Abstract
Study Design
Retrospective cohort study.
Objectives
The objective of this study was to characterize the association between cell-salvage and allogeneic transfusion rate in pediatric patients undergoing posterior arthrodesis for scoliosis.
Methods
NSQIP Pediatric database years 2016-2022 was used. Patients under the age of 18 who received posterior arthrodesis with 7 or more surgical levels for spinal deformity correction were included. Rates of cell-salvage and allogeneic transfusion were determined. We assessed the impact of cell-salvage on the rate of allogeneic transfusion using chi-square test and multivariable logistic regression.
Results
There were 34,241 patients in this study. The rate of allogeneic transfusion was 21.6% (n = 7407). The allogeneic transfusion rates for idiopathic, neuromuscular, and congenital/syndromic scoliosis were 12.3%, 50.8%, and 25.9%, respectively. Cell-salvage was used in 71.1% of patients (n = 24,344). In the multivariable regression analysis, longer operative time (P < .001), non-idiopathic scoliosis (P < .001), hematocrit less than 35 (P < .001), and ≥13 surgical levels (P < .001) were associated with higher odds of allogeneic transfusion. Use of cell-salvage (P < .001), increasing age (P < .001), and increasing patient weight (P < .001) were associated with significantly lower odds of allogeneic transfusion. In a subanalysis, use of cell-salvage was associated with reduced rate of allogeneic transfusion in patients with idiopathic scoliosis. Cell-salvage was not associated with reduced rates of allogeneic transfusion in neuromuscular and congenital/syndromic scoliosis.
Conclusion
This is the largest study investigating the impact of cell-salvage on rate of allogeneic transfusion in pediatric spinal deformity surgery. Use of cell-salvage is associated with reduced allogeneic transfusion rates in idiopathic scoliosis surgery.
Introduction
Posterior arthrodesis for scoliosis in pediatric patients is associated with major blood loss from extensive soft tissue dissection and instrumentation of the vertebrae, often necessitating allogeneic blood transfusion.1-3 Idiopathic scoliosis is the most common type of scoliosis, accounting for up to 85% of cases. 4 Neuromuscular scoliosis arises from underlying neuromuscular conditions, such as cerebral palsy, muscular dystrophy, and spinal muscular atrophy. 5 Congenital scoliosis is caused by congenital vertebral anomalies, such as hemivertebra, block vertebra, and unsegmented unilateral bar. 6 A pooled meta-analysis estimated average blood loss of 500 to 1500cc for adolescent idiopathic scoliosis and 2000 to 3500cc for neuromuscular scoliosis. 7 Several studies have reported an association between allogeneic blood transfusion and higher infection rates, poor postoperative functional recovery, and reduced long-term survival.8-10 Consequently, techniques and strategies have evolved to improve blood management to minimize the need for allogeneic blood transfusion.11,12
Intraoperative cell-salvage involves the collection and processing of blood from the operative field for reinfusion. 13 Similarly, postoperative cell-salvage collects blood from postsurgical drains for reinfusion. 12 While use of cell-salvage may reduce the need for allogeneic blood transfusion, results have been conflicting.14-19 The primary aim of this study was to investigate whether the use of cell-salvage in pediatric spinal deformity surgery impacts the rate of allogeneic blood transfusion. The secondary aim was to identify risk factors associated with allogeneic blood transfusion.
Methodology
Source of Data
The American College of Surgeons National Surgical Quality Improvement Program (NSQIP) Pediatric database years 2016 to 2022 was used for this retrospective cohort study. The dataset was harmonized with the procedure targeted spinal fusion dataset. NSQIP is a validated, risk-adjusted, outcomes-based program that measures the quality of surgical care internationally. In each participating hospital, a trained Surgical Clinical Reviewer collects preoperative through 30-day post-operative data on randomly selected patients. Therefore, the database does not record consecutive cases. Data are entered online in a secure, web-based platform available for open access by request.
