Abstract
Study Design
Retrospective cohort study.
Objectives
This study aimed to evaluate the survival period in patients with a single spinal metastasis (SSM), subsequently comparing those with isolated-single spinal metastasis (I-SSM) and single spinal metastasis with other metastasis (O-SSM) after surgery, and to identify prognostic factors affecting their survival.
Methods
A total of 135 patients were included, with 24 patients in the I-SSM group and 111 in the O-SSM group. Survival analysis was utilized to assess the survival of SSM patients, followed by a comparison of survival rates between the two groups. Univariate and multivariate analyses were conducted to identify significant prognostic factors for survival.
Results
The overall median survival period for patients with single spinal metastasis (SSM) was 10.2 ± 1.8 months. Specifically, the median survival was 15.7 ± 5.7 months in the I-SSM group and 10.2 ± 1.5 months in the O-SSM group. The difference in survival periods between the two groups was not statistically significant (
Conclusions
Patients with SSM have a median survival of 10.2 months, with no significant difference in postoperative survival between the I-SSM and O-SSM groups. Significant prognostic factors influencing the survival period after surgery include preoperative KPS, postoperative ambulatory status, and the primary malignancy site.
Keywords
Introduction
The spine is the most common site of bone metastasis, with nearly one-third of all bone metastases occurring in the spine.1,2 Spinal metastasis frequently leads to deterioration of spinal structure and cord compression, necessitating appropriate treatment modality.3,4 Despite remarkable advancements in immunotherapy, targeted therapy, and radiotherapy in recent decades, surgery still plays a critical role in the treatment of metastatic spinal tumors, especially in situations of spinal instability and cord compression.5-9
When determining the appropriate surgical approach for spinal metastasis, life expectancy is a significant consideration.9-11 In previous studies, Tokuhashi et al, 12 Tomita et al, 10 and Sioutos et al. 13 have introduced a scoring system to predict the survival period for patients with spinal metastases. In this scoring system, extensive tumor involvement suggests a larger tumor burden and generally results in a shorter anticipated lifespan. 14 Therefore, even if in single spinal metastasis (SSM), if there are multiple metastases to other organs, palliative surgery tends to be performed because it is believed to have a relatively poor prognosis with a comparatively short life expectancy.2,12,15 Conversely, an isolated SSM without involvement of other organs may potentially warrant more aggressive surgical interventions such as total en-bloc spondylectomy, given the possibility of a more favorable prognosis.16-18 However, current research on survival outcomes in patients with SSM is still limited, and it is not well understood whether there exists a difference in survival between patients with isolated-single spinal metastases (I-SSM) and those with SSM involving metastases to other organs (O-SSM).
Therefore, the purpose of this study is to evaluate postoperative survival in patients with SSM, and subsequently compare I-SSM to O-SSM groups, and to identify prognostic factors that exert significant influence on the survival of patients with SSM after surgery.
Materials and Methods
Study Design
The study was approved by the Institutional Review Board of Samsung Medical Center (IRB No. 2023-12-120), and informed consent was waived due to the retrospective nature of the data. We reviewed the medical records of patients who underwent surgery for SSM at our institution between September 2015 and December 2021. The indications for surgical treatment were carefully determined through a multilateral, inter-department conference based on the following criteria: 1) refractory pain despite conservative treatment, and 2) neurological deterioration or the potential for neurological deficits with spinal column instability.
The inclusion criteria in this study cohort were as follows: 1) confirmed diagnosis of SSM based on radiological findings; 2) meeting the criteria for surgery; 3) patients who have been consecutively treated at our institution and followed up for at least 3 months. Meanwhile, patients with multiple spinal metastases, those with life expectancies of less than 3 months, and those with poor medical conditions deemed unable to tolerate surgery by three spine surgeons were excluded.
In this study, SSM was confirmed by radiology specialists at our institution following comprehensive imaging studies, including whole-spine magnetic resonance imaging (MRI), computed tomography (CT) of the chest, abdomen, and pelvis, and whole-body PET-CT. SSM is defined as a primary tumor found elsewhere in the body, not originating in the spine, with metastasis to one vertebral body in the whole spine. I-SSM refers to cases where there is no metastasis to other organs, and metastasis occurs in only one vertebra throughout the whole spine. On the other hand, O-SSM refers to cases where there is metastasis to one vertebral body in the whole spine, along with metastases to other internal organs.
