Abstract
Study Design
Retrospective cohort study.
Objectives
Preoperative risk stratification using frailty is common for adults but difficult to apply to pediatric populations. We aimed to identify risk factors indicating physiologic vulnerability and predict perioperative complications in children with neuromuscular scoliosis (NMS) and to create a prediction model for physiological vulnerability (PV-5).
Methods
Patients with NMS were identified from the American College of Surgeons National Surgical Quality Improvement Program Pediatric database. The 9442 patients identified were randomly divided into training and testing cohorts. Univariate and multivariable logistic regression were performed; variables significantly associated with complications were evaluated using the Akaike information criterion and area under the curve. Significant variables received weighted scores, and a patient-specific prediction model was generated and evaluated using the Brier score.
Results
Patients with central nervous system abnormality (OR 1.32 [95%CI 1.13-1.53]), hematologic disorder (OR 1.40 [1.06-1.85]), congenital malformation (OR 1.30 [1.1-1.54]), nutritional support (OR 2.21 [1.91-2.57]), and preoperative wound infection (OR 2.3 [1.4-3.76]) were more likely to develop complications after spinal fusion surgery. PV-5 scores were calculated from these risk factors to generate a prediction model. PV-5 scores of 1 (OR: 2.0 [1.27-3.43], P < 0.004), 2 (OR: 2.75 [1.63-4.64], P < 0.001), 3 (OR: 3.67 [2.18-6.19], P < 0.001), 4 (OR: 4.09 [2.39-6.99], P < 0.001), and 5+ (OR: 3.58 [1.35-9.47], P = 0.01) predicted greater complication risk than PV-5 of zero (accuracy = 89.65%, Brier score = 0.09).
Conclusions
Using factors associated with complications in children with NMS undergoing spinal fusion surgery, we created a prediction model to illustrate physiologic vulnerability and morbidity. Our model serves as a foundation for further body system-specific investigation.
Keywords
Introduction
Scoliosis is common in children with neuromuscular disorders or severe neurological impairments. 1 Indications for surgery to address neuromuscular scoliosis (NMS) vary by deformity, severity, and underlying condition. These cases are complex, in part because of comorbid conditions that may increase the likelihood of intraoperative or postoperative complications.2-4 The benefits of surgery can be significant, including preserved upright posture, improved pulmonary function, reduced aspiration, and overall better health-related quality of life. 5 Patients may choose to pursue surgical intervention based on their understanding of preoperative assessment, risk stratification, and perceived benefit.
Recently, physiologic vulnerability, often conflated with frailty, has become a topic of interest in spine surgery, including in adult patients undergoing surgery for spinal deformity correction.6-8 A common thread is that aging triggers multisystem body dysregulation and greater susceptibility to adverse health outcomes, known as frailty.9,10 The Fried frailty phenotype was developed to standardize the definition of frailty in the geriatric population and is based on 5 domains: unintentional decrease in body weight, fatigue, decreased grip strength, slow walking, and decline in physical activity. 10 Fried’s phenotype was described in the geriatric population, but the concept of physiologic vulnerability may not be age-specific. An attempt has been made to operationalize the term “frailty” to emphasize clinical rather than physiologic phenotypes. 10 Frailty scores, such as the 5- and 11-factor modified frailty indexes (mFI-5 and mFI-11), are based on clinical characteristics that have been shown to increase a patient’s risk for poor outcomes. However, it is difficult to extrapolate these indices outside their intended populations, and these brief indices may combine disease-related factors with age-related factors, which distinguish the Fried phenotype.11-13
Children with NMS harbor many of the traits traditionally considered reflective of the frailty clinical phenotype, including low body weight and osteosarcopenia; additionally, children with neuromuscular disease often have comorbidity profiles and conditions that would suggest “frailty.”11-13 To date, no study has aimed to operationalize physiologic vulnerability in the population of pediatric patients with NMS. To do so, we sought a measure of physiologic vulnerability independent of age and age-related decline in health and function.
In this study, we performed an exploratory analysis to identify factors associated with greater risk of complications to implicate key body systems associated with physiologic vulnerability in children with NMS. We then developed a preoperative risk calculator to predict the incidence of complications after pediatric deformity correction for NMS. We hypothesized that there is a specific set of factors that identify physiologically vulnerable patients at risk for complications in this specific population, which differs from adults, and can be used to predict morbidity after spinal fusion surgery.
Methods
Data Source
We performed a retrospective cohort study using the American College of Surgeons National Surgical Quality Improvement Program Pediatric (NSQIP-P) database (https://www.facs.org/quality-programs/data-and-registries/pediatric/). NSQIP-P tracks and audits 30-day perioperative outcomes (perioperative and surgical) for individuals ≤18 years. Variables are collected based on a systematic sampling process that allows for proportional diversity in selection by using an 8-day sampling cycle from more than 60 pediatric institutions. The NSQIP-P data are audited and validated continually and organized into participant user files for quality improvement and outcomes research. Because NSQIP-P participant user file datasets do not have patient-identifying information, the study was exempt from ethics review as determined by the local institutional review board and human subjects protection office.
Patient Population
ICD-9, ICD-10, and CPT Codes Used in the Study With Their Definitions.
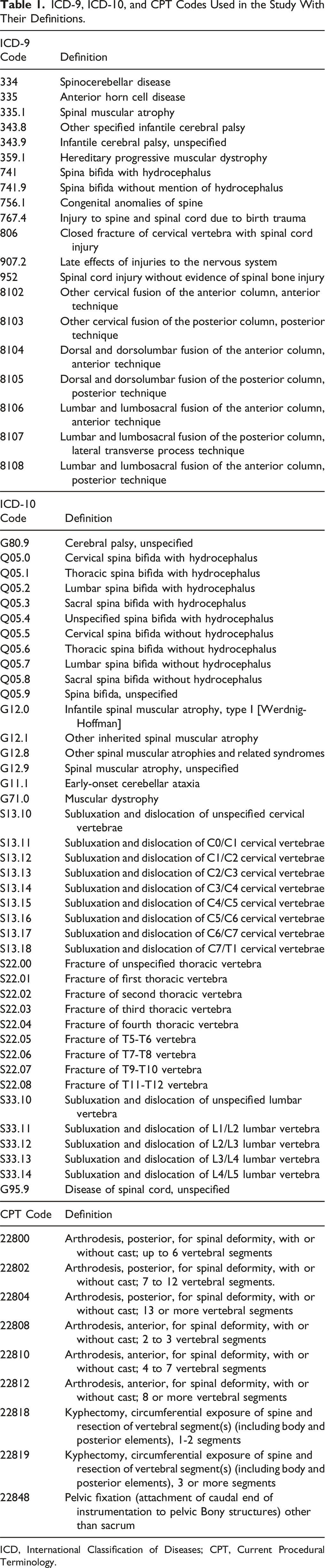
ICD, International Classification of Diseases; CPT, Current Procedural Terminology.
