Abstract
Study Design
Prospective genetic cohort study.
Objective
Adolescent idiopathic scoliosis (AIS) is a common spinal disorder affecting individuals aged 10-18 years without other underlying health conditions. This study aimed to examine the potential etiologic association between AIS and polymorphisms in the LBX1 (rs11190870, rs625039, rs11598564), TIMP2 (rs8179090), GPR126 (rs6570507), and CHD7 (rs121434341) genes in Turkish patients. Additionally, the relationships of these polymorphisms with sex, age, age at diagnosis, and Cobb angle were evaluated.
Methods
The study included 301 individuals: 201 patients with AIS (aged 10-18 years, Cobb angle ≥10°, no genetic disorders or related diseases), and 100 healthy controls (aged 10-18 years, no scoliosis diagnosis). The study analyzed rs625039, rs11598564, rs6570507, rs121434341, rs11190870, and rs8179090 polymorphisms in patients with AIS and controls using RT-PCR, confirmed the SNP regions through DNA sequencing, and performed statistical analysis.
Results
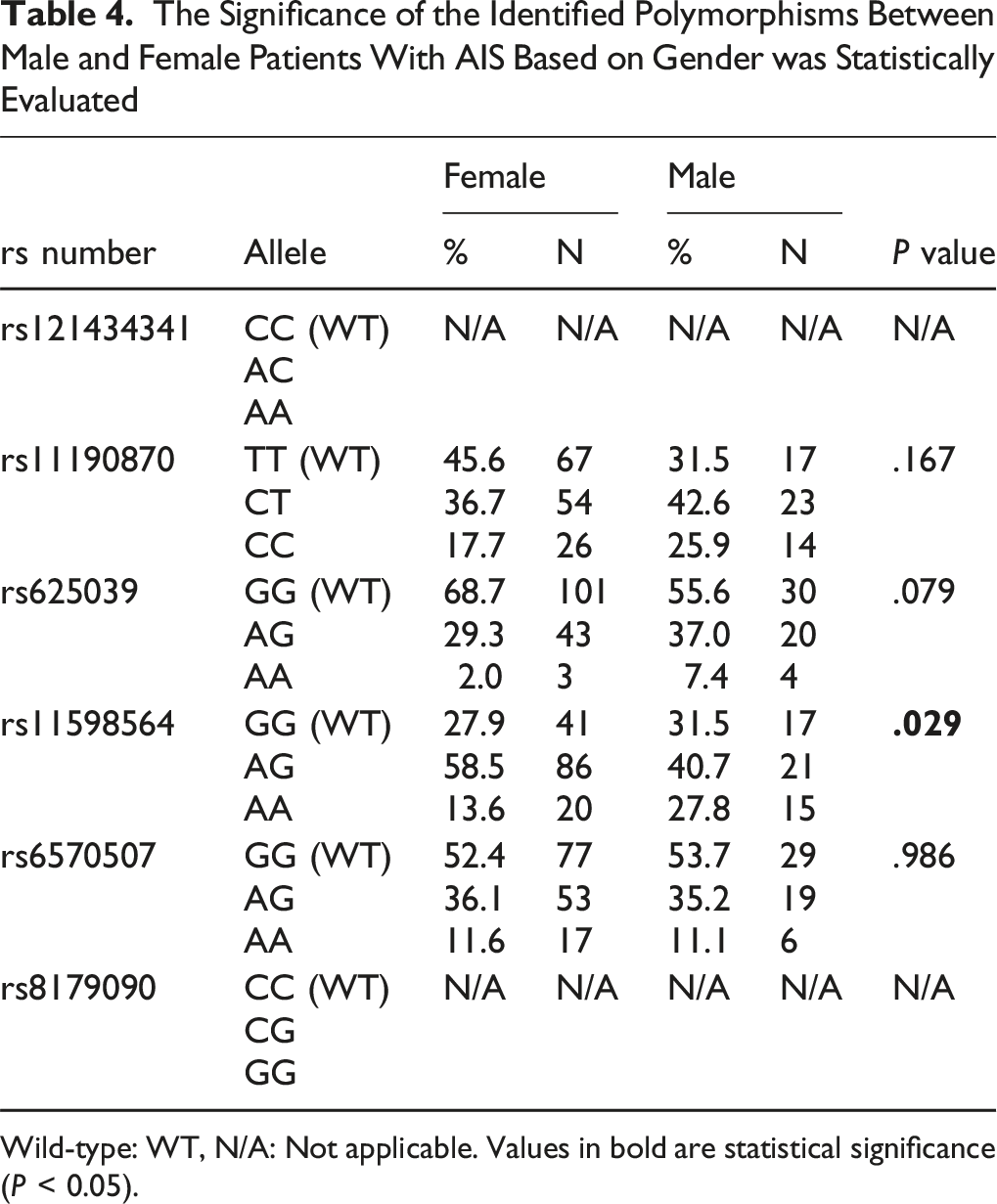
In this study, the rs11190870 polymorphism of the LBX1 gene demonstrated a statistically significant difference between patients with AIS and the control group (P < .001), but no significance was observed for the other polymorphisms analyzed. Sex-based analysis revealed a significant association for the LBX1 rs11598564 polymorphism, with a higher frequency observed in females (P = .029); no significant differences were identified for the other polymorphisms in terms of sex. The rs8179090 and rs121434341 polymorphisms, previously associated with AIS in other populations, showed no statistically significant association in the present study cohort.
