Abstract
Study Design:
Retrospective cohort study.
Objective:
Complication profiles for lateral approaches to the spine are well established. However, the influence of level of surgery on complication rates and subtypes are less well established. To determine risk factors for complications as determined by level and surgery type in patients undergoing a lateral (retroperitoneal or retropleural approach) to the thoracolumbar spine.
Methods:
All adult patients undergoing a lateral thoracolumbar fusion with or without posterior instrumentation performed at a single institution were identified. Primary outcomes assessed were presence of complication, complication subtype, and need for reoperation. The primary independent variables were spinal level (thoracic, thoracolumbar, or lumbar) and type of surgery (discectomy or corpectomy). Categorical outcomes were compared using chi-square test. Unadjusted and adjusted odds ratios for corpectomy status were calculated to determine risk of complication by level. P < .05 was considered statistically significant.
Results:
A total of 165 patients aged 18 to 75 years were identified as having undergone a lateral fusion. Complication rates were 28.6%, 36.4%, and 11% for thoracic, thoracolumbar, and lumbar lateral approach fusions, respectively. Under univariate analysis, patients undergoing lateral approach in the thoracic spine group had significantly higher rates of postoperative complications than those in the lumbar group (P = .005). After adjusting for corpectomy status, there was no difference in complication rates.
Conclusions:
Lateral (retroperitoneal or retropleural) approaches to the thoracic and thoracolumbar spine may be used with complication rates comparable to well-established lumbar approaches. Extent of surgery (corpectomy vs discectomy) rather than level of surgery may represent the primary driver of complications.
Introduction
The thoracolumbar spine is a common location for traumatic and nontraumatic pathologies and multiple approaches have been described, ranging from open approaches to more minimally invasive techniques. 1 -3 Retroperitoneal and retropleural approaches were first described in 1925 by Fey 4 and 21 years later by Francioli 5 for sympathectomy. Moskovich et al 6 later described the lateral (retroperitoneal and retropleural) approach to the thoracolumbar junction and thoracic spine. While advances in minimally invasive spine surgery have popularized the lateral transpsoas lumbar interbody fusion (LTIF) through a retroperitoneal approach at the lumbar spine, anatomical differences at different levels of the spine affect the complication profiles of these approaches and have created polarizing opinions about its safety. This is true especially at the thoracolumbar junction (TLJ) given the unique anatomical challenges with the diaphragm and convergence of the retroperitoneal and retropleural space. 7,8
Several authors have mentioned the lateral approach to the TLJ and thoracic spine using the same expandable retractor system as used in the lumbar spine in different pathologies. 3,9,10 Although minimally invasive lateral approaches to the thoracic, thoracolumbar, and lumbar spine are unique with respect to anatomical dissection, surgical principles and techniques with regard to retractor systems, discectomies, and fusion techniques are analogous. The advantages of this approach are similar to those of the more established lateral transpsoas approach (LTPA), which include direct visualization of disc space with more extensive end plate preparation, larger interbody devices that mitigate the likelihood of subsidence, greater restoration of disc height and indirect decompression, and an ability to avoid the thecal sac, mitigating the risk of cerebrospinal fluid (CSF) leak. 11 -15 As the use of far lateral procedures continue to increase, it is important for surgeons to remain aware of procedural complications. The incidence of neurological complications is well known and has been published and described extensively in the lumbar spine but is less commonly reported regarding the thoracolumbar junction and thoracic spine. 16 -25
This study analyzes the complications of patients undergoing fusion through a minimally invasive lateral approach (retroperitoneal or retropleural) in the lumbar, thoracolumbar and thoracic spine and compares the complications associated with the lateral approach in these three spinal regions.
Methods
Study Design
All adult patients age 18 to 75 from 2006 to 2016, who underwent a lateral arthrodesis of the thoracic or lumbar spine using lateral approach technique at a single institution were included. All patients had a minimum follow-up of 6 months. The median follow-up was 9.6 months (range 6-14.2 months) patients with missing data or were lost to follow-up were excluded. Patients were separated into 3 groups based on spinal level: thoracic spine (T10 and above), TLJ (T11-L2), and lumbar (L2 to L5) groups. Demographic information was collected, including age, gender, presenting symptoms, body mass index, smoking status, comorbidities. Surgical details were evaluated including number of surgical levels, and presence of an indication for corpectomy. Postoperative improvement in radiculopathy was documented. Complications were assessed, including the incidence of postoperative nerve root palsies and other surgical complications. A major complication was defined as any conditions necessitating re-operation or which might cause significant morbidity and mortality. When present, reoperation reasons and postoperative complications were recorded. Neurological complication was defined as any new neurological compromise (motor or sensory) persisting beyond 6 months.
Surgical Technique
The lateral approach at the TLJ and the thoracic spine were both performed using the standard minimally invasive lateral spine access retractors as is used for the transpsoas lumbar approach (MIS Lateral Platform Depuy Synthes). After positioning the patient in a lateral decubitus position and confirming the surgical level, a 4- to 6-cm oblique incision is made. The rib overlying the affected level is located, which is usually the 10th rib for a T12 target level, 11th rib for L1, and the 12th rib for the L2 level. The intercostal space above the desired rib is dissected carefully exposing the superior aspect of the rib. The periosteum is incised and elevated. Further separation of the periosteum is performed with the aid of an Alexander or Cobb elevator. The intercostal bundle is identified and protected. After complete separation from the underlying pleura the rib is partially removed using a rib cutter (Figure 1). Depending on the planned procedure, 4 to 6 cm of rib is removed, which can then be used as graft material. Then a blunt dissection is carried out between the endothoracic fascia and the parietal pleura and further advanced with the aid of a finger down to the spine. Care must be taken while bluntly deflecting the pleura from the inner thoracic wall down to the thoracic spine especially in cases where there is scar tissue. Once retropleural access to the thoracic spine is achieved the retractors are placed while avoiding tension on the intercostal bundle. In the event of a corpectomy, ventral reconstruction is performed using expandable titanium cages and bone autograft. Spinal instrumentation was completed by posterior pedicle screw instrumentation.