Data Analysis and Prediction Modeling
All variables in the NSQIP-P database were evaluated by 4 independent reviewers for relevance. Patient demographic factors (age, sex, race) and 44 preoperative variables were selected in advance for evaluation. The primary study outcome was the incidence of any postoperative complication within 30 days of surgery, which was defined as a composite of the following outcome variables: urinary tract infection, pneumonia, vein thrombosis requiring therapy, superficial incisional surgical site infection (SSI), deep incisional SSI, organ/space SSI, deep wound disruption/dehiscence, unplanned intubation, progressive renal insufficiency, acute renal failure, coma >24 hours, cerebrovascular accident/stroke or intracranial hemorrhage, seizure disorder, nerve injury, cardiac arrest requiring cardiopulmonary resuscitation, Clostridium difficile colitis, sepsis, septic shock, and central line-associated bloodstream infection. Categorical or dichotomous variables were compared using the Chi-squared test of independence.
A training cohort comprising 70% of the overall cohort was randomly selected. Univariate analysis was performed to identify which variables were associated with complications (P-value <0.10). An Akaike information criterion (AIC) analysis was then used to estimate the relative predictive quality. Additionally, an area under the curve (AUC) analysis was performed to identify the combination of variables predicting the primary outcome. Subsequent multivariable analysis was performed to identify independent associations with the outcome of complications after controlling for confounding variables. A high-throughput approach involving multiple iterations of logistic regression analyses was used to find the best model. The model with a combination of variables that produced the lowest AIC value and highest AUC score was used to create our final patient-specific prediction model. A variance inflation factor (VIF) was calculated for each of the variables in the final prediction model to assess multicollinearity between features. A VIF value >5 was considered as contributing considerably to multicollinearity.
The remaining 30% of patients in the cohort were allocated to a testing/validation group (ie, a “naïve” patient population used to validate our final prediction model). The model with a combination of variables that produced the lowest AIC value/highest AUC score was used. Odds ratios (ORs) were converted to integers and used to assign a weighted score for each variable—a score of 1 for an OR of 1-2 and a score of 2 for OR >2. ORs were converted to probabilities and used to create a prediction scoring model termed the 5-factor physiologic vulnerability score (PV-5) to predict complications.14,15 A score greater than the reference score of zero indicated a higher risk of postoperative complications. This scale was applied to the validation cohort. Subsequent analysis was performed to evaluate our model’s predictive performance as a risk stratification model.
To evaluate our model’s diagnostic performance, a confusion matrix was generated to compare true positives with predicted positives. Accuracy, sensitivity, and specificity were calculated at various pediatric physiological vulnerability scores to determine the best score cutoff. This analysis workflow was performed in an all-encompassing patient age group and in 2 separate age groups (0-9 and ≥10 years old). To validate our model’s predictive capabilities, all possible combinations of predictors were analyzed to create a risk calculator that provided a probabilistic output. Our model’s predictive performance was evaluated using a Brier score to generate an accuracy and Brier score loss measurement. 16
