Abstract
Study Design
Retrospective Cohort Study.
Objectives
Unilateral biportal endoscopy (UBE) provides a minimally invasive option for treating lumbar spinal stenosis, enabling faster recovery but requiring surgical precision. This study aims to establish a learning curve model for UBE in central lumbar spinal stenosis, focusing on surgical duration and neuromonitoring data.
Methods
Data were collected from a senior orthopedic spine surgeon’s cases involving lumbar spinal stenosis. Neuromonitoring was performed during UBE, recording each dural and root irritation as a risk. Surgical details, including operative time (from first incision to final suture) and neuromonitoring events, were documented. The RA-CUSUM method in Python was used to evaluate proficiency, adjusting for procedure time.
Results
Seventy-eight patients with central lumbar spinal stenosis were included. RA-CUSUM analysis, integrating neuromonitoring and surgical time, indicated significant proficiency gains by the 16th case, with reductions in procedural time and neural irritations. CUSUM analysis based solely on operative time showed stabilization and efficiency improvements by the 35th case.
Conclusion
Proficiency in UBE surgery is generally achieved by the 16th case regarding neuromonitoring management and by the 35th case regarding time efficiency. These findings are valuable for surgeons transitioning to UBE, although the results may not fully generalize to those without prior surgical experience. Continued research and structured training are essential to optimize UBE outcomes.
Introduction
The prevalence of lumbar spinal stenosis increases with age, leading to significant disabilities due to associated pain. These conditions can be effectively managed through the unilateral biportal endoscopy (UBE) technique, which offers substantial advantages. 1 UBE’s benefits include providing a wide field of view, a minimally invasive approach, less bleeding, reduced traumatization, and rapid functional recovery.2-4Given that UBE is a novel and evolving technique performed around the central nervous system, even experienced spine surgeons may face significant risks and challenges. Surgeons must balance their procedures to be both safe and adequately surgical. Understanding the learning curve for surgeons who have not previously performed UBE surgery will serve as a guide during the application of this surgical principle. The learning curve is defined as the improvement in surgical procedure performance with increased experience and training.
There are quite different opinions regarding learning curve studies on UBE, with claims that the learning curve is completed between 20 and 89 cases.5-9 However, the results found in these studies not only show a wide range but may also be insufficient regarding a risk-based learning curve. In this study, neuromonitoring was used during UBE surgery, and each dural and root stimulus recorded during neuromonitoring was defined as a risk.
The aim of this study was to establish a learning curve model for treating central lumbar spinal stenosis using the UBE technique, focusing on the duration of surgery and neuromonitoring data. This model is intended to provide insights for spine surgeons on the learning process for this surgical technique.
Materials and Methods
This study received ethical approval from the institutional review board and adhered to the guidelines outlined in the Declaration of Helsinki. Conducted from December 2022 to December 2024, the research comprised a retrospective analysis of prospectively collected data. These cases were derived from the clinical practice of an orthopedic spine surgeon with over 10 years of extensive experience in both microscopic and open spinal procedures. Furthermore, the surgeon’s expertise in arthroscopic surgery contributed essential triangulation skills, which are crucial for effectively performing the unilateral UBE technique utilized in this study.
The study focused exclusively on patients diagnosed with central lumbar spinal stenosis, all of whom failed to find relief through physical therapy and conservative treatment, and suffered from neurological claudication that limited their walking distance to less than 100 meters. It excluded those with disc herniation, multilevel stenosis, or a history of open or endoscopic spinal surgery to maintain a uniform cohort. During each procedure, meticulous records were kept on the number of dural irritations caused by the radiofrequency probe and root irritations noted during more invasive stages of the surgeries. Comprehensive assessments were conducted preoperatively and postoperatively to document changes in neurological status. The duration of each surgery was precisely measured from the first incision to the final suture, ensuring detailed tracking of surgical efficiency. The decompression levels of the patients were recorded. Additionally, the preoperative VAS scores and postoperative 1st-month VAS scores were noted. The difference between these two VAS scores was considered as “pain relief.”
