Abstract
Study Design
Rabbit model study.
Objective
To examine whether cartilaginous endplate (CEP) destruction leads to endplate and vertebral marrow signal changes (Modic changes, MCs) on MR images.
Methods
Sixteen adult rabbits were used in the study and underwent an anterolateral procedure to expose the L2-6 intervertebral discs. The L4/5, L3/4 and L2/3 discs underwent annulotomy, annulotomy and CEP curettage, or annulotomy and chemonucleolysis, respectively, while the L5/6 disc served as a shame control. MR imaging was performed preoperatively and at 1, 3, and 6 months postoperatively to evaluate the presence or absence of MCs. After the last imaging, animals were sacrificed for histological study, focusing on endplate pathologies and their associations with MCs.
Results
Among the 64 endplates that underwent CEP curettage or were exposed to chemonucleolysis, there were 6 (9.4%), 19 (29.7%), and 32 (50%) endplates with MCs at 1, 3 and 6 months, respectively. No MCs developed in the sham controls. Both surgical curettage and chymopapain injection successfully induced CEP destruction. Endplates with full layer CEP defects were most likely to develop MCs (59.6% vs 11.4%, P < 0.001). Moreover, endplates with MCs had a greater histological degeneration score than those without (8.97 ± 1.92 vs 5.35 ± 2.28, P < 0.001) and higher expression levels of inflammatory factors (IL-1β, TNF-α, and IL-6, P < 0.05 for all) in the subchondral vertebral marrow.
Conclusions
CEP destruction, induced either by physical curettage or chemical lysis, can lead to long-lasting inflammation in the vertebral marrow and Modic-like signal changes on MR images. CEP destruction may be a root pathology underlying MCs.
Introduction
Modic changes (MCs), signal variations in the vertebral trabeculae adjacent to the endplate, 1 are common findings on lumbar spine magnetic resonance (MR) images. 2 MCs have long been suspected as a pathological cause of back pain and numerous clinical studies on MCs have been conducted, yet to date the association between MCs and back pain remains controversial. On the other hand, the pathogenesis of MCs is less studied, and a validated animal model of MCs has not been established. A number of theories have been proposed to explain the occurrence of MCs, including mechanical stress alteration, 3 low-virulent infection,4,5 and autoimmune inflammation. 6 Yet, none is universally accepted.
Clues from clinical and histological studies suggest that the cartilaginous endplate (CEP) is often damaged in the presence of MCs. Early histological studies of tissue samples observed that MCs are often accompanied by endplate fissures and disruptions, with vascular granulation in Type-I MCs and fatty degeneration in Type-II MCs.1,7,8 In patients with lumbar disc herniation who underwent discectomy, cartilage fragments are more commonly identified in the removed disc tissues when adjacent MCs are present,9-11 as compared with those without adjacent MCs, suggesting that CEP avulsion is closely associated with MCs. Chemonucleolysis, a therapeutic strategy for treating disc herniation by injecting chymopapain into the disc, often leads to Type-I MCs in the adjacent vertebral marrow within 6-12 weeks. 1 One explanation is that chymopapain is a non-specific proteolytic enzyme which dissolves both nucleus tissues and CEP, resulting in CEP damage and inflammation in the adjacent vertebral marrow and thus signal changes on MR images. Moreover, in patients with pyogenic discitis, the infection usually destroys or dissolves the endplate, resulting in a spread of infection to the vertebral trabeculae seen as vertebral signal changes on MR images. 12 In fact, recent studies also suggest that infections of low-virulent bacteria, such as propionibacterium acnes, are associated with Type I MCs.4,5 In addition, MCs are more common in spondylolisthesis 3 and surgical cases of intervertebral fusion 13 where the CEP is disrupted by either mechanical alteration or surgical curettage. It seems that CEP disruption may be a key pathology underlying vertebral marrow signal changes on MR images, though not all these signal changes are called MCs.
Given the current evidence, we hypothesized that CEP disruption is a root pathological cause underlying MR signal changes at the endplate and adjacent vertebral marrow, and the causes of CEP disruption may be biological, chemical, physical, or a combination of these factors. The current study aims to examine whether CEP destruction leads to endplate and vertebral marrow signal changes on MR images by using physical curettage and chemical lysis to induce CEP destruction in the lumbar spines of rabbits. As biological etiologies, such as pyogenic discitis, are well-known causes for signal changes in the vertebral marrow,14,15 and MCs are generally regarded as degenerative findings, we were interested in the effects of mechanical or chemical CEP disruption, rather than infection and discitis, on vertebral marrow signal changes on MR images.
Materials and methods
Animals
Sixteen 6-month-old male New Zealand rabbits, with a mean body weight of 5 kg (range 4.4 to 5.3 kg), were used. The study protocol was reviewed and approved by the review board and ethics committee at the authors’ institution (ZJU2020-053-05).
Surgical Procedures