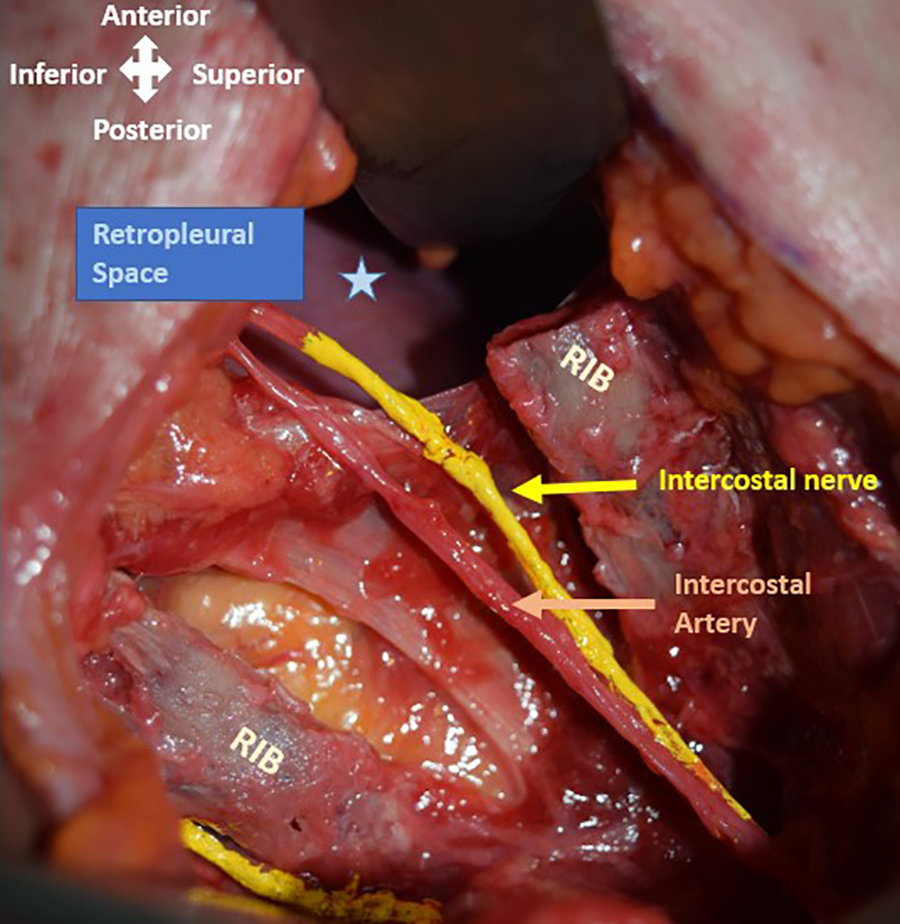
Cadaveric specimen: Lateral view of into retropleural space after exposure and rib resection. Neurovascular bundle exposed (yellow arrow—intercostal nerve; orange arrow—intercostal artery; blue star—retropleural space).
Statistical Analysis
Descriptive statistics were used to describe demographic and surgical characteristics. For categorical variables, frequency counts were computed and presented along with their percentages. For continuous variables, means were computed and presented along with their standard deviation. Complications were analyzed using chi-square test. Unadjusted odds ratios (ORs) and adjusted odds ratios (aORs) and their 95% confidence intervals were calculated using logistic regression to compare the proportion of individuals experiencing one or more complications by level of surgery. Since corpectomy is a known risk factor for complications, 26 and since the frequency of corpectomy was unequally distributed across surgical levels, we computed adjusted odds ratios controlling for corpectomy. Analyses were performed using Stata software, version 9.0 (College Station, TX, USA).
Results
Demographics
A total of 165 patients were included. The mean age was 66.0 ± 10.4 years. The gender ratio was equal with 50.0% male and 50.0% female patients. The thoracic group had 7 patients, the TLJ group had 22 patients, and the lumbar group had 136 patients, Demographics and preoperative symptoms are summarized in Table 1.
Patient Demographics and Preoperative Symptoms.
