Abstract
Study Design
Human cadaver study.
Objectives
To provide a qualitative and quantitative evaluation by demonstrating measurements of the proximity of vital structures involved and assessed injuries during a T12-corpectomy and cage implantation via a far lateral approach.
Material and Methods
Six fresh-frozen adult cadaveric specimens were dissected according to standardized protocol. A formal left-sided far lateral T12-corpectomy was carried out by trained experienced spine fellows. Upon completion of the procedure, a cage was placed between T11 and L1. We then turned the patient supine and performed a formal celiotomy and sternotomy to allow for an open anterior central inspection of all structures concerned. Vital structures as in vessels, diaphragm, pleural membranes, neural elements, important foramina of the diaphragm (Bochdalek, Morgagni) and the thoracic duct were identified. Any injuries to these structures were recorded and proximity to key relevant structures to this exposure were measured.
Results
We were able to quantify the actual diaphragm excursions and describe its origins to the spine. There was no actual diaphragm injury in any of the cadavers and there were no injuries to the neurovascular structures. We found expected parietal but no visceral pleural injuries.
Conclusion
Our cadaver study identified the feasibility of performing a T12-corpectomy through a far lateral approach with no violation of the actual diaphragm and expected limited injuries to the parietal pleura only.
Keywords
Introduction
In recent years far lateral surgeries have gained an increasingly important role in spine surgery. Intra- and postoperative complications such as bleeding, wound healing issues and infection rates, as well as duration of hospital stay, and rehabilitation time may be improved by adopting these new procedural techniques.1–3 Even more extensive surgeries such as corpectomies with cage-implantation have become feasible with far lateral approaches. 4 Although these techniques have become increasingly utilized in routine clinical settings, there have been only few studies that have looked at potential risk to vital structures during these procedures.
The thoracolumbar junction is a common area of concern for fracture, oncologic disease, de novo spinal infection and complex deformity of the spine. At this fragile spinal transition zone there is a complex juxtaposition of the diaphragm, its attachments, and proximity of vascular and critical organ structures that can affect anterior/lateral surgical access.4,5 To date, there have been few studies that qualitatively assessed which structures are at risk and none have provided quantitative measurements of the proximity of vital organs.
The purpose of this study was to assess the spatial relations of vital organs, the diaphragm, pleural membranes, as well as injury potential to vital structures surrounding the thoracolumbar junction during a far lateral complete T12-corpectomy and cage reconstruction. The secondary goal of this cadaveric study was to specifically measure the proximity of key vascular structures during this type of surgery.
Finally, we hypothesized that injuries to the diaphragm and vital structures can be avoided during access and decompression with reconstruction surgery of the thoracolumbar junction.
Methods
Six fresh-frozen adult cadaveric specimens (torsos) were thawed per conventional tissue management protocol and then dissected by Neurological and Orthopedic Spine surgeons to simulate real life surgery. All specimens were verified to have been without prior thoracic, retroperitoneal, and abdominal surgeries. Dissections were performed at the Cadaveric Laboratory at the Bioskills Lab at the Seattle Science Foundation by the investigative team. Procedures were carried out by a senior spine fellow under supervision after having received focused training by an experienced attending surgeon.
We followed previously described protocols for a far lateral corpectomy. In brief, the cadaver was firmly positioned with the left side up on a radiolucent table with a pillow placed under the thoracolumbar junction. The position of the C-arm was then adjusted to obtain a orthogonal view until true anteroposterior and lateral images could be verified. Then, a targeted approach was carried out over the 11th rib with a 5 cm lateral rib dissection under preservation of the neurovascular bundle. The procedure was performed using dedicated retractors and other specialized instruments provided by a manufacturer (Globus Medical Inc.) of minimally invasive far lateral access technologies. After blunt dissection to the lateral vertebral body, the base of the left T12 rib was resected and upon identification of the foramen, full discectomies of T11/12 and T12/L1were carried out except for the anterior anulus. Then a T12 corpectomy was carried out using power drills and manual fragment removal, including the far side of the cortex. After completion of the corpectomy, an expandable titanium cage was placed until press fit to the rostral and caudal denuded endplates was achieved. Satisfactory cage placement using far lateral access instrumentation was confirmed in all cadavers via biplanar fluoroscopy.
Following cage implantation, the cadaver was placed in a supine position and a combined midline sternotomy and laparotomy was carried out to allow for full exposure of the anterior situs. We then explored and measured the distances to the vital structures of the thoracoabdominal junction including arterial and venous vessels, diaphragm, pleural membranes, neural elements, the main foramina of the diaphragm (Bochdalek, Morgagni) as well as the thoracic duct. Any injuries to these structures were recorded and proximity of the T12 surgical dissection to key relevant structures were measured. Representative illustrative pictures of the entire procedure were obtained and used for educational purposes (Figure Reference).
Results
Overview of the Demographic Characteristics of the Cadavers.
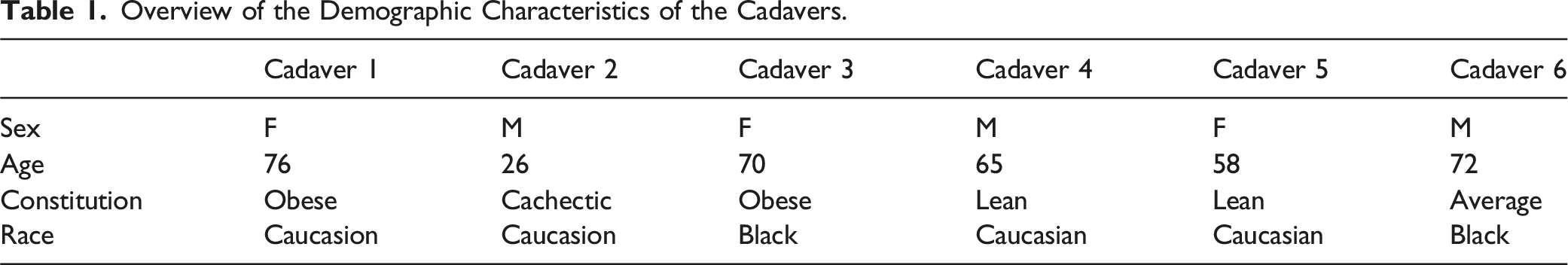
Anterior view of the thoracolumbar junction, pointing out the anatomical position and dimensions of the diaphragm and its close relationship to the most important neighboring vital structures.
