Abstract
Study Design:
Retrospective observational.
Objectives:
This study aimed to document the safety and efficacy of lumbar corpectomy with reconstruction of anterior column through posterior-only approach in complete burst fractures.
Methods:
In this retrospective study, we analyzed complete lumbar burst fractures treated with corpectomy through posterior only approach between 2014 and 2018. Clinical and intraoperative data including pre and post-operative neurology as per the ISNCSCI grade, VAS score, operative time, blood loss and radiological parameters, including pre and post-surgery kyphosis, height loss and canal compromise was assessed.
Results:
A total of 45 patients, with a mean age of 38.89 and a TLICS score 5 or more were analyzed. Preoperative VAS was 7-10. Mean operating time was 219.56 ± 30.15 minutes. Mean blood loss was 1280 ± 224.21 ml. 23 patients underwent short segment fixation and 22 underwent long segment fixation. There was no deterioration in post-operative neurological status in any patient. At follow-up, the VAS score was in the range of 1-3. The difference in preoperative kyphosis and immediate post-operative deformity correction, preoperative loss of height in vertebra and immediate post-operative correction in height were significant (p < 0.05).
Conclusion:
The posterior-only approach is safe, efficient, and provides rigid posterior stabilization, 360° neural decompression, and anterior reconstruction without the need for the anterior approach and its possible approach-related morbidity. We achieved good results with an all posterior approach in 45 patients of lumbar burst fracture (LBF) which is the largest series of this nature.
Introduction
Burst fractures involve the anterior and middle column of a vertebra causing bony fragments to compromise the spinal canal.1,2 The goals of surgery in LBF include stabilizing the spine, avoiding kyphosis and providing pain free mobilization. 3 There are only a few reports that describe the operative indications, risks, and benefits associated with the various treatment options for unstable lumbar burst fractures in currently published literature, particularly in reference to the lower lumbar segments. Furthermore, there is limited literature on lumbar corpectomy for LBF through a “posterior-only” approach. A load sharing classification (LSC) scores of 7 to 9 indicate severe vertebral comminution resulting in loss of anterior support. In this circumstance, posterior-only stabilization results in additional load on pedicle screws and thereby, a higher chance of instrumentation failure. 4 However, following developments in techniques and implants over the last decade, it remains unclear whether LSC can accurately predict the need for anterior fixation.
Patients with unstable lumbar burst fracture require surgical management with spinal instrumentation to relieve pain, address neurological deficits, and stabilize the spine to prevent further deformity. However, the optimal surgical treatment for unstable lumbar burst fractures remains controversial. Literature exists regarding posterior only approach for unstable burst fractures of the dorsal spine; however, it is less commonly used for unstable lumbar burst fractures. This is because, placement of a cage is usually difficult from the posterior approach owing to the need to navigate around the lumbar nerve roots.
In the present single-center study, we report single-stage posterior-only vertebrectomy with circumferential decompression and stabilization in unstable lumbar burst fractures. We have focused on the indications, surgical techniques, complications and outcomes of patients treated with this technique. The authors hypothesize that, posterior-only lumbar corpectomy reduces complications secondary to anterior and anterior plus posterior (combined) approach. Our study aimed at documenting the efficacy of lumbar corpectomy and reconstruction of anterior column using a “posterior-only” approach.
Materials and Methods
This retrospective study was conducted at a tertiary level spine surgery center. Approval from the institutional review board was taken before commencing recruitment (ISIC/ RP/ 2019/ 019). Written informed consent was obtained from each patient. We analyzed hospital records between 2014 and 2017. Patients with a lumbar corpectomy carried out via “all posterior approach” for burst fractures were assessed and followed. Only patients who have completed atleast 2 years’ follow-up after the surgery were included. Plain radiographs, CT and MRI studies were evaluated. TLICS score and AO spine classification of thoracolumbar burst fractures was noted. Patients with LBF (AO type A4-complete burst), TLICS score >5, corpectomy performed via posterior approach, who completed at least 2-year follow-up following surgery were included. Clinical data assessed included neurology as per the ISNCSCI grade5,6 and VAS.7,8 Intra-operative data including operative time and blood loss was noted. Radiological variables assessed were kyphosis, vertebral height loss and canal compromise. Short segment fixation was defined as fixation extending not more than 1 level proximal and distal to fractured vertebra. Any fixation extending beyond this was considered as long segment fixation.
The amount of kyphosis at the fractured level was measured between superior endplate of vertebra above and inferior endplate of vertebra below affected level. (Figure 1) Loss of height was calculated using the following method: 100 − 2F/ (A+ B) 100, where F represents height of fractured segment; A, height of the proximal segment; and B, height of the distal segment (segmental height, height of vertebra plus the height of adjacent 2 discs, or the height between midpoint of lower endplate of proximal vertebra and that of upper endplate of distal vertebra. Both measurements were made on a lateral X-ray. (Figure 2) Percentage canal encroachment was calculated using the formula: a = (1- x/y) 100; where a is percentage of canal encroachment, x is midsaggital diameter of spinal canal at fractured vertebra; and y is the mean midsaggital diameter of spinal canal at the levels superior and inferior to the fractured vertebra on computer tomography (Figure 3). 9 Each radiological parameter was independently assessed by 2 observers and the mean value for each variable was considered.
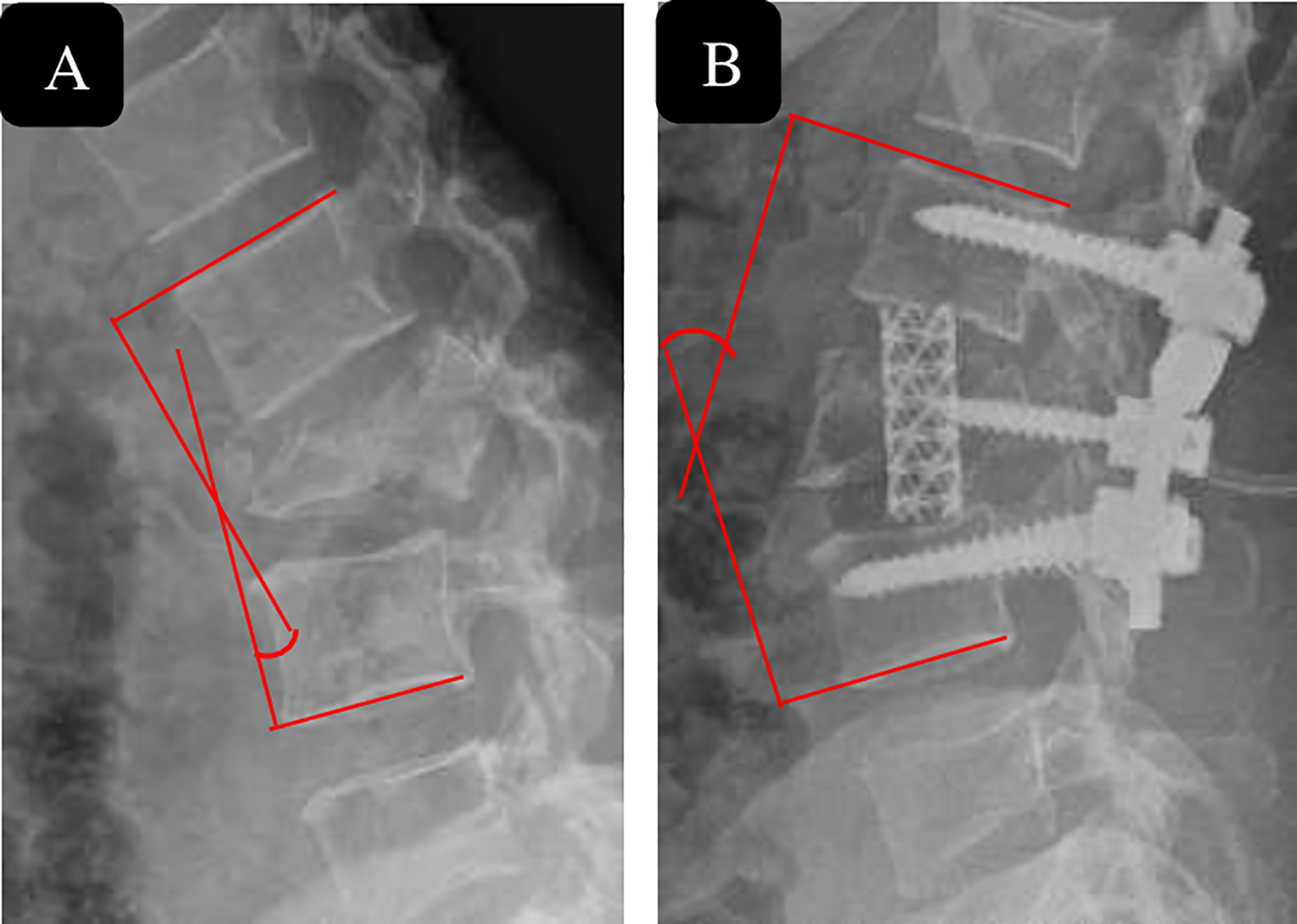
Measurement of the kyphotic and lordosis angle. (A) Shows measurement of the angle of kyphotic deformity of the fractured segment was measured as the angle between the superior endplate of the vertebral body above the affected level and the inferior endplate of the vertebral body below the affected level, where kyphosis is recorded as a positive one, and (B) lordosis is recorded as a negative one.
