Abstract
Study design
Retrospective Study.
Objectives
Postoperative gastrointestinal dysfunction (POGD) adversely affects patient outcomes. ERAS protocols can improve postoperative outcomes by optimizing gastrointestinal (GI) function. This study aims to evaluate the effectiveness of a GI management ERAS protocol in patients with posterior lumbar interbody fusion (PLIF) surgery.
Methods
A retrospective analysis was conducted on patients who underwent PLIF between 2017 and 2020 in a single institution. The control group included patients treated before the institution adopted updated ERAS protocols including GI management, and the intervention group included patients treated after, with special intestinal preparation, intake and GI management. Demographic data, intraoperative and postoperative variables, and GI assessments were analyzed.
Results
The study included 163 patients: 78 in the intervention group and 85 in the control group. No significant differences were found in demographics, perioperative variables, comorbidities, or postoperative VAS and ODI scores. In the intervention group, the postoperative length of stay and ambulation time were reduced by 45.0% (P = 0.02) and 68.0% (P = 0.01), respectively. They also had lower rates of postoperative complications, including poor feeding (11.6%, P = 0.02), nausea and vomiting (11.6%, P = 0.03), hypoalbuminemia (10.3%, P = 0.04), and constipation (24.4%, P < 0.01). The time to first postoperative flatus and defecation was reduced by 41.5% (P < 0.01) and 30.1% (P = 0.02). PAC-SYM was decreased by 31.9% (P < 0.01), and GIS was decreased by 41.2% (P < 0.01).
Conclusions
Implementing a GI management ERAS protocol accelerates postoperative recovery in PLIF surgery. Integrating these strategies into standard perioperative care may not only diminish the incidence of postoperative GI dysfunction but also reduce complications, enhancing surgical outcomes.
Keywords
Introduction
With a global aging population, the incidence of lumbar spine disease is increasing rapidly. Posterior lumbar interbody fusion (PLIF) is the gold standard treatment,1-3 but due to the invasive nature of the surgery and the effects of anesthesia, postoperative complications may result in delayed recovery.4,5 Postoperative gastrointestinal dysfunction (POGD) is a common disorder with an incidence of up to 8.2% after PLIF surgery, which significantly impacts patient outcomes, 6 as it can lead to electrolyte disorders and hypoproteinemia, which prolong wound healing and increase the risk of infection and bony nonunion.7-9 These electrolyte imbalances can exacerbate gastrointestinal (GI) issues, creating a vicious cycle. The relationship between POGD and orthopedic surgeries has been well-documented, with many studies observing the adverse effects of postoperative ileus,10,11 such as pain, nausea, vomiting, severe constipation, and other GI symptoms. 12 Post-PLIF patients are more prone to constipation than other patients undertook general anesthesia due to their defecation posture being greatly affected by the surgery, and the incision site being closer to the digestive system, thus entering the vicious circle of “difficulty in defecation - reduced food intake - nutritional deficiency – POGD”. In addition, it has been shown that patients who experience POGD are at greater risk of other complications, prolonged hospital stays, and increased financial burden. Traditional GI protocols in postoperative care recommend withholding oral intake until patients have normal bowel and gut function,13,14 delaying a return to normal diets for at least 48 hours postoperatively. However, significant, decades-long evidence 15 contests this standard practice, leading to the recent creation of the Enhanced Recovery After Surgery (ERAS) protocol,16-18 aiming to improve the management of GI function and nutrition against the shortcomings of the historical postoperative course.
ERAS is a multidisciplinary set of guidelines that emphasizes the role of care in all periods of surgery, from preoperative to postoperative. It has been shown to significantly improve GI function and general postoperative outcome of orthopedic patients, and is associated with reduced length of stay (LOS) and postoperative complications in spinal fusion surgery.19-21 Furthermore, as the first 2 postoperative weeks are critical for wound healing, infection prevention, and mitigation of eating dysfunction, a relatively fast return to a normal diet (given no unexpected symptoms such as nausea or vomiting), as outlined by ERAS, has been shown to accelerate recovery and decrease complications. Alongside other detailed changes, such as greater preoperative nutritional support and effective pain management, ERAS has been reported to significantly reduce digestive complications.22-24
Although factors, including but not limited to, obesity, diabetes, operative time, and larger incision size, are highly associated with postoperative complications,25-27 the impact of GI management is less investigated. As such, the relationship between GI management and POGD remains poorly characterized. While improved surgical techniques are essential for preventing complications, the role of perioperative GI management should be of great importance in patient care. Thus, we have improved the traditional ERAS protocols, especially by adding special intestinal preparation, postoperative intake and GI management and GI rehabilitation programs. The purpose of this study is to evaluate the effectiveness of ERAS protocols in mitigating postoperative complications, specifically focusing on the impact of updated GI management on various indicators of postoperative recovery, including nausea, vomiting, hypoalbuminemia, and more.
