Abstract
Study Design
Randomized Controlled Trial.
Objective
Chronic low back pain (CLBP) is a major public health concern that will continue to grow with the expected aging of the population. The purpose of this study was to examine the clinical effect of a personalized, home-based biomechanical intervention compared to traditional physical therapy in patients with CLBP.
Methods
This was a randomized controlled trial. One-hundred and sixty-two patients were randomized in a 2:1 ratio to a home-based biomechanical intervention (HBBI, AposHealth) or traditional physical therapy (TPT), respectively. Patients were assessed at baseline and after 12 weeks and 52 weeks. The primary outcome measure was pain at 52 weeks, using a standard Numeric Rating Scale (NRS). Secondary outcomes included pain and function metrics, quality of life and objective spatio-temporal gait test. A Linear Mixed Model assessed changes over time across all study visits.
Results
A significant reduction in NRS was found after 52 weeks with a superiority effect of the HBBI arm compared to TPT (F = 13.82, P < 0.001). Patients in the HBBI arm demonstrated a marginal mean reduction of 3.5 points, from 6.2 to 2.7 (a 56% reduction), while patients in the TPT arm reported a mean decrease of 1.8 points from 6.9 to 5.1 (a 26% reduction).
Conclusions
A new foot-worn, home-based, biomechanical intervention for patients with chronic non-specific back pain was found to be clinically effective. Given the lack of non-surgical, non-pharmacological interventions for this populations, this treatment might serve as an adjunct to the current standard of care.
Keywords
Introduction
Low back pain (LBP) is a common musculoskeletal disorder that affects a large proportion of the global population. 1 About 15% of adults will develop chronic low back pain (CLBP), defined as LBP lasting more than 3 months, resulting in periods of pain and many will have significant physical limitations that affect a patient’s quality of life. 2 Patients with CLBP are characterized by greater comorbidity and higher direct and indirect costs compared with those without CLBP. This poses a significant burden on the individual, healthcare, and society.2-4 Frequently, patients with CLBP are characterized by work absenteeism 5 and reliant on pain relief medications, predominantly opioids. 6 LBP is a major public health concern that will continue to grow with the expected aging of the population, hence there is a need for improvement of LBP assessment, non-surgical treatments and prevention programs.
Biomechanics plays an essential role in the occurrence of CLBP. Poor posture, altered spinal alignment, and muscle imbalances are some of the reasons for chronic pain. Since surgical interventions have a poor record of benefit, non-surgical interventions have been widely used to manage LBP, with the aim of reducing pain and disability, improving function and quality of life, and preventing recurrence. 7 Because non-surgical interventions have some effect in managing CLBP, they are considered a first-line treatment option before surgical interventions are considered. Among the most commonly used non-surgical approaches are pharmacological interventions, bracing, epidural steroid injections, manual manipulation, physical therapy, acupuncture, and mindfulness meditation and education. However, since the effectiveness of these interventions is limited with varying responses, there is a dire need to develop more effective interventions and personalize them for the specific needs of each patient. 8 Recently, in December 2023 The World Health Organization (WHO) released its first-ever guidelines on managing CLBP in primary and community care settings, listing interventions for health workers to use and also to not use during routine care. 9 WHO recommends a holistic care plan addressing a mix of factors (physical, social and psychological) that may influence the patient’s condition. Non-surgical interventions include education programs, exercise, some physical therapies such as spinal manipulation therapy and massage, psychological therapies such as cognitive behavioral therapy and medications such as NSAIDs. WHO also advised against some non-surgical interventions including lumbar braces, belts and/or supports, some physical therapies such as traction and some medications such as opioids. 9
Utilizing the ability to alter biomechanics, a personalized foot-worn biomechanical device for gait rehabilitation combined with a home-based program was shown to be effective for patients with knee OA.10,11 Using similar principles to correct altered biomechanics, and train neuromuscular control, preliminary studies using the aforementioned biomechanical device for patients with CLBP have demonstrated clinical efficacy,12,13 including our experience with the device. 14 The biomechanical footwear causes alterations to the ground reaction forces going through the biomechanical chain up to the lower back. As the device can normalize gait and posture, we hypothesized that the use of the device in CLBP may help to restore normal low back mechanics and improve pain and function. To our knowledge, there are no randomized controlled trials that compared this intervention with a sham device or standard care. Therefore, the current study aimed to examine the clinical effect of a personalized, home-based biomechanical intervention compared to traditional physical therapy.