Statistical analysis was performed with the Sci-Kit Learn machine learning package in Python (version 1.1.2, Python Software Foundation, https://www.python.org/).
Results
There were 9442 patients with NMD who underwent posterior spinal fusion for spinal deformity identified. Of this total, 6609 and 2833 patients were allocated to the training and validation cohorts, respectively (Figure 1). Patient baseline demographic, clinical, and surgical characteristics for the entire study cohort are reported in Table 2. Patient characteristics were further subdivided to include the percentages within the complication and non-complication cohorts of patients. Diagram of analysis workflow. Baseline Demographic, Clinical, and Surgical Variables for Children With Neuromuscular Disease Undergoing Spinal Deformity Correction (n = 9442). Values reported as number (%) unless otherwise indicated. P values in boldface type are significant at P < 0.05. aFive without ASA classification assigned. bSee Table 1. BMI, body mass index; CNS, central nervous system; ASA, American Society of Anesthesiologists; CPR, cardiopulmonary resuscitation; CPT, Common Procedural Terminology.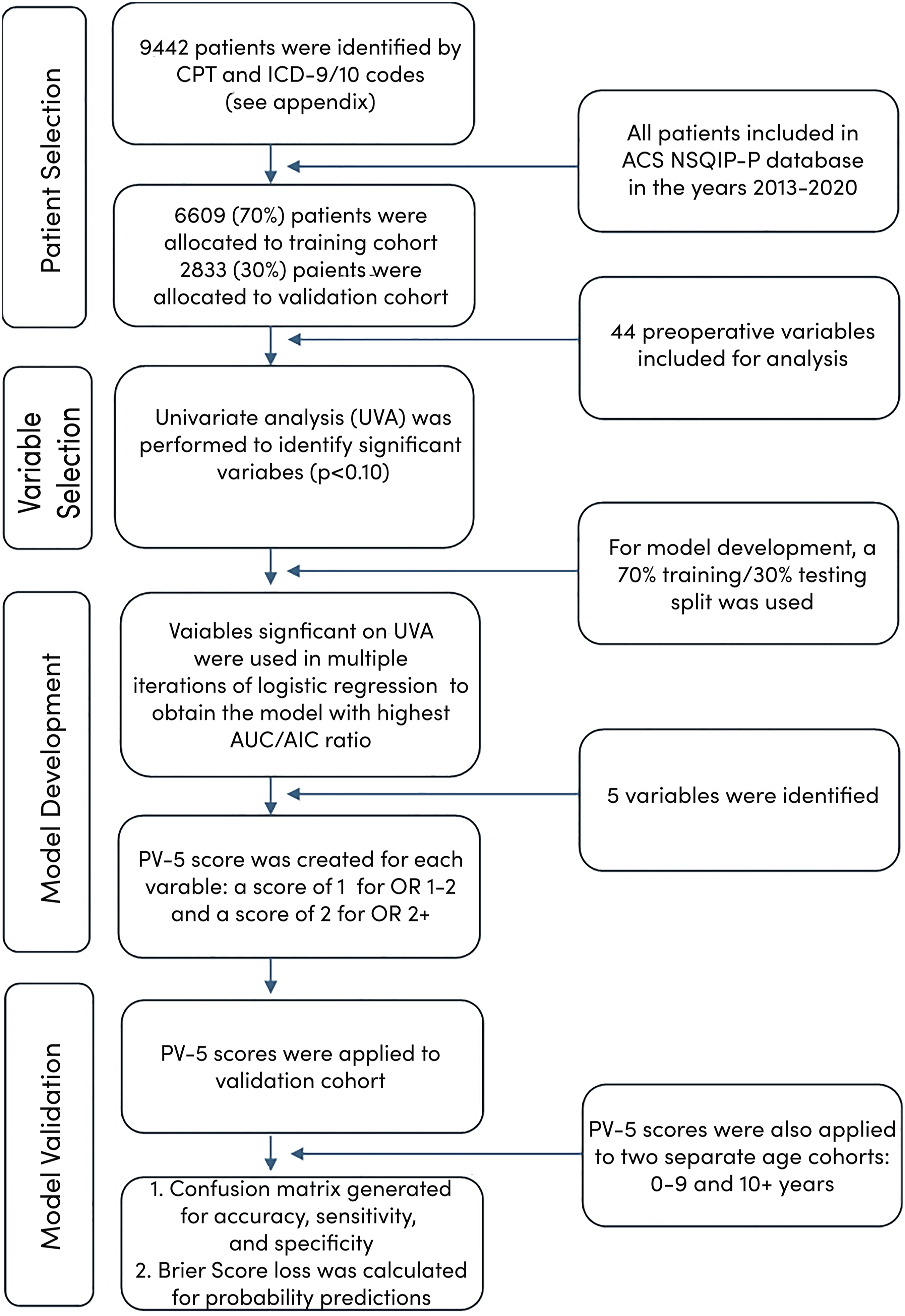
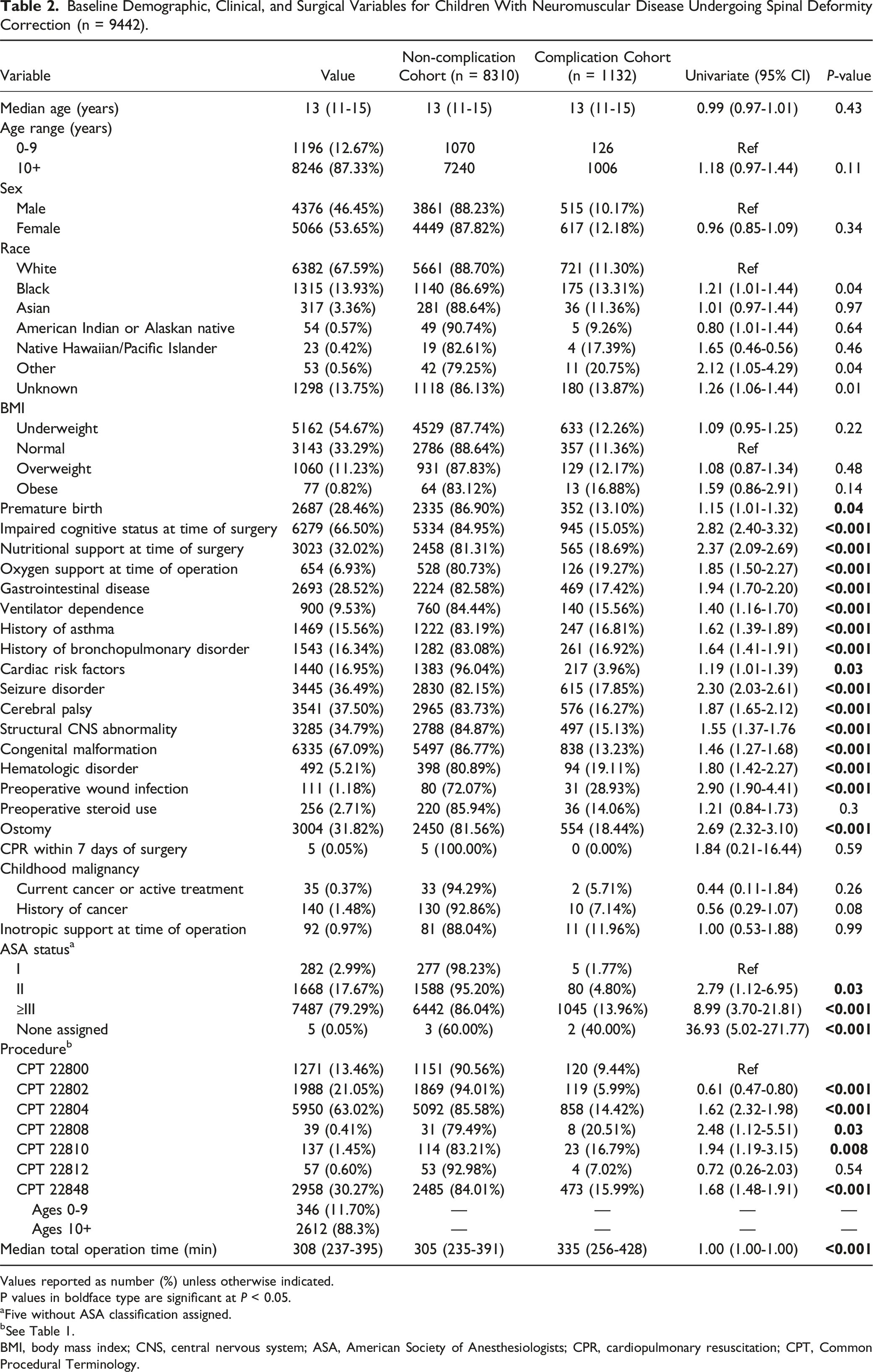
The median age of patients in the study (across both cohorts) was 13 years (11-15). Most children were ≥10 years old, but 12.67% of patients were <10 years old (Table 2). Overall, 46.45% were male and 53.65% were female. Most patients identified as White (67.59%), followed by Black (13.93%), Asian (3.36%), American Indian or Alaskan Native (0.57%), Native Hawaiian/Pacific Islander (0.42%), and other races (0.56%), with 13.75% unknown. Most patients were underweight (54.67%), 33.29% were within normal range, 11.23% were overweight, and 0.82% were obese. Many patients underwent pelvic fixation procedures (2958 [30.27%]), 346 (11.70%) of which were performed in the 0-9 age group and 2612 (88.3%) in the 10+ age group.
Reported preoperative medical comorbidities included premature birth (28.46%), impaired cognitive status at the time of surgery (66.50%), ostomy (31.82%), nutritional support at the time of surgery (32.02%), oxygen support at the time of operation (6.93%), gastrointestinal disease (28.52%), ventilator dependence (9.53%), history of asthma (15.56%), and history of bronchopulmonary disorder (16.34%) (Table 2). Previous neurological conditions included seizure disorder (36.49%), cerebral palsy (37.50%), and structural central nervous system (CNS) abnormality (34.79%). Congenital malformations were reported in 67.09% of patients, along with hematologic disorder (5.21%) and history of wound infection (1.18%). Most patients (83.05%) had no cardiac risk factors, but 8.48% had minor, 7.83% had major, and 0.64% had severe cardiac risk factors. Most patients (79.29%) had an American Society of Anesthesiologists (ASA) classification ≥III, 17.67% were ASA II, 2.99% were ASA I, and 0.05% had no ASA score assigned.
Postoperative Outcomes for Children With Neuromuscular Scoliosis Undergoing Spinal Deformity Correction (n = 9442).