Methods
Study Design
Retrospective analysis was performed on data collected from patients who underwent PLIF between January 2017 and July 2020 in a single institution. Institutional review board approval (No. 2020-ke-32) was obtained on 2020.04.11 before data collection and informed consent was obtained from each patient included in the study. Statistical analysis was performed by researchers who did not participate in data collection in order to minimize potential bias.
Patient Selection:
Patients were included in the database if they were between 18 and 75 years of age. The flow chart for patient selection was shown in Figure 1. The exclusion criteria were: • Patients receiving treatment for spinal tumor; • Patients receiving treatment for spinal infection; • Patients receiving treatment for congenital spine deformity; • Patients undergoing revision surgery; • Patients undergoing combined cervical and/or thoracic surgery; • Patients’ data was incomplete; • Patients noncompliant with ERAS protocols; The flow chart of patient selection.
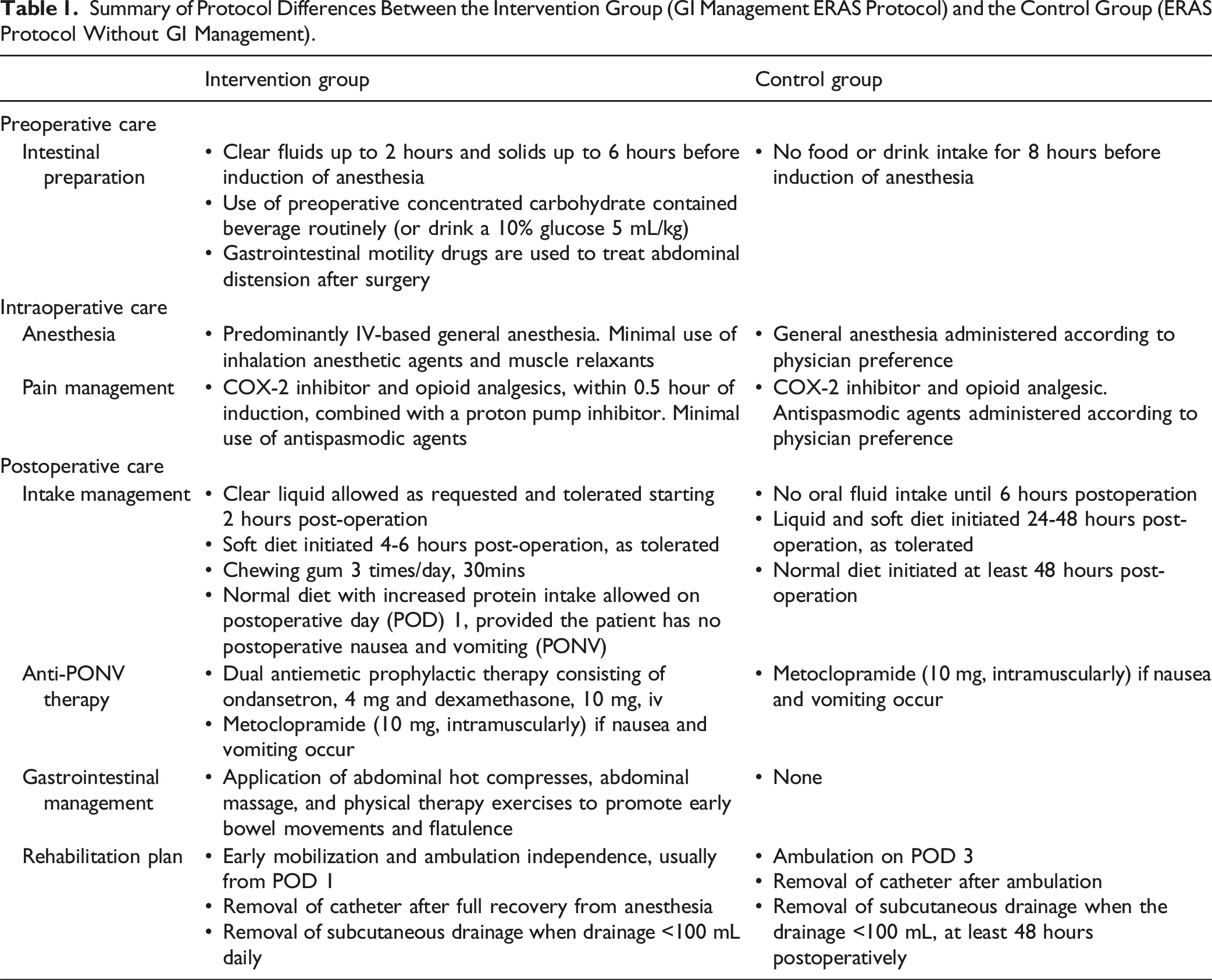
Summary of Protocol Differences Between the Intervention Group (GI Management ERAS Protocol) and the Control Group (ERAS Protocol Without GI Management).