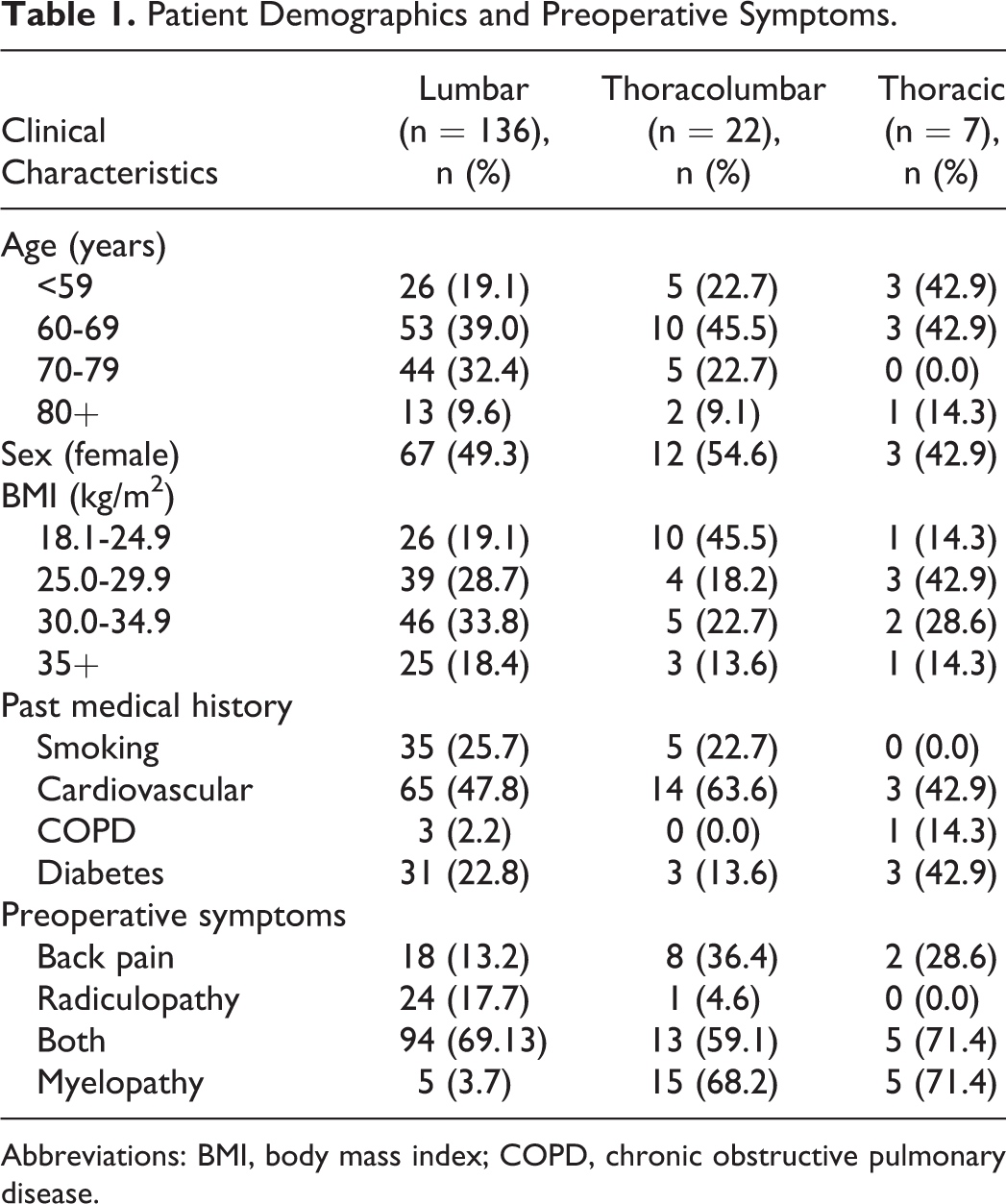
Abbreviations: BMI, body mass index; COPD, chronic obstructive pulmonary disease.
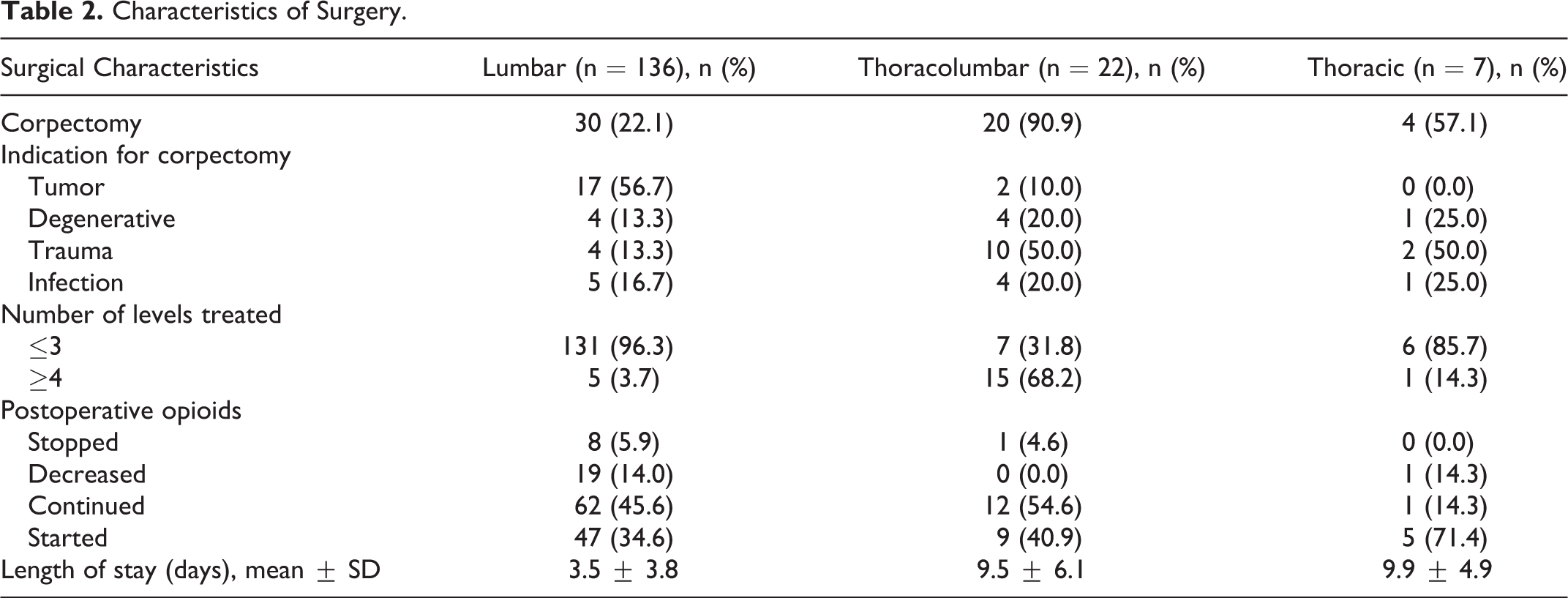
Characteristics of Surgery
In total, 54 corpectomies were done, the indications for which varied by location. In the lumbar spine, the majority of corpectomies were done for tumor resection (Figure 2) while the leading indication in the TLJ (10 out of 20) and the thoracic spine (2 out of 4) was trauma (Figure 3). While only 20% of lumbar approaches included corpectomies, a greater proportion of the TLJ and thoracic approaches were done with corpectomies (90.9% and 57.1%, respectively). Furthermore, the number of levels treated was four or more in 68.2% at the TLJ while 14.3% in the thoracic spine and 3.7% in the lumbar spine. Surgical characteristics are summarized in Table 2.