The rabbits were anaesthetized with sodium pentobarbital (30 mg/kg) via the ear vein. A 5 cm longitudinal incision was made along the tips of the left lumbar vertebral transverse processes (Figure 1(A)). Through the inter-muscle space between obliquus externus abdominis and erector spinae, the peritoneal membrane was pushed anteriorly to expose the lumbar spine. Then, the psoas was detached to expose the anterolateral portion of the lumbar intervertebral discs (Figure 1(B)). Surgical procedure of exposing the L2-6 lumbar discs for various experimental interventions. (A) A 5 cm longitude incision was made along the tips of the lumbar vertebral transverse processes on the left side. (B) Through the inter-muscle space between obliquus externus abdominis and erector spinae, the anterolateral side of the lumbar spine was exposed. The intervertebral discs are bulging and in white. (C) Annulotomy was performed using an 18G needle. The needle punctured through the full layer of the AF, and punctures were repeated to create a 3 mm AF breach. (D) Followed annulotomy, CEP curettage was performed using a dental curette. Cartilage of both cranial and the caudal endplates of the L3/4 disc was scratched. The top view of a disc that underwent CEP scratching. While most of the CEP interfacing the NP was removed, the osseous endplate remained largely intact (white arrow).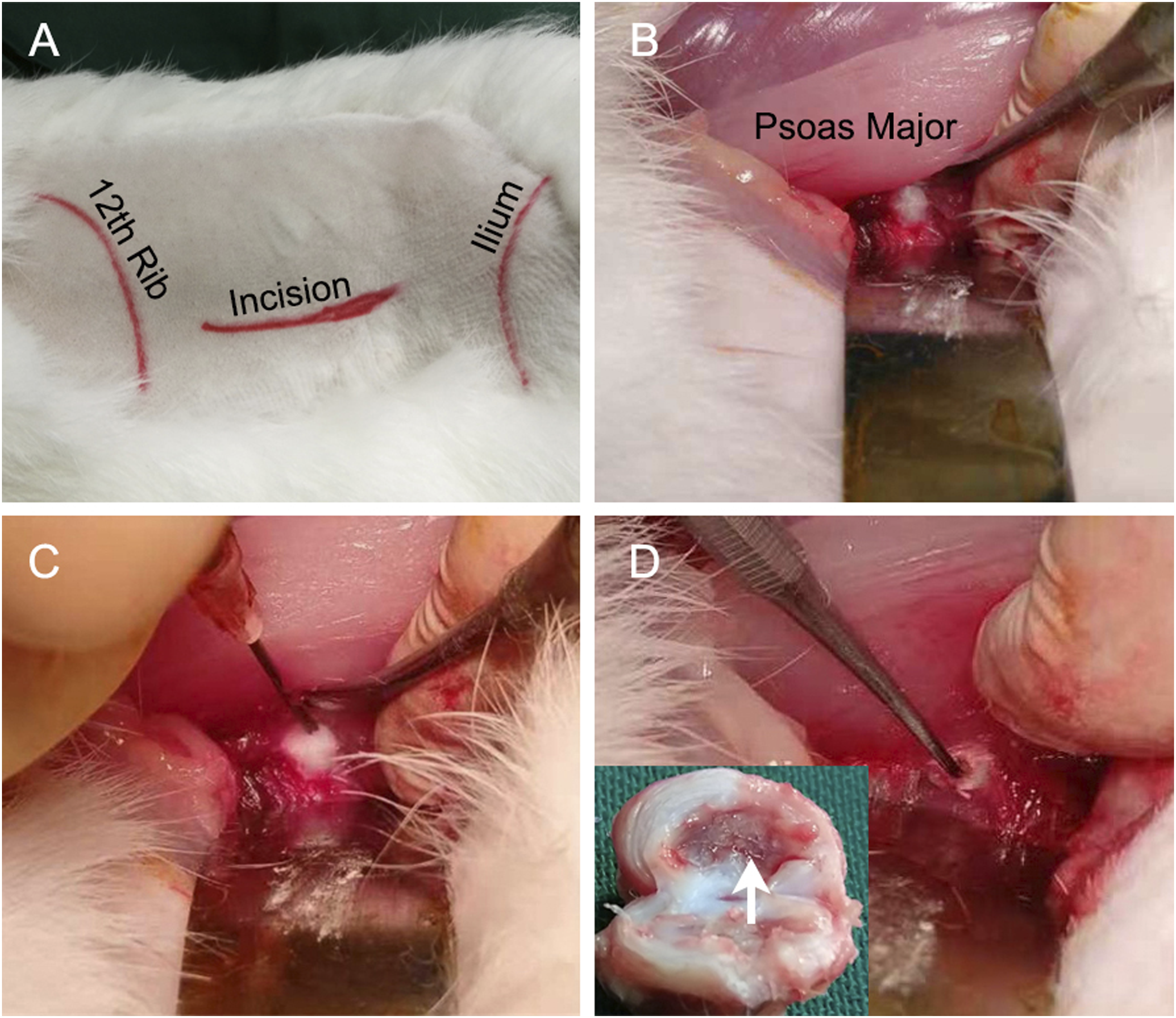
For each animal, the L2-6 discs were used. The L5/6 disc was exposed but there was no other intervention (sham control). For the 3 discs at the L2-5 segments, annulotomy was first performed. An 18 G needle was punctured through the anterolateral portion of the annulus fibrosus (AF) to the nucleus pulposus (NP). A full layer puncture of AF was then repeated to enlarge the AF breach until it was approximately 3 mm in diameter (Figure 1(C)). For the L4/5 disc, only annulotomy was performed and was also taken as a control. Following annulotomy, CEP curettage was performed at the L3/4 disc. A dental curette was inserted through the AF breach to the center of the disc to scratch both the cranial and the caudal CEP of the disc (Figure 1(D)). Usually, the tip of the dental curette was stopped by the contralateral AF and the aim of CEP curettage was to remove the CEP interfacing the NP tissues. The curettage was performed softly back and forth for 10 times with same force. By this way, CEP can be peeled off from the vertebral endplate and the bony endplate can remain basically uninjured. For the L2/3 disc, chemonucleolysis was performed by injecting 100 μL of 0.25% chymopapain (Merck, Germany) after annulotomy.
Tramadol was administered orally to relieve pain and penicillin (Haerbin, China) was intramuscularly injected (0.2 million U, once per day) for 3 days. The rabbits were then raised individually with free access to food and water for further experiments. After the last MR imaging, which was performed 6 months postoperatively, the rabbits were sacrificed with an overdose of pentobarbital sodium (30 mg/kg) for histological study and PCR analysis.
MR Imaging
Each animal underwent MR imaging before surgery, and 1, 3, and 6 months after surgery. MR imaging of the lumbar spine was performed using a 3.0 T MR scanner (Philips, the Netherland) and a knee-joint surface coil. T1W sagittal images were acquired with a TE/TR of 8/442 ms, and T2W sagittal images with a TE/TR of 120/2500 ms. The matrix size was 268 × 191with a field of view of 24 × 24 cm. A slice thickness of 3 mm and intersection gap of 0.3 mm were used. For each animal, there were 5 T1W and 5 T2W MR images.
Evaluation of MCs and Disc Degeneration
A senior radiologist and a spinal surgeon independently evaluated all MR images blinded to disc level, surgical procedure, and timepoint. When disagreements occurred, the case was re-evaluated and discussed to reach a consensus. MCs were first assessed as present or absent, and then, if present, by specific subtype of MCs.
On T2W sagittal images, quantitative measurements of signal intensity were further obtained for the adjacent vertebral marrow and disc to assess the changes in signal over time. Using Image J (Version 1.53, NIH, USA), signal intensity was measured as the mean signal measurement within a defined region of interest (ROI). On the mid-sagittal T2W sagittal images, the ROIs were manually drawn for the disc, subchondral vertebral marrow 2 mm from the CEP, and the spinal cord immediately behind the disc (Figure S1). Signal intensity of the spinal cord was used as a reference to adjust signal intensity measurements of the disc and subchondral marrow.
Histology Study of the Endplate and Disc
After the last MR imaging at 6 months, the animal was sacrificed. The lumbar spine was excised and soft tissues were removed. Each spine was sagittally bisected into 2 halves with a band saw. One half was dissected into ‘vertebra-disc-vertebra’ segmental specimens, fixed with 4% paraformaldehyde at 4°C for 48 hours and then decalcified with EDTA for 10 days. Using a Leica microtome (Leica, RM2125), serial 5μm-thick sagittal sections were obtained. Sections were then stained with hematoxylin and eosin (HE) and safranin O-fast green for histological examination.
Histological evaluation of the endplate was performed using Boos’ scale, 16 which incorporates 3 variables to assess CEP (cell proliferation, disorganization, and cracks, each rated on a zero to 4-point scale) and another 3 for osseous endplate (microfracture, new bone formation, and sclerosis, graded as zero to 2). The grades were then summed for each endplate, with a greater score indicating a greater degree of endplate degeneration (maximum 18 points). In addition, full layer CEP defects, which were defined as remarkable focal or extensive CEP absence upon the osseous endplate, were recorded. Disc degeneration was evaluated using Lai’s approach. 17
Two spine surgeons independently assessed all histological slices blinded to disc level. Data were merged to identify disagreements, which were resolved by re-evaluation and discussion.