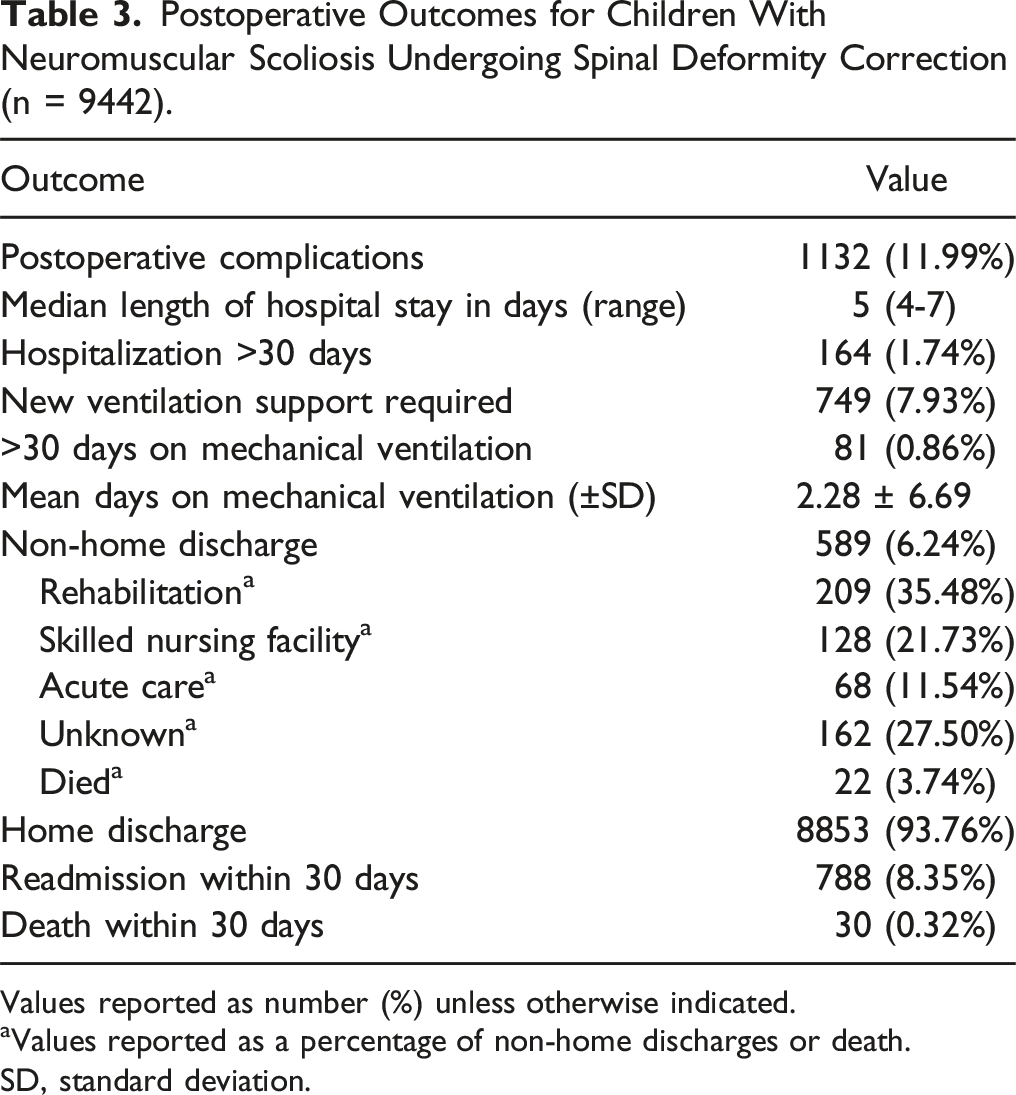
Values reported as number (%) unless otherwise indicated.
aValues reported as a percentage of non-home discharges or death.
SD, standard deviation.
Postoperative Complication Rates of Patients Having at Least One Complication (N = 1132).

SSI, surgical site infection; CPR, cardiopulmonary respiration; C. diff, Clostridium difficile; CVA, cerebrovascular accident.
Univariate Analysis
Black patients were significantly more likely to develop complications compared with White patients (OR = 1.21, P = 0.04) (Table 2). Patient age, sex, and body mass index were not significant predictors of complications. Of the 44 preoperative variables studied on univariate analysis, premature birth (P = 0.04), impaired cognitive status at time of surgery (P < 0.001), nutritional support at time of surgery (P < 0.001), oxygen support at time of operation (P < 0.001), gastrointestinal disease (P < 0.001), ventilator dependence (P < 0.001), history of asthma (P < 0.001), history of bronchopulmonary disorder (P < 0.001), cardiac risk factors (P = 0.03), seizure disorder (P < 0.001), cerebral palsy (P < 0.001), structural CNS abnormality (P < 0.001), congenital malformation (P < 0.001), hematologic disorder (P < 0.001), preoperative wound infection (P < 0.001), ostomy (P < 0.001), ASA grade II and ASA grade ≥ III (P = 0.03 and P < 0.05, respectively), pelvic fixation (P < 0.001), and total operating room time (P < 0.001) were all associated with the occurrence of complications (Table 2). OR time and pelvic fixation rates portray the variation of surgeries being performed but were not included in the final PV model.
Multivariable Analysis
Multivariable Logistic Regression Results of Model With Comorbidity Probabilities as Identified by Area Under the Curve (AUC) and Akaike Information Criteria (AIC) Analysis.
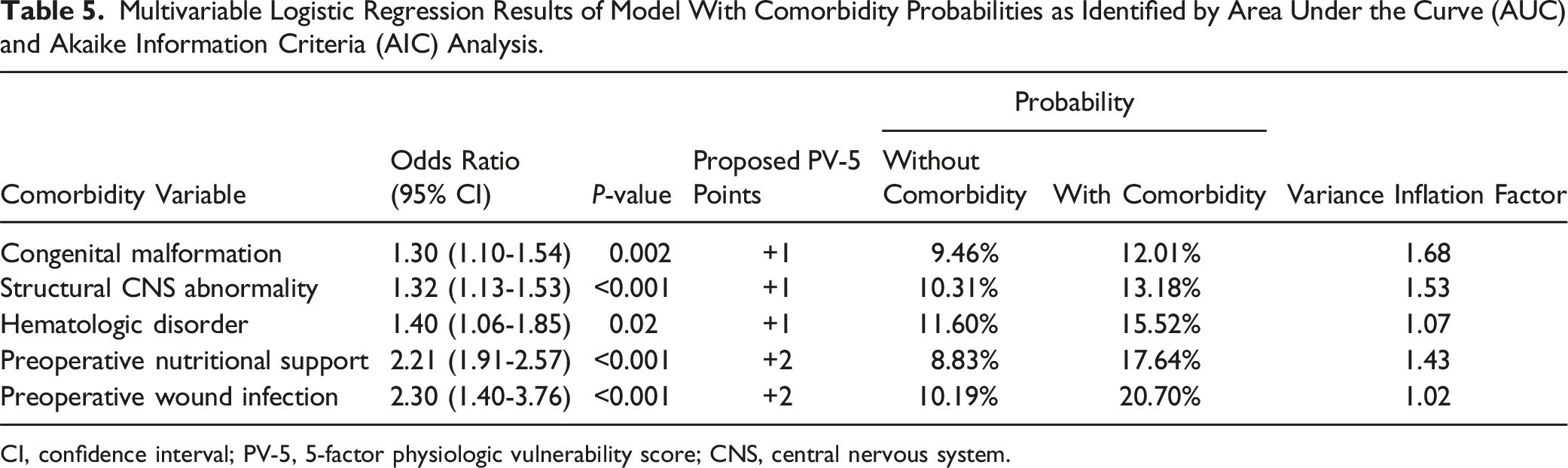
CI, confidence interval; PV-5, 5-factor physiologic vulnerability score; CNS, central nervous system.
