Abstract
Study Design
Retrospective propensity score–matched cohort study.
Objectives
To investigate whether the use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) is associated with a reduced risk of lumbar degenerative disease (LDD) and lumbar spine surgery in patients with type 2 diabetes mellitus (T2DM).
Methods
Data were obtained from the TriNetX Global Collaborative Network. Adult patients with T2DM initiating GLP-1 RA therapy were matched 1:1 with non-users based on demographics, comorbidities, and medication use. Patients with pre-existing LDD or prior lumbar spine surgery were excluded. Primary outcome was the incidence of LDD; secondary outcome was lumbar spine surgery. Outcomes were assessed at 3- and 5-year intervals, and odds ratios (ORs) with 95% confidence intervals (CIs) were calculated.
Results
A total of 196 435 matched pairs were analyzed. At 3 years, GLP-1 RA users had a lower incidence of LDD (11.9% vs 13.3%; OR 0.89, 95% CI: 0.87-0.91). At 5 years, this difference widened (14.6% vs 18.7%; OR 0.74, 95% CI: 0.73-0.74). Lumbar spine surgery rates were similar at 3 years (0.4% in both groups; OR 1.03, 95% CI: 0.93-1.14), but significantly lower in GLP-1 RA users at 5 years (0.5% vs 0.6%; OR 0.86, 95% CI: 0.78-0.92).
Conclusions
GLP-1 RA use is associated with a lower long-term risk of developing LDD and undergoing lumbar spine surgery in patients with T2DM. These findings support further investigation of GLP-1 RAs as potential disease-modifying agents in spinal health.
Introduction
Lumbar degenerative diseases (LDD), including lumbar spondylosis, intervertebral disc degeneration, spinal stenosis, and spondylolisthesis, are common musculoskeletal conditions that frequently affect individuals with type 2 diabetes mellitus (T2DM) and obesity.1,2 These degenerative changes often lead to chronic low back pain, radicular symptoms, and functional decline. In more advanced stages, surgical intervention such as lumbar decompression or spinal fusion may become necessary to alleviate symptoms and restore quality of life. 3
Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) are pharmacologic agents initially developed for glycemic control in patients with T2DM. 4 In recent years, their indications have expanded to include weight management, 5 given their effectiveness in promoting weight loss, improving metabolic profiles, and modulating inflammatory pathways.6,7 These systemic effects have raised interest in the potential musculoskeletal benefits of GLP-1 RAs beyond glycemic control.
Recent studies have shown that GLP-1 RA use may be associated with improved surgical outcomes in orthopedic and spine surgery, including reduced complication rates and enhanced postoperative recovery.8,9 However, whether GLP-1 RAs can reduce the development of LDD or the eventual need for lumbar spine surgery has not been fully established.
In this study, we used a large-scale, real-world dataset and a propensity score–matched cohort design to evaluate the association between GLP-1 RA use and the long-term risk of developing LDD and undergoing lumbar spine surgery in patients with T2DM.
Methods
We performed a retrospective cohort analysis using the TriNetX Global Collaborative Network, a global federated health research network. Adult patients with type 2 diabetes mellitus (T2DM) who initiated treatment with glucagon-like peptide-1 receptor agonists (GLP-1 RAs) were included and compared to T2DM patients not receiving GLP-1 RAs. Our dataset included patients enrolled between January 1, 2019 and January 1, 2020. GLP-1 receptor agonists were identified using the ICD ATC code A10BJ, which encompasses the following FDA-approved agents during the study period: Exenatide (A10BJ01), Liraglutide (A10BJ02), Lixisenatide (A10BJ03), Albiglutide (A10BJ04), Dulaglutide (A10BJ05), and Semaglutide (A10BJ06). All of these agents had FDA approval at the time, although some (eg, Albiglutide and Lixisenatide) have since been withdrawn from the U.S. market. In addition, Tirzepatide, a dual GLP-1/GIP agonist, was captured separately using its specific medication code (2601723).
Patients were excluded if they had a prior diagnosis of lumbar degenerative disease (LDD) or had undergone lumbar spine surgery before cohort entry. A comparator group of T2DM patients not receiving GLP-1 RAs was selected. Propensity score matching was performed at a 1:1 ratio to balance demographics, comorbidities, and medication use. The primary outcome was the development of LDD, identified using diagnostic codes for lumbar spondylosis, disc degeneration, spinal stenosis, or spondylolisthesis. The secondary outcome was lumbar spine surgery, defined by procedure codes for decompression or fusion. Outcomes were assessed at 3-year and 5-year follow-up intervals. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated to estimate risk differences between groups. We also performed stratified analyses on Semaglutide users, given its relatively recent introduction and rapidly increasing prescription rates.