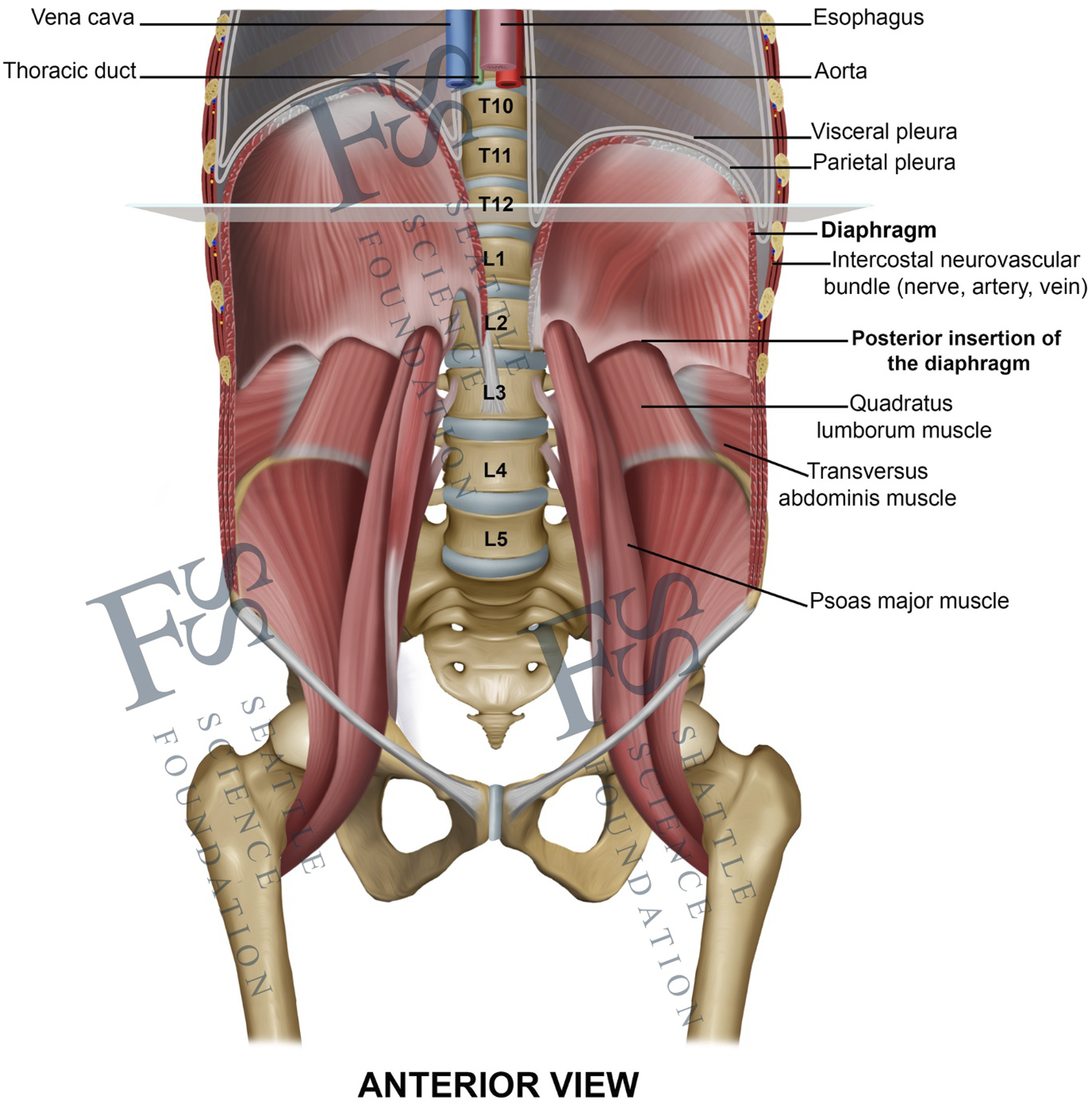
Lateral view of the thoracolumbar junction, pointing out the anatomical position and dimensions of the diaphragm and its close relationship to the most important neighboring vital structures.
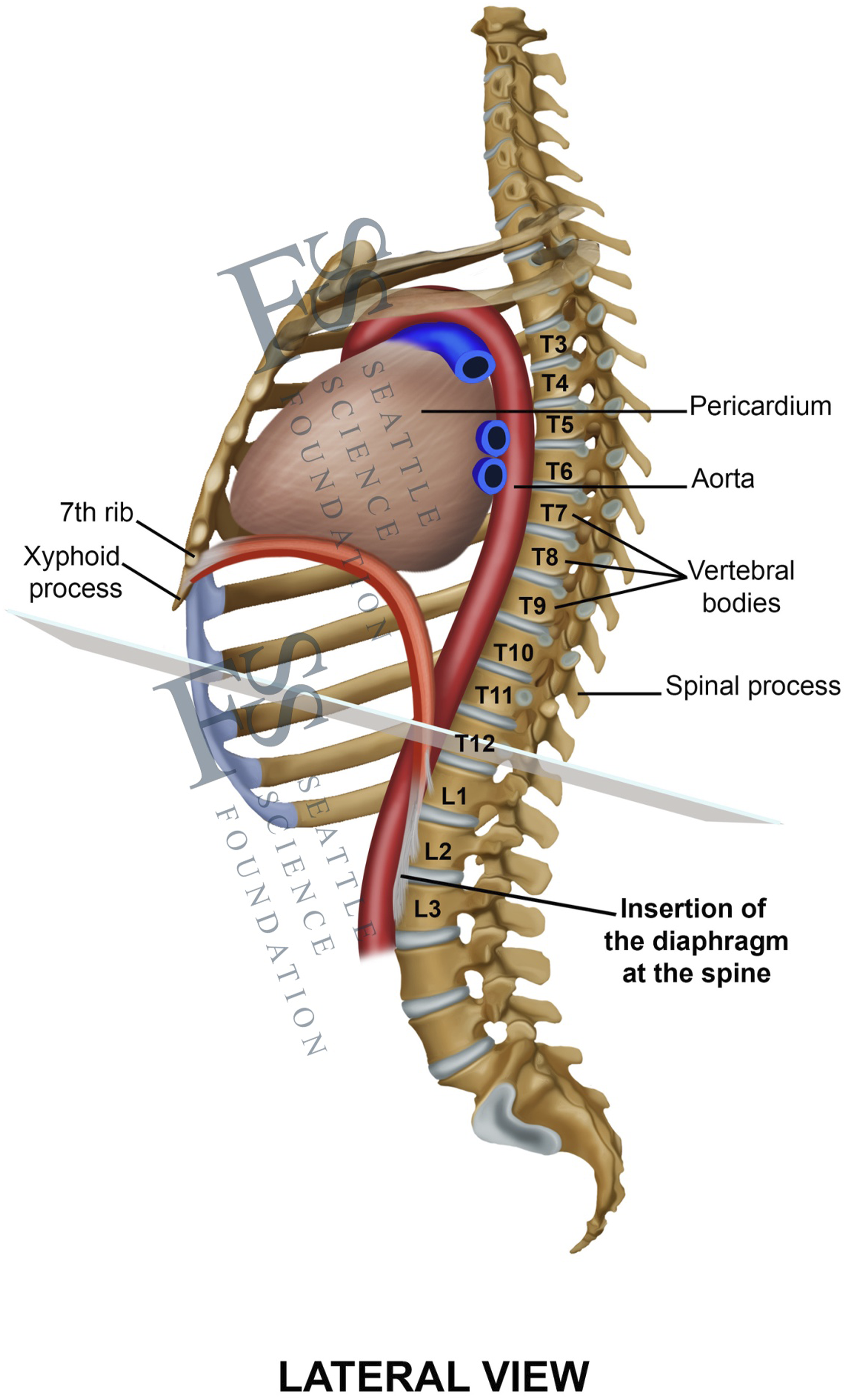
Cross-sectional view at the height of the Mid-T12-vertebral body, pointing out the anatomical position and dimensions of the diaphragm and its close relationship to the most important neighboring vital structures.
Therefore, there was no actual diaphragm injury in any of the cadavers.