Patients with SSM were categorized into I-SSM and O-SSM groups. Baseline data and potential survival-related variables were compared between these groups to identify any significant differences. Additionally, among patients who were ambulatory postoperatively, we compared I-SSM and O-SSM group patients based on whether they received chemotherapy or not. Survival analysis was performed to assess the overall survival of SSM patients and to determine if there was a difference in survival rates between the two groups. Furthermore, prognostic factors for SSM (including both I-SSM and O-SSM) were investigated to identify variables significantly influencing survival.
Baseline Data and Presumed Survival-Related Factors
This study evaluated various presumed survival-related factors as follows: 1) neurological deficits (Frankel grade) which categorized patients into 3 subgroups (A-B, C-D, and E),19,20 2) preoperative Karnofsky Performance Scale (KPS), scores were grouped into three categories (10-40, 50-70, and 80-100),6,21 3) pre- and postoperative ambulatory status, 22 4) primary malignancy site factor using Tomita’s scoring system categorized into Groups A (slow growth), Group B (moderate growth), and Group C (rapid growth), 10 5) four surgical types: Type 1, fixation only; Type 2, laminectomy and fixation; Type 3, partial corpectomy and fixation; and Type 4, total corpectomy and total en-bloc spondylectomy, 6) modified Tokuhashi score, 7) the presence of major internal organ metastases, and the presence of other bone metastasis, 8) adjuvant chemotherapy and radiation therapy before and after surgery.
Statistical Analysis
To compare the presumed risk factors between I-SSM and O-SSM, the chi-squared test and Fisher’s exact test were conducted, and survival differences between the two groups were assessed using Kaplan-Meier survival analysis. To investigate significant survival-related factors in patients with single spinal metastasis, a univariate analysis using the Log-rank test was conducted. Following this, a multivariate analysis was performed using the Cox proportional hazards model. All statistical evaluations were executed using SPSS software (version 22.0.0; IBM Corp.), with a threshold of
Results
Baseline Data and Presumed Survival-Related Factors
Comparison of the Baseline Data and Presumed Survival-Related Factors Between I-SSM and O-SSM Groups.
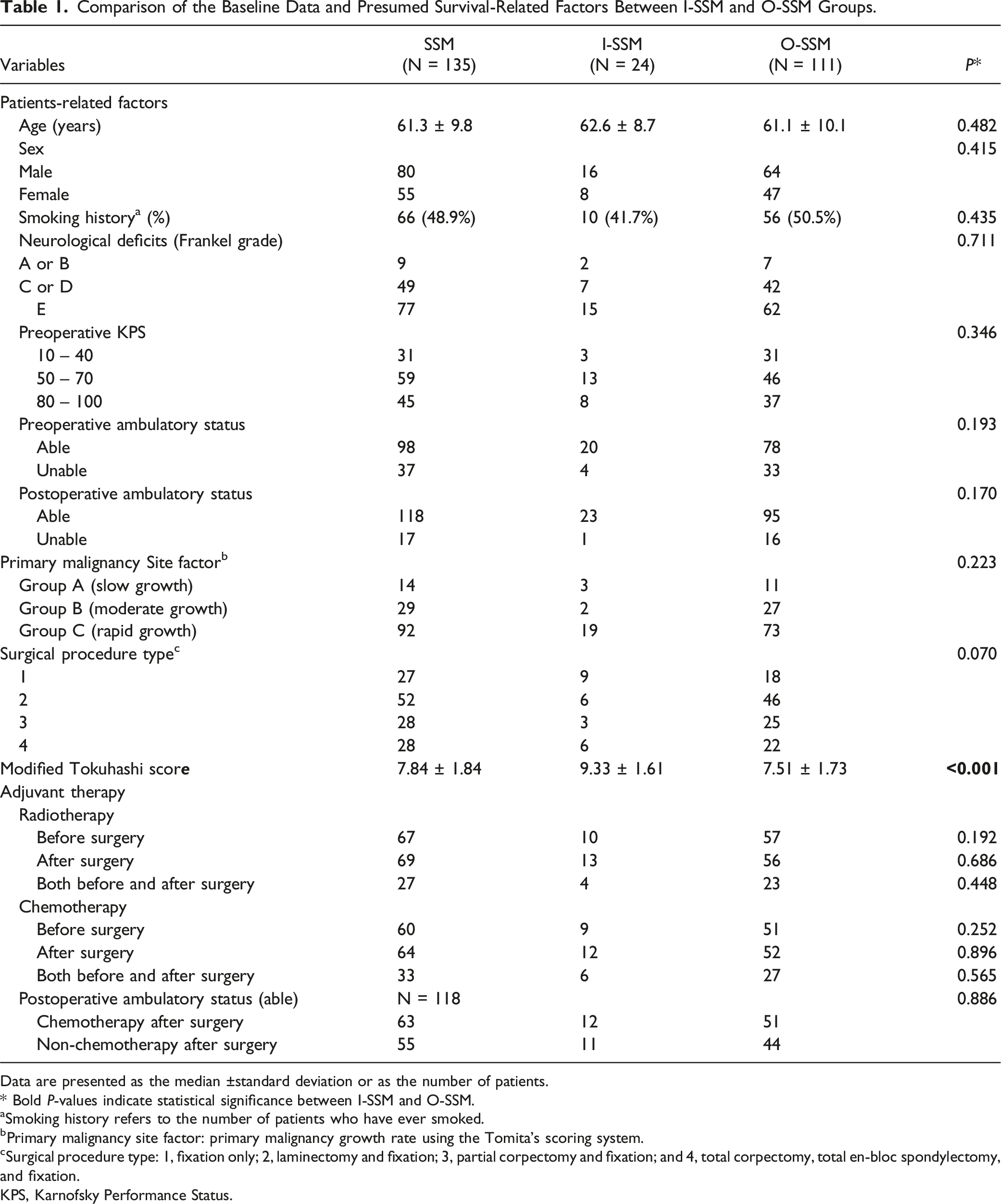
Data are presented as the median ±standard deviation or as the number of patients.