Surgical Technique
The patient was placed under general anesthesia and positioned prone. Necessary portal markings were made using C-arm fluoroscopy. The UBE procedure was performed through two distinct paramedian portals (viewing and working), created via 1 cm incisions under fluoroscopic guidance. The operating surgeon (AG) was right-hand dominant. In all cases, the surgeon stood on the left side of the patient, using the left hand for the viewing portal and the right hand for the working portal. Blunt dissection was performed with the aid of a dilator. A 0° arthroscope was introduced through the viewing portal, while surgical instruments were manipulated through the working portal, with continuous irrigation maintained to ensure optimal visualization throughout the decompression procedures. Particular attention was paid to maintaining an interportal distance of 2-3 cm to avoid instrument crowding and to preserve an adequate working space. Meticulous hemostasis was achieved using bipolar coagulation. Once the spinolaminar junction was identified, bony resection was initiated using a burr. Using a plasma radio-frequency knife, the inferior edge of the superior lamina, the root of the spinous process, the superior margin of the inferior lamina, the inner margin of the facet joint, and the interlaminar ligamentum flavum were carefully exposed. All compressive bony structures were resected to an extent deemed unlikely to cause postoperative instability. The same procedures were then applied from the sublaminar to the contralateral side. After the completion of bony work, the ligamentum flavum was fully exposed. During the excision of the ligamentum flavum, the contralateral side was addressed first, followed by the ipsilateral side. Throughout the procedure, care was taken to avoid contact with the dura and nerve roots as much as possible.
Neuromonitoring
Intraoperative neuromonitoring was performed using a 16-channel multimodal neuromonitoring system. The modalities employed included motor evoked potentials (MEP), somatosensory evoked potentials (SSEP), and free-running electromyography (F-EMG). MEP was elicited through subcutaneous needle electrodes delivering constant voltage (400-500 V) in trains of 5-7 pulses with a duration of 200-400 ms per pulse and an interstimulus interval of 2.0-4.0 ms. Responses were recorded from muscles innervated by corresponding nerve roots. SSEP monitoring was conducted via stimulation of the posterior tibial nerve at the ankle, with stimulus intensities of 35-45 mA at 2 Hz. Responses were recorded at Cz and Fz electrode positions with 160-300 averages per trace. F-EMG continuously monitored spontaneous muscle activity in corresponding myotomes.
Dural or nerve root irritation events were defined based on established intraoperative neuromonitoring thresholds: an event was recorded when there was an 80% or greater reduction in MEP amplitude or a complete waveform loss; for SSEP, a 50% or greater decrease in amplitude and/or a latency increase of 10% or more compared to baseline was considered significant; and for F-EMG, sustained neurotonic discharges lasting more than 5 s or continuous abnormal burst activity were interpreted as evidence of neural irritation. These events were recorded in real-time and logged as discrete occurrences, representing procedural risk and forming the basis for the risk-adjusted learning curve analysis.
Data Analysis
The analysis was conducted using Python programming language (version 3.12.7). Data loading and preprocessing were performed using the pandas library, while mathematical operations utilized the numpy library. Visualization was accomplished with the matplotlib.pyplot library, and the Savitzky-Golay filter and gradient analyses were implemented using functions from the scipy.signal module.
A Risk-Adjusted Cumulative Sum (RA-CUSUM) analysis was employed to monitor and evaluate proficiency gains in surgical procedures. In this method, the ‘duration of surgery (min)’ was the primary factor, representing expected reductions in procedural time as experience increased. Performance was assessed using the ‘number of total root and dural irritations obtained by neuromonitoring’, which was adjusted for variations in procedural duration. This adjustment highlighted performance changes while accounting for risks associated with procedural time variability, providing a detailed perspective on learning and proficiency gains over time.
To enhance visualization and interpretability of the RA-CUSUM data, a Savitzky-Golay filter was applied. Various combinations of window sizes (21, 31, 41, 47) and polynomial orders were tested to optimize data smoothing, with odd window sizes ensuring symmetry around the central point. The selected window sizes balanced the length of the data series (78 measurements) with the need for comprehensive smoothing. The optimal smoothing parameters were determined based on maximizing the R2 value, ensuring the best fit of the smoothed data to the original series.
The stabilization point, representing the plateau of the learning curve, was identified by calculating the gradient of the smoothed RA-CUSUM curve. The stabilization point was defined as the first instance where the absolute gradient fell within the lowest 10% of values, reflecting minimal performance changes. This critical point was visually highlighted on the graphs with a vertical marker, providing a clear indicator of the transition to stable performance.
The Mann-Whitney U test was used to compare pain relief between the mastery phase and learning phase. Differences in level distributions were assessed using Chi-square tests.
Results
This study analyzed the learning curve associated with the UBE technique in 78 patients diagnosed with central lumbar spinal stenosis. Among these patients, 38 were female. The mean age of the cohort was 68.9 years, with a standard deviation of 5.8 years. This demographic setup provides a basis for evaluating the proficiency level of UBE technique on a standard patient group. No intraoperative or postoperative major complications such as dural tear, epidural hematoma, deep infection, or nerve root injury were observed in any of the patients included in the study. Only three patients experienced minor complications in the form of superficial wound infections. However, all three cases were successfully managed with wound care and antibiotic therapy, resulting in complete recovery without recurrence.