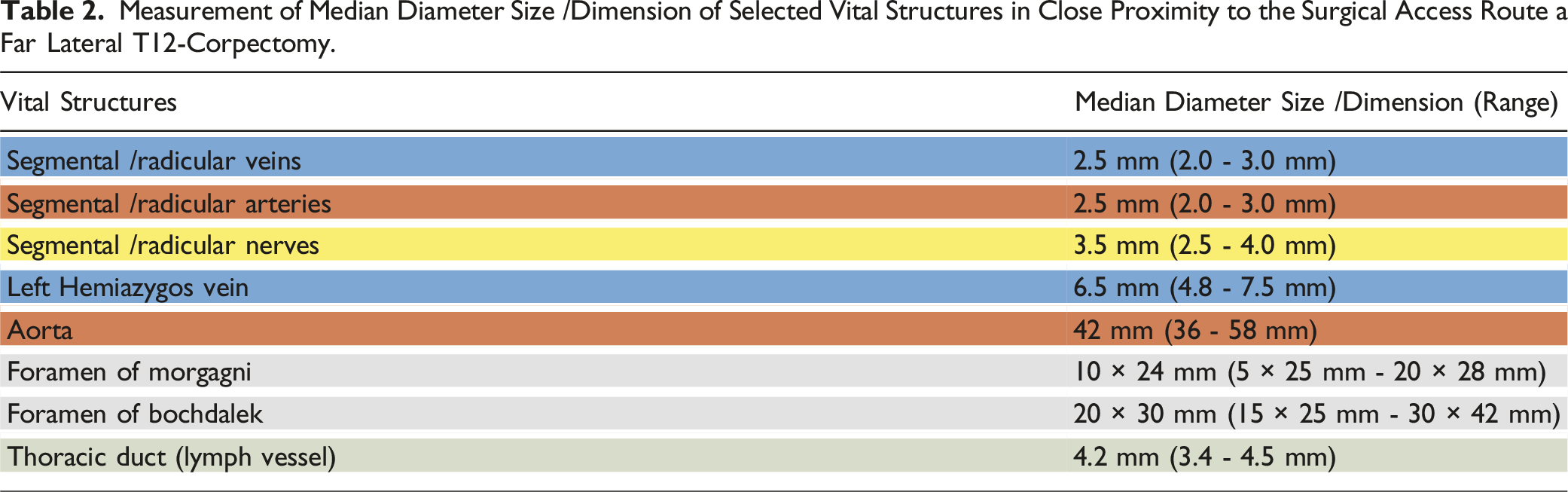
Measurement of Median Diameter Size /Dimension of Selected Vital Structures in Close Proximity to the Surgical Access Route a Far Lateral T12-Corpectomy.
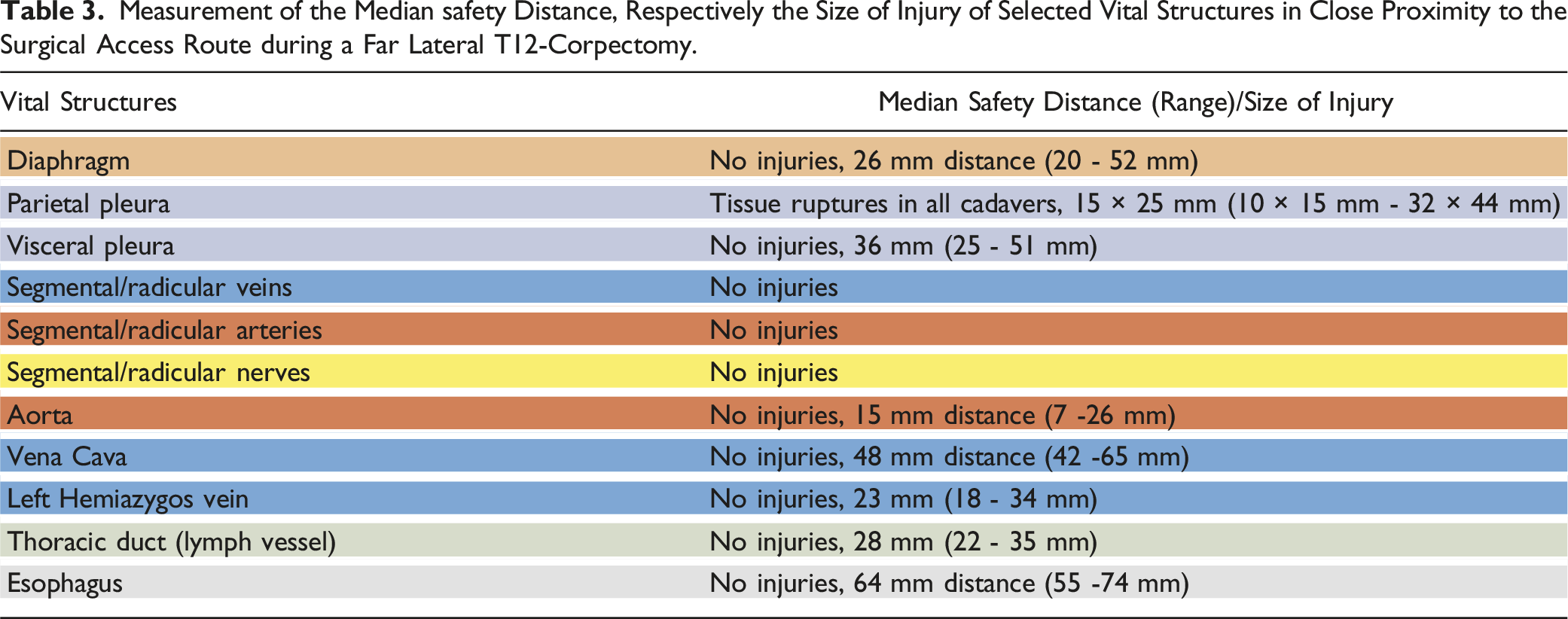
Measurement of the Median safety Distance, Respectively the Size of Injury of Selected Vital Structures in Close Proximity to the Surgical Access Route during a Far Lateral T12-Corpectomy.
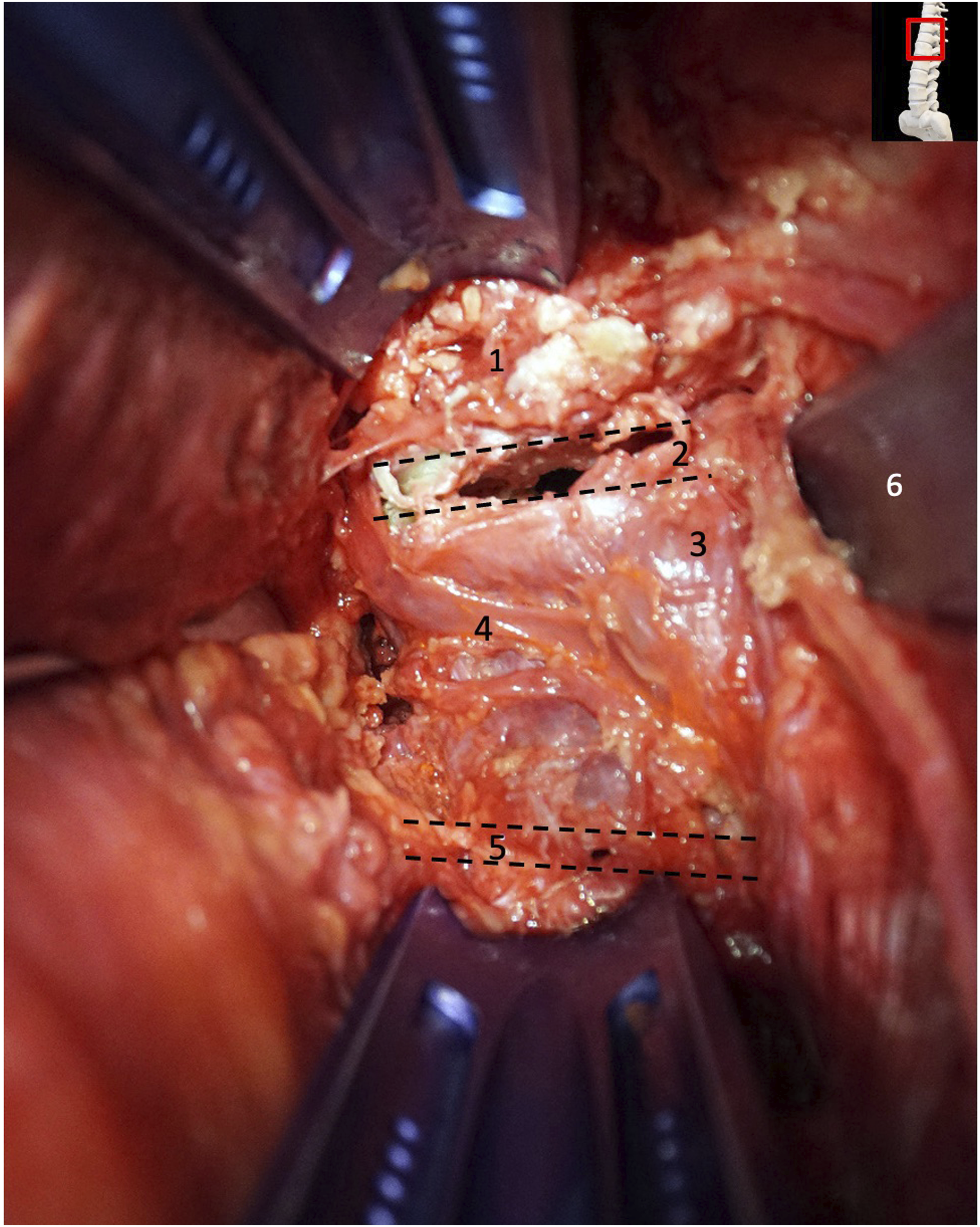
Exposure of T12 vertebrae prior to T12 corpectomy and cage implantation. 1: T11 vertebrae body; 2: disk space between T11 and T12; 3: T12 vertebral body; 4: segmental vein; 5: disc space between T12 and L1; 6: retractor blade.
