Abstract
Study Design
Multicenter retrospective cohort study.
Objectives
Cement-augmented pedicle screws (CAPS) enhance fixation in osteoporotic patients, but carry a risk of cement leakage (CL), potentially leading to pulmonary embolism and neurological deficits. This study aimed to investigate the incidence and classification of CL and identify risk factor.
Methods
We included 140 patients undergoing thoracic or lumbar spinal fusion with CAPS at 7 institutions. CL was assessed by postoperative CT and classified as type S (segmental vein), type B (basivertebral vein), or type I (malpositioned screws). Multivariate logistic regression was performed to identify independent risk factors for intravenous CL.
Results
CL was observed in 88/140 patients (62.9%) and 190/650 screws (29.2%). Type S leakage was most common (77.9%), followed by type B (11.6%) and type I (7.9%). No symptomatic pulmonary cement embolism occurred; however, 1 patient experienced CL-related nerve root injury. Lower femoral neck T-score (aOR: 0.61; P = 0.009) was an independent risk factor for intravenous CL, while diffuse idiopathic skeletal hyperostosis was protective (aOR: 0.24; P = 0.009). Type S leakage was associated with shorter distance from the cement hole to the vertebral sidewall (aOR: 0.77; P < 0.001) and smaller vertebral body cross-sectional area (aOR: 0.87; P = 0.030). Type B leakage was associated with shorter distance to the vertebral posterior wall (aOR: 0.71; P < 0.001) and lower pedicle Hounsfield units (aOR: 0.99; P = 0.041).
Conclusions
Asymptomatic CL was frequently observed following CAPS fixation. Preoperative imaging assessments and precise screw placement may reduce the CL risk.
Keywords
Introduction
Pedicle screw fixation is a cornerstone of spine surgery, particularly for spinal stabilization in degenerative disease, trauma, infection, tumor, or deformities. However, in patients with compromised bone quality, conventional pedicle screw fixation may be insufficient due to poor screw purchase and increased risk of loosening, potentially leading to mechanical failure and revision surgery.1,2 Among the various augmentation techniques proposed to address this issue, cement-augmented pedicle screws (CAPS) have emerged as a viable solution to enhance screw stability by injecting bone cement into the vertebral body. 3 CAPS have been shown to significantly improve screw pull-out strength, resulting in reduced rates of screw loosening and revision surgery, especially in osteoporotic patients.4-6
Despite these biomechanical and clinical advantages, CAPS carries the inherent risk of cement leakage (CL). CL is a common complication, which can occur into the epidural space, vertebral veins, or even the pulmonary circulation. Of particular concern is intravenous CL, which has been linked to serious complications such as pulmonary embolism and neurological deficits. Previous studies have identified several risk factors for intravenous CL, including lower bone mineral density (BMD), a higher number of augmented screws, and increased cement volume.7-9 However, detailed imaging analysis and patient-specific risk factors remain underexplored in the literature.
Recently, novel imaging-based methods for evaluating spinal bone quality, such as Hounsfield units (HU) on computed tomography (CT) and vertebral bone quality (VBQ) scores on magnetic resonance imaging (MRI), have gained attention as potential alternatives to dual-energy X-ray absorptiometry (DEXA) for assessing BMD.10,11 These methods provide the advantage of localized bone quality assessment at the site of instrumentation. Given the known association between poor bone quality and CL risk, we hypothesized that HU and VBQ scores could serve as predictive tools for intravenous CL.
This study aimed to investigate the incidence and classification of CL in patients undergoing spinal fusion surgery with CAPS and to identify specific risk factors contributing to intravenous CL. By integrating detailed imaging analysis with clinical and surgical variables, we sought to improve the safety profile of CAPS in spine surgery.
Materials and Methods
Patients
This multicenter, retrospective cohort study was approved by our institutional review board (Approval No. E24-0465). Given the retrospective nature of the study and the use of de-identified data, the requirement for informed consent was waived by the ethics committee. We included 140 consecutive patients who underwent thoracic or lumbar spinal fusion with CAPS between March 2022 and January 2025 at 7 institutions, including 4 university and 3 community hospitals. There were no exclusion criteria applied beyond the need for complete clinical and radiographic data. CAPS were primarily indicated in patients with compromised bone quality to enhance screw fixation. Diagnoses included osteoporotic vertebral fracture in 112 cases (80%), pyogenic spondylitis in 9 cases (6.4%), lumbar spinal stenosis in 7 cases (5%), spinal deformity in 6 cases (4.3%), and spinal tumor in 6 cases (4.3%). A total of 650 CAPS instrumentations were performed.
