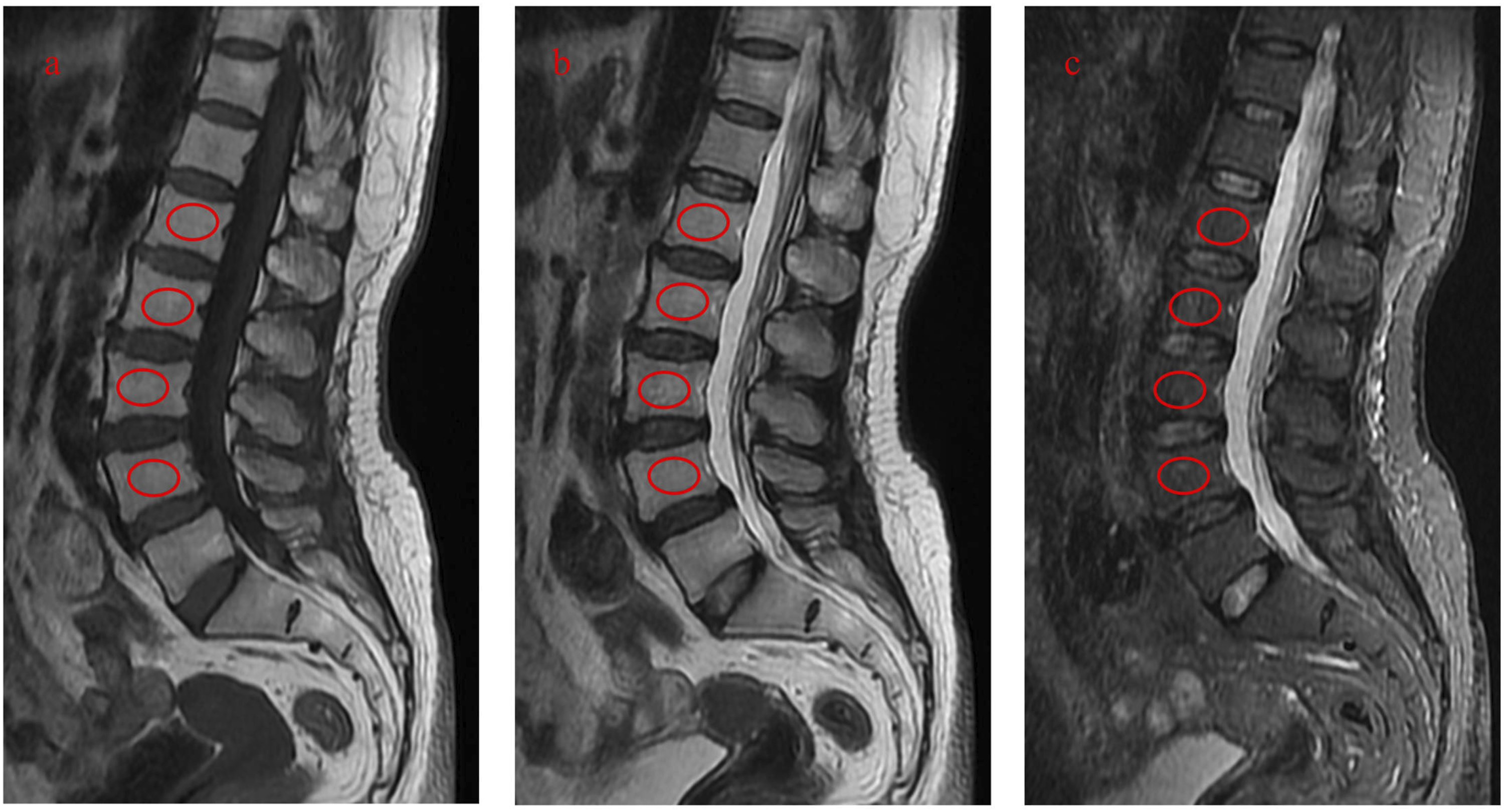
Finite Element Analysis
The assembled model was imported into Ansys software. Model incorporated intervertebral discs as simplified homogeneous structures, while excluding all other soft tissues (ligaments/muscles). A finite element mesh was generated within Ansys. Crucially, material properties for the bone were assigned based on QCT-derived Bone Mineral Density (QCT-BMD, ρQCT in mg/cm3). This involved converting CT Hounsfield Units (HU) to ρQCT using a linear relationship calibrated with a CT phantom, as established in prior literature.