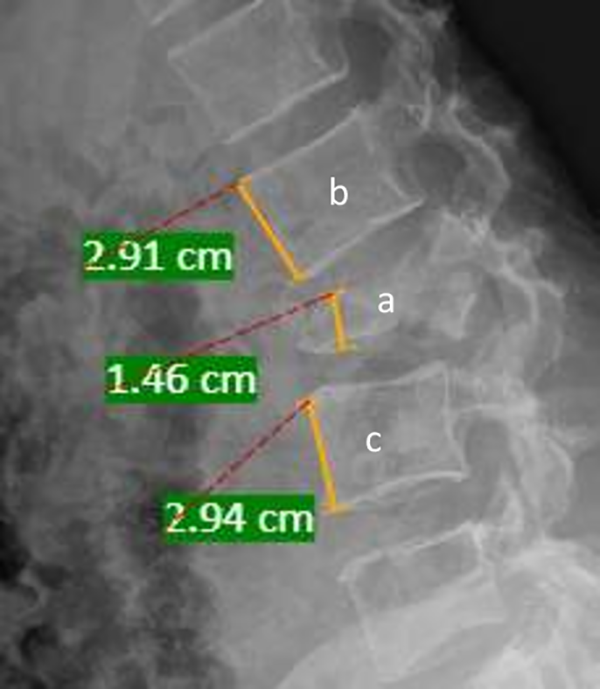
Percentage of anterior body height compression (% ABC). Percentage of anterior body height compression (% ABC) is calculated by the formula: % ABC = 100 − 2a/ (b + c) 100, where a is the height of fractured vertebra; b is the height of the proximal vertebra; and c is the height of the distal vertebra (measured by the PACS measurement software).
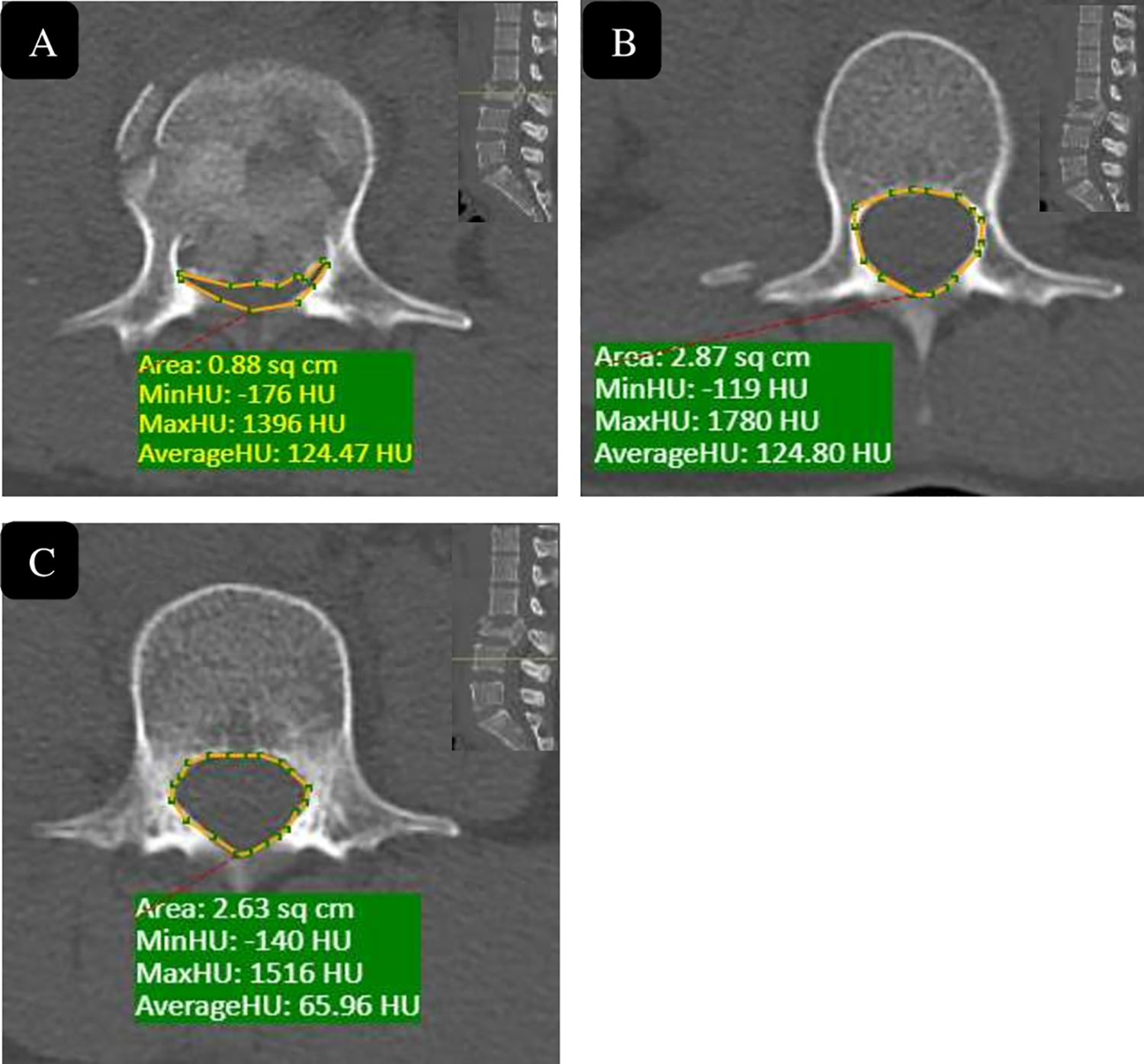
Canal encroachment and the percentage of canal encroachment. Canal area measured by the PACS measurement software. (A) is the area of the injured vertebra, (B) and (C) are that of the 2 adjacent vertebrae, and the percentage of canal encroachment (%CE) was calculated as the area of the protrusion into the canal of the injured vertebra divided by the mean of the maxim canal area of the adjacent 2 vertebrae, that is %CE = 100-2A/ (B + C) × 100.