Participants
Patients under the age of 18 years who received posterior arthrodesis for spinal deformity correction were included in this study. Current Procedural Technology (CPT) codes (CPT 22802 and 22804) and ICD-10 codes (M40, M41, M42, M96, Q67, and Q76) were used to identify the study population. Patients were excluded if they had anterior only approaches for deformity correction or fewer than 7 surgical levels. The NSQIP Pediatric database collection excludes: (1) patients 18 years of age or older, (2) cases involving hyperthermic intraperitoneal chemotherapy, (3) ASA score of 6, (4) concurrent case by different surgical team under the same anesthetic, (5) multiple cases within 30 days, (6) transplant cases, and (7) trauma and abuse cases.
Outcomes of Interest and Statistical Analysis
The primary outcome of this study was receipt of allogeneic blood transfusion within 30-days of surgery. Patient demographics, surgical metrics, volume of allogeneic blood transfusion, and volume of cell-salvage transfusion were characterized using descriptive statistics. Results are presented as means ± standard deviation. The rates of cell-salvage use and allogeneic blood transfusions were reported as proportions. A multivariable logistic regression analysis was performed to identify risk factors associated with allogeneic blood transfusion. Independent variables were chosen a priori based on literature review and clinical relevance. The variables included for adjustment were age in years, weight in pounds, preoperative hematocrit less than 35, type of scoliosis (idiopathic, neuromuscular, and congenital/syndromic), operative time in hours, number of surgical levels (7-12, ≥13 levels), addition of pelvic fixation, use of intraoperative antifibrinolytics, and use of cell-salvage. A secondary analysis was performed in stratified subgroups by type of scoliosis and number of surgical levels. If cell-salvage transfusion is associated with allogeneic transfusion in the regression analysis, a post-hoc chi-square test was performed to compare the rates of postoperative allogeneic transfusion between patients that did and did not receive cell-salvage transfusion.
The alpha value for significance was 0.05 a priori. Listwise deletion was used for missing data for complete case analysis. Statistical analysis was performed using IBM SPSS Statistics version 29.0.2.0.
Results
Participants and Surgical Metrics
Summary of Patient Characteristics and Surgical Factors.
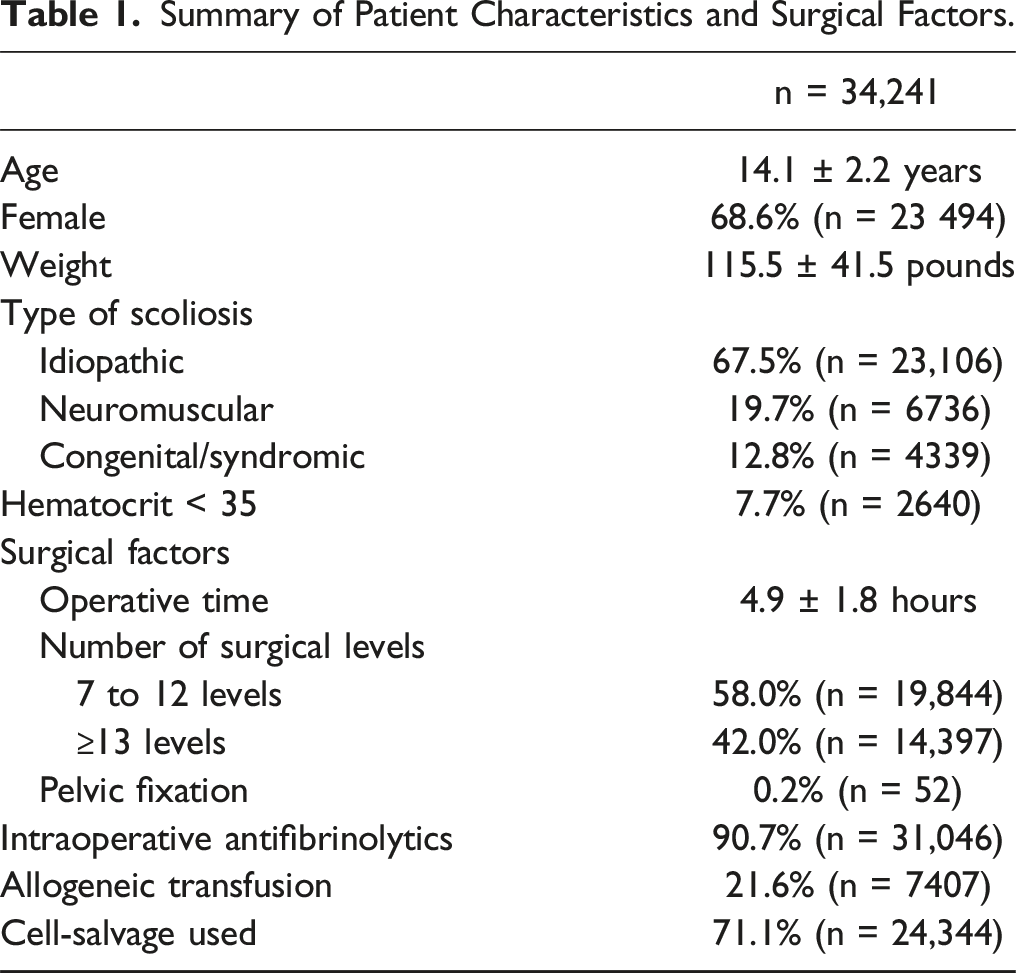
Summary of Rate of Transfusion and Transfusion Volumes for Idiopathic, Neuromuscular, and Congenital/Syndromic Scoliosis.
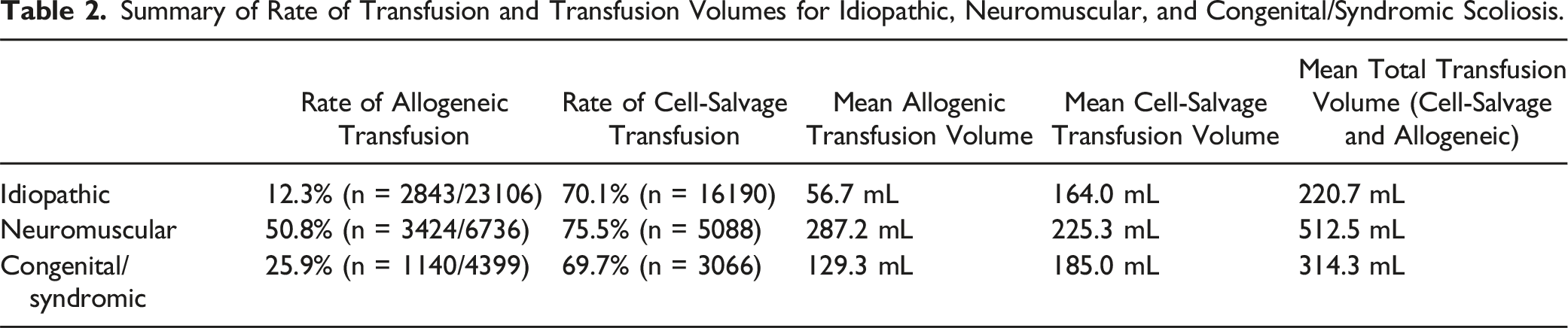
Risk Factors for Allogeneic Blood Transfusion and Impact of Cell-Salvage for Entire Cohort
Multivariable Logistic Regression Analysis for Allogeneic Transfusion for Pediatric Spinal Deformity Surgery.
