Abstract
Study Design
A Retrospective real-world study.
Objective
This study aimed to investigate whether paraspinal muscle degeneration (PMD) is a predictor of chronic low back pain (CLBP) in elderly patients with lumbar spondylolisthesis after posterior lumbar interbody fusion (PLIF).
Methods
A comparative cohort analysis was performed on 86 patients aged >60 with Meyerding grade I–II spondylolisthesis. Patients were stratified into PMD and non-PMD groups based on psoas major muscle index (PMI) and multifidus fat infiltration (MFI). Clinical outcomes were assessed using Visual Analog Scale (VAS) and Oswestry Disability Index (ODI) scores, alongside lumbosacral sagittal parameters including lumbar lordosis (LL), pelvic tilt (PT), pelvic incidence (PI), PI-LL (PL) mismatch. Propensity score matching for listhesis degree and osteoporosis yielded 30 patients per group for analysis. Multivariate analysis identified risk factors for CLBP at 2 years, and a machine learning model was developed as a clinically applicable predictive tool based on an expanded cohort of 649 patients.
Results
The PMD group demonstrated significantly worse outcomes at 2-year follow-up, with higher VAS (2.40 ± 1.55 vs 1.23 ± 1.48) and ODI scores (29.40 ± 6.83 vs 20.57 ± 6.38), and poorer sagittal alignment. PMI and MFI were identified as major risk factors for CLBP both pre- and postoperatively. The Random Forest Model achieved high accuracy (97.1%), sensitivity (95.9%), specificity (90.6%), and an F1 score (88.9%) in predicting postoperative CLBP.
Conclusions
PMD significantly impacts both preoperative symptom severity and postoperative recovery. Assessment of PMD provides valuable prognostic information, and the established predictive model can aid in managing surgical expectations and guiding rehabilitation.
Keywords
Introduction
Lumbar spondylolisthesis, frequently observed in the lower lumbar region, is a common condition that often leads to chronic low back pain (CLBP). In addition to back pain, some patients experience lower limb discomfort and nerve dysfunction, which significantly reduce their quality of life. Epidemiological studies estimate the prevalence of lumbar spondylolisthesis in the general population to be approximately 3%-6%, with a higher occurrence in women compared to men. 1 Furthermore, as the population ages, the prevalence of this condition is expected to rise. 2 Lumbar fusion surgery is the most commonly used method for treating lumbar spondylolisthesis. However, many studies have reported that some patients still experience CLBP after surgery, and the reasons may be diverse.3-5
Sarcopenia refers to the age-related, progressive, and widespread loss of skeletal muscle mass, strength, and function. 6 Epidemiological studies indicate that the prevalence of sarcopenia increases with age, affecting approximately 10%-40% of individuals aged 60 and older. 7 This condition not only impairs daily life and mobility in older adults but is also linked to numerous adverse health outcomes, including a heightened risk of falls, fractures, hospitalization, and mortality. 8 In the lumbar spine, sarcopenia primarily manifests as paraspinal muscle degeneration (PMD), also referred to as atrophy. PMD is characterized by both a reduction in the volume of the lumbar paraspinal muscles—typically assessed through measurements of muscle area or relative area—and a decline in muscle quality, often evaluated by the extent of fat infiltration. Recent studies have highlighted the high prevalence of sarcopenia and PMD in lumbar spine diseases and their significant impact on surgical outcomes, drawing increasing attention from researchers and clinicians.9-13 In particular, the significance of PMD in CLBP after lumbar spine surgery has been explored by some studies. 14
At present, the research on the impact of PMD on lumbar spondylolisthesis is limited. Can PMD increase the severity of symptoms? And patient-reported outcomes? In particular, will it increase the occurrence of CLBP after surgery? Understanding these issues can enable clinical staff to assess potential treatment risks in advance and provide more accurate predictive management and intervention. Machine learning algorithms are powerful tools for predicting clinical outcomes based on big data analysis.15,16 In our previous research, there have been successful experiences in establishing machine learning models for predicting short-term residual pain after lumbar fusion surgery and predicting the sinking of the fusion device.17,18
Based on the above, this study aims to explore the role of PMD in patients with lumbar spondylolisthesis, and establish a clinically practical predictive model. Understanding these influences provides valuable insights into preoperative planning and postoperative management strategies, and further assist in the formulation of individualized treatment plans and ultimately improve the prognosis of patients.
Methods
Ethical Statement
This study was approved by the institutional ethics committee (Lot No. 20250120054246102), and all patients provided written informed consent. The study adhered to the ethical principles outlined in the Declaration of Helsinki for human research, ensuring the confidentiality and privacy of patient information.
Study Design
This retrospective real-world study consists of 2 parts. The first part involves a cohort comparative analysis of 86 elderly patients with low lumbar spondylolisthesis who were hospitalized at our center between January 2020 and June 2021. This analysis aims to evaluate the effects of PMD on low back pain, functional outcomes, and sagittal balance before and after posterior lumbar interbody fusion (PLIF) surgery. The second part expands the sample size by including 649 elderly patients with lumbar spondylolisthesis who underwent PLIF at our center between January 2020 and June 2022. With an expanded dataset, this phase applies machine learning models for multi-class prediction to address the limitations of a small sample size, validate the preliminary findings, identify key predictors of low back pain, and enhance the accuracy of the prediction model through self-training.
Inclusion Criteria
For the first part of the study, the inclusion criteria were as follows: patients diagnosed with mild to moderate (Meyerding grade I–II) 19 lower lumbar spondylolisthesis (L4 or L5) who underwent single-level PLIF; age over 60 years; and a minimum postoperative follow-up period of 24 months. Patients with incomplete statistical data or a history of other lumbar surgeries were excluded.
In the second part, the machine learning dataset still adopts retrospective, non-random cases obtained by full sampling according to the screening conditions. The inclusion criteria were broadened to encompass spondylolisthesis at any lumbar segment (L1–L5), and the surgical segment was no longer restricted to a single level. All other criteria remained unchanged. The age should be over 60 years, the endpoints should be complete and the follow-up period should be more than 24 months. They are largely comparable to the first cohort.
Parameters
Demographic data included age, gender, body mass index (BMI), bone mineral density (BMD), and a history of chronic diseases, like hypertension, diabetes, coronary heart disease (CHD), and stroke. Surgical data encompassed the spondylolisthesis level, degree of spondylolisthesis, presence of spondyloschisis, and length of postoperative hospital stay.
Preoperative lumbar MRI was used to evaluate PMD, with the psoas major muscle index (PMI) and multifidus fat infiltration (MFI) serving as key assessment metrics. PMI (cm2/m2) was calculated as the total psoas major muscle area (cm2) divided by height squared (m2) at the L3 level, providing an index of muscle mass in the para-lumbar region (Figure 1). This evaluation is based on an authoritative prospective study endorsed by the German Spine Society.