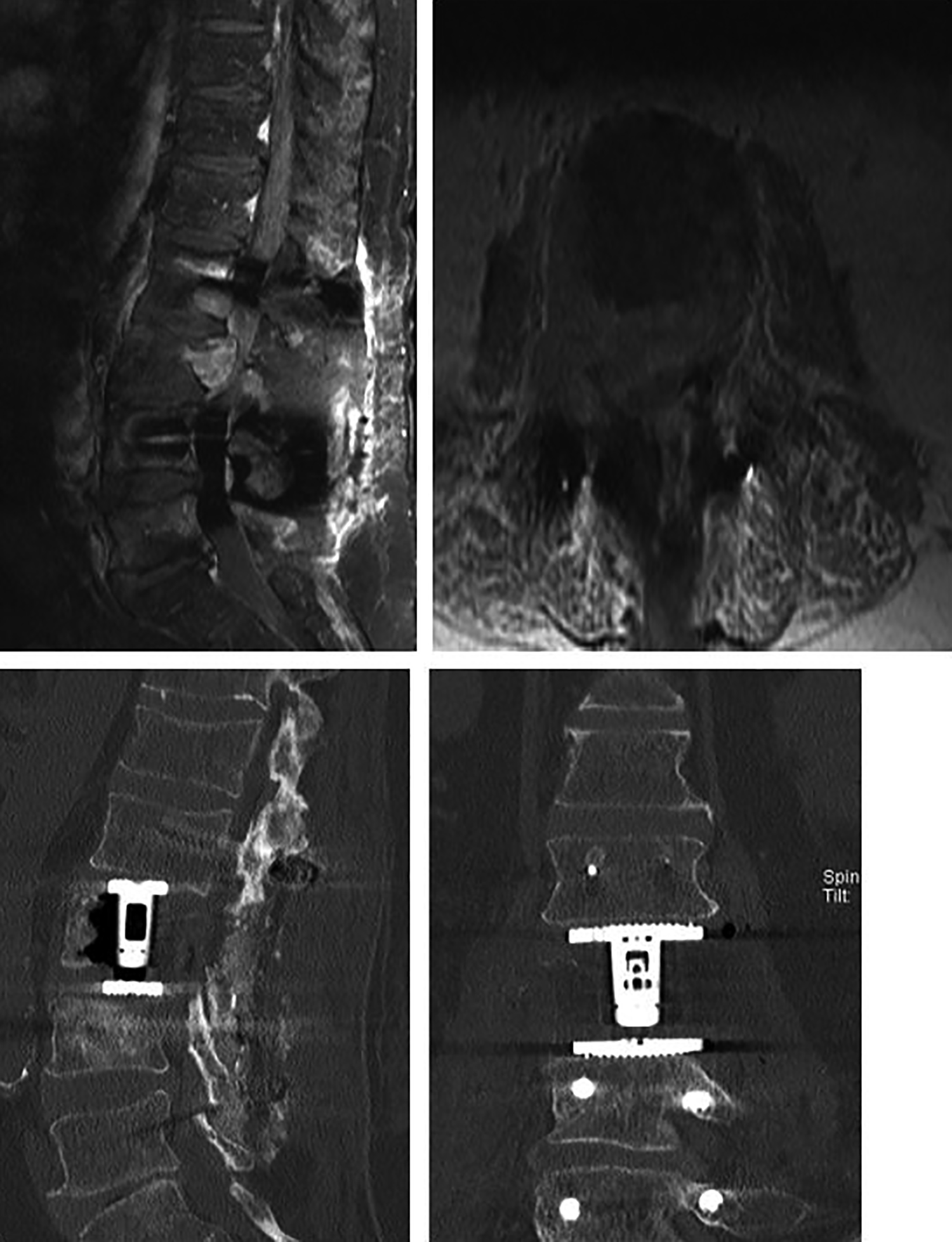
Patient X with worsening low back and radicular pain with L3 metastasis and retropulsion underwent L3 lateral corpectomy and posterior stabilization. (Top panel) Magnetic resonance imaging (sagittal and axial views): preoperative images. Computed tomography lumbar (sagittal and coronal views): postoperative images.