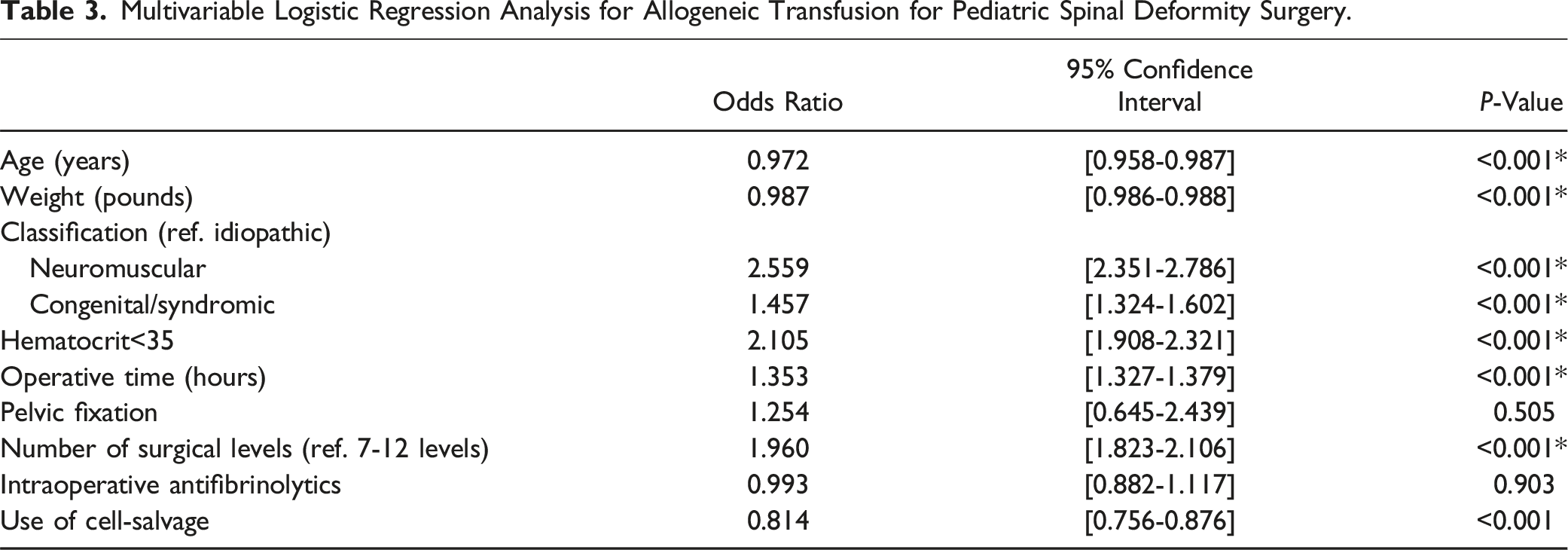
Secondary Analyses
Idiopathic Scoliosis
Multivariable Logistic Regression Analysis for Allogeneic Transfusion for Idiopathic Scoliosis Surgery Stratified by Number of Surgical Levels.
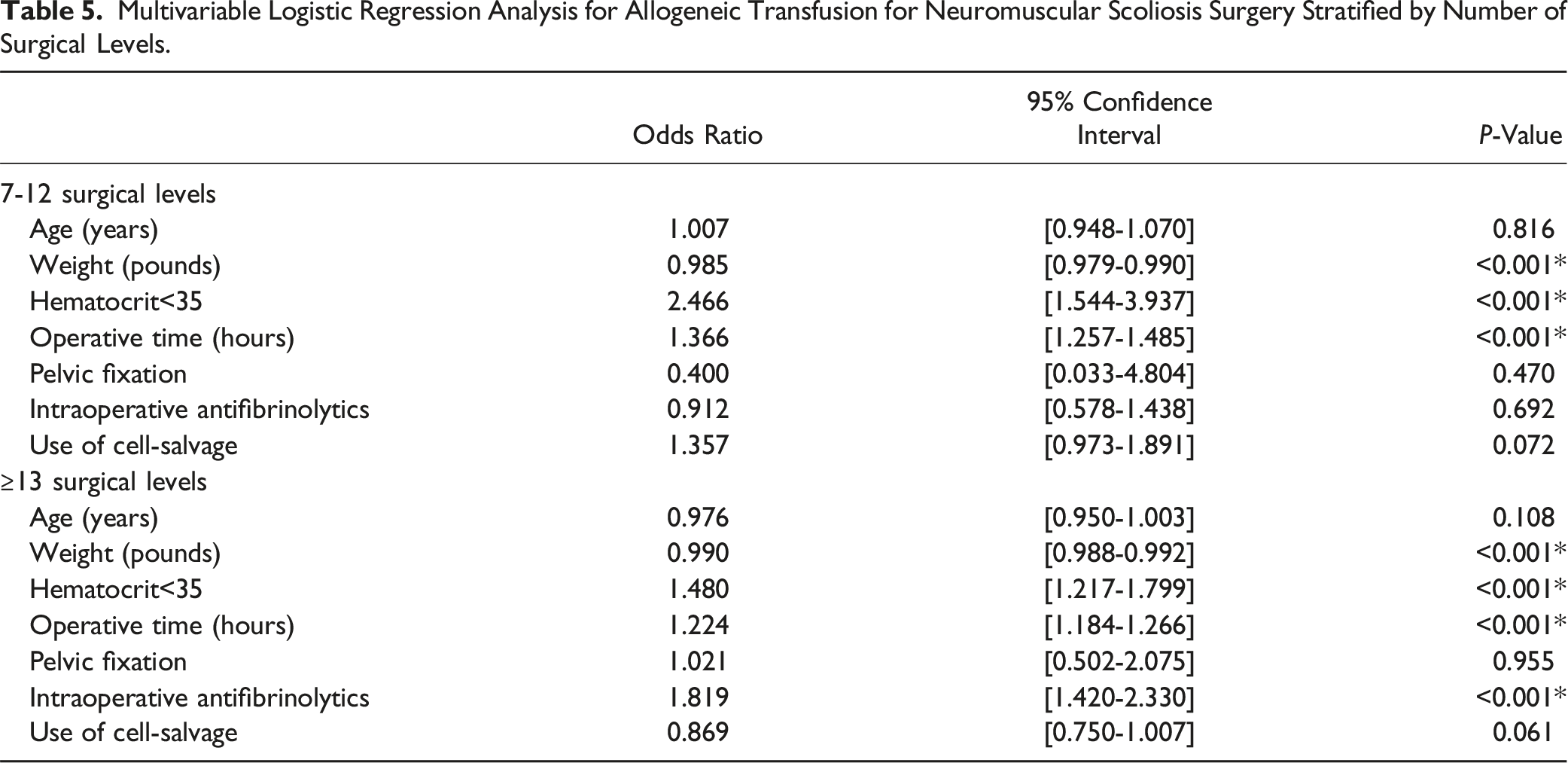
Neuromuscular Scoliosis
Multivariable Logistic Regression Analysis for Allogeneic Transfusion for Neuromuscular Scoliosis Surgery Stratified by Number of Surgical Levels.
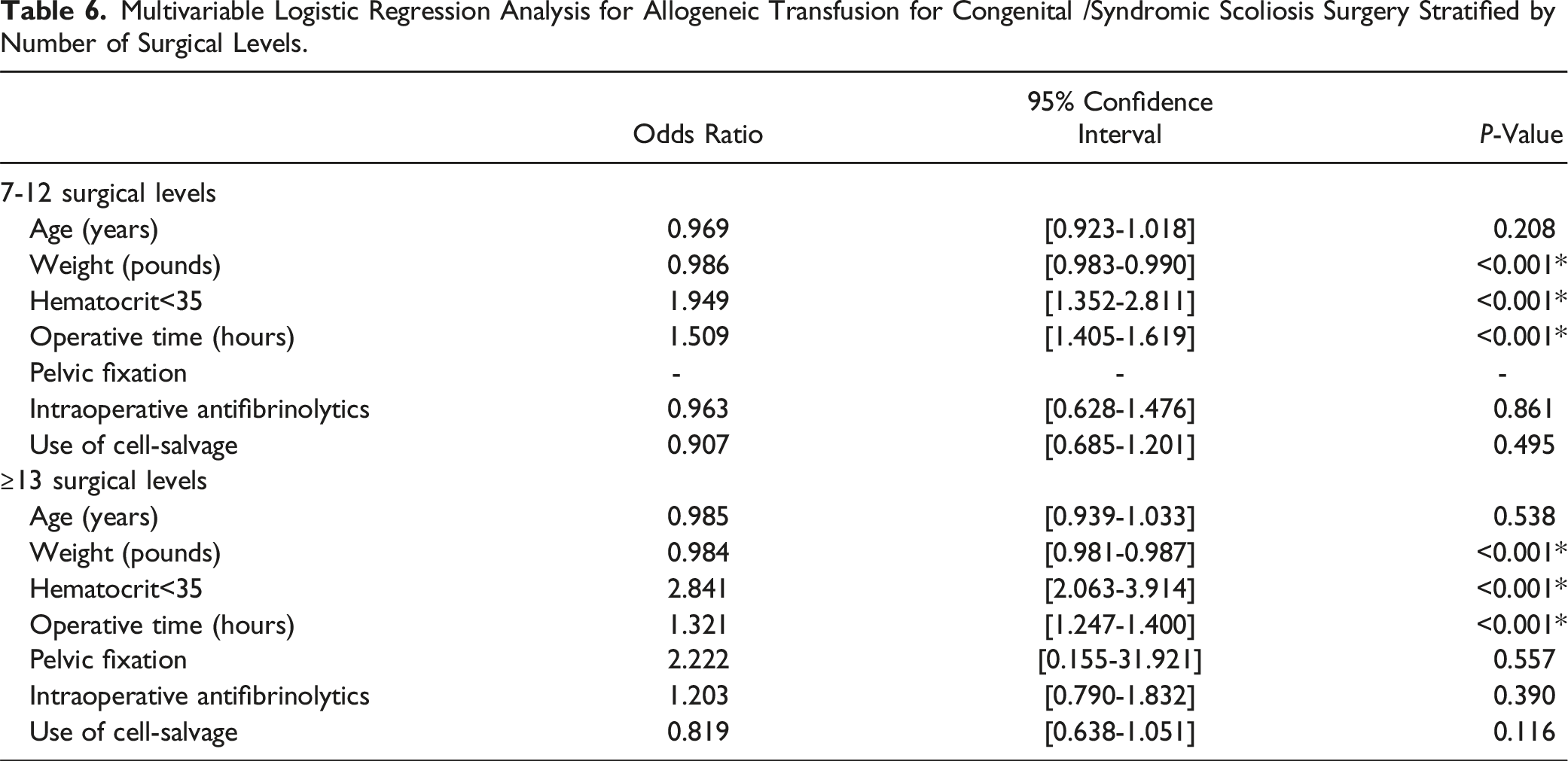
Congenital /Syndromic Scoliosis
Multivariable Logistic Regression Analysis for Allogeneic Transfusion for Congenital /Syndromic Scoliosis Surgery Stratified by Number of Surgical Levels.
Discussion
Posterior arthrodesis for pediatric spinal deformity correction is typically associated with significant blood loss, often requiring allogeneic blood transfusion. 20 Techniques and strategies have evolved over the past few decades to optimize blood management to minimize the use of allogeneic transfusion. 11 Preoperative strategies include autologous blood donation and red cell augmentation with erythropoietin. 12 Intraoperatively, optimal positioning, hemodilution, hypotensive anesthesia, antifibrinolytics, topical thrombotic agents, and cell salvage are utilized for blood management. 12 However, it remains unclear how effective these strategies are at reducing the rate of allogeneic blood transfusion. The focus of this study was to investigate the impact of cell salvage on the rate of peri-operative allogeneic blood transfusion.