* Bold
aSmoking history refers to the number of patients who have ever smoked.
bPrimary malignancy site factor: primary malignancy growth rate using the Tomita’s scoring system.
cSurgical procedure type: 1, fixation only; 2, laminectomy and fixation; 3, partial corpectomy and fixation; and 4, total corpectomy, total en-bloc spondylectomy, and fixation.
KPS, Karnofsky Performance Status.
Overall Survival of Patients with SSM and Within Groups Between I-SSM and O-SSM
Kaplan-Meier survival analysis revealed a median survival of 10.2 ± 1.8 months (95% confidence interval [CI] 6.8 to 13.6) and a mean survival of 23.3 ± 2.6 months (95% CI 18.2 to 28.4) for all patients with SSM. The overall cumulative survival rate at 12 months was 48.2%, and it further declined to 29.9% at 24 months (Figure 1). Kaplan-Meier survival curve in patients with SSM.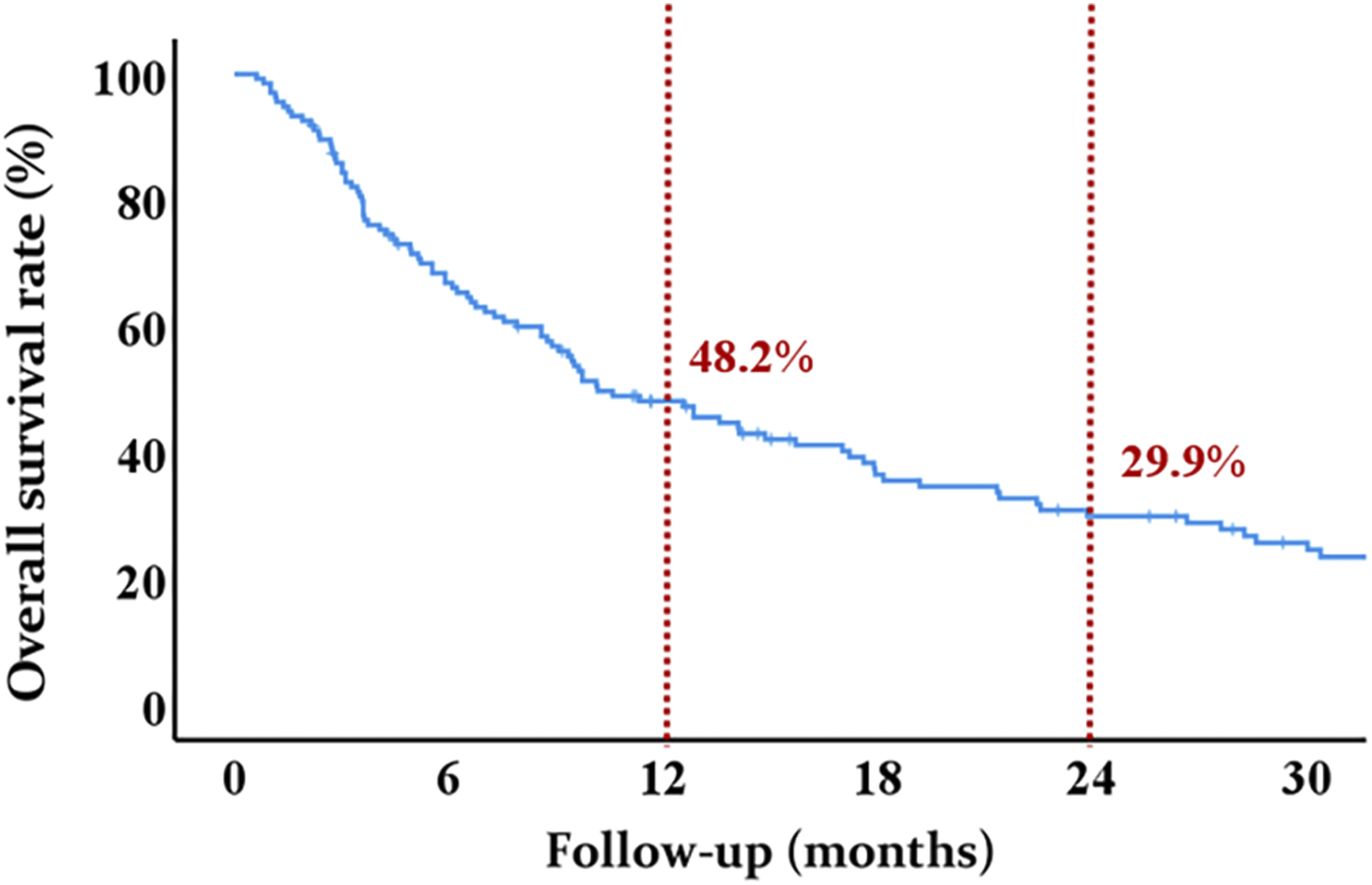
Survival Analysis of Patients With SSM and Comparison Between Patients With I-SSM and Those With O-SSM.
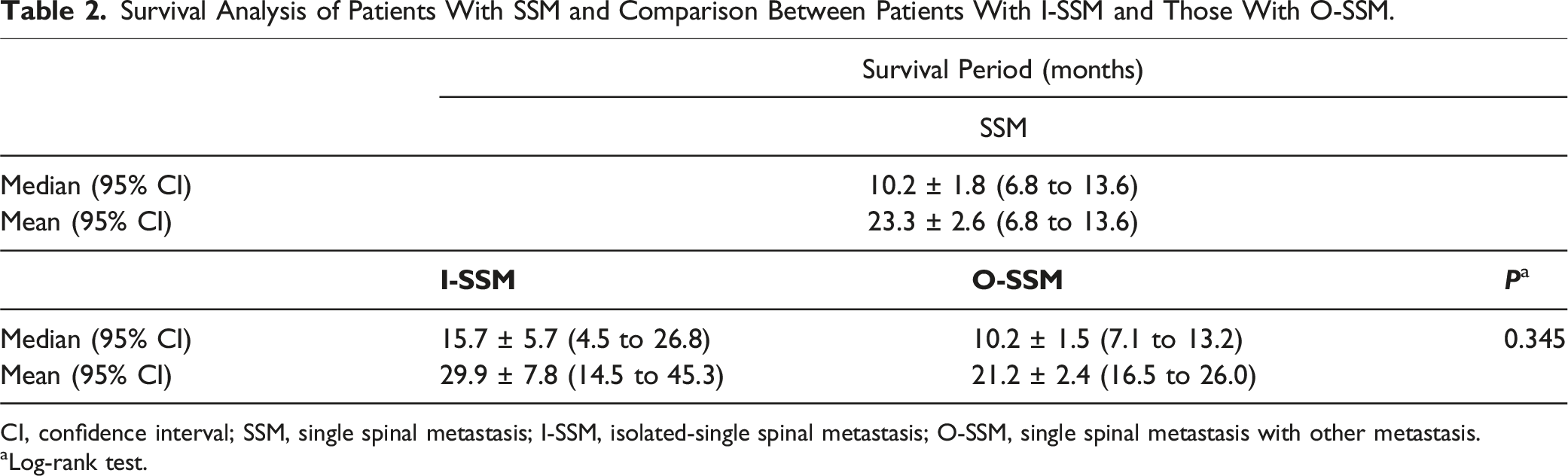
CI, confidence interval; SSM, single spinal metastasis; I-SSM, isolated-single spinal metastasis; O-SSM, single spinal metastasis with other metastasis.
aLog-rank test.