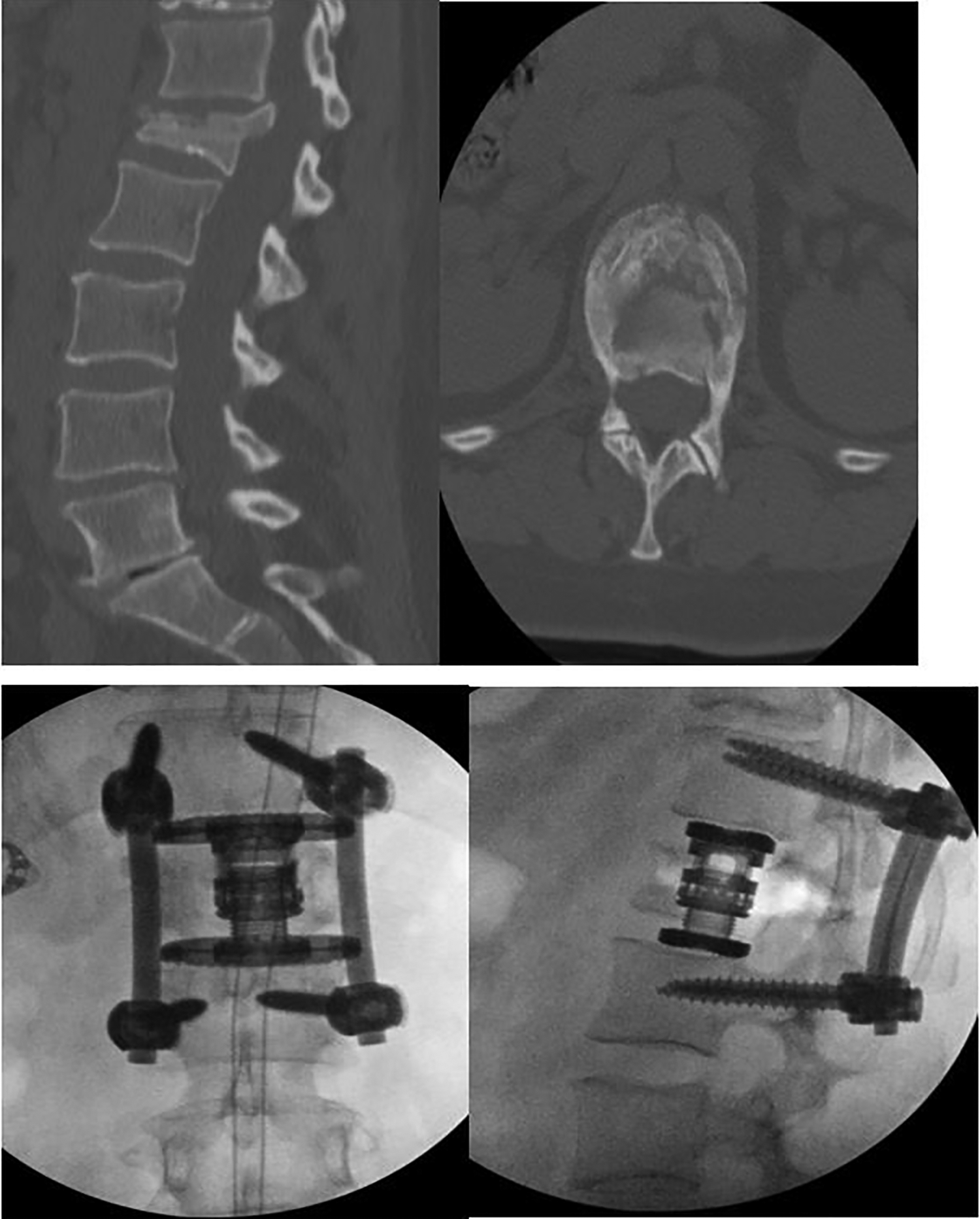
Patient Y with L1 burst fracture who underwent lateral L1 corpectomy with interbody cage placement and posterior T12-L2 decompression and fusion. (Top panel) Computed tomography (sagittal and axial views): preoperative images. (Bottom panel) Lateral and anterior/posterior lumbar radiographs: postoperative films.
Characteristics of Surgery.
Complications
There were significantly more overall postoperative complications with lateral approach in the thoracic spine compared with the lumbar spine (P = .005) (Table 3). However, when the odds ratio was adjusted for corpectomy, there was no difference in complication rates amongst the 3 groups (Table 4). We encountered 3 pneumothoraces, all at the TLJ (P = .003). Neurological complications and reoperations can be found in Table 3. We found a significant difference in infection when comparing the 3 groups, with the highest infection rate in the TLJ group (P = .015). There was also a significant difference frequency of hardware complications, with the highest rate of hardware complication in the thoracic spine. The reoperation did not differ significantly in the 3 spinal regions (P = .581).
Complications in Lumbar, Thoracolumbar, and Thoracic Regions.