In our study of 34,241 patients, 21.6% received allogeneic blood transfusion. The rate of transfusion in our study was comparable to rates previously reported.3,11,20 Similar to previous studies investigating risk factors associated with allogeneic blood transfusion,7,11,20 we found non-idiopathic scoliosis to be associated with a higher rate of allogeneic transfusion, most notably with neuromuscular scoliosis (Idiopathic: 12.3%; Neuromuscular: 50.8%; Congenital/syndromic: 25.9%). Potential reasons for this finding include higher degree of spine curvature, more surgical levels, use of osteotomies for correction, bone quality and longer operative time. However, in our study we found non-idiopathic scoliosis was independently associated with a higher risk of allogeneic blood transfusion when controlling for operative time and number of surgical levels, which were both independently associated with a higher risk of transfusion. Studies have suggested other potential reasons for increased blood loss in neuromuscular scoliosis including osteopenic bone, decreased coagulation factor reserve, altered vascular smooth muscle, and increased fibrinolytic activity.21-25 These potential underlying mechanisms were beyond the scope of this paper; however, future research to understand the underlying mechanisms for increased blood loss in neuromuscular scoliosis is required to allow for targeted management strategies.
Higher weight in pounds and older age in years were associated with a lower risk of allogeneic transfusion. A study by Rodriguez et al. investigating compensatory reserve index in pediatric patients using a computational algorithm found weight and age to be significant predictive factors for hemodynamic vascular adaptation during acute blood loss. 26 They found higher weight to be associated with improved compensatory reserve index, likely due to larger blood volume associated with larger patient size.26,27 Improved compensatory reserve index with increasing age, independent of blood volume and weight, may be due to improved physiological adaptation mechanisms such as vascular elastic properties, adrenergic response, and pleural/pulmonary pressures. 26 We found preoperative hematocrit less than 35 was associated with a higher risk of allogeneic blood transfusion. Similarly, a study by Nuttall et al. identified preoperative hemoglobin concentration as a predictor for allogeneic transfusion. 28 Age, weight, and hematocrit are important factors to consider for risk stratification and implementation of additional blood management strategies. Use of erythropoietin and iron supplementation could be considered for preoperative optimization or autologous preoperative blood donation in preparation for surgery in high risk patients.11,12
Use of intraoperative antifibrinolytics has been widely implemented across various surgical specialities. 29 Tranexamic acid and ε-aminocaproic acid are common antifibrinolytic agents used. Multiple studies, systematic reviews, and meta-analyses have reported reduced perioperative blood loss, transfusion rate, and transfusion requirements with use of intraoperative antifibrinolytics in pediatric scoliosis surgery.29-36 Use of intraoperative antifibrinolytics was not a significant variable in our multivariable logistic regression analysis, except in neuromuscular scoliosis patients with ≥13 surgical levels, in which it was associated with an increased odds of transfusion. The variable in the dataset was collected as a binary variable of whether or not a patient received intraoperative antifibrinolytics. Therefore, it is difficult to draw conclusions based on this dichotomized variable. A study by Farrokhi et al. investigating the impact of a low dose tranexamic acid protocol reported no difference in intraoperative blood loss or blood transfused compared to control. 37 In contrast, several studies investigating the more commonly used high dose protocol of 100 mg/kg at the start of surgery followed by 10 mg/kg/hr until skin closure have reported reduced blood loss and transfusion requirements.33,38,39 To properly study the impact of intraoperative antifibrinolytics, more granular data than this database provides would be required, such as agent, dose, and protocol.