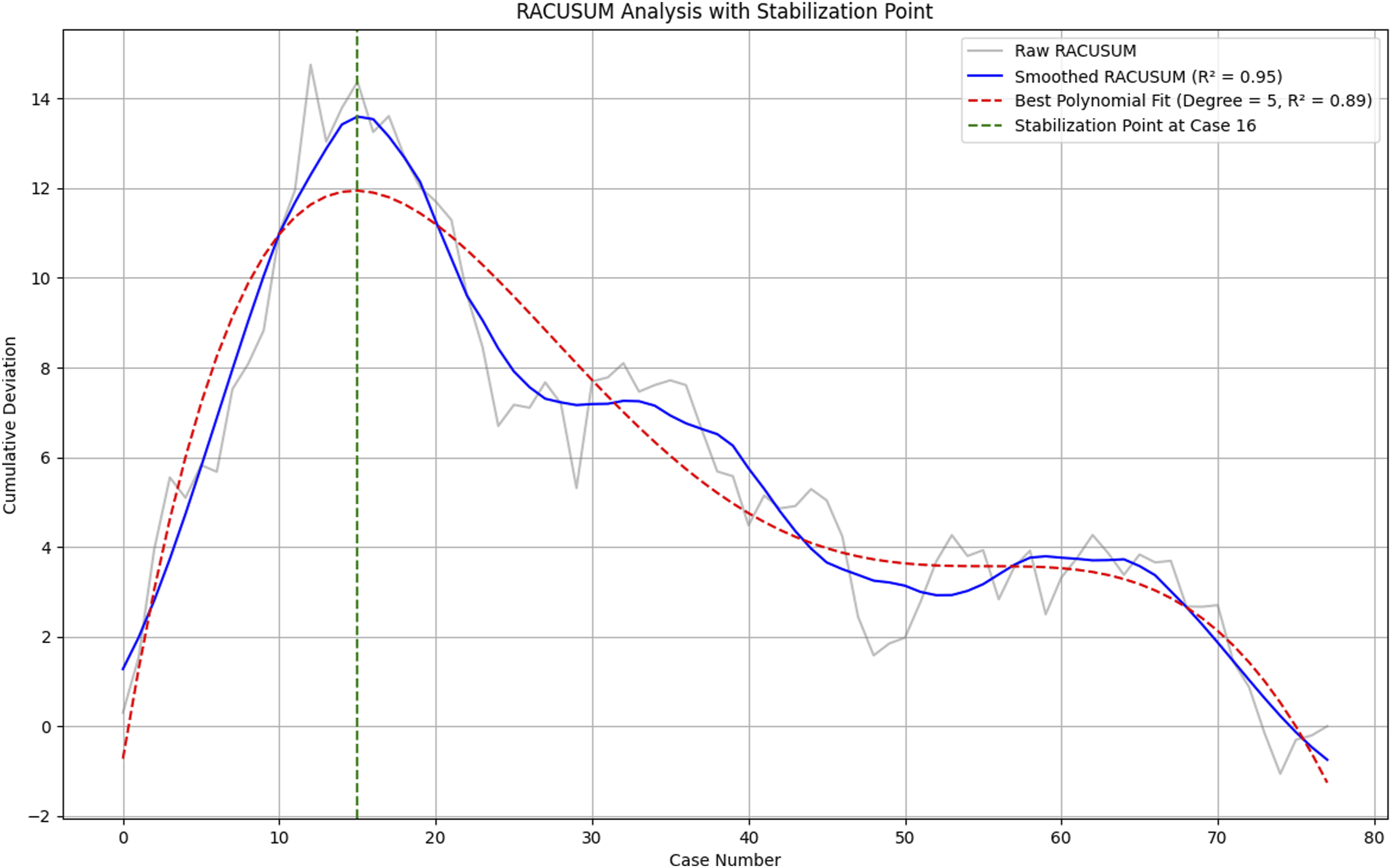
This retrospective analysis evaluated the evolution of surgical proficiency using both CUSUM and RA-CUSUM methods, aiming to identify the stabilization point in learning. Stabilization reflects a consistent level of proficiency based on procedural time and neuromonitoring metrics, such as dural and total root irritations. The CUSUM analysis focused solely on surgical time, offering an unadjusted learning curve that highlighted efficiency gains over time. Stabilization was observed around the 35th case (Figure 1), where the gradient of the learning curve plateaued, indicating consistency in reducing surgical time. In contrast, the RA-CUSUM analysis integrated both procedural time and neuromonitoring metrics, adjusting for variations in case complexity. This risk-adjusted approach provided a more nuanced understanding of proficiency development by capturing deviations in performance relative to expected outcomes. Stabilization in the RA-CUSUM curve was identified by the 16th case (Figure 2), where significant reductions in irritations indicated improved precision and expertise. Polynomial regression modeling was applied to both analyses, yielding high R2 values (0.89 for RA-CUSUM and 0.98 for CUSUM), confirming