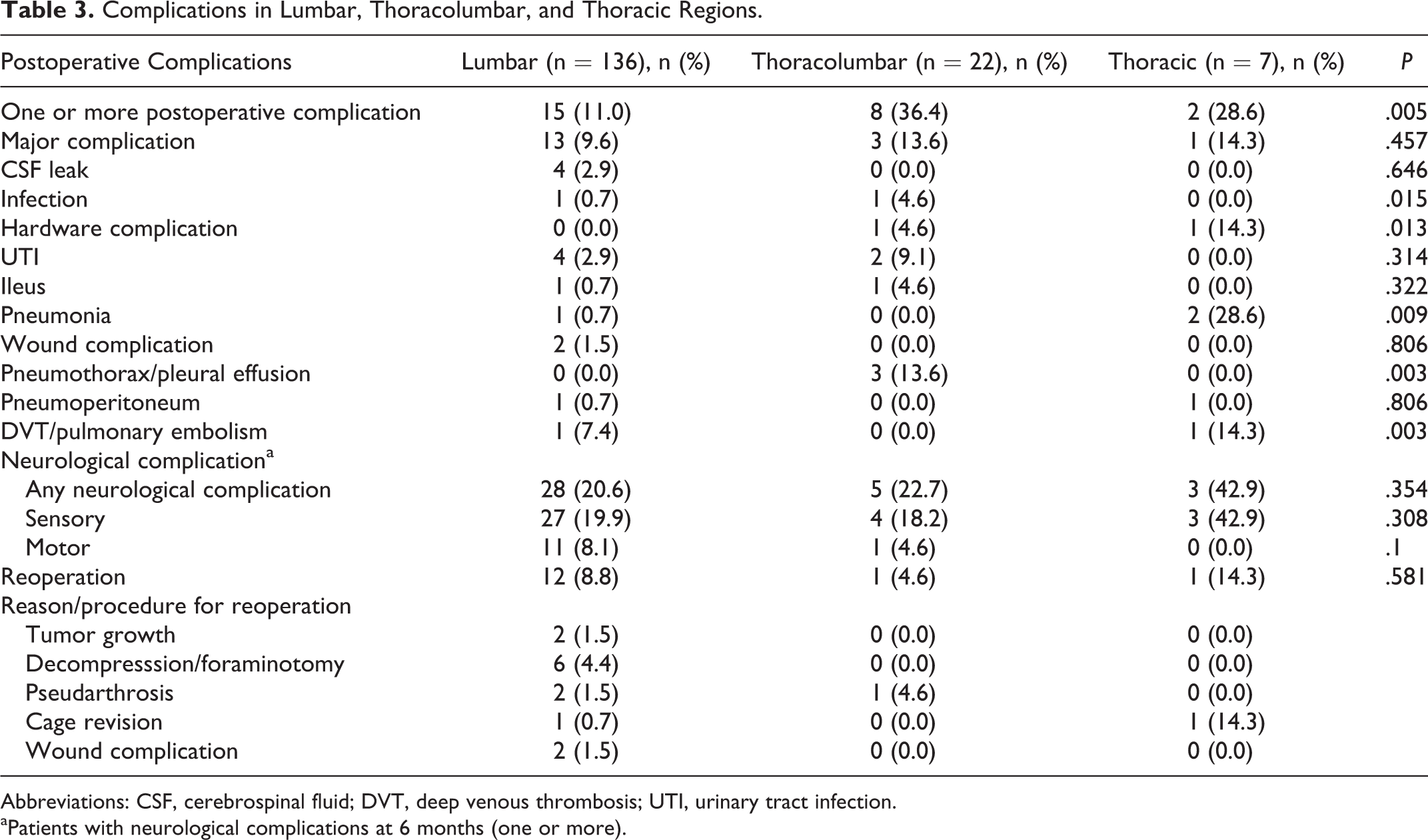
Abbreviations: CSF, cerebrospinal fluid; DVT, deep venous thrombosis; UTI, urinary tract infection.
aPatients with neurological complications at 6 months (one or more).
Univariate Analysis for Postoperative Complication Among Lumbar, Thoracolumbar Junction, and Thoracic Approaches.

Abbreviations: aOR, adjusted odds ratio for corpectomy; CI, confidence interval; OR, odds ratio; TLJ, thoracolumbar junction.
Discussion
The aim of the study was to differentiate the risk profile of the lateral approach to the spine at different regions. It was found that the highest rate of neurological complications (42.9%) (sensory only) and instrumentation failures (14.3%) after 6 months occurred in the thoracic spine region. The highest rate of pleural injury (13.6%) was present at the TLJ.
Instrumentation failure rates, which were 4.6% in the TLJ and 14.3% in the thoracic spine, were also comparable to previous studies. Karikari et al 10 had one