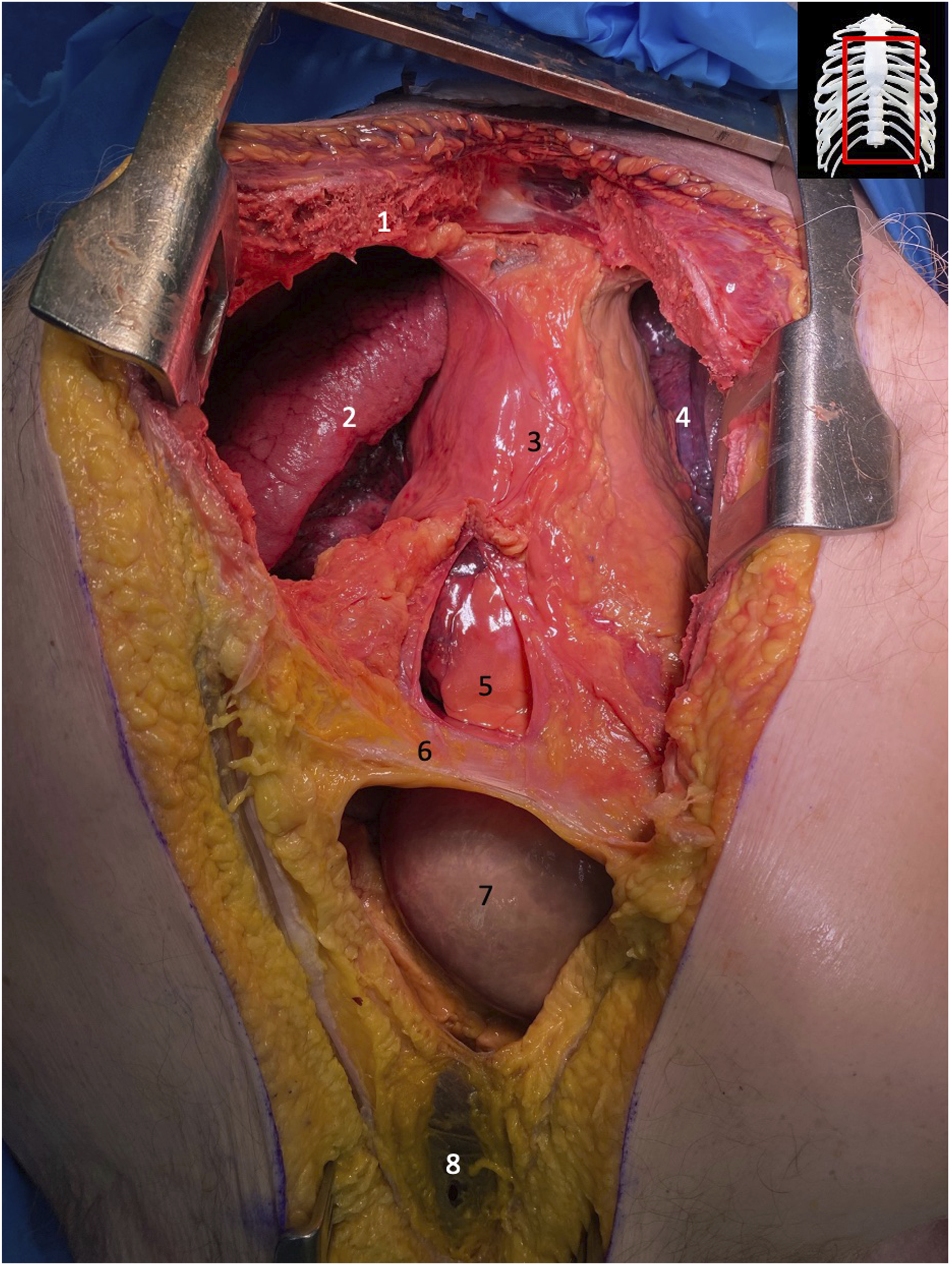
Exposure of mediastinum and adjacent structures post median sternotomy. 1: right portion of sternum post sternotomy; 2: superior lobe of right lung; 3: mediastinum; 4: superior lobe of left lung; 5: pericardium underneath opened mediastinum; 6: diaphragm; 7: section of transverse colon underneath opened peritoneum; 8: peritoneum.
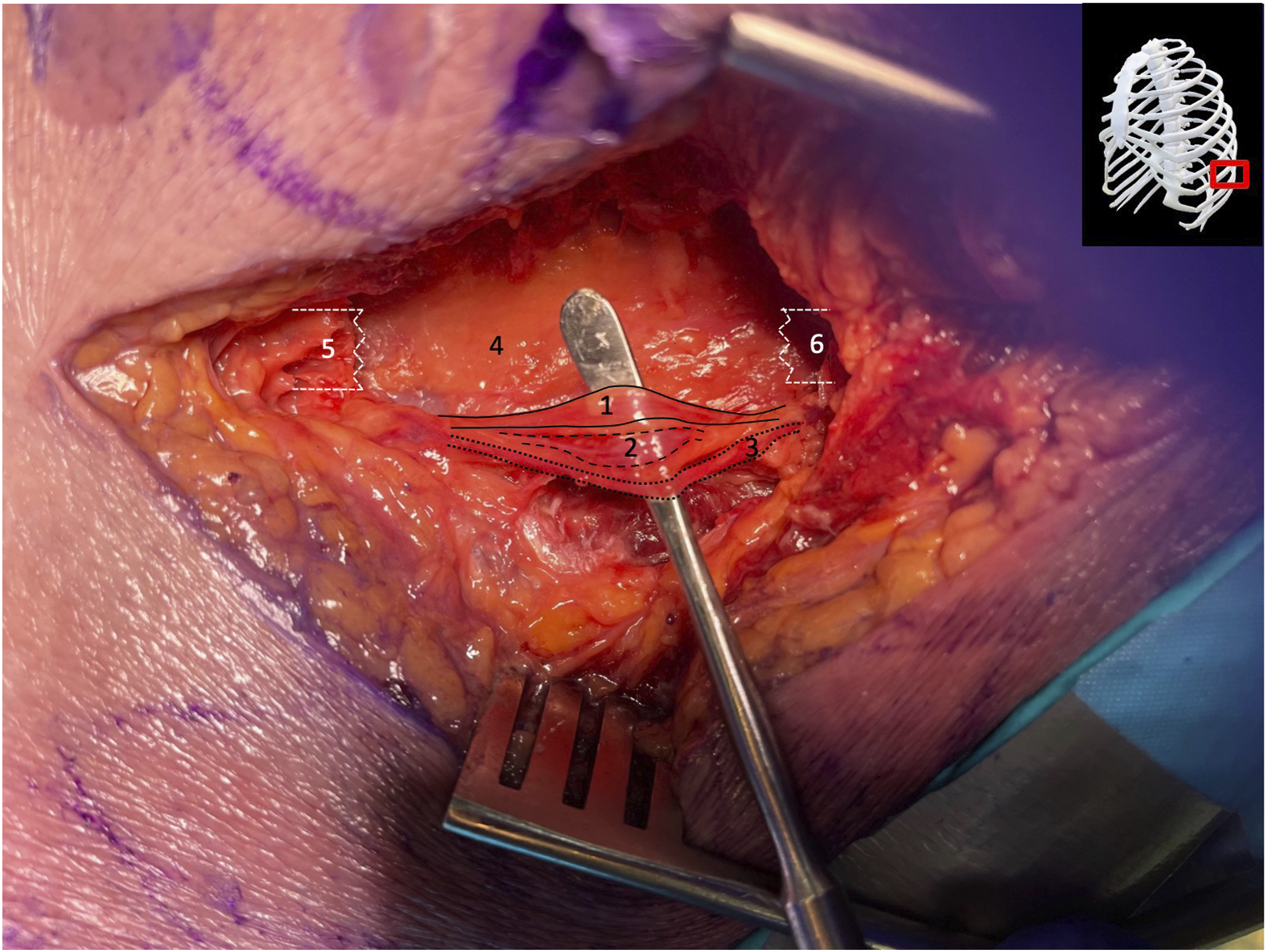
Exposure of T11/T12 intercostal neurovascular bundle after removal of a 5 cm portion of the left 11th rib for far lateral approach. 1: intercostal nerve; 2: intercostal vein; 3: intercostal artery; 4: intact parietal pleura; 5: anterior end of 11 rib; 6: posterior end of 11th rib.