Data Collection
We collected baseline demographic data, including age, sex, body mass index (BMI), smoking status, alcoholism, American Society of Anesthesiologists (ASA) classification, diabetes, dyslipidemia, chronic kidney disease, hemodialysis, hematological disease, rheumatoid arthritis, long-term steroid use (>3 months), osteoporosis treatment, DEXA T-score of the lumbar spine and femoral neck, and diffuse idiopathic skeletal hyperostosis (DISH). DISH was evaluated according to the Resnick classification, which defines DISH as continuous bony bridging involving 4 or more vertebral bodies. 12 Additionally, we recorded operative parameters and perioperative complications, including the number of fused levels, number of CAPS utilized, operative time, estimated blood loss, dural tear, nerve root injury, epidural hematoma, surgical site infection, symptomatic pulmonary cement embolism (PCE), and revision surgery. Indications for revision surgery included screw misplacement, screw loosening, surgical site infection, and the need for extended fixation or decompression.
Operative Procedures
Posterior instrumentation was performed using either a conventional open approach or a minimally invasive percutaneous technique at the experienced surgeon’s discretion. CAPS placements were conducted utilizing the Expedium Verse® or Viper Prime® Fenestrated Screw System with Vertecem V+ cement (DePuy Synthes, MA, USA). The screw design has 3 fenestrations at 120-degree intervals, centered at 8 mm from the tip for screws ≤40 mm, and 6 fenestrations centered at 8 and 14 mm for screws ≥45 mm. Polymethylmethacrylate (PMMA) was injected through the fenestrated screws under serial lateral fluoroscopic guidance with a target cement volume of 1 to 2 mL per screw. Cement injection was discontinued if CL was observed. Operative data were collected, including screw position (right or left), location of augmented vertebrae (thoracic, lumbar, or sacrum), screw diameter, screw length, and cement volume.
CT Assessments