Surgical procedure: Multimodal neuromonitoring (SSEP/ MEP) was used intra-operatively in all patients (except 1 patient with AIS-A neurology). Baseline values after induction prior to positioning were taken in all cases. Tranexamic acid was utilized to minimize blood loss with a loading dose of 50 mg/kg and maintenance dose of 5 mg/kg/hr. Under general anesthesia and in the prone position, using, pedicle screws were inserted through the posterior midline approach and placement confirmed by intraoperative image intensifier. A temporary rod was inserted on the side opposite to area of work. Laminectomy was done along with unilateral facetetcomy of affected level and the level above. After discectomy and defining the nerve roots, transpedicular corpectomy was carried out through the window between nerve roots using a burr. Dura and the exiting and traversing roots were protected using nerve root retractors. The temporary rod was then exchanged to opposite side and procedure was repeated on contralateral side. The endplates were denuded while ensuring no damage to the underlying bone. The size of reconstruction cage was estimated using a K-wire cut approximately to size of void created and checked under image intensifier.
For insertion of the mesh cage, exiting as well as traversing nerve roots were gently retracted using nerve-root retractors. A Harm’s mesh cage was filled with bone autograft and inserted obliquely, first engaging the upper end plate of the lower end vertebra followed by gradual insertion of the proximal end of the mesh cage using cage pusher with prongs under direct vision and image intensification. The cage was adjusted until it was adequately placed on the both the endplates (Figure 4). A contoured connecting rod was placed to recreate local lordosis. Harm’s titanium mesh cage filled with autograft was used in all cases. Controlled compression through the pedicle screws ensured that the cage was secured between the 2 bony end plates, achieving good contact. A concurrent posterolateral spinal fusion was carried out in all cases after decortication of the laminae, transverse processes and facet joints and local autografting.
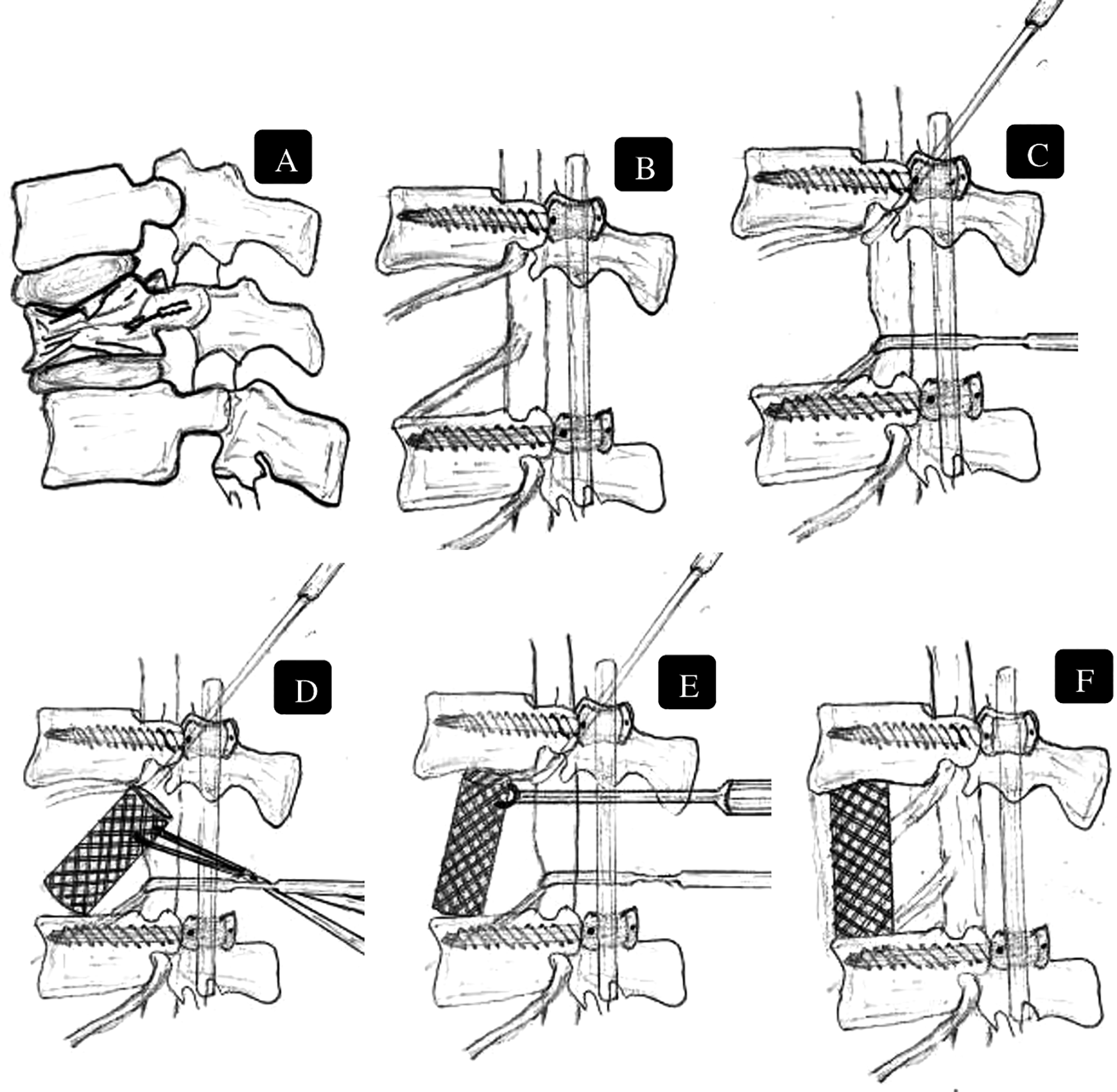
Sequential steps (A-F) involving vertebrectomy and retraction of nerve roots and dura to insert appropriate size titanium mesh cage using cage holder by engaging on the upper end plate of lower end vertebra and realigning it to desired position using cage pusher.
From the second post-operative day, patients were mobilized without a brace as per the pain tolerance (sitting/ wheelchair mobilization or walking) depending neurological status. Radiographs were taken on first post-operative day. Follow-up consultations were conducted post-operatively at 2 weeks, 3 months, 6 months, 12 months, and then every year. Fusion was assessed on plain radiographs by assessing the bridging bony trabeculae without lucencies between adjacent endplates. In the instance where the Harm’s cage masked the bridging bone inside, and stability of the construct was assessed by evaluating any change in cage position on serial follow-up images (Figures 5 and 6).