Conclusion
The LBX1 gene rs11190870 polymorphism was found to be associated with AIS in Turkish patients.
Introduction
Adolescent idiopathic scoliosis (AIS) is the most prevalent nondegenerative spinal deformity in pediatric populations.1,2 Its prevalence, as determined through plain radiography, 3 is estimated to range between 1% and 4%.4-6 Sex and family history have been identified as significant risk factors associated with AIS. 7 Moreover, the distribution of the disease appears to vary across different populations. 8 Despite extensive research, the precise etiology of AIS remains unclear, with no definitive evidence pointing to either genetic or environmental causes. However, multiple studies have reported an increased prevalence of AIS in families with at least 1 affected first-degree relative, a finding corroborated by twin studies,7,9 suggesting a substantial genetic component in the pathogenesis of AIS. 10 Patients with AIS who remain untreated during the early stages of the disease, as characterized by the Cobb angle and skeletal maturity, typically experience worse clinical outcomes compared with those who undergo timely intervention. 11 Genetic studies hold the potential to identify AIS predisposition prior to the manifestation of overt clinical symptoms, enabling earlier diagnosis and therapeutic intervention.
Encoding ladybird homeobox 1 gene (LBX1) plays a critical role in neuromuscular development, and its dysregulation may contribute to imbalances in spinal alignment, thereby predisposing individuals to AIS.12,13 The replication of these findings in diverse populations, including Turkish individuals, is essential to confirm the role of LBX1 in the pathophysiology of AIS and its potential as a universal genetic risk factor. G protein-coupled receptor 126 (GPR126), a member of the adhesion G protein-coupled receptor family, is strongly associated with human height and skeletal growth. Heightened vertebral growth and skeletal asymmetry, hallmark features of AIS, suggest that dysregulated GPR126 activity could influence susceptibility. 14 Patients with AIS often exhibit disproportionate growth rates, particularly in the anterior vertebral column, which contributes to thoracic hypokyphosis, a key biomechanical factor in AIS progression.15-17 Tissue inhibitor of metalloproteinases 2 (TIMP2), a member of the tissue inhibitor of metalloproteinases family, regulates extracellular matrix remodeling through its inhibitory effects on matrix metalloproteinases (MMPs).18,19 Endochondral ossification, a critical process for vertebral development, relies on the tight regulation of matrix remodeling, and any disruption in this balance may lead to skeletal abnormalities.20,21 Chromodomain helicase DNA-binding protein 7 (CHD7) is associated with scoliosis in CHARGE syndrome, a condition involving growth and developmental anomalies. Although CHARGE syndrome represents a syndromic form of scoliosis, CHD7 variants may also contribute to idiopathic scoliosis.22,23 Investigating CHD7 polymorphisms in patients with AIS may reveal shared genetic mechanisms across syndromic and idiopathic cases.
This study aims to investigate whether there is an etiologic relationship between AIS and the polymorphisms of the LBX1, TIMP2, GPR126, and CHD7 genes, and determine the relationship between these polymorphisms and factors such as sex, age, age at diagnosis, and Cobb angle in these patients.
In this study, we conducted an analysis of genetic polymorphism in the LBX1 gene to assess its potential as a candidate gene for AIS susceptibility in Turkish patients, marking a first-time evaluation in this population. We performed extensive screening of LBX1 gene through direct sequencing to detect polymorphisms, and statistical analysis to examine the genetic effects on AIS in Turkish patients.
Materials and Methods
Study Design
In this study, blood samples were obtained from 201 patients with AIS and 100 control individuals following the acquisition of informed consent. The samples were collected in purple-capped EDTA tubes and stored at −80°C until the DNA isolation process was performed. The patient group included individuals aged 10 to 18 years who were diagnosed as having AIS, with a Cobb angle of 10° or greater on direct radiographs, and who had no known genetic disorders or conditions recognized to play a role in scoliosis etiology (such as degenerative, neuromuscular, or congenital scoliosis, among others). The control group consisted of healthy individuals aged 10 to 18 years with no scoliosis diagnosis, as determined through physical examinations and/or imaging studies. As part of the workflow in this study, DNA was isolated from the collected blood samples. Following isolation, DNA quality was assessed, and polymorphism analyses were performed using primers specific to the single nucleotide polymorphism (SNP) regions under investigation. This analysis was performed using a real-time polymerase chain reaction (RT-PCR) device, in accordance with the study protocol.
DNA Isolation
In this study, DNA isolation from peripheral blood samples, which were collected and stored at −80°C, was performed using the E.Z.N.A.® Blood DNA Mini Kit (Omega Bio-tek, USA) protocol. The isolated DNA was then diluted with 100 µL of ddH2O. The DNA samples were kept at 4°C until the next stage of the experiment.