All patients underwent preoperative and immediate postoperative CT scans. CL was defined as cement leakage extending beyond the vertebral body in at least 2 consecutive slices on postoperative CT imaging. The classification of CL followed the report of Guo et al,
7
categorizing 3 types: type S (leakage into segmental veins), type B (leakage into the spinal canal via basivertebral veins), and type I (leakage through malpositioned fenestrated screw instrumentation) (Figure 1). Patients with any type of CL were categorized into the CL group, while those without CL were assigned to the non-CL group. Postoperative CT measurements included screw tip position (lateral or medial relative to the inner edge of the pedicle), distance from the cement fenestration hole to both the vertebral body sidewall and posterior wall. The cement fenestration hole was determined at 8 and 14 mm from the tip for screws ≤40 mm and ≥45 mm, respectively. Additionally, we measured the vertebral body cross-sectional area (CSA) and HU values along the pedicle screw trajectory on preoperative axial CT images using an elliptical region of interest. Mean HU values were also calculated for the superior, middle, and inferior regions of the vertebral body as previously described.
10
Imaging was independently reviewed by trained spine surgeons at each institution. Training sessions with representative cases were held to standardize assessments. Cases with uncertain classification were jointly reviewed by experienced senior spine surgeons. Postoperative CT Images Illustrating the Classification of CL. (A) Type S: Leakage Into the Segmental Veins. (B) Type B: Leakage Into the Spinal Canal via Basivertebral Veins. (C) Type I: Leakage Through Malpositioned Fenestrated Screw Instrumentation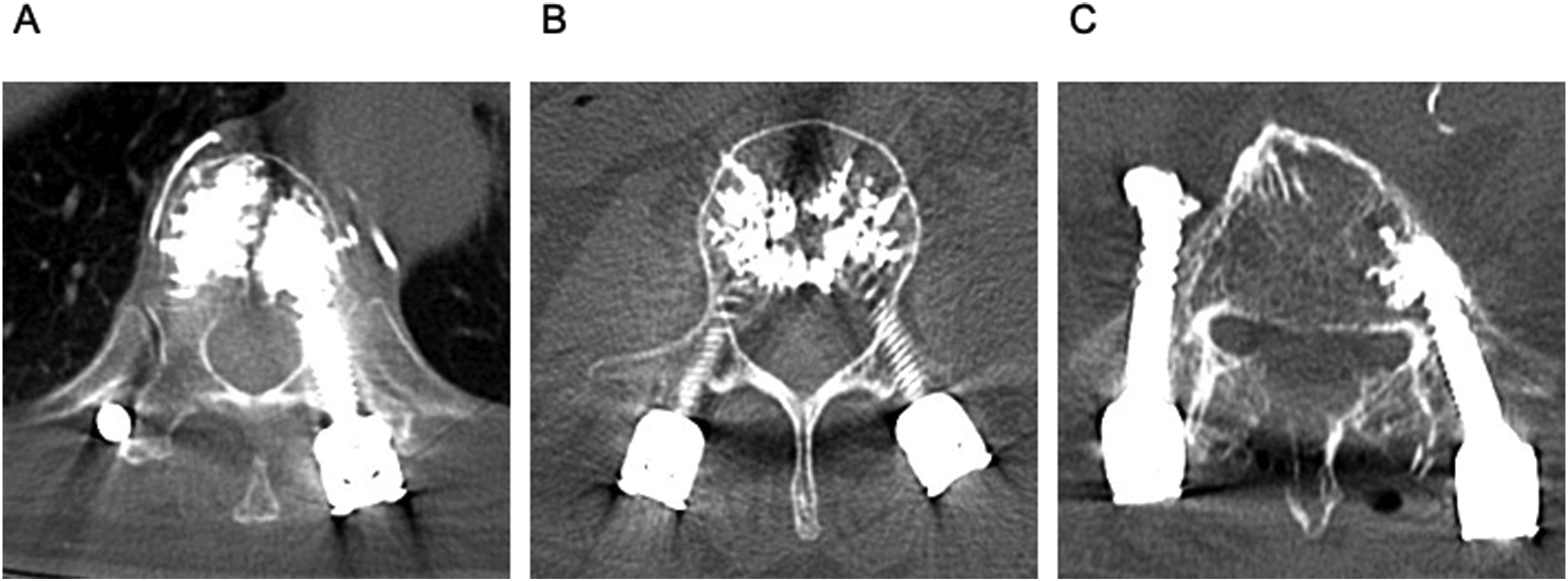
VBQ Score Calculations