PCR Analysis of Inflammatory Factors
The other half of the spinal segmental units was used for qPCR analysis of the subchondral trabeculae. Disc tissues and CEP were removed and approximately 3 mm thick subchondral trabeculae, including the osseous endplate, were dissected. Total RNA was isolated using Trizol Reagent (Takara Bio, Japan). Reverse transcription was performed using Prime Script Master Mix (Takara Bio, Japan). The obtained cDNA was amplified with a Real-time PCR System (7900; Applied Biosystems, USA) and a SYBR Green Real time PCR Master Mix (Takara, Japan). The primers of related genes are listed in Table S1. PCR reactions began at 95°C for 10 minutes for activation, followed by amplification with 40 cycles at 95°C for 10 seconds, 55°C for 20 seconds, and 72°C for 20 seconds, and a final extension at 72°C for 60 seconds.
Statistical Analyses
The differences in the occurrence rate of MCs, signal intensity of subchondral vertebral marrow and disc, histological scores and RNA expression among groups were examined using t-tests and Chi-square tests, as appropriate. The relationship between endplate degeneration and the occurrence of MCs was examined using a multivariable logistic regression model. Statistical analyses were performed using SPSS (Version 26.0, SPSS Inc, USA).
Results
The Occurrence Rate of MCs on MR Images
The Presence of MCs at Adjacent Vertebral Marrow on 1, 3, and 6-month MR images (n = 32 Endplates for each group).
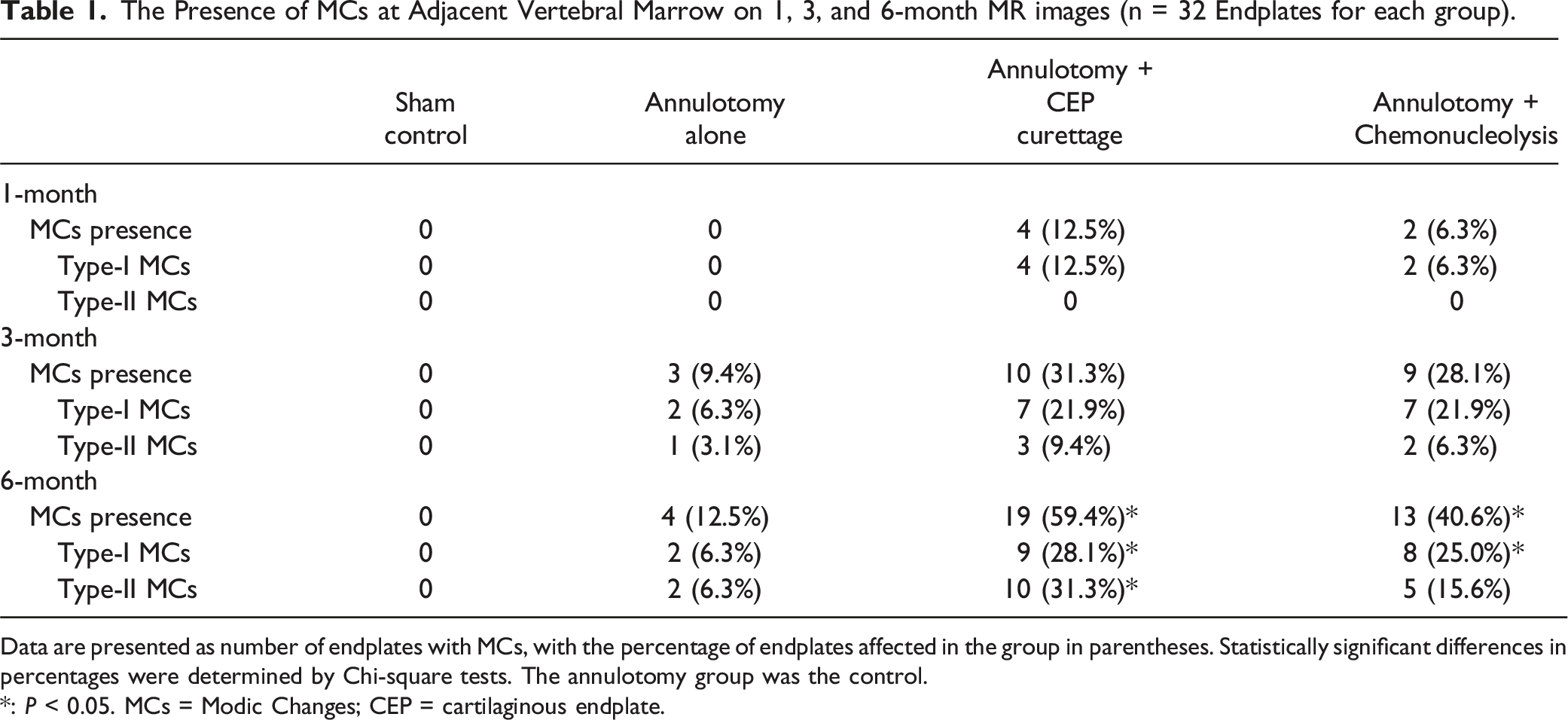
Data are presented as number of endplates with MCs, with the percentage of endplates affected in the group in parentheses. Statistically significant differences in percentages were determined by Chi-square tests. The annulotomy group was the control.
*: P < 0.05. MCs = Modic Changes; CEP = cartilaginous endplate.
For discs that underwent annulotomy and CEP curettage (the L3/4 disc), there were 4 (12.5%) endplates with type-I MCs 1 month after surgery, which increased to 10 (31.3%) at 3 months and 19 (59.4%) at 6 months. Notably, 3 out of 4 type-I MCs at 1-month MR imaging transformed to type-II at 3 months. Between 3 and 6 months, 5 out of 7 type-I MCs transformed to type-II, and the size of most MCs increased during this period.