Methods
Enrollment Criteria.

Protocol
Patients in both groups underwent an initial evaluation by a physical therapist that included a physical examination, patient-reported outcome measures (PROMs) and a computerized gait assessment. Patients in the intervention arm were fitted with the biomechanical device and were given a home-based treatment plan. Patients in the TPT arm received up to 20 in-clinic PT sessions, utilizing the department rehabilitation protocol for patients with LBP (Appendix 1). Patients were asked to return to study visits for outcomes re-assessments at 12 and 52 weeks which also included tracking of compliance and adverse events.
Intervention
All patients in the intervention group received a personalized, non-invasive, home-based, biomechanical intervention that aims to alleviate pain and improve function (AposHealth®). AposHealth is an FDA-cleared biomechanical device that utilizes a shoe with an interface to attach two convex pods to the plantar surface of the sole (Figure 1). The device is personally calibrated (i.e., adjusted in a customized manner) to the individual to alleviate pain while walking, accounting for symptoms, physical examination, and gait patterns. By adjusting the location of the pods, the ground reaction force vectors are altered via changes in the center of pressure, which changes the moments on the lower extremity and lower back. The convex nature of the elements also induces a level of controlled perturbation that provides neuromuscular training. Overall, the device setup aims to off-load and destabilized the joint thorough mirco instability and retraining of the neuromuscular system and utilizes gait rehabilitation protocol to achieve healthier gait pattern and clinical relief simultaneously. This is fundamentally different than insoles, orthotics, wedges and braces which aim to support the joint. Apos system.
Following the initial evaluation and calibration of the device in the clinic, patients received a home-based treatment plan which included wearing the device for approximately 30 min per day while doing regular tasks at home or work. Patients were instructed to do intervals of standing/walking while accumulating 15 min in total, out of the 30 min (50% of wear time). Typically, patients were instructed to gradually increase the wear time for up to 2-3 h per day indoors. Some patients were advised to add outdoor walking as tolerated. In addition, patients were advised to return to follow-up appointments as scheduled in the protocol where the device was re-calibrated as needed and the ongoing treatment plan adjusted as needed. There is no need for special training on device usage. During the initial evaluation and calibration of the device, patients get acquainted with the device and walk with it under the supervision of the physical therapist. Thereafter, the patient receives home-based usage instructions verbally and in writing. The device is not a smart shoe and is not connected to power. Moreover, the device does not have internal or external sensors attached to it, nor does it have a supporting patient application and patients are not required to upkeep the device technically.
Adherence to the treatment plan was assessed at 3 months by the physical therapist. The clinician inquired about adherence to wearing the device, including average use pattern (frequency and time), and documented whether patients were compliant with the program or not. Compliance was classified into 3 groups: compliant (using the device per instructions), partially compliant (self-managing with the device), and non-compliant (not using the device at all).
Traditional PT
Physical therapy for CLPB included two to 3 visits a week for 4 to 8 weeks (up to 20 sessions), depending on the progression of the patient. Each session takes about 1 h. Physical therapists followed the department’s standard care protocol with minor adjustments as needed. A typical physical therapy regimen for CLBP includes core strengthening, stretching exercises, therapeutic modalities and education. Compliance with the TPT program was assessed by counting the number of visits patients attended. Patients were classified into one of 3 groups: compliant – those who attended at least 7 TPT appointments; partially compliant – those who attended between 1 - 7 appointments; non-compliant – those who did not attend any TPT appointment.
Patients in both groups were allowed to consume non-surgical interventions during the study period.