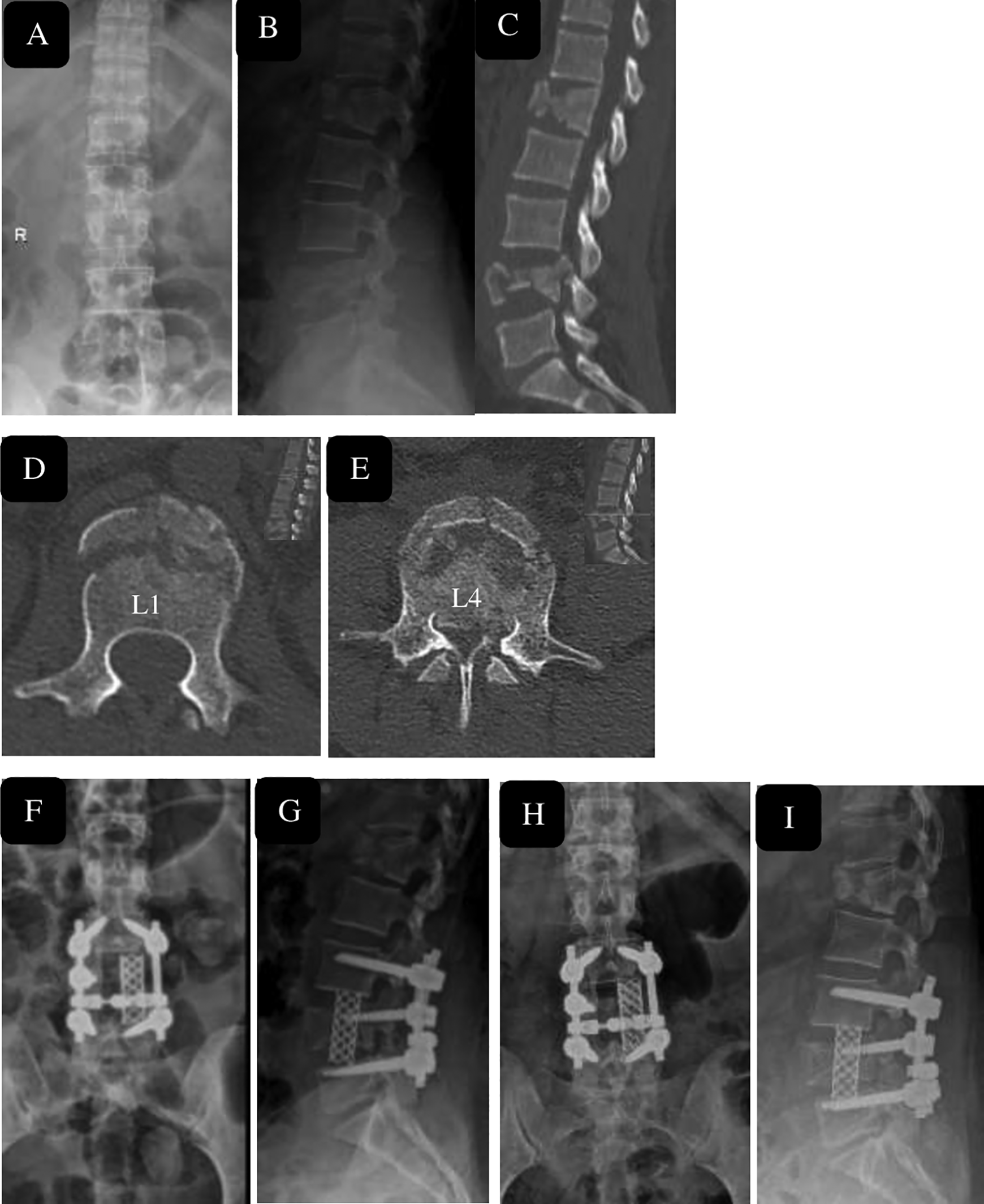
29 year female after fall from height, with burst Fracture L4 and L1 (A, B) with canal compromise at L4 level seen on sagittal and axial Computer tomography images (C-E). conservatively managed L1 fracture with short segment stabilization and corpectomy L4 with restoration of height and segmental lordosis immediate post operatively (F, G) and maintenance of height regained and lordosis with fusion seen at 2 year follow-up (H, I).
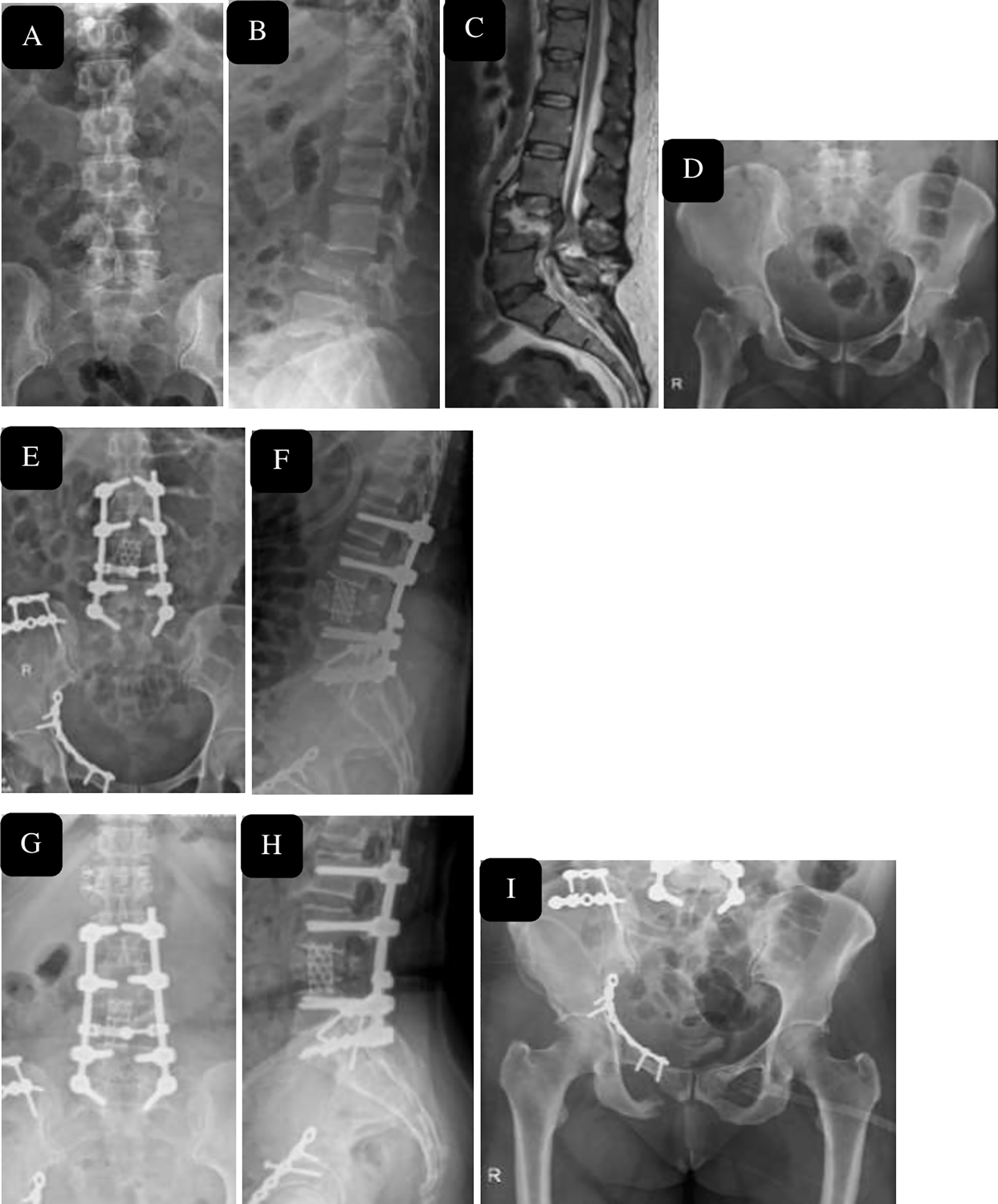
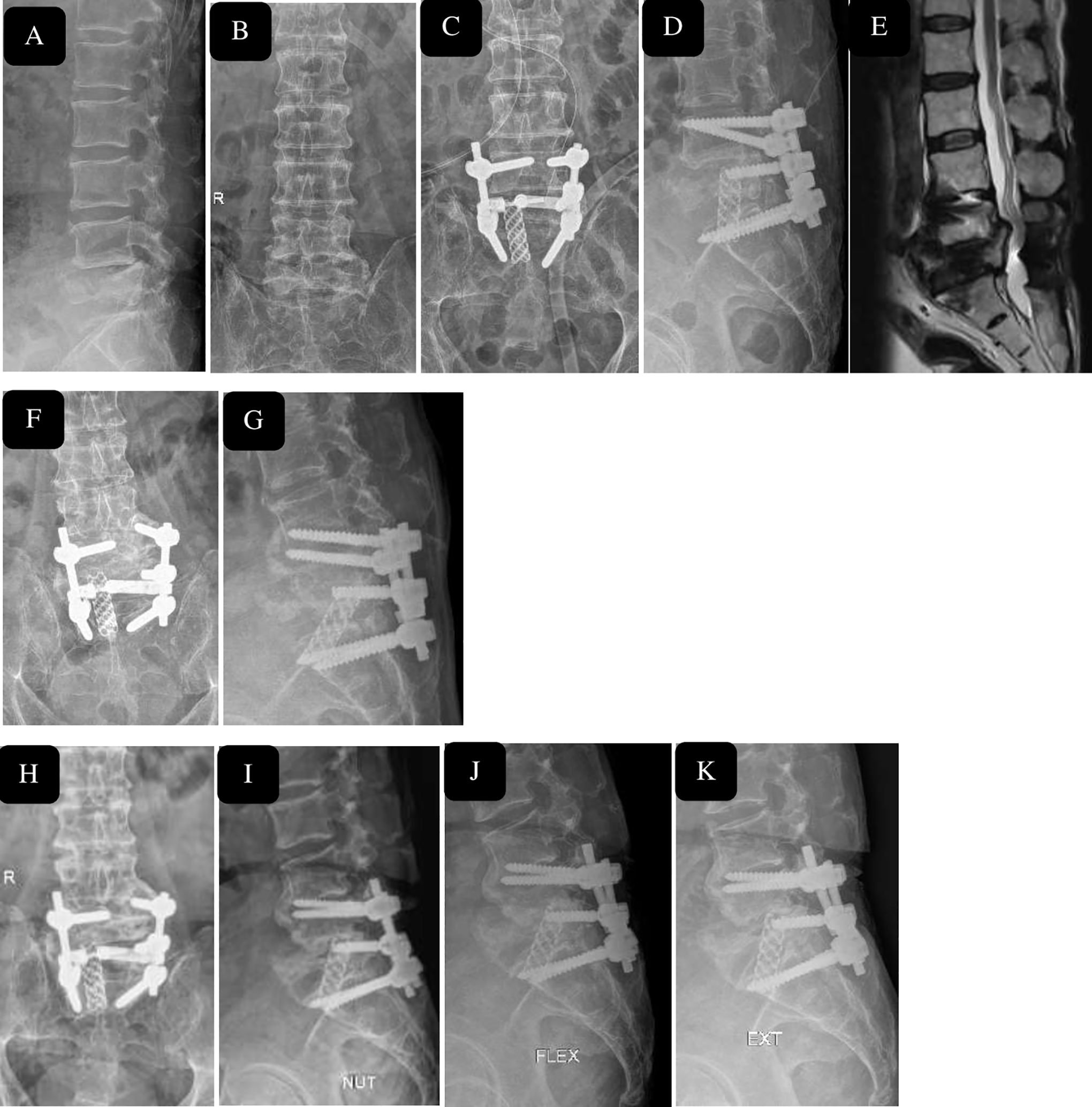
47 year female after fall from height, with burst Fracture L4 (A, B) with canal compromise at L4 level seen on sagittal magnetic resonance imaging (C) and acetabulum/ iliac wing fracture (D), managed with long segment stabilization and corpectomy L4, open reduction internal fixation of right acetabulum, ilium with restoration of height and segmental lordosis immediate post operatively (E, F) and maintenance of height regained and lordosis seen at 1 year follow-up (I-K).
Statistical analysis: Qualitative data was described as a percentage. The parametric quantitative data was presented as mean and standard deviation. Comparison between each of the 2 groups was made using Fisher exact test for qualitative data and paired t-test for parametric quantitative data. Statistical analysis was performed using SPSS software (Version 21.0. Armonk, NY: IBM Corp).
Interobserver and intraobserver reliability: The kappa coefficients for intraobserver reliability for percentage loss of vertebral height, focal kyphosis and percentage of canal compromise were 0.85 (95% confidence interval 0.74–0.95) and 0.89 (95% confidence interval 0.79–0.99), respectively. The kappa coefficients for interobserver reliability were 0.74 (95% confidence interval 0.59– 22 0.88) and 0.75 (95% confidence interval 0.64–0.87), respectively, indicating high conformity.
Results
We had a total of 49 patients who satisfied the inclusion criteria of which 45 patients completed the follow-up duration and were included in the analysis (Table 1). Mean age was 38.89 ± 11.03 years. (Range 17-60 years). We had 60% male patients (27/45) in our series. Follow-up period ranged from 2- 6 years with the mean follow-up duration of 37 months. The most common mode of injury was motor vehicle accident (34 patients); while the remaining 11 patients sustained a fall from height.
Demographics, Perioperative Characteristics and Follow-Up Data of the Patients.