Of the L2/3 discs that underwent annulotomy and chemonucleolysis, only 2 (6.3%) endplates had type-I MCs on 1-month MR images, which transformed to type-II at 3 months. The occurrence of MCs increased to 9 (28.1%) at 3 months and to 13 (40.6%) at 6 months. Similarly, 3 of the 9 type-I MCs on 3-month MR images transformed to type-II at 6 months. Representative MR images are presented in Figure 2. Representative T1W and T2W images of the lumbar discs under various conditions. The sham control L5/6 disc was exposed without additional intervention, and no MCs were observed on the cranial or caudal endplates at follow-up MR images. There are also no MCs observed on the Annulotomy only L4/5 disc. The L3/4 disc underwent annulotomy and CEP curettage demonstrates type-I MCs on the caudal endplate relative to the disc on 1- and 3-month MR images, which converted to a typical type-II MC at 6 months and increased in size. Meanwhile, the cranial endplate also had type- II MCs. The endplates adjacent to the L2/3 disc, which underwent annulotomy and chemonucleolysis, had type-I MCs on 3-month MR images, and type-II MCs at 6 months. Hollow arrows indicate endplates with type-I MCs and solid arrows indicate endplates with type-II MCs. MCs = Modic Changes; CEP = cartilaginous endplate; T1W = T1-weighted; T2W = T2-weighted.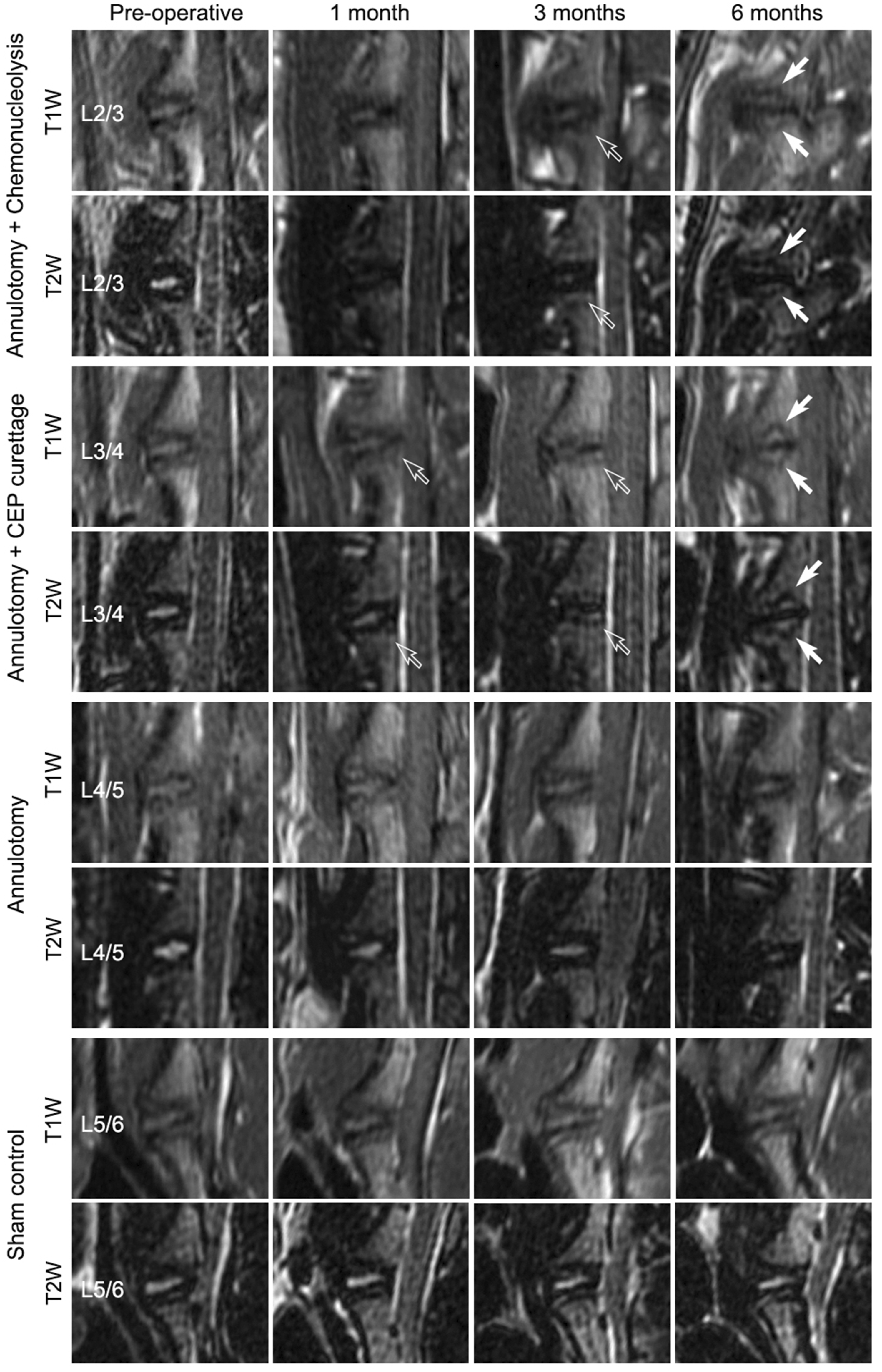
At 6 months postoperatively, the occurrence rate of MCs was significantly higher in endplates adjacent to discs that underwent CEP curettage and chemonucleolysis, as compared with those that underwent annulotomy alone (P < 0.05 for both, Figure 3). While there were more endplates with MCs following CEP curettage and chemonucleolysis than annulotomy at postoperative 1 and 3 months, differences were not statistically significant (Figure 3). The occurrence rates of MCs on 1, 3, and 6-month MR images (n = 32 endplates for each group). Chi-square tests were used and the reference was the group that underwent annulotomy alone. *: P < 0.05; MCs = Modic Changes.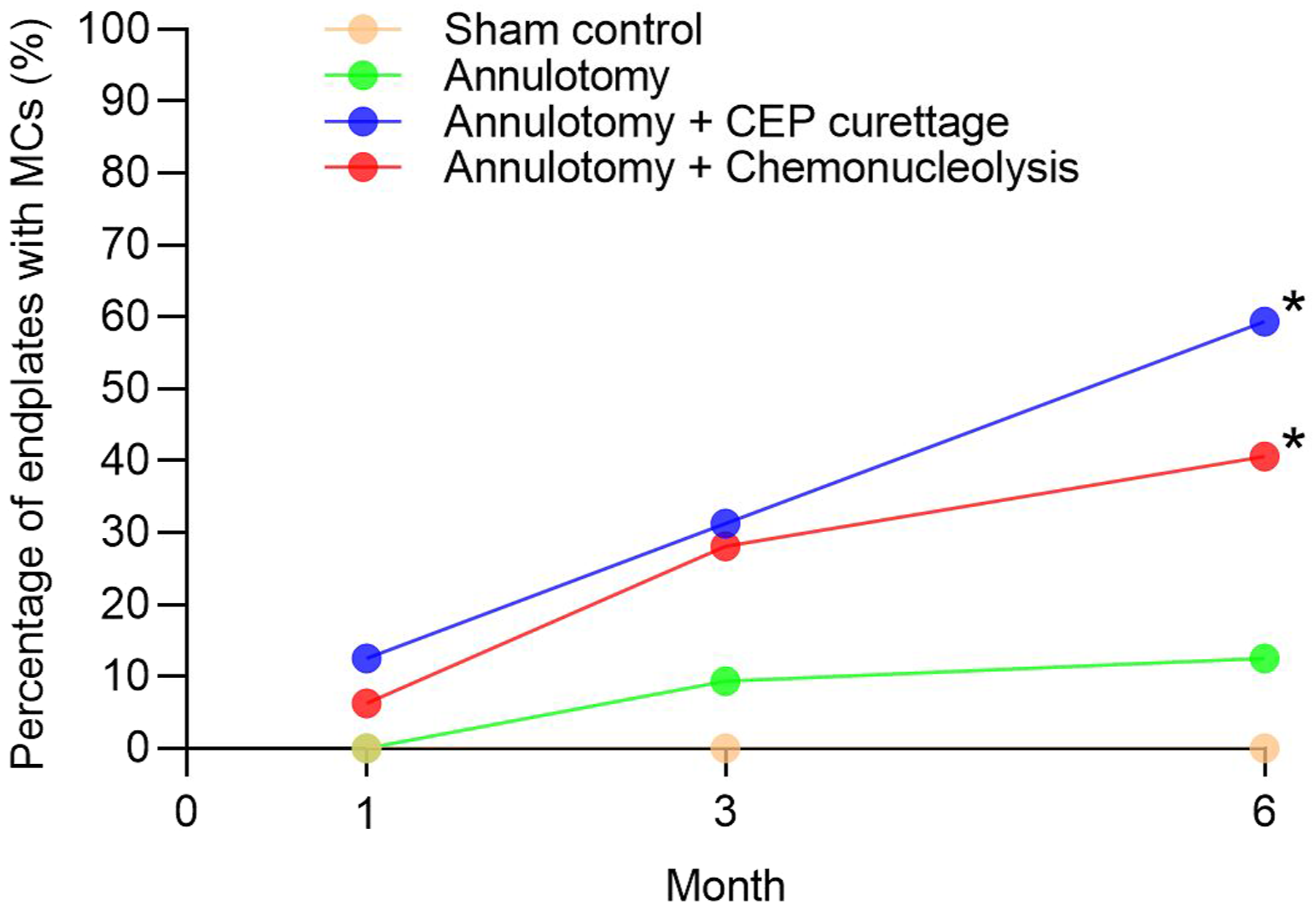
Signal Intensity Measurements
Adjusted Signal Intensity Measurements of Subchondral Vertebral Marrow and Intervertebral Disc on T2W MR Images.