21
This approach was chosen due to the demonstrated superiority of QCT-BMD over DEXA-BMD for vertebral biomechanical analysis.
21
The relationships between yield stress (σys, MPa) vs ρQCT for the vertebral bone of Chinese were determined based on previous study.
21
The boundary conditions entailed coupling the superior surface nodes of the L1 vertebra and the inferior surface nodes of the L5 vertebra to distinct reference points located at the respective superior and inferior endplates. A compressive load of 500 N was applied perpendicularly to the superior endplate of L1 through the upper reference point, while the lower reference point was fully constrained. A nonlinear analysis utilizing a linear elastic, perfectly plastic material model was performed to calculate vertebral deformations under the applied load. The maximum yield strength for the L1-L5 segment, along with the corresponding stresses, was subsequently determined based on the material’s yield stress criterion.
Statistical Analysis
The normality of continuous data was evaluated using the Shapiro-Wilk test. For variables that were normally distributed and exhibited homogeneous variance, paired t-tests were employed to compare differences between matched groups. For variables that were not normally distributed, Wilcoxon signed-rank tests were utilized. All statistical analyses were conducted using SPSS 26.0 (Chicago, IL), with statistical significance defined as P < .05, unless otherwise indicated.
Results
Baseline Characteristics
Baseline Characteristics of Subjects
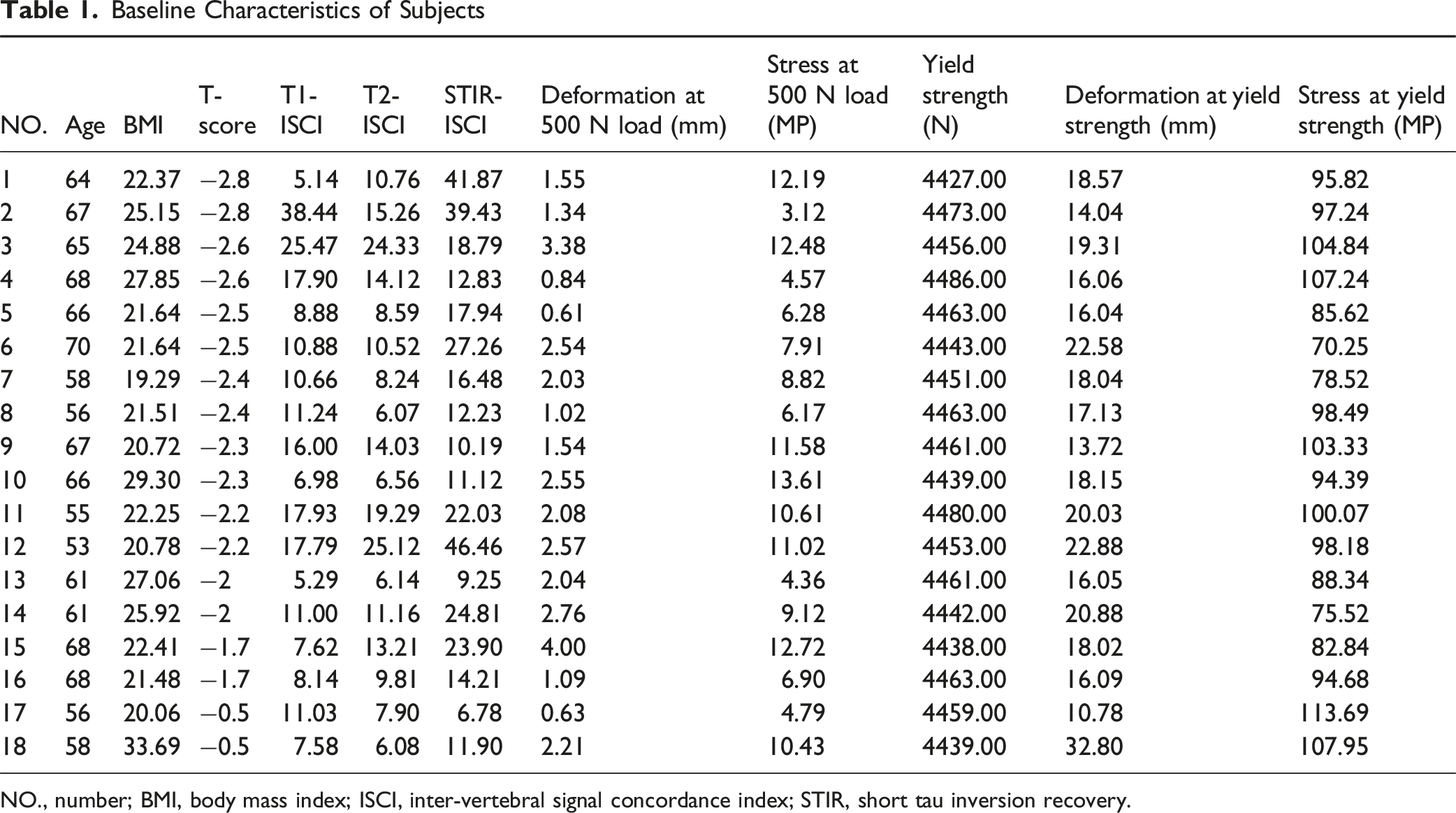
NO., number; BMI, body mass index; ISCI, inter-vertebral signal concordance index; STIR, short tau inversion recovery.
The uniformity in baseline parameters confirms the suitability of this cohort for isolating the biomechanical impact of ISCI, independent of BMD and age effects.
Subgroup Analysis by MRI Sequence
To assess the predictive utility of ISCI derived from various MRI sequences (T1-weighted, T2-weighted, and STIR) for OVCF risk, the paired subgroups were categorized into higher and lower ISCI groups based on relative values within each matched pair.
T1-ISCI Subgroups Analysis by MRI Sequence
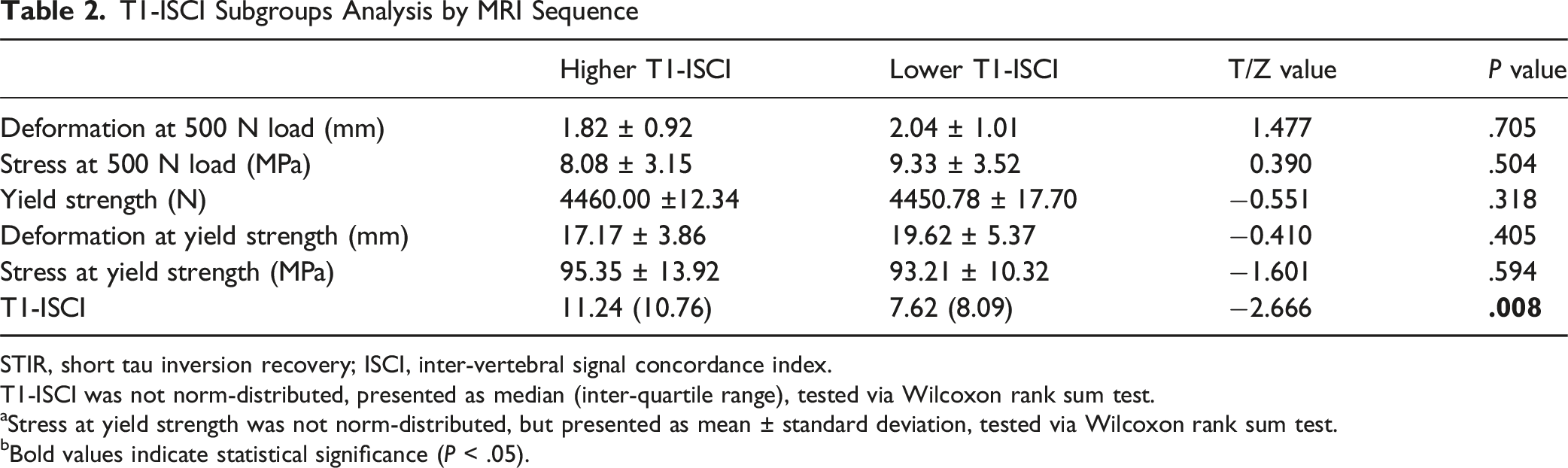
STIR, short tau inversion recovery; ISCI, inter-vertebral signal concordance index.