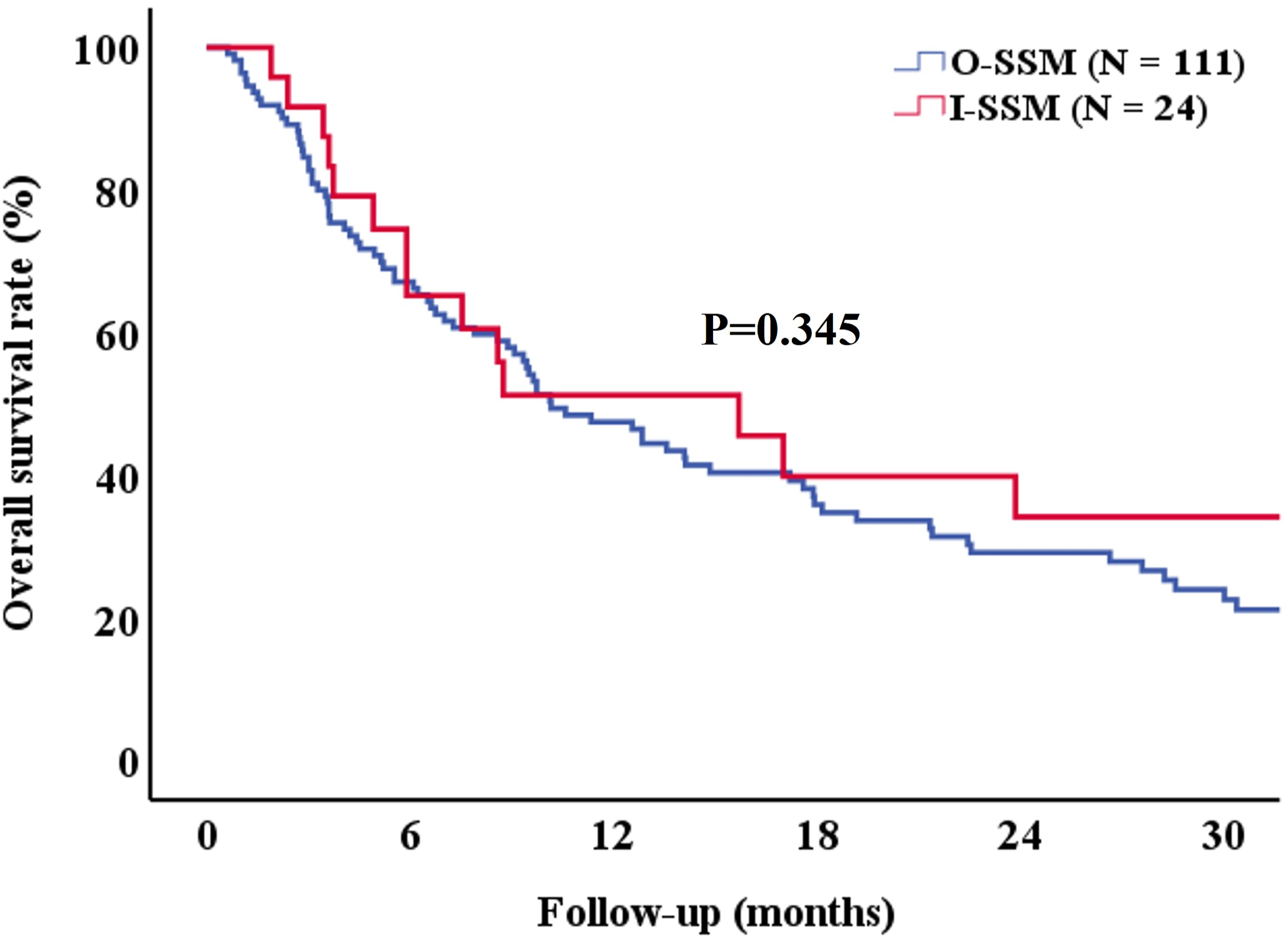
Kaplan-Meier survival analysis for overall survival between patients with I-SSM and those with O-SSM.
Prognostic Factors for Survival After Surgery
Univariate Analysis of the Presumed Prognostic Factors for Survival.