case of subsidence of the graft in 22 patients who underwent lateral surgery for thoracic and thoracolumbar spine disease (4.5%), and Meridith et al 27 reported 2 in 18 (11.1%) instrumentation failures (Figure 4 and Figure 5).
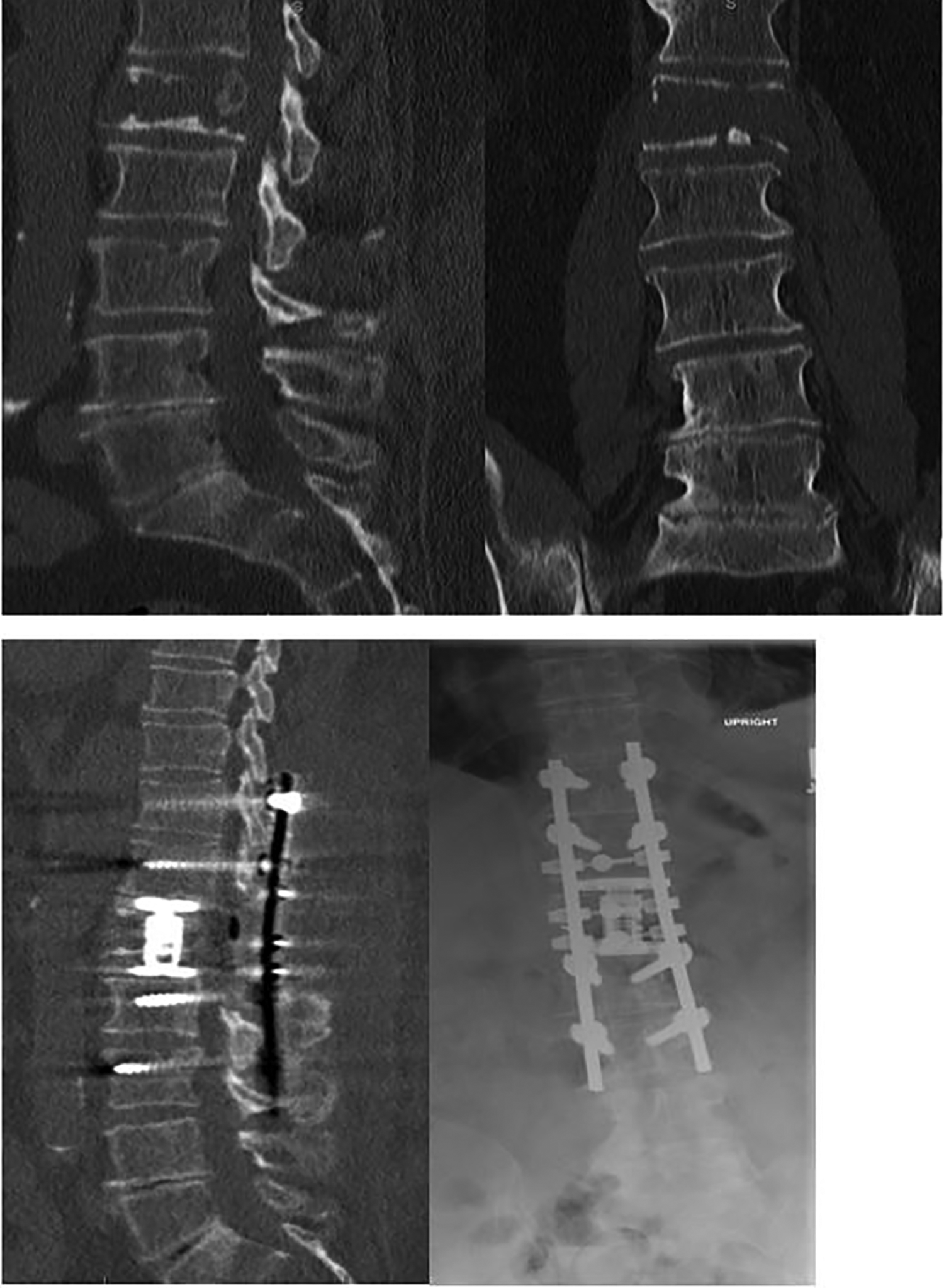
Patient Z with intractable back pain found to have L1 osteomyelitis who underwent lateral L1 corpectomy with posterior decompression and fusion. (Top panel) Computed tomography (sagittal and axial views): preoperative images. (Bottom panel) Lateral and anterior/posterior lumbar radiographs: postoperative films.
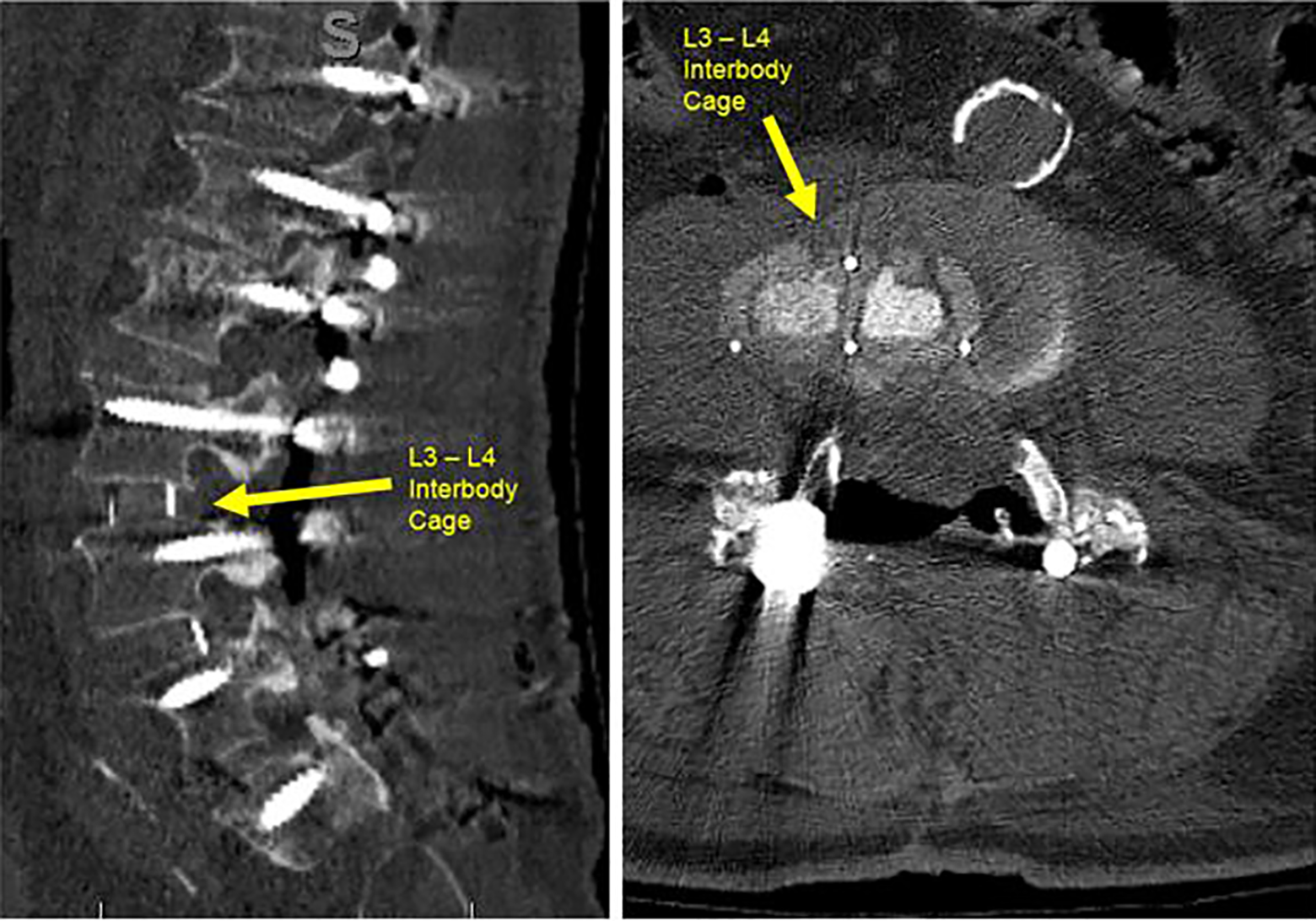
Patient W with leg pain and kyphoscoliosis who underwent staged L2-L5 lateral interbody fusion with posterior T10-pelvic fusion, then subsequent revision L3-L4 lateral interbody fusion due to L3-L4 cage migration between staged procedures. Computed tomography lumbar images (sagittal and axial views) of cage displacement eccentric to the right side (yellow arrow).