T1-ISCI was not norm-distributed, presented as median (inter-quartile range), tested via Wilcoxon rank sum test.
aStress at yield strength was not norm-distributed, but presented as mean ± standard deviation, tested via Wilcoxon rank sum test.
bBold values indicate statistical significance (P < .05).
T2-ISCI Subgroups Analysis by MRI Sequence
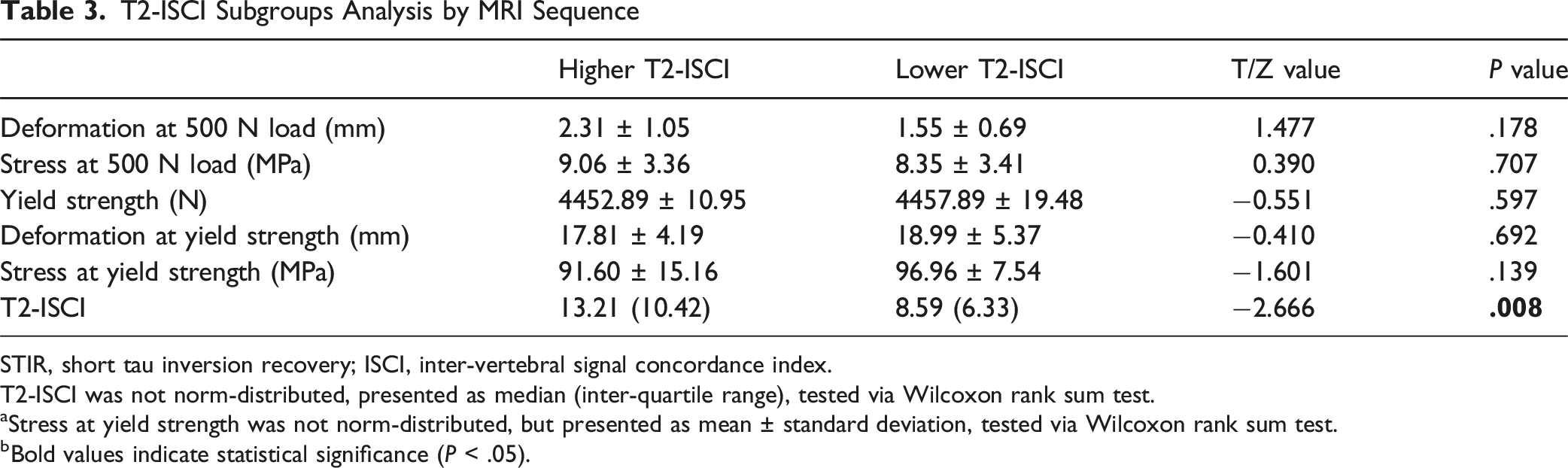
STIR, short tau inversion recovery; ISCI, inter-vertebral signal concordance index.
T2-ISCI was not norm-distributed, presented as median (inter-quartile range), tested via Wilcoxon rank sum test.
aStress at yield strength was not norm-distributed, but presented as mean ± standard deviation, tested via Wilcoxon rank sum test.
bBold values indicate statistical significance (P < .05).