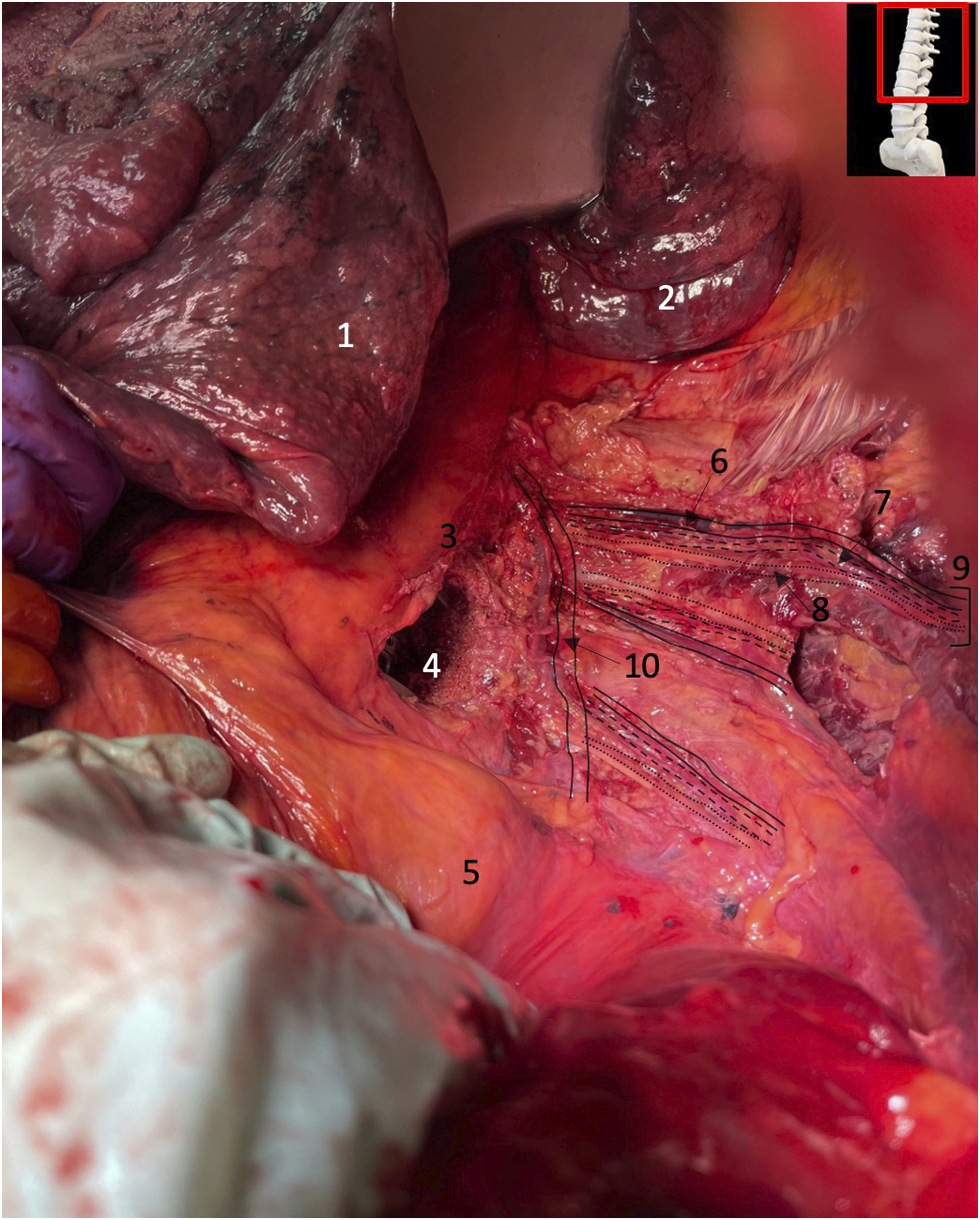
Exposure of T11 – L1 neurovascular bundles post T12 corpectomy and cage implantation. 1: inferior lobe of left lung; 2: superior lobe of left lung; 3: intact aorta; 4: cage implant; 5: intact diaphragm; 6: intercostal vein (solid line); 7: intercostal artery (dashed line); 8: intercostal nerve (dotted line); 9: T11 neurovascular bundle; 10: hemiazygos vein.