The VBQ score was determined using preoperative non-contrast T1-weighted MRI, as previously reported. 11 Signal intensities (SI) within the medullary regions of each vertebral body and the cerebrospinal fluid (CSF) space were measured on mid-sagittal slice. The VBQ score was then calculated as the ratio of the average SI of each vertebra to the SI of CSF. If the vertebral region was not clearly visible on the mid-sagittal slice due to scoliosis, a parasagittal slice was used. While the original VBQ method utilizes the median of L1-L4, our study measured scores for each instrumented vertebra.
Statistical Analysis
Descriptive analyses were performed to assess the incidence and classification of CL. Normality of distribution for continuous variables was assessed using the Shapiro-Wilk test. Depending on the distribution, comparisons were performed using the Student’s t-test or Mann-Whitney U test. Categorical variables were compared using the chi-square test. To explore independent risk factors for intravenous CL and its subtypes (type S and type B), univariate logistic regression analyses were initially conducted to assess potential associations. Variables considered clinically relevant and those previously identified as risk factors in the literature, regardless of univariate significance, were included in the multivariable logistic regression models to ensure robust model specification and minimize bias from data-driven selection. P < 0.05 was considered statistically significant. All statistical analyses were conducted using JMP Pro software (version 18.0; SAS Institute, NC, USA).
Results
Incidence and Classification of CL
Cement leakage was observed in 88/140 patients (62.9%) and in 190/650 screws (29.2%). Among the 190 screws, type S was identified in 148 screws (77.9%), type B in 22 screws (11.6%), type S + B in 5 screws (2.6%), and type I in 15 screws (7.9%). The distribution of CAPS-instrumented vertebrae is shown in Figure 2. The Distribution of CAPS Instrumented Vertebrae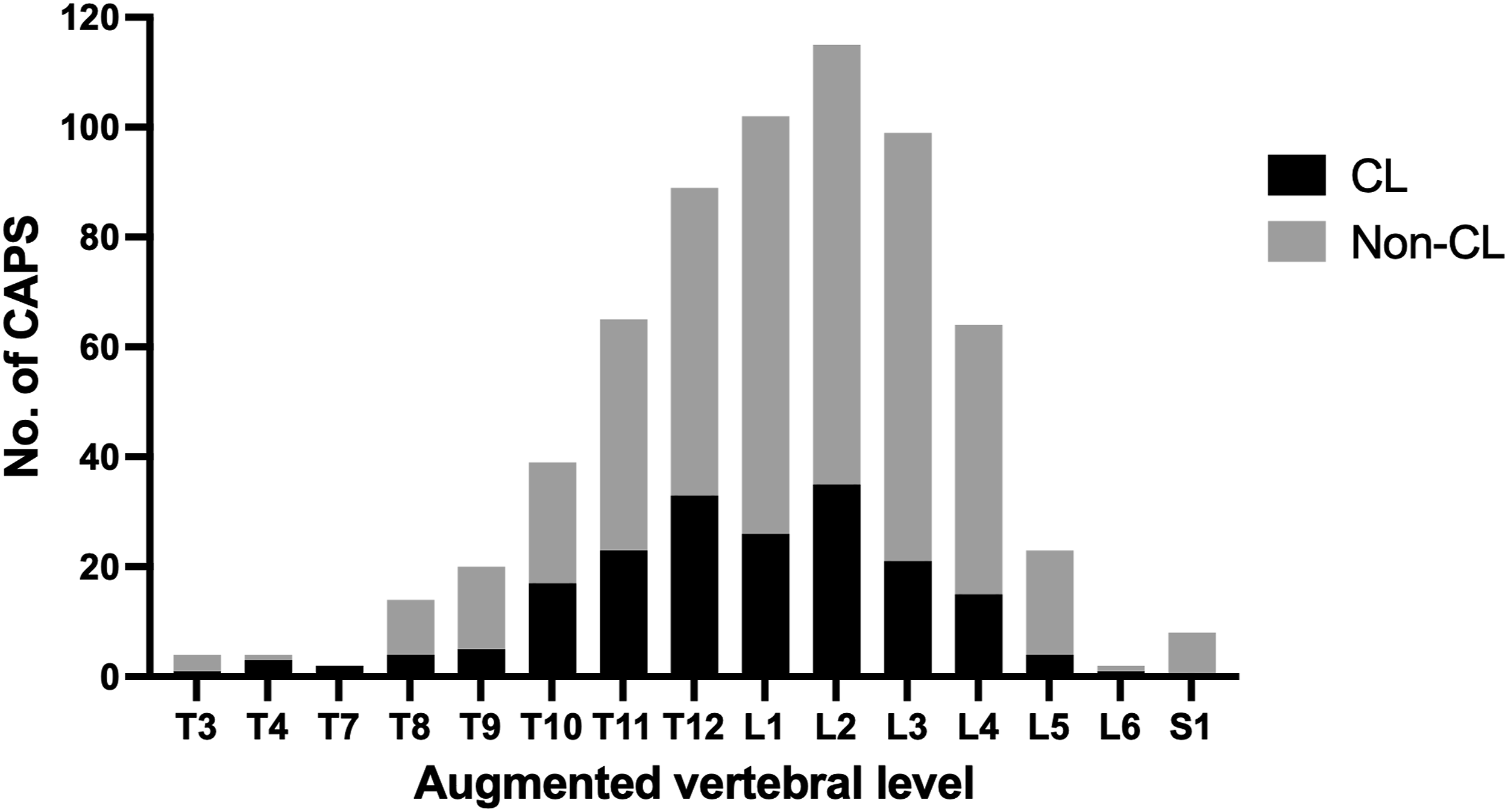
Patient Demographics and Perioperative Data
Patient Demographics (N = 140)
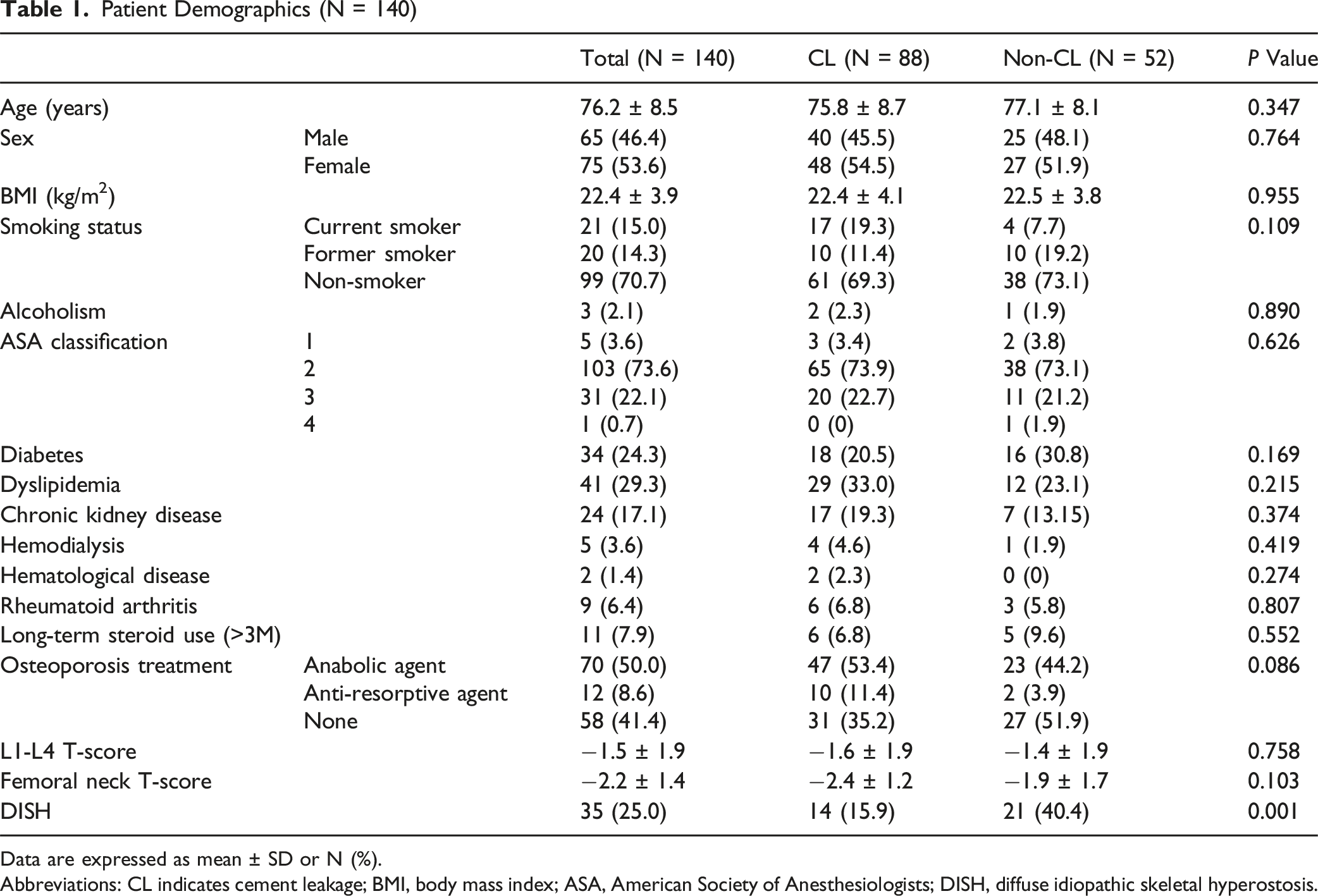
Data are expressed as mean ± SD or N (%).