13
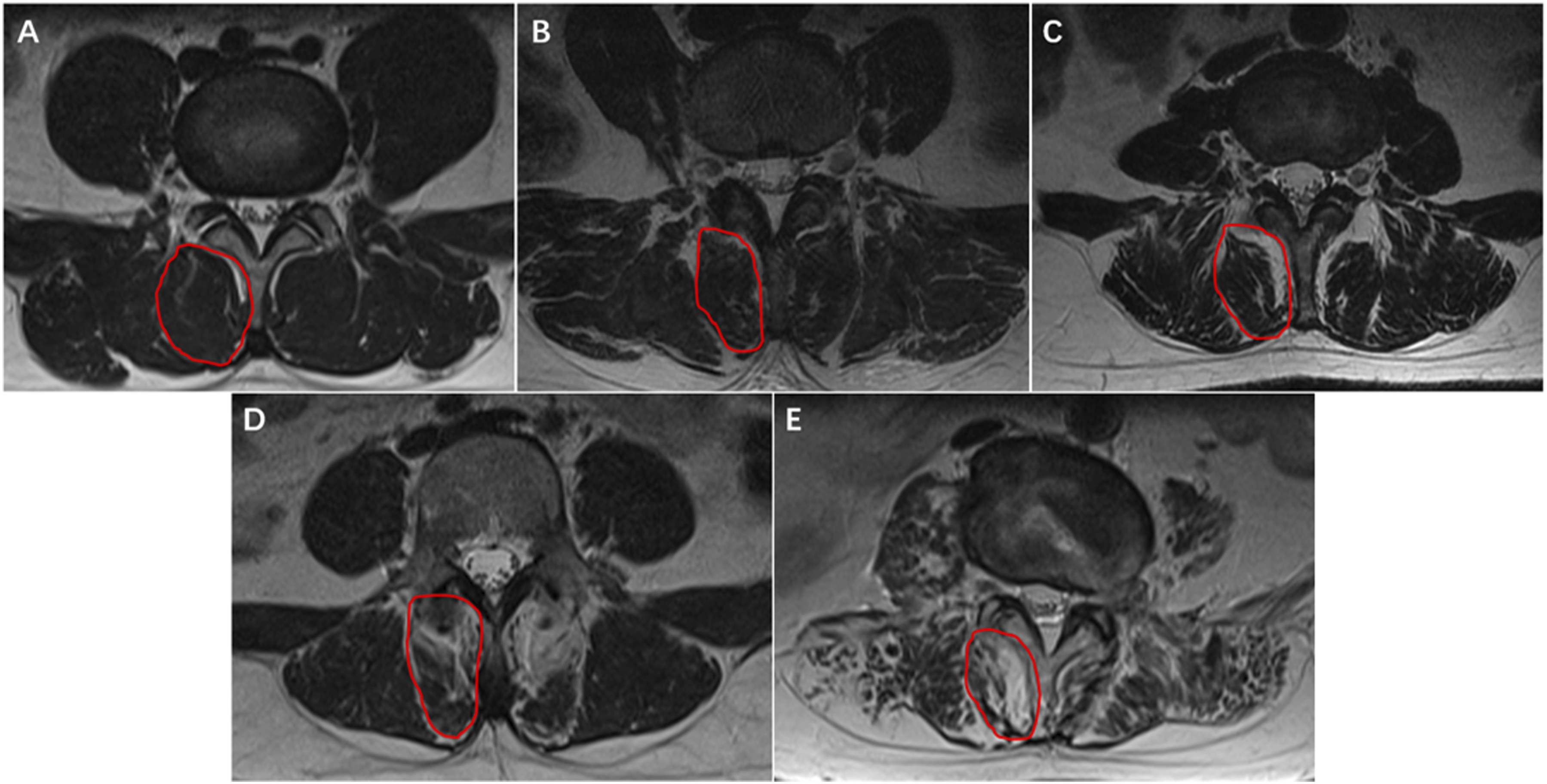
MFI at the L3 level was graded using the Goutallier classification
20
(Figure 2), where grades 0-1 indicate mild infiltration and grades 2-4 indicate moderate to severe infiltration.
21
Measurement of Bilateral Psoas Major Muscle Area at L3 Level Fat Infiltration Grade in the L3 Multifidus Muscle Using the Goutallier Classification. (A) Grade 0; (B) Grade 1; (C) Grade 2; (D) Grade 3; (E) Grade 4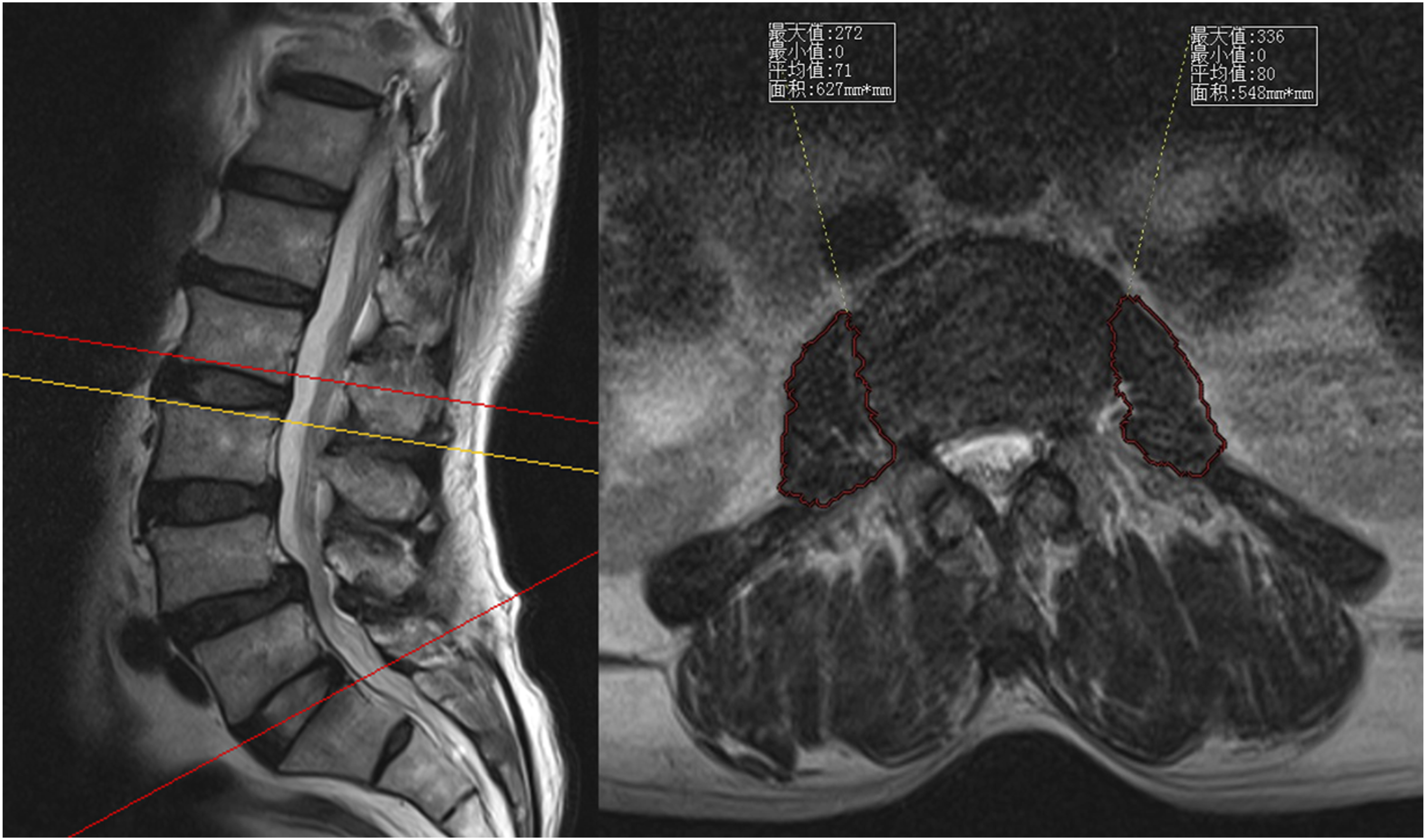
To determine diagnostic thresholds, ethnic differences were considered by referencing relevant domestic studies. According to Kong et al, 22 the critical PMI values for individuals over 60 years of age are ≤5.89 for males and ≤4.22 for females. Patients meeting both the PMI threshold and exhibiting moderate to severe MFI were classified into the PMD group, while those not meeting these criteria were assigned to the non-PMD group.