Genotyping Analysis
Location of the Examined SNPs in the DNA Sequence, rs Numbers, Polymorphism and Location Information

A 10 µL reaction mixture was used for RT-PCR, and this volume contained 5 µL each of TaqPath™ ProAmp™ Master Mix, 0.25 µL of TaqMan™ SNP Genotyping Assay (20X), 2 µL genomic DNA (minimum 0.2 ng/µL), and 2.75 µL of ddH2O. The PCR thermal cycling conditions were as follows: pre-PCR denaturation at 60°C for 30 s, followed by an initial holding stage at 95°C for 10 min. The cycling stage consisted of 40 cycles, each including denaturation at 95°C for 15 s and annealing/extension at 60°C for 1 min. The post-PCR stage involved a final hold at 60°C for 30 s. The presence of wild-type and variant alleles was determined by comparing the relative end-point fluorescence, which resulted from the degradation of each fluorescently labeled TaqMan probe (FAM and VIC).
Sanger Sequencing Analysis
The Forward and Reverse Primers Utilized in the Study, Along With Their Respective Annealing Temperatures

Statistical Analysis
Statistical analysis of the outcomes was performed using the SPSS software, version 29.0 (SPSS Inc., USA). Categorical variables were compared between the groups using Pearson’s Chi-square test. The Bonferroni test was used as a multiple comparison test. P-values of <.05 were considered statistically significant.
Results
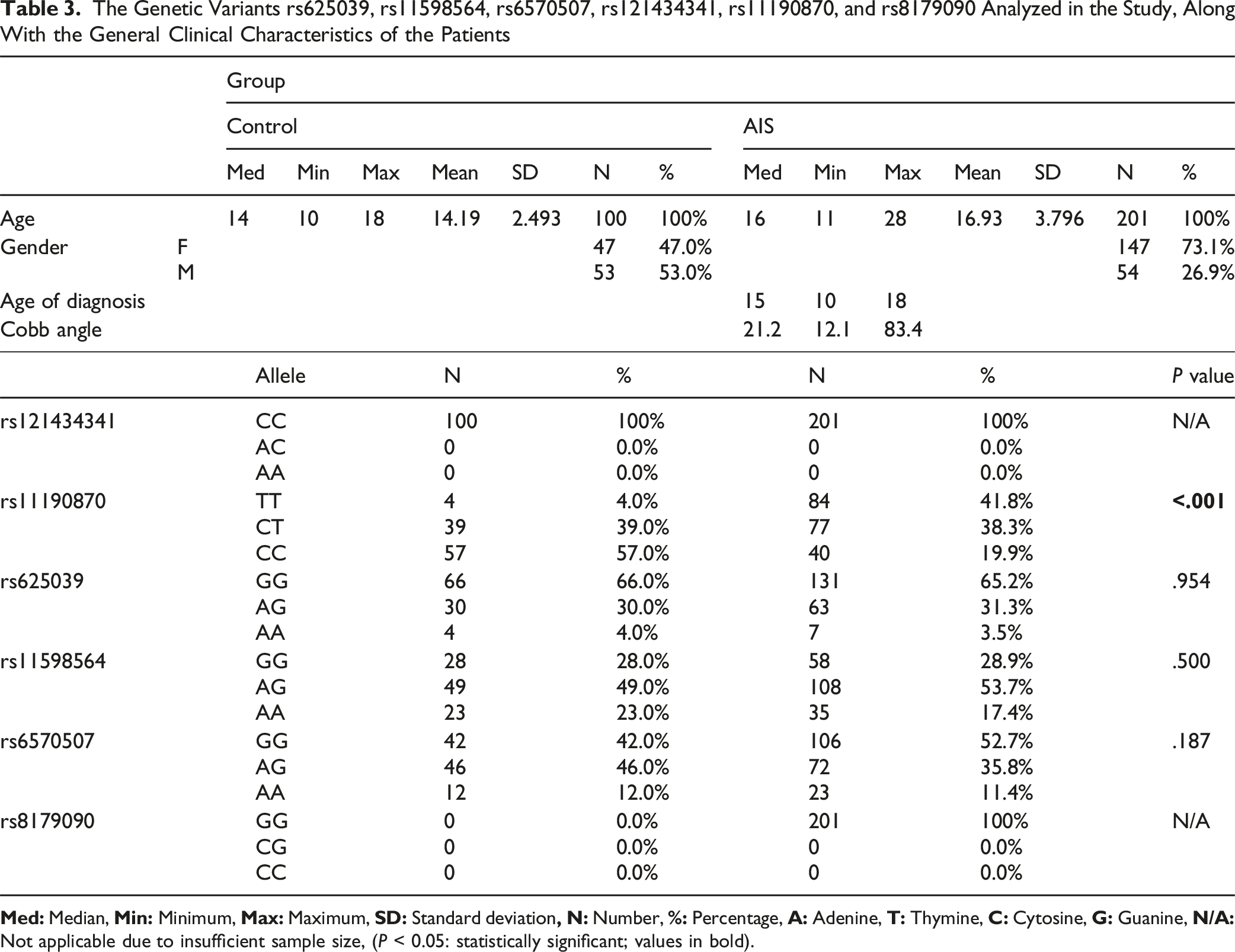
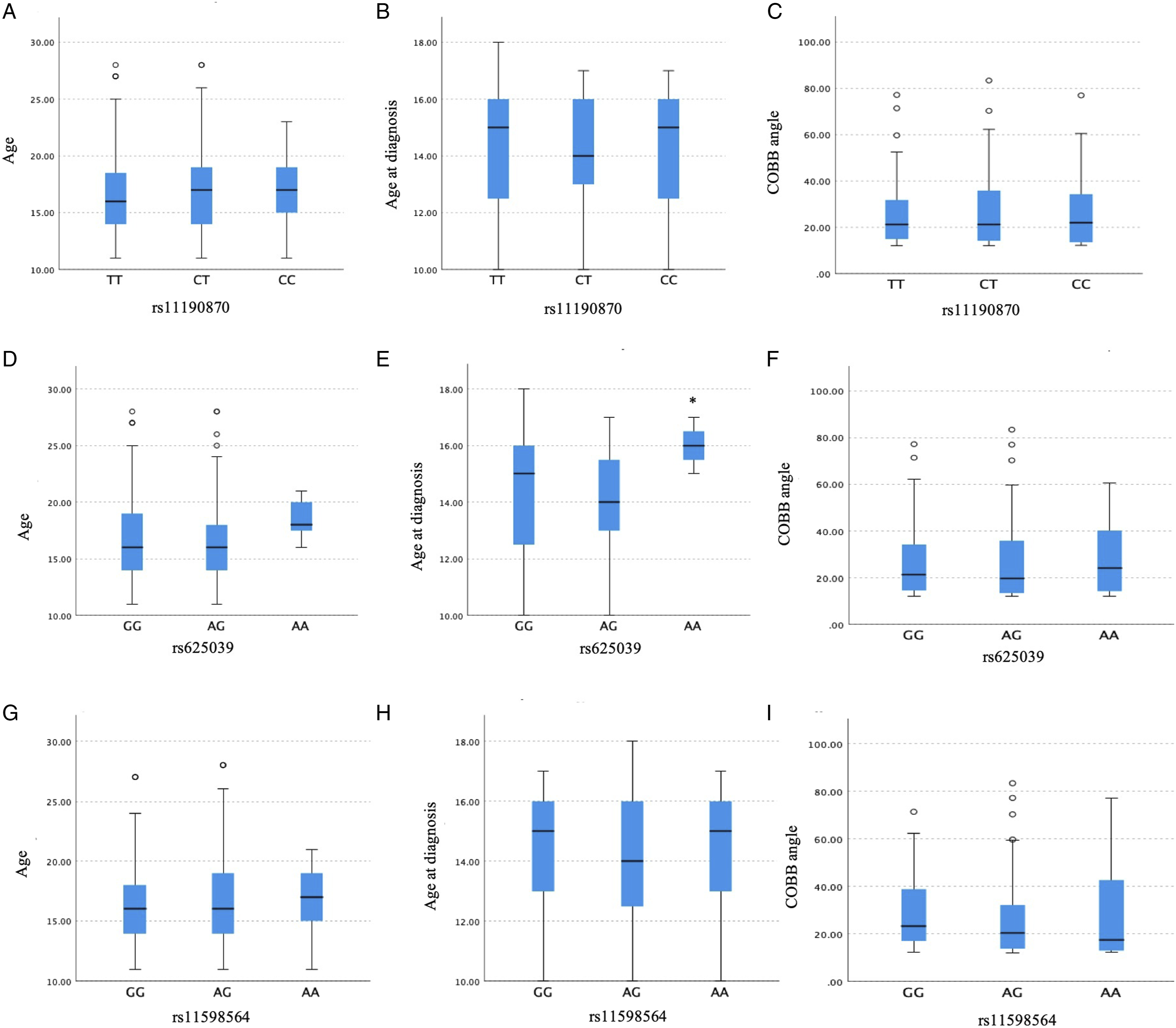
This study is the first to investigate the association between gene polymorphisms in LBX1 (rs11190870, rs625039, rs11598564), TIMP2 (rs8179090), GPR126 (rs6570507), and CHD7 (rs121434341) with AIS susceptibility in Turkish patients. The study included 201 patients with AIS (54 males and 147 females; mean age, 