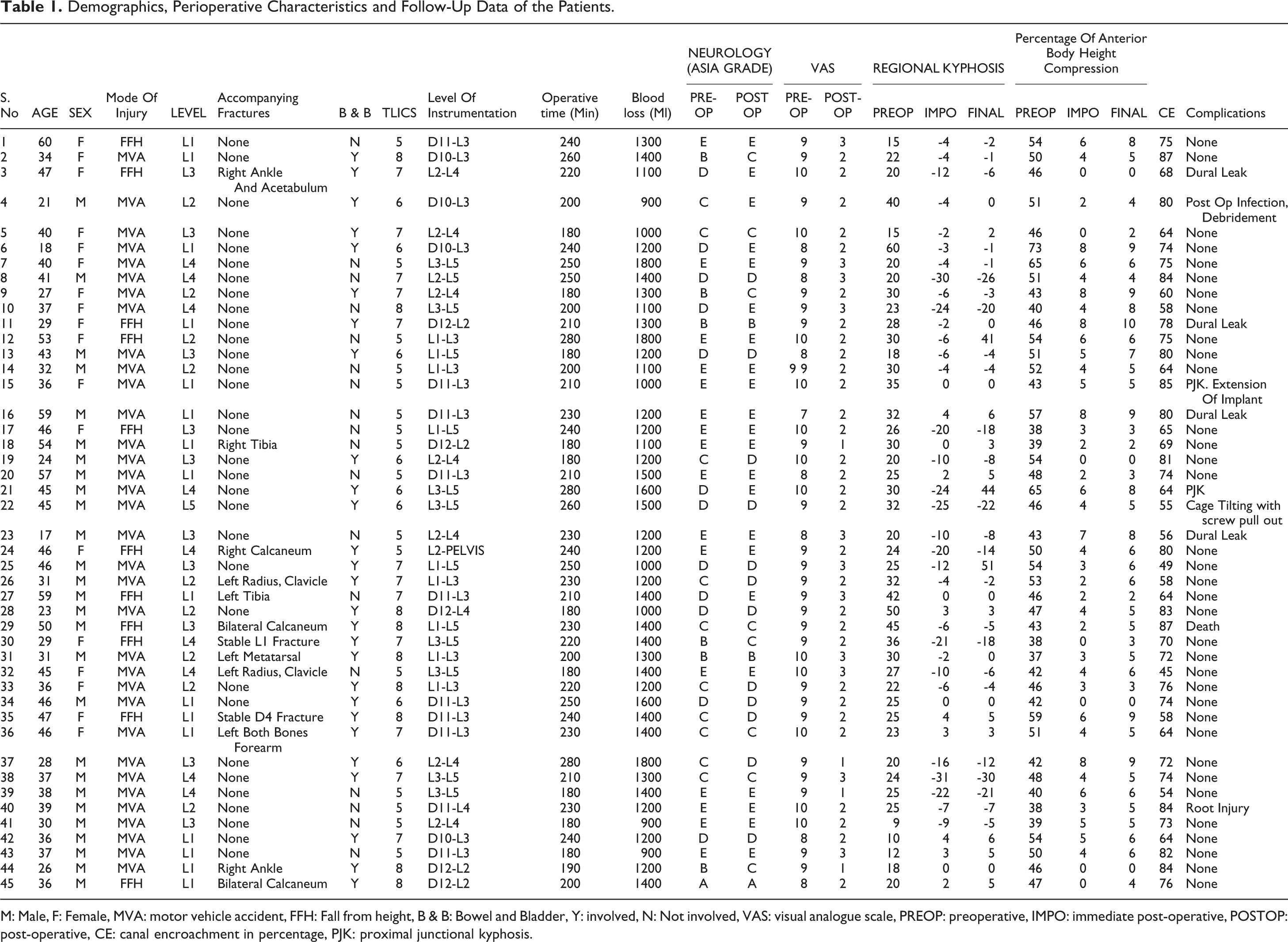
M: Male, F: Female, MVA: motor vehicle accident, FFH: Fall from height, B & B: Bowel and Bladder, Y: involved, N: Not involved, VAS: visual analogue scale, PREOP: preoperative, IMPO: immediate post-operative, POSTOP: post-operative, CE: canal encroachment in percentage, PJK: proximal junctional kyphosis.
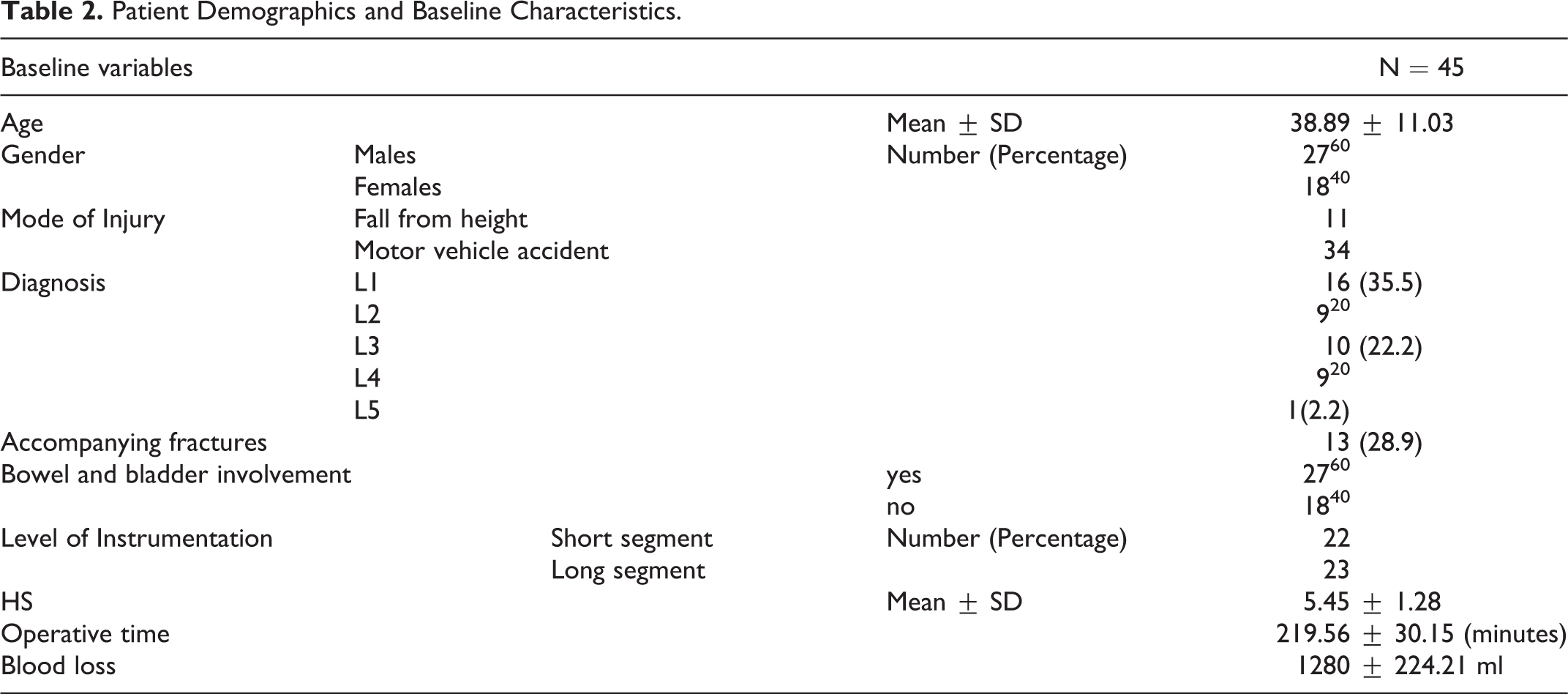
The most commonly injured vertebral level was L1, accounting for 35.6% of cases. According to the AO classification, every case was classified as AO type A4 (complete burst) (Table 2). All subjects scored 5 or more when scored using TLICS. Mean preoperative kyphosis was 26.44° ± 9.95° (range 9°- 60°). Mean preoperative loss in vertebral height was 47.33 ± 7.25% (range 37-73%). The mean canal compromise was 70.98 ± 10.66% (range 45-87%). The mean surgical duration was 219.56 ± 30.15 minutes (range: 180-280 minutes). Mean blood loss was 1280 ± 224.21 ml (range: 900-1800 ml). 23 (51.1%) patients underwent short segment fixation and 22 (48.9%) long segment fixation. The mean correction of kyphosis in the immediate post-operative period was 34.02° ± 13.40° (range 6°-63°) and correction of kyphosis on final follow-up was 31.80° ± 13.35° (range 4°-61°). The change in kyphosis, preoperative and immediate post-operative deformity correction was significant. Also, change in kyphosis, preoperative and final follow-up correction was significant. Mean correction in the height in immediate post-operative period was 43.47 ± 7.01% (range 34-65%) and in follow-up period was 42.16 ± 7.04% (range 32-64%). The change in vertebral height, preoperative and immediate post-operative correction in height was significant. The change in vertebral height from preoperative to follow-up was significant. The preoperative VAS ranged from 7-10 in all cases. At follow-up, VAS was in the range of 1-3. In majority of cases, the score was 2 (30 patients, 66.7%) followed by 3 (11 patients, 24.4%) and 1 (4 patients, 8.9%).