The Visual Analogue Scale (VAS) of low back pain and Oswestry Disability Index (ODI) were used to assess clinical efficacy, served as the primary endpoints. Scores were recorded preoperatively, postoperatively, and at the final follow-up. Radiographic evaluation of sagittal balance parameters included measurements of lumbar lordosis (LL), pelvic tilt (PT), and pelvic incidence (PI), which were assessed using EOS imaging system preoperatively and at the final follow-up. The mismatch between PI and LL, defined as the value of PI minus LL (PI-LL), was calculated. A PI-LL (PL) value greater than 10° was classified as a mismatch, indicating spinal-pelvic misalignment. 23 The LL, PT, PI, PL and PL-mismatch were served as secondary endpoints.
Statistic Analysis
Independent sample t-tests were conducted to compare continuous variables between the 2 groups, while chi-square tests were used for categorical variables. Paired t-tests were applied to evaluate changes in the same variables before and after surgery. Based on the demographic results with inconsistent baselines identified through the initial comparison, secondary matching was conducted between the 2 groups to ensure baseline consistency. Moreover, covariance analyses were conducted in endpoints with inconsistent baselines. Multiple linear regression analysis was performed to identify independent factors influencing preoperative and convalescent low back pain levels. Shapiro-wilk test was conducted to ensure the model assumptions are verified. All statistical analyses were two-tailed, with a significance level set at
For the machine learning model, we pre-classified VAS scores into 4 levels: 0 points (no pain), 1-3 points (mild), 4-6 points (moderate), and 7-10 points (severe). Synthetic Minority Over-sampling Technique (SMOTE) was employed for data preprocessing, and the lbfgs solver was utilized for model training. To address the issue of data imbalance, we defined an AdaptiveSampler, which dynamically selects either SMOTE or RandomOverSampler based on class distribution to enhance the minority class data. Class imbalance handling——The multiclass distribution of the 4 VAS outcome categories was imbalanced. We therefore applied multiclass SMOTE (imblearn; k_neighbors = 5, fixed random_state) within the training folds only, using sampling_strategy = ‘not majority’ to increase each minority class toward the majority-class size per fold. When a class had fewer than 6 cases in a fold (insufficient neighbors for SMOTE), we used RandomOverSampler for that class (AdaptiveSampler). No resampling was applied to the internal hold-out validation or test sets. Algorithms supporting class weights were trained with class_weight = “balanced”. This procedure was embedded in the cross-validation loop to avoid information leakage. Subsequently, we selected 13 machine learning models for the classification task, including Logistic Regression, Random Forest, Support Vector Machine (SVM), K-Nearest Neighbors (KNN), Decision Tree, XGBoost, LightGBM, Gaussian Naïve Bayes (GaussianNB), Neural Network (MLP), AdaBoost, Gradient Boosting (GB), Histogram-based Gradient Boosting (HGB), and Gaussian Process Classifier (GPC). During cross-validation, we dynamically adjusted the number of folds, ensuring it does not exceed 5 but is at least 2 to maintain a reasonable distribution of training and testing data.
Results
Demographics
A total of 86 patients with lower lumbar spondylolisthesis who underwent single-level PLIF surgery were included in this study. The patients had an average age of 68.0 ± 5.8 years and comprised 30 males and 56 females. Based on the diagnostic threshold for the PMI, 14 males and 33 females met the criteria. Regarding the degree of MFI, 12 patients (13.95%) were classified as grade 0, 24 (27.91%) as grade 1, 21 (24.42%) as grade 2, 27 (31.40%) as grade 3, and 2 (2.33%) as grade 4.
In total, 39 patients met both diagnostic criteria and were classified into the PMD group, while 47 were included in the non-PMD group. There were no statistically significant differences between the groups in terms of gender distribution, age, BMI, or the prevalence of hypertension, diabetes, coronary heart disease, or cerebral infarction. However, the average bone density (T-score) was significantly lower in the PMD group (−2.18 ± 0.78) compared to the non-PMD group (−1.55 ± 0.83) (
Demographic Results of Two Matched Groups
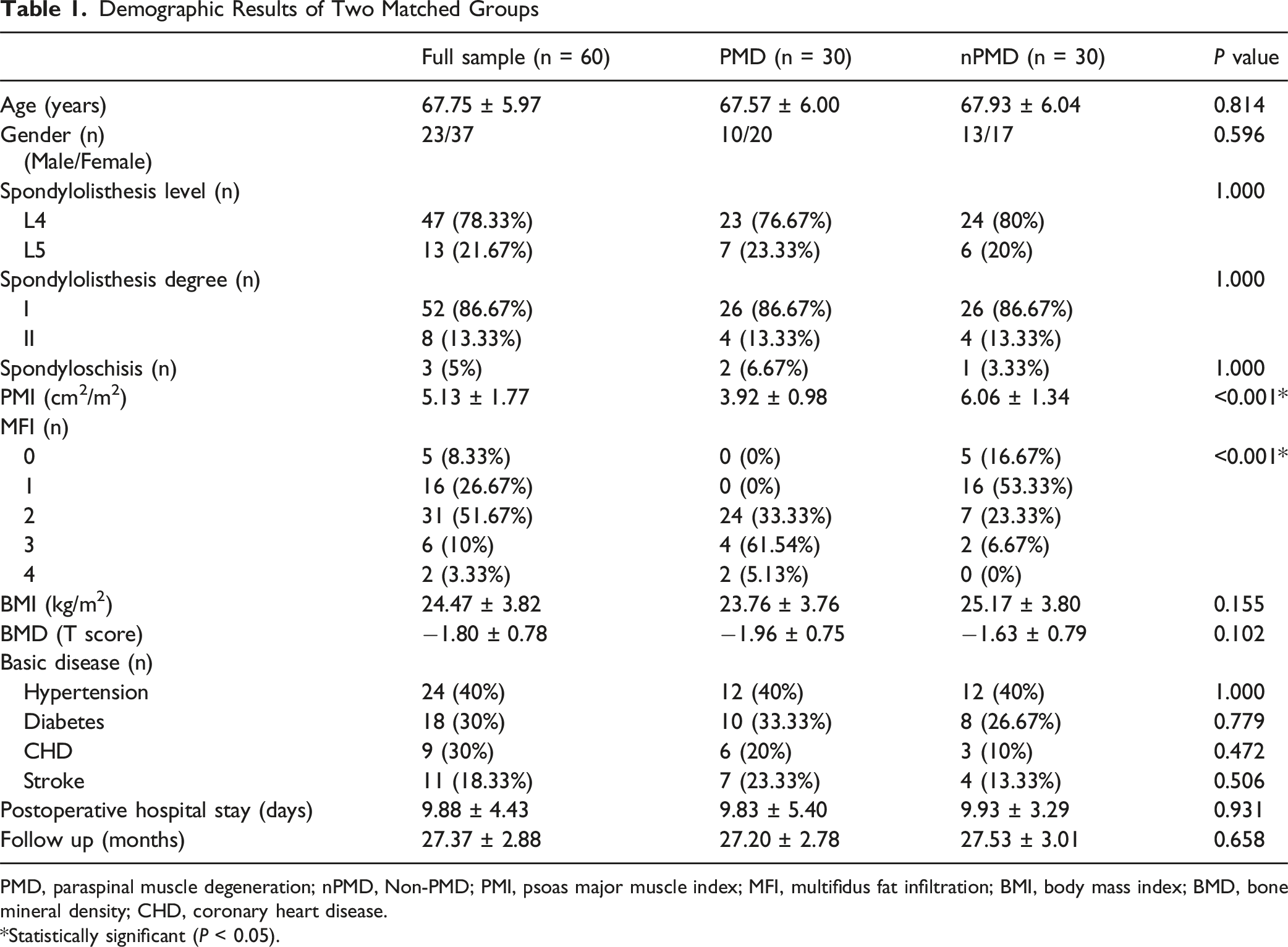
PMD, paraspinal muscle degeneration; nPMD, Non-PMD; PMI, psoas major muscle index; MFI, multifidus fat infiltration; BMI, body mass index; BMD, bone mineral density; CHD, coronary heart disease.