Outcomes
Basic demographic information included age, sex, body mass index (BMI), ethnicity, race, and baseline assessment of all outcome measures. The primary outcome measure was changes in the Numeric Rating Scale (NRS) for lower back pain at inception and 52 weeks. Secondary outcome measures were: NRS at 12 weeks, Oswestry Disability Index (ODI), PROMIS Pain Interference and Physical Function and the MOS item of the Short Form (SF)-36 at baseline, 12 weeks, and 52 weeks. Similarly, a computerized gait test was performed to evaluate gait velocity at baseline, 12 weeks, and 52 weeks. Patients walked barefoot at a self-selected speed over a four-meter walkway mat, with two meters before and after to allow for sufficient acceleration and deceleration time outside the measurement area (Zeno walkway, ProtoKinetics).
Statistical Analysis
Based on previous research,12-14 we estimated that a sample size of 147 patients would yield 80% power to detect an expected difference of 1.5 at a 2-sided alpha of 0.05 on a pain scale from 0 to 10. The difference corresponds to a clinically meaningful moderate effect size of 0.4 standard deviation units assuming a typical standard deviation of 2.65. Anticipating an attrition rate of 10%, we aimed to recruit 162 patients for the trial (53 patients will be assigned to the TPT arm and 109 patients assigned to the HBBI arm in a 2:1 ratio. In designs where one group serves as a control (e.g., standard treatment), increasing the size of the experimental group can help ensure that the trial has sufficient power to detect differences between the treatments and therefore a more conservative approach was used with respect to the experimental group.
Descriptive statistics of baseline measurements and group differences were measured using t test. Primary outcome measures were assessed using a linear mixed model analysis which compared the first and last visit. A Linear Mixed Model assessed changes over time across all study visits, controlling for group differences in case of significant differences at baseline. The p-level was set to P < 0.05. To avoid various misleading artifacts, we checked our hypotheses based on the intention to treat (ITT) analysis. All patients were included in the analyses, and multiple imputation was used for missing outcome data.
Results
One-hundred and sixty-two patients were enrolled to the study in a 2:1 ratio, 109 patients were allocated to the HBBI arm, and 53 patients were allocated to the TPT arm. Ninety-one patients (83%) from the HBBI group and 39 (74%) from the TPT group completed the study. The difference in drop-out rate between groups was not significant (P = 0.138). Of the 109 patients enrolled in the active group, 18 patients (17%) dropped out. Two patients never started, 7 patients dropped out before 3 months, and 9 patients attended the 3 months assessment and dropped out afterward. Of the 53 patients enrolled in the control group, 14 patients (26%) dropped out. Ten patients were enrolled and never started TPT (one of them due to COVID-19), and four patients declined to continue and dropped out before 3 months. At 3 months there were 85 patients and 30 patients with complete data for the intervention and control group, respectively. For the intervention group, 77% of the patients were compliant, 5% were partially compliant and 17% were non-compliant. In comparison, 6% of patients treated with TPT were compliant, 49% were partially compliant, and 37% were noncompliant.
Baseline Characteristics. Results are Presented as Mean (SD).