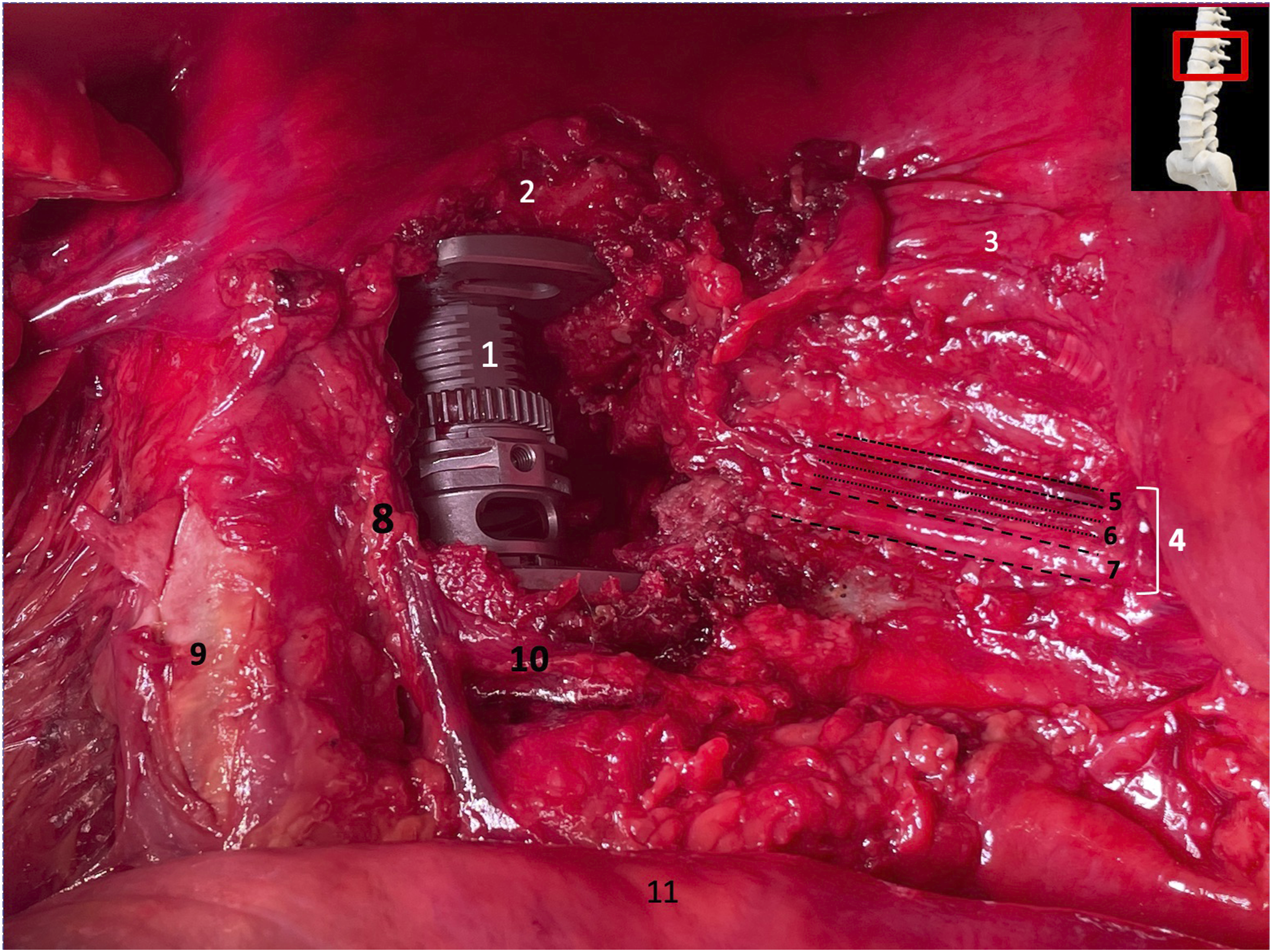
Overview of some of the most crucial structures of the surgical site after performing a T12 corpectomy and cage implantation via a far-left lateral approach. 1: implanted cage at the level of T12; 2: 11th thoracic vertebra (T11); 3: parietal pleura covering the left side of the rib cage; 4: intercostal neurovascular bundle; 5: intercostal vein; 6: intercostal artery; 7: intercostal nerve; 8: external anterior vertebral venous plexus; 9: aorta; 10: segmental vein of L1; 11: diaphragm.
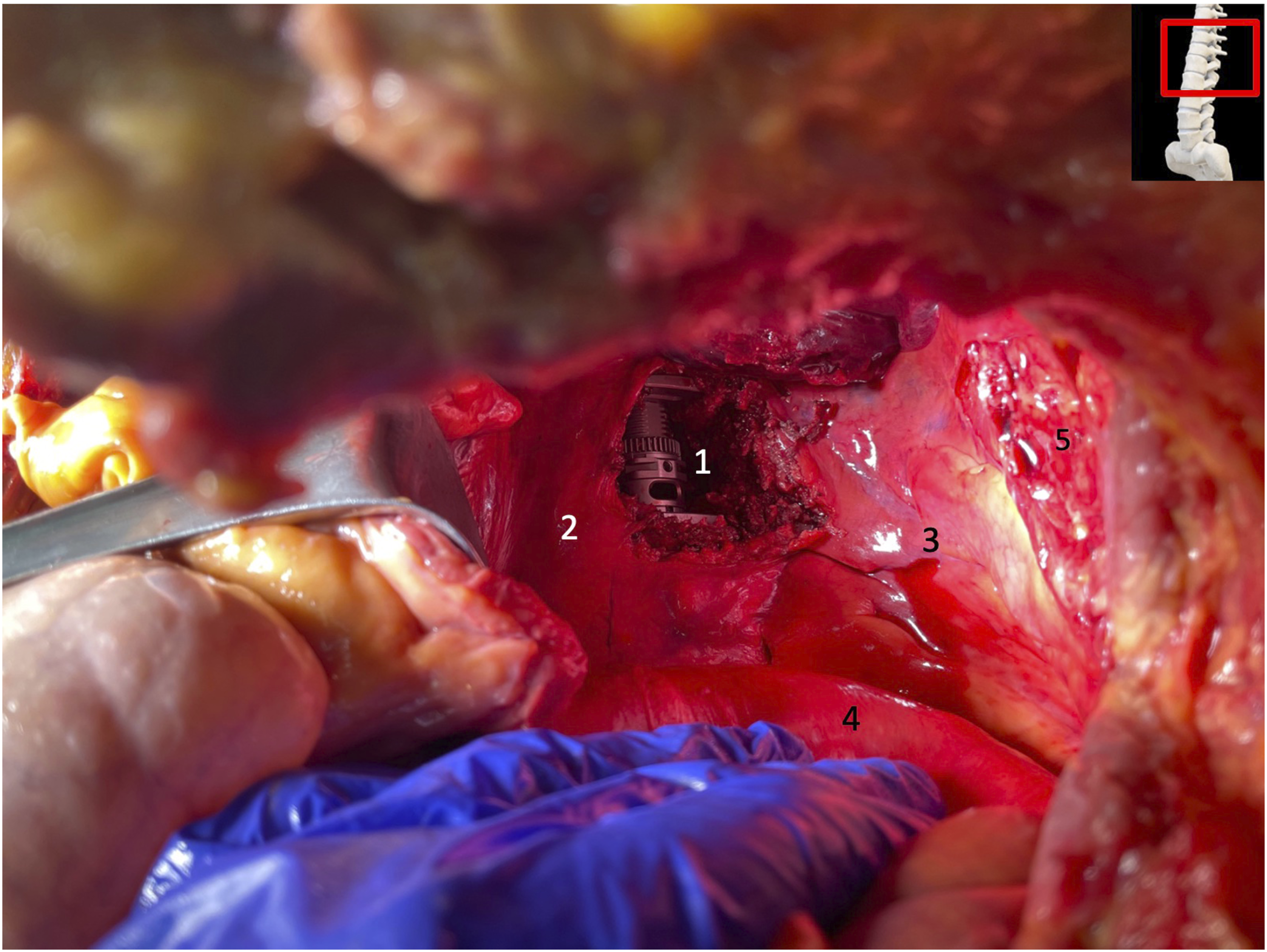