the robustness of the results. The smoothed RA-CUSUM curve demonstrated the effectiveness of the risk-adjusted approach, capturing both efficiency gains and error reductions over time. The convergence of findings from these analyses—stabilization of irritations by the 16th case and procedural time by the 35th case—provides a comprehensive view of the surgeon’s learning curve. These results underscore the importance of incorporating risk-adjusted metrics to accurately evaluate proficiency and support targeted training strategies in surgical practice. The Figure Illustrates the CUSUM Analysis of Surgical Time. The Grey Line Represents the Raw CUSUM Data, Capturing Cumulative Changes in Surgical Time. The Blue Line Depicts the Smoothed CUSUM Curve, Optimized Using a Savitzky-Golay Filter With an R2 Value of 0.99, Highlighting Trends in Procedural Efficiency. A Polynomial Fit (Red Dashed Line) of Degree 5 With an R2 Value of 0.98 is Overlaid to Model the Learning Curve. The Stabilization Point, Identified at Case 35, is Marked by the Green Dashed Line, Indicating the Plateau in the Learning Process Where Surgical Times Become Consistent The Figure Presents the RACUSUM Analysis Incorporating Both Surgical Time and Neuromonitoring Metrics. The Grey Line Illustrates the Raw RACUSUM Data, Representing Cumulative Deviations in Performance. The Blue Line Represents the Smoothed RACUSUM Curve (R2 = 0.95), Optimized Using a Savitzky-Golay Filter, Providing a Clearer Visualization of Trends in Surgical Precision. A Polynomial Fit (Red Dashed Line) of Degree 5 With an R2 Value of 0.89 Models the Learning Curve. The Stabilization Point is Identified at Case 16 (Green Dashed Line), Marking the Point Where Performance Metrics Stabilize, Indicating Proficiency in Surgical Execution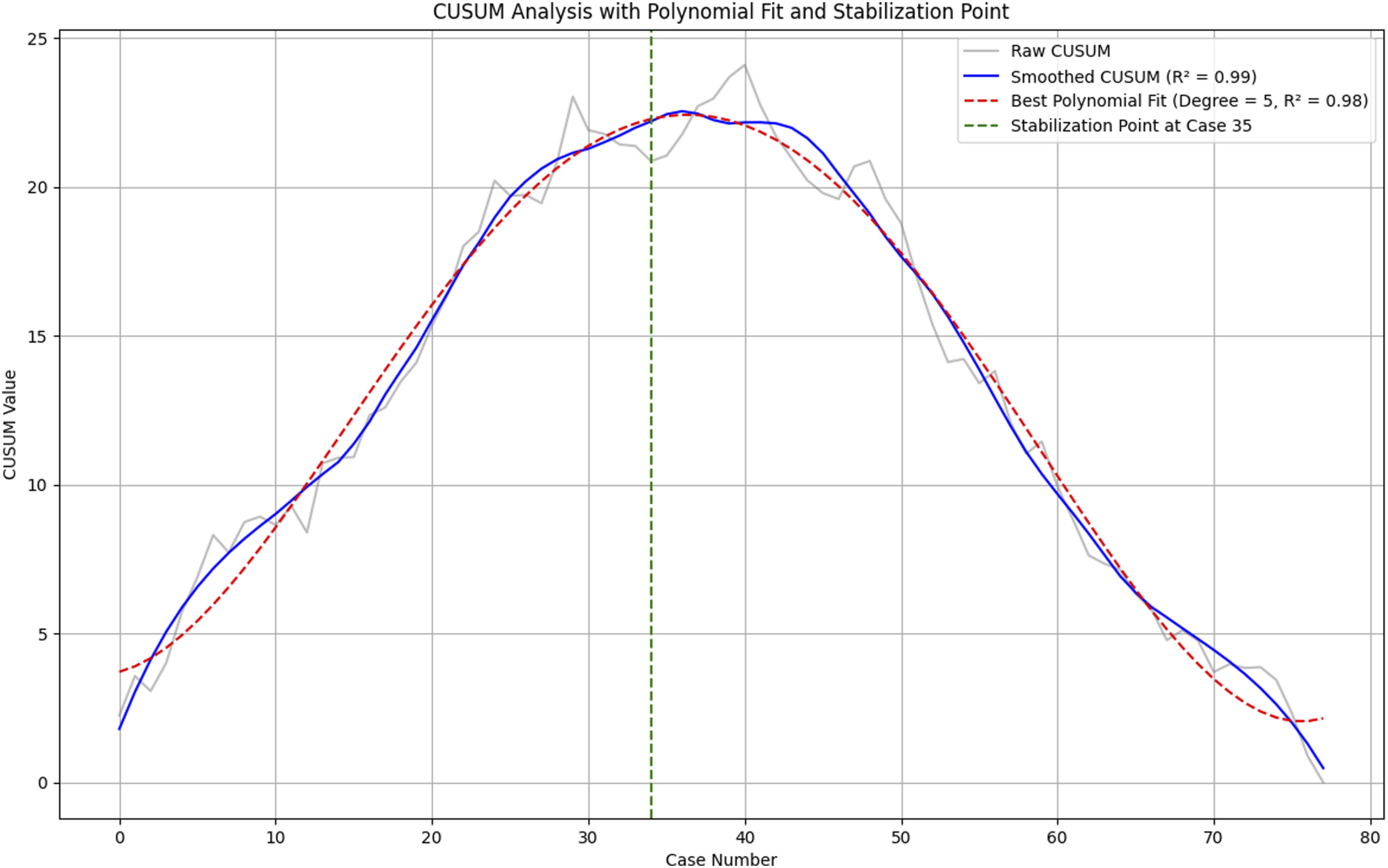
This Table Compares Surgical Time and Total Root Irritations Before and After Stabilization Points Identified Through CUSUM and RACUSUM Analyses
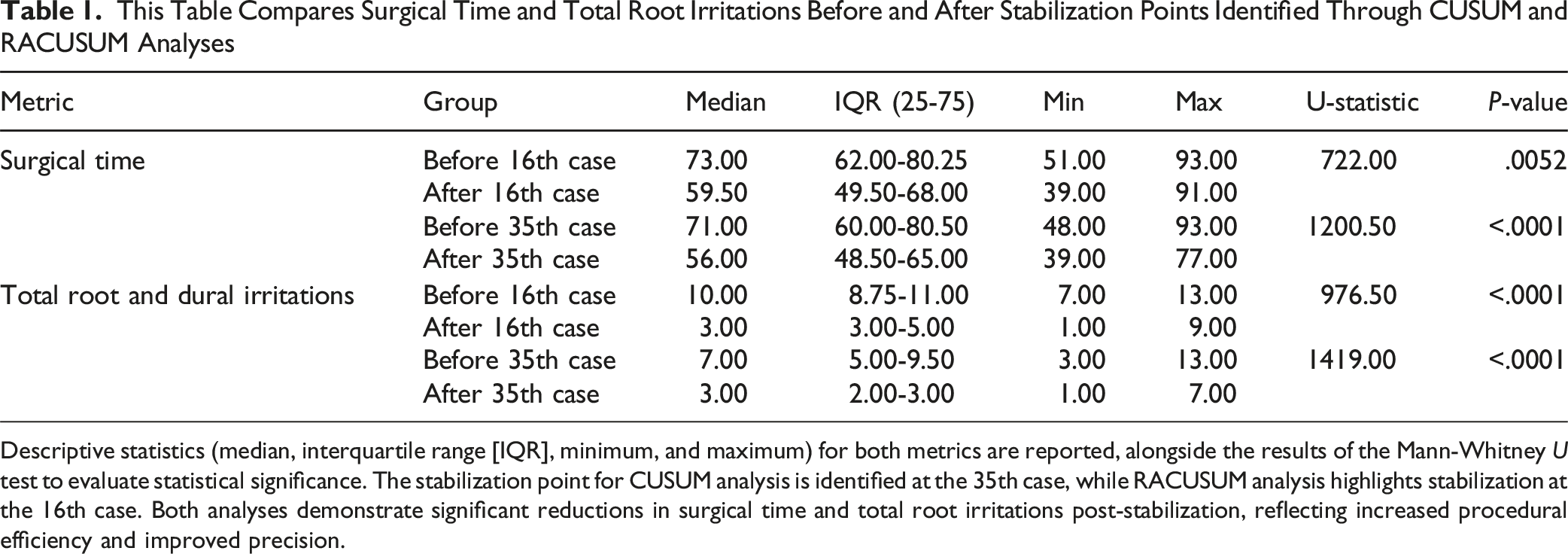
Descriptive statistics (median, interquartile range [IQR], minimum, and maximum) for both metrics are reported, alongside the results of the Mann-Whitney U test to evaluate statistical significance. The stabilization point for CUSUM analysis is identified at the 35th case, while RACUSUM analysis highlights stabilization at the 16th case. Both analyses demonstrate significant reductions in surgical time and total root irritations post-stabilization, reflecting increased procedural efficiency and improved precision.