Patient Demographics and Baseline Characteristics.
16 patients presented with intact neurology, 12 with AIS grade D, 10 with grade C, 6 with grade B and 1 patient with grade A neurology. 27 patients had involvement of bladder or bowel and in 18 cases, bowel and bladder function was intact. Based on the ASIA impairment scale, there was no deterioration in post-operative neurological status in any of the patients. In 15 (33.3%) patients the neurological status improved and it remained unchanged in others. (Table 3)
Preoperative and Post-Operative Neurological Status.
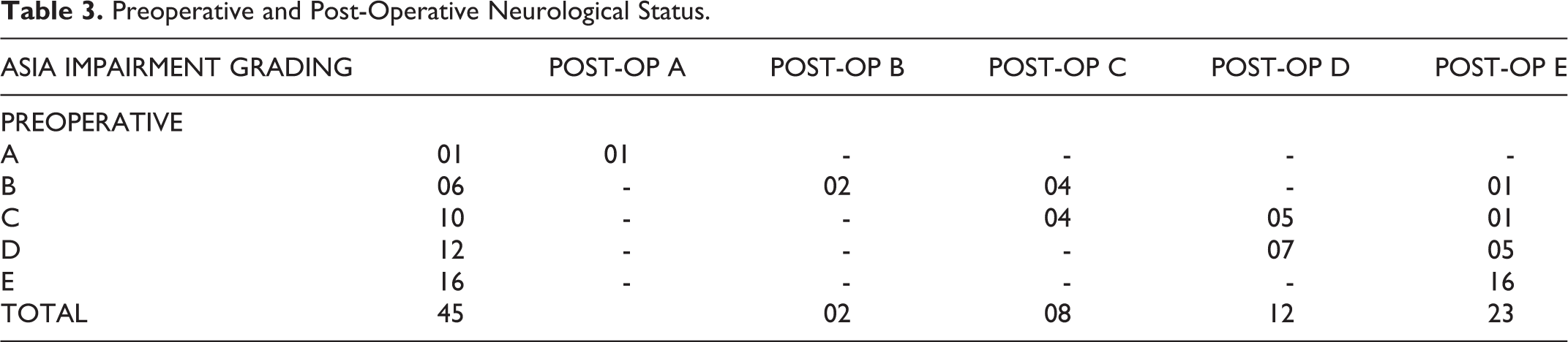
Two cases of PJK were observed, of which one was revised with extension of the construct. 4 incidences of dural leak were observed due to preexisting trauma leading to bone impingement, 1 case of root injury during cage insertion which recovered in follow-up, indicating neuropraxia, 1 case of tilted cage with screw pull out (patient denied revision procedure) (Figure 7) and 1 superficial infection requiring debridement and resuturing. All patients achieved bony fusion confirmed by radiographic evaluation.
45 year old male after motor vehicle accident with L5 burst fracture, (A, B), was treated with short segment posterior stabilization and L5 corpectomy and titanium mesh cage, immediate post op image showing restoration of height (C, D) and canal decompression on Sagittal MR image (E). Cage tilting with screw cut out seen at 1 year follow-up, patient denied revision procedure. Fusion seen with no further loss of height and alignment at the end of 2 years (H-K).
Discussion
The reconstruction of unstable LBF represents a complex challenge depending on the type of fracture, level of injury, and degree of neurologic impairment. Treatment options for LBF are less familiar owing to their low prevalence, lack of conclusive clinical studies and existing controversies regarding the optimal treatment strategy. 10 The goals of surgery in LBF are decompression of neural tissue, restoration of vertebral body height, allowing early pain free mobilization, avoiding problems of recumbency and limiting the number of instrumented vertebrae.11-13
Direct access to a ventral lesion, sparing of paraspinal musculature, avoiding previous posterior surgical scars, placement of bone graft in predominant load-bearing anterior segment of the spine, placement of larger cages with endplate to endplate apposition, direct decompression of the spinal canal, intervertebral distraction for height restoration are some of the features that have popularized anterior procedures. However, in untrained hands, this approach is associated with severe complications such as vascular trauma, ureteric injury, retrograde ejaculation and impotence, injury to the lumbosacral plexus, post-operative ileus and peritoneal injuries to name a few. Avoiding these complications may require services of an access surgeon, which may not be available at all centers. The lowest lumbar levels (L4 and L5) have vital structures (bifurcating common iliac vessels) in the vicinity. Shousha et al. reported multiple technical difficulties during L5 corpectomy. 14 An anterior approach is indicated only in scenarios where the posterior column remains intact following the injury; however, the operative risk is higher as compared to posterior approaches in the hands of a surgeon unfamiliar with the approaches. 1 The combined approach, though associated with superior construct rigidity, also has higher morbidity, longer operative time, more blood loss, prolonged hospital stay and rehabilitation. The posterior approach provides clear visualization of the neural elements, and allows multisegmental fixation and correction of deformities. However, significant mechanical stress exposes a posterior-only construct to a high risk of failure because of rod-screw fracture or pullout. However, a posterior approach can avoid the disadvantages of the anterior approach, such as the limited ability to repair an injured posterior column and the ruptured dural sac. Additionally, most surgeons are better acquainted with the posterior approaches.
In the present study, we reconstructed the anterior column via the posterior approach; reducing the morbidity of the surgery. Our study highlights safety and efficacy of this technique. One of the challenges encountered is placement of the cage without injuring the nerve root. Unlike the thoracic spine, where the nerve roots can be sacrificed for the purpose of cage insertion, the same is not possible with the lumbar roots. However, when proper surgical steps are followed, the chances of root injury are low. We achieved a good kyphosis correction and restoration of height of the vertebral body (Figures 5, 6, 8). Clinically, the VAS score improved significantly and no patient deteriorated neurologically following the surgery.