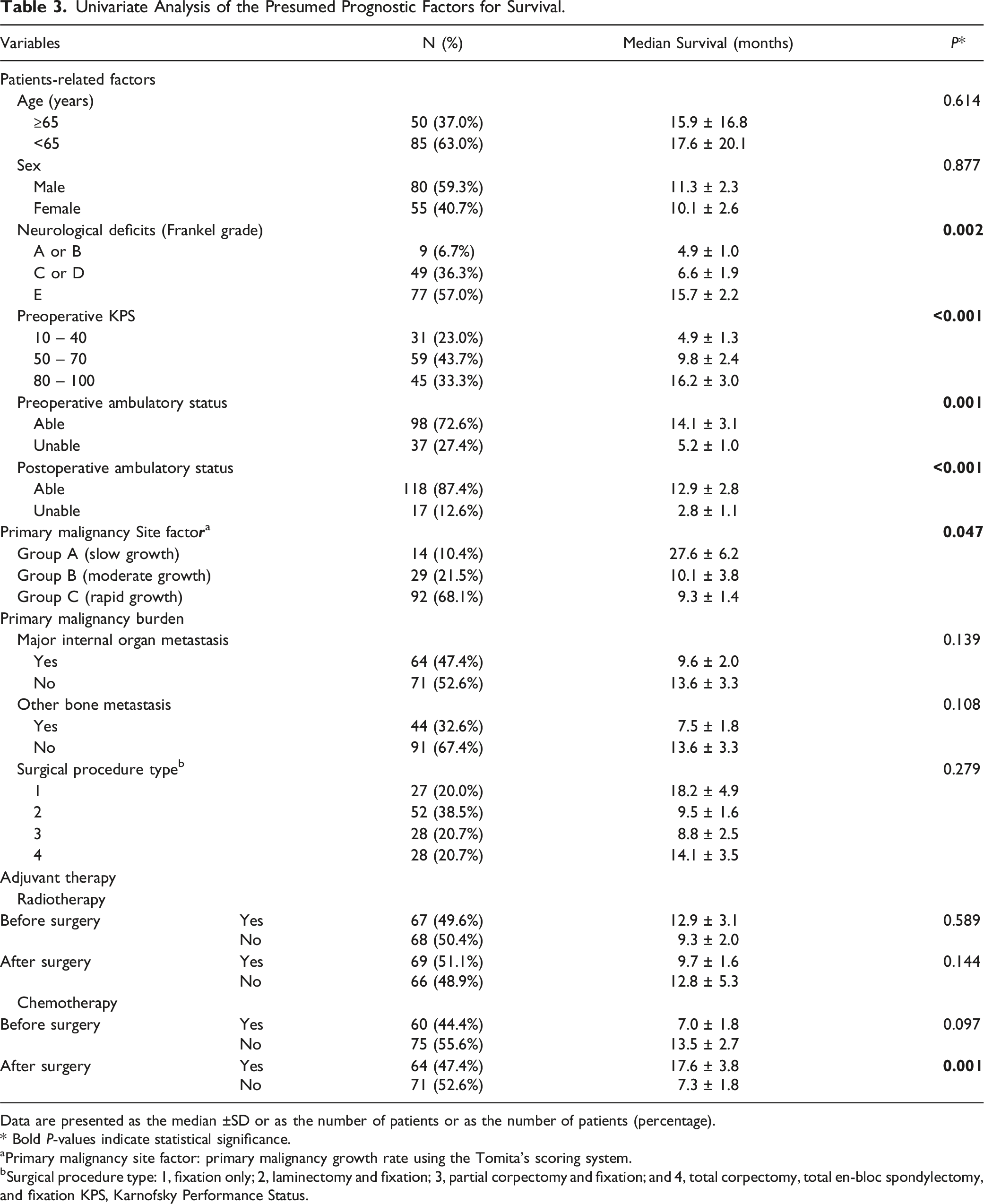
Data are presented as the median ±SD or as the number of patients or as the number of patients (percentage).
* Bold
aPrimary malignancy site factor: primary malignancy growth rate using the Tomita’s scoring system.
bSurgical procedure type: 1, fixation only; 2, laminectomy and fixation; 3, partial corpectomy and fixation; and 4, total corpectomy, total en-bloc spondylectomy, and fixation KPS, Karnofsky Performance Status.
Multivariate Analysis for Prognostic Factors of Survival in Patients With SSM.