Abbreviations: CL indicates cement leakage; BMI, body mass index; ASA, American Society of Anesthesiologists; DISH, diffuse idiopathic skeletal hyperostosis.
Perioperative Data

Data are expressed as mean ± SD or N (%).
Abbreviations: CL indicates cement leakage; CAPS, cement-augmented pedicle screw.
Patient-Specific Risk Factors for Intravenous CL
Multivariate Logistic Regression Analysis for Intravenous CL Patients
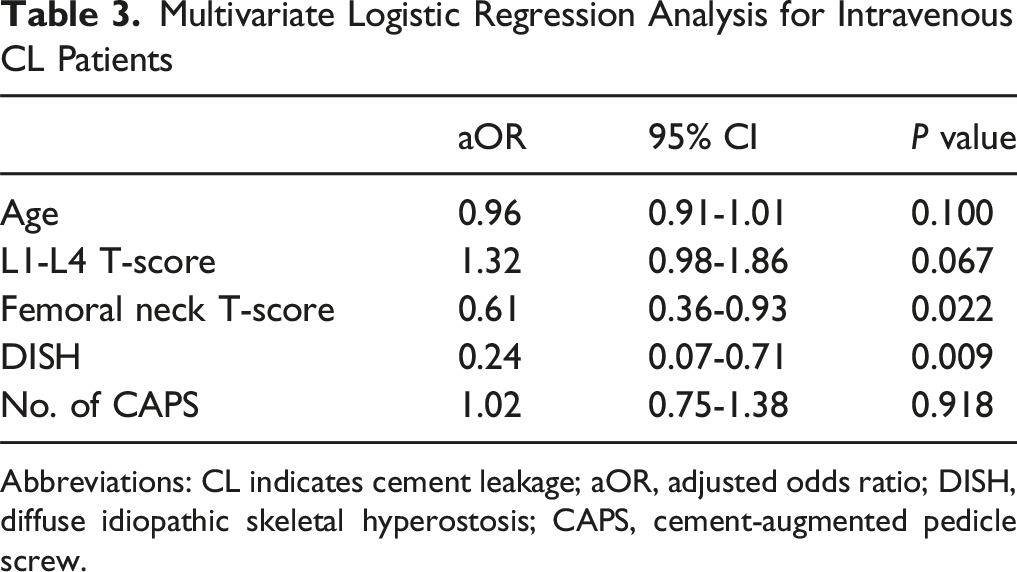
Abbreviations: CL indicates cement leakage; aOR, adjusted odds ratio; DISH, diffuse idiopathic skeletal hyperostosis; CAPS, cement-augmented pedicle screw.