Demographic data including age, gender, body mass index (BMI), bone mineral density (BMD), comorbidities, and preoperative laboratory values were obtained preoperatively. Intraoperative variables included the number of fused segments, estimated blood loss (EBL) and OR time. Postoperative variables included postoperative complications (SSI, poor wound healing, poor feeding, vomiting, hypoalbuminemia) within 30 days after surgery, length of stay (LOS), and 30-day readmission rate. GI assessments included thirst and hunger assessment (10-point Visual Analog Scale),
28
time to first postoperative flatus, time to defecation, presence of constipation, and length of postoperative hospitalization, Patient Assessment of Constipation Symptoms (PAC-SYM),
29
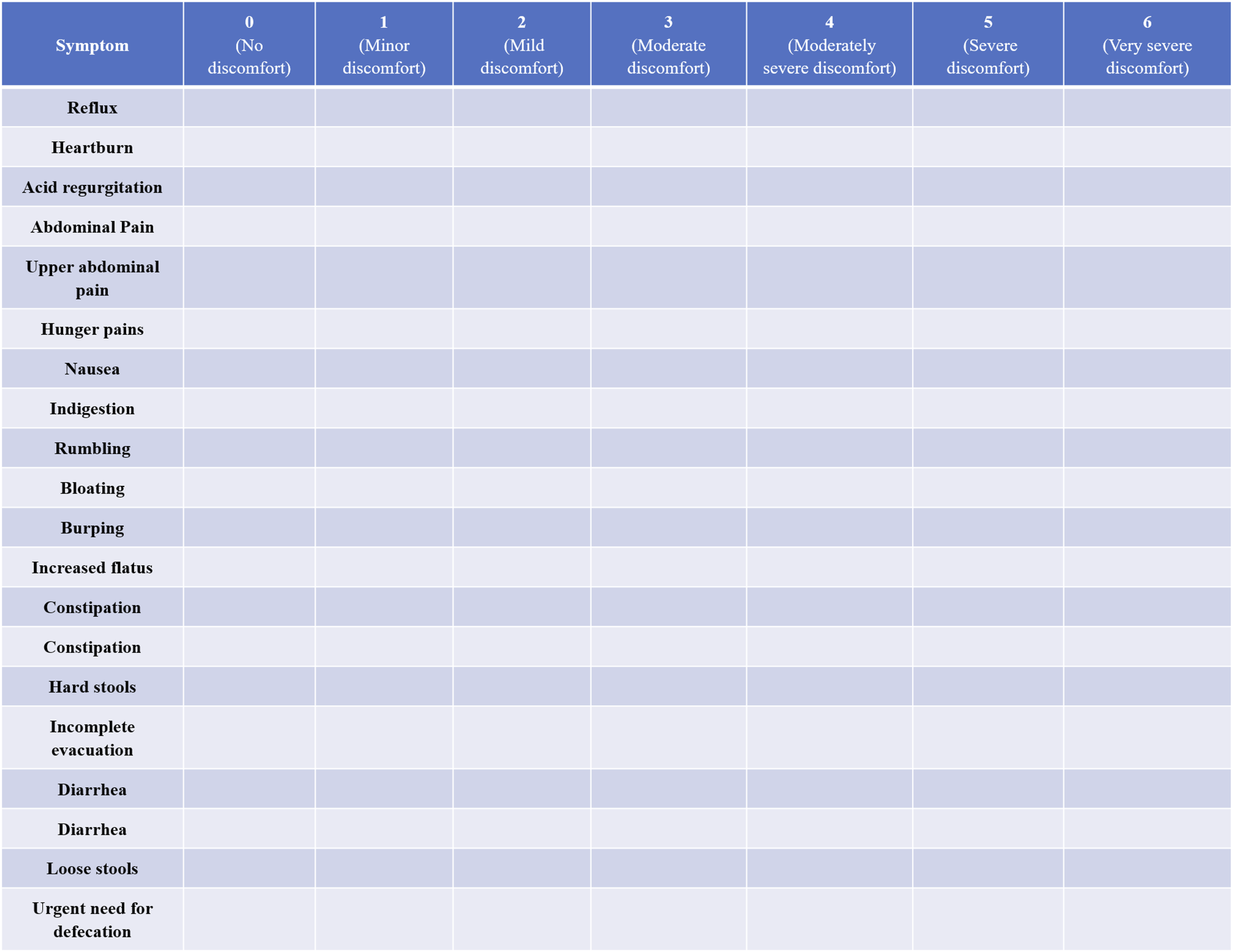
and Gastrointestinal Symptom Score (GIS)
30
(Figures 2 and 3). The legend of the PAC-SYM The legend of the GIS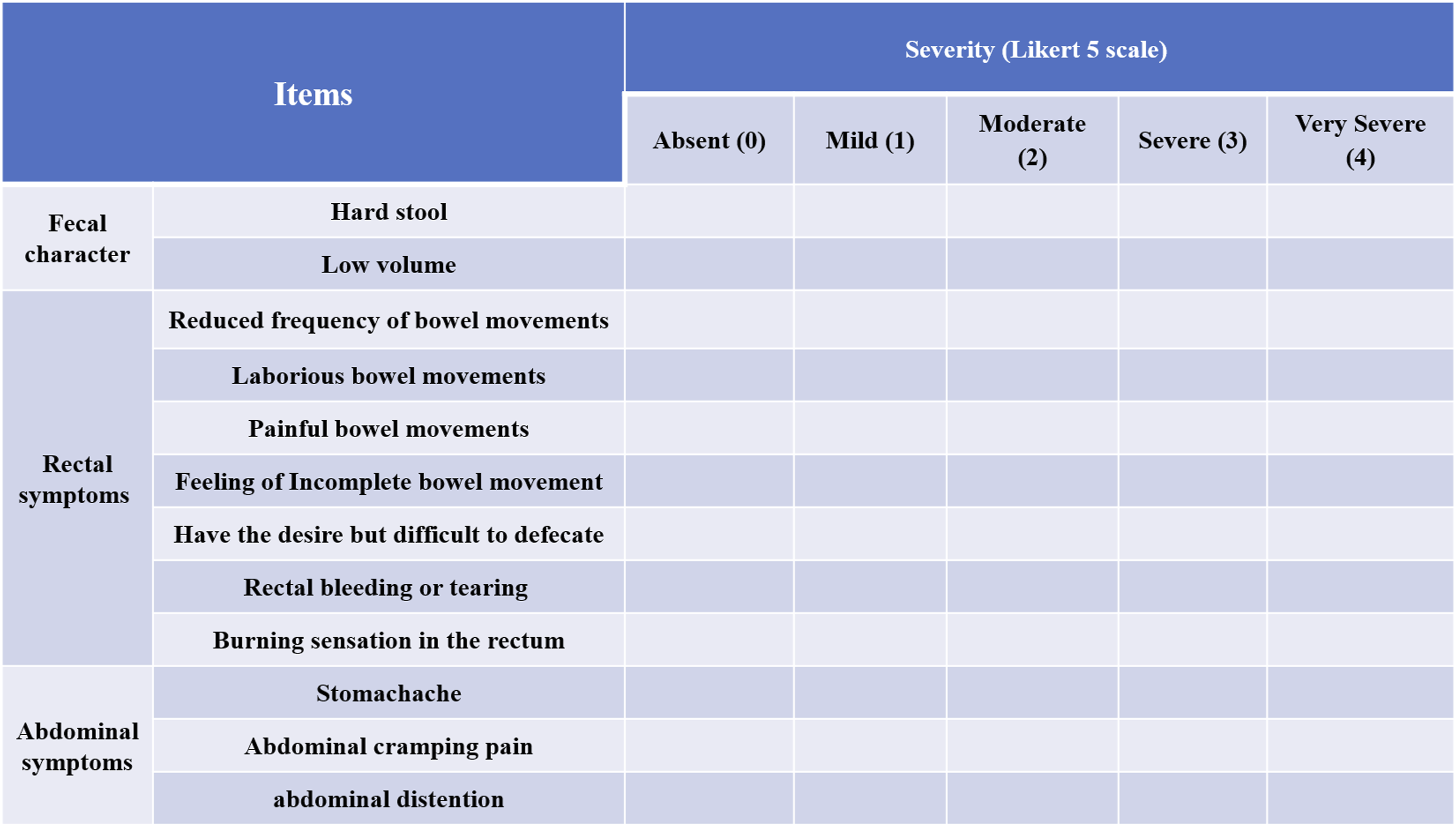
Statistical Analysis
Statistical analyses were conducted using SPSS Version 22 (IBM Corp., Armonk, NY) by an independent researcher (J.W.) who was not involved in data collection and group assignments. All patients included in this study had complete and valid data. Continuous variables were assessed for normality using the Shapiro-Wilk test. Variables conforming to a normal distribution with homogeneity of variance were analyzed using the independent samples t-test (two-tailed, α = 0.05), while non-normally distributed variables or those with unequal variances were analyzed using the Mann-Whitney U test. Categorical variables were compared using the chi-square test, with Fisher’s exact test substituted when expected cell frequencies were <5. All hypothesis tests were two-tailed. Continuous variables are reported as mean ± standard deviation, and categorical variables as frequency (percentage). The significance threshold was set at α = 0.05. A P-value of ≤0.05 was considered indicative of statistical significance a priori. This study adheres to STROBE guidelines.