16.93 ± 3.796 years) and 100 healthy control individuals (53 males and 47 females; mean age, 14.19 ± 2.493 years). Blood samples were collected from these 301 participants and analyzed using RT-PCR (Figure 1). The data obtained from the comparison of the other polymorphisms with the control group are presented in Table 3. No variations were observed for the rs121434341 and rs8179090 polymorphisms in either the patient or control groups. Among the polymorphisms examined, the rs11190870 polymorphism in the LBX1 gene showed a significant association with AIS, as it was found at a statistically higher frequency in patients with AIS compared with the control group (P < .001). In addition, when evaluating the rs11598564 polymorphism in the LBX1 gene in relation to sex, it was observed that this polymorphism occurred more frequently in female patients with AIS (P = .029) (Table 4). The findings from the 3 polymorphisms (rs11190870, rs625039, and rs11598564) in the LBX1 gene suggest that these genetic variants do not exhibit a statistically significant association with age, age at diagnosis, or Cobb angle in patients with AIS (Figure 2). Age (P = .520), age at diagnosis (P = .873), and Cobb angle (P = .949) showed no significant association with the rs11190870 polymorphism. Similarly, there was no significant correlation with age (P = .177), age at diagnosis (P = .165), and Cobb angle (P = .954) and the rs625039 polymorphism. There was also no meaningful relationship between the rs11598564 polymorphism and age (P = .851), age at diagnosis (P = .630), and Cobb angle (P = .314). Evaluation of Results From TaqMan® SNP Genotyping Analysis Based on Fluorescence (Graphical Representations on the Left Side) and Amplification Plots (Graphical