Exposure of cage implantation post T12 corpectomy and cage insertion and proximity to vital structures. 1: T12 cage implant; 2: Aorta; 3: intact parietal pleura; 4: intact diaphragm; 5: defect of the parietal pleura
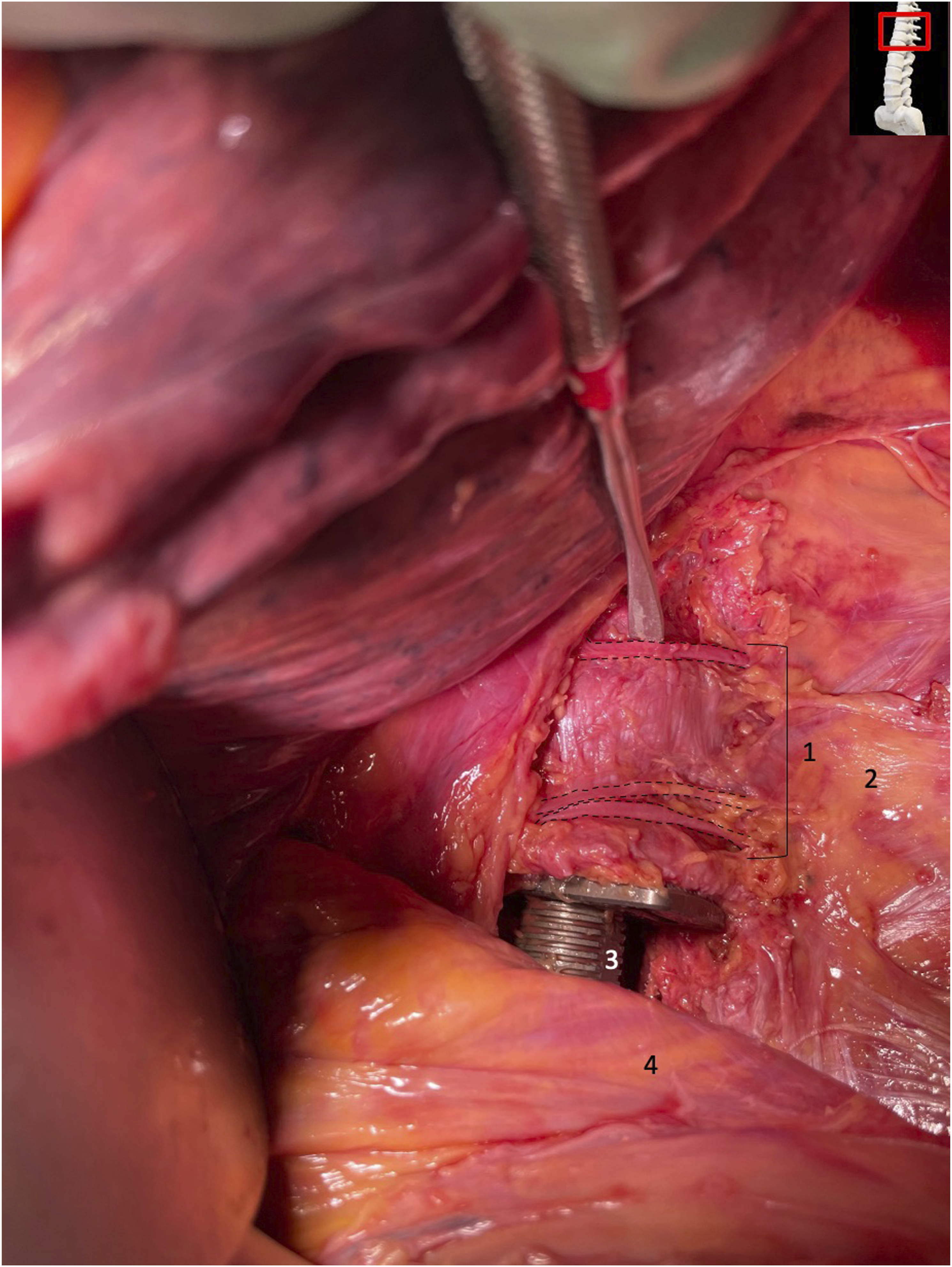
Exposure of the T12 cage implantation, associated segmental veins and shape of the diaphragm. 1: segmental veins; 2: intact parietal pleura; 3: cage implant; 4: intact diaphragm.
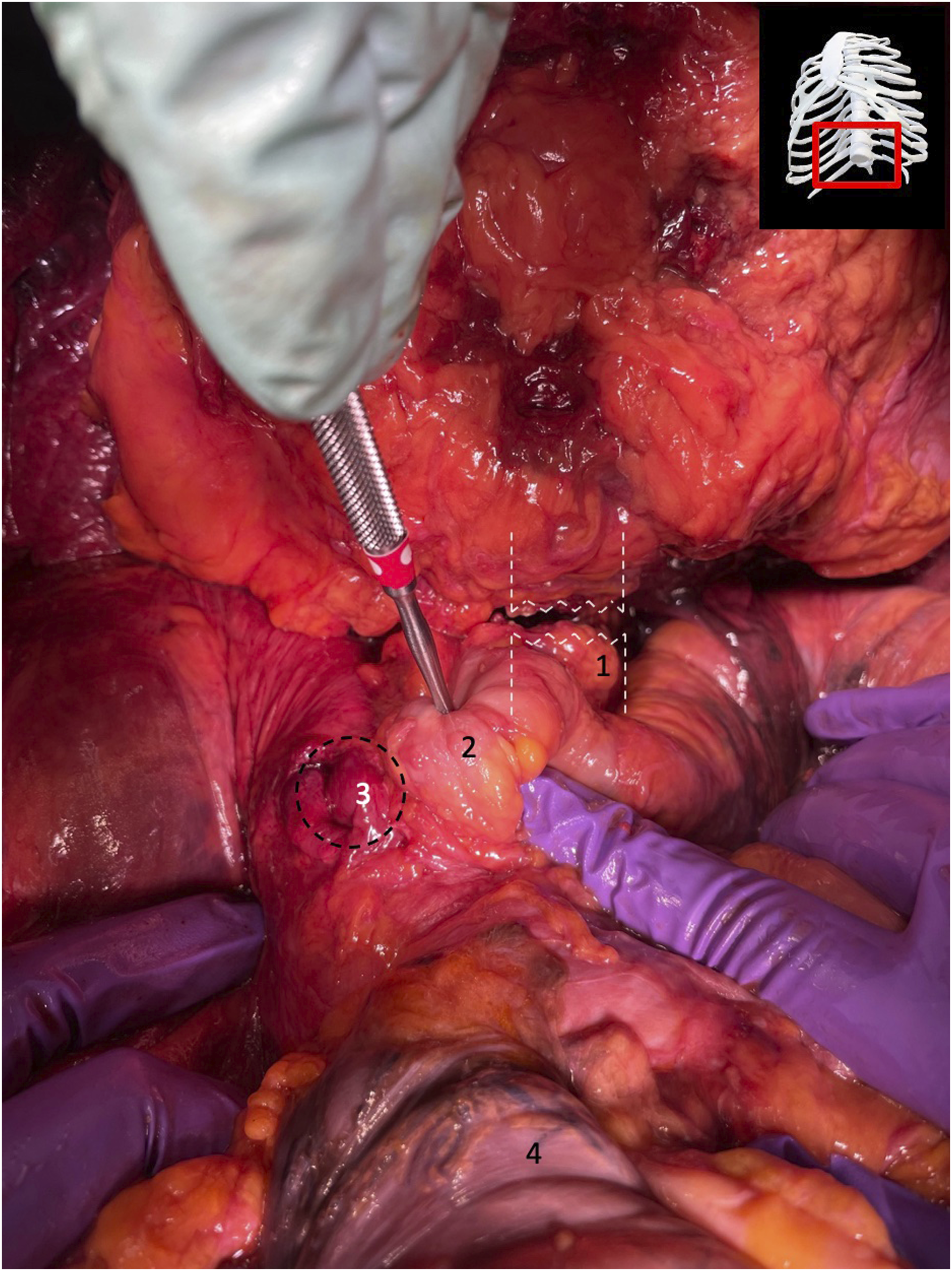
Exposure of adjacent internal structures of the diaphragm in an oblique superior view. 1: dissected calcified aorta; 2: esophagus; 3: inferior vena cava; 4: transverse colon.