This study was approved by the Institutional Review Board of National Cheng-Kung University Hospital (Approval No. B-ER-114-116; April 14, 2025). The requirement for informed consent was waived, as all data were de-identified and the study was deemed to pose minimal risk. All procedures involving human participants were conducted in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its subsequent amendments or comparable ethical guidelines.
Results
Patient Demographics and Comorbidities for GLP-1 RA Users and Control Cohorts Before and After Propensity Score Matching
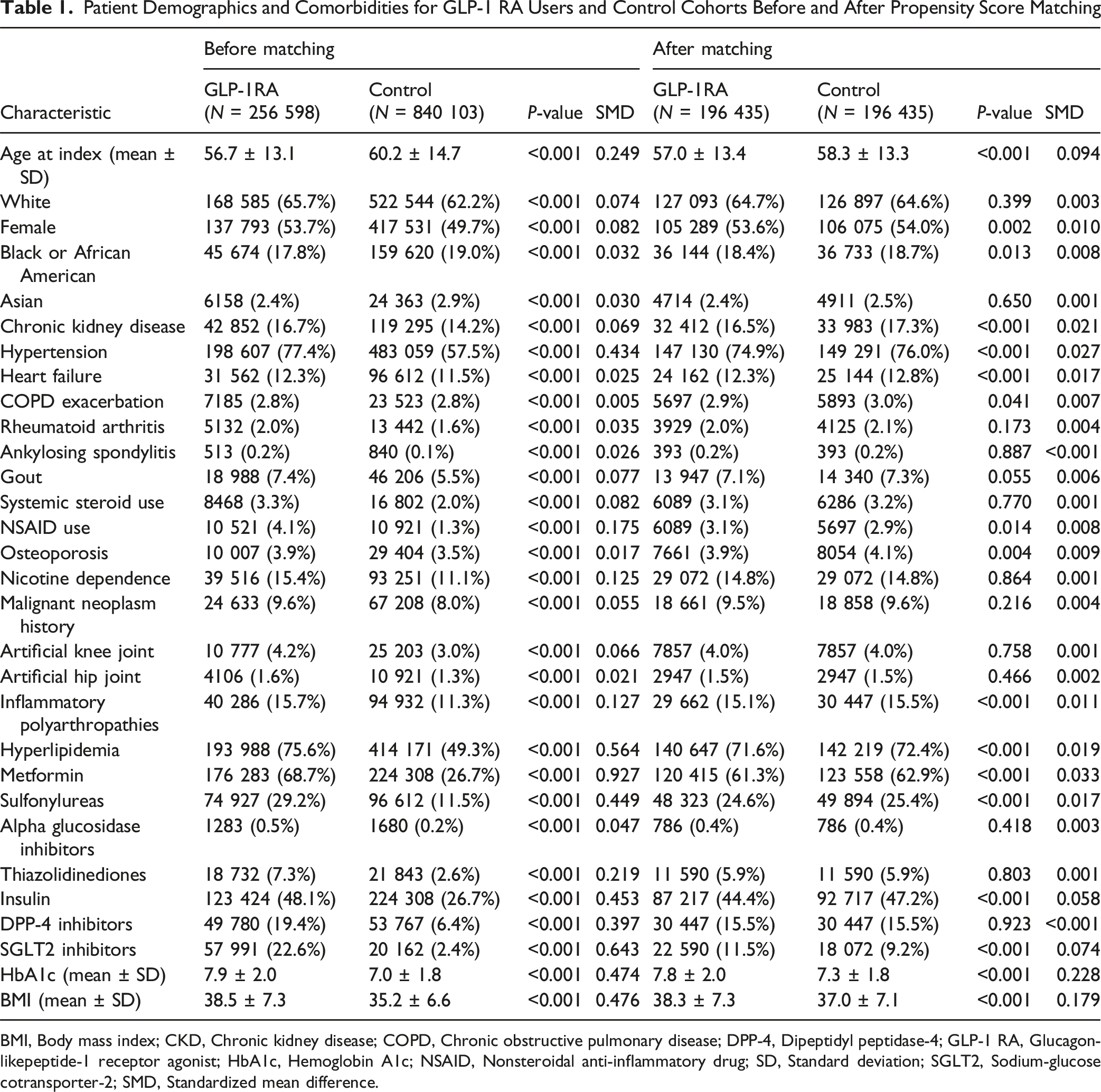
BMI, Body mass index; CKD, Chronic kidney disease; COPD, Chronic obstructive pulmonary disease; DPP-4, Dipeptidyl peptidase-4; GLP-1 RA, Glucagon-likepeptide-1 receptor agonist; HbA1c, Hemoglobin A1c; NSAID, Nonsteroidal anti-inflammatory drug; SD, Standard deviation; SGLT2, Sodium-glucose cotransporter-2; SMD, Standardized mean difference.