Validation of the PV-5 Score
The validation cohort consisted of 2361 patients with demographic profiles (age, sex, comorbid conditions) like those of the training cohort (data not shown). Most patients in the validation cohort had PV-5 scores of 1 or 2 (Figure 2). Patients of all ages with PV-5 scores of 1 (OR: 2.08, CI: 1.27-3.43, P < 0.004), 2 (OR: 2.75, CI: 1.63-4.64, P < 0.001), 3 (OR: 3.67, CI: 2.18-6.19, P < 0.001), 4 (OR: 4.09, CI: 2.39-6.99, P < 0.001), and 5+ (OR: 3.58, CI: 1.35-9.47, P = 0.01) were significantly more likely to experience complications than those with a score of zero (Table 6). Patients ≥10 years with PV-5 scores of 1 (OR: 2.23, CI: 1.34-3.71, P = 0.002), 2 (OR: 2.71, CI: 1.57-4.67, P < 0.001), 3 (OR: 3.95, CI: 2.29-6.81, P < 0.001), 4 (OR: 4.27, CI: 2.43-7.51, P < 0.001), and 5+ (OR: 3.02, CI: 0.96-9.50, P = 0.04) were significantly more likely to experience complications than those with a score of zero (Table 6). Within the 0-9 age group, PV-5 scores failed to significantly predict complication rates. Distribution of 5-factor physiological vulnerability (PV-5) scores within validation cohort. Univariate Logistic Regression of Proposed PV-5 Scoring System Using the Validation Dataset. PV-5, 5-factor physiologic vulnerability score; OR, odds ratio; CI, confidence interval.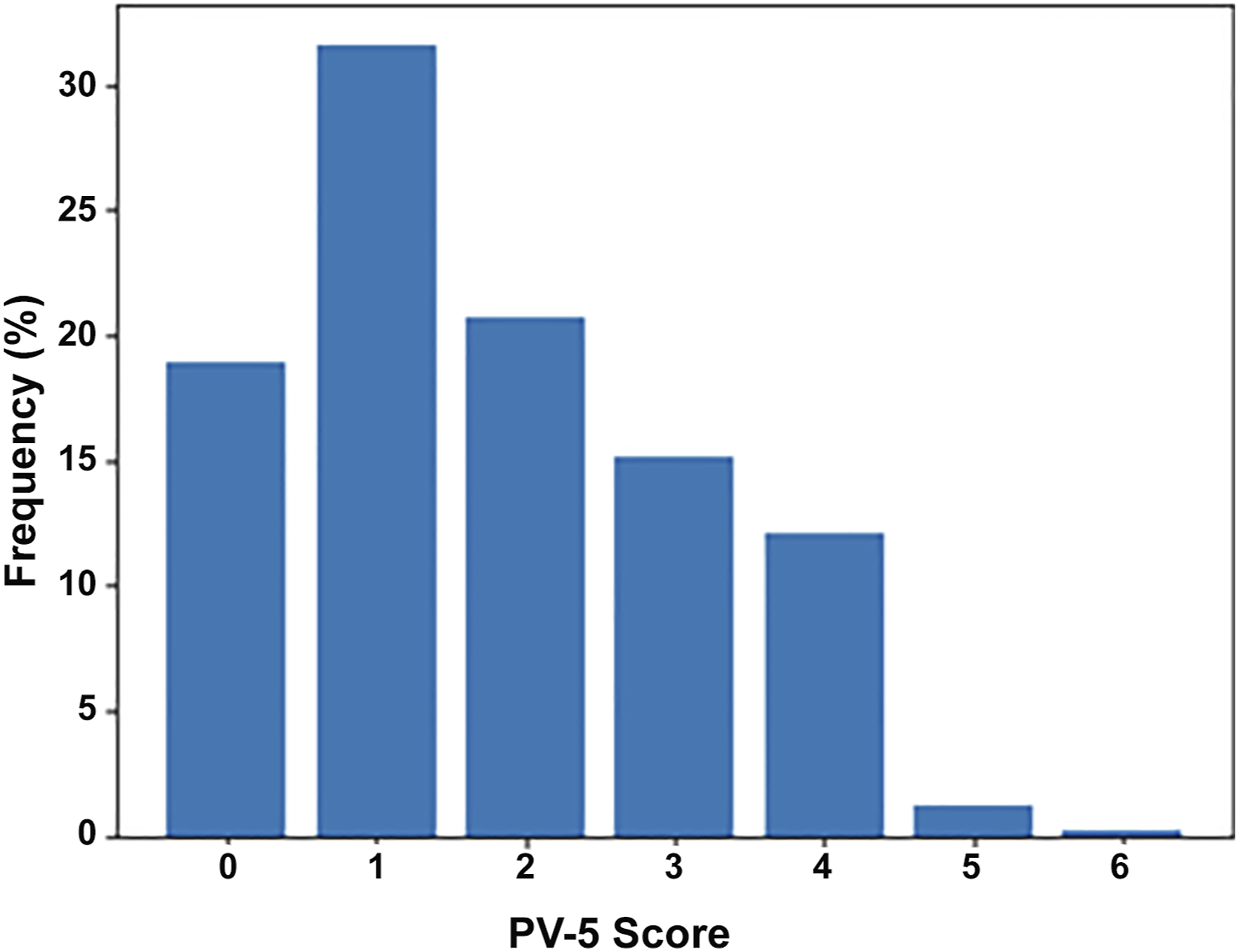
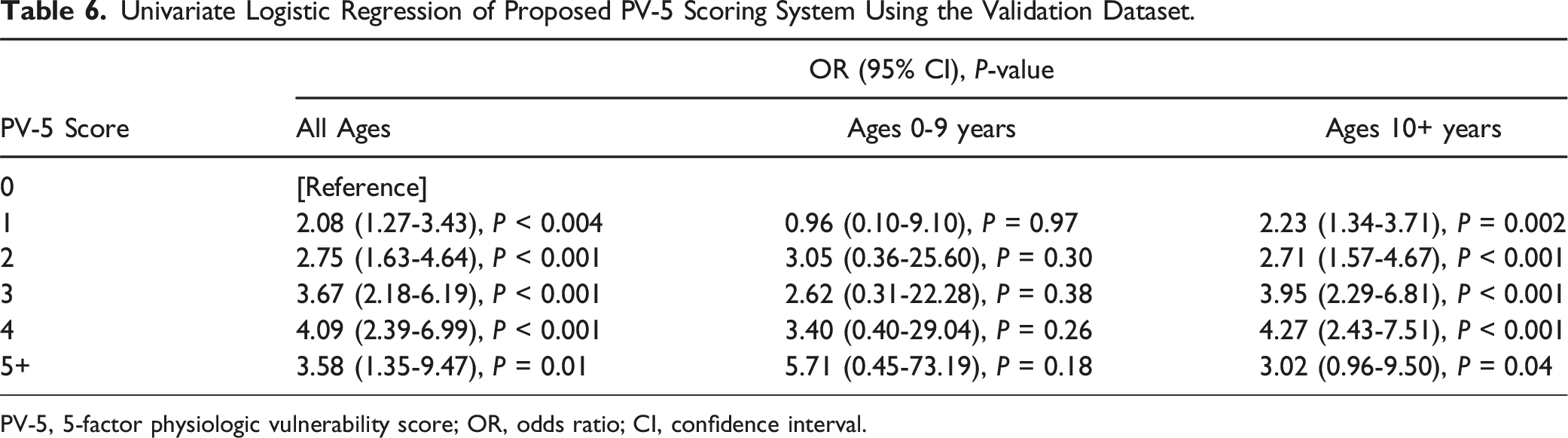
Postoperative Complication Rates Stratified by PV-5 Score (N = 9442).