Representations on the Right Side): (A) Homozygous Normal/Allele 1 (Green Peak), (B) Heterozygous Variant/Alleles 1 and 2 (Green and Blue Peaks Together), (C) Homozygous Variant/Allele 2 (Blue Peak) The Genetic Variants rs625039, rs11598564, rs6570507, rs121434341, rs11190870, and rs8179090 Analyzed in the Study, Along With the General Clinical Characteristics of the Patients The Significance of the Identified Polymorphisms Between Male and Female Patients With AIS Based on Gender was Statistically Evaluated Wild-type: WT, N/A: Not applicable. Values in bold are statistical significance (P < 0.05). The Statistical Analysis Results for the LBX1 Gene Polymorphisms, rs11190870, rs625039, and rs11598564, in Patients With AIS, Were Assessed in Relation to Various Clinical Parameters, including Age (A, D, G), Age at Diagnosis (B, E, H), and Cobb Angle (C, F, I). The Independent Samples Kruskal-Wallis Test was Used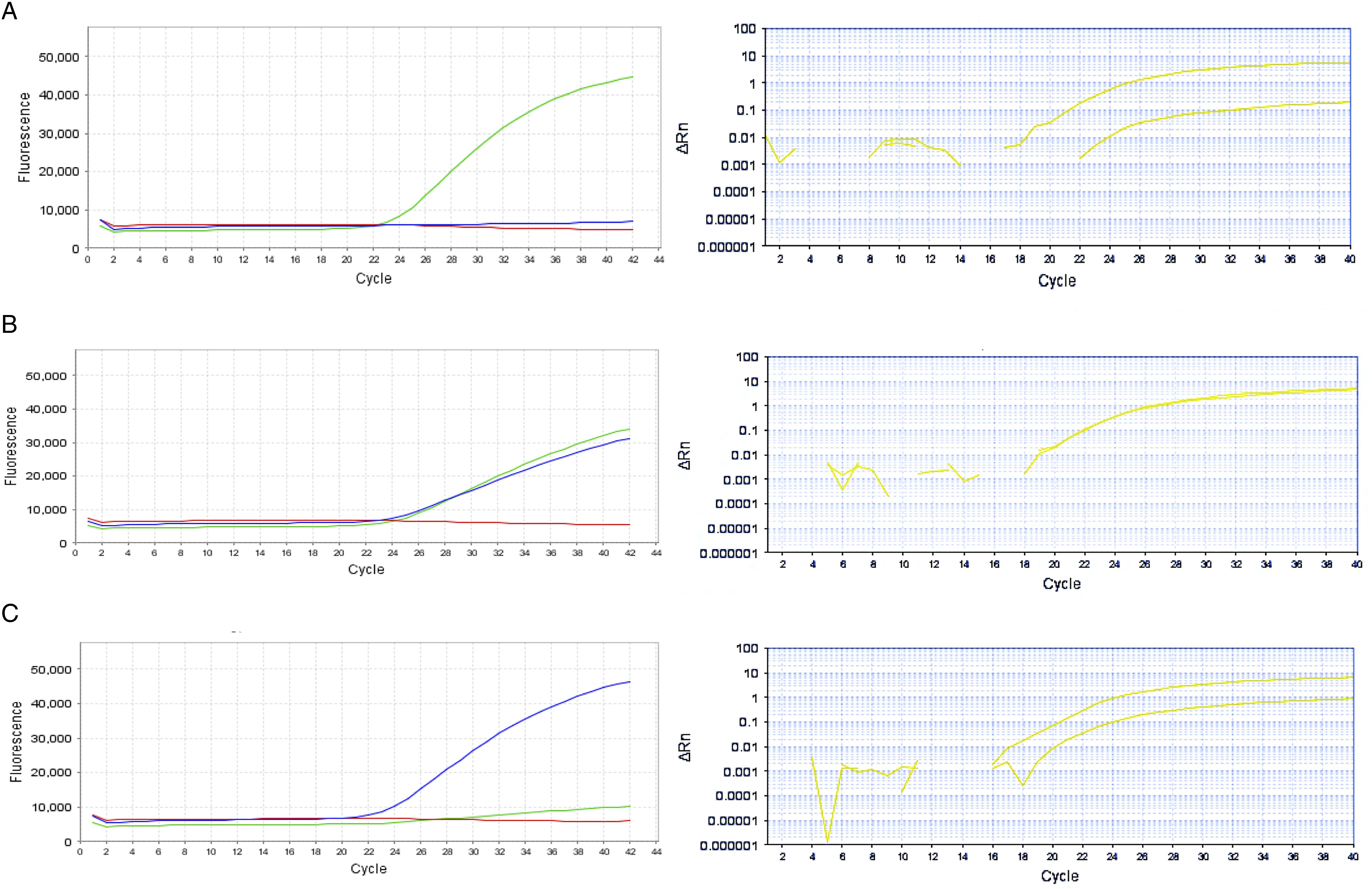
The variants identified through RT-PCR in this study were found to be congruent with the results obtained from DNA sequencing analysis (Figure 3). DNA Sequence Analysis of the CHD7 Gene (rs121434341), TIMP2 Gene (rs8179090), LBX1 Gene (rs11190870, rs625039, rs11598564), and GPR126 Gene (rs6570507), Illustrating Wild-type Sequences as Well as Heterozygous and Homozygous Variations (A-N)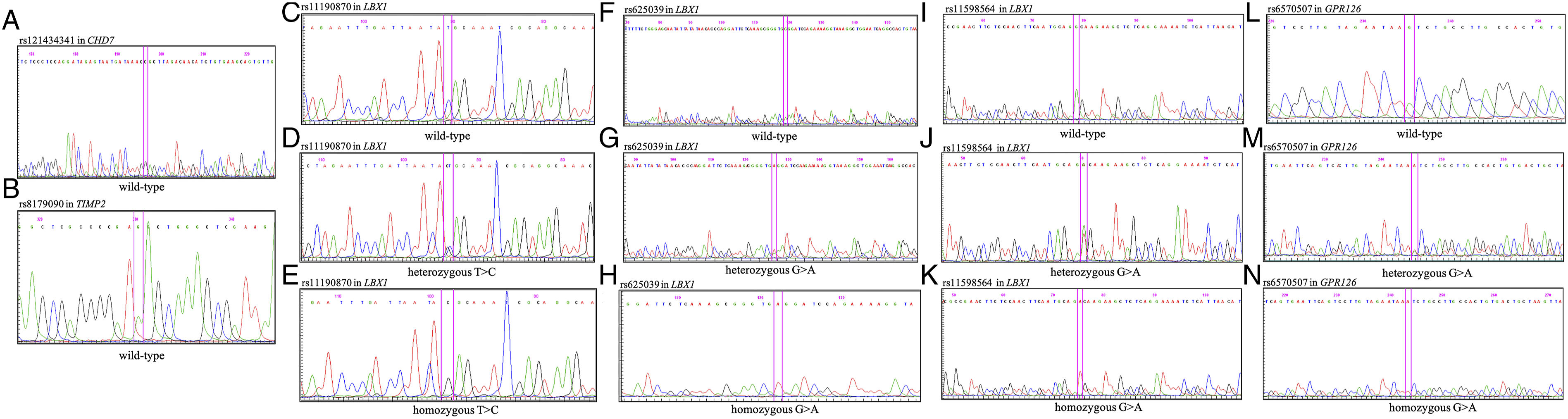
Discussion
AIS is the most prevalent spinal deformity in adolescents, yet its underlying etiology remains largely unclear. Although several factors, including genetics, hormones, and mechanical stresses, contribute to its development, a comprehensive understanding of the genetic mechanisms involved is still lacking. 