Although the risk of complications was higher in the thoracic and thoracolumbar region compared with the lumbar spine, after adjustment for corpectomy there was no difference in risk among the 3 groups. Therefore, the invasiveness of the procedure, which is greatly increased with corpectomy, is an important factor for risk of complication.
Compared to open approaches, the application of lateral approach in the thoracic and thoracolumbar spine has many advantages, including decreased blood loss, decreased length of hospital stay, chest tube duration, and lower complication rates. 13,28 -30 Lateral approach avoids the prolonged lung deflation that is required in wide-open techniques, and may lower the risk of pulmonary complications, especially in patients with preexisting pulmonary disease. 27 Nevertheless, the retropleural approaches require intimate navigation of the critical respiratory structures, and patients are at risk of experiencing pneumothorax, diaphragm injury, rib pain, and pleural effusion. 31
In our series, 3 of the TLJ patients had pneumothorax, in contrast to 0% in the lumbar and thoracic spine (Table 3). The combined thoracic and TLJ rates of pleural effusion, pneumothorax, and hemothorax after lateral approach was 10.3%, which was comparable to rates reported in the literature, with most ranging from 0% to 10.2%. 10,32,33 One explanation for our rate of 10.3% compared with those in the study by Kasliwal et al, 33 which had no pleural complications, is that 57% of thoracic patients and 90% of TLJ patients underwent corpectomy compared with 0% of the patients in Kasliwal et al.
Patients undergoing lateral approaches to the spine at the TLJ may also experience postoperative neuropathy. In a retrospective review of 20 patients who underwent minimally invasive corpectomy at the TLJ for traumatic burst fractures, 5 patients had L3 distribution pain lasting at least 5 months. 34 In another study that evaluated patients who underwent XLIF (extreme lateral interbody fusion) in the lumbar and TLJ region, 4 patients experienced transient L4 weakness while 3 had transient L4 hypoesthesia. 35 While none of the patients in the thoracic spine group had a post-operative motor deficit in the present study, 11 (8.1%) of the lumbar and 1 (4.6%) of the TLJ patients had postoperative sensory deficits. There was no significant difference in neurological complications among the 3 spinal regions. Although the intercostal nerve is not compromised during the approach, tension caused by retractor placement may contribute to the high number of postoperative sensory deficits.
Although our series had a high percentage of preexisting conditions, with 63.6% cardiovascular diseases in the TLJ group, 42.9% in the thoracic group, overall we had comparable complication rates even to studies in which patients were younger and healthier, with mean age of 33 years in Li et al 36 and mean age of 45.9 years in Yu et al. 34 Therefore, elderly patients and patients with multiple comorbidities who require thoracic interbody fusion may benefit from the minimally invasive extreme lateral approach.
The mean length of stay (LOS) in our thoracic and TLJ patients was 9.5 and 9.9 days, respectively. While this is longer than those reported in patients who underwent thoracic XLIF for thoracic discectomy (2.6-5.3 days), 30,33 it was comparable to a study by Yu et al 34 in which patients underwent lateral approaches and corpectomy in the thoracic spine (8.7 days). Yu et al point out that one reason for the discrepancy in LOS in patients undergoing XLIF in the thoracic spine may be posterior instrumentation, which leads to a greater operative time, estimated blood loss (EBL), and therefore a longer LOS. 34 While Yu et al 34 and all of our patients underwent posterior instrumentation, studies that reported significantly shorter LOS did not. Furthermore, patients in the thoracic group and 5 patients in the TLJ group had much longer LOS due to social reasons that were unrelated to health status.
Manipulation of the diaphragm is usually necessary when accessing the thoracolumbar junction during a lateral procedure. Therefore, an understanding of the anatomy of the diaphragm and its attachments are crucial to avoid diaphragmatic injury. In a cadaveric study investigating diaphragm attachments relevant to lateral procedures, Sun et al 37 showed that the attachments are typically between the inferior edge of the 10th rib and the superior edge of the 12th rib. Based on these attachments, the authors recommend placing the incision above the 10th rib for the retropleural approach and below the 12th rib for the retroperitoneal approach to avoid injury to the diaphragm. 37 In a retropleural approach, the posterior attachments of the diaphragm can be swept inferiorly using monopolar cautery.
Limitations
We recognize the limitations in our study. First, the populations were heterogeneous, in large part due to the indications for surgery. Second, our study sample was small limiting our ability to control for potential confounding variables. Nevertheless, the ability to control for corpectomy given the frequency of this procedure was accomplished. Given the retrospective nature of the study, we were limited in our ability to describe the patient and surgical characteristics by those variables present for analysis. Future studies may include sample sizes with validated outcome measures with prospectively collected data.
Conclusion
Patients who receive lateral (retroperitoneal or retropleural) approaches in the lumbar, thoracic and TLJ are different form each other. Nevertheless, their complications are likely a result of surgical factors and regional anatomy as well as the relationship of the diaphragm to the chest cavity with larger procedures. While surgical risk may be increased with more invasive procedures like corpectomy, the risk does not differ significantly in the various levels of the spine. Lateral approach in the thoracic and TLJ may be beneficial in patients who are elderly or have multiple comorbidities.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