Results
Noninferiority criteria was used to calculate the appropriate sample size, as this was a retrospective, clinical superiority trial in patients undergoing posterior lumbar surgery based on primary outcome of POGD rate and clinical recovery. The expected POGD and other complications absence rate of the control group applying general care was 80%, which agreed with several studies. For noninferiority of updating ERAS vs general protocols in PLIF with two-sided 5% significance, power of 80%, and noninferiority margin of 15%, a sample of 73 patients for each group was necessary.
Comparison of Demographic Data Between Intervention Group (GI Management ERAS Protocol) and Control Group (ERAS Protocol Without GI Management).
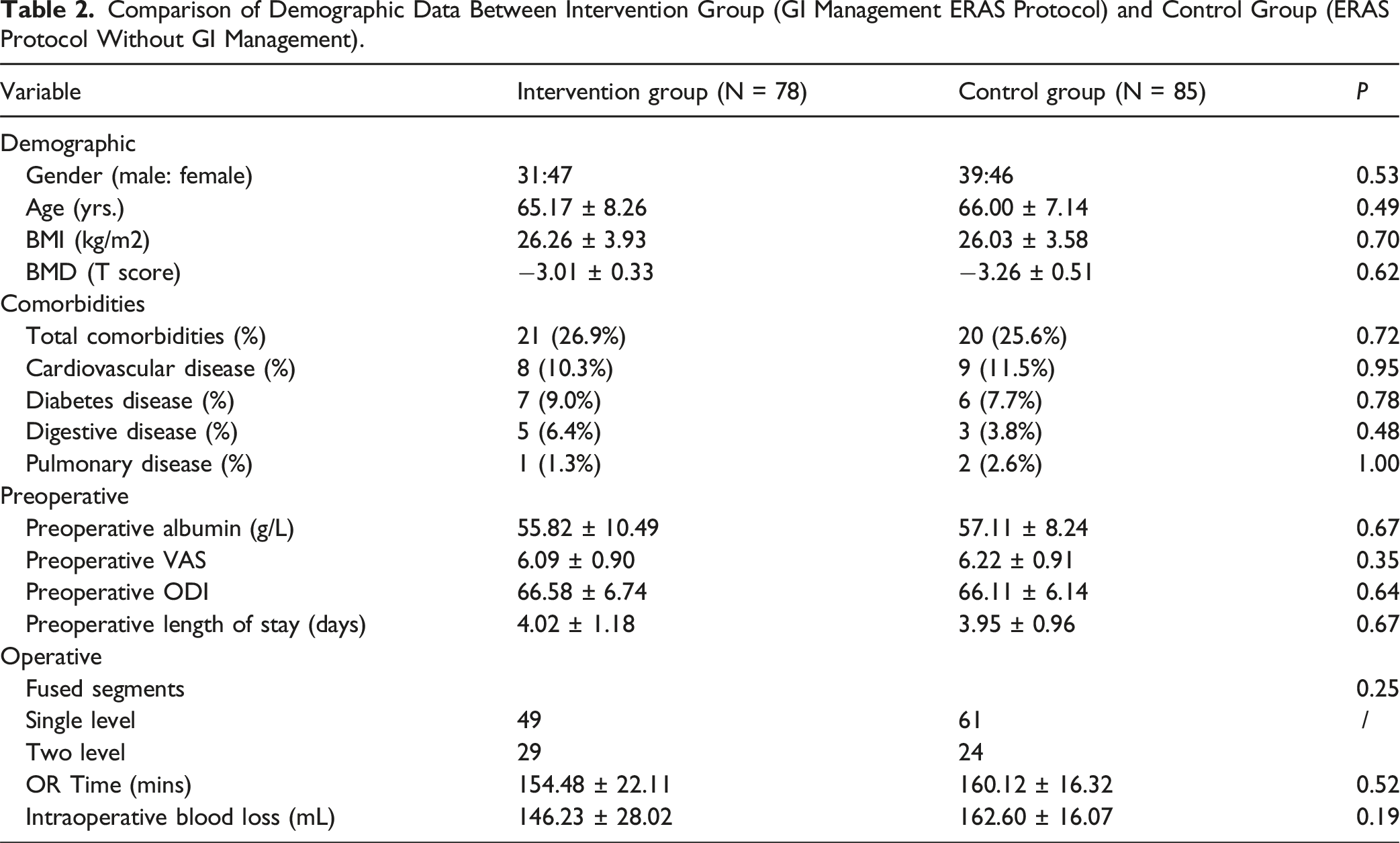
No differences were observed in postoperative pain and disability between the intervention group and control group with respect to VAS and ODI scores(2.97 ± 0.74 vs2.96 ± 0.73, 32.64 ± 5.28 vs 33.83 ± 6.32, respectively). However, the intervention group had statistically significantly shorter postoperative lengths of stay and earlier ambulation time (3.39 vs 6.16 days, P = 0.02; 1.16 vs 3.62 days, P = 0.01, respectively). We propose that the intervention directly improved GI symptoms, which enhanced postoperative comfort, and consequently led to early mobilization as well as a shorter LOS.