9 This study aimed to investigate the genetic factors influencing AIS by examining the role of 4 candidate genes LBX1 (rs11190870, rs625039, rs11598564), TIMP2 (rs8179090), GPR126 (rs6570507), and CHD7 (rs121434341) in a cohort of Turkish patients. These genes were selected based on their involvement in processes such as skeletal growth, muscle development, and extracellular matrix regulation, all of which are integral to AIS pathogenesis.
The LBX1 gene, due to its well-documented involvement in muscle precursor cell migration and nervous system development, is considered the most promising candidate in the etiology of AIS. 24 It is essential for the development of somatosensory relay neurons, which could influence spinal deformities such as AIS. Studies have shown that abnormalities in LBX1 expression may lead to dysfunctional somatosensory systems and asymmetric paraspinal muscles, which are associated with the pathogenesis of AIS. 25
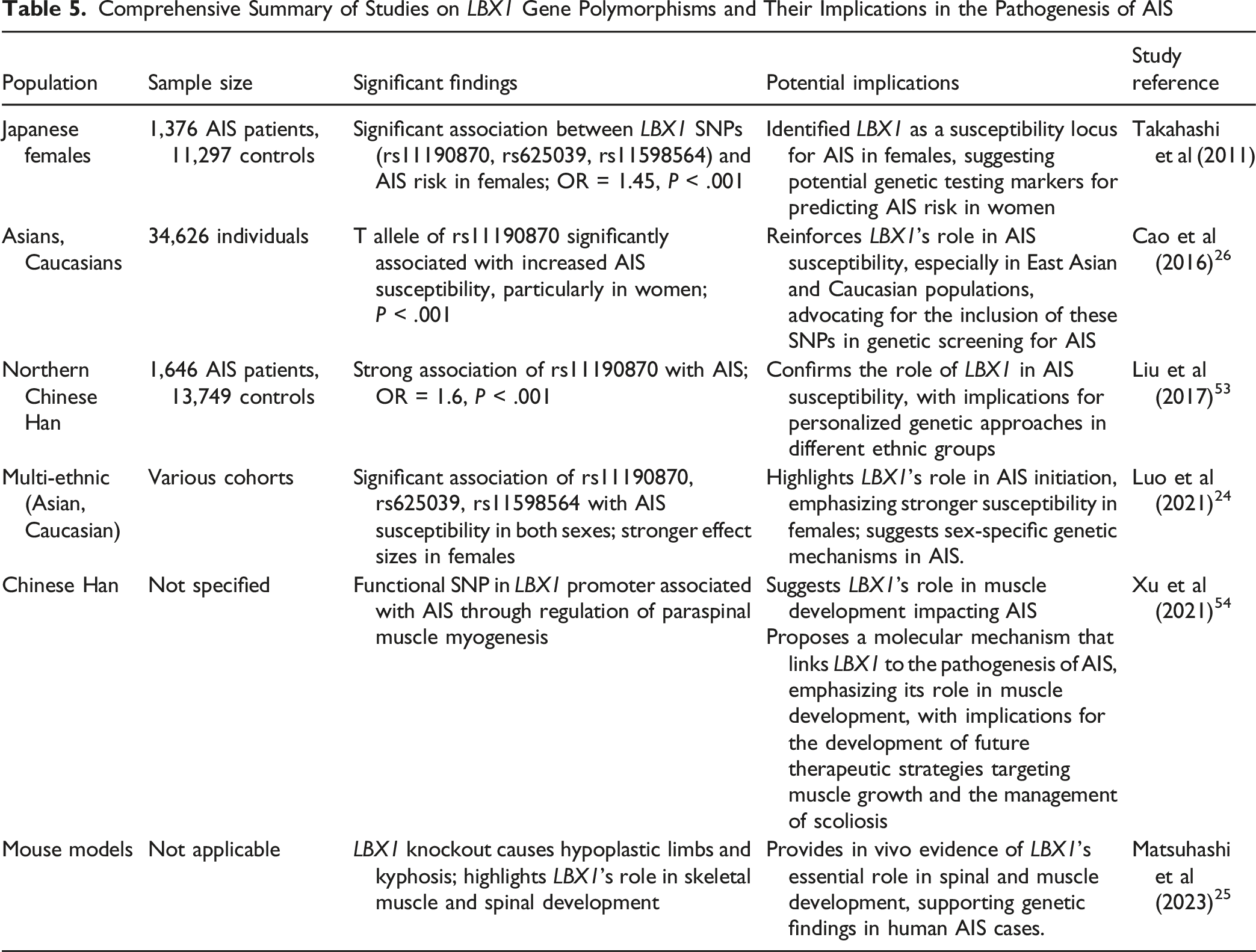
Genome-wide association studies (GWAS) have identified susceptibility loci on chromosome 10q24.31, particularly polymorphisms rs11190870, rs625039, and rs11598564, which are strongly associated with AIS in various populations, especially in East Asian cohorts. 12 Takahashi et al (2011) performed one of the pivotal studies reporting a strong correlation between LBX1 gene polymorphisms and an increased risk of developing AIS. The study was based on a GWAS comparing 1,376 Japanese female patients with AIS with 11,297 female controls, providing compelling evidence that these genetic variants might play a critical role in AIS susceptibility. 