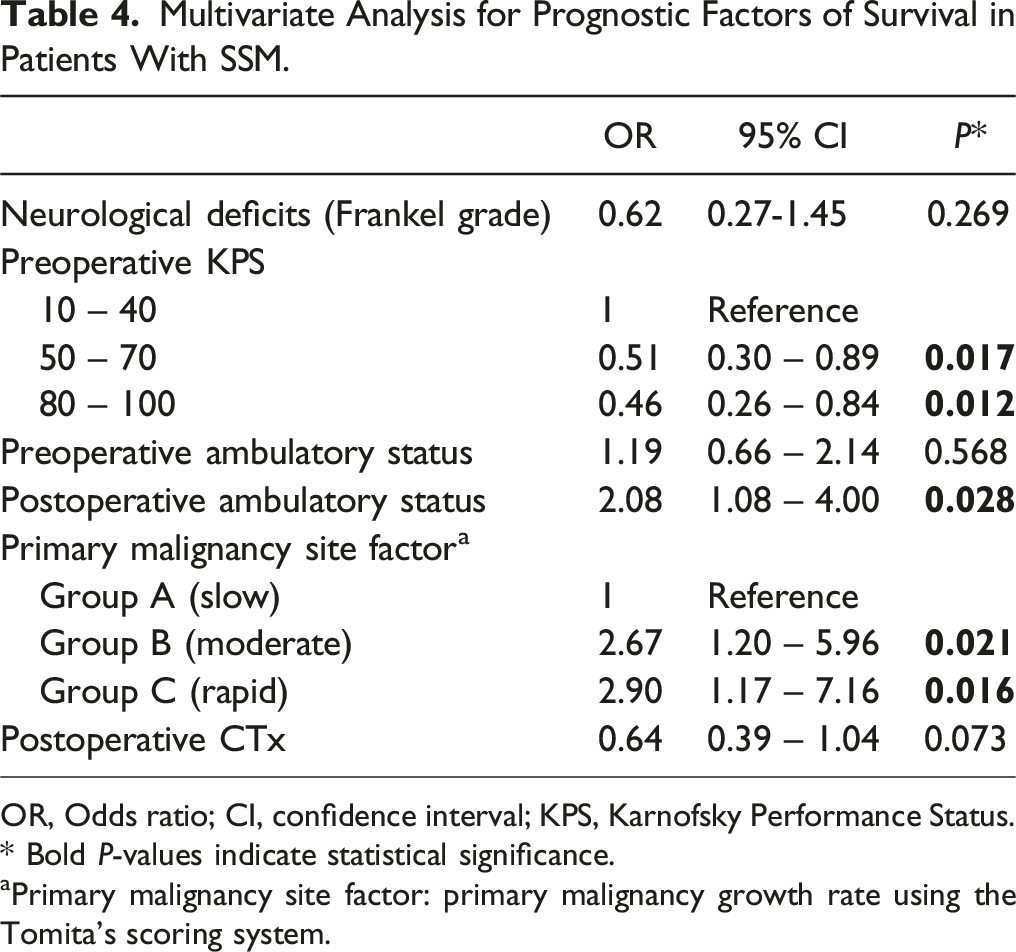
OR, Odds ratio; CI, confidence interval; KPS, Karnofsky Performance Status.
* Bold
aPrimary malignancy site factor: primary malignancy growth rate using the Tomita’s scoring system.
Discussion
Surgical decision-making in SSM remains challenging despite advances in radiotherapy, targeted therapy, and immunotherapy. 23 This study aimed to guide surgeons in navigating the dilemma of surgical aggressiveness for SSM patients. However, the inherent heterogeneity of this population further complicates decision-making, especially considering the limited lifespan associated with SSM. Existing research underscores the short median survival for patients with SSM. Yang et al reported 6 months, 22 Lun et al. 8.2 months in 78 patients, 14 and Lee et al. 10.8 months in 200 patients. 24 Our study aligns with these findings, revealing a median survival of 10.2 months. This indicates that the median survival for patients with an SSM is relatively short, typically around 12 months. Moreover, Arrigo et al and Bollen et al documented similar median survival times (8.0 and 10.7 months, respectively) even in cohorts including patients without visceral metastases.25,26 This consistently calls into question whether the presence of visceral metastases should be a definitive factor in surgical decision-making for SSM.
Recent studies have challenged traditional prognostic factors for spinal metastasis, emphasizing the importance of patient’s general health and functional status over previously considered factors. Arrigo et al and Chong et al found that visceral metastasis, generally considered important for prognosis, was not significant for surgically treated patients.26,27 Lun et al 14 reported no difference in survival between patients with single vs multiple spinal metastases, suggesting that the number of metastases may not impact prognosis. Instead, Switlyk et al and Chen et al identified serum albumin and lactate dehydrogenase levels as significant indicators of general condition and survival prognosis.21,28 Our study aligns with these findings, identifying preoperative KPS and postoperative ambulatory status as significant prognostic factors, surpassing even visceral metastasis or the number of metastases. Notably, KPS is significant as a preoperative assessment tool that can be used to determine the aggressiveness of surgical intervention.