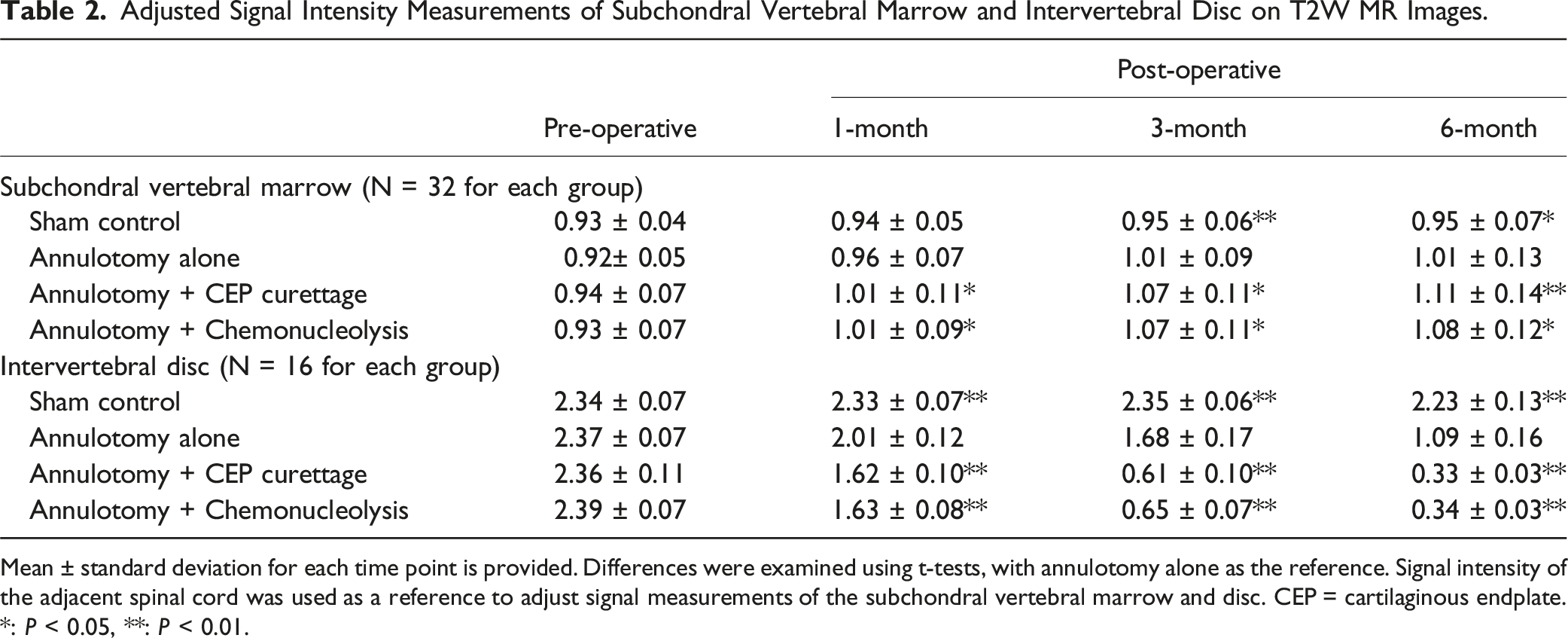
Mean ± standard deviation for each time point is provided. Differences were examined using t-tests, with annulotomy alone as the reference. Signal intensity of the adjacent spinal cord was used as a reference to adjust signal measurements of the subchondral vertebral marrow and disc. CEP = cartilaginous endplate.
*: P < 0.05, **: P < 0.01.