Cell-salvage involves the collection of blood from the operative field for transfusion after appropriate processing to remove cellular, biochemical, and noncellular debris.12,15 Several studies have reported red blood cell return as high as 60% of red cell loss during surgery.12,40,41 However, it is unclear when cell-salvage should be utilized since results from various studies have been conflicting.14-19 Additionally, it can be costly and requires technical personnel to operate.15,42,43 Therefore, the purpose of our study was to study the utility of cell-salvage in pediatric spine deformity surgery using a large database. Our multivariable logistic regression analysis for the entire study cohort found use of cell-salvage was associated with reduced odds of allogeneic transfusion (OR = 0.814, P < .001). We performed a subanalysis stratified by etiology and number of surgical levels to investigate when cell-salvage may be useful. In our stratified subanalysis, we found use of cell-salvage was associated with reduced odds of allogeneic transfusion in idiopathic scoliosis regardless of number of surgical levels fused. However, use of cell-salvage was not associated with the allogeneic transfusion rate in congenital/syndromic scoliosis or neuromuscular scoliosis. Non-idiopathic scoliosis, particularly neuromuscular scoliosis, has high intraoperative blood loss and is associated with high rates of allogeneic transfusion (Neuromuscular: 50.8%, Congenital/syndromic: 25.9%) and larger transfusion volumes (Neuromuscular: 512.5 mL, Congenital/syndromic: 314.3 mL). The high transfusion requirements for non-idiopathic scoliosis may mean cell-salvage alone cannot prevent the need for allogeneic transfusion. In comparison, idiopathic scoliosis is associated with relatively low blood loss. Our study found a mean total transfusion volume of 220.7 mL. Use of cell-salvage in surgeries with low transfusion requirements, such as idiopathic scoliosis may be adequate to reduce the risk of allogeneic transfusion. Liberal allogeneic transfusion is associated with increased morbidity and cost.1,44-47 Improved screening for transfusion-transmissible infections and hospital risk reduction protocols have significantly reduced the risk of communicable disease transmission and ABO mismatch (Vitale et al, 2002) (3). However, allogeneic transfusion has been increasingly associated with negative outcomes such as higher rates of infectious complications, non-infectious adverse events, immunological reactions, and poor functions recovery.1,3,20,45-47 Therefore, use of cell-salvage in idiopathic scoliosis should be considered to reduce the use of allogeneic transfusion and its associated morbidities.
This is the largest study investigating the impact of cell-salvage on rate of allogeneic transfusion in pediatric deformity surgery. We found non-idiopathic scoliosis to have a higher rate of transfusion and require higher transfusion volumes than idiopathic scoliosis. Use of cell-salvage in idiopathic scoliosis was associated with reduced likelihood of allogenic transfusion and should be considered to avoid adverse events associated with allogeneic transfusion. Additional blood management strategies such as preoperative erythropoietin, iron supplementation, and donation should be considered in patients with risk factors for requiring allogeneic transfusion, such as low weight, younger age, and hematocrit <35. This study has limitations. This is a retrospective study using administrative data, which may be susceptible to unmeasured confounding secondary to data elements that were not captured such as pre-operative hemoglobin levels or pre-operative bone density. Additionally, there are likely between-center differences in transfusion rates and use of cell-salvage which we are unable to study or control for using this database. There are various surgical variables that can impact surgical blood loss that could not be studied using the data available, such as increased blood loss associated with osteotomies or resections of congenital vertebral anomalies. The technique used for facetectomies and osteotomies can also impact blood loss; ultrasonic bone cutting devices have been shown to be associated with reduced blood loss compared to other techniques. 48 Further, we were unable to quantify the association between occurrence of allogeneic transfusion and longer-term outcomes such as pain, delayed complications or readmissions. There are several strengths to this study. This study uses a national registry database with minimal missing data, allowing for more robust statistical power and generalizability. We included a large number of patients with varying scoliosis etiologies, which allowed us to perform a stratified analyses to provide more specific findings to aid in clinical decision making. Future directions include studying the implementation of preoperative optimization protocols in patients with risk factors for requiring allogeneic transfusion and studying the cost-effectiveness of using cell-salvage for idiopathic scoliosis surgery.
Footnotes
Author Contributions
V.K.C. contributed to conceptualization of the project, developing the methodology, analyzing the data, drafting the manuscript, and editing the manuscript. G.S. contributed to conceptualization of the project, developing the methodology, drafting of the manuscript, and editing the manuscript. A.K.M. contributed to developing the methodology and editing the manuscript. D.E.L. contributed to editing the manuscript. D.L.S. contributed to conceptualization of the project, developing the methodology, and editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: V.K.C., G.S., A.K.M., and D.E.L. declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. D.L.S. is a consultant for Highridge and Globus Medical and receives royalties from Medtronic, Highridge, Globus Medical, and Wolters Fluwer Health.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