Long-Term Follow-Up of Risk of Development of LDD and Need for Surgery for LDD
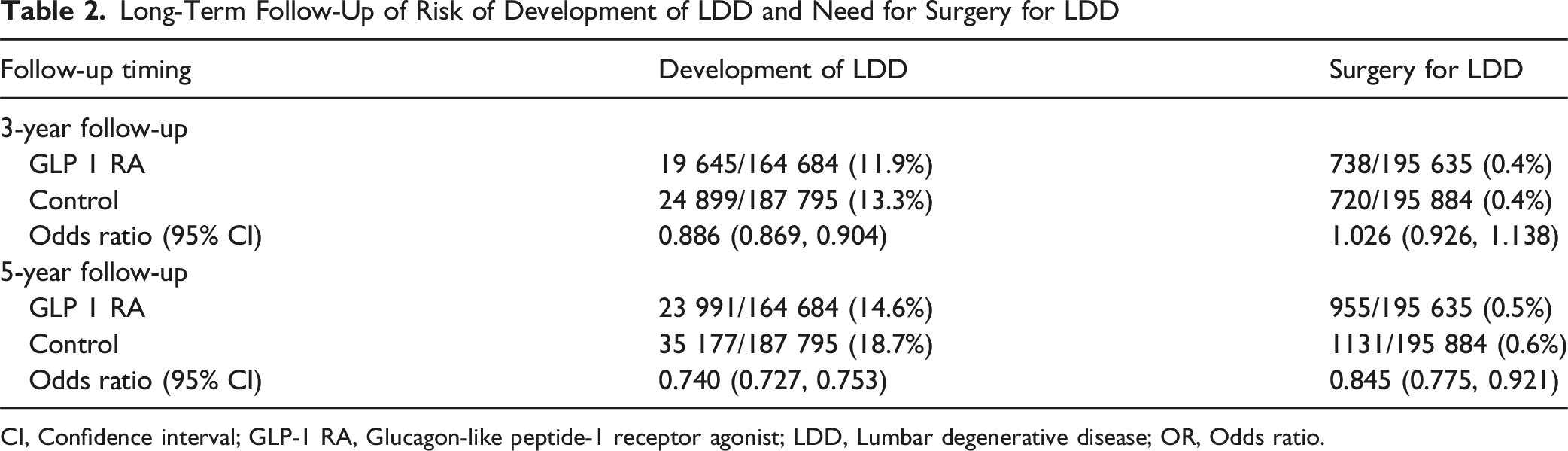
CI, Confidence interval; GLP-1 RA, Glucagon-like peptide-1 receptor agonist; LDD, Lumbar degenerative disease; OR, Odds ratio.
Discussion
In this large-scale, propensity score–matched retrospective cohort study, the use of glucagon-like peptide-1 receptor agonists (GLP-1 RAs) in patients with T2DM was associated with a significantly lower incidence of both developing lumbar degenerative disease (LDD) and undergoing lumbar spine surgery. These findings suggest that GLP-1 RAs may offer protective benefits at multiple stages of the degenerative spine disease continuum. At both 3- and 5-year follow-up intervals, GLP-1 RA users exhibited consistently lower rates of LDD diagnosis compared to non-users. Although the rate of lumbar spine surgery did not differ at 3 years, a significant reduction emerged at 5 years, suggesting that the musculoskeletal benefits of GLP-1 RAs may accumulate over time. These results are particularly meaningful given the high prevalence of spinal degeneration and the increasing surgical burden among patients with diabetes and obesity.