Risk Factors for Type S Leakage
Univariate Logistic Regression Analysis for Type S and Type B Leakage Screws
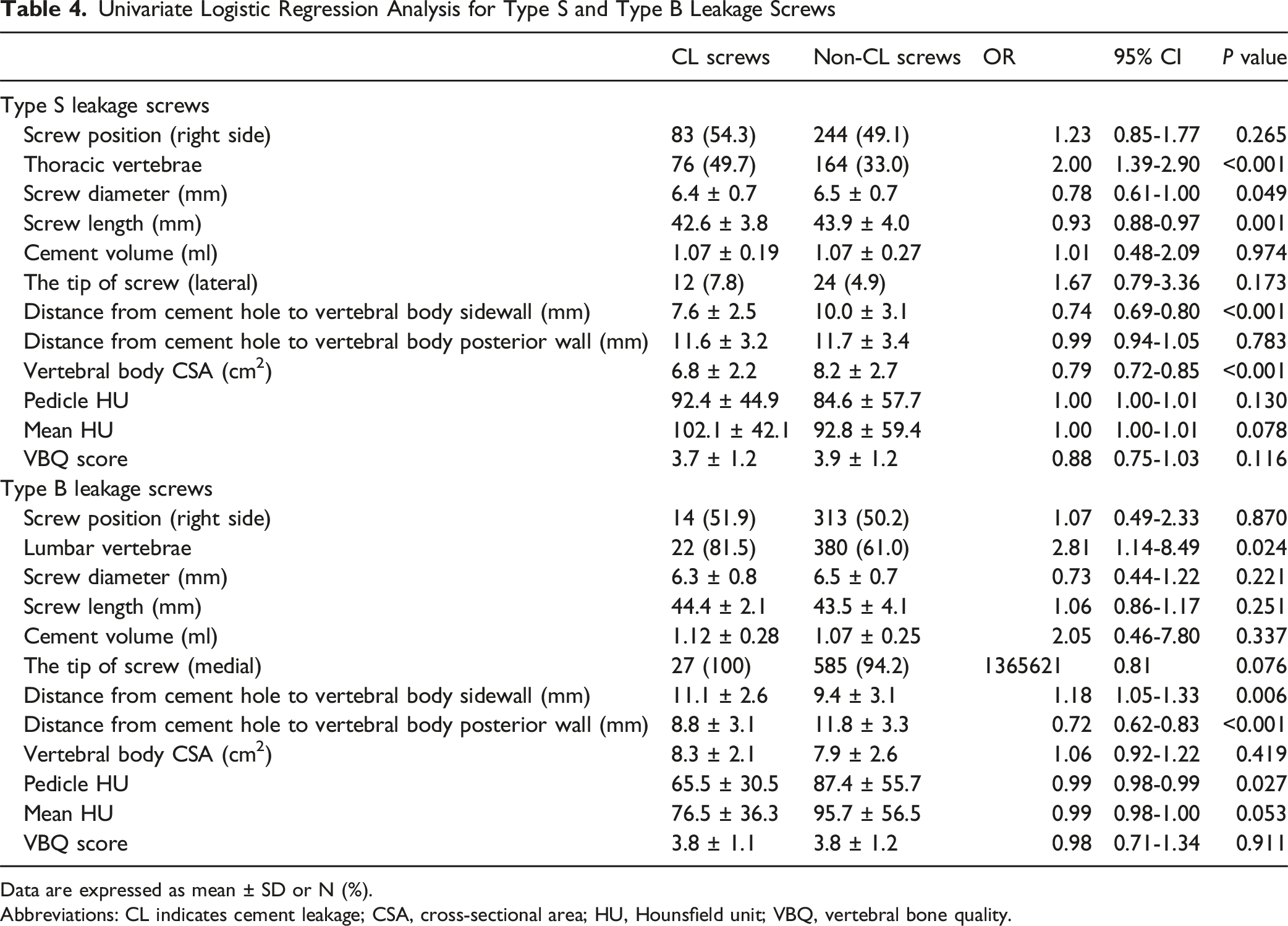
Data are expressed as mean ± SD or N (%).
Abbreviations: CL indicates cement leakage; CSA, cross-sectional area; HU, Hounsfield unit; VBQ, vertebral bone quality.
Multivariate Logistic Regression Analysis for Type S and Type B Leakage Screws
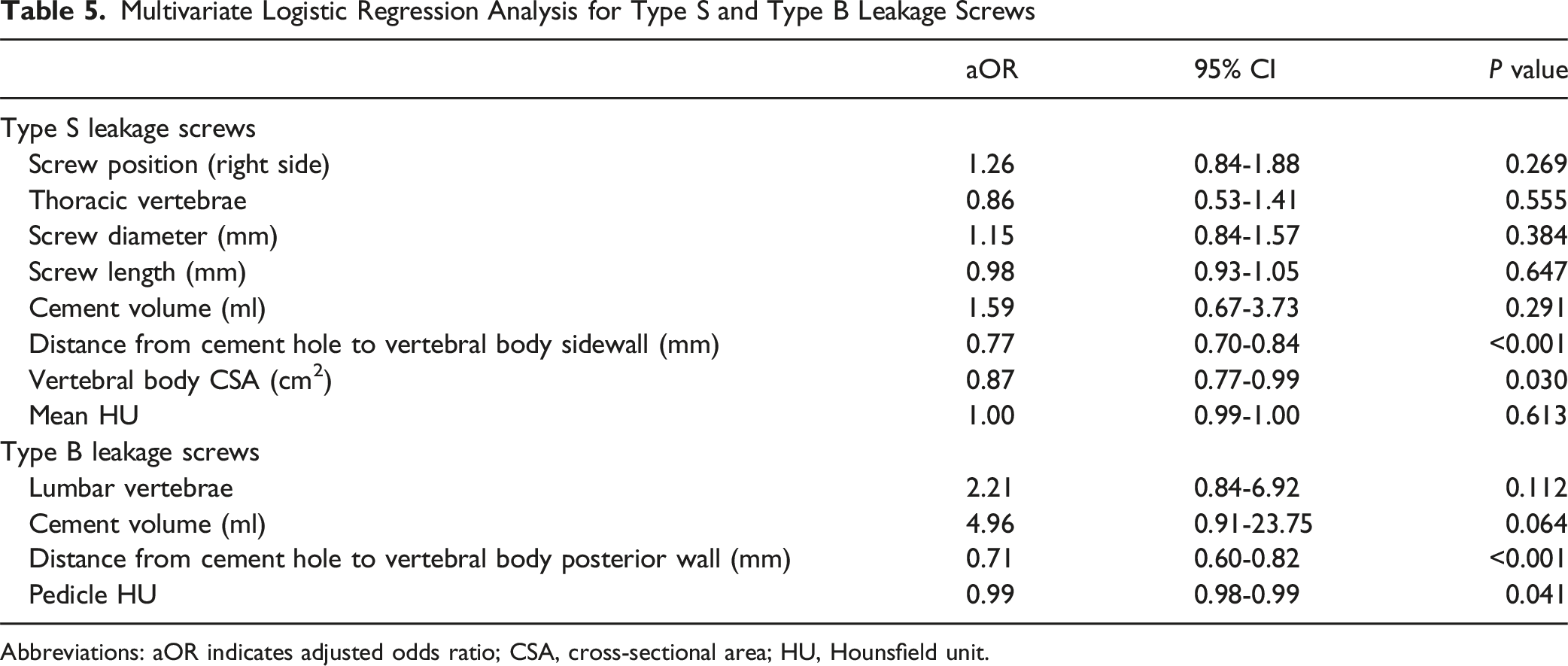
Abbreviations: aOR indicates adjusted odds ratio; CSA, cross-sectional area; HU, Hounsfield unit.
Risk Factors for Type B Leakage
Univariate analysis showed that lumbar vertebrae, longer distance from cement hole to vertebral body sidewall, shorter distance from cement hole to vertebral body posterior wall, and lower pedicle HU were significant risk factors for type B leakage (Table 4). Multivariate analysis identified shorter distance from cement hole to vertebral body posterior wall (aOR: 0.71; 95% CI: 0.60-0.82; P < 0.001) and lower pedicle HU (aOR: 0.99; 95% CI: 0.98-0.99; P = 0.041) as independent risk factors for type B leakage (Table 5).
Discussion
This study demonstrated the incidence and classification of CL in patients undergoing spinal fusion with CAPS, revealing that CL occurred in 62.9% of patients and 29.2% of augmented screws. Notably, type S leakage was the most prevalent, followed by type B and type I leakage. Furthermore, we found that the presence of DISH was independently associated with a reduced risk of intravenous CL, while the femoral neck T-score emerged as a critical patient-specific risk factor. Additionally, this study underscored the significance of screw