P-values in boldface type are significant at P < 0.05.
PV-5, 5-factor physiologic vulnerability score; SSI, surgical site infection; CVA, cerebrovascular accident; CPR, cardiopulmonary respiration; C. diff, Clostridium difficile.
Univariate Logistic Regression and Frequency Analysis Depicting the Association of PV-5 Scores With Mortality.
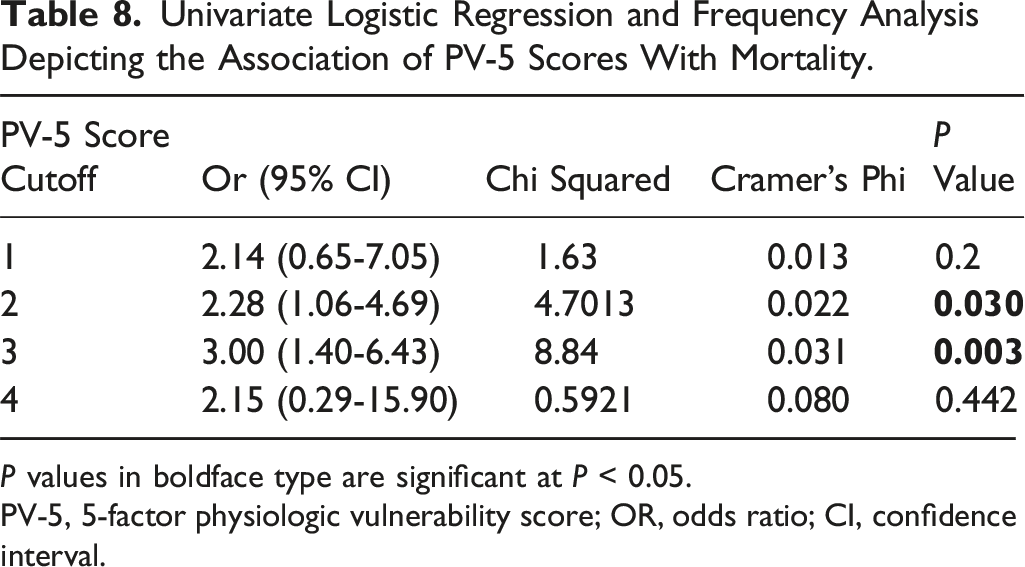
P values in boldface type are significant at P < 0.05.
PV-5, 5-factor physiologic vulnerability score; OR, odds ratio; CI, confidence interval.
Multivariate Logistic Regression Results of Prediction Model With Combined Odds Ratios and Probabilities.
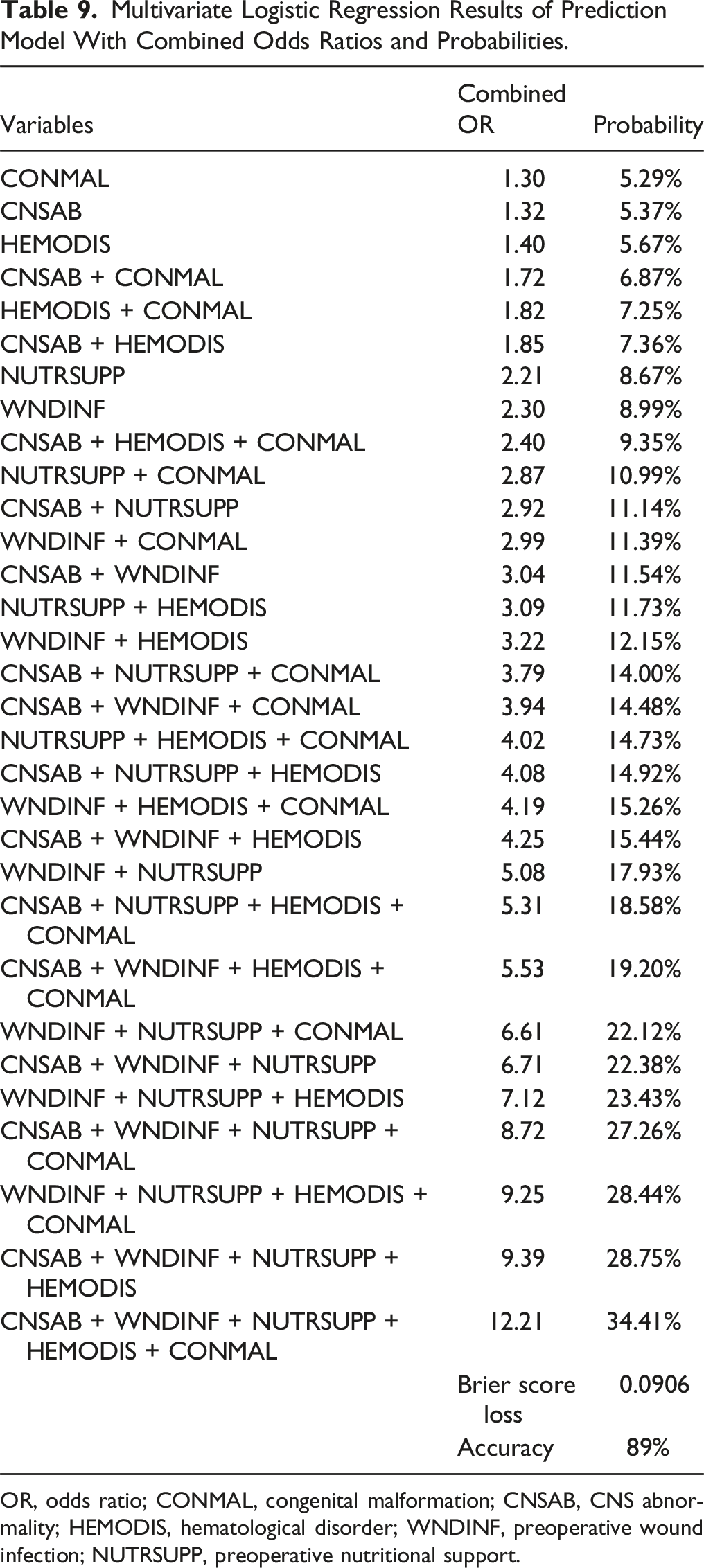
OR, odds ratio; CONMAL, congenital malformation; CNSAB, CNS abnormality; HEMODIS, hematological disorder; WNDINF, preoperative wound infection; NUTRSUPP, preoperative nutritional support.
Area Under the Curve Analysis to Evaluate PV-5 Model Performance Using the Validation Dataset.