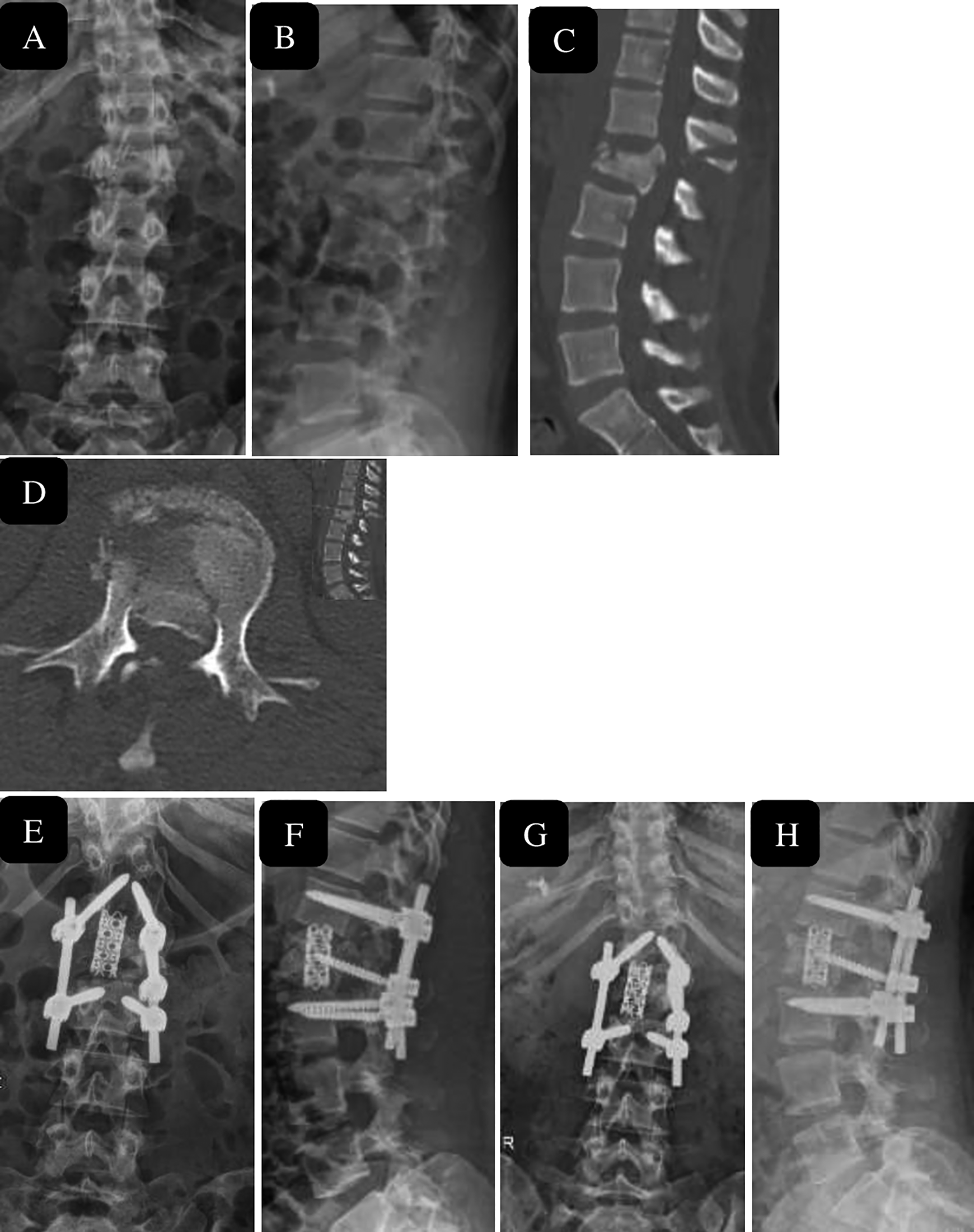
32 year male after road traffic accident, with burst Fracture L2 (A, B) with canal compromise at L2 level seen on sagittal and axial Computer tomography images (C, D). managed with short segment stabilization and corpectomy L2 with restoration of height and segmental lordosis immediate post operatively (E, F) and maintenance of height regained and lordosis with fusion seen at 1.5 year follow-up (G, H).
Gentle manipulation of the cage between nerve roots allows proper insertion without significant traction of the neural elements. Perhaps, inserting a cage after corpectomy via anterior approach is difficult due to limitation of space by the vascular anatomy and other vital structures in this region.15,16 In this series, all patients were ambulated as early as tolerated due to rigid fixation, most often on the first post-operative day. This has physical and psychological advantages for patients and avoids hazards of prolonged recumbency. However, in the lower lumbar spine, the window for inserting a cage between the upper and lower nerve roots without sacrificing a root is narrow. However, complications involving cage insertion or expansion, such as dural tear, can be prevented by adequate use of a retractor. Cage placement requires oblique insertion, which alters the orientation of the inter nerve root corridor with rotation of cage being done once it is ventral to the neural elements.
We used local autograft for filling the mesh cage, thus conferring osteoconductive as well as osteoinductive properties. The cages confer resistance to axial compression, rotation and lateral flexion. A potential downside of this technique is the limited opportunity to insert cages exceeding a diameter of 20 mm, which is small compared to vertebral body end plate footprint in the lumbar spine. The lumbosacral junction, transitional zone between mobile lumbar spine and relatively fixed sacrum, subjects implants to higher loads and greater stress with a high rate of pseudoarthrosis.17,18 However, when combined with the rigid posterior fixation, a titanium mesh cage supporting the anterior column provides good fixation is able to tolerate maximum loads over the implants till the bony fusion occurs. This may explain the tilting of cage and screw pull out in our series using short segment stabilization at lumbosacral junction.
Instead of using an expandable cage as Ayberk et al 19 and Sasani and Ozer 20 described, we used non-expandable cage to reconstruct anterior and middle column. Previous investigations have failed to demonstrate any significant difference in biomechanical properties of expandable and non-expandable cages.21,22 Our overall clinical and radiographic results were promising. Although a small fusion bed and possible implant failure could be drawbacks to the use of smaller cage compared to lumbar endplate footprint, we placed bone chips surrounding the cage, and the lack of fusion failure in our series demonstrates the relatively good stability and safety of the construct. Our experience demonstrates the feasibility of using a single posterior approach to insert mesh cage that is not eccentrically located without sacrificing the nerve roots for unstable LBF. In our opinion, the key to performing this operative technique is wide dissection and thorough mobilization of the lumbar spinal nerve roots to create a corridor for cage insertion, as it allows for less traumatic insertion using narrower inserters.
We encountered 2 cases of PJK in the present study, one of which was managed by extending fixation to a proximal level. There were 4 patients with dural leak of which 3 could be repaired. One patient had a post-traumatic leak located anteriorly which couldn’t be sutured. However, the CSF leak subsided with time. We had 1 patient with dehiscence of the wound who required debridement and suturing under local anesthesia. 1 patient had a tilted cage on follow-up. However, there was no further tilting on serial radiographs during follow-up. The cage fused in position and patient had no functional problems; hence a revision was not considered. One patient had neuropraxia which completely recovered during postoperative follow-up. In this study, the average blood loss was 1280 ml, which is less than that reported in combined approaches, which reached 3.2 L in some studies. 14 In general, the posterior approach is a simpler technique that is familiar to most spine surgeons, carries little risk for vascular or visceral injury, and minimizes recovery time. Also, the single-stage posterior approach allows for effective decompression of neural elements, anterior distraction, pedicle screw stabilization and fusion.
Our study is not without limitations as it is retrospective in nature with a prospective follow-up. The absence of a control group and small sample size due to lower incidence of these injuries is a major limitation. A short follow-up duration, no evaluation of other outcome measures and absence of post-operative CT scan at 2-year follow-up to evaluate fusion and canal clearance due to economic constraints are further shortcomings of this study. The main goals of surgery in cases of LBF are to decompress neural structures and provide spinal stability for immediate rehabilitation. Currently, any procedure that meets these 2 aims with minimum complications is to be preferred over other methods. We achieved good results with an all posterior approach in 45 patients of LBF, which is the largest series of this nature in current literature. However, our study is by no means suggesting that the single-stage posterior corpectomy and circumferential reconstruction should be the preferred treatment over other techniques; it is merely one of the surgical options.
Conclusion
The posterior-only approach for corpectomy is safe, efficient, and provides rigid posterior stabilization, 360° neural decompression, and anterior reconstruction obviating the need for the anterior approach.
Footnotes
Acknowledgments
We express special thanks of gratitude for providing English language editing services and grammatical corrections to 1. Dr. Brett Rocos, MD, FRCS Spine services, University of Toronto. 2. Dr. Tarush Rustagi. MD. Department of Neurosurgery, Ohio State University 3. Dr. Anuradha Mohapatra, “Quick-Stats solutions,” which is a professional agency providing English language editing services for medical research (email-
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