trajectory, particularly the distance from cement hole to the lateral and posterior walls of the vertebral body. Moreover, vertebra-specific risk factors, such as smaller vertebral body CSA and lower pedicle HU, were identified as key determinants of type S and B leakage, respectively. These findings provide valuable insights that may contribute to reducing intravenous CL and enhancing the safety of spinal fusion surgery utilizing CAPS.
The reported incidence of CL in patients undergoing spinal fusion with cement-augmented fenestrated pedicle screws varies considerably across studies, with rates ranging from 9.3% to 95.7%. These variations stem from differences in patient characteristics, surgical indications, operative techniques, and the definitions and imaging modalities used to identify CL.8,13-20 The lack of a standardized definition across studies contributes to this heterogeneity. In this study, CL was rigorously defined as cement leakage extending beyond the vertebral body on at least 2 consecutive CT slices obtained immediately postoperatively. While a meta-analysis reported a pooled CL prevalence of 34.4% (95% CI: 1.8%-66.9%), 6 our study observed a relatively higher incidence with 62.9% of patients experiencing CL. Additionally, our reported incidence of 29.2% of augmented screws is consistent with published rates ranging from 8.9% to 45.8%.8,18,20-23 Moreover, the distribution patterns of type S and type B leakages observed in this study align with previous studies,7-9,24 indicating that type S leakage is more common in thoracic vertebrae, whereas type B leakage occurs more frequently in lumbar vertebrae.