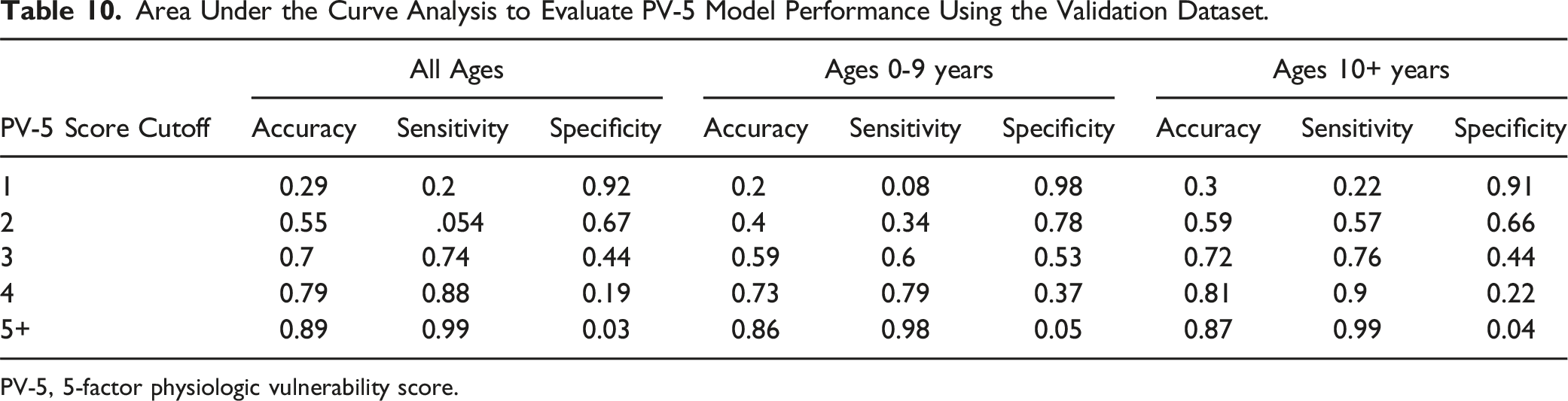
PV-5, 5-factor physiologic vulnerability score.
Discussion
In this study, we have identified a set of variables that suggest physiological vulnerability in children undergoing spinal fusion surgery for neuromuscular disease. Frailty is a complex term defined as a physiologic state of greater vulnerability to adverse health outcomes, and it was originally used to describe individuals in the geriatric population.10,17 Frail patients comprise a significant proportion of the elderly population, but patients of all ages can exhibit dysregulation of multiple physiologic systems, increasing their risk of adverse outcomes after spinal fusion.9,18 The Fried frailty phenotype was created to standardize the definition of frailty, distinguish it from comorbidity and disability, and pose a link between frailty and multisystem dysfunctions (ie, pulmonary dysfunction with low forced expiratory volume).10,19 The Canada Study of Health and Aging Frailty Index (CSHA-FI), which included 70 measurements (eg, cognitive function, nutritional status), was created to evaluate these functions. 17 Although it effectively predicts surgical morbidity, collecting the numerous variables proved to be time consuming, which inspired more concise frailty measurements such as the mFI-11 and mFI-5. 20
The mFI-11 was developed using NSQIP data and included 11 items from the CSHA-FI. However, its effectiveness has been questioned because of the frequently reported irregular recordings of the 11 variables. 20 This prompted the development of the mFI-5, which consists of just 5 variables: congestive heart failure, diabetes mellitus, chronic obstructive pulmonary disease or pneumonia, dependent functional health status, and hypertension requiring medication. Both mFI-5 and mFI-11 correlate frailty with morbidity and mortality in 9 different surgical specialties across various indications and case severities. 21 However, many of these measurements are not as relevant for the pediatric NMS population. The prevalence of many different frailty measurements underscores the need for an improved understanding of the physiologic basis of frailty and how it applies to various patient populations. 9
The heterogeneity of various disease states limits the creation of a single generalizable and clinically relevant frailty metric. 9 Our results suggest that complications in pediatric patients with NMS are not associated with the same factors as those in adult patients in general but instead have a unique profile of factors contributing to postoperative outcomes. Within the context of adult spinal deformity, preoperative risk stratification tools such as the Charlson Comorbidity Index, the mFI-5, and the adult spinal deformity frailty index generate robust postoperative complication predictions.22-29 However, these models were not created for children, including those with neuromuscular disorders. Recently developed risk stratification calculators, such as the BrAIST-Calc, have demonstrated significant prognostic benefits for the adolescent idiopathic scoliosis (AIS) population. 30 However, these calculators are geared more towards predicting curve progression than assessing surgical risk. Unlike the AIS population, children with NMS represent a high-risk surgical group, and every effort should be made to reduce the potential morbidity for posterior spinal fusion surgery.3,31-34
In our study, we found that postoperative complications were associated with 5 variables that can easily be obtained and used to stratify the risk for patients with NMS requiring deformity correction surgery. Furthermore, we have identified factors that beget vulnerability in this population, which is our key outcome of interest. The final PV-5 score includes congenital factors (malformations, structural CNS abnormalities), preoperative suggestions of physiologic impairment (nutritional support, previous wound infection), and hematologic conditions. Importantly, preoperative wound infection was defined as an open wound or an open wound infection present before surgery and did not refer to any procedure performed to address a surgical site infection. Interestingly, Jain et al. 35 reported similar factors (ie, seizures, nonverbal status, gastrostomy tube) that effectively predicted complications and health-related quality of life in patients with Gross Motor Function Classification System Level 5 cerebral palsy. These results align with previous studies that have reported higher complication rates for pediatric patients with NMS with similar preoperative comorbidities.3,34,36-39 Furthermore, patients with PV-5 scores of 2 or greater demonstrated 2-3 times higher odds of mortality, although the relatively low frequency of mortality observed in our population must be considered. These unique features imply a population-specific risk stratification profile for children with neuromuscular diseases requiring posterior spinal fusion that is different from that of adults undergoing spine surgery.26-28,40-42 For example, cardiac factors (excluding congenital cardiac malformations)—which have been highly correlated with frailty status in adults—were found in 16% of patients in our study and did not significantly predict complications. This underscores the complexity of composite measures that may require more granular analyses of individual variables to interrogate relevant associations. Interestingly, the presence of hematologic disorder or preoperative wound infection (unrelated to previous NMS surgery) was rare, but these were significant contributors in our final model. These results suggest that patients with NMS exhibit a unique set of factors contributing to their vulnerability phenotype.