Postoperative Outcomes and Complications
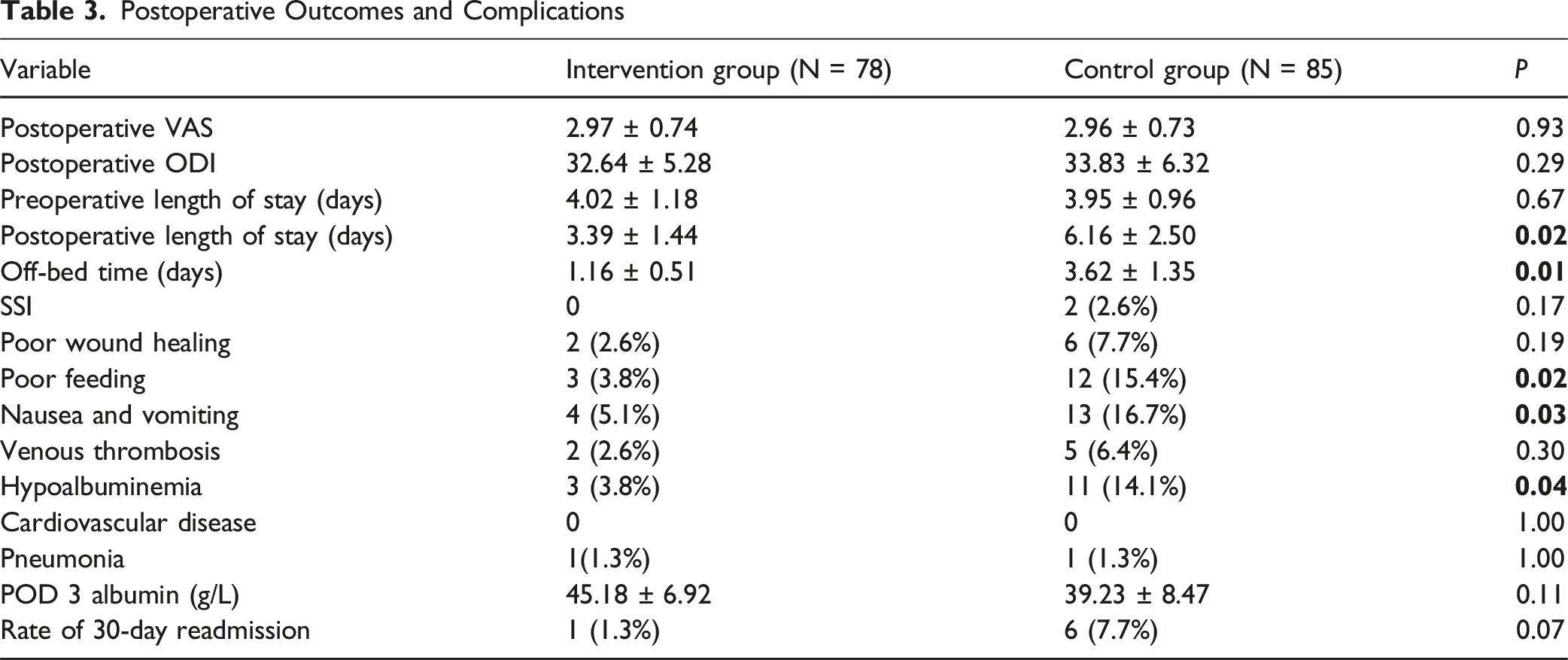
Gastrointestinal Self-Assessments in Intervention and Control Group
Discussion
Traditional GI protocols advise surgeons to withhold patients from solid diets until it is clear that normal bowel and digestive function have returned 13 ; however, recent evidence contradicts this commonly held belief and show clear improvement in accordance with changes outlined in the new Enhanced Recovery After Surgery (ERAS) protocol. By emphasizing a multidisciplinary approach and a rapid return to normal diet, given no other obvious GI symptoms, ERAS protocols have improved postoperative outcomes across various surgical fields, including cardiac, colorectal, and gynecological surgeries.31-33 Within orthopedics, many works have reported an impressive overall reduction in the average length of hospital stay for hip and knee arthroplasty,34,35 as well as total joint replacement (TJR) surgery. 36 However, most studies caution that more robust research is needed to investigate the success of ERAS in other types of orthopedic surgeries,34,37 as few papers have evaluated the implementation of ERAS outside of arthroplasty and TJR. Included in this sphere of understudied surgeries is spinal surgeries, such as posterior lumbar interbody fusion (PLIF). In this study, we focused on the central role of GI management in postoperative recovery for patients who underwent PLIF, and demonstrated that comprehensive ERAS GI changes significantly enhance postoperative outcomes, particularly by reducing POGD, minimizing hospital stay, and accelerating recovery.