STIR-ISCI Subgroups Analysis by MRI Sequence
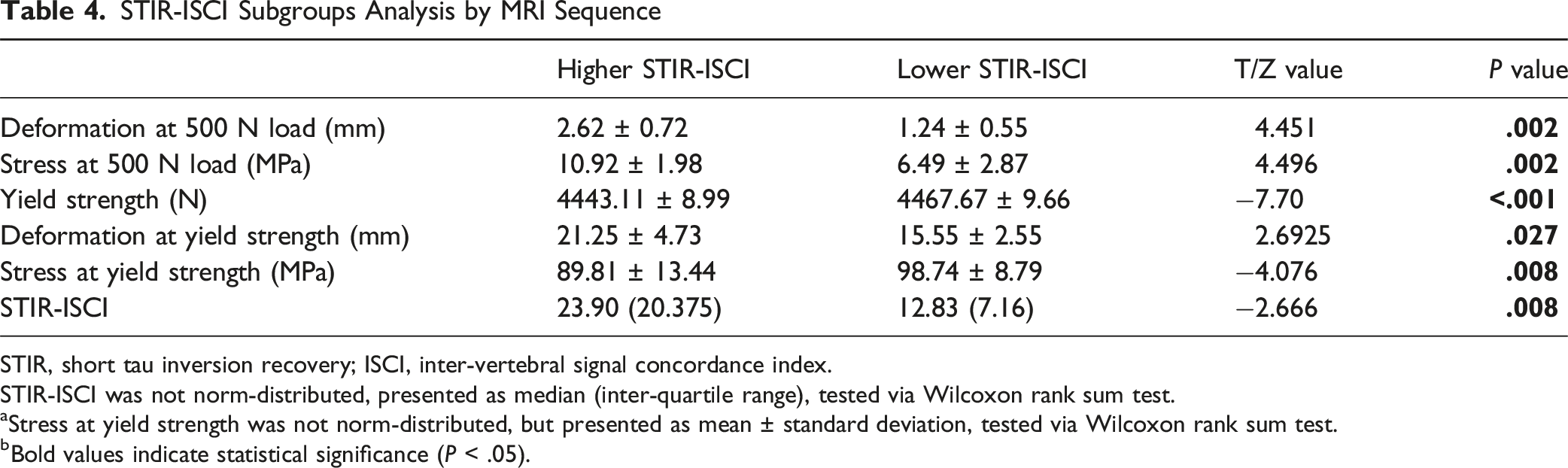
STIR, short tau inversion recovery; ISCI, inter-vertebral signal concordance index.
STIR-ISCI was not norm-distributed, presented as median (inter-quartile range), tested via Wilcoxon rank sum test.
aStress at yield strength was not norm-distributed, but presented as mean ± standard deviation, tested via Wilcoxon rank sum test.
bBold values indicate statistical significance (P < .05).
The sequence-specific findings of this study demonstrate that only the STIR-derived ISCI is associated with a reduction in vertebral load-bearing capacity, independent of BMD and age, thereby serving as a biomarker for susceptibility to OVCF, which was consistent with the retrospective findings (Supplemental Table 2).
Discussion
This research provides the first mechanistic validation that an elevated STIR-derived ISCI independently predicts compromised vertebral bone strength, unaffected by BMD confounding, as determined through finite element analysis in rigorously matched subjects. Under physiological loading conditions (refer to Table 4), subjects exhibiting elevated STIR-ISCI demonstrated significantly greater vertebral deformation (P = .002), increased von Mises stress (P = .002), and a notable reduction in yield strength (P < .001). These pronounced biomechanical changes substantiate ISCI as a direct biomarker for fracture susceptibility. Importantly, this biomechanical deterioration is attributed to signal heterogeneity-induced microstructural disorganization: elevated ISCI reflects discordant marrow signaling across vertebrae, leading to stress concentrations that accelerate microdamage accumulation and diminish load-bearing capacity. The lack of observable effects in the T1/T2-ISCI subgroups (P > .05, Tables 2 and 3) further highlights the distinctive sensitivity of STIR to pathophysiological changes that occur before fracture. By isolating the impact of ISCI through meticulous triple matching (identical T-scores, age caliper ≤4 years, voltage variance ≤20 kV), we demonstrate a causal relationship between marrow signal discordance and biomechanical failure that is independent of conventional determinants.
Considering the mechanistic link between elevated STIR-ISCI and biomechanical compromise, ISCI effectively addresses significant deficiencies in current bone strength assessment paradigms, especially in identifying microstructural deterioration independent of BMD.