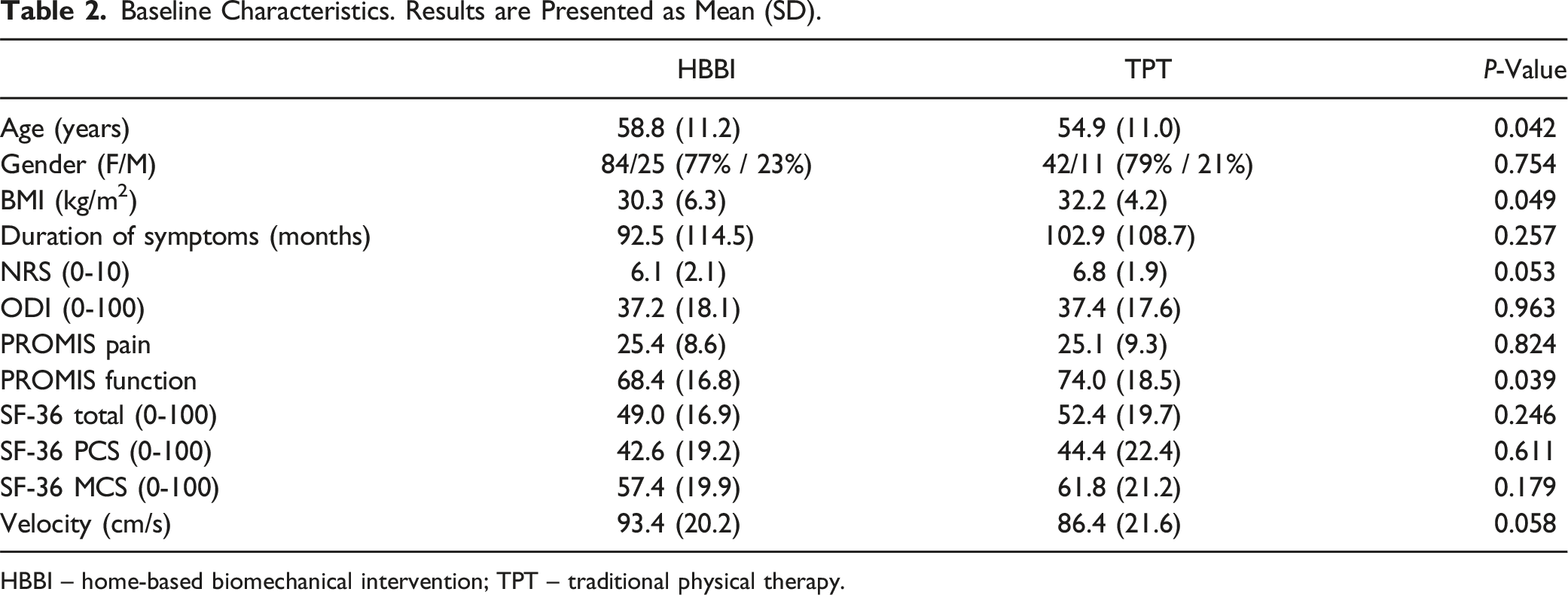
HBBI – home-based biomechanical intervention; TPT – traditional physical therapy.
Primary Outcome Measures
A significant reduction in NRS was found after 52 weeks with a superiority effect of the HBBI arm compared to TPT (F = 13.82, P < 0.001). Patients in the HBBI arm demonstrated a marginal mean reduction of 3.5 points, from 6.2 to 2.7 (a 56% reduction), while patients in the TPT arm reported a mean decrease of 1.8 points from 6.9 to 5.1 (a 26% reduction). Patients in the HBBI met The Minimally Clinically Important Change for NRS in patients with CLBP, which is defined as a reduction of at least 2.5 points.15,16
Secondary Outcome Measures
Changes at 52 Weeks. Results are Presented as Mean (SE).