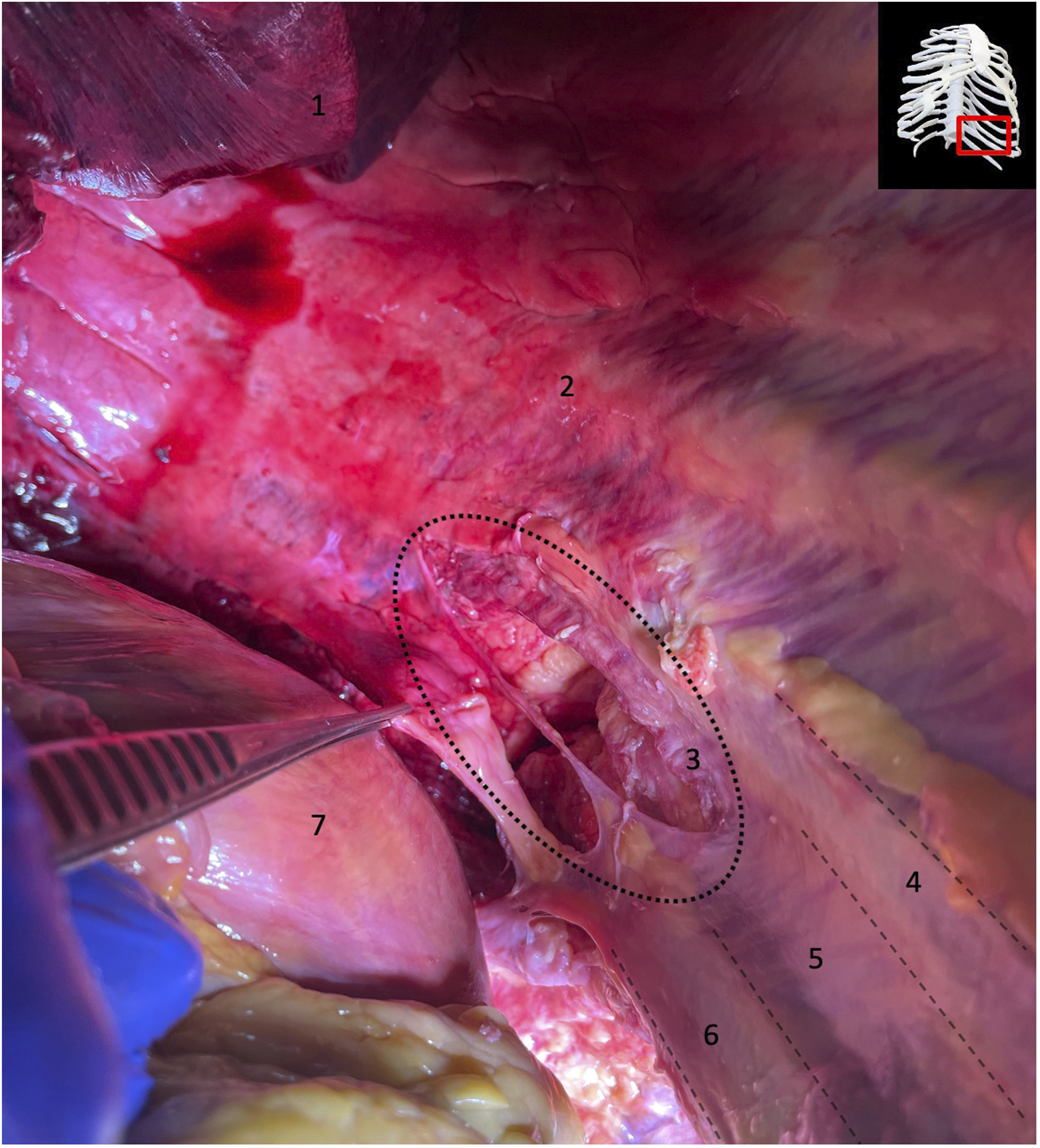
Exposure of an intraoperative defect of the parietal pleura after a T12-corpectomy and cage implantation. 1: left lower lobe of the lung; 2: intact parietal pleura covering the inner surface of the left-sided rib cage; 3: intraoperatively injured / ruptured parietal pleura above the 11th and 12th left rib; 4: 11th rib underneath the parietal pleura; 5: 11th intercostal space; 6: 12th rib underneath the parietal pleura; 7: diaphragm (partly detached from the 12th rib).
Rarely mentioned structures in the diaphragm such as the foramen of Morgagni (median size 10 × 24 mm) and the foramen of Bochdalek (median size 20 × 30 mm) were also measured (Table 2). Furthermore, we found no need to ligate the exiting segmental nerve roots.
Discussion
In clinical practice far lateral surgery to thoracolumbar spine has been relatively safe with reported complications mainly involving temporary lumbar plexus impairment in up to 20% and with reported 7% permanent postoperative neurologic deficits.6,7 Injuries to the great vessels, ureters and bowel are reportedly rare (approximately 0.6%). 8 Thus far, only few basic research projects have critically assessed far lateral spine surgery and the possible vital structures at risk. In the light of less invasive surgery with tubular retractor systems, impeded direct visualization of critical organ structures has given way to neuromonitoring and image guidance assistance to make these procedures safer. A recent solitary cadaveric study by Uribe et al categorized generalized approach considerations for three anatomic thoracolumbar regions without finding any specific qualitative concerns, however, lacked detailed measurement of the vital structures involved. 9
To date, there have been no dedicated anatomic studies pertaining to actual safety distances to vital structures at risk during less-invasive far lateral spine surgery.
In our study, the parietal pleural injuries varied in size and outside of an intentional segmental vein ligation we could prevent vessel violations while preserving the key vessels and lymphatics. These findings are perhaps maybe even more remarkable considering that all the procedures were performed by surgical trainees (fellows) after having received dedicated training.
Our ventral formal exploration through a celiotomy provided a very clear foundation to observe the surgical dissection path and all structures of the left and right of the torso. The number of 6 cadavers is similar to other studies discovering the anatomical relations of the vital thoracolumbar structures. 10 A major limitation of our study inherent to its cadaveric nature is the fact that the thoracic cavity had no inflated lungs and no physiologic inspirations making defects at the visceral pleura very unlikely.
Further the potential of vessel injuries in our cadaver project may underestimate the actual risk for arterial/venous injuries in a living human being, given these structures would be filled with blood and show pulsations, possibly increasing the risk of injuries during actual surgery.
Additionally, the location of the base of the visceral pleura was more cranially located at the height of T10 level due to the post mortal collapse of the lungs and therefore did not interfere with the surgical access. Considering the parietal injuries in our study, visceral injuries could therefore be an issue in vivo as in clinical practice. We usually do not deflate the lungs.
Another important issue to consider is the position of the cadaver, given that we performed the anterior approach via a median thoracotomy and laparotomy, but the surgical procedure is executed in a right lateral decubitus position. The relationships of the anatomical structures and shifting of structures during surgical position remain controversial. Yet, there has not been an established standard measurement of these vital structures because our study is the first one measuring concrete values regarding the approximity of the vital organs of the thoracolumbar junction to the surgical approach. We used a supine position for our median sternotomy and laparotomy because of the better feasability and reproducability of this position, compared to a lateral decubitus position. It helps to provide direct visualization for possible damage to the neurovascular structures and other vital organs. Our study carefully utilized the anterior approach celiotomy to adequately visualize all the vital organs adjacent to the surgical site pertaining to the procedure.
The use of torso cadavers instead of full body cadavers as it pertains to the procedure and potential impact on the vital structures is not known to negatively influence the results. There have been no studies which examined possible differences between torsos and fullbody cadavers for this procedure so far and for the sake of saving valuable cadaver material, we focussed on the torsos.
One of the most compelling findings in our study was the identification of the unique shape of the diaphragm with an attachment of the posterior portion as low as the height of the L2 vertebral body and beyond that the L2/3 disc space ridge formed by the endplates. In the anatomic literature, an attachment of the posterior fibers of the diaphragm at the height of T12-L1 is frequently described. Still, considering our small number of cadavers, further studies could be needed to further verify our finding across larger population samples.
Conclusion
Our cadaver study identified the feasibility of performing a T12-corpectomy through a far lateral less invasive tubular approach with no violation of the actual diaphragm and expected limited injuries to the parietal pleura only. Formal measurements of distances to the vital structures were obtained, are listed, and are presented together with a detailed graphic depiction. This cadaveric study did support the fact that one can perform a T12-corpectomy without a diaphragm injury. In fact, we could have concluded a L1 corpectomy without diaphragm injuries since the diaphragm was found to originate at the L2 vertebra and below. Moreover, our study did demonstrate the reproducibility of performing a surgical technique considering only Spine surgeons in learning performed these procedures after receiving structured training.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