12 A meta-analysis involving 34,626 individuals revealed that the T allele of rs11190870, located near the LBX1 gene, was significantly associated with an increased susceptibility to AIS in Asian, Caucasian, and particularly female populations, suggesting that sex-specific genetic mechanisms might play a crucial role in AIS development. 26 In addition to these findings, another GWAS conducted in a Japanese cohort further confirmed the association between the aforementioned LBX1 polymorphisms and AIS, reinforcing their role in the pathogenesis of AIS. 27 Furthermore, additional studies conducted in populations from China, Bulgaria, Caucasia, and French-Canada have also validated the relationship between rs11190870 and AIS.26,28-31 A subsequent meta-analysis, which included 5,159 patients with AIS and 17,840 controls, reported a highly significant association with this SNP, with a P = 1.22 × 10-43 for both men and women, and an even stronger P = 2.94 × 10-48 for women only. 32
The present study established a significant association between the LBX1 polymorphism rs11190870 and AIS susceptibility (P < .001), consistent with prior research conducted across diverse populations, particularly in East Asian cohorts. LBX1 has consistently been identified as a key gene in the pathogenesis of AIS, with rs11190870 emerging as a reliable genetic marker linked to AIS susceptibility across various ethnic groups. Furthermore, a sex-specific association was demonstrated for the LBX1 rs11598564 polymorphism, with significantly higher frequencies in female patients with AIS (P = .029), supporting the well-documented higher prevalence of AIS in females.12,24,26 These findings indicate that genetic factors, particularly those influencing LBX1 expression, may contribute to the sex disparity in AIS, potentially through mechanisms that modulate spinal development and curvature progression in a sex-specific manner.