*Statistically significant (
Clinical Function Assessed by VAS and ODI in Two Matched Groups
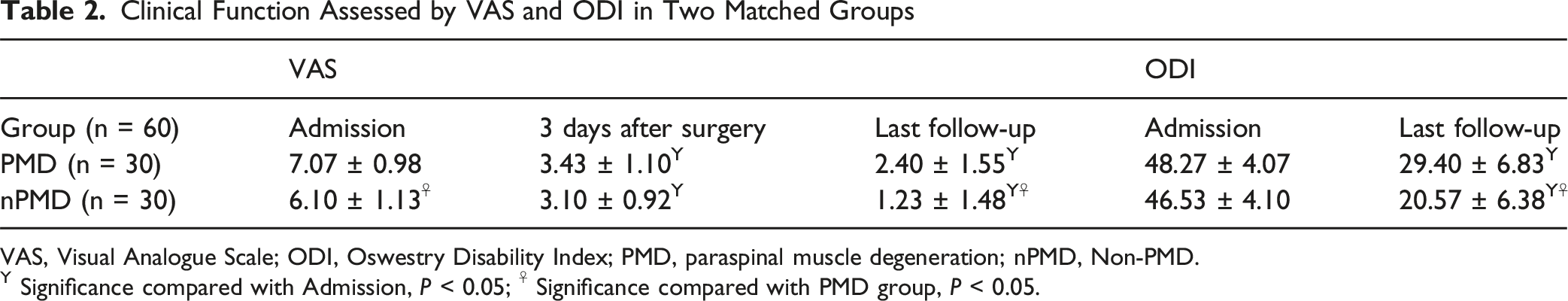
VAS, Visual Analogue Scale; ODI, Oswestry Disability Index; PMD, paraspinal muscle degeneration; nPMD, Non-PMD.
Y Significance compared with Admission,
Lumbosacral Sagittal Balance Parameters in Two Matched Groups
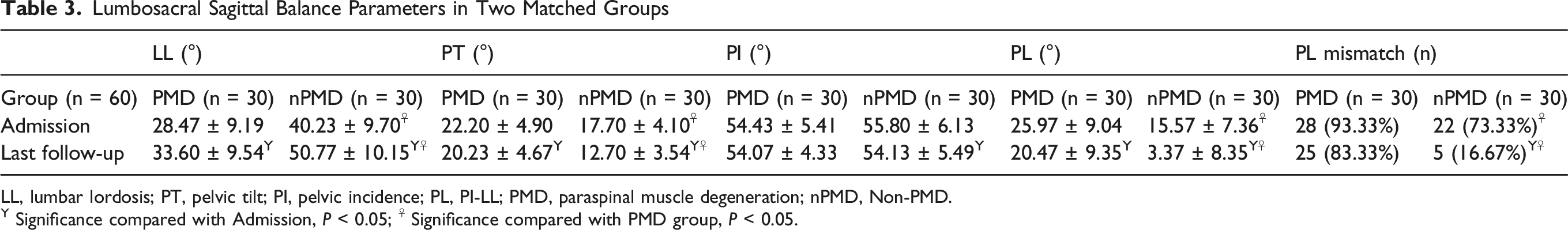
LL, lumbar lordosis; PT, pelvic tilt; PI, pelvic incidence; PL, PI-LL; PMD, paraspinal muscle degeneration; nPMD, Non-PMD.
Y Significance compared with Admission,
Preoperative Functional and Radiographic Assessment
The average preoperative VAS score in the PMD group was 7.07 ± 0.98, while in the non-PMD group it was 6.10 ± 1.13, with a statistically significant difference (
There were significant differences in preoperative sagittal balance between the PMD and non-PMD groups. Specifically, LL in the PMD group was significantly lower than in the non-PMD group (28.47 ± 9.19° vs 33.60 ± 9.54°,
Perioperative Indexes
Typically, patients are encouraged to walk with the support of a waist protector starting from the third day post-surgery, at which point low back pain levels are re-evaluated. The results indicated a significant improvement in postoperative low back pain levels in both groups; however, no significant difference was observed between the 2 groups (3.43 ± 1.10 vs 3.10 ± 0.92,
Functional Outcomes as Primary Endpoints
All patients were followed for at least 2 years, with a mean follow-up duration of 27.4 months postoperatively. The results of VAS and ODI were adjusted by covariance analysis. At the last follow-up, both the PMD and non-PMD groups showed improvements in low back pain levels and dysfunction; however, the improvement was less pronounced in the PMD group. The pain level in the PMD group was significantly higher than in the non-PMD group (2.40 ± 1.55 vs 1.23 ± 1.48,
Sagittal Balance Parameters as Secondary Endpoints
The results of LL, PT, PL were adjusted by covariance analysis. At the last follow-up, sagittal balance parameters had improved in both groups. When comparing the 2 groups, LL in the PMD group remained significantly lower than in the non-PMD group (33.60 ± 9.54° vs 50.77 ± 10.15°,
Analysis of Related Factors of Low Back Pain
Preoperative and convalescent low back pain levels were analyzed using multiple-factor analysis in the initial 86 people. The analysis of preoperative pain included the following factors: gender, age, BMI, bone mineral density, hypertension, diabetes, coronary heart disease, stroke, degree of spondylolisthesis, spondyloschisis, PMI, MFI, and preoperative sagittal balance parameters (LL, PT, PI, PL). The factors of interest for convalescent pain included gender, age, BMI, bone mineral density, hypertension, diabetes, coronary heart disease, stroke, degree of spondylolisthesis, spondyloschisis, PMI, MFI, and latest sagittal balance parameters (LL, PT, PI, PL).
Multivariate Linear Regression Analysis of Factors Associated With the Last VAS
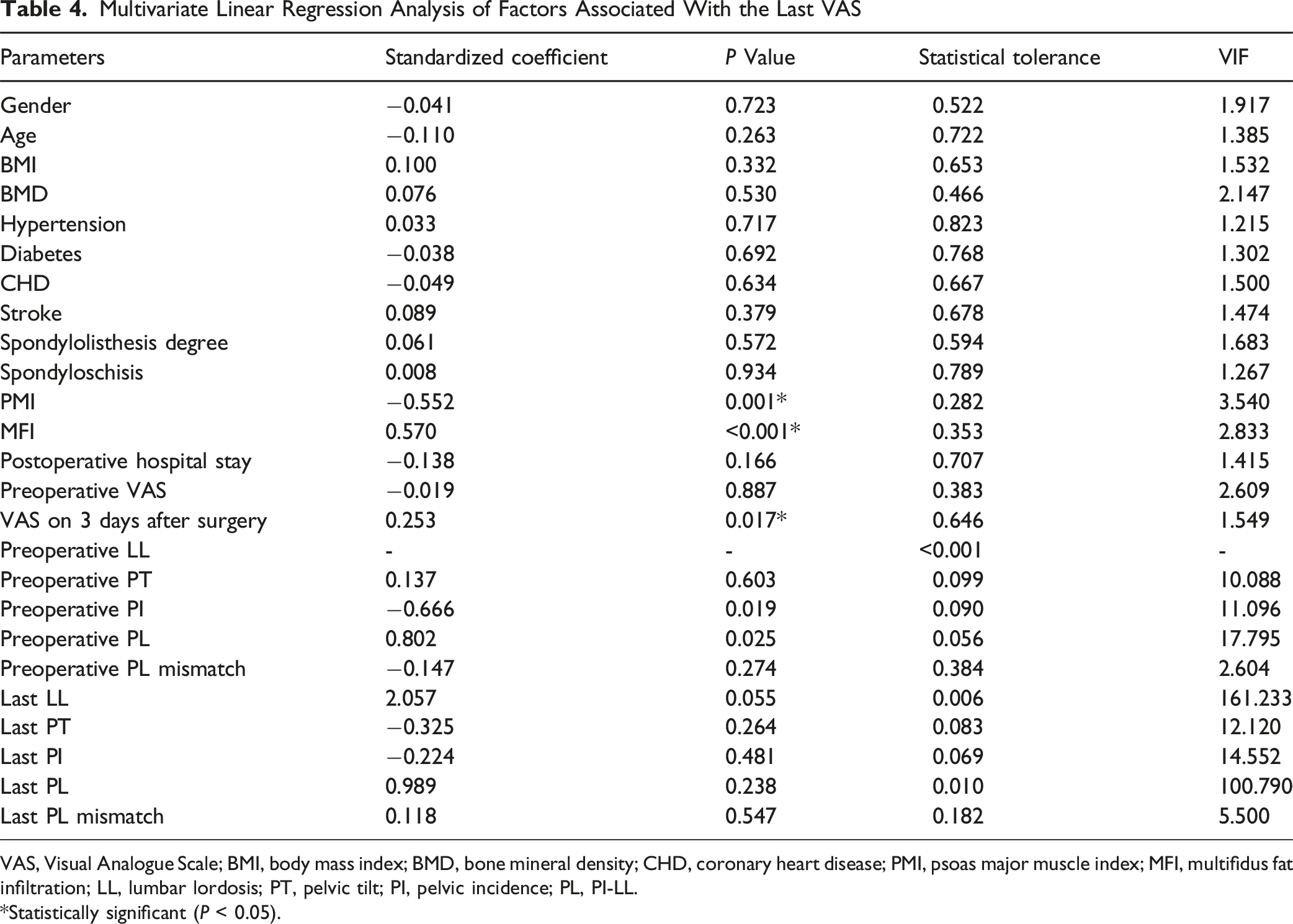
VAS, Visual Analogue Scale; BMI, body mass index; BMD, bone mineral density; CHD, coronary heart disease; PMI, psoas major muscle index; MFI, multifidus fat infiltration; LL, lumbar lordosis; PT, pelvic tilt; PI, pelvic incidence; PL, PI-LL.