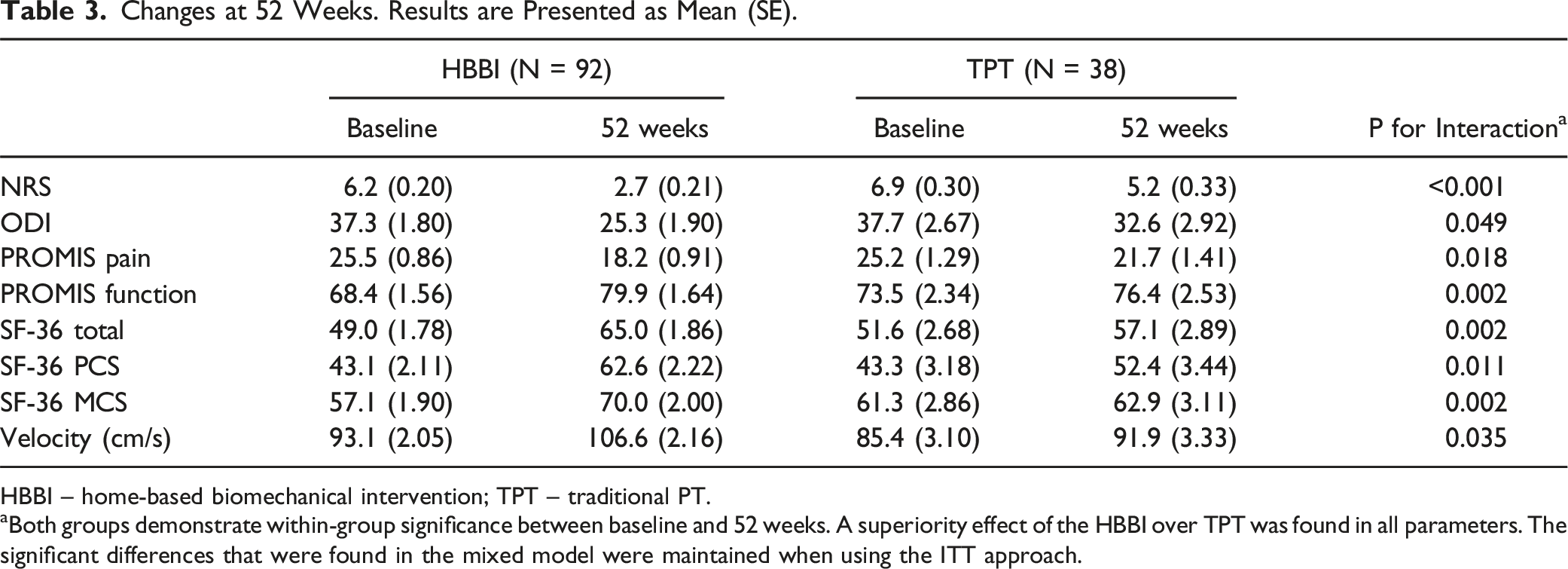
HBBI – home-based biomechanical intervention; TPT – traditional PT.
aBoth groups demonstrate within-group significance between baseline and 52 weeks. A superiority effect of the HBBI over TPT was found in all parameters. The significant differences that were found in the mixed model were maintained when using the ITT approach.
Mixed Model Analysis for Changes Over Time Across all Study Visits. Results are Presented as Mean (SE) [95% CI].
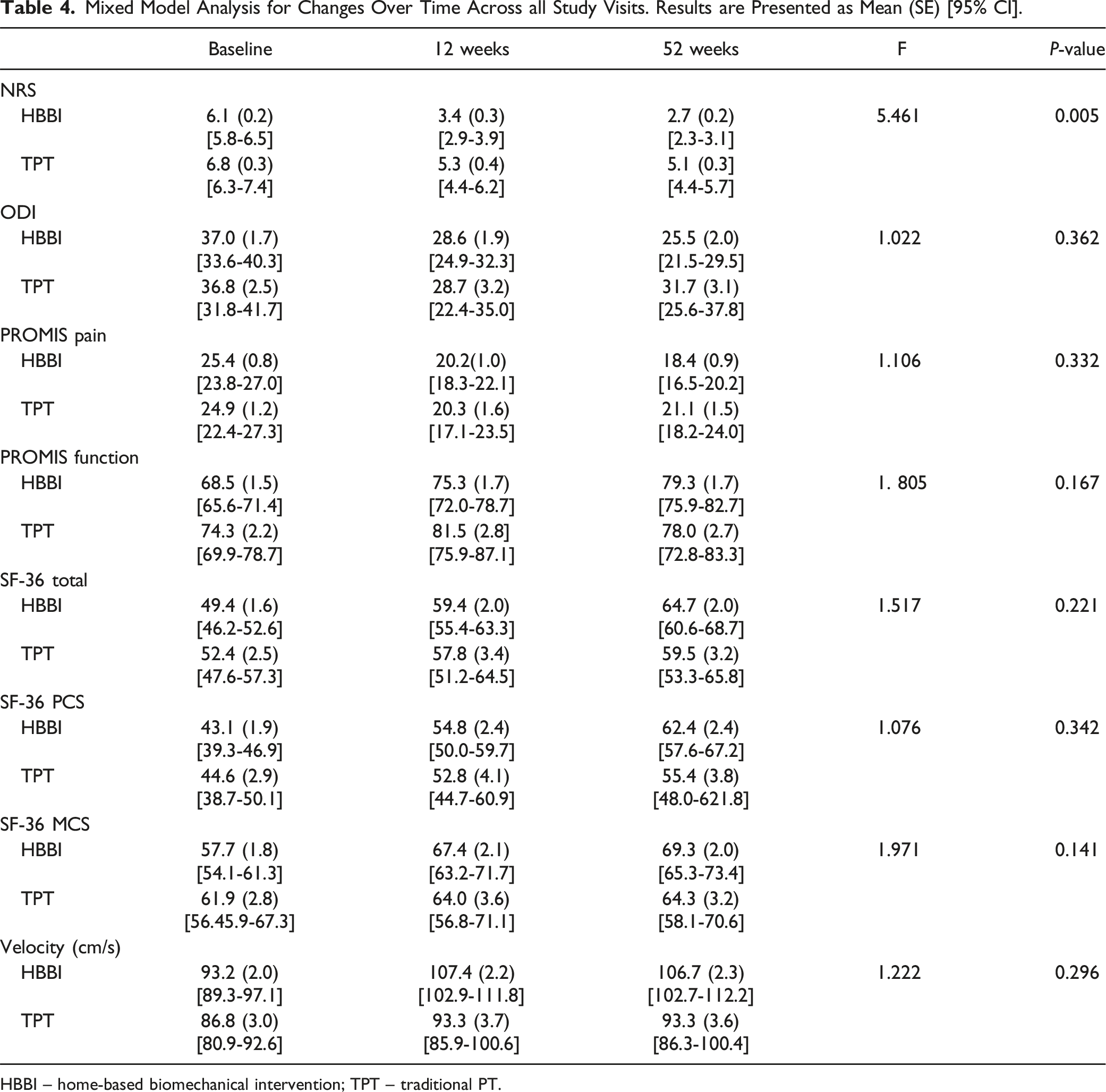
HBBI – home-based biomechanical intervention; TPT – traditional PT.
Discussion
Our primary finding in this study is that HBBI had a greater beneficial effect on pain in individuals with non-specific low back pain at the 12 and 52 week time points. Of note, both groups had improvement at 12 weeks, consistent with the known benefit of TPT and showing that HBBI also had similar effects in early improvement of symptoms. The persistent improvement in HBBI over traditional PT was likely due in part to the continued use of the device over the duration of the study and may reflect the easier sustainment of treatment benefits with HBBI over the commonly seen loss of adherence to exercise in TPT. Since the study population is complex and characterized by chronic back pain, without ongoing care and compliance with a treatment plan, it is reasonable to expect that most patients will experience a decline in symptoms. The benefits that were achieved during the first 3 months of TPT were a result of improved mobility, strength, flexibility, pain management techniques, home-based exercise, education, lifestyle modifications and perhaps even some placebo effect. However, those fade with improper follow-up and maintenance, which are a result of system constraints and patient motivation. McLeod