Regarding patient-specific risk factors for intravenous CL, this study reinforces the association between lower BMD and increased CL risk, as previously documented.8,20,25 Furthermore, we present a novel finding: the presence of DISH was independently associated with a reduced risk of intravenous CL. This unique observation may be attributed to the increased rigidity of the vertebral cortical bone due to continuous bony bridging in DISH patients, which may serve as a barrier to cement migration. Alternatively, structural alterations in the vertebral venous system may lead to impaired venous reflux, but further studies are needed to elucidate these mechanisms. While previous studies have indicated that a greater number of augmented screws is a risk factor for CL,7-9 this study did not find a significant association. A plausible explanation for this discrepancy is that the majority of cases in our cohort involved 4 augmented screws, thereby minimizing variability in the number of screws and its potential impact on CL risk.
Regarding risk factors for type S leakage, this study identified smaller screw diameter and shorter screw length as potential contributors. These findings suggest that inserting greater diameter and longer length screws may help mitigate type S leakage. Notably, multivariate analysis revealed that a shorter distance from cement hole to vertebral body sidewall was an independent risk factor, emphasizing the importance of precise screw placement. Additionally, a smaller vertebral body CSA was independently associated with an increased risk of type S leakage, indicating that smaller vertebrae have a reduced capacity to contain injected cement. Previous studies have reported higher cement volume and thoracic vertebrae as independent risk factors for type S leakage7,8; however, our study did not find a clear association. Based on the study by Weiser et al, 26 which demonstrated no significant difference in screw stability between augmentation with 1 mL and 3 mL of cement, most cases in this study employed 1 mL of cement per augmented screw. Our results suggest that when a standardized cement volume is used, vertebral morphology, particularly CSA, may have a greater influence on the risk of type S leakage than the anatomical factor of thoracic vertebrae.
Regarding risk factors for type B leakage, this study identified a shorter distance from cement hole to vertebral body posterior wall as an independent risk factor. This finding corroborates previous studies showing that shallower screw implantation and a smaller distance between the screw tip and vertebral body midline are significant predictors for epidural CL.7,9 This may be attributed to the anatomical structure of the basivertebral veins, which course horizontally through the center of the vertebral body and converge posteriorly to drain into the ventral internal venous plexus.27,28 Furthermore, this study identified lower pedicle HU values as an independent risk factor for type B leakage. HU is a well-established indicator of BMD, and lower HU values indicate compromised bone integrity. 10 This finding suggests that weaker trabecular structures allow greater cement migration, increasing the risk of leakage into the basivertebral venous system. Given this, preoperative assessment of pedicle HU could serve as a valuable tool to predict and mitigate the risk of type B leakage. Moreover, other studies have reported additional risk factors for Type B leakage, including higher cement volume, lumbar vertebrae, and the presence of magistral-type basivertebral foramen, suggesting that vertebral morphology plays a crucial role in leakage patterns.7-9
PCE and neurological complications associated with CL represent the most serious adverse events in CAPS fixation. A review of 1974 cases reported a 6% incidence of PCE, with symptomatic cases accounting for 1.3%. 29 The variability in PCE rates is attributed to differences in patient selection, cement injection technique, and imaging modalities. Additionally, thermal injury from cement polymerization, mechanical compression of neural structures, and inflammatory responses contribute to neurological complications. In this study, no symptomatic PCE cases were observed. However, 1 patient (0.7%) developed nerve root injury due to CL into the spinal canal associated with type I leakage. Notably, all cases of type S and B leakage were asymptomatic, highlighting the critical importance of accurate screw placement.
This study has several limitations. First, as a multicenter retrospective study, inherent selection bias and variations in surgical technique across institutions may influence the findings. Second, although imaging was reviewed by trained surgeons at each institution after standardized instruction, interobserver variability may have affected the classification of CL. Third, this study primarily focuses on immediate postoperative CL and related complications, and does not evaluate long-term complications such as adjacent vertebral fractures and screw loosening. Fourth, variations in cement viscosity and injection speed among surgeons were not fully standardized, which could impact the CL rates. Finally, routine postoperative chest CT was not performed, so asymptomatic PCE cases may have been overlooked.
Conclusions
This study shows a high incidence of asymptomatic CL, occurring in 62.9% of patients and 29.2% of augmented screws, with type S leakage being the most prevalent. Notably, we were the first to identify that the presence of DISH was independently associated with a reduced risk of intravenous CL. Furthermore, our findings underscore the critical importance of careful screw placement with optimal pedicle trajectory and preoperative imaging assessments (including vertebral morphology and HU values) to mitigate the risk of CL. Future prospective studies with longer follow-up are warranted to further validate these results and assess the long-term clinical outcomes of CAPS fixation.
Footnotes
Ethical Approval
This study was approved by the institutional review board (Approval No. E24-0465).
Author Contribution
All authors contributed to the conception and design of the study. Material preparation, data collection, and analysis were performed by S.T., H.T., A.I., R.Y., T.S., K.I., T.T., T.K., and Y.I. The first draft of the manuscript was written by S.T., and all authors commented on previous versions of the manuscript. All authors read and approved the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available, but are available from the corresponding author upon reasonable request.