While DEXA measures areal BMD, it fails to account for microstructural deterioration, which is precisely the pathway through which elevated ISCI diminishes bone strength. 22 Our findings indicate that ISCI can identify vulnerability in patients with normal T-scores, elucidating why a significant proportion of fragility fractures occur in individuals who are not classified as osteoporotic. By detecting signal heterogeneity indicative of trabecular disconnection, ISCI provides microarchitectural insights that are absent in density-based metrics. VBQ scores, while promising, are hindered by variability dependent on the equipment used. 23 The STIR-ISCI approach addresses these limitations: it converts routine lumbar MRI into a comprehensive biomarker capable of quantifying microstructural instability that predisposes to fractures, thereby resolving the standardization issues associated with fat fraction and VBQ methodologies.
Previous research has shown elevated VBQ-STIR levels in OVCF cohorts, likely indicating marrow fat infiltration similar to PDFF. 20 However, our identification of STIR-ISCI as a biomechanical predictor independent of BMD suggests different underlying mechanisms. Importantly, ISCI detects signal discrepancies that may be caused by non-adipose pathophysiological processes, including micro-edema, inflammatory activation, or clusters of trabecular micro-damage. Future studies should aim to elucidate the molecular mechanisms responsible for this heterogeneity to broaden therapeutic targets beyond lipid modulation. The biomechanical deterioration associated with elevated STIR-ISCI likely originates from its reflection of trabecular microarchitectural failure. STIR sequences detect free water protons in bone marrow with high sensitivity, enabling visualization of pre-fracture pathologies such as micro-edema, vascular leakage, and inflammatory infiltration.16,24 These pathologies are frequently concomitant with trabecular disconnection and micro-fractures, which impair load distribution and generate stress concentrations—consistent with our FEA data showing elevated von Mises stress (10.92 vs 6.49 MPa, P = .002) in high-STIR-ISCI subjects. Critically, this mechanism diverges from fat-based biomarkers: while PDFF and VBQ-STIR measure marrow adiposity,12,16 STIR-ISCI quantifies signal discordance arising from non-adipose microstructural disorganization. This explains the biomechanical irrelevance of T1/T2-ISCI (P > .05) and highlights STIR-ISCI as a unique biomarker of structural vulnerability. Besides, STIR-ISCI holds promise for augmenting fracture prediction models (eg, FRAX®) by quantifying microstructural deterioration. Whereas FRAX® incorporates clinical risk factors and BMD, STIR-ISCI detects early trabecular disconnection and microdamage—particularly crucial for the >50% of fragility fractures occurring in individuals with normal T-scores. Future large-scale studies should quantify STIR-ISCI’s incremental utility via net reclassification improvement (NRI) and decision curve analysis.
Despite the clear advantages of STIR-ISCI over traditional metrics in predicting biomechanical deterioration, our results should be considered within the context of certain methodological limitations. Chief among these is the relatively small cohort size (n = 9/9 matched pairs), which limits the statistical power to detect subtle biomechanical effects and prevents robust subgroup analyses across different vertebral levels (eg, comparing vulnerabilities of L1 vs L4). Future multi-center prospective studies are warranted to validate these preliminary findings. Through rigorous matching of identical T-scores and age differences of no more than 4 years, finite element modeling effectively isolates the impact of ISCI. However, we recognize the presence of unmeasured confounding variables, such as prolonged glucocorticoid use (exceeding 5 mg/day prednisone equivalents) and sedentary lifestyles, which may independently influence bone microarchitecture.24,25 To directly resolve the microstructural correlates of STIR-ISCI, future studies should integrate this biomarker with high-resolution micro-CT to quantify trabecular parameters (eg, number, spacing, and connectivity), validating its association with specific architectural deterioration. Besides, our study utilized standardized 3.0 T MRI systems with fixed STIR parameters, which controls scanner variability but limits generalizability. While STIR sequences exhibit lower cross-platform heterogeneity than quantitative MRI biomarkers, future work should validate ISCI in multi-center cohorts with diverse scanners. Finally, while our FEA results validate STIR-ISCI as a BMD-independent biomechanical risk factor, diagnostic threshold derivation requires larger studies. The current matched-pair design (n = 9 pairs) intentionally controlled confounding factors but lacks statistical power for cut-off analysis. Future large-scale studies are needed to define clinically applicable thresholds.