et al suggest that the two main factors influencing adherence to a home-based exercise program are the need for practitioners to develop effective communication skills that focus on the individual’s needs, including personalization of exercise programs aimed at an individual’s symptoms, lifestyle and treatment goals. The second factor recommends follow-up feedback and support strategies via electronic (virtual) or face-to-face in-clinic and home visits to increase compliance. 17 Those factors are not part of the standard of care and might be the reason for the lack of compliance and deterioration in symptoms. Interestingly, the investigational device addresses those two recommendations and might be a reason for the sustainment of results. More research on non-surgical interventions that utilize the above recommendations and patient adherence is warranted. Additionally, the patients in the study had improvement in function in both groups but had greater improvement in the HBBI group for many of the parameters. Interestingly, patients in both groups had a significant increase in gait velocity, which reflects an objective improvement in mobility. There is a negative correlation between the level of pain and gait velocity, i.e., patients with higher pain intensity demonstrate slower gait, which might reflect a pain minimization strategy. 18 The reduction in pain might explain the improvement in gait velocity. In addition, we speculate that a higher increase in gait velocity, bringing them to a normal walking speed, in the HBBI is a result of neuromuscular improvement. However, there was no superiority effect. The significant improvement in mobility may also contribute to improved adherence and easier ability to comply with the home exercise program in order to sustain benefits.