In cases 1-35, decompressions were performed at L2–L3 in six patients, L3–L4 in eight patients, L4–L5 in 18 patients, and L5–S1 in three patients. In cases 36-78, the levels included L1–L2 in one patient, L2–L3 in five patients, L3–L4 in 14 patients, L4–L5 in 15 patients, and L5–S1 in eight patients. Statistical analysis revealed no significant difference in the distribution of operated levels between these two phases (P = .342). Additionally, we performed a separate comparison between the first 16 cases and cases 17-78. In the first 16 cases, decompression was performed at L2–L3 in three patients, L3–L4 in three patients, and L4–L5 in 10 patients. Among cases 17-78, the operated levels were L1–L2 in one patient, L2–L3 in eight patients, L3–L4 in 19 patients, L4–L5 in 23 patients, and L5–S1 in 11 patients. Again, no statistically significant difference was found between these groups (P = .200). The pain relief for cases 1-35 was found to be a median of 6 (range: 3 to 8), and for cases 36-78, the median was also 6 (range: 5 to 8), with no statistically significant difference between the two periods (P = .127). Similarly, the median pain relief for cases 1-16 was 6 (range: 3 to 8), and for cases 17-78, the median was also 6 (range: 3 to 8), with no statistically significant difference observed (P = .647)
Discussion
Spinal stenosis is defined as the narrowing of the spinal canal, lateral recess, or neural foramen due to bone or soft tissue compression. Treatments vary based on the severity of the disease. In patients with mild symptoms, conservative treatments may be attempted, while surgical interventions are applicable in cases where conservative measures fail and symptoms are advanced. The goal of surgical treatment is to alleviate compression, which can be addressed through open surgeries, microscopic surgeries, and percutaneous transforaminal surgeries. In cases without stability issues, decompression with fixation is not considered superior to simple decompression. 10 Traditionally, open decompressive surgeries are the most commonly used method for treating lumbar spinal stenosis. However, the secondary tissue damage, pain, atrophy, and potential instabilities caused by large incisions in open surgeries can be concerning. To mitigate these issues, microscopic procedures have been defined, though their limitations include poor visualization and minor muscle-bone damage. 7 To further minimize secondary damages, single-portal procedures have been developed. Nonetheless, these procedures still have high complication rates and steep learning curves.7,11
UBE offers various advantages, the most significant being the elimination of the need for extensive soft tissue stripping and massive dissections. Instruments and scopes can be directly delivered to the target area through minimal soft tissue channels. UBE surgery also results in less destruction to facet joints and fewer complications, including postoperative pain. The use of two portals in UBE surgery—one for visualization and one for the procedure—provides a broad range of motion. It is feasible to successfully access the area even in obese patients. 5 The UBE technique also allows for visualization and magnification of the contralateral spinal canal and intervertebral foramen. Compared to percutaneous transforaminal endoscopic surgery, UBE involves less radiation exposure. 12 Damage to the multifidus muscle is also minimal. 13 Compared to microscopic surgeries, UBE has been observed to have shorter hospital stays and operation times, and higher success rates. 14 One of the significant advantages of UBE is that it can be performed with general arthroscopic lenses and requires less specialized equipment. However, despite similarities to arthroscopy, UBE involves working in an artificial rather than a true space, which can be a significant disadvantage for those new to the technique. 8 This technique requires the use of both hands, and surgeons without endoscopy-arthroscopy experience may face difficulties in achieving visualization, establishing triangulation, and manipulating surgical instruments. If not performed correctly, there is a risk of complications such as dural and nerve injuries. Therefore, achieving successful decompression while avoiding complications should be the primary goal of UBE. It is crucial for surgeons aiming for this goal to have an understanding of the learning curve to navigate the process and complete their training.