Age, when analyzed as a continuous variable, did not significantly influence complication rate; however, when categorizing patients by age, we found differences in the prognostic abilities of PV-5. The PV-5 score based on children of all ages significantly predicted complications at all levels for those ≥10 years old. Prognostication of complications in patients younger than this, however, was not effective with the PV-5 score. Limited sample size may have skewed these results, although it is possible that younger patients undergoing surgery (ie, before the end of growth potential) carry a different risk profile than older children. This may be due to a differential maturity in immune profile as children age that may influence the risk of infection and poor wound healing. 43 Additionally, differences in scoliosis severity may warrant earlier treatment, which is not reflected within the PV-5 and may account for its poor performance in this population. Such risk profiles may be associated with a different state of vulnerability and warrant further investigation, especially given the known high rate of complications with growth-friendly instrumentation in the early-onset scoliosis population. 10
In children ≥10 years old, a PV-5 score of >3 accurately predicted complications for 74% of patients (accuracy = 70%, sensitivity = 74%, specificity = 44%). Patients with a PV-5 score of 5+ had complications nearly 100% of the time, although this accuracy is largely driven by sensitivity rather than specificity. Receiver operating curve analysis confirmed these findings, demonstrating our model’s ability to effectively distinguish between true positives and true negatives, as evidenced by our high sensitivity and accuracy. It is important to note that our VIF results demonstrated minimal multicollinearity among variables in our multivariable model, indicative of the accuracy of our model, particularly with minimization of overfitting and the reduction of variance between our predictive variables.
After using PV-5 to guide our model development, we were able to compare the risk of developing a complication given every combination of comorbidities from PV-5. This allowed us to develop a risk calculator that uses binary inputs (“yes” or “no”) to generate probabilistic risk outputs for every comorbidity. This model can enable earlier identification of patients with high-risk, physiologically vulnerable NMS, allowing clinicians to implement practices that can mitigate the incidence of postoperative complications. These include having appropriate discussions with family, deferring surgery, medically optimizing patients, diverting patients to higher-volume centers, and more appropriately allocating resource usage to support physiologically vulnerable patients. Patients with higher PV-5 scores may benefit from more postoperative follow-up and a lower threshold for intensive care unit admission or extended intensive care unit stay.
These broad clinical factors alone do not suggest frailty in the NMS population but rather serve as a preliminary analysis to better understand which body systems broadly predispose children to adverse events after surgery. This study builds upon the limited understanding of physiologic vulnerability within the pediatric population, a domain not well investigated using traditional frailty metrics. Physiologic vulnerability within the pediatric NMS population is composed of a distinct subset of comorbidities that may not directly align with those traditionally found in adults. We identified domains of interest that operationalize the vulnerable phenotype within pediatric patients with NMS, establishing a relationship between physiologic vulnerability and morbidity after deformity surgery. Future studies are warranted to interrogate more specific metrics within physiologic systems to potentially enhance the predictive capabilities of PV-5 and other measures of physiologic vulnerability in pediatric patients.
Limitations
The retrospective design of this study limits the ability to draw definitive conclusions regarding the prognostic abilities of the PV-5. Additionally, the use of the NSQIP database, although it is robust and well documented, may limit the generalizability and performance of the PV-5 score in other populations; however, most frailty studies are retrospective database studies with similar limitations.21,40,44,45 The NSQIP data also have limited follow-up times, preventing the analysis of long-term complications. Further, because variables to be considered were identified by a group consensus, there is a small risk of selection bias; we aimed to reduce this by including nearly all preoperative variables.
It is important to note that we only looked at patients with NMS who underwent spinal fusion. These patients undergo a variety of surgeries of varying complexities, which may include risk factors and complications distinct from those found in our heterogeneous sample. We did not stratify complications by type or severity but did choose to report the incidence of each. Although this may limit the granularity of the PV-5, the goal of this study was to robustly report associations with any complication and use this as a proxy for morbidity. Specific system-based complications would be of interest but are outside the scope of this exploratory analysis; however, the complications chosen in the composite profile are well-established, tracked metrics and thus justifiable within our chosen methodology. Lastly, we relied on reported CPT and ICD-9 and -10 codes to refine our patient population. This may lead to an underreporting of the number of patients with NMS and their potential risk of postoperative complications.
Predictive Risk Factor Definitions.

CNS, central nervous system; TPN, total parenteral nutrition.
Conclusion
Physiologic vulnerability in children with neuromuscular disease, as indicated by higher PV-5 scores, was associated with significantly greater incidence of complications after pediatric NMS deformity correction surgery. The PV-5 score and prediction model can be used as an effective risk-stratification tool to help surgeons optimize patients preoperatively and reduce adverse postoperative complications. In the future, task-based variables applicable to the NMS population will be useful to further enhance the definition of physiologic vulnerability in this population.
Footnotes
Appendix
Author Contributions
Muhammad S. Ghauri: Data curation; Formal analysis; Investigation; Methodology; Project administration; Software; Validation; Visualization; Roles/Writing - original draft; and Writing - review & editing. Sujay Rajkumar: Data curation; Formal analysis; Investigation; Methodology Software; Validation; Visualization; Roles/Writing - original draft; and Writing - review & editing. Lauren E. Stone: Conceptualization, Methodology, Formal analysis; Investigation; Validation; Visualization Roles/Writing - original draft; and Writing - review & editing. Michael P. Kelly: Validation; Roles/Writing - original draft; and Writing - review & editing. Rajiv R. Iyer: Validation; Roles/Writing - original draft; and Writing - review & editing. Jennifer Bauer: Writing - review & editing. Christopher Ames: Writing - review & editing. Peter Newton: Writing - review & editing. David D. Gonda: Validation; Roles/Writing - original draft; and Writing - review & editing. Michael L. Levy: Validation; Roles/Writing - original draft; and Writing - review & editing. Vijay M. Ravindra: Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Roles/Writing - original draft; and Writing - review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Jennifer Bauer is a consultant for Orthopaediatrics, Depuy Synthes, and Proprio; serves on the Board of Directors of the Pediatric Orthopaedic Society of North America; and her institution has received a quality improvement grant from POSNA. Christopher P. Ames is a consultant for DePuySynthes, Medtronic, Medicrea, K2M, Agada Medical, and Carlsmed; receives royalties from Stryker, Biomet Zimmer Spine, DePuy Synthes, Nuvasive, Next Orthosurgical, Medicrea, and K2M; has research support from Titan Spine, DePuy Synthes, and ISSG; and has a grant from the Scoliosis Research Society. Peter O. Newton is a consultant for DePuy Synthes Spine, Stryker/K2M, MiRus, Globus Medical, and Pacira; has received honoraria from DePuy Synthes Spine and Medtronic; receives royalties from DePuy Synthes Spine, Thieme Publishing, and Stryker/K2M; has patents with DePuy Synthes Spine (anchoring systems and methods for correcting spinal deformities [8540754], low-profile spinal tethering systems [8123749], screw placement guide [79811117], compressor for use in minimally invasive surgery [7189244]) and Stryker/K2M (posterior spinal fixation); is on the leadership of the Scoliosis Research Society, the Setting Scoliosis Straight Foundation, the Rady Children’s Specialists of San Diego, and the International Pediatric Orthopedic Think Tank; has equity interest in Accelus and Spinology; and his institution receives grant funding from DePuy Synthes Spine, the Scoliosis Research Society, EOS Imaging, Nuvasive, Orthopediatrics, Stryker/K2M, Alphatech, Mazor Robotics, and the Setting Scoliosis Straight Foundation. The other authors report no conflicts of interest.
Ethical Statement
Data Availability Statement
Data are available upon reasonable request to the corresponding author.