Patients with CLBP are in pain and frequently have some degree of functional disability. Unfortunately, in many cases, patients rely on pain relief medications, primarily opioids, to relieve pain and improve function and quality of life. The overuse and prescription of opioids are thought to be in part responsible for the constant increase in opioid and drug overdose mortality in the US over the last 2 decades, which is now a national public health crisis. 19 Because current evidence does not support surgical interventions as the first line of treatment for most cases of CLBP, non-surgical interventions are still the first line of treatment. 20 However, non-surgical interventions only have a moderate clinical effect at best 8 and there is a need for new non-surgical interventions that will help this population. Recent evidence suggests that physical manipulations is the preferred non-surgical intervention among patients with CLBP, followed by supervised body-mind physical activities and self-management courses. Supervised sports and physical activities had a positive but slight effect. Corticosteroid injections and psychotherapy were generally rejected by patients. Given the preference heterogeneity, it is important to facilitate a decision between the patient and the physicians. 21 The results of our randomized controlled trial of a home-based biomechanical intervention show that using a foot-worn device to alter leg and back biomechanics while providing neuromuscular training may provide another non-invasive alternative for managing low back pain. 22 One of the advantages of this intervention is that it is a home-based program with a personally calibrated device that is specifically adapted to a patient based on their clinical symptoms and gait patterns. With no adverse effects, all of our patients were able to safely use the device in daily activities such as walking indoors and outdoors and climbing stairs. Another advantage is the ability to re-calibrate the device to increase or reduce perturbation and change neuromuscular training intensity as the patient has improved. We have observed that this intervention has a high level of engagement and compliance because it is a home-based program that integrates with the patient’s routine and produces an immediate clinical effect. In addition, a recent meta-analysis of RCTs by Sitthipornvorakul et al. suggests that walking, which is easy to perform and highly accessible, is recommended to manage chronic LBP to reduce pain and disability. 23
Another benefit of this HBBI is its mechanism of action. It is known that patients with CLBP present altered lumbar muscle activation patterns, including co-contraction of agonistic and antagonistic muscles compared to healthy controls. 24 These variations could be due to the compensation mechanism required to stabilize the spine. Since the device changes muscle activation patterns and allows a more synchronized activity that reduces the bracing mechanism at the lumbar spine,25-27 the use of the device may be beneficial in direct pain relief and also in motor re-education to normalize the altered gait patterns seen in back pain. More specifically, the translation of COP along the mediolateral axis significantly impacts the sagittal and frontal-plane pelvis and spine kinematics, external oblique activity, and frontal and transverse-plane lumbar moments. 27 The current study did not evaluate changes in muscle activation patterns, and we can not determine and quantify the neuromuscular changes in the lower back. We recommend that future studies will focus on biomechanical aspects of the lower back before and after the HBBI to better understand the reasons for the clinical effect.