Histological Study
In gross examination, while the sham control discs retained their gelatinous NP tissues, diminished NP and decreased disc height occurred in most interventional discs (Figure 4(A) to (D)). Moreover, NP invasion through the endplate to the adjacent marrow were common in discs treated with CEP curettage (12/32) or chemonucleolysis (6/32) (Figure 4(C) and(D)). Gross histology, HE and SO staining of lumbar segments that underwent different procedures. Images in the “20×” rows are magnifications of the regions in black rectangles in corresponding low-magnification images. A, E, I: Sham control disc. NP tissues are gelatinous and disc height is normal. An intact layer of CEP (dotted line), which is well-structured hyaline cartilage, is tightly attached to osseous endplate (OEP). B, F, J: Disc treated with annulotomy only. NP tissues are fragmented and shrunk, and CEP is thinned and disorganized (triangle arrow in F). However, the connection of OEP-CEP is intact (black arrow in J). Tears of AF (black arrow in F) can be observed. (C, G) K: Annulotomy and curettage performed. Full layer defects of CEP (asterisk in G and K) are extensive and microfracture can be observed in the OEP (arrow in G and K). Furthermore, invasion of NP tissues into the defects of the OEP are common (arrow in K). In the OEP, there were some cracks and fissures, with vacuoles inside (triangle arrow in G). (D, H) L: Annulotomy and chemonucleolysis. Extensive CEP lysis with localized full layer CEP defect can be observed, especially at the corner of the endplate. OEP = osseous endplate; CEP = cartilaginous endplate; NP = nucleus pulposus; HE = Haematoxylin and eosin.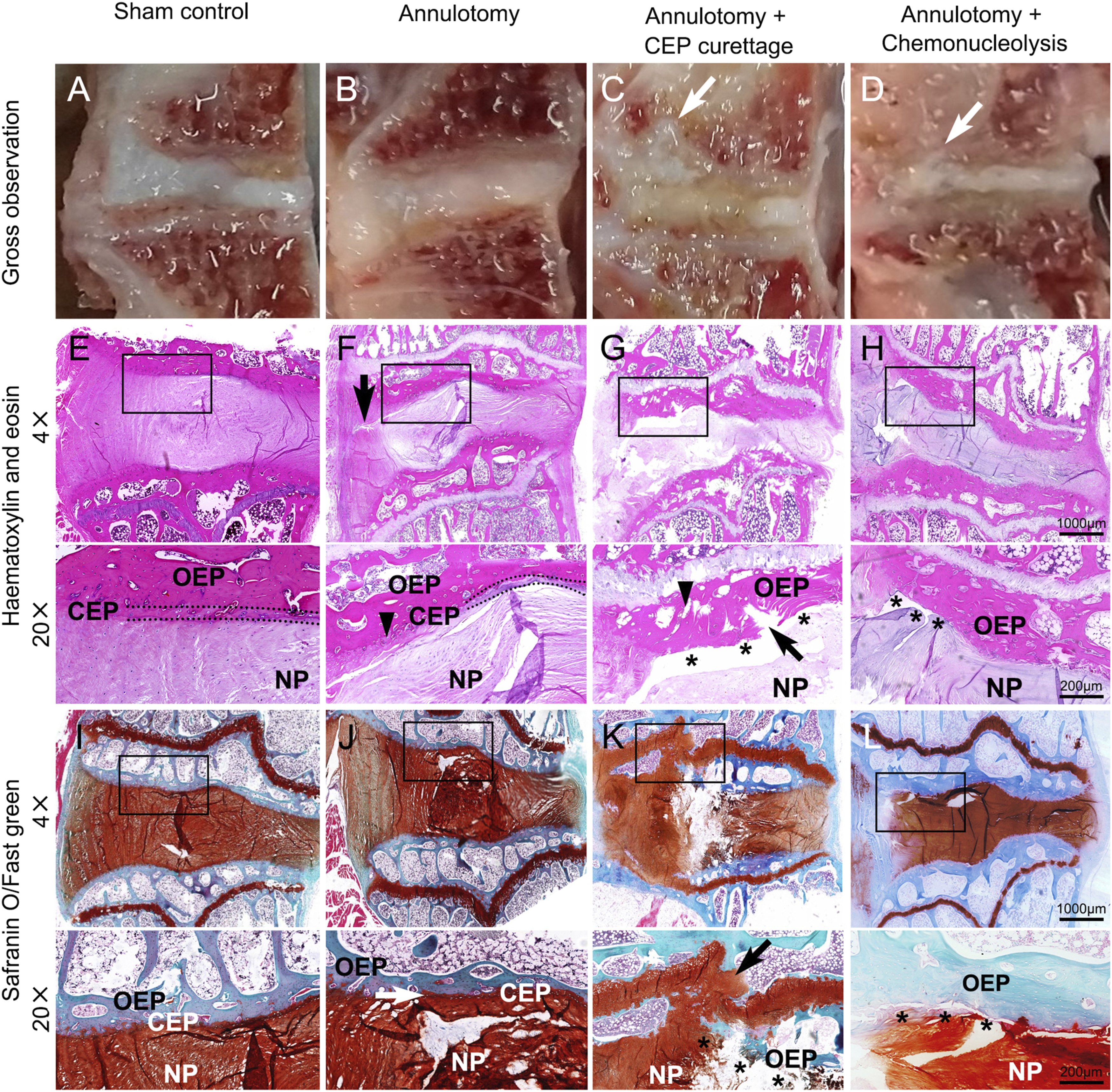
In histological examination, disc degeneration was apparent in most discs that underwent intervention, along with varying degrees of CEP destruction. For discs with annulotomy alone, a main histological feature was CEP thinning, along with small endplate defects and CEP disorganization (Figure 4(F) and (J)). On the other hand, the connection between the cartilaginous and osseous endplate was intact, and full layer CEP defects and osseous endplate microfracture were rare (Figure 4(F) and (J)). For discs that underwent annulotomy and CEP curettage, full layer CEP defects were observed for all endplates, which also presented with severe osseous endplate microfracture (Figure 4(G) and (K)). For discs that underwent annulotomy and chemonucleolysis, endplates presented extensive CEP lysis or avulsion, especially at the corner of an endplate (Figure 4(H) and (L)). Microfracture on the osseous endplate occurred in 24 of the 32 endplates studied (75%), with most rated as mild microfracture according to Boos’ definition.
Histological Evaluation of the Endplate (n=32 for each group).
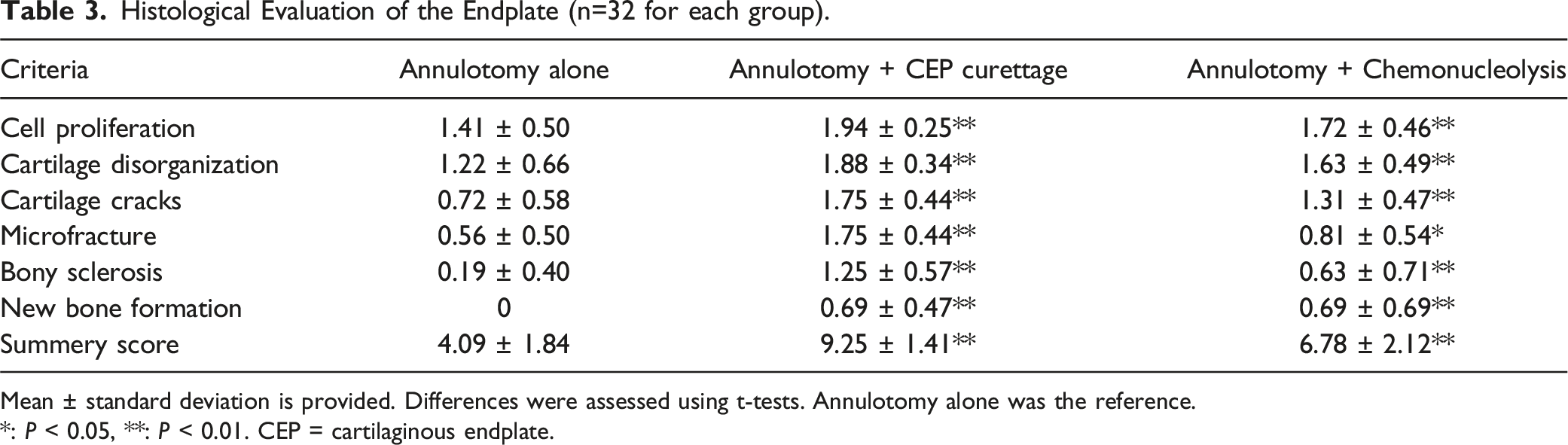
Mean ± standard deviation is provided. Differences were assessed using t-tests. Annulotomy alone was the reference.*: P < 0.05, **: P < 0.01. CEP = cartilaginous endplate.