*Statistically significant (
Machine Learning Model
Visualization Analysis
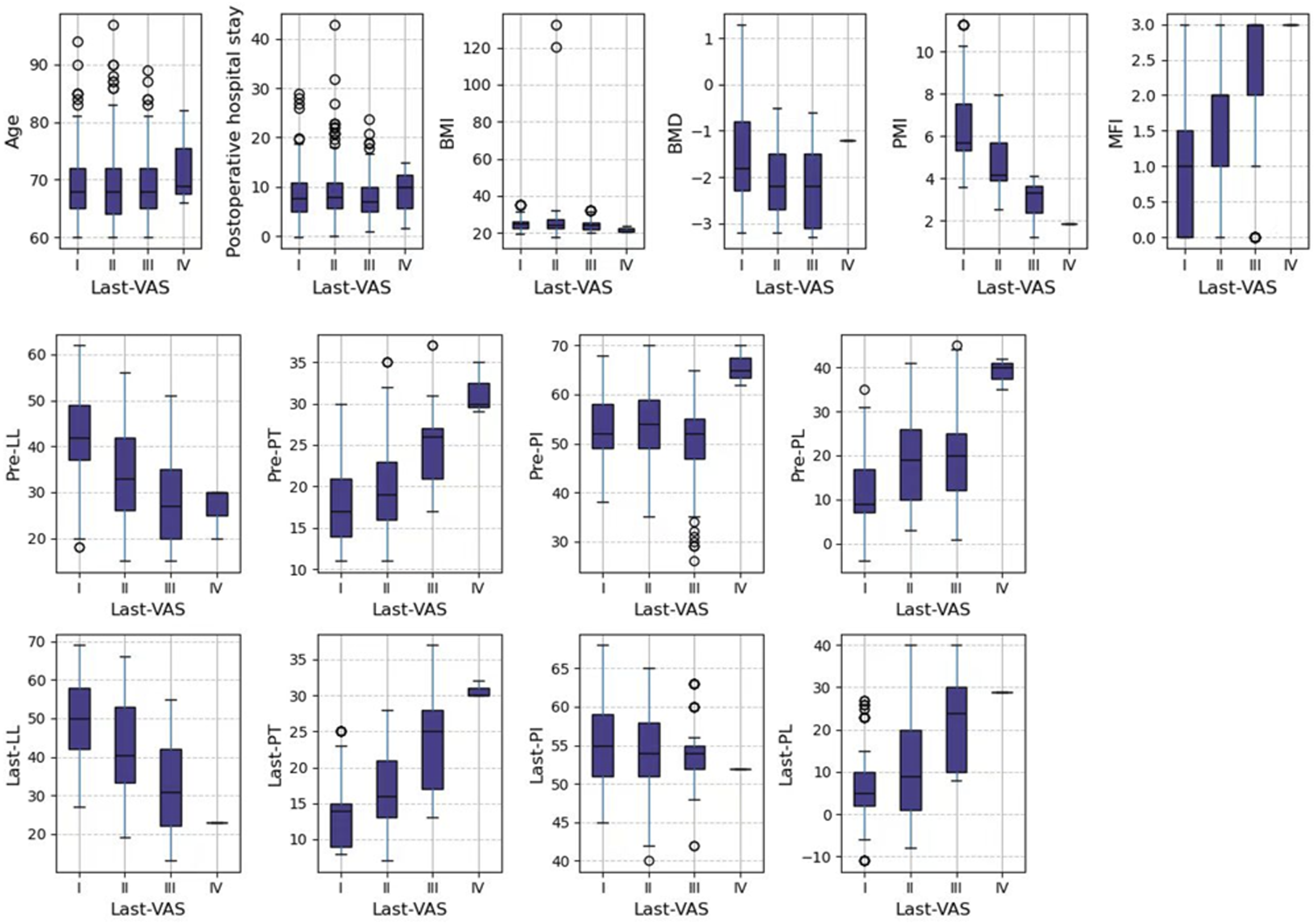
We established a classification system for low back pain severity based on the VAS. A score of 0 was classified as class 0 (no pain), a score between 1-3 was classified as class 1 (mild), a score between 4-6 as class 2 (moderate), and a score between 7-10 as class 3 (severe). The distribution of Last-VAS (VAS at the last follow-up) categories is shown in Figure 3. It can be seen that the most people suffer from mild CLBP, followed by those with no pain, then those with moderate pain, and only a very small number of people complain of severe pain. Specifically, the breakdown of Last-VAS categories by factors such as age, PMI, MFI and sagittal balance parameters is shown in Figure 4. It can be seen that PMI, MFI, LL, PT, PL and show correlations with Last-VAS categories. Distribution of Last-VAS Categories Breakdown of Influencing Factors by Last-VAS Category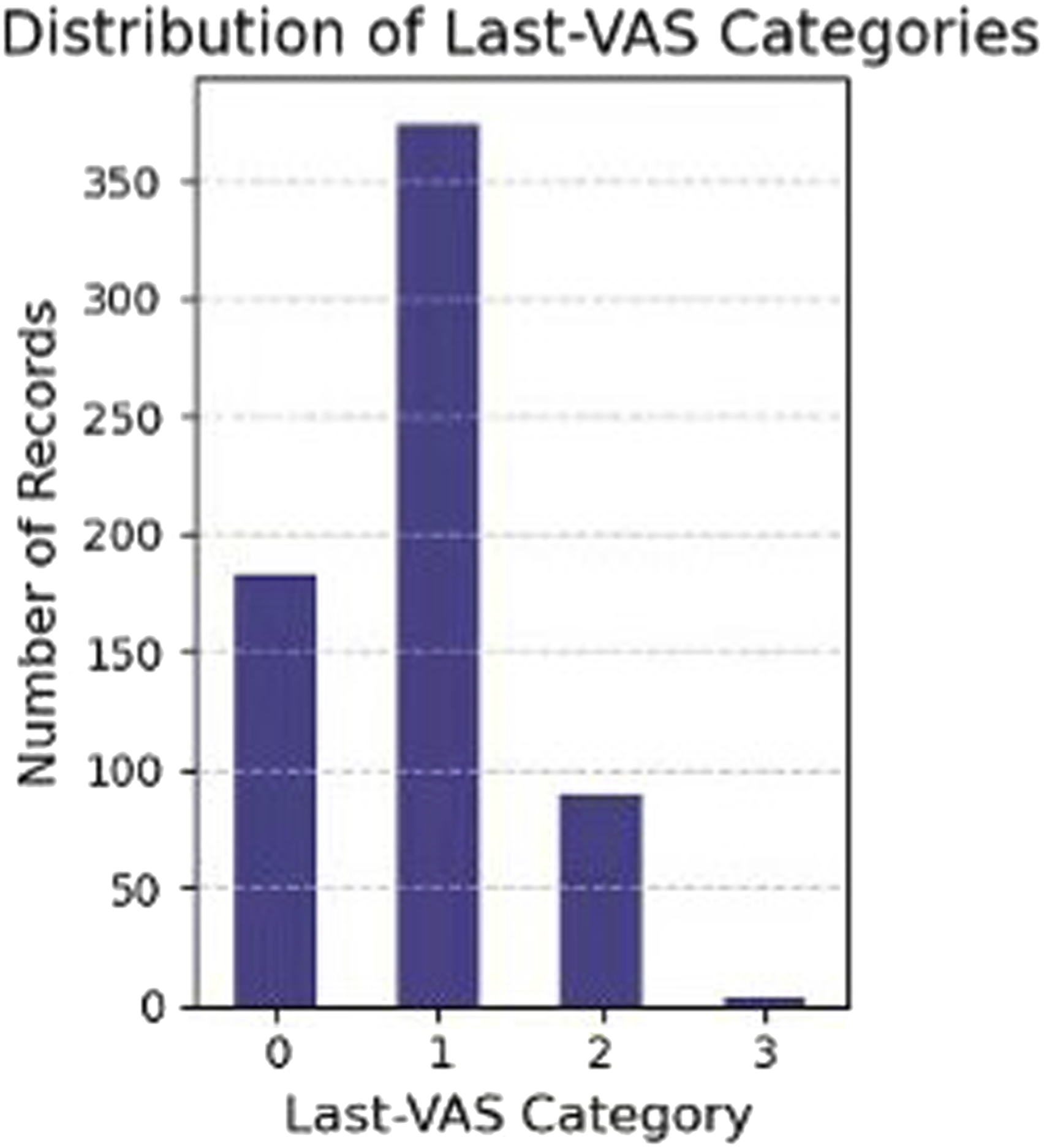
Multi-Class Prediction and Permutation Importance of Variables