Comprehensive Summary of Studies on LBX1 Gene Polymorphisms and Their Implications in the Pathogenesis of AIS
Despite significant findings related to LBX1, no statistically significant associations were observed between TIMP2 and CHD7 gene polymorphisms and AIS in our cohort. The TIMP2 gene, located at chromosomal region 17q25.3, has been previously implicated in AIS susceptibility. 40 Moreover, TIMP2 has been associated with various pathologic conditions including chronic obstructive pulmonary disease, oral squamous cell carcinoma, and endometriosis.41-44 Prior research found that the rs8179090 polymorphism in the TIMP2 promoter region was not associated with AIS onset but might be linked to thoracic AIS progression in a Chinese cohort. 43 Nevertheless, subsequent studies, such as those by Ogura et al (2013) in a Japanese cohort and Andrusiewicz et al (2019) in a Caucasian population, failed to replicate this association.45,46 Consistent with these findings, our study found no significant association between the rs8179090 polymorphism and AIS, suggesting that the involvement of TIMP2 in the pathogenesis of AIS may be population-specific or modulated by additional genetic factors beyond the scope of this study.
In recent years, the potential role of the CHD7 gene in the development of AIS has been highlighted.47-50 A study conducted by Gao et al (2007) identified CHD7 as a susceptibility gene for AIS in a cohort of 52 European families. 48 Supporting the findings reported by Gao et al (2007), a study by Borysiak et al (2020), involving 211 female patients with AIS, found that the rs1017861 polymorphism in the CHD7 gene was associated with AIS in Caucasian Polish female patients. 50 However, Tilley et al (2013) analyzed 22 genotyped SNPs in the CHD7 gene across 244 families with familial idiopathic scoliosis and found no significant association between these SNPs and familial idiopathic scoliosis. 49 Furthermore, a meta-analysis of these cohorts confirmed the lack of a genetic association between CHD7 and familial idiopathic scoliosis. 49 Consistent with these findings, our study detected no statistically significant association between the rs121434341 polymorphism in CHD7 and AIS in our population. Comparisons were made in patients with AIS regarding age, age at diagnosis, and Cobb angle for the 6 SNPs; however, no statistically significant differences were identified for any of these variables.
The limitations of our study, when compared with previous studies, include being a single-center study, the limited number of patients enrolled, the lack of sex correlation between the AIS patient group and the control group, and the absence of an evaluation of the SNPs in relation to curve progression. Despite these limitations, Turkey’s genetic landscape, characterized by significant heterogeneity due to its strategic location at the crossroads of Europe, Asia, and the Middle East, as well as its diverse ethnic composition and historical migration patterns, offers a rich context for exploring genetic factors. 51 This genetic diversity is particularly evident in the southern Marmara region, which is home to approximately 25 million people, reflecting a significant aspect of Turkey’s broader genetic variability and offering a valuable context for further genetic research. 52 Our center operates in this region, providing healthcare services to a population that mirrors the genetic diversity of the country. Furthermore, the findings of our study suggest that, although common genetic variations associated with AIS have been identified, further meta-analytic studies are necessary to explore population-specific variations and their role in the pathogenesis of AIS.
The present study contributes to the understanding of genetic factors associated with AIS in the Turkish population, particularly through the examination of the LBX1 rs11190870 polymorphism. Our data indicate a significant association of this polymorphism with AIS, suggesting its relevance in the genetic profile of the condition among our cohort. However, other polymorphisms studied, such as those in TIMP2 and CHD7, showed no significant differences, underscoring the need for further research to clarify their roles in AIS etiology across different populations.
Conclusion
We anticipate that our study will make a significant contribution to the literature by uncovering the potential genetic basis of AIS, a condition with an etiology that remains largely undefined. Given the high prevalence of AIS among adolescents and its considerable burden on public healthcare systems, clarifying its genetic foundation is of paramount importance. The LBX1 gene plays a pivotal role in both the onset and potential progression of AIS, particularly through its involvement in muscle and neuronal development. To gain a comprehensive understanding of the complex mechanisms linking LBX1 to AIS, future research should employ integrated genomic and epigenomic approaches, investigating the potential interactions between LBX1, hormonal fluctuations, and environmental factors. It is expected that the findings of the study will serve as a foundation for future research with larger cohorts, particularly in Turkey, and will help to advance the understanding of the genetic mechanisms contributing to AIS.
Footnotes
Acknowledgments
Special thanks to Ayten Haciefendi (MSc) for her valuable assistance with the laboratory work. We also appreciate the financial support provided by [Project No: TTU-2023-1349, ID: 1349 and THIZ-2024-1850, ID: 1850] for the thesis project of E.B.
Statement of Ethics
This study followed the tenets of the Declaration of Helsinki and was approved by the Ethics Committee of the university (Decision number: 2024-09/13 date: 05.06.2024) and registered with ![]() (NCT06540924). The methods were performed in accordance with the approved guidelines. Written consent forms were obtained from all participants/parents before participating in the study.
(NCT06540924). The methods were performed in accordance with the approved guidelines. Written consent forms were obtained from all participants/parents before participating in the study.
Author Contributions
E.B., B.A. and H.T.U. designed the study. E.B. and H.T.U. drafted the article. E.B., B.A., H.T.U., G.C., Y.B., and H.S.A. performed data analyses, wrote the manuscript and were involved in data collection and statistical analyses. All authors contributed to the interpretation of data and revision of the manuscript and also approved final approval of the version to be published.
Funding
This study was supported by a grant from the Scientific Research Projects Foundation (BAP) of Bursa Uludag University [Project No: TTU-2023-1349, ID: 1349 and THIZ-2024-1850, ID: 1850].
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