Associations of MCs with Histological Evaluations and Inflammatory Factors
We pooled endplate data from the 3 experimental discs (excluding those in the sham control group) to examine endplate pathologies in relation to MCs on 6-month MR images. As a result, 96 endplates from 48 discs were analyzed. Among them, 36 (37.5%) endplates presented with MCs on 6-month MR images.
Histology Analysis of Endplates With or Without MCs on MR Images.
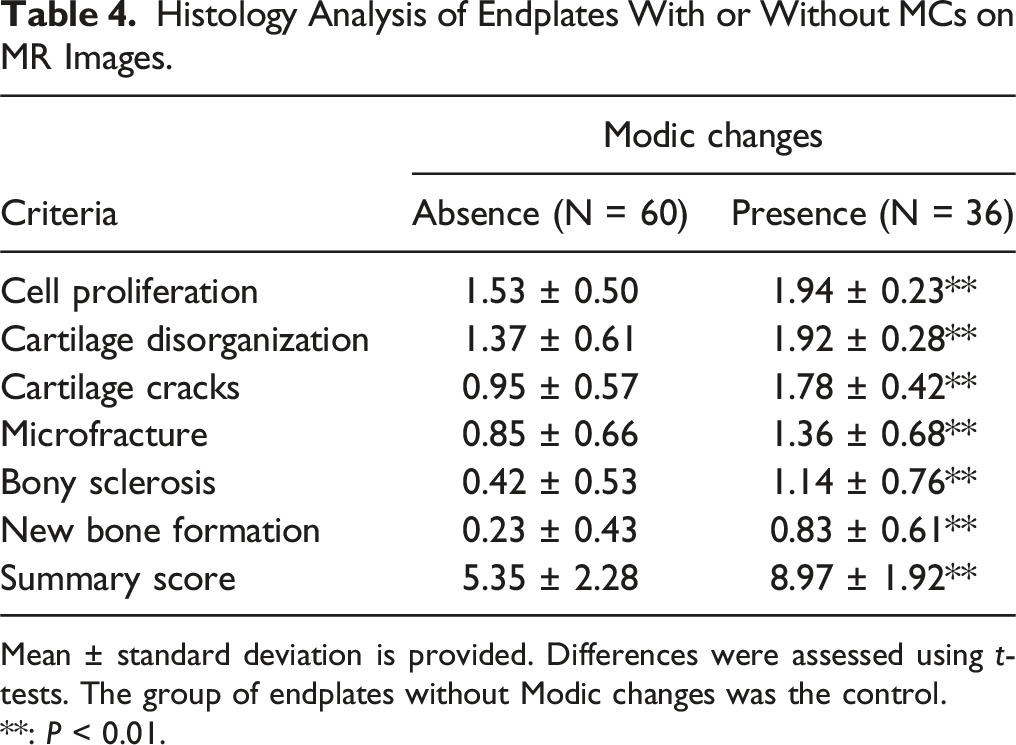
Mean ± standard deviation is provided. Differences were assessed using t-tests. The group of endplates without Modic changes was the control.**: P < 0.01.
Associations of Histological Degeneration of the Disc and Endplate with MCs.
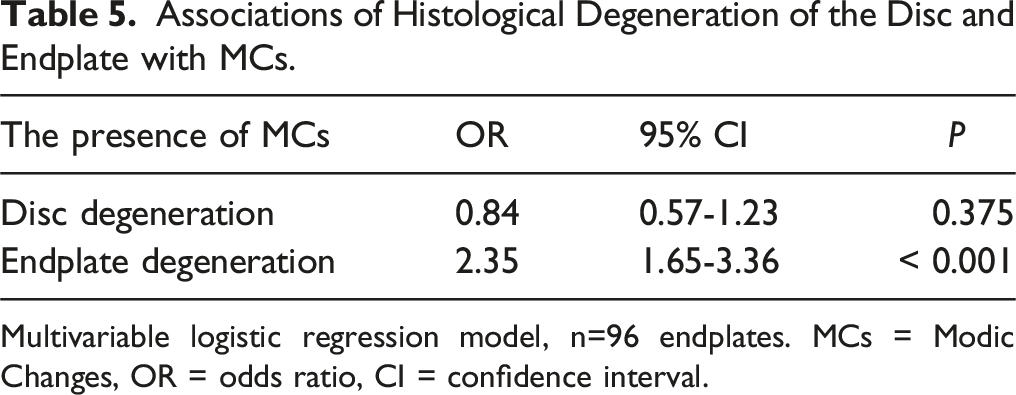
Multivariable logistic regression model, n=96 endplates. MCs = Modic Changes, OR = odds ratio, CI = confidence interval.
In addition, the expression levels of IL-1β, TNF-α, and IL-6 were significantly higher in the subchondral vertebral marrow with MCs than those without (P < 0.05 for all, Figure 5). The expression of IL-1β (A), TNF-α (B), and IL-6 (C) in the subchondral vertebral marrow with or without MCs. MCs = Modic changes; IL-1β = interleukin 1beta, TNF-α = tumor necrosis factor alpha; IL-6 = interleukin 6. ***P < 0.001.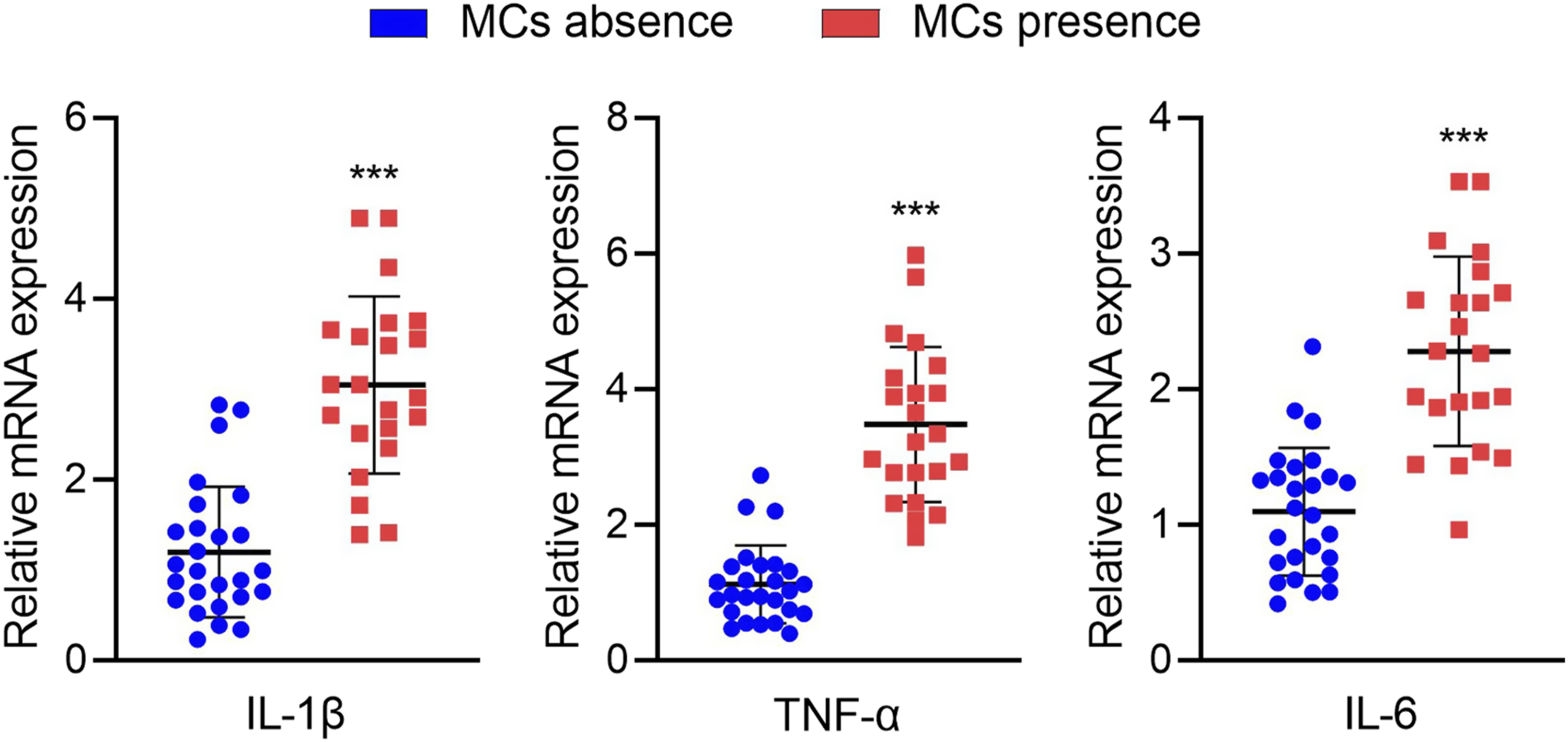
Discussion
Using surgical curettage and chymopapain, we induced CEP destruction in rabbit lumbar discs and found that, overall, 29.7% of adjacent endplates developed Modic-like signal changes at 3 months, which increased to 50.0% at 6 months. Moreover, histology demonstrated that CEP damage, particularly full layer CEP destruction, was closely associated with the occurrence of inflammation and MCs in the adjacent vertebral marrow. These findings suggest that CEP destruction, induced either by physical damage or chemical lysis, may lead to persistent inflammation in the vertebral marrow reflected as signal changes on MR images. In other words, CEP destruction may be a root pathology underlying MCs.
Our findings are congruent with clinical observations of an association between the presence of MCs and CEP avulsion, with CEP fragments identified more commonly in discectomy tissue from discs with adjacent MCs.9-11 Other studies have revealed an association of the presence and size of vertebral endplate defects (lesions) with MCs and disc degeneration.18,19 In addition, a clinical study of 386 patients with lumbar disc herniation suggested that the presence of CEP avulsion was associated with adjacent MCs and residual back pain. 10 It seems that MCs, endplate defects, and disc degeneration are parallel phenomena, and that endplate pathologies, which can influence both the vertebral trabeculae and the disc, play a key role in the development of MCs and disc degeneration.
As most studies on MCs have been based on clinical observations, the pathogenesis of MCs has remained elusive. Attempts to establish animal models of MCs, such as NP tissue transplantation to vertebral trabeculae 6 and Propionibacterium acnes injection to the disc,4,5 induced MCs that were neither typical nor repeatable. 20 In contrast, high-virulent germs, such as tuberculosis and staphylococcus aureus, which are capable of dissolving the CEP, often lead to vertebral signal changes on MR images,14,15 although these signal changes usually are not called MCs. A recent animal study also reported that surgically induced osseous endplate microfracture can lead to Modic-like signal changes. 21 It seems that disruption of endplate integrity, either the CEP or the osseous endplate, is related to signal changes of the vertebral marrow.