Our findings demonstrate the positive effect of early feeding on alleviating GI symptoms. Early postoperative feeding can significantly enhance the recovery of GI motility through multiple mechanisms. 38 Firstly, it activates the vagal reflex by stimulating oral and GI receptors, promoting motility and secretion. Secondly, it provides essential nutrients for mucosal repair and stimulates insulin secretion, improving metabolic function. Additionally, the balance of gut microbiota, psychological well-being and appetite indirectly aiding GI recovery. Overall, these mechanisms highlight the importance of early feeding in reducing postoperative ileus and accelerating GI function recovery.
In the intervention group, the enhanced ERAS protocol led to notable improvements in several critical postoperative parameters compared with the control group. The postoperative LOS was significantly reduced (3.39 vs 6.16 days, P = 0.02), highlighting the effectiveness of the enhanced protocol in accelerating recovery. This reduction in LOS aligns with existing literature that underscores the benefits of ERAS protocols in various surgical settings.18,39-41 The early mobilization and tailored intake management strategies likely contributed to these outcomes by promoting quicker return to normal activities and physiological functions.19,42
One of the key findings of this study is the significant reduction in postoperative GI complications in the intervention group. The incidence of poor feeding (3 vs 12, P = 0.02), nausea and vomiting (4 vs 13, P = 0.03), and hypoalbuminemia (3 vs 11, P = 0.04) were markedly lower in the group receiving GI management ERAS protocols. The early initiation of oral intake and the use of GI motility drugs and antiemetic prophylaxis appear to have played crucial roles in these improvements.8,9,43 Additionally, the application of abdominal hot compresses, abdominal massage, and physical therapy exercises facilitated early bowel movements and reduced the risk of constipation, as evidenced by the lower incidence of constipation (33.33% vs 57.68%, P < 0.01) and better GI symptom scores (PAC-SYM and GIS) in the intervention group. Compared with prior studies, the incidence of postoperative nausea and vomiting (PONV) in control group (ERAS protocol without GI management) in our study (13/85, 16.7%) was similar with that in traditional ERAS group (5/35, 14.3%) reported by Yang, et al, both of which were much higher than 5.1% (4/78) in intervention group (GI management ERAS protocol) in our study. 44
The early postoperative feeding protocol, including the use of clear liquids and soft diets shortly after surgery, contrasts with traditional practices of delayed oral intake. 18 This approach not only mitigated the severity of thirst and hunger postoperatively but also expedited GI recovery, as indicated by the shorter time to first flatus (8.29 vs 14.18 hours, P < 0.01) and defecation (2.74 vs 3.92 days, P = 0.02). These findings align with previous studies suggesting that early nutrition supports GI motility and reduces the duration of postoperative ileus.45,46
Despite the significant improvements in GI-related outcomes, it is notable that there were no statistically significant differences between the groups in terms of surgical site infections (0 vs 2, P = 0.17) and poor wound healing (2 vs 6, P = 0.19). This suggests that the sample size may be too small to detect a definitive impact in these areas, although the results are approaching statistical significance. Further research with a larger sample size is necessary to conclusively determine the effect of GI management on these specific postoperative complications.