Research on learning curves can be conducted through graphical, grouping, cumulative, and regression methods. Among these, CUSUM analysis is a highly effective statistical method for measuring the learning curve. The CUSUM curve distinctly illustrates the temporal relationship between a surgeon’s proficiency in a specific surgical task and the chronological order of cases performed. The curve splits into two phases at its peak: all cases on the initial side of the peak take longer than the average operation time, while those on the other side are shorter. The number of cases needed to reach the peak reflects the surgeon’s learning phase, while the number of cases where the curve progresses negatively thereafter represents the mastery phase. According to our CUSUM curve, the case at the top of the curve is 35th cases. Therefore, cases 1-35 can be grouped as the learning phase, and cases 36-78 as the mastery phase. However, operation time alone is not sufficient to simulate the learning curve for a procedure. The ability to perform the procedure safely is one of the most critical factors in shaping the learning curve. Therefore, in this study, we conducted an RA-CUSUM analysis based on the amount of dural and root irritations in patients undergoing UBE surgery with neuromonitoring. RA-CUSUM incorporates the failure rate as a parameter in the analysis. In this study, neuromonitoring was used in all patients undergoing UBE surgery for spinal stenosis, and all dural and root stimulations obtained during neuromonitoring were defined as risks. The analyses showed that the surgeries stabilized in terms of irritations by the 16th case. The irritation rate in the learning phase cases (1-16) was significantly lower than in the mastery phase cases (17-78).
There are different opinions in the literature regarding the learning curve for UBE. According to Park et al, the learning curve for UBE interventions for spinal stenosis was completed by the 58th case. 7 Chen et al suggest that 24 cases provide sufficient experience for the learning curve. 5 Wang et al 8 researched the learning curve on fresh frozen cadavers across three surgeons and determined that the learning curve was completed between the 16th and 20th cases. Guo et al 6 reached the mastery phase by the 29th case in their CUSUM analyses, while RA-CUSUM analyses indicated reaching this goal only by the 41st case. Xu et al found in their RA-CUSUM analysis that at least 89 cases are needed to achieve a consistent success rate in surgery. 9 There are varying perspectives in the literature regarding the learning curve of UBE, with reports indicating that it may be completed at different stages. This variability may be attributed to the fact that previous studies often did not focus on a single pathology or procedure but rather included a range of applications such as disc herniation, spinal stenosis, transforaminal lumbar interbody fusion, or cadaveric simulations. Additionally, a surgeon’s prior experience in spinal surgery, as well as their familiarity with endoscopic or arthroscopic techniques, undoubtedly influences the progression and completion of the learning curve. The use of neuromonitoring in spinal surgeries is instrumental in recognizing and intervening in potential neurological injuries in a timely manner. 15 It is also utilized in minimally invasive spinal surgeries, providing surgeons with a sense of security by preventing possible complications.16-18 In our study, neuromonitoring was consistently applied across all cases. The initial CUSUM analysis indicated a stabilization point around the 35th case, suggesting a steady continuation of the learning process until this point. In the RA-CUSUM analysis, which incorporated neuromonitoring data related to root and dural irritations, an earlier stabilization around the 16th case was observed.-This suggests that neuromonitoring may have accelerated the learning process by providing real-time feedback and boosting the surgeon’s confidence during the early cases.- This focus on neuromonitoring data as a risk factor is due to its direct relevance in detecting complications that are critical during the learning curve of surgical procedures. While complications can be influenced by multiple factors, and the early stages of a learning curve are typically more prone to complications, it is noteworthy that in this study, no patients experienced a postoperative neuro-motor status that was worse than their preoperative condition. Moreover, while improvements in pain and the need for subsequent surgeries could also reflect the surgeon’s initial cautious approach due to inexperience, they may equally be influenced by the natural progression of degenerative conditions. Our clinical approach aims to decompress each case as thoroughly and safely as possible using endoscopic methods (Figure 1). Furthermore, we believe that the use of neuromonitoring, particularly in the initial surgeries, helps reduce the surgeon’s anxiety about potential complications, thereby providing a more secure environment to achieve necessary decompression. This methodological choice ensures a balanced interpretation of the learning curve by focusing on direct intraoperative risks rather than postoperative symptoms, which can be ambiguous and influenced by a variety of factors beyond the surgeon’s control. Moreover, the continued decrease in surgical time observed in the RA-CUSUM curve up to the 35th case indicates that proficiency was still improving, albeit at a slower rate. This underscores the role of neuromonitoring not only as a safety tool but also as a facilitator of accelerated learning, particularly in the initial cases. Based on these findings, we recommend the use of neuromonitoring during the first 15-20 cases to provide a secure environment for the surgeon, mitigate risks, and enhance the learning process. However, it is noteworthy that the surgeon who conducted the surgeries in this study later discontinued the use of neuromonitoring. This decision was influenced by the absence of complications in previous cases and the surgeon’s belief that he had completed the learning phase. This scenario highlights the evolving judgment and confidence of the surgeon as proficiency increased, demonstrating a critical evaluation of the necessity and benefits of continuous neuromonitoring. Therefore, we feel that integrating the use of neuromonitoring into the early stages of learning for UBE surgery is crucial for ensuring patient safety and enhancing surgical outcomes. This approach aligns with our study’s findings, where neuromonitoring played a significant role in the rapid stabilization of surgical techniques and outcomes. Clearly, neuromonitoring not only aids in avoiding complications but also significantly contributes to the surgeon’s mastery of new surgical techniques, such as UBE, which requires high precision and skill.