Ambulatory status is closely linked to KPS, and in non-ambulatory patients, the risks associated with chemotherapy often outweigh its potential benefits due to an increased risk of adverse effects. Consequently, chemotherapy is generally not administered to non-ambulatory patients at our institution. In this study, postoperative chemotherapy was closely related to ambulatory status and appeared as a factor influencing survival in the univariate analysis but was excluded from the multivariate analysis. We hypothesized that this may be attributed to two factors: 1) postoperative ambulation status may serve as a stronger prognostic factor, reflecting the patient’s overall general condition, 2) patients who were capable of ambulation were more likely to receive aggressive chemotherapy. and 3) even when chemotherapy is administered, it may not uniformly improve survival across all patients, as individual variations in chemotherapy resistance may affect treatment efficacy. Interestingly, patients who were unable to ambulate preoperatively but regained ambulatory function postoperatively had a mean survival of 16.0 ± 4.1 months and a median survival of 9.6 ± 3.4 months. This was not statistically significant (
Finally, the primary malignancy site emerged as another significant prognostic factor that can be assessed preoperatively. This aligns with findings by Constans et al, 33 who reported that primary tumor type influences both survival and quality of short-term outcomes in patients with spinal metastases. This study’s finding regarding visceral metastasis adds a noteworthy counterpoint to existing literature. While previous research has consistently identified their presence as a significant prognostic factor,25-27,34,35 our analysis among surgically treated patients found no such association. One potential explanation for this discrepancy lies in study populations. Previous studies have included patients who did not undergo surgery, introducing heterogeneity, whereas this study only included patients who underwent surgical treatment. Not all spinal metastases necessitate surgical treatment. However, by the time they can reach that point where surgery is necessary, the tumor burden is substantial, and at that point, the overall performance status and general condition may become more important than the tumor burden itself. From the surgeon’s perspective, whether the primary origin site is well controlled is often not one of the primary concerns. Instead, understanding prognostic factors that assist in determining the extent of surgery at the time of surgery is a greater concern. It is therefore worth noting that there appears to be no significant difference in prognosis between I-SSM and O-SSM groups. This implies that, even in the case of a patient with I-SSM, there may not be an imperative need to consider more aggressive surgical treatment.
There are several limitations to this study. First, the study has a relatively small sample size of 135 patients. We focused on patients with SSM who underwent surgical treatment, which further reduced the number of cases available for analysis. In this study, we used the Tomita score (A, B, or C) for grouping; however, this may not fully reflect the diverse biological characteristics and prognoses of individual tumor types. Moreover, this study had a high proportion of Group B (21.5%) and Group C (68.1%), which together constitute a larger fraction (89.6%) compared to Group A (10.4%). This uneven distribution may be due to differences in racial, genetic, lifestyle, and dietary factors. Additionally, because this study focused on patients who underwent surgical treatment, there might be a tendency toward including more aggressive tumor types. Consequently, the findings may not be generalizable to other geographic locations. Future studies with larger cohorts might benefit from analyzing survival and prognostic factors specific to particular tumor types rather than broad categories. Second, there was a lack of data regarding the prognosis of non-surgical patients. Our study aimed to assess postoperative prognosis based on the timing of the need for surgical treatment. Terminal-stage patients with spinal metastases exhibit significant heterogeneity due to varying general conditions and comorbidities, potentially introducing bias in survival data. However, indications for surgical treatment were carefully determined through multidisciplinary collaboration. Therefore, this study is significant for providing valuable insights into prognostic factors relevant to determining the appropriate extent of surgical treatment, particularly in patients with SSM.
Conclusion
Patients with SSM have a median survival of 10.2 months, with no significant difference in postoperative survival between I-SSM and O-SSM groups. Significant prognostic factors for survival after surgery in SSM patients included preoperative KPS, postoperative ambulatory status, and the primary malignancy site. Therefore, even in the case of I-SSM, aggressive surgery should be undertaken cautiously with careful consideration of these identified factors.
Supplemental Material
Supplemental Material - Survival and Prognostic Factors After Surgery in Single Spinal Metastasis: Comparison of Isolated-Single Spinal Metastasis and Single Spinal Metastasis With Other Metastasis
Supplemental Material for Survival and Prognostic Factors After Surgery in Single Spinal Metastasis: Comparison of Isolated-Single Spinal Metastasis and Single Spinal Metastasis With Other Metastasis by Hyun-Jun Kim, MD, Jin-Sung Park, Se-Jun Park, Dong-Ho Kang, and Chong-Suh Lee in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