While this study mechanistically validates STIR-ISCI as a BMD-independent biomarker for vertebral fragility, determining its diagnostic threshold requires further investigation. Our matched-pair design prioritized isolating causal biomechanical effects over clinical cut-off derivation. The limited sample size (n = 9 pairs) and intentional control of BMD/age confounders preclude robust threshold analysis. Exploratory data from the retrospective cohort (Supplemental Table 1) showed significantly elevated STIR-ISCI in fracture patients (19.31 ± 14.98) vs controls (15.67 ± 12.58, P < .001), with overlapping distributions suggesting a potential risk transition zone around 16-20. Future large-scale studies are needed to define clinically applicable thresholds.
These factors are crucial as they could exacerbate signal heterogeneity beyond the pathophysiological scope of ISCI, potentially inflating effect sizes within high-ISCI subgroups. Future research endeavors with larger sample sizes should incorporate comprehensive profiling of medication and lifestyle factors to elucidate these interactions.
Despite the limitations of a modest sample size and the aforementioned unmeasured confounders, STIR-ISCI exhibits immediate clinical applicability as a biomarker independent of BMD, derived from routine lumbar MRI. By quantifying marrow signal heterogeneity on standard STIR sequences, it identifies high-risk individuals with normal T-scores, who account for over 50% of fragility fractures, thereby facilitating early preventive interventions without incurring additional imaging costs. 26
To fully realize the clinical utility of STIR-ISCI as a practical screening tool, the following key aspects of work are worth future exploration. Firstly, comprehensive multi-center validation involving significantly larger patient cohorts is crucial. This approach will robustly affirm its predictive capacity for fracture risk and mitigate the limitations associated with initial smaller studies, thereby ensuring that findings are reliable and generalizable across diverse populations. Integral to this validation is incorporating STIR-ISCI into established FRAX®/BMD models to rigorously quantify its added clinical predictive power through metrics like AUC improvement and Net Reclassification Index (NRI). Furthermore, standardization of MRI protocols across platforms is essential to ensure consistent, reliable results applicable in varied clinical settings. Secondly, it is essential to investigate its biomechanical applicability to other critical fracture sites. Building upon its foundation in assessing bone stress distribution—a fundamental determinant of fracture susceptibility—research must evaluate the performance of STIR-ISCI at high-risk locations such as the hip and wrist, beyond its initial application site.
These coordinated efforts aim to translate ISCI from a localized biomarker into a comprehensive diagnostic framework for assessing systemic fracture risk.
Conclusion
This study confirms that elevated STIR-derived ISCI independently predicts reduced vertebral bone strength, without BMD interference, using finite element analysis in well-matched subjects. The ISCI demonstrates potential as a supplementary tool for identifying individuals at high risk who exhibit normal DEXA results, thereby potentially enabling earlier intervention for osteoporotic vertebral fractures.
Supplemental Material
Supplemental Material - Inter-Vertebral Signal Concordance Index is a Risk Factor for Osteoporotic Vertebral Compressed Fracture: Validated by Finite Element Analysis
Supplemental Material for Inter-Vertebral Signal Concordance Index is a Risk Factor for Osteoporotic Vertebral Compressed Fracture: Validated by Finite Element Analysis by Chenhui Cai, Xianming Huang, Song Huang, Chao Tang, Xu Zhao, Zaoqing Zhang, Xuan Wen, Hao Qiu, Sizhen Yang, Ying Zhang, Tongwei Chu in Global Spine Journal
Footnotes
Author Contributions
All authors listed meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors, and all authors agree with the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Project of Chongqing Technology Innovation and Application Development Special Project (Grant No. CSTB2022TIAD-KPX0192), and Graduate Student Scientific Research Innovation Projects of Chongqing (Grant No. CYB23290).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during this study are available from the corresponding author upon reasonable request.
IRB Approval
IRB approval was obtained (2024-255-01).
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.