The model performance summary (Figure 5) provides a quantitative evaluation of various classifiers. The Random Forest model exhibited the highest ROC AUC (0.937 ± 0.031) and specificity (0.968 ± 0.020), indicating strong discriminative power. The Gradient Boosting and Gaussian Process classifiers also achieved competitive performance. Neural networks and SVM performed relatively well, while Logistic Regression and Gaussian Naïve Bayes demonstrated lower sensitivity and overall predictive power. Comparison of Model Performance Metrics. (A) The Random Forest Model Demonstrates Superiority in ROC AUC, Specificity and Sensitivity. (B) A Summary of all Evaluation Indicators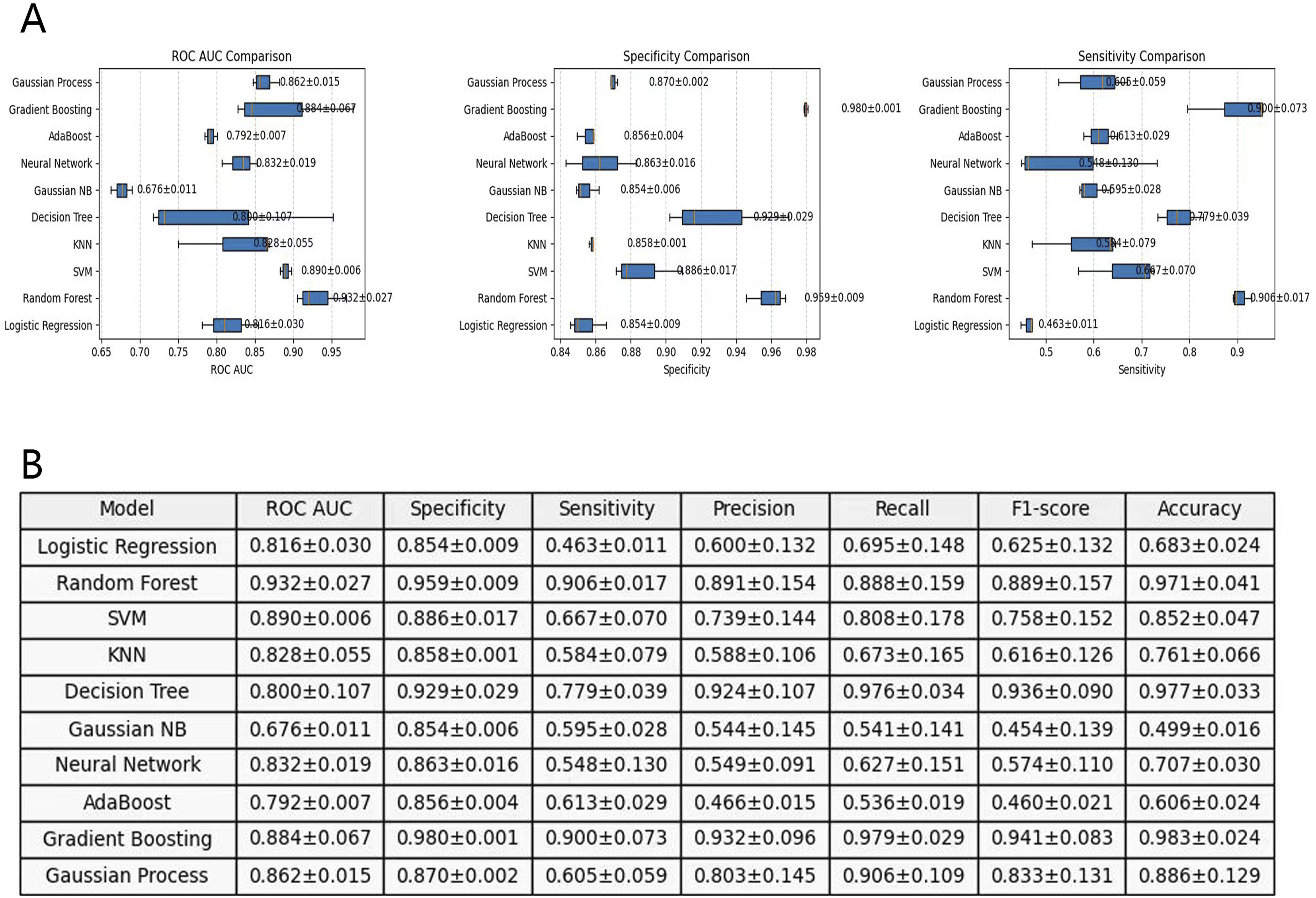
To further interpret the model’s predictions, Figure 6 illustrates the feature importance analysis using the Random Forest model. PMI, BMD, MFI and BMI emerged as the most influential features, contributing significantly to the classification performance. Additionally, VAS-related variables (Pre-VAS, Post-VAS) and spondylolisthesis-related attributes also played an important role. Feature Importance Analysis by Random Forest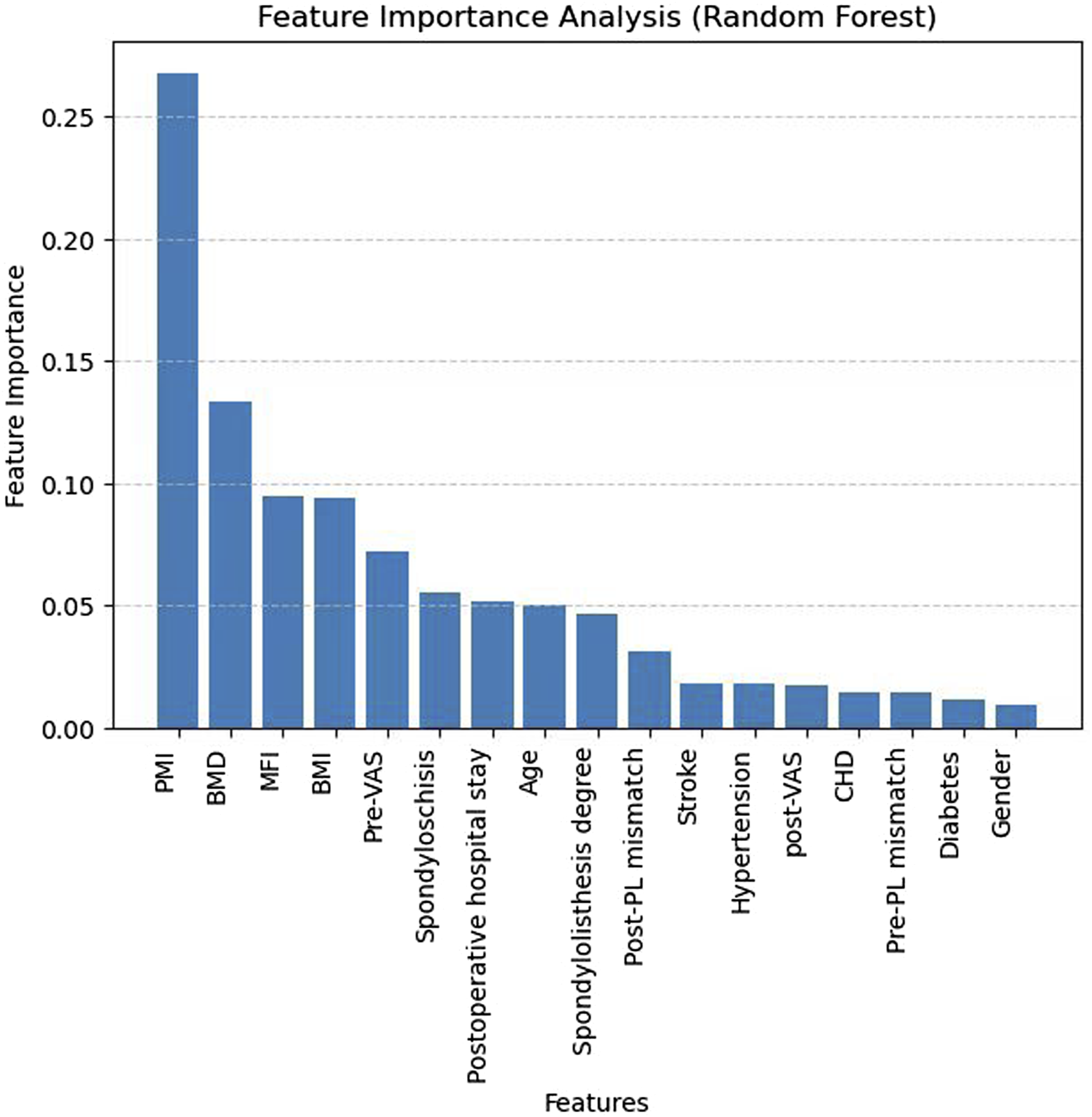