Consisting of osseous and cartilaginous components, the endplate is a physical shield between the vertebral trabeculae and disc. It is well documented that disruption of the endplate integrity leads to adjacent disc degeneration, likely through mechanisms of direct loss of nucleus contents, and consequent inflammation, nutrient supply impairment, and mechanical alternations. 22 It is noteworthy that the adjacent vertebral trabeculae also undergo similar biological and mechanical alternations as the disc. When the endplate is destroyed, the immune-privileged NP tissues may invade the adjacent trabeculae, as commonly observed in the current study, resulting in persistent inflammation. Moreover, efflux of cytokines to the vertebral trabeculae, as driven by intradiscal pressure, increase. 23 Indeed, we detected remarkably elevated IL-1β, IL-6 and TNF-α in the vertebral trabeculae. As a result, CEP destruction may initiate long-lasting inflammation in the adjacent vertebral marrow, reflected as signal changes on MR images.
On the other hand, osseous endplate pathologies have also been investigated in relation to MCs.19,24 Consistent with previous studies that CEP destruction can cause osseous endplate defects, 25 histological analysis in this study revealed that 60.9% of endplates with full layer CEP defects had various degrees of osseous endplate microfracture or defects, of which 59.6% presented with MCs, suggesting a close association of CEP destruction with both osseous endplate defects and MCs. Collectively, findings suggest that CEP destruction may be a key link between endplate defects and MCs.
The current study has many strengths, including establishing an animal model of MCs by destroying the CEP via physical curettage or chemical lysis, but there are also limitations that must be noted. The Modic-like signal changes we modelled may not fully reflect MCs in the human spine, as MCs are generally thought to be degenerative in nature. While a series of follow-up MR imaging was performed to understand the occurrence of MCs over time, we only followed 6 months. Given the increase in the occurrence of MCs from 1 to 6 months postoperatively, a longer follow-up may have revealed more MCs and better demonstrated variations in their temporal development. Also, detailed molecular pathomechanisms of CEP destruction and MCs were not studied.
In conclusion, this study demonstrated that CEP destruction can lead to persistent inflammation of the adjacent vertebral marrow and MCs on MR images, supporting CEP destruction as a root pathology underlying MCs. This study also provides a practical animal model for future basic science research on MCs.
Supplemental Material
Supplemental Material - Cartilaginous Endplate Damage May be a Root Pathology Underlying Modic Changes on Lumbar Spine MR Images
Supplemental Material for Cartilaginous Endplate Damage May be a Root Pathology Underlying Modic Changes on Lumbar Spine MR Image by Zhiyun Feng, Shuchen Ding, Honghao Wu, Miao Liu, Xiaojian Hu, Lunhao Chen, Kai Wu, Michele C. Battie, and Yue Wang in Global Spine Journal
Footnotes
Author Contributions
ZYF and YW conceived and designed the study. The animal model establishment and magnetic resonance imaging were performed by ZYF, SCD, ML, KW and XJH. The MCs evaluation was performed by XJH, YW, and ML. Histology and PCR were performed by HYW and SCD and then evaluated by XJH and ZYF. The data analyses were performed by SCD and ZYF. ZYF, SCD, YW and MCB wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (82102599), Science Technology Department of Zhejiang Province (2020C03042), and Zhejiang Provincial Natural Science Foundation of China (LTGY24H060003).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