Some limitations should be acknowledged. The study started in 2017 and enrolled patients until February 2022. During the time of the study, the COVID-19 pandemic compromised the continuance of clinical studies in New York City from March 2020 to January 2021. During the height of the pandemic in New York, it was impossible to maintain the study visits as planned as the program was not essential care and therefore many study visits were missed. This, however, affected both groups equally. Moreover, a higher (but statistically non-significant) dropout rate was observed in the TPT group which can potentially be attributed to several factors including limited clinical efficacy and the duration of the study. Despite maximum efforts invested to promote adherence to the research protocol, the 12-month follow-up period represents a significant commitment for patients, and may reflect the known loss of adherence to exercise of up to 70% seen in traditional physical therapy.28-30 The challenges inherent in long-term treatment adherence, including potential side effects, competing life responsibilities, and perceived lack of immediate benefits, could contribute to the observed dropout rate. This highlights a need for tailored strategies to enhance retention in long-term treatment programs, especially for interventions perceived to have limited clinical impact. We recommend that future studies will account for this to improve future adherence rates and ensure the validity of the results. Secondly, there were differences between groups in baseline characteristics, including age and BMI. Although all statistical tests accounted for and controlled for those differences, there is still a possible bias. Previous studies suggest conflicting outcomes regarding the effect of age on clinical response in patients with LBP.31-34 Thirdly, CLBP has various underlying causes. Some of the most common etiologies include pain which results from soft tissue damage (muscle and ligament strain), degenerative disc disease, frequently a result of aging, herniated or bulging disc, frequently a result of acute injury, spinal stenosis, facet joint dysfunction, sacroiliac joint dysfunction, spondylolisthesis, and osteoarthritis. This study did not account for etiology but rather focused on symptoms, and therefore, a sub-group analysis to assess the clinical response in different etiologies was not performed. Additionally, there was not sufficient number of patients to adequately do a sub analysis based on etiology. Future studies should explore the correlation between the clinical effect and the underlying pathology. Fourth, since this study was long-term, patients were allowed to continue with other non-surgical interventions to treat their symptoms. We believe that the bias, if it exists, will be equally distributed across both groups, however we cannot rule out a potential bias. Lastly, this trial focuses on clinical outcomes only and does not include economic aspects. Future studies should examine the cost-effectiveness of this HBBI compared to traditional care.
Some considerations should be dedicated to the selected methodology. The design of the trial relies on a pragmatic methodology principle. That is, the control group (traditional PT) performs the prescribed program as if it was done in commercial settings. The rationale for a pragmatic design is to be able to measure real-life behavior when using traditional PT as an intervention for patients with CLBP to test the true effect of the device compared to TPT. The 3 months timeframe was selected to reflect the average time of a TPT program. Of note, during those 3 months, the intervention group had fewer touchpoints with the physical therapist. Thereafter, patients in both groups were advised to continue with self-managing, while patients in the intervention group had 2 follow-up appointments with the physical therapist to re-calibrate the device as needed. All in all, patients in both groups had roughly the same number of touchpoints with the physical therapist.
Conclusion
The findings of this study indicate that HBBI has a significant clinical effect in terms of pain reduction and functional improvement. The outcomes associated with HBBI could be attributed to the device’s unique design and ease of usage, which may facilitate better adherence and maintenance of therapeutic effects relative to TPT. Given the persistent challenges surrounding the management of chronic low back pain, including the reliance on pharmacological interventions, the integration of innovative non-surgical approaches such as HBBI presents a potentially valuable addition to existing treatment options.
Supplemental Material
Supplemental Material - Clinical Outcomes of a New Foot-Worn Non-invasive Biomechanical Intervention Compared to Traditional Physical Therapy in Patients With Chronic Low Back Pain. A Randomized Clinical Trial
Supplemental Material for Clinical Outcomes of a New Foot-Worn Non-invasive Biomechanical Intervention Compared to Traditional Physical Therapy in Patients With Chronic Low Back Pain. A Randomized Clinical Trial by Ratnakar Veeramachaneni, Andrew Gitkind, Sandeep Yerra, Michael Hagan, Asude N. Hasanoglu, Natnael Akile, Hannah Kareff, Derek Ho, and Matthew N. Bartels in Global Spine Journal.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by AposHealth provided devices at no cost for the conduct of the trial.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