Figure 7 provides a broader feature importance distribution across trees, highlighting the variability of importance scores in ensemble models. PMI and spondylolisthesis degree exhibited the highest variability, suggesting their strong yet dataset-dependent predictive capability. Feature Importance Distribution Across Trees by Random Forest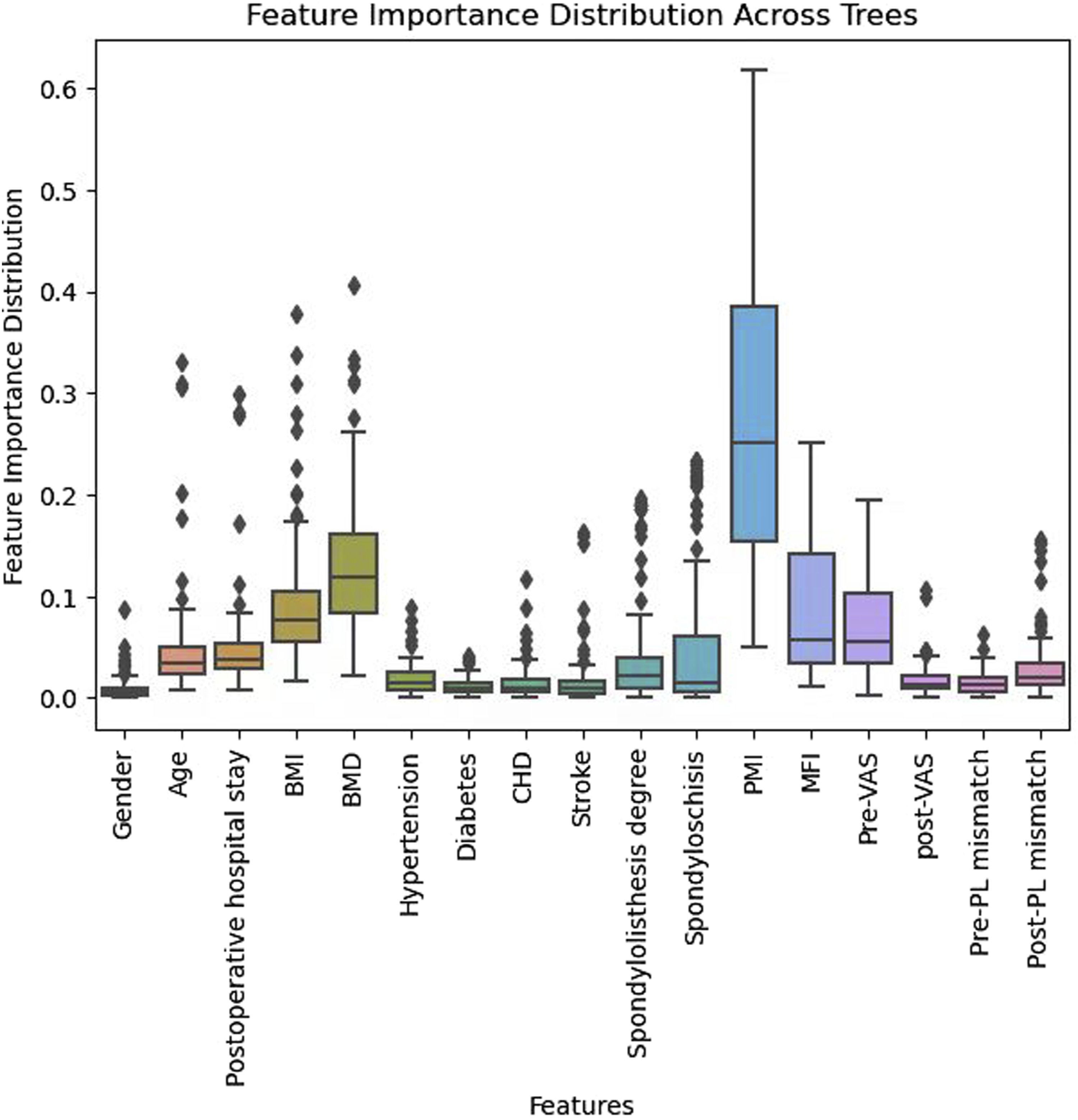
Discussion
The results show that PMD has significant effects on preoperative symptom severity and surgical outcomes in elderly patients with lower lumbar spondylolisthesis undergoing single-level PLIF. Patients with PMD exhibit more severe preoperative symptoms and greater sagittal imbalance. During the postoperative recovery period, these patients experience poorer low back pain relief and functional recovery, and their improvement in sagittal balance is less pronounced compared to non-PMD patients. Through factor analysis and machine learning model, we predict that PMD plays an important role in CLBP of the lumbar spondylolisthesis population. We believe that the model shows promising internal performance and could evaluate postoperative risk stratification, waiting for external validation.