Interestingly, the study found no significant differences in the incidence of cardiovascular complications, pneumonia, or venous thrombosis between the groups. This indicates that the enhanced ERAS protocol did not adversely impact these outcomes, which is consistent with the holistic approach of ERAS protocols in addressing various aspects of postoperative care without compromising overall patient safety.17,24,47
Our findings underscore the value of a GI management ERAS protocol for patients undergoing PLIF. By managing GI function with perioperative care, the protocol effectively reduces postoperative GI complications, shortens hospital stays, and facilitates accelerated functional recovery. Integrating these strategies into standard perioperative care may not only diminish the incidence of postoperative GI dysfunction but also reduce complications, enhancing surgical outcomes. Future work should involve prospective, multicenter trials with larger and more diverse patient populations to validate these preliminary results, while also exploring the cost-effectiveness of comprehensive ERAS adoption. Such investigations may further refine protocol elements, delineate patient subgroups most likely to benefit, and guide implementation strategies that optimize patient safety, resource utilization, and overall quality of spine surgery care.
There are several limitations of this study, including retrospective design, single-institution data, and interpretation of missing data. First, retrospective studies may introduce selection bias, as researchers must set exclusion criteria that impact the generalizability and strength of the results. In this study, patients were excluded if they underwent surgeries that may introduce confounding factors; for example, patients undergoing combined cervical and thoracic surgery experience larger operating room times than patients admitted for PLIF only, and may skew the results if included in analysis. However, in future works with greater sample sizes, the correlation between the effectiveness of ERAS and length of time under anesthesia may be an interesting relationship to investigate, and shine light on the robustness of GI management under more strenuous conditions. Second, single-institution data may introduce a greater risk of Type II error; but in this work, by selecting patients over a long period of time, we hoped to increase sample size and increase the power of the statistical analysis. What’s more, many patients were excluded due to “non-compliance to ERAS protocols” and “incomplete data”. We attributed the reasons to insufficient publicity and education provided by medical staff to patients admitted to hospital, so that patients could not correctly and completely understand the new ERAS standard. With the gradual implementation of the GI management ERAS protocol, the admission education provided by medical staff has become more accurate, and patients have also paid more attention to the GI management ERAS protocol and adhered to it in a standardized manner. The patients in the GI management ERAS group included in the study all came from the period after the protocol was fully implemented. In the future, greater sample sizes and prospective investigation across multiple institutions would increase the reliability of results, especially as variations in surgical practices and patient populations across different centers may influence results. Finally, missing data is an additional potential source of error. Increasing the sample size and investigating multi-institutional results would help mitigate this error and better reveal the long-term benefit of enhanced GI management in ERAS protocol.
Nevertheless, given these limitations, this study investigates a deeply understudied aspect of ERAS success. This enhanced protocol has been met with wide success throughout a variety of specialties, yet, partially due to its recent implementation in many medical centers, its impact on surgeries such as PLIF has not yet been studied in detail. Given 3 years of recent data (pre-ERAS to post-ERAS), we are presented with the unique opportunity to investigate the effectiveness of GI management ERAS protocol for patients undergoing PLIF, thus shining light on the central role of rapid GI care in postoperative recovery in spinal surgery.
Conclusion
The implementation of an ERAS protocol with a specific focus on GI management significantly improves postoperative recovery in patients undergoing PLIF surgery. Orthopedists should seriously consider the effectiveness of ERAS when performing postoperative care, as this enhanced protocol effectively reduces POGD, shortens LOS, and accelerates postoperative recovery. These findings underscore the importance of comprehensive perioperative care that includes targeted GI management as a critical component of ERAS protocols. Adopting such enhanced protocols could lead to better patient outcomes, and improved overall surgical experiences. Future research should focus on multicenter, prospective studies to validate these results and further explore the long-term benefits of such protocols in diverse surgical settings. A holistic approach to perioperative care that incorporates targeted GI management is essential for improving patient outcomes and optimizing recovery.
Footnotes
Ethical Consideration
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee of Beijing Jishuitan Hospital (2020-ke-32). The authors confirm that written informed consent has been obtained from the involved patients and they have given approval for this information to be published.
Author Contributions
All authors had full access to the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. Conceptualization, CZ, DH; Methodology, AYX, BGD, AHD; Investigation, BGD, AHD; Formal Analysis, JW; Resources, CZ, DH; Writing - Original Draft, HD; Writing - Review & Editing, AYX, AHD; Visualization, HD; Supervision, DH; Funding Acquisition, CZ. All authors have read and agreed to the published version of the manuscript. Statistical analysis was performed by researchers who did not participate in data collection in order to minimize potential bias. All authors meet the authorship criteria according to the latest guidelines of the International Committee of Medical Journal Editors, and all authors are in agreement with the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Beijing Hospitals Authority’s Ascent Plan, grant number DFL20240401.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