To enhance the learning curve for surgeons new to unilateral biportal endoscopy (UBE), we believe it is crucial for these surgeons to have prior experience in open spinal surgeries. Additionally, experience in arthroscopy and endoscopy can significantly aid in developing proficiency in UBE, as these skills translate well to the techniques used in UBE. Familiarity with the surgical instruments and their selection can also simplify the learning process. We recommend opting for a 0-degree scope over a 30-degree scope for initial cases, as it can facilitate easier image adaptation and enhance visibility during the procedure. Ensuring clean visualization is also crucial, which can be achieved with experienced anesthesiologists who are adept at maintaining appropriate blood pressure levels. The operating room team’s experience with spine and arthroscopy surgeries can expedite the setup process, enhancing the overall efficiency of surgical operations. Training on models and cadavers is also of paramount importance. Although the authors of this study have not experienced it first-hand, virtual reality (VR) training for UBE can be beneficial before real surgical procedures. This modern training tool can provide a realistic and interactive environment for surgeons to hone their skills without the risks associated with live surgeries. In terms of patient selection, it is advantageous to start with simpler cases that are relatively easier to manage. Managing patients with minimal degeneration and unilateral symptoms can be more straightforward, providing an ideal learning environment for early surgical cases. This strategy allows surgeons to gradually build confidence and skill while minimizing the risk of complications as they progress to more complex cases.
This study has significant limitations. The primary limitation is the inherent variability among spinal stenosis cases; each stenosis has a unique structure, and variations in patients’ anatomy, condition levels, general health, and the surgical team can limit the generalizability of the study’s findings. Furthermore, the use of UBE is not confined to spinal stenosis alone but extends to disc herniations, 5 fractures, 19 cystic structures, 20 epidural abscess treatments, 21 lumbar interbody fusions, 22 and revision surgeries. 23 Therefore, the learning curve established in this study for UBE cannot be universally applied to all its indications. Another limitation of this study is the absence of dural tears in this case series, despite the fact that dural tears are a significant and limiting complication in UBE surgery. One of the key limitations of this study concerns its generalizability, as the surgeon performing the procedures has over 10 years of experience in microscopic and open spine surgeries, as well as arthroscopic procedures, which are critical for achieving triangulation in UBE. Studies like this are often based on cases handled by experienced spinal surgeons, and the learning curve data presented might overly encourage early-career surgeons. Consequently, surgeons with less experience should proceed with caution, as the learning curve values demonstrated here might not be directly replicable in their practices without similar levels of experience and guidance.
To conclude, in UBE surgery mastery is typically achieved by the 16th case in terms of managing irritations and by the 35th case in terms of operational time efficiency. These findings are particularly valuable for surgeons with experience in spinal surgeries who are new to the UBE technique, as they provide crucial feedback and guidance on the progression of surgical proficiency. However, the results might not be fully applicable to surgeons lacking prior experience in spinal surgeries, emphasizing the necessity of foundational skills in overcoming the complexities inherent in UBE procedures. This study highlights the critical need for ongoing research and tailored training that takes into account a surgeon’s specific background and experience to enhance outcomes in UBE surgeries.
Footnotes
Ethical Approval
This study was performed in line with the principles of the Declaration of Helsinki. Ethical approval was obtained from the Selcuk University Local Ethics Committee (Date: 25.02.2025, Number: 2025/104).
Author Contributions
A.G. Conceptualization, Methodology, Formal analysis, Writing - Original Draft, Writing - Review & Editing, Project administration, E.E. Investigation, Methodology, Writing - Original Draft, Writing - Review & Editing, S.C. Methodology, Formal analysis, Writing - Original Draft, S.S. Methodology, Formal analysis, Writing - Original Draft, Resources, A.Ö. Data Curation, Resources, Writing - Original Draft , F.D. Data Curation, Resources, Writing - Original Draft, Validation, B.K.A Writing - Original Draft, Writing - Review & Editing, Supervision. All authors have read and approved the final submitted manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available due to the fact that they involve patient-related information obtained from the hospital but are available from the corresponding author on reasonable request.