Factor analysis reveals a strong correlation between PMI and MFI with both preoperative and convalescent low back pain levels, a finding that is further validated by the machine learning model. Patients with sarcopenia or PMD tend to experience higher levels of chronic pain and dysfunction, a conclusion supported by existing literature.24,25 However, whether these conditions are a cause or a consequence of pain remains inconclusive. Some researchers suggest that this correlation may be explained by the tendency of individuals with degenerative lumbar diseases and chronic pain to deliberately reduce their physical activity, including movement of the body and waist, in an effort to avoid exacerbating pain.26,27 The PMD group exhibited poorer pain relief and slower functional recovery during the postoperative period, which aligns with other studies, highlighting the negative impact of PMD on postoperative rehabilitation. 28 This may be attributed to the reduced muscle strength and mass in PMD patients, resulting in a diminished capacity for recovery following surgery. Studies have shown that a reduced MRI lumbar cross-sectional area is associated with functional prognosis after posterior lumbar surgery, with the psoas index being particularly reliable, especially at the L3/4 level. 29 Furthermore, the degree of multifidus fat infiltration and atrophy before surgery is a strong predictor of postoperative pain and disability. 30 It was observed that patients with PMD generally exhibited a higher degree of lumbar spondylolisthesis compared to the normal population. Xu et al 31 developed a finite element model to analyze the impact of sarcopenia on spinal injury risk, revealing that sarcopenia primarily increased the risk of injury at the L4-L5 intervertebral disc, L1 vertebral body, and L3-S1 joint capsule under significant stress or strain changes. This provides a reasonable explanation for the development of lumbar spondylolisthesis due to abnormal stress in the intervertebral joints. A recent study examining the relationship between the severity of slippage in degenerative lumbar spondylolisthesis and PMD suggested that progressive muscular imbalance between the posterior and anterior paraspinal muscles could contribute to the progression of lumbar slippage. 32 Wang et al 33 confirmed that multifidus muscle atrophy could contribute to lumbar spondylolisthesis. Another biomechanical experiment further supported the idea that PMD exacerbates lumbar spondylolisthesis. 34 In a comparison of patients with degenerative lumbar spondylolisthesis and degenerative lumbar kyphosis, Ding et al 35 found that multifidus fat infiltration was more common in patients with lumbar spondylolisthesis. Moreover, our machine learning model revealed a significant effect of the degree of spondylolisthesis on convalescent pain levels. Specifically, the model demonstrated that more severe slippage was more likely to result in postoperative chronic pain at moderate to severe levels. These findings highlight the importance of fully understanding the role and potential impact of PMD in patients with lumbar spondylolisthesis, and emphasize the need for careful surgical planning and enhanced patient education on back muscle exercises.
Spinal sagittal imbalance is a common condition in patients with degenerative spinal diseases, particularly those with lumbar spondylolisthesis. Research has shown that sagittal imbalance is not only associated with low back pain and functional impairment but also significantly impacts the effectiveness and long-term outcomes of spinal surgery. 36 Patients with PMD often experience postural instability due to diminished muscle strength and mass. To maintain balance, these individuals may adopt compensatory postures, such as leaning forward or backward, which can result in abnormal sagittal balance parameters.37,38 In this study, preoperative sagittal balance parameters, including LL, PT, and PL mismatches, were worse in the PMD group compared to the non-PMD group. After more than 2 years of observation, the improvement of sagittal balance parameters in the PMD group was also less pronounced than in the non-PMD group. This may be attributed to the lack of muscle strength in individuals with PMD, resulting in poor spinal stability and postural adjustment. 39 Previous studies have indicated that poor sagittal or coronal balance of the lumbar spine is closely associated with PMD.40-44 On the other hand, the functional outcome of lumbar spondylolisthesis surgery is closely related to both preoperative and postoperative sagittal spinal balance, as supported by the literature.45-47 Therefore, assessing PMD may be useful in predicting the need for surgical correction of sagittal balance.
There are several limitations to this study. First, the study only included individuals over the age of 60, primarily based on the following considerations. While sarcopenia is not exclusive to the elderly, its incidence is notably high within this age group.6,48,49 Numerous studies have attempted to explain the relationship between aging and sarcopenia at the molecular biological level.50,51 Research in the spinal field has further confirmed a clear correlation between PMD and aging.52-54 Therefore, we aimed to control for this prominent confounding factor within a manageable range. Second, some preoperative parameters differ between the 2 groups, and these baseline inconsistencies could potentially affect the accuracy of our conclusions. For example, significant differences in listhesis degree and bone mineral density were observed between the groups. Extensive studies have shown a close relationship between sarcopenia and osteoporosis. 55 Sarcopenia and decreased bone mineral density frequently co-occur due to shared common causes and risk factors, such as aging, poor nutrition, reduced physical activity, chronic diseases, and the use of certain medications. 56 There is a mutually supportive relationship between muscle strength and bone health, and neither should be assumed as the sole cause or effect of the other. For this reason, the concept of “osteosarcopenia” has been introduced, referring to the coexistence of both conditions.57-59 To minimize the influence of confounding factors as much as possible, we matched the 2 groups by listhesis degree and BMD. To more accurately identify the main factor influencing outcomes, we conducted a correlation factor analysis and confirmed that poor muscle condition was the primary cause of CLBP. Third, the follow-up period of this study was limited to 2 years post-operation, preventing an evaluation of long-term efficacy and complications. Meanwhile, due to the lack of unified and systematic postoperative management, we were unable to study the impact of interventions (eg, targeted muscle strengthening or nutritional interventions) on the prognosis improvement of the PMD population. Fourth, the lack of external validation using an independent cohort may affect the model’s cross-center transferability and pose a risk of spectral bias beyond the composition of cases in this single center. Additionally, this model is intended for elderly patients with degenerative lumbar spondylolisthesis (Meyerding I–II grade) undergoing PLIF treatment; its generalizability to other disease spectra (such as those primarily characterized by disc herniation), higher grades of spondylolisthesis, or different treatment modalities has not been validated. In the future, we aim to conduct prospective multicenter studies with different age groups, consistent baseline levels, and longer follow-up periods to further refine the research content. In this regard, we have registered a prospective study with the National Center for Clinical Trials (Registration number: ChiCTR2300072052). As part of this further research, the included cases are being evaluated according to sarcopenia-related diagnostic guidelines, including muscle strength, grip strength, and walking speed. This research is currently underway.
Supplemental Material
Supplemental Material - Role of Paraspinal Muscle Degeneration as a Predictor of Chronic Low Back Pain in Lumbar Spondylolisthesis: A Machine Learning Model is Developed
Supplemental Material for Role of Paraspinal Muscle Degeneration as a Predictor of Chronic Low Back Pain in Lumbar Spondylolisthesis: A Machine Learning Model is Developed by Junxin Zhang, Tianci Fang, Xingyu You, Yingchuang Tang, Xingbang Ruan, Haifu Sun, Liu Hao, and Huilin Yang in Global Spine Journal.
Footnotes
Acknowledgements
Thanks to all patients involved in this study.
Funding
This study is supported by the National Natural Science Foudation of China (82502912) and the Natural Science Foundation of Jiangsu Province (BK20250355).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