The mechanisms underlying this association may be multifactorial. First, GLP-1 RAs promote sustained weight loss, thereby reducing mechanical loading on the lumbar spine. 5 Obesity is a known contributor to disc degeneration and facet joint overload, and weight reduction has been associated with decreased low back pain and improved spine biomechanics.10,11 Second, GLP-1 RAs possess anti-inflammatory properties. Experimental and clinical studies have demonstrated that GLP-1 RAs reduce levels of pro-inflammatory cytokines such as TNF-α and IL-6, which are implicated in the pathogenesis of disc degeneration and nerve root irritation. 6 Third, improved glycemic control and insulin sensitivity may enhance tissue perfusion and metabolic health.4,7 Collectively, these pathways suggest a plausible disease-modifying role for GLP-1 RAs in degenerative spine conditions.
In addition to these systemic effects, GLP-1 RAs may exert direct protective actions on musculoskeletal tissues. GLP-1 receptors are expressed in chondrocytes and synovial membranes, where their activation reduces pro-inflammatory cytokines and matrix-degrading enzymes via NF-κB inhibition and PKA/CREB signaling.12-15 These effects attenuate cartilage degeneration and suggest relevance to intervertebral disc and facet joint preservation. Additionally, GLP-1Rs on osteoblasts and osteoclasts mediate bone remodeling; liraglutide promotes osteoblastogenesis and reduces resorption by modulating Runx2, Col1A1, and RANKL pathways.16,17 These mechanisms may support vertebral integrity and delay spinal instability, aligning with our observed reduction in lumbar spine surgery among GLP-1 RA users.
Previous research on GLP-1 RAs in spinal pathology has primarily focused on their perioperative use. Seddio et al reported a reduction in 90-day postoperative complications among semaglutide users. 9 Moreover, Ghali et al found no increase in adverse events and a significantly lower rate of pseudoarthrosis one year after lumbar fusion. 8 Addressing a different phase of care, our study demonstrates that GLP-1 RA use is associated with a reduced risk of both developing lumbar degenerative disease and progressing to surgical intervention in a large real-world cohort of patients with type 2 diabetes. These findings underscore the multifaceted role of GLP-1 RAs in spine care and highlight the need for further research to clarify their long-term musculoskeletal effects and therapeutic potential. In addition, beyond currently available single-agonist agents, emerging dual agonists (targeting GLP-1 and GIP receptors) and triple agonists (targeting GLP-1, GIP, and GCGR) hold the potential for enhanced metabolic and musculoskeletal benefits through broader mechanisms of action. Evaluating these drug classes separately in long-term matched studies would allow future trials to quantify whether distinct pharmacological profiles translate into meaningful differences in musculoskeletal degeneration.
Several limitations must be acknowledged. First, the retrospective nature of the study limits causal inference. While propensity score matching mitigates observed confounding, unmeasured variables such as lifestyle factors, physical activity, genetic predispositions, symptom severity, imaging findings, or adherence to medications may still influence the observed associations. Second, the identification of outcomes relied on administrative coding, which may not fully capture the clinical nuances of disease progression or surgical decision-making. Third, the study population consisted of patients with T2DM; thus, generalizability to non-diabetic individuals remains uncertain. Finally, the potential impact of unmeasured confounders highlights the need for future studies incorporating more granular clinical data, including lifestyle, genetic, and imaging information.
Conclusions
The use of GLP-1 RAs in patients with T2DM is associated with a reduced risk of LDD and lumbar surgical intervention over long-term follow-up. These findings suggest potential musculoskeletal benefits and support further investigation of GLP-1 RAs as disease-modifying agents in spinal health. Integrating metabolic therapies into early musculoskeletal care strategies may represent a promising approach to reducing the long-term burden of degenerative spine disease.
Supplemental Material
Supplemental Material - Association of GLP-1 Receptor Agonist Use With Risk of Lumbar Degenerative Disease and Spine Surgery in Patients With Type 2 Diabetes: A Propensity Score–matched Cohort Study
Supplemental Material for Association of GLP-1 Receptor Agonist Use With Risk of Lumbar Degenerative Disease and Spine Surgery in Patients With Type 2 Diabetes: A Propensity Score–matched Cohort Study by Yu Chang, Kuan-Yu Chi, Junmin Song, Chien-Min Chen, Hong-Min Lin in Global Spine Journal
Footnotes
Ethical Considerations
This study was approved by the National Cheng-Kung University Hospital institutional review board (approval number: B-ER-114-116; approval date: April 14, 2025).
Consent to Participate
Patient consent is waived, because the study is classified as minimal risk and all data used in the